Sanitary Emergencies at the Wild/Domestic Caprines Interface in Europe
Simple Summary
Abstract
1. Introduction
2. Major Emergencies in Native Wild Caprinae in Europe
3. Conclusions
- (1)
- sheep and goats can no longer be considered the exclusive or the main reservoir of M. conjunctivae for wild Caprinae;
- (2)
- following suspected infection from livestock, chamois (Rupicapra spp.) and ibex (Capra spp.) have rapidly turned into the exclusive reservoir hosts of S. scabiei wherever SM outbreaks have been reported in wild ruminant hosts; and
- (3)
- similar to (2), Southern chamois R. pyrenaica pyrenaica is currently deemed to be the single reservoir of the specific BDV4 variant, and the Alpine ibex is considered the single reservoir of B. melitensis in the French Alps.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Carranza, J. Ungulates and their management in Spain. In European Ungulates and Their Management in the 21th Century, 1st ed.; Apollonio, M., Andersen, R., Putman, R., Eds.; Cambridge University Press: Cambridge, UK, 2010; pp. 419–439. [Google Scholar]
- Apollonio, M.; Ciuti, S.; Pedrotti, L. Ungulates and their management in Italy. In European Ungulates and Their Management in the 21th Century, 1st ed.; Apollonio, M., Andersen, R., Putman, R., Eds.; Cambridge University Press: Cambridge, UK, 2010; pp. 475–506. [Google Scholar]
- Carnevali, L.; Pedrotti, L.; Riga, F.; Toso, S. Banca Dati Ungulati: Status, distribuzione, consistenza, gestione e prelievo venatorio delle popolazioni di Ungulati in Italia. Rapporto 2001–2005. Biol. Cons. Fauna 2009, 117, 1–168. [Google Scholar]
- Tosi, G.; Pedrotti, L.; Mustoni, A.; Carlini, E. Lo stambecco in Lombardia e sull’Arco Alpino; Istituto Oikos: Padova, Italy, 2012; p. 250. ISBN 978-88-908266-1-0. [Google Scholar]
- Latini, R.; Gentile, L.; Asprea, A.; Pagliaroli, D.; Di Pirro, V.; Argenio, A.; Monaco, A. Life is a risky business: Status and conservation perspectives of the last ancient population of endemic Apennine chamois (Rupicapra pyrenaica ornata). In Proceedings of the Chamois International Congress, Majella National Park, Italy, 17–19 June 2014; Antonucci, A., Di Domenico, G., Eds.; 2014. [Google Scholar]
- Pérez, J.M.; Granados, J.E.; Soriguer, R.C.; Fandos, P.; Márquez, F.J.; Crampe, J.P. Distribution, status and conservation problems of the Spanish Ibex, Capra pyrenaica Mammalia: Artiodactyla. Mammal Rev. 2002, 32, 26–39. [Google Scholar] [CrossRef]
- Rüttimann, S.; Giacometti, M.; McElligott, A.G. Effect of domestic sheep on chamois activity, distribution and abundance on sub-alpine pastures. Eur. J. Wildl. Res. 2008, 54, 110–116. [Google Scholar] [CrossRef]
- Richomme, C.; Gauthier, D.; Fromont, E. Contact rates and exposure to inter-species disease transmission in mountain ungulates. Epidemiol. Infect. 2006, 134, 21–30. [Google Scholar] [CrossRef] [PubMed]
- European Wildlife Disease Association. Available online: https://ewda.org/ (accessed on 24 October 2019).
- Groupe d’Etude sur l’Ecopathologie de la Faune Sauvage de Montagne. Available online: https://www.geefsm.eu/ (accessed on 24 October 2019).
- Gauthier, D.; Hars, J.; Rossi, L. Brucellosis in free ranging chamois Rupicapra rupicapra and its relationships with domestic breeding. In Proceedings of the Third Conference of European Wildlife Disease Association, Edinburg, Scotland, 16–20 September 1998. [Google Scholar]
- Ferroglio, E.; Gennero, S.; Rossi, L.; Tolari, F. Monitoraggio di un focolaio di brucellosi nel camoscio alpino. J. Mt. Ecol. 2003, 7, 229–232. [Google Scholar]
- Ferroglio, E.; Gennero, S.; Pasino, M.; Bergagna, S.; Dondo, A.; Grattarola, C.; Rondoletti, M.; Bassano, B. Cohabitation of a Brucella melitensis infected Alpine ibex Capra ibex with domestic small ruminants in an enclosure in Gran Paradiso National Park, in Western Italian Alps. Eur. J. Wildl. Res. 2007, 53, 158–160. [Google Scholar] [CrossRef]
- Godfroid, J.; Garin-Bastuji, B.; Saegerman, C.; Blasco, J.M. Brucellosis in terrestrial wildlife. Rev. Sci. Tech. 2013, 32, 27–42. [Google Scholar] [CrossRef]
- National Research Council. Brucellosis in the Greater Yellowstone Area; The National Academies Press: Washington, DC, USA, 1998; pp. 107–120. [Google Scholar]
- Rhyan, C.J.; Nol, P.; Quance, C.; Gertonson, A.; Belfrage, J.; Harris, L.; Straka, K.; Robbe-Austerman, S. Transmission of Brucellosis from Elk to Cattle and Bison, Greater Yellowstone Area, USA, 2002–2012. Emerg. Infect. Dis. 2013, 19, 1992–1995. [Google Scholar] [CrossRef]
- Garin-Bastuji, B.; Hars, J.; Drapeau, A.; Cherfa, M.A.; Game, Y.; Le Horgne, J.M.; Rautureau, S.; Maucci, E.; Pasquier, J.J.; Jay, M.; et al. Reemergence of Brucella melitensis infection in wildlife, France. Emerg. Infect. Dis. 2014, 20, 1570–1571. [Google Scholar] [CrossRef]
- Mick, V.; Le Carrou, G.; Corde, Y.; Game, Y.; Jay, M.; Garin-Bastuji, B. Brucella melitensis in France: Persistence in wildlife and probable spillover from Alpine ibex to domestic animals. PLoS ONE 2014, 9, e94168. [Google Scholar] [CrossRef]
- Hars, J.; Rautureau, S.; Vaniscotte, A.; Herbaux, J.P.; Pasquier, J.J.; Depecker, A.; Le Bourg, V.; Game, Y.; Toigo, C.; Mick, V.; et al. La brucellose des bouquetins du massif du Bargy Haute-Savoie: Oùenest-on en 2015? Bull. Épidémiol. Santé Anim. Aliment. 2015, 70, 14–18. [Google Scholar]
- Ponsart, C.; Garin-Bastuji, B.; Riou, M.; Locatelli, Y.; Fadeau, A.; Jaÿ, M.; Jacques, I.; Simon, R.; Perrot, L.; Freddi, L.; et al. Vaccination anti-brucellique du Bouquetin des Alpes (Capra ibex), une option pour l’assainissement du massif du Bargy? Comparaison du niveau d’innocuité conjonctivale du vaccin rev.1 chez le Bouquetin des Alpes et la chèvre domestique (C. hircus). Épidémiol. Santé Anim. 2019, 74, 25–42. [Google Scholar]
- Anses. Avis de l’Agence nationale de sécurité sanitaire de l’alimentation, de l’environnement et du travail relatif à «l’évaluation approfondie et réactualisée de mesures de maîtrise du foyer de brucellose chez les bouquetins du Bargy. 2017. Available online: https://www.anses.fr/fr/system/files/SABA2016SA0229.pdf (accessed on 4 November 2019).
- Rushton, J.; Lyons, N. Economic impact of Bluetongue: A review of the effects on production. Vet. Ital. 2015, 51, 401–406. [Google Scholar] [PubMed]
- Coetzer, J.A.W.; Tustin, R.C. Infectious Diseases of Livestock, 2nd ed.; Oxford University Press: Cape Town, South Africa, 2004. [Google Scholar]
- Rossi, S.; Pioz, M.; Beard, E.; Durand, B.; Gibert, P.; Gauthier, D.; Klein, F.; Maillard, D.; Saint-Andrieux, C.; Saubusse, T.; et al. Bluetongue Dynamics in French Wildlife: Exploring the Driving Forces. Transbound Emerg. Dis. 2014, 61, 12–24. [Google Scholar] [CrossRef]
- García-Bocanegra, I.; Arenas-Montes, A.; Lorca-Oró, C.; Pujols, J.; González, M.Á.; Napp, S.; Gomez-Guillon, F.; Zorrilla, I.; San Miguel, E.; Arenas, A. Role of wild ruminants in the epidemiology of bluetongue virus serotypes 1, 4 and 8 in Spain. Vet. Res. 2011, 421, 88. [Google Scholar] [CrossRef]
- López-Olvera, J.R.; Falconi, C.; Férnandez-Pacheco, P.; Fernández-Pinero, J.; Sánchez, M.A.; Palma, A.; Herruzo, I.; Vicente, J.; Jiménez-Clavero, M.A.; Arias, M.; et al. Experimental infection of European red deer Cervus elaphus with bluetongue virus serotypes 1 and 8. Vet. Microbiol. 2010, 145, 148–152. [Google Scholar] [CrossRef]
- Lorca-Oró, C.; Pujols, J.; Arenas, A.; Gómez-Guillamón, F.; Zorrilla, I.; Domingo, M.; Arenas-Montés, A.; Ruano, M.J.; García-Bocanegra, I. Epidemiological surveillance of bluetongue virus serotypes 1, 4 and 8 in Spanish ibex Capra pyrenaica hispanica in southern Spain. Vet. Microbiol. 2011, 149, 230–235. [Google Scholar] [CrossRef]
- Corbière, F.; Nussbaum, S.; Alzieu, J.P.; Lemaire, M.; Meyer, G.; Foucras, G.; Schelcher, F. Bluetongue virus serotype 1 in wild ruminants, France, 2008–2010. J. Wildl. Dis. 2012, 484, 1047–1051. [Google Scholar] [CrossRef]
- Casaubon, J.; Chaignat, V.; Vogt, H.R.; Michel, A.O.; Thür, B.; Ryser-Degiorgis, M.P. Survey of bluetongue virus infection in free-ranging wild ruminants in Switzerland. BMC Vet. Res. 2013, 9, 166. [Google Scholar] [CrossRef]
- Grego, E.; Sossella, M.; Bisanzio, D.; Stella, M.C.; Giordana, G.; Pignata, L.; Tomassone, L. Wild ungulates as sentinel of BTV-8 infection in piedmont areas. Vet. Microbiol. 2014, 174, 93–99. [Google Scholar] [CrossRef]
- Lorca-Oró, C.; Pujols, J.; García-Bocanegra, I.; Mentaberre, G.; Granados, J.E.; Solanes, D.; Fandos, P.; Galindo, I.; Domingo, M.; Lavín, S.; et al. Protection of Spanish Ibex (Capra pyrenaica) against Bluetongue Virus Serotypes 1 and 8 in a Subclinical Experimental Infection. PLoS ONE 2012, 7, e36380. [Google Scholar] [CrossRef] [PubMed]
- Rossi, S.; Viarouge, C.; Faure, E.; Gilot-Fromont, E.; Gache, K.; Gibert, P.; Verheyden, H.; Hars, J.; Klein, F.; Maillard, D.; et al. Exposure of Wildlife to the Schmallenberg Virus in France (2011–2014): Higher, Faster, Stronger (than Bluetongue)! Transbound. Emerg. Dis. 2017, 64, 354–363. [Google Scholar] [CrossRef] [PubMed]
- Miller, D.S.; Weiser, G.C.; Aune, K.; Roeder, B.; Atkinson, M.; Anderson, N.J.; Roffe, T.; Keating, K.A.; Chapman, P.L.; Kimberling, C.; et al. Shared Bacterial and Viral Respiratory Agents in Bighorn Sheep (Ovis canadensis), Domestic Sheep (Ovis aries), and Goats (Capra hircus) in Montana. Vet. Med. Int. 2011, 2011, 162520. [Google Scholar] [CrossRef] [PubMed]
- Citterio, C.V.; Luzzago, C.; Sala, M.; Sironi, G.; Gatti, P.; Gaffuri, A.; Lanfranchi, P. Serological study of a population of Alpine chamois Rupicapra rupicapra affected by an outbreak of respiratory disease. Vet. Rec. 2003, 15319, 592–596. [Google Scholar] [CrossRef]
- Giacometti, M.; Janovsky, M.; Belloy, L.; Frey, J. Infectious keratoconjunctivitis of ibex, chamois and other Caprinae. Rev. Sci. Tech. 2002, 212, 335–345. [Google Scholar] [CrossRef]
- Crampe, J.-P.; (Parc National des Pyrénées, Tarbes, France). Personal communication, 2008.
- Jones, G.E. Infectious keratoconjunctivitis. In Diseases of Sheep, 2nd ed.; Blackwell Scientific Publications: Oxford, UK, 1991; pp. 280–283. [Google Scholar]
- Gauthier, D. La kérato-conjonctivite infectieuse du chamois; étude épidémiologique dans le département de la Savoie 1983–1990. Ph.D. Thesis, National Veterinary School of Lyon, University of Lyon, Lyon, France, 1991. [Google Scholar]
- Degiorgis, M.P.; Frey, J.; Nicolet, J.; Abdo, E.M.; Fatzer, R.; Schlatter, Y.; Reist, S.; Janovsky, M.; Giacometti, M. An outbreak of infectious keratoconjunctivitis in Alpine chamois in Simmental-Gruyeres, Switzerland. Schweiz. Archiv. Tierheilkd. 2000, 1429, 520–527. [Google Scholar]
- Arnal, M.; Herrero, J.; de la Fe, C.; Revilla, M.; Prada, C.; Martínez-Durán, D.; Gómez-Martín, A.; Fernández-Arberas, O.; Amores, J.; Contreras, A.; et al. Dynamics of an infectious keratoconjunctivitis outbreak by Mycoplasma conjunctivae on Pyrenean Chamois Rupicapra p. pyrenaica. PLoS ONE 2013, 8, e61887. [Google Scholar] [CrossRef]
- Degiorgis, M.; Obrecht, E.; Ryser, A. The possible role of eye-frequenting flies in the transmission of Mycoplasma conjunctivae. J. Swiss Entomol. Soc. 1999, 72, 3–4. [Google Scholar]
- Gelormini, G.; Gauthier, D.; Vilei, E.M.; Crampe, J.P.; Frey, J.; Ryser-Degiorgis, M.P. Infectious keratoconjunctivitis in wild Caprinae: Merging field observations and molecular analyses sheds light on factors shaping outbreak dynamics. BMC Vet. Res. 2017, 13, 67. [Google Scholar] [CrossRef]
- Giacometti, M.; Janovsky, M.; Jenny, H.; Nicolet, J.; Belloy, L.; Goldschmidt-Clermont, E.; Frey, J. Mycoplasma conjunctivae infection is not maintained in alpine chamois in eastern Switzerland. J. Wildl. Dis. 2002, 38, 297–304. [Google Scholar] [CrossRef]
- Mavrot, F.M.; Vilei, E.; Marreros, N.; Signer, C.; Ryser-Degiorgis, M.P. Occurrence, quantification, and genotyping of Mycoplasma conjunctivae in wild Caprinae with and without infectious keratoconjunctivitis. J. Wildl. Dis. 2012, 48, 619–631. [Google Scholar] [CrossRef]
- Ryser-Degiorgis, M.P.; Bischof, D.F.; Marreros, N.; Willisch, C.; Signer, C.; Filli, F.; Brosi, G.; Frey, J.; Vilei, E.M. Detection of Mycoplasma conjunctivae in the eyes of healthy, free-ranging Alpine ibex: Possible involvement of Alpine ibex as carriers for the main causing agent of infectious keratoconjunctivitis in wild Caprinae. Vet. Microbiol. 2009, 134, 368–374. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Aguilar, X.; Cabezon, O.; Frey, J.; Velarde, R.; Serrano, E.; Colom-Cadena, A.; Gelormini, G.; Marco, I.; Mentaberre, G.; Lavin, S.; et al. Long-term dynamics of Mycoplasma conjunctivae at the wildlife-livestock interface in the Pyrenees. PLoS ONE 2017, 10, e0186069. [Google Scholar] [CrossRef] [PubMed]
- Frolich, K.; Jung, S.; Ludwig, A.; Lieckfeldt, D.; Gibert, P.; Gauthier, D.; Hars, J. Detection of a newly described pestivirus of Pyrenean chamois (Rupicapra pyrenaica pyrenaica) in France. J. Wildl. Dis. 2005, 41, 606–610. [Google Scholar] [CrossRef] [PubMed]
- Alzieu, J.P.; Deplanche, M.; Moulignie, M.; Letellier, C.; Lacroux, C.; Duquesnel, R.; Baranowski, E.; Meyer, G.; Kerkhofs, P.; Schelcher, F. First description of pestivirus disease in Rupicapra pyrenaica pyrenaica. Bull. Acad. Vét. France 2004, 157, 49–54. [Google Scholar] [CrossRef]
- Marco, I.; Rosell, R.; Cabezón, O.; Mentaberre, G.; Casas, E.; Velarde, R.; Lavín, S. Border disease virus among chamois, Spain. Emerg. Infect. Dis. 2009, 15, 448–451. [Google Scholar] [CrossRef] [PubMed]
- Widen, F.; Meredith, A. Weissen-Boch, H. Other virus infection. In Infectious Diseases of Wild Mammals and Birds in Europe, 1st ed.; Gavier-Widen, D., Duff, P., Meredith, A., Eds.; Wliley-Blackwell: West Sussex, UK, 2012; pp. 249–281. [Google Scholar]
- Fernández-Sirera, L.; Cabezón, O.; Allepuz, A.; Rosell, R.; Riquelme, C.; Serrano, E.; Lavín, S.; Marco, I. Two different epidemiological scenarios of border disease in the populations of Pyrenean chamois Rupicapra p. pyrenaica after the first disease outbreaks. PLoS ONE 2012, 10, e51031. [Google Scholar] [CrossRef]
- Cavallero, S.; Marco, I.; Lavín, S.; D’Amelio, S.; López-Olvera, J.R. Polymorphisms at MHC class II DRB1 exon 2 locus in Pyrenean chamois Rupicapra pyrenaica pyrenaica. Infect. Genet. Evol. 2012, 5, 1020–1026. [Google Scholar] [CrossRef]
- Luzzago, C.; Ebranati, E.; Cabezón, O.; Fernández-Sirera, L.; Lavín, S.; Rosell, R.; Veo, C.; Rossi, L.; Cavallero, S.; Lanfranchi, P.; et al. Spatial and Temporal Phylogeny of Border Disease Virus in Pyrenean Chamois Rupicapra p. pyrenaica. PLoS ONE 2016, 11, e0168232. [Google Scholar] [CrossRef]
- Gilot-Fromont, E.; Garel, M.; Gibert, P.; Lambert, S.; Menaut, P.; Bonetti, B.; Game, Y.; Reynaud, G.; Foulché, K. Self-clearance of Pestivirus in a Pyrenean Chamois Rupicapra pyrenaica Population. J. Wildl. Dis. 2018, 54, 335–341. [Google Scholar] [CrossRef]
- Olde Riekerink, R.G.; Dominici, A.; Barkema, H.W.; de Smit, A.J. Seroprevalence of pestivirus in four species of alpine wild ungulates in the High Valley of Susa, Italy. Vet. Microbiol. 2005, 108, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Martin, C.; Letellier, C.; Caij, B.; Gauthier, D.; Jean, N.; Shaffii, A.; Saegerman, C. Epidemiology of Pestivirus infection in wild ungulates of the French South Alps. Vet. Microbiol. 2011, 27, 320–328. [Google Scholar] [CrossRef] [PubMed]
- Gaffuri, A.; Giacometti, M.; Tranquillo, V.M.; Magnino, S.; Cordioli, P.; Lanfranchi, P. Serosurvey of roe deer, chamois and domestic sheep in the central Italian Alps. J. Wildl. Dis. 2006, 423, 685–690. [Google Scholar] [CrossRef] [PubMed]
- Falconi, C.; Oleaga, A.; López-Olvera, J.R.; Casais, R.; Prieto, M.; Gortázar, C. Prevalence of antibodies against selected agents shared between Cantabrian chamois Rupicapra pyrenaica parva and domestic goats. Eur. J. Wildl. Res. 2010, 56, 319–325. [Google Scholar] [CrossRef]
- Fernández-Sirera, L.; (Univerdad Autonoma de Barcelona, Bellaterra, Spain). Personal communication, 2001.
- Alasaad, S.; Rossi, L.; Heukelbach, J.; Pérez, J.M.; Hamarsheh, O.; Otiende, M.; Zhu, X.Q. The neglected navigating web of the incomprehensibly emerging and re-emerging Sarcoptes mite. Infect. Genet. Evol. 2013, 17, 253–259. [Google Scholar] [CrossRef]
- Miller, C. Die Gamsraude in den Alpen. Z. Jagdwiss. 1986, 321, 42–46. [Google Scholar] [CrossRef]
- Pérez, J.M.; Ruiz-Martinez, I.; Granados, J.E.; Soriguer, R.; Fandos, P. The dynamics of sarcoptic mange in the ibex population of Sierra Nevada in Spain–Influence of climatic factors. J. Wild. Res. 1997, 2, 86–89. [Google Scholar]
- Leon-Vizcaino, L.; Ruiz de Ybañez, M.; Cubero, M.J.; Ortiz, J.M.; Espinosa, J.; Pérez, L.; Simon, M.; Alonso, F. Sarcoptic mange in spanish ibex from Spain. J. Wildl. Dis. 1999, 35, 647–659. [Google Scholar] [CrossRef]
- Fernández-Morán, J.; Gómez, S.; Ballesteros, F.; Quirós, P.; Benito, J.L.; Feliu, C.; Nieto, J.M. Epizootiology of sarcoptic mange in a population of cantabrian chamois Rupicapra pyrenaica parva in northwestern Spain. Vet. Parasitol. 1997, 15, 163–171. [Google Scholar] [CrossRef]
- González-Candela, M.; León-Vizcaíno, L.; Cubero-Pablo, M.J. Population effects of sarcoptic mange in barbary sheep Ammotragus lervia from Sierra Espuña Regional Park, Spain. J. Wildl. Dis. 2004, 40, 456–465. [Google Scholar] [CrossRef]
- Rossi, L.; Fraquelli, C.; Vesco, U.; Permunian, R.; Sommavilla, G.M.; Carmignola, G.; Da Pozzo, M.; Meneguz, P.G. Descriptive epidemiology of a scabies epidemic in chamois in the Dolomite Alps, Italy. Eur. J. Wildl. Res. 2007, 53, 131–141. [Google Scholar] [CrossRef]
- Pérez-Barbería, F.J.; Palacios, B.; González-Quirós, P.; Cano, M.; Nores, C.; Díaz, A. La evolución de la población del rebeco en la cordillera Cantábrica. In El Rebeco Cantábrico (Rupicapra pyrenaica parva). Conservación y Gestión de sus Poblaciones, 1st ed.; Pérez-Barbería, F.J., Palacios, B., Eds.; Ministerio de Medio Ambiente, Medio Rural y Marino. Naturaleza y Parques Nacionales: Madrid, Spain, 2009; pp. 106–125. [Google Scholar]
- Leon Vizcaino, L.; Astorga, R.; Escos, J.; Alonso, F.; Alados, C.; Contreras, A.; Cubero, M.J. Epidemiologıa de la sarna sarcoptica en el Parque Natural de las Sierras de Cazorla, Segura y Las Villas. In Proceedings of the International Congress on the Genus Capra in Europe, Ronda, Spain, 20–22 October 1992; Junta Rectora del Parque Natural Sierra de las Nieves, Consejerıa de Medio Ambiente, Junta de Andalucıa, Eds.; Junta de Andalucia: Sevilla, Spain, 1993. [Google Scholar]
- Rossi, L.; Meneguz, P.G.; De Martin, P.; Rodolfi, M. The epizootiology of sarcoptic mange in chamois, Rupicapra rupicapra, from the Italian eastern Alps. Parassitologia 1995, 37, 233–240. [Google Scholar] [PubMed]
- Mona, S.; Crestanello, B.; Bankhead-Dronnet, S.; Pecchioli, E.; Ingrosso, S.; D’Amelio, S.; Rossi, L.; Meneguz, P.G.; Bertorelle, G. Disentangling the effects of recombination, selection, and demography on the genetic variation at a major histocompatibility complex class II gene in the alpine chamois. Mol. Ecol. 2008, 1718, 4053–4067. [Google Scholar] [CrossRef] [PubMed]
- Schaschl, H.; Suchentrunk, F.; Morris, D.L.; Ben Slimen, H.; Smith, S.; Arnold, W. Sex-specific selection for MHC variability in Alpine chamois. BMC Evol. Biol. 2012, 15, 12–20. [Google Scholar] [CrossRef]
- Lavin, S.; Ruiz-Bascaran, M.; Marco, I.; Fondevila, M.D.; Ramis, A.J. Experimental infection of chamois Rupicapra pyrenaica parva with Sarcoptes scabiei derived from naturally infected goats. J. Vet. Med. B Infect. Dis. Vet. Public Health 2000, 479, 693–699. [Google Scholar] [CrossRef]
- Menzano, A.; Rambozzi, L.; Rossi, L. A severe episode of wildlife-derived scabies in domestic goats in Italy. Small Rum. Res. 2007, 70, 154–158. [Google Scholar] [CrossRef]
- Sandvik, T. Border disease virus: Time to take more notice? Vet. Rec. 2014, 174, 65–66. [Google Scholar] [CrossRef]
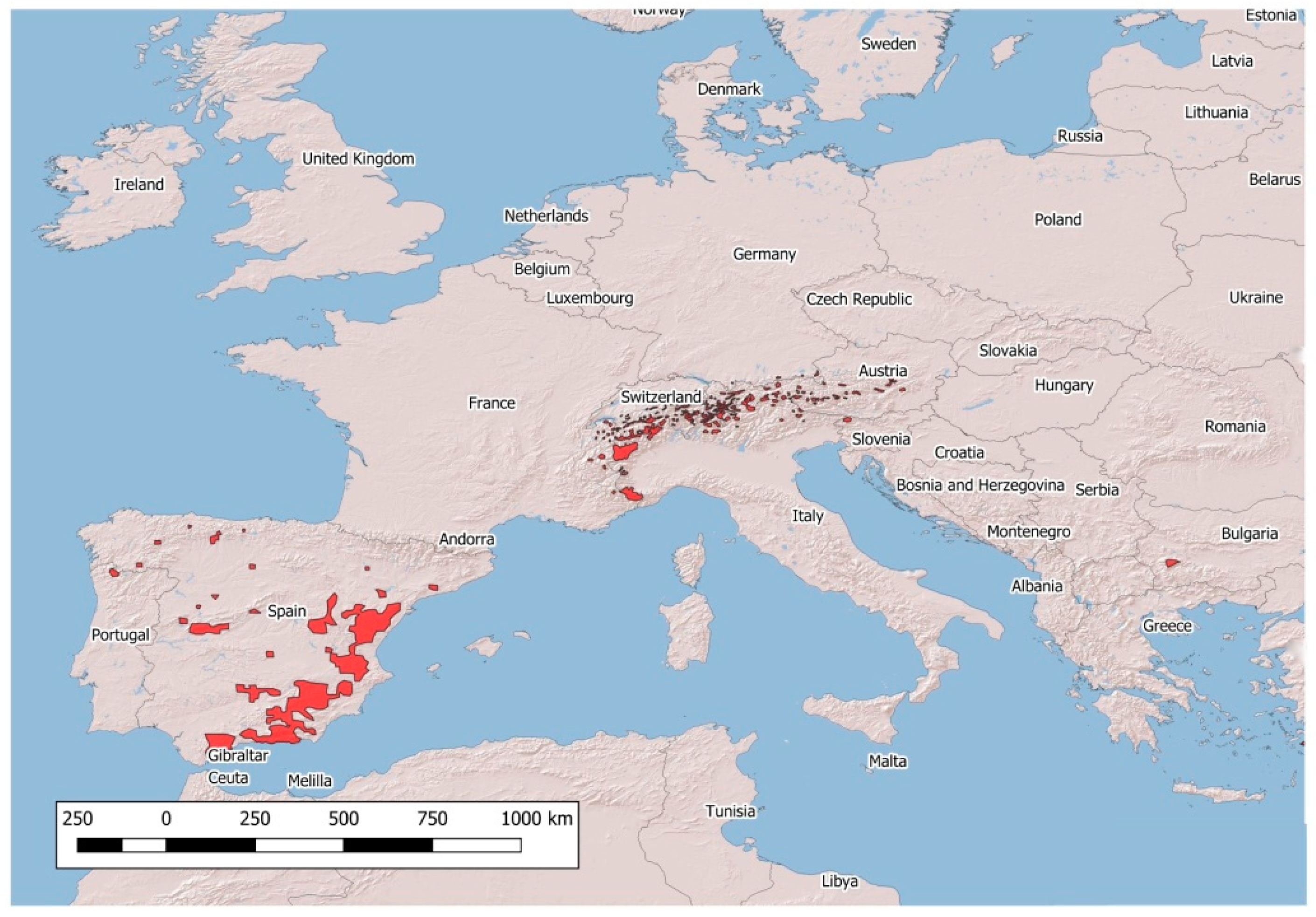
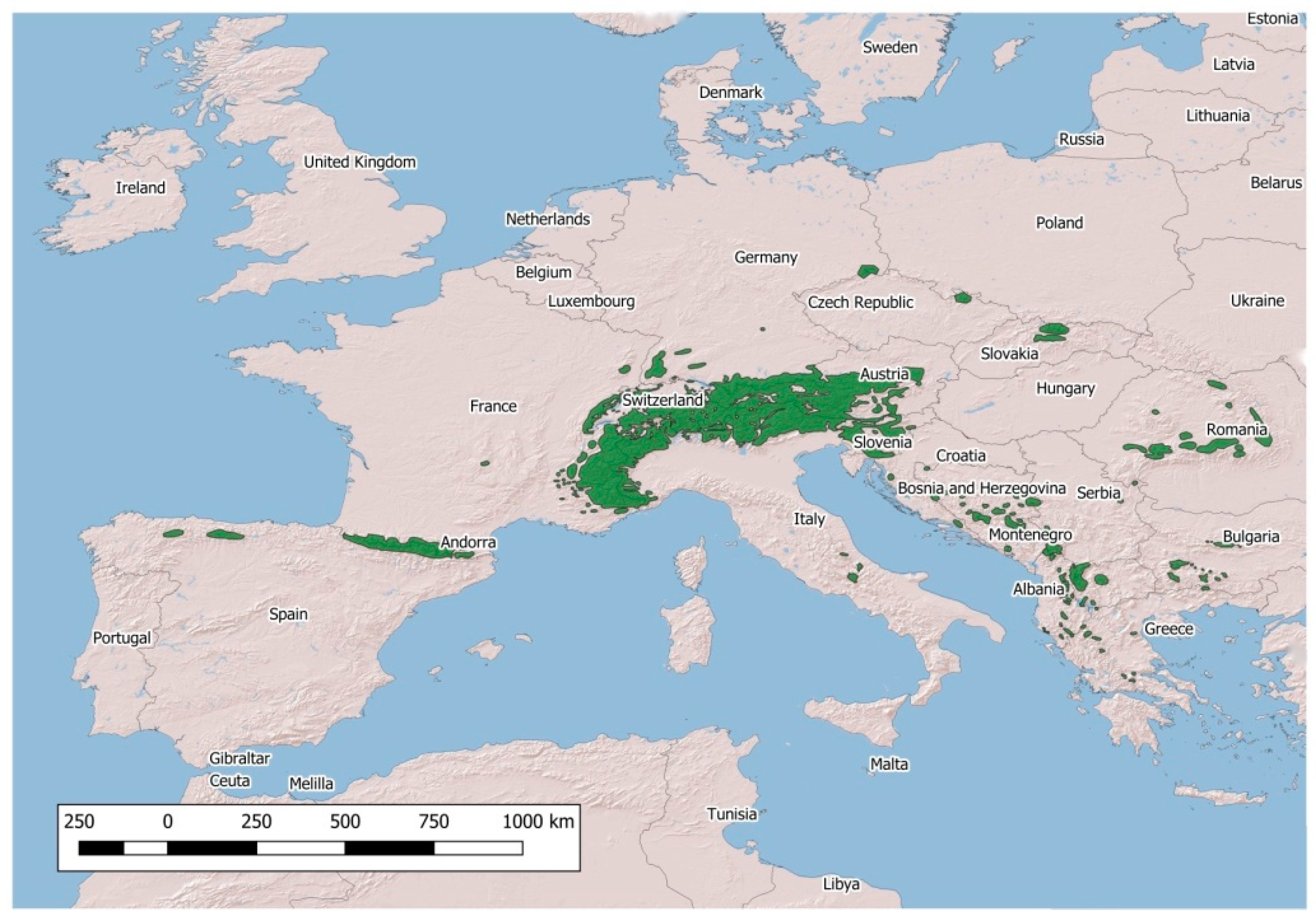
| Name of the Disease | Pathogen | Wild Hosts | Eradication Program in Livestock |
|---|---|---|---|
| Blue tongue | Blue tongue virus (Reoviridae) | Northern Chamois Southern Chamois Alpine Ibex Iberian ibex | yes |
| Pestivirosis | Border disease virus (strain 4) | Southern Chamois | no |
| Brucellosis | Brucella melitensis | Northern Chamois Alpine Ibex Iberian ibex | yes |
| Infectious kerato-conjunctivitis | Mycoplasma conjunctivae | Northern Chamois Southern Chamois Alpine Ibex Iberian ibex | no |
| Sarcoptic mange | Sarcoptes scabiei | Northern Chamois Southern Chamois Alpine Ibex Iberian Ibex | no |
| (1) |
| Mountain System | Capra spp. | ||||
|---|---|---|---|---|---|
| BRC | BT | IKC | PV | SM | |
| Penibaetic System | + | + | + | - | + |
| Central System | - | - | - | - | - |
| Iberian System | - | - | - | - | + |
| Pyrenees (E) | - | - | - | - | - |
| Pyrenees (F) | - | - | + | - | - |
| Western Alps (F) | + | - | + | - | - |
| Western Alps (I) | + | - | + | - | - |
| Western Alps (CH) | - | - | + | - | - |
| Eastern Alps (I) | - | - | - | - | + |
| Eastern Alps (A, D) | - | - | + | - | + |
| Eastern Alps (SLO) | - | - | - | - | + |
| Mountain System | Rupicapra spp. | ||||
|---|---|---|---|---|---|
| BRC | BT | IKC | PV | SM | |
| Cantabrian Mountains | - | - | + | - | + |
| Pyrenees (E) | - | - | + | + | - |
| Pyrenees (F) | - | - | + | + | - |
| Western Alps (F) | + | - | + | - | - |
| Western Alps (I) | + | - | + | - | - |
| Western Alps (CH) | - | - | + | - | - |
| Eastern Alps (I) | - | - | + | - | + |
| Eastern Alps (A, D) | - | - | + | - | + |
| Eastern Alps (SLO) | - | - | + | - | + |
| Tatra Mountains | - | - | - | - | - |
| Carpathian Mountains | - | - | - | - | - |
| Balkan Mountains | - | - | - | - | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rossi, L.; Tizzani, P.; Rambozzi, L.; Moroni, B.; Meneguz, P.G. Sanitary Emergencies at the Wild/Domestic Caprines Interface in Europe. Animals 2019, 9, 922. https://doi.org/10.3390/ani9110922
Rossi L, Tizzani P, Rambozzi L, Moroni B, Meneguz PG. Sanitary Emergencies at the Wild/Domestic Caprines Interface in Europe. Animals. 2019; 9(11):922. https://doi.org/10.3390/ani9110922
Chicago/Turabian StyleRossi, Luca, Paolo Tizzani, Luisa Rambozzi, Barbara Moroni, and Pier Giuseppe Meneguz. 2019. "Sanitary Emergencies at the Wild/Domestic Caprines Interface in Europe" Animals 9, no. 11: 922. https://doi.org/10.3390/ani9110922
APA StyleRossi, L., Tizzani, P., Rambozzi, L., Moroni, B., & Meneguz, P. G. (2019). Sanitary Emergencies at the Wild/Domestic Caprines Interface in Europe. Animals, 9(11), 922. https://doi.org/10.3390/ani9110922





