Canine Endogenous Oxytocin Responses to Dog-Walking and Affiliative Human–Dog Interactions
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Human–Dog Bond Questionnaire
2.3. Experimental Conditions
2.3.1. Dog-Walking (DW)
2.3.2. Affiliative Human–Dog Interaction (H-DI)
2.4. Hormone Analysis
2.4.1. Urinary Oxytocin
2.4.2. Urinary Creatinine
2.5. Statistical Methods
3. Results
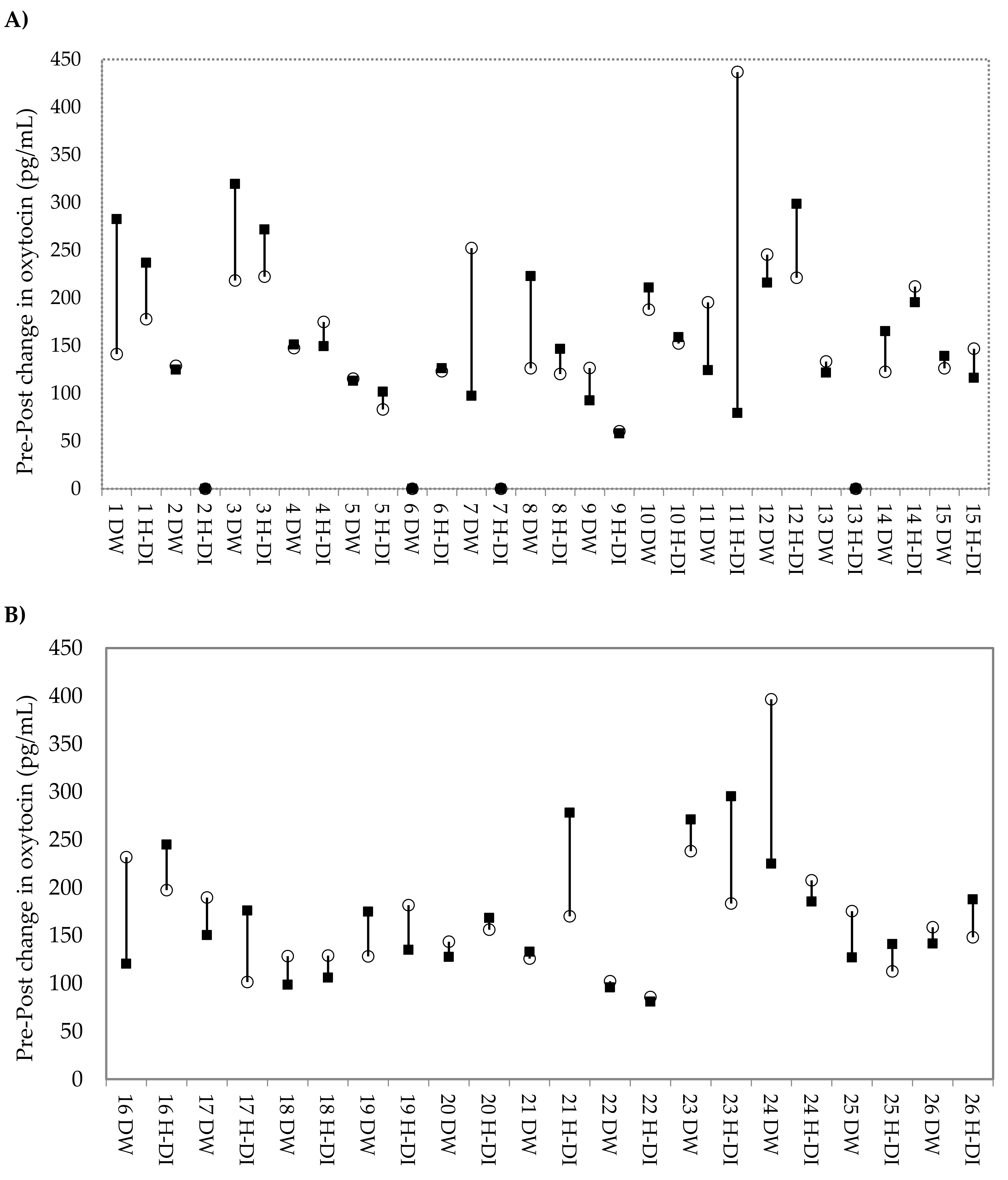
3.1. Differences in Urinary Oxytocin
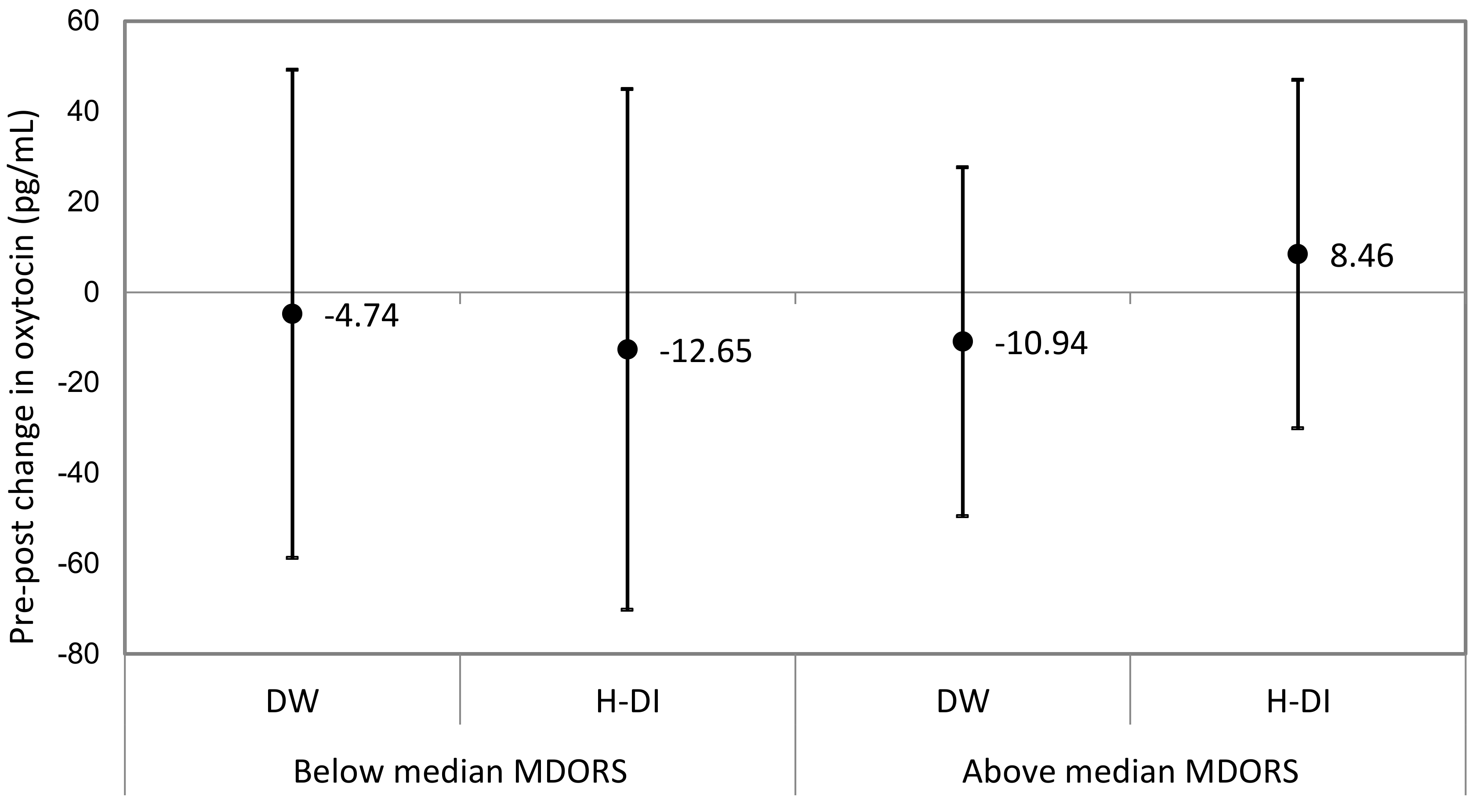
3.2. MDORS
3.3. Sensitivity Analysis
3.3.1. Dog Breed Group
3.3.2. Dog Age
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Miklósi, Á.; Topál, J. What does it take to become ‘best friends’? Evolutionary changes in canine social competence. Trends Cogn. Sci. 2013, 17, 287–294. [Google Scholar]
- Payne, E.; Bennett, P.C.; McGreevy, P.D. Current perspectives on attachment and bonding in the dog–human dyad. Psych. Res. Behav. Manag. 2015, 8, 71. [Google Scholar] [CrossRef] [PubMed]
- Buttner, A.P. Neurobiological underpinnings of dogs’ human-like social competence: How interactions between stress response systems and oxytocin mediate dogs’ social skills. Neurosci. Biobehav. Rev. 2016, 71, 198–2014. [Google Scholar] [CrossRef] [PubMed]
- Gimpl, G.; Fahrenholz, F. The oxytocin receptor system: structure, function, and regulation. Physiol. Rev. 2001, 81, 629–683. [Google Scholar] [CrossRef] [PubMed]
- Powell, L.; Guastella, A.J.; McGreevy, P.; Bauman, A.; Edwards, K.M.; Stamatakis, E. The physiological function of oxytocin in humans and its acute response to human–dog interactions: A review of the literature. J. Vet. Behav. 2018. [Google Scholar] [CrossRef]
- Kis, A.; Kanizsár, O.; Gácsi, M.; Topál, J. Intranasally administered oxytocin decreases heart rate and increases heart rate variability in dogs. J. Vet. Behav. 2014, 9, e15. [Google Scholar] [CrossRef]
- Dölen, G.; Darvishzadeh, A.; Huang, K.W.; Malenka, R.C. Social reward requires coordinated activity of nucleus accumbens oxytocin and serotonin. Nature 2013, 501, 179. [Google Scholar]
- Nagasawa, M.; Ogawa, M.; Mogi, K.; Kikusui, T. Intranasal oxytocin treatment increases eye-gaze behavior toward the owner in ancient Japanese dog breeds. Front. Psychol. 2014, 8, 1624. [Google Scholar] [CrossRef]
- Romero, T.; Nagasawa, M.; Mogi, K.; Hasegawa, T.; Kikusui, T. Oxytocin promotes social bonding in dogs. Proc. Natl. Acad. Sci. USA 2014, 111, 9085–9090. [Google Scholar] [CrossRef]
- Kis, A.; Ciobica, A.; Topál, J. The effect of oxytocin on human-directed social behaviour in dogs (Canis familiaris). Horm. Behav. 2017, 94, 40–52. [Google Scholar] [CrossRef]
- Odendaal, J.S.; Meintjes, R.A. Neurophysiological correlates of affiliative behaviour between humans and dogs. Vet. J. 2003, 165, 296–301. [Google Scholar] [CrossRef]
- Handlin, L.; Hydbring-Sandberg, E.; Nilsson, A.; Ejdebäck, M.; Jansson, A.; Uvnäs-Moberg, K. Short-term interaction between dogs and their owners: Effects on oxytocin, cortisol, insulin and heart rate—An exploratory study. Anthrozoös 2011, 24, 301–3015. [Google Scholar] [CrossRef]
- Mitsui, S.; Yamamoto, M.; Nagasawa, M.; Mogi, K.; Kikusui, T.; Ohtani, N.; Ohta, M. Urinary oxytocin as a noninvasive biomarker of positive emotion in dogs. Horm. Behav. 2011, 60, 239–243. [Google Scholar] [CrossRef] [PubMed]
- Nagasawa, M.; Mitsui, S.; En, S.; Ohtani, N.; Ohta, M.; Sakuma, Y.; Onaka, T.; Mogi, K.; Kikusui, T. Oxytocin-gaze positive loop and the coevolution of human–dog bonds. Science 2015, 348, 333–336. [Google Scholar] [CrossRef] [PubMed]
- Rehn, T.; Handlin, L.; Uvnäs-Moberg, K.; Keeling, L.J. Dogs’ endocrine and behavioural responses at reunion are affected by how the human initiates contact. Physiol. Behav. 2014, 124, 45–53. [Google Scholar] [CrossRef] [PubMed]
- MacLean, E.L.; Gesquiere, L.R.; Gee, N.R.; Levy, K.; Martin, L.W.; Carter, C.S. Effects of affiliative human–animal interaction on dog salivary and plasma oxytocin and vasopressin. Front. Psychol. 2017, 8, 1606. [Google Scholar]
- Kerepesi, A.; Dóka, A.; Miklósi, Á. Dogs and their human companions: the effect of familiarity on dog–human interactions. Behav. Process. 2015, 110, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Horn, L.; Range, F.; Huber, L. Dogs’ attention towards humans depends on their relationship, not only on social familiarity. Animal Cogn. 2013, 16, 435–443. [Google Scholar] [CrossRef] [PubMed]
- Handlin, L.; Nilsson, A.; Ejdebäck, M.; Hydbring-Sandberg, E.; Uvnäs-Moberg, K. Associations between the psychological characteristics of the human–dog relationship and oxytocin and cortisol levels. Anthrozoös 2012, 25, 215–228. [Google Scholar] [CrossRef]
- Dwyer, F.; Bennett, P.C.; Coleman, G.J. Development of the Monash dog owner relationship scale (MDORS). Anthrozoös 2006, 19, 243–256. [Google Scholar] [CrossRef]
- Stamatakis, E.; Ekelund, U.; Wareham, N.J. Temporal trends in physical activity in England: The health survey for England 1991 to 2004. Prev. Med. 2007, 45, 416–423. [Google Scholar] [CrossRef] [PubMed]
- Saghaei, M. An overview of randomization and minimization programs for randomized clinical trials. J. Med. Signals Sens. 2011, 1, 55. [Google Scholar] [PubMed]
- Strava Inc. Strava Tracker: Record Running, Cycling & Swimming [mobile application software]. 2017. Available online: https://www.strava.com/ (accessed on 6 July 2017).
- Askenazi, D.J.; Moore, J.F.; Fineberg, N.; Koralkar, R.; Clevenger, S.; Sharer, J.D. Comparison of methods, storage conditions, and time to analysis of serum and urine creatinine measured from microsamples by liquid chromatography mass spectrometery (lc/ms) vs. Jaffe. J. Clin. Lab. Anal. 2014, 28, 405–408. [Google Scholar] [CrossRef] [PubMed]
- Reyes, T.L.; Galinsky, A.M.; Hoffmann, J.N.; You, H.M.; Ziegler, T.E.; McClintock, M.K. Social peptides: measuring urinary oxytocin and vasopressin in a home field study of older adults at risk for dehydration. J. Gerontol. B Psychol. Sci. Soc. Sci. 2014, 69 Suppl. 2, S229–S237. [Google Scholar] [CrossRef] [PubMed]
- Trial Review. Available online: https://www.anzctr.org.au/Trial/Registration/TrialReview.aspx?id=373188&isReview=true (accessed on 6 September 2018).
- Dog Breeds. Available online: https://www.akc.org/dog-breeds/ (accessed on 30 August 2018).
- McGreevy, P.D.; Starling, M.; Branson, N.; Cobb, M.L.; Calnon, D. An overview of the dog–human dyad and ethograms within it. J. Vet. Behav. 2012, 7, 103–117. [Google Scholar] [CrossRef]
- Rilling, J.K.; DeMarco, A.C.; Hackett, P.D.; Chen, X.; Gautam, P.; Stair, S.; Haroon, E.; Thompson, R.; Ditzen, B.; Patel, R. Sex differences in the neural and behavioral response to intranasal oxytocin and vasopressin during human social interaction. Psychoneuroendocrinology 2014, 39, 237–248. [Google Scholar] [CrossRef] [PubMed]
- Kovács, K.; Kis, A.; Kanizsár, O.; Hernádi, A.; Gácsi, M.; Topál, J. The effect of oxytocin on biological motion perception in dogs (Canis familiaris). Animal Cogn. 2016, 19, 513–522. [Google Scholar]
- Odendaal, J. Animal-assisted therapy—Magic or medicine? J. Psychosom. Res. 2000, 49, 275–280. [Google Scholar] [CrossRef]
- Herzog, H. The impact of pets on human health and psychological well-being: Fact, fiction, or hypothesis? Curr. Dir. Psychol. Sci. 2011, 20, 236–239. [Google Scholar] [CrossRef]
- Easterbrook, P.J.; Gopalan, R.; Berlin, J.; Matthews, D.R. Publication bias in clinical research. Lancet 1991, 337, 867–872. [Google Scholar] [CrossRef]
- McCullough, M.E.; Churchland, P.S.; Mendez, A.J. Problems with measuring peripheral oxytocin: Can the data on oxytocin and human behavior be trusted? Neurosci. Biobehav. Rev. 2013, 37, 1485–1492. [Google Scholar] [CrossRef] [PubMed]
- Neumann, I.D.; Landgraf, R. Balance of brain oxytocin and vasopressin: implications for anxiety, depression, and social behaviors. Trends Neurosci. 2012, 35, 649–659. [Google Scholar] [CrossRef] [PubMed]
- Rehn, T.; Keeling, L.J. Measuring dog-owner relationships: Crossing boundaries between animal behaviour and human psychology. Appl. Anim. Behav. Sci. 2016, 183, 1–9. [Google Scholar] [CrossRef]
- Persson, M.E.; Trottier, A.J.; Bélteky, J.; Roth, L.S.; Jensen, P. Intranasal oxytocin and a polymorphism in the oxytocin receptor gene are associated with human-directed social behavior in golden retriever dogs. Horm. Behav. 2017, 95, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Pekkin, A.-M.; Hänninen, L.; Tiira, K.; Koskela, A.; Pöytäkangas, M.; Lohi, H.; Valros, A. The effect of a pressure vest on the behaviour, salivary cortisol and urine oxytocin of noise phobic dogs in a controlled test. Appl. Anim. Behav. Sci. 2016, 185, 86–94. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Powell, L.; Edwards, K.M.; Bauman, A.; Guastella, A.J.; Drayton, B.; Stamatakis, E.; McGreevy, P. Canine Endogenous Oxytocin Responses to Dog-Walking and Affiliative Human–Dog Interactions. Animals 2019, 9, 51. https://doi.org/10.3390/ani9020051
Powell L, Edwards KM, Bauman A, Guastella AJ, Drayton B, Stamatakis E, McGreevy P. Canine Endogenous Oxytocin Responses to Dog-Walking and Affiliative Human–Dog Interactions. Animals. 2019; 9(2):51. https://doi.org/10.3390/ani9020051
Chicago/Turabian StylePowell, Lauren, Kate M. Edwards, Adrian Bauman, Adam J. Guastella, Bradley Drayton, Emmanuel Stamatakis, and Paul McGreevy. 2019. "Canine Endogenous Oxytocin Responses to Dog-Walking and Affiliative Human–Dog Interactions" Animals 9, no. 2: 51. https://doi.org/10.3390/ani9020051
APA StylePowell, L., Edwards, K. M., Bauman, A., Guastella, A. J., Drayton, B., Stamatakis, E., & McGreevy, P. (2019). Canine Endogenous Oxytocin Responses to Dog-Walking and Affiliative Human–Dog Interactions. Animals, 9(2), 51. https://doi.org/10.3390/ani9020051





