Different Diets Change the Expression of Bovine Serum Extracellular Vesicle-miRNAs
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Animals and Management
2.3. Serum Collection
2.4. Preparation of EVs from Serum
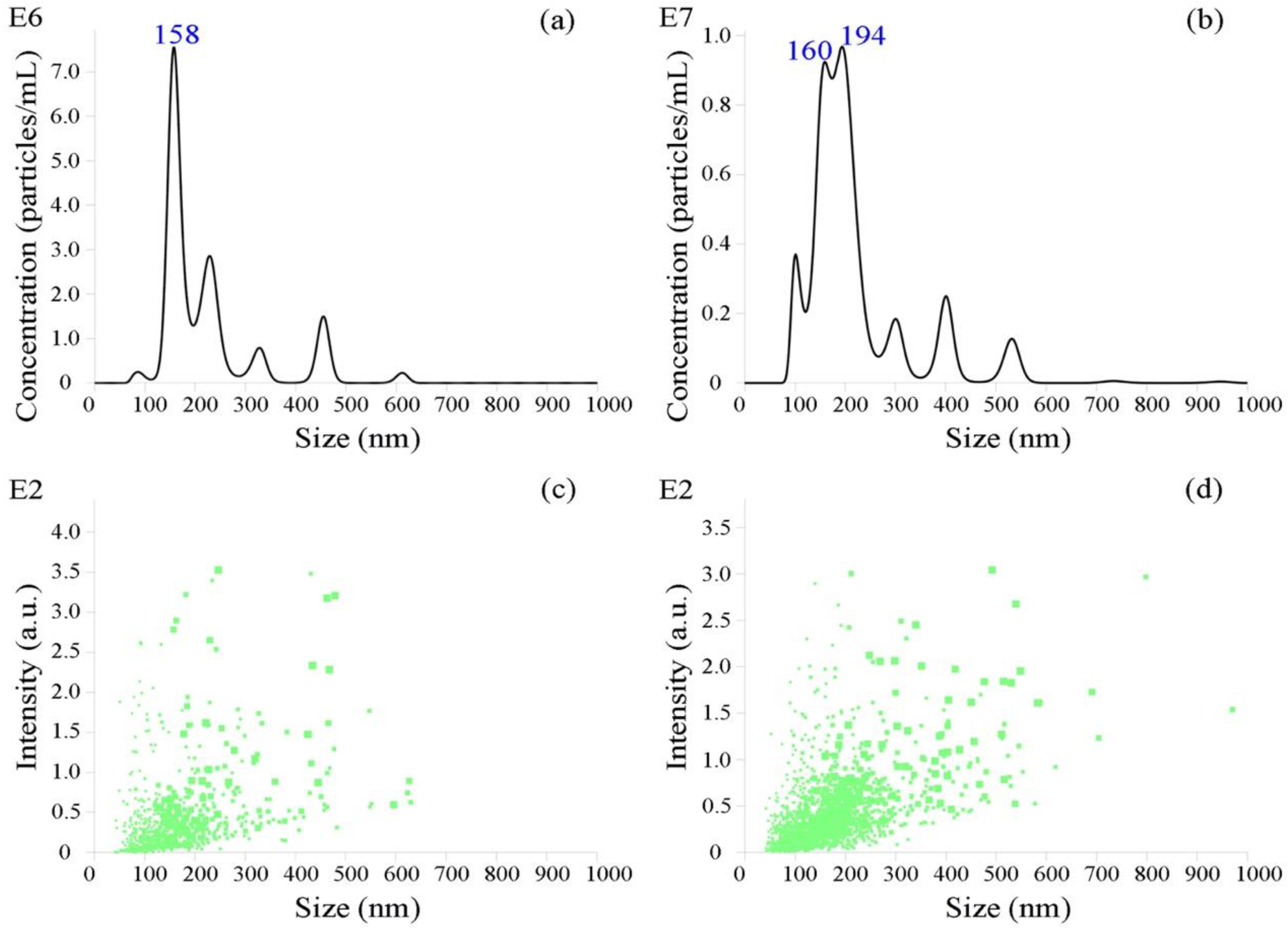
2.5. Nanoparticle Tracking Analysis
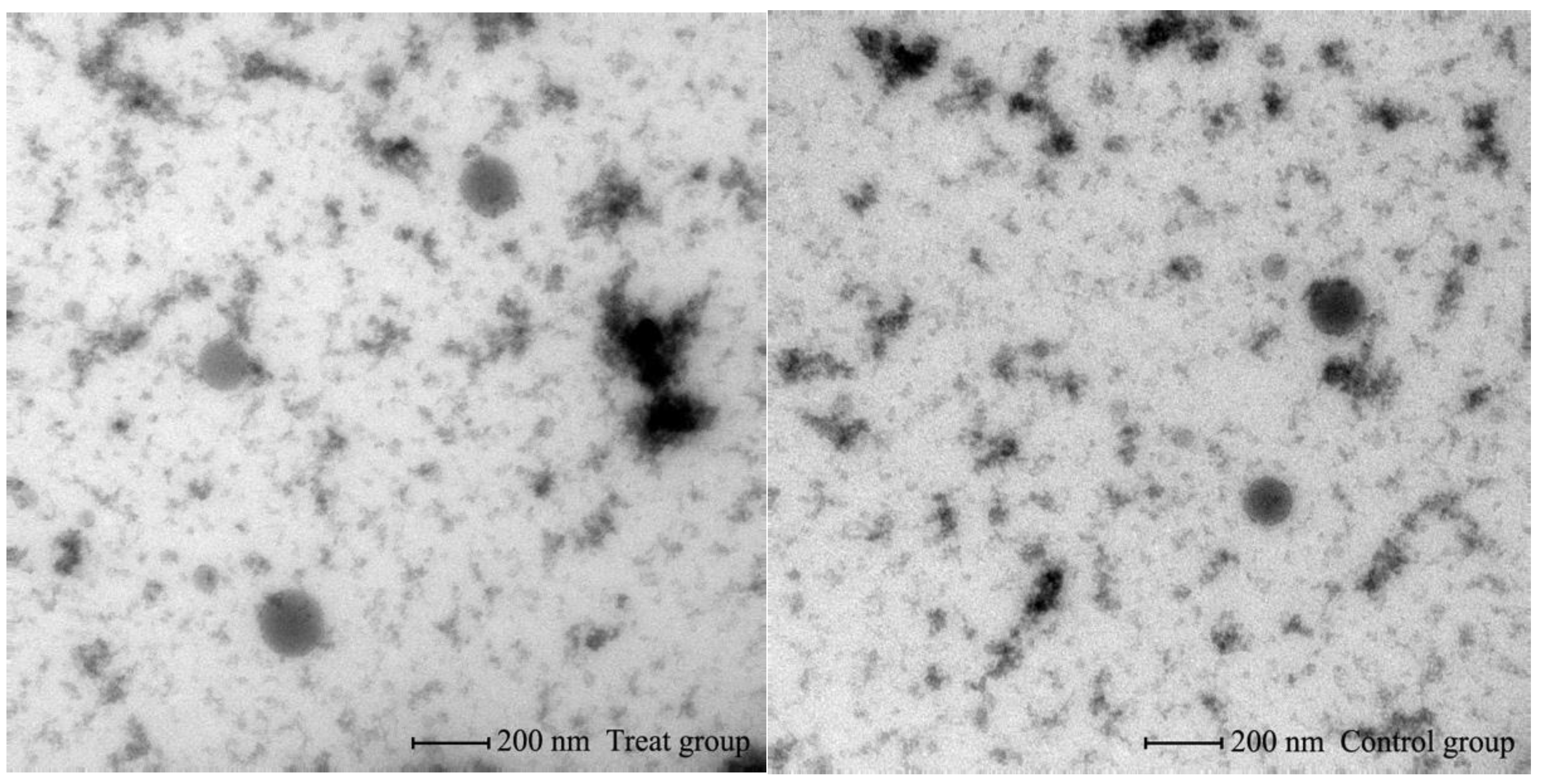
2.6. Transmission Electron Microscopy
2.7. Small RNA Library Construction and Sequencing
2.8. Bioinformatics Analysis
3. Results
3.1. Identification of Bovine Serum EVs
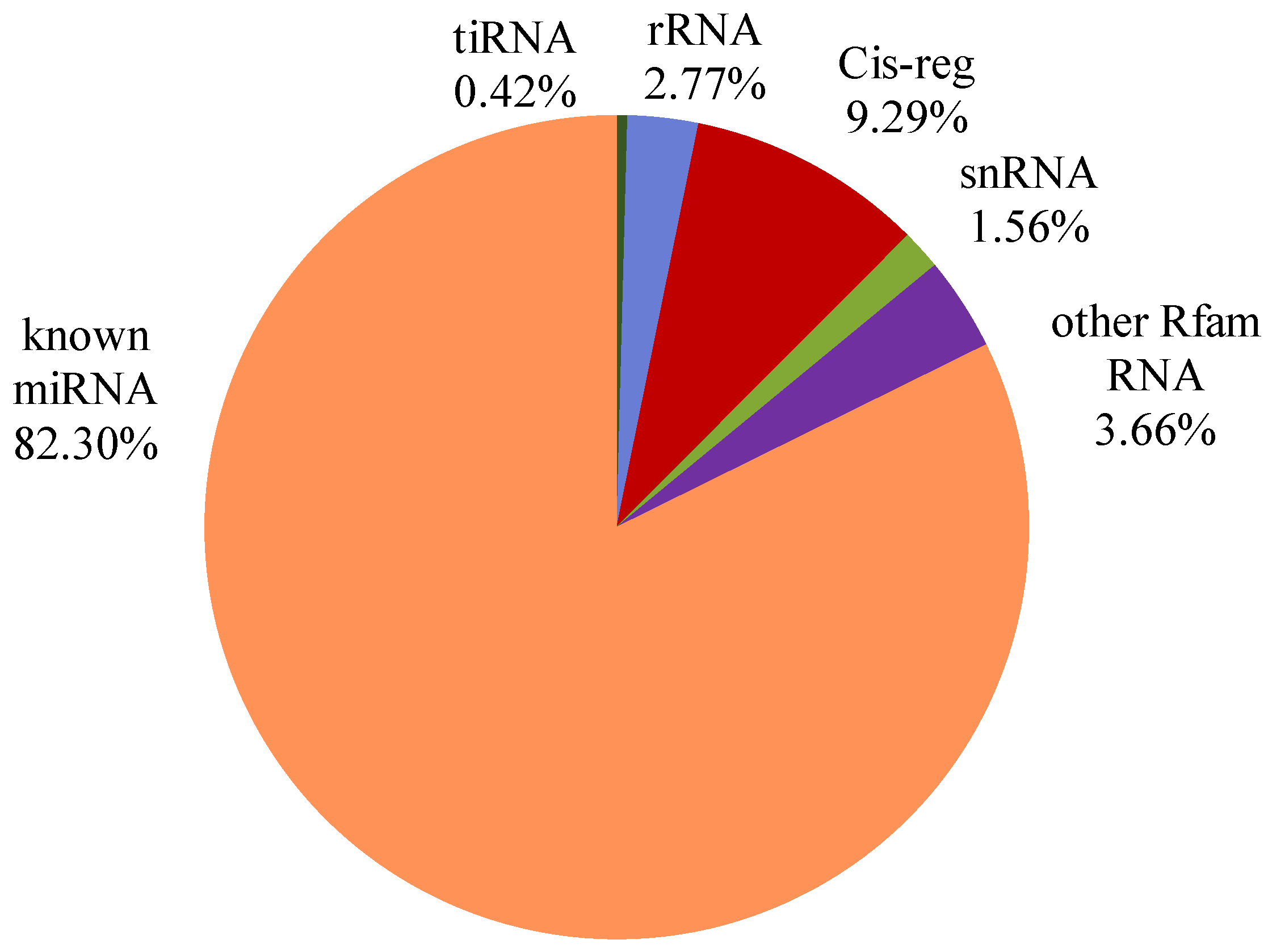
3.2. Small RNAs Are Abundant in Bovine Serum EVs
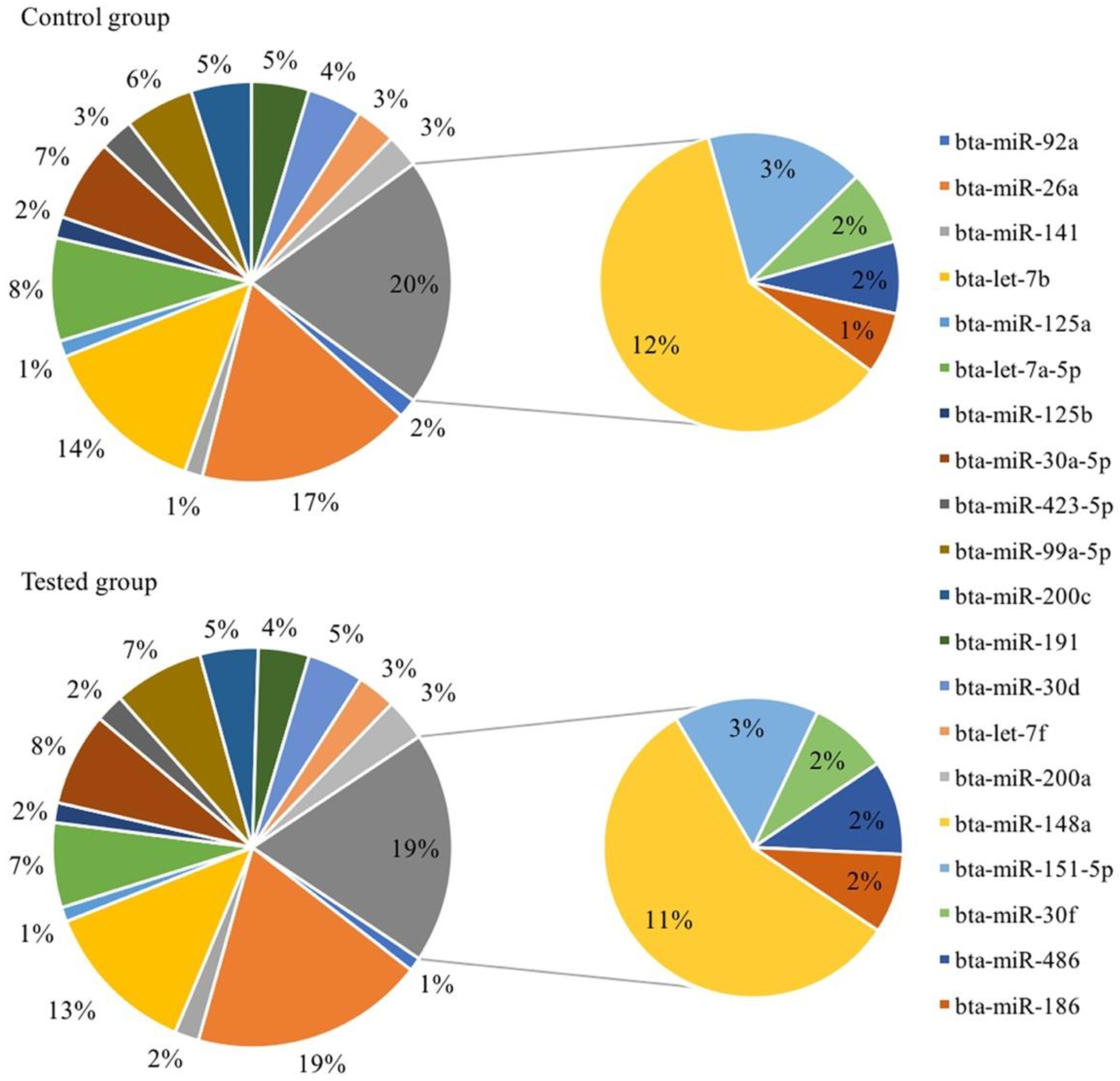
3.3. Immune-Related miRNAs Are Highly Expressed in Bovine Serum EVs
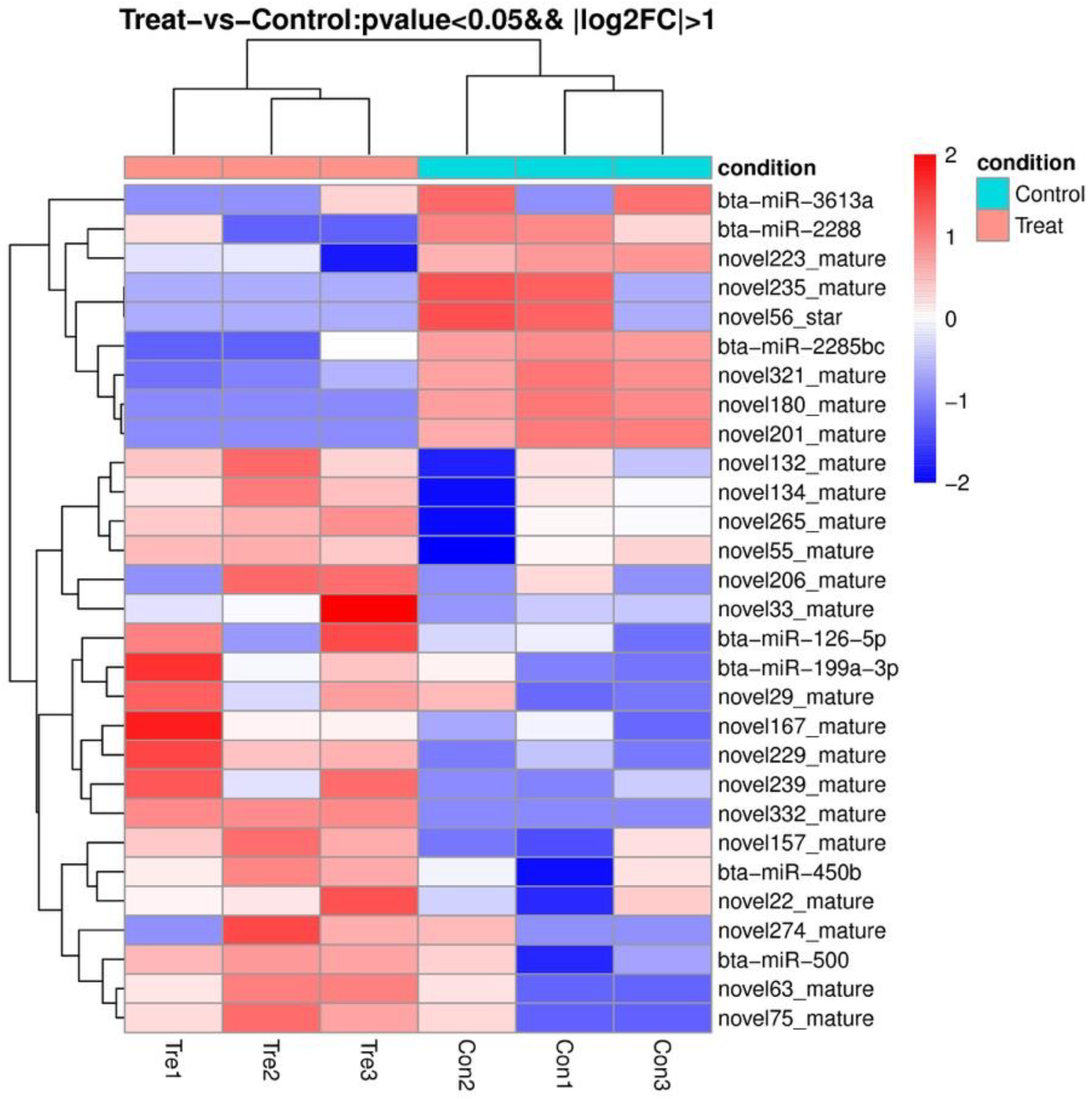
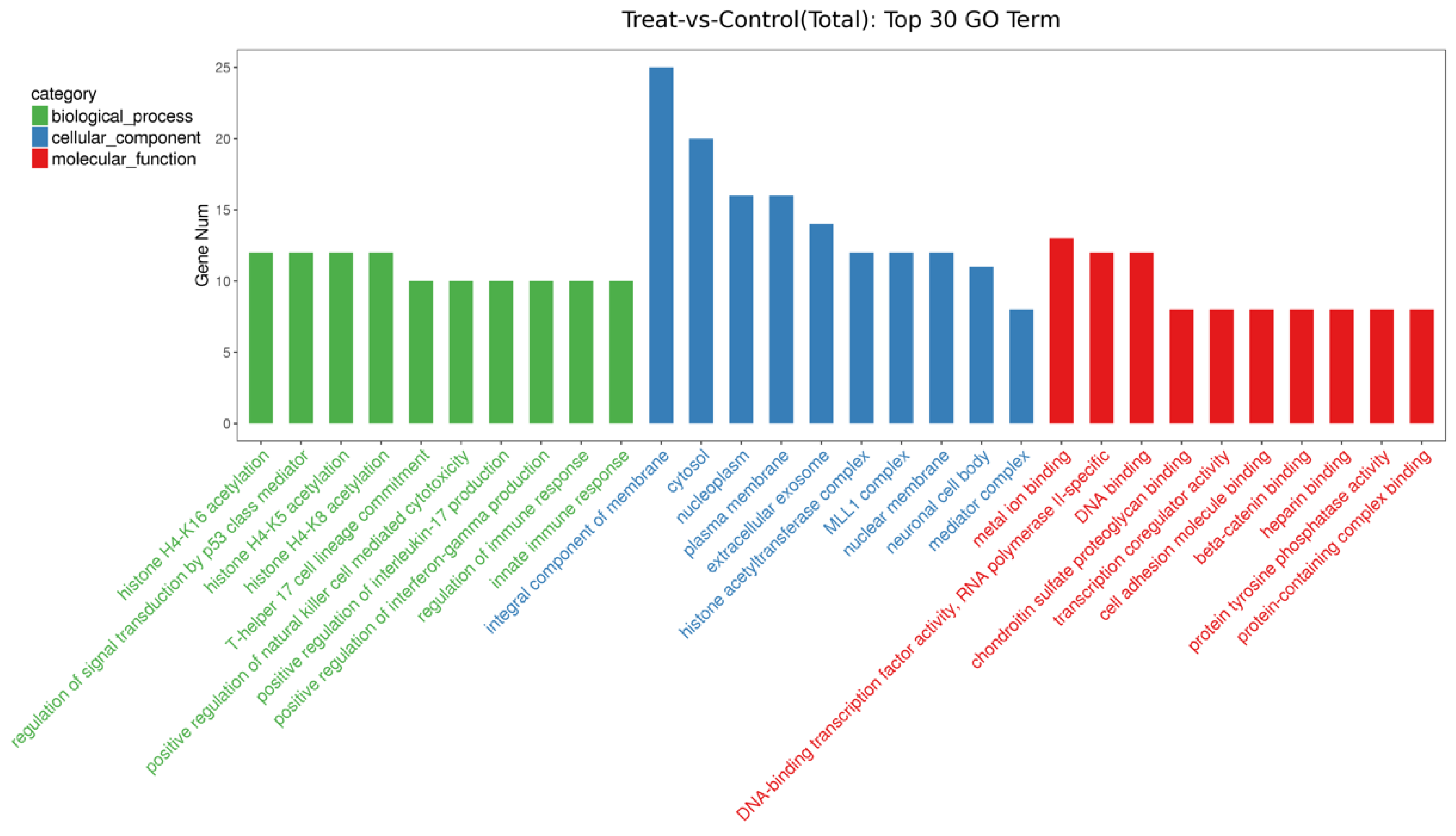
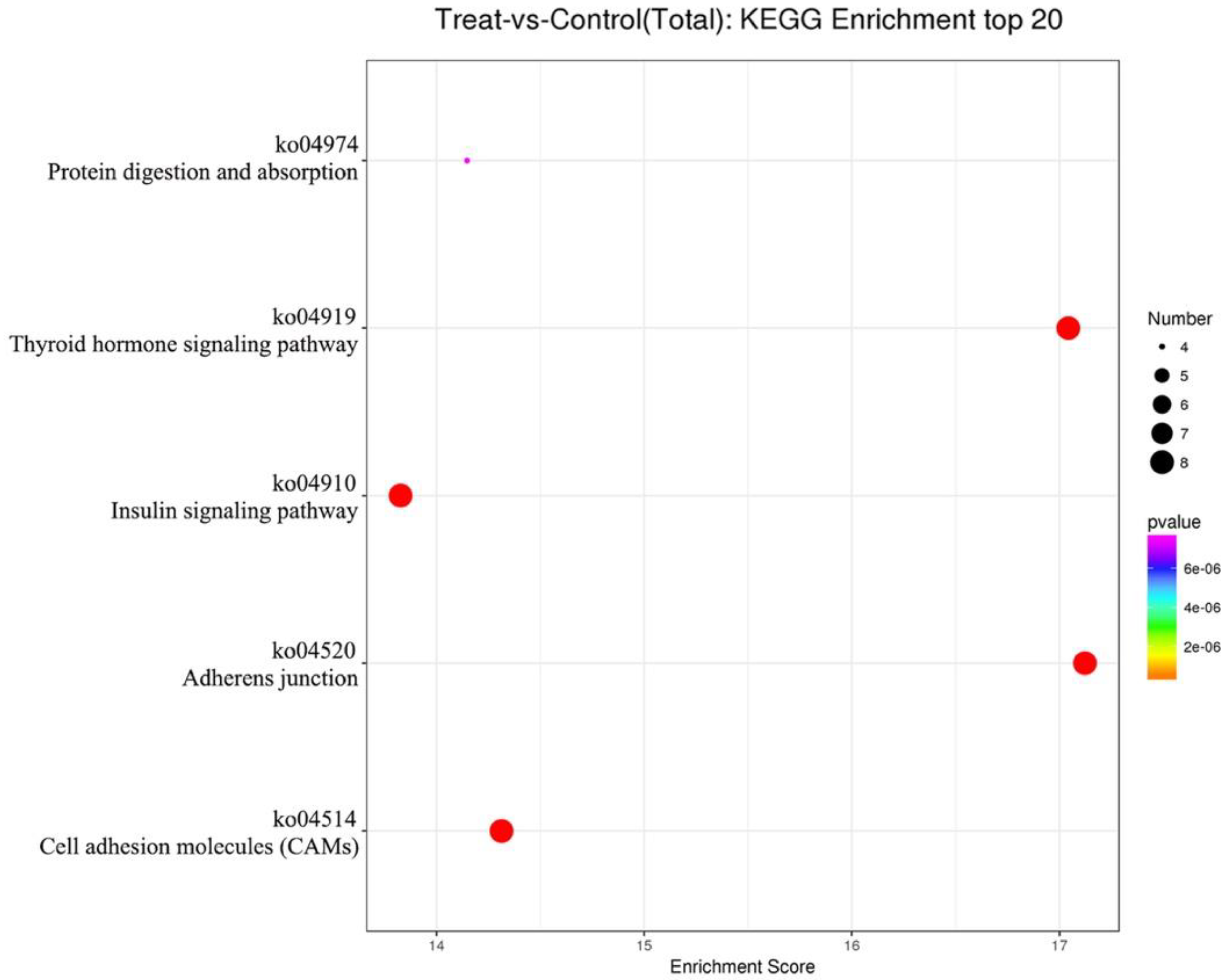
3.4. Effects of Different Diets on the Expression of EV-miRNAs
4. Discussion
5. Conclusions
Data Availability
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| miRNA | microRNA |
| EV | Extracellular Vesicle |
| TEM | Transmission Electron Microscope |
| GO | Gene Ontology |
| KEGG | Kyoto Encyclopedia of Genes and Genomes |
| CAM | Cell Adhesion Molecule |
| NTA | Nanoparticle Tracking Analysis |
| TMR | Total Mixed Ration |
References
- Yanez-Mo, M.; Siljander, P.R.; Andreu, Z.; Zavec, A.B.; Borras, F.E.; Buzas, E.I.; Buzas, K.; Casal, E.; Cappello, F.; Carvalho, J.; et al. Biological properties of extracellular vesicles and their physiological functions. J. Extracel. Vesicles 2015, 4, 27066. [Google Scholar] [CrossRef] [PubMed]
- Ciardiello, C.; Cavallini, L.; Spinelli, C.; Yang, J.; Reissobreiro, M.; Candia, P.D.; Minciacchi, V.R.; Vizio, D.D. Focus on Extracellular Vesicles: New Frontiers of Cell-to-Cell Communication in Cancer. Int. J. Mol. Sci. 2016, 17, 175. [Google Scholar] [CrossRef] [PubMed]
- Andaloussi, S.E.; Mäger, I.; Breakefield, X.O.; Wood, M.J. Extracellular vesicles: Biology and emerging therapeutic opportunities. Nat. Rev. Drug Discov. 2013, 12, 347. [Google Scholar] [CrossRef] [PubMed]
- Taylor, D.D.; Gercel-Taylor, C. The origin, function, and diagnostic potential of RNA within extracellular vesicles present in human biological fluids. Front Genet 2013, 4, 142. [Google Scholar] [CrossRef] [PubMed]
- Weber, J.A.; Baxter, D.H.; Zhang, S.; Huang, D.Y.; How Huang, K.; Jen Lee, M.; Galas, D.J.; Wang, K. The MicroRNA Spectrum in 12 Body Fluids. Clin. Chem. 2010, 56, 1733–1741. [Google Scholar] [CrossRef] [PubMed]
- Psychogios, N.; Hau, D.D.; Peng, J.; Guo, A.C.; Mandal, R.; Bouatra, S.; Sinelnikov, I.; Krishnamurthy, R.; Eisner, R.; Gautam, B.; et al. The human serum metabolome. PLoS ONE 2011, 6, e16957. [Google Scholar] [CrossRef] [PubMed]
- Samanta, S.; Rajasingh, S.; Drosos, N.; Zhou, Z.; Dawn, B.; Rajasingh, J. Exosomes: New molecular targets of diseases. Acta Pharmacol. Sin. 2018, 39, 501–513. [Google Scholar] [CrossRef]
- Bartel, D.P. MicroRNAs: Genomics, biogenesis, mechanism, and function. Cell 2004, 116, 281–297. [Google Scholar] [CrossRef]
- Zhang, B.; Wang, Q.; Pan, X. MicroRNAs and their regulatory roles in animals and plants. J. Cell. Physiol. 2007, 210, 279–289. [Google Scholar] [CrossRef]
- Pritchard, C.C.; Cheng, H.H.; Tewari, M. MicroRNA profiling: Approaches and considerations. Nat. Rev. Genet. 2012, 13, 358. [Google Scholar] [CrossRef]
- Zhao, K.; Liang, G.; Sun, X.; Guan le, L. Comparative miRNAome analysis revealed different miRNA expression profiles in bovine sera and exosomes. BMC Genom. 2016, 17, 630. [Google Scholar] [CrossRef] [PubMed]
- Fenech, M.; El-Sohemy, A.; Cahill, L.; FePleasrguson, L.R.; French, T.-A.C.; Tai, E.S.; Milner, J.; Koh, W.-P.; Xie, L.; Zucker, M. Nutrigenetics and nutrigenomics: Viewpoints on the current status and applications in nutrition research and practice. Lifestyle Genom. 2011, 4, 69–89. [Google Scholar] [CrossRef] [PubMed]
- García-Segura, L.; Pérez-Andrade, M.; Miranda-Ríos, J. The Emerging Role of MicroRNAs in the Regulation of Gene Expression by Nutrients. J. Nutrigenet. Nutrigenom. 2013, 6, 16–31. [Google Scholar] [CrossRef] [PubMed]
- Bradford, B.J.; Mullins, C.R. Invited review: Strategies for promoting productivity and health of dairy cattle by feeding nonforage fiber sources. J. Dairy Sci. 2012, 95, 4735–4746. [Google Scholar] [CrossRef] [PubMed]
- Mowrey, A.; Spain, J. Results of a nationwide survey to determine feedstuffs fed to lactating dairy cows. J. Dairy Sci. 1999, 82, 445–451. [Google Scholar] [CrossRef]
- Ipharraguerre, I.; Clark, J. Soyhulls as an alternative feed for lactating dairy cows: A review. J. Dairy Sci. 2003, 86, 1052–1073. [Google Scholar] [CrossRef]
- Ipharraguerre, I.; Ipharraguerre, R.; Clark, J. Performance of lactating dairy cows fed varying amounts of soyhulls as a replacement for corn grain. J. Dairy Sci. 2002, 85, 2905–2912. [Google Scholar] [CrossRef]
- Ehrlich, W.K.; Cowan, R.T.; Reid, A. Use of whole cotton seed as a dietary supplement for grazing dairy cows. Aust. J. Exp. Agr. 1993, 33, 283–286. [Google Scholar] [CrossRef]
- Müller, M.; Kersten, S. Nutrigenomics: Goals and strategies. Nat. Rev. Genet. 2003, 4, 315. [Google Scholar] [CrossRef]
- Bionaz, M.; Osorio, J.; Loor, J.J. Triennial lactation symposium: Nutrigenomics in dairy cows: nutrients, transcription factors, and techniques. J. Anim. Sci. 2015, 93, 5531–5553. [Google Scholar] [CrossRef]
- Toral, P.G.; Hervás, G.; Frutos, P. Role of nutrigenomics in ruminant milk production. Albéitar 2013, 163, 48–49. [Google Scholar]
- Dolganiuc, A.; Petrasek, J.; Kodys, K.; Catalano, D.; Mandrekar, P.; Velayudham, A.; Szabo, G. MicroRNA expression profile in Lieber-DeCarli diet-induced alcoholic and methionine choline deficient diet-induced nonalcoholic steatohepatitis models in mice. Alcohol Clin. Exp. Res. 2009, 33, 1704–1710. [Google Scholar] [CrossRef] [PubMed]
- Drummond, M.J.; Glynn, E.L.; Fry, C.S.; Dhanani, S.; Volpi, E.; Rasmussen, B.B. Essential amino acids increase microRNA-499, -208b, and -23a and downregulate myostatin and myocyte enhancer factor 2C mRNA expression in human skeletal muscle. J. Nutr. 2009, 139, 2279–2284. [Google Scholar] [CrossRef] [PubMed]
- Dey, N.; Das, F.; Mariappan, M.M.; Mandal, C.C.; Ghosh-Choudhury, N.; Kasinath, B.S.; Choudhury, G.G. MicroRNA-21 orchestrates high glucose-induced signals to TOR complex 1, resulting in renal cell pathology in diabetes. J. Biol. Chem. 2011, 286, 25586–25603. [Google Scholar] [CrossRef] [PubMed]
- Davidson, L.A.; Wang, N.; Shah, M.S.; Lupton, J.R.; Ivanov, I.; Chapkin, R.S. n-3 Polyunsaturated fatty acids modulate carcinogen-directed non-coding microRNA signatures in rat colon. Carcinogenesis 2009, 30, 2077–2084. [Google Scholar] [CrossRef] [PubMed]
- Nikolova, G.; Lammert, E. Interdependent development of blood vessels and organs. Cell Tissue Res. 2003, 314, 33–42. [Google Scholar] [CrossRef]
- Robbins, P.D.; Morelli, A.E. Regulation of immune responses by extracellular vesicles. Nat. Rev. Immunol. 2014, 14, 195. [Google Scholar] [CrossRef]
- Li, M.; Zeringer, E.; Barta, T.; Schageman, J.; Cheng, A.; Vlassov, A.V. Analysis of the RNA content of the exosomes derived from blood serum and urine and its potential as biomarkers. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2014, 369, 20130502. [Google Scholar] [CrossRef]
- Siomi, H.; Siomi, M.C. Posttranscriptional regulation of microRNA biogenesis in animals. Mol. Cell. 2010, 38, 323–332. [Google Scholar] [CrossRef]
- Vegh, P.; Magee, D.A.; Nalpas, N.C.; Bryan, K.; McCabe, M.S.; Browne, J.A.; Conlon, K.M.; Gordon, S.V.; Bradley, D.G.; MacHugh, D.E. MicroRNA profiling of the bovine alveolar macrophage response to Mycobacterium bovis infection suggests pathogen survival is enhanced by microRNA regulation of endocytosis and lysosome trafficking. Tuberculosis 2015, 95, 60–67. [Google Scholar] [CrossRef]
- Lykken, E.A.; Li, Q.-J. The microRNA MIR-191 supports T cell survival following common γ chain signaling. J. Biol. Chem. 2016, 291, 23532–23544. [Google Scholar] [CrossRef] [PubMed]
- Gaziel-Sovran, A.; Hernando, E. miRNA-mediated GALNT modulation of invasion and immune suppression: A sweet deal for metastatic cells. Oncoimmunology 2012, 1, 746–748. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Zhang, S.; Li, Z.; Wang, H.; Li, Z.; Hu, Y.; Chen, H.; Zhang, X.; Cui, L.; Zhang, J. miR-125b-5p and miR-99a-5p downregulate human γδ T-cell activation and cytotoxicity. Cell. Mol. Immunol. 2019, 16, 112. [Google Scholar] [CrossRef] [PubMed]
| Items | Experiment Diet | |
|---|---|---|
| Control Group | Tested Group | |
| Ingredient, % of DM | ||
| Alfalfa hay | 18.67 | 9.70 |
| Corn silage | 27.39 | 24.88 |
| Steam-flaked corn | 23.20 | 24.25 |
| Soybean meal | 8.22 | 8.21 |
| Cottonseed meal | 9.54 | 9.54 |
| Beet pulp | 5.68 | 5.68 |
| Distillers dried grains with solubles | 3.78 | 3.77 |
| Whole cotton seed | — | 5.72 |
| Soybean hull | — | 4.73 |
| Bergagat T300 | 1.04 | 1.04 |
| Premix | 1.86 | 1.84 |
| Chemical composition, % of DM | ||
| CP | 16.02 | 16.34 |
| EE | 4.20 | 5.18 |
| RDP(%CP) | 58.61 | 56.64 |
| NDF | 31.01 | 31.84 |
| f NDF | 20.92 | 15.67 |
| peNDF | 11.61 | 11.63 |
| ADF | 21.78 | 22.23 |
| NFC | 40.48 | 38.92 |
| Starch | 25.35 | 25.49 |
| NEL, Mcal/kg | 1.61 | 1.62 |
| Items | Concentration (pg/ μL) | miRNAs Content (%) |
|---|---|---|
| Control 1 | 759.7 | 76 |
| Control 2 | 3045.6 | 75 |
| Control 3 | 301.1 | 62 |
| Tested 1 | 950.8 | 72 |
| Tested 2 | 505.0 | 61 |
| Tested 3 | 358.8 | 52 |
| Item | Control Group | Tested Group | ||||
|---|---|---|---|---|---|---|
| Control 1 | Control 2 | Control 3 | Tested 1 | Tested 2 | Tested 3 | |
| Number | 230 | 263 | 240 | 258 | 242 | 238 |
| Mean | 244 | 246 | ||||
| miRNA-ID | Base Mean | Fold Change | p-Value |
|---|---|---|---|
| bta-miR-126-5p | 55.80 | 5.01 | 0.03 |
| bta-miR-199a-3p | 80.07 | 5.94 | 0.01 |
| bta-miR-2285bc | 17.35 | 0.01 | 0.00 |
| bta-miR-2288 | 17.49 | 0.02 | 0.01 |
| bta-miR-3613a | 9.23 | 0.02 | 0.04 |
| bta-miR-450b | 13.45 | 20.10 | 0.02 |
| bta-miR-500 | 69.45 | 2.52 | 0.05 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Quan, S.; Nan, X.; Wang, K.; Jiang, L.; Yao, J.; Xiong, B. Different Diets Change the Expression of Bovine Serum Extracellular Vesicle-miRNAs. Animals 2019, 9, 1137. https://doi.org/10.3390/ani9121137
Quan S, Nan X, Wang K, Jiang L, Yao J, Xiong B. Different Diets Change the Expression of Bovine Serum Extracellular Vesicle-miRNAs. Animals. 2019; 9(12):1137. https://doi.org/10.3390/ani9121137
Chicago/Turabian StyleQuan, Suyu, Xuemei Nan, Kun Wang, Linshu Jiang, Junhu Yao, and Benhai Xiong. 2019. "Different Diets Change the Expression of Bovine Serum Extracellular Vesicle-miRNAs" Animals 9, no. 12: 1137. https://doi.org/10.3390/ani9121137
APA StyleQuan, S., Nan, X., Wang, K., Jiang, L., Yao, J., & Xiong, B. (2019). Different Diets Change the Expression of Bovine Serum Extracellular Vesicle-miRNAs. Animals, 9(12), 1137. https://doi.org/10.3390/ani9121137






