Horses Categorize Human Emotions Cross-Modally Based on Facial Expression and Non-Verbal Vocalizations
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects and Husbandry
2.2. Ethical Note
2.3. Visual and Acoustic Stimuli
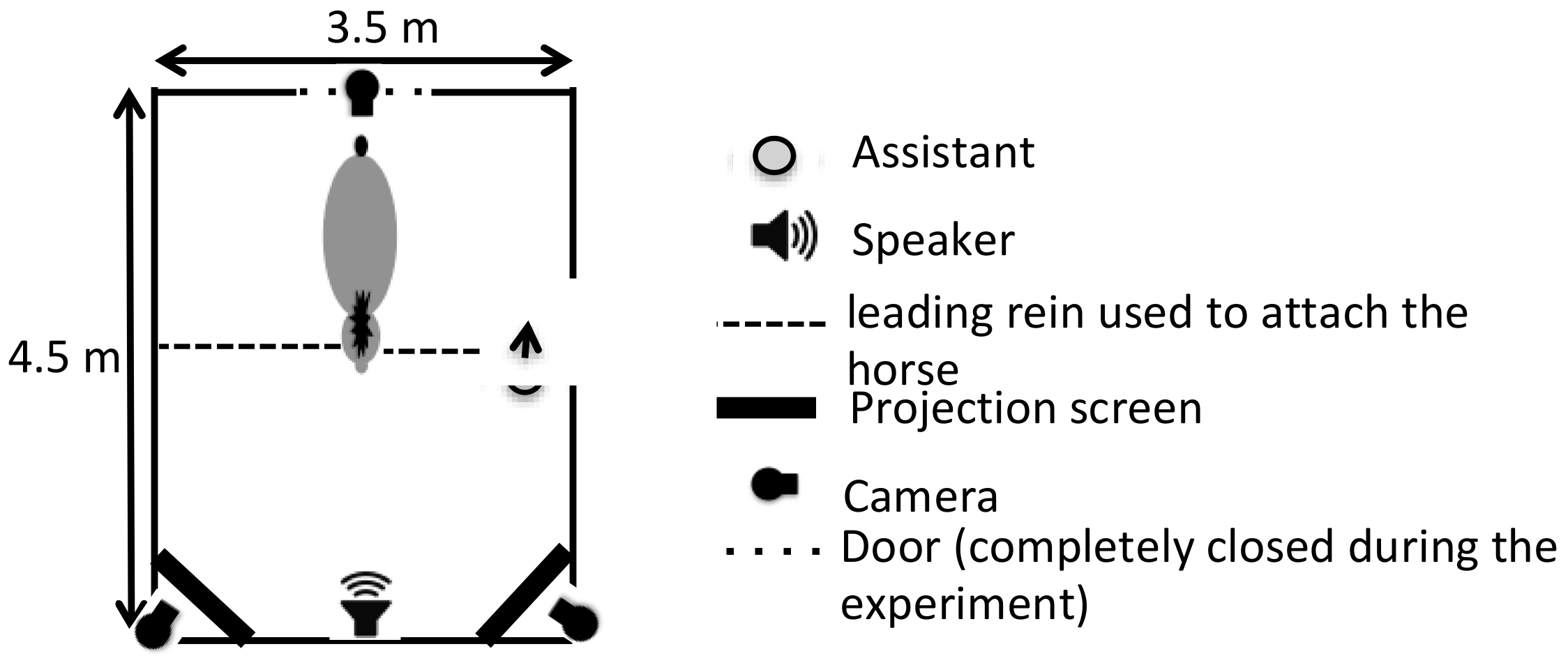
2.4. Set-Up and Procedure
- Familiarization. Horses were first familiarized with the experimental set-up by presenting two random pictures of nature while playing a recorded birdsong. The duration and number of sessions depended on the individual horse, but each horse was submitted to at least two familiarization sessions of a minimal duration of 5 min each. Familiarization stopped when the horse could stay attached in front of screen for 1 min without constraint with a heart rate lower than 80 bpm.
- Test session. Horses were tested only once, always in the afternoon. A test session consisted of 6 trials (three with the “joy” vocalization and three with the “anger” vocalization) with 5 s breaks between trials (with black screens and no sound played). Each trial lasted 15 s: the same vocalization was repeated, and the animated pictures were played on a loop, one on the left and one on the right of the horse. Thus, for each vocalization, a horse had two trials with the matching picture on one side and one trial with the matching picture on the other side. The sides of the matching picture were counterbalanced between the horses so that at the group level, there were an equal number of trials with the matching picture on the right and on the left side. The order of the vocalizations and the side of each picture were semi-randomly distributed between the trials: the same vocalization and the same configuration of pictures were never presented more than twice in a row.
2.5. Coding and Statistical Analyses
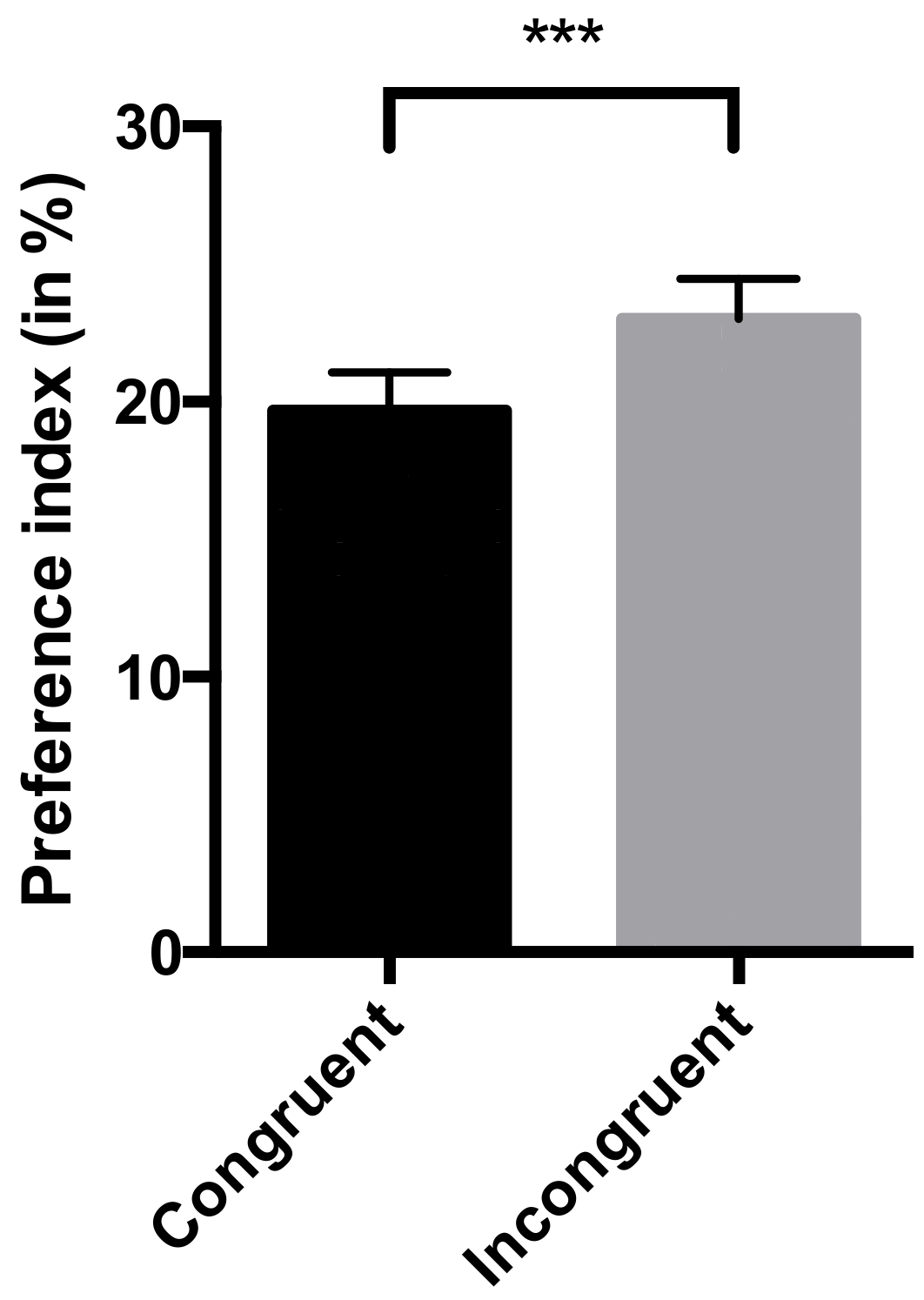
- Preference index: this measure was calculated for each picture (congruent and incongruent with the vocalization played) to investigate the multimodality of the recognition of human emotions. This preference index was defined as the percentage of time spent looking at one picture across the total duration of the trial (15 s). Horses were considered to be looking at a picture when their muzzle was directed towards this picture (within 45° [25]). The ICC was 0.92 (lower bound = 0.89), which is considered as an excellent interobserver reliability [40].
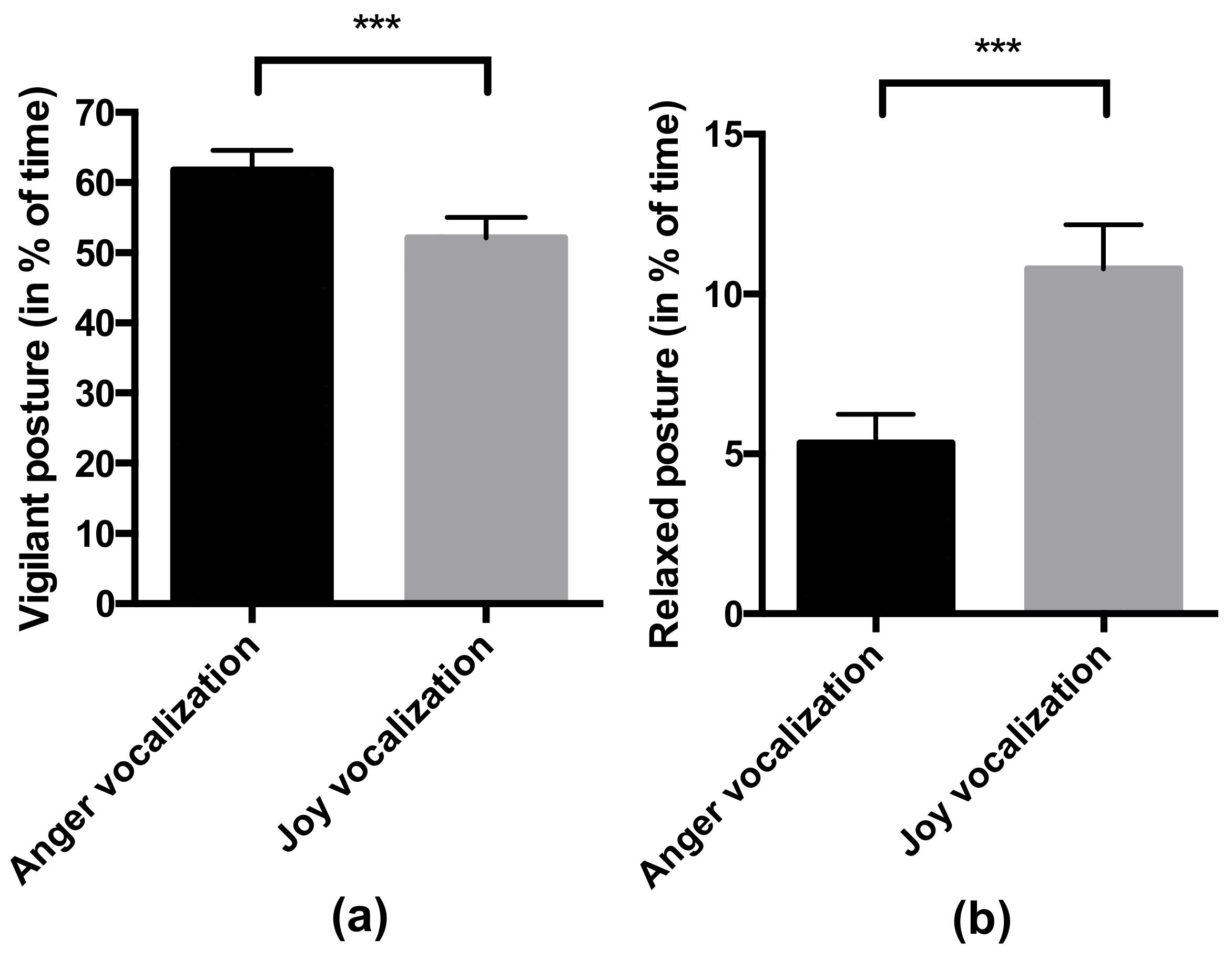
- Behavior in response to the valence of the vocalization: the percentage of time spent in a vigilant posture (the horse freezes, with the two ears oriented forwards and the head high [41,42,43]) and percentage of time spent in a relaxed posture (the horse relaxes its neckline muscles and puts its head down: angle between the neck and the withers is wider than 165°). For the vigilant posture, the ICC was 0.93 (lower bound = 0.87), which is considered as an excellent interobserver reliability [40]. For the relaxed posture, the ICC was 0.89 (lower bound = 0.80), considered as a good reliability [40].
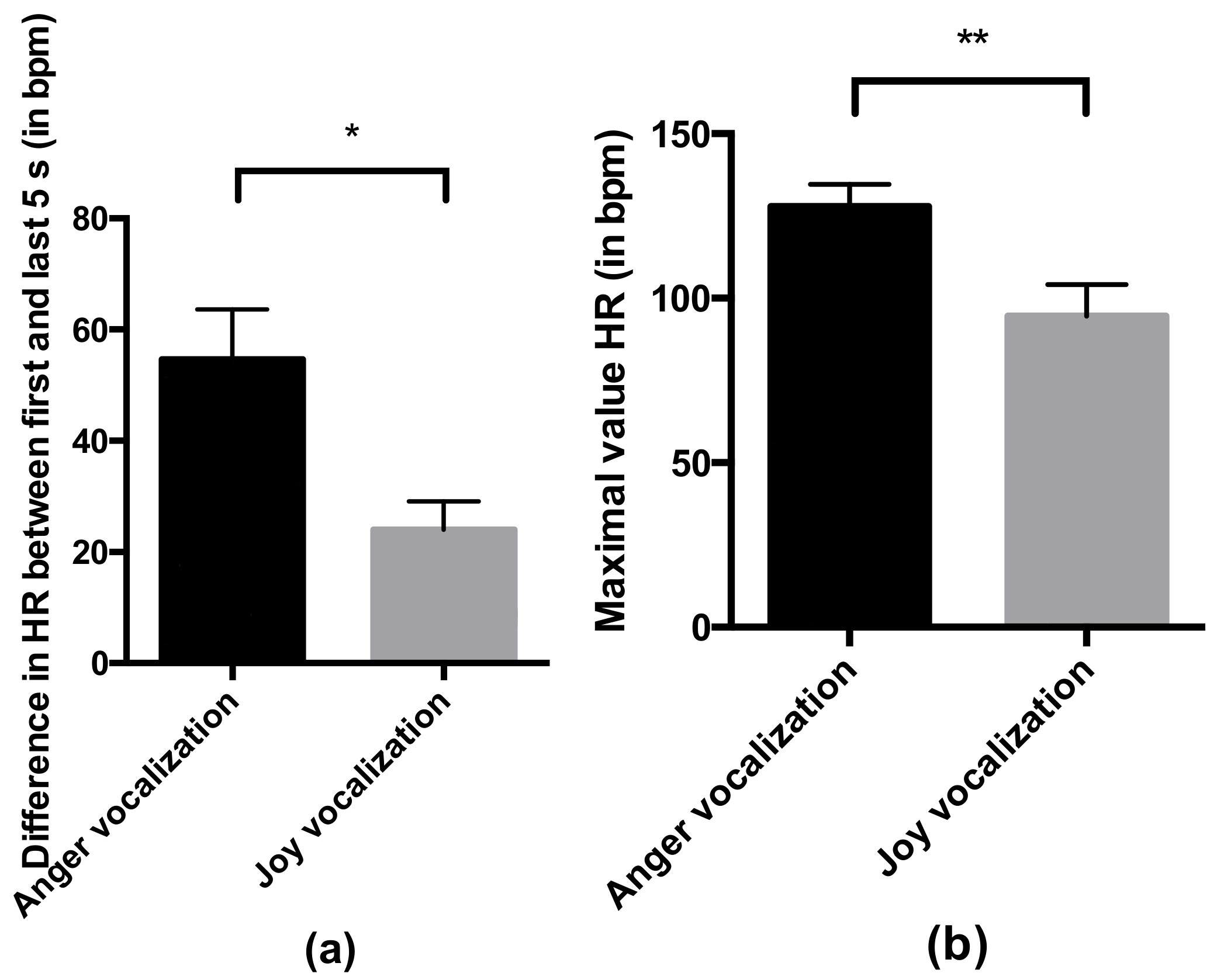
- Heart rate in response to the valence of the vocalization: the mean heart rate and the maximal value across the duration of the whole trial, and the difference in the mean heart rate between the first and last 5 s of the trial. As the trials followed each other closely and the heart rate might not have been reset to a basal level between the trials, we analyzed these variables during the first trial only. Due to technical issues with the heart monitor system, data were missing for several individuals (30 individuals were used to analyze the mean and maximal value of the heart rate, and 22 were used to analyze the difference in the heart rate between the first and last 5 s). The excluded individuals were average in their behavior (mean ± SEM of excluded individuals for the relaxed posture: 0.080 ± 0.023; global mean: 0.081 ± 0.008; mean of excluded individuals for the vigilant posture: 0.593 ± 0.075; global mean: 0.572 ± 0.020) and came from the two conditions (anger vocalization and joy vocalization). All horses were familiarized with the heart monitor system before the beginning of the experiment during the familiarization.
3. Results
4. Discussion
4.1. Horses are Capable of Cross-Modal Recognition of Human Emotions
4.2. Horses Responded Behaviorally and Physiologically to the Valence of Non-Verbal Vocalizations
5. Conclusions and Implications for the Horse-Human Relationship
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Waller, B.M.; Micheletta, J. Facial Expression in non-human animals. Emot. Rev. 2013, 5, 54–59. [Google Scholar] [CrossRef]
- Briefer, E.F. Vocal contagion of emotions in non-human animals. Proc. R. Soc. B Biol. Sci. 2018, 285, 20172783. [Google Scholar] [CrossRef] [PubMed]
- Kikusui, T.; Takigami, S.; Takeuchi, Y.; Mori, Y. Alarm pheromone enhances stress-induced hyperthermia in rats. Physiol. Behav. 2001, 72, 45–50. [Google Scholar] [CrossRef]
- Bredy, T.W.; Barad, M. Social modulation of associative fear learning by pheromone communication. Learn. Mem. 2009, 16, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Špinka, M. Social dimension of emotions and its implication for animal welfare. Appl. Anim. Behav. Sci. 2012, 138, 170–181. [Google Scholar] [CrossRef]
- Waller, B.M.; Whitehouse, J.; Micheletta, J. Macaques can predict social outcomes from facial expressions. Anim. Cogn. 2016, 19, 1031–1036. [Google Scholar] [CrossRef] [PubMed]
- Parr, L.A.; Heintz, M. Facial expression recognition in rhesus monkeys, Macaca mulatta. Anim. Behav. 2009, 77, 1507–1513. [Google Scholar] [CrossRef]
- Micheletta, J.; Whitehouse, J.; Parr, L.A.; Waller, B.M. Facial expression recognition in crested macaques (Macaca nigra). Anim. Cogn. 2015, 18, 985–990. [Google Scholar] [CrossRef]
- Parr, L.A. The discrimination of faces and their emotional content by chimpanzees (Pan troglodytes). Ann. N. Y. Acad. Sci. 2003, 1000, 56–78. [Google Scholar] [CrossRef]
- Albuquerque, N.; Guo, K.; Wilkinson, A.; Savalli, C.; Otta, E.; Mills, D. Dogs recognize dog and human emotions. Biol. Lett. 2016, 12, 20150883. [Google Scholar] [CrossRef]
- Müller, C.A.; Schmitt, K.; Barber, A.L.A.; Huber, L. Dogs can discriminate emotional expressions of human faces. Curr. Biol. 2015, 25, 601–605. [Google Scholar] [CrossRef] [PubMed]
- Siniscalchi, M.; D’Ingeo, S.; Fornelli, S.; Quaranta, A. Lateralized behavior and cardiac activity of dogs in response to human emotional vocalizations. Sci. Rep. 2018, 8, 77. [Google Scholar] [CrossRef] [PubMed]
- Tate, A.J.; Fischer, H.; Leigh, A.E.; Kendrick, K.M. Behavioural and neurophysiological evidence for face identity and face emotion processing in animals. Philos. Trans. R. Soc. B Biol. Sci. 2006, 361, 2155–2172. [Google Scholar] [CrossRef] [PubMed]
- Bellegarde, L.G.A.; Haskell, M.J.; Duvaux-Ponter, C.; Weiss, A.; Boissy, A.; Erhard, H.W. Face-based perception of emotions in dairy goats. Appl. Anim. Behav. Sci. 2017, 193, 51–59. [Google Scholar] [CrossRef]
- Nawroth, C.; Albuquerque, N.; Savalli, C.; Single, M.S.; McElligott, A.G. Goats prefer positive human emotional facial expressions. R. Soc. Open Sci. 2018, 5, 180491. [Google Scholar] [CrossRef]
- Massaro, D.W.; Egan, P.B. Perceiving affect from the voice and the face. Psychon. Bull. Rev. 1996, 3, 215–221. [Google Scholar] [CrossRef]
- Faragó, T.; Pongrácz, P.; Miklósi, À.; Huber, L.; Virányi, Z.; Range, F. Dogs’ expectation about signalers’ body size by virtue of their growls. PLoS ONE 2010, 5, e15175. [Google Scholar] [CrossRef]
- Bálint, A.; Faragó, T.; Dóka, A.; Miklósi, Á.; Pongrácz, P. Beware, I am big and non-dangerous!—Playfully growling dogs are perceived larger than their actual size by their canine audience. Appl. Anim. Behav. Sci. 2013, 148, 128–137. [Google Scholar] [CrossRef]
- Ratcliffe, V.F.; McComb, K.; Reby, D. Cross-modal discrimination of human gender by domestic dogs. Anim. Behav. 2014, 91, 127–135. [Google Scholar] [CrossRef]
- Izumi, A.; Kojima, S. Matching vocalizations to vocalizing faces in a chimpanzee (Pan troglodytes). Anim. Cogn. 2004, 7, 179–184. [Google Scholar] [CrossRef]
- Payne, C.; Bachevalier, J. Crossmodal integration of conspecific vocalizations in rhesus macaques. PLoS ONE 2013, 8, e81825. [Google Scholar] [CrossRef] [PubMed]
- Evans, T.A.; Howell, S.; Westergaard, G.C. Auditory-visual cross-modal perception of communicative stimuli in tufted capuchin monkeys (Cebus apella). J. Exp. Psychol. Anim. Behav. Process. 2005, 31, 399–406. [Google Scholar] [CrossRef] [PubMed]
- Turcsán, B.; Szánthó, F.; Miklósi, Á.; Kubinyi, E. Fetching what the owner prefers? Dogs recognize disgust and happiness in human behaviour. Anim. Cogn. 2015, 18, 83–94. [Google Scholar] [CrossRef] [PubMed]
- Trösch, M.; Ringhofer, M.; Yamamoto, S.; Lemarchand, J.; Parias, C.; Lormant, F.; Lansade, L. Horses prefer to solicit a person who previously observed a food-hiding process to access this food: A possible indication of attentional state attribution. Behav. Process. 2019, 166, 103906. [Google Scholar] [CrossRef]
- Ringhofer, M.; Yamamoto, S. Domestic horses send signals to humans when they face with an unsolvable tas. Anim. Cogn. 2016, 20, 397–405. [Google Scholar] [CrossRef]
- Smith, A.V.; Proops, L.; Grounds, K.; Wathan, J.; McComb, K. Functionally relevant responses to human facial expressions of emotion in the domestic horse (Equus caballus). Biol. Lett. 2016, 12, 20150907. [Google Scholar] [CrossRef]
- Catala, A.; Mang, B.; Wallis, L.; Huber, L. Dogs demonstrate perspective taking based on geometrical gaze following in a Guesser-Knower task. Anim. Cogn. 2017, 20, 581–589. [Google Scholar] [CrossRef]
- Chijiiwa, H.; Kuroshima, H.; Hori, Y.; Anderson, J.R.; Fujita, K. Dogs avoid people who behave negatively to their owner: Third-party affective evaluation. Anim. Behav. 2015, 106, 123–127. [Google Scholar] [CrossRef]
- Schuetz, A.; Farmer, K.; Krueger, K. Social learning across species: Horses (Equus caballus) learn from humans by observation. Anim. Cogn. 2017, 20, 567–573. [Google Scholar] [CrossRef]
- Briefer, E.F.; Mandel, R.; Maigrot, A.L.; Briefer Freymond, S.; Bachmann, I.; Hillmann, E. Perception of emotional valence in horse whinnies. Front. Zool. 2017, 14, 8. [Google Scholar] [CrossRef]
- Lansade, L.; Nowak, R.; Lainé, A.L.; Leterrier, C.; Bonneau, C.; Parias, C.; Bertin, A. Facial expression and oxytocin as possible markers of positive emotions in horses. Sci. Rep. 2018, 8, 14680. [Google Scholar] [CrossRef] [PubMed]
- Proops, L.; Grounds, K.; Smith, A.V.; McComb, K. Animals Remember Previous Facial Expressions that Specific Humans Have Exhibited. Curr. Biol. 2018, 28, 1428–1432.e4. [Google Scholar] [CrossRef] [PubMed]
- Baba, C.; Kawai, M.; Takimoto-Inose, A. Are horses (Equus caballus) sensitive to human emotional cues? Animals 2019, 9, 630. [Google Scholar] [CrossRef] [PubMed]
- Wathan, J.; Proops, L.; Grounds, K.; McComb, K. Horses discriminate between facial expressions of conspecifics. Sci. Rep. 2016, 6, 38322. [Google Scholar] [CrossRef]
- Smith, A.V.; Proops, L.; Grounds, K.; Wathan, J.; Scott, S.K.; McComb, K. Domestic horses (Equus caballus) discriminate between negative and positive human nonverbal vocalisations. Sci. Rep. 2018, 8, 13052. [Google Scholar] [CrossRef]
- Nakamura, K.; Takimoto-Inose, A.; Hasegawa, T. Cross-modal perception of human emotion in domestic horses (Equus caballus). Sci. Rep. 2018, 8, 8660. [Google Scholar] [CrossRef]
- Calvo, M.G.; Fernández-Martín, A.; Recio, G.; Lundqvist, D. Human observers and automated assessment of dynamic emotional facial expressions: KDEF-dyn database validation. Front. Psychol. 2018, 9, 2052. [Google Scholar] [CrossRef]
- Lima, C.F.; Castro, S.L.; Scott, S.K. When voices get emotional: A corpus of nonverbal vocalizations for research on emotion processing. Behav. Res. Methods 2013, 45, 1234–1245. [Google Scholar] [CrossRef]
- Friard, O.; Gamba, M. BORIS: A free, versatile open-source event-logging software for video/audio coding and live observations. Methods Ecol. Evol. 2016, 7, 1325–1330. [Google Scholar] [CrossRef]
- Koo, T.K.; Li, M.Y. A Guideline of Selecting and Reporting Intraclass Correlation Coefficients for Reliability Research. J. Chiropr. Med. 2016, 15, 155–163. [Google Scholar] [CrossRef]
- Lansade, L.; Bouissou, M.F.; Erhard, H.W. Fearfulness in horses: A temperament trait stables across time and situations. Appl. Anim. Behav. Sci. 2008, 115, 182–200. [Google Scholar] [CrossRef]
- Le Scolan, N.; Hausberger, M.; Wolff, A. Stability over situations in temperamental traits of horses as revealed by experimental and scoring approaches. Behav. Process. 1997, 41, 257–266. [Google Scholar] [CrossRef]
- Seaman, S.C.; Davidson, H.P.B.; Waran, N.K. How reliable is temperament assessment in the domestic horse (Equus caballus)? Appl. Anim. Behav. Sci. 2002, 78, 175–191. [Google Scholar] [CrossRef]
- Kuznetsova, A.; Brockhoff, P.B.; Christensen, R.H.B. lmerTest: Tests for random and fixed effects for linear mixed effect models. Available online: https://CRAN.R-project.org/package=lmerTest (accessed on 23 October 2019).
- Proops, L.; McComb, K. Cross-modal individual recognition in domestic horses (Equus caballus) extends to familiar humans. Proc. R. Soc. B Biol. Sci. 2012, 279, 3131–3138. [Google Scholar] [CrossRef] [PubMed]
- Lampe, J.F.; Andre, J. Cross-modal recognition of human individuals in domestic horses (Equus caballus). Anim. Cogn. 2012, 15, 623–630. [Google Scholar] [CrossRef]
- Proops, L.; McComb, K.; Reby, D. Cross-modal individual recognition in domestic horses (Equus caballus). Proc. Natl. Acad. Sci. USA 2009, 106, 947–951. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Trösch, M.; Cuzol, F.; Parias, C.; Calandreau, L.; Nowak, R.; Lansade, L. Horses Categorize Human Emotions Cross-Modally Based on Facial Expression and Non-Verbal Vocalizations. Animals 2019, 9, 862. https://doi.org/10.3390/ani9110862
Trösch M, Cuzol F, Parias C, Calandreau L, Nowak R, Lansade L. Horses Categorize Human Emotions Cross-Modally Based on Facial Expression and Non-Verbal Vocalizations. Animals. 2019; 9(11):862. https://doi.org/10.3390/ani9110862
Chicago/Turabian StyleTrösch, Miléna, Florent Cuzol, Céline Parias, Ludovic Calandreau, Raymond Nowak, and Léa Lansade. 2019. "Horses Categorize Human Emotions Cross-Modally Based on Facial Expression and Non-Verbal Vocalizations" Animals 9, no. 11: 862. https://doi.org/10.3390/ani9110862
APA StyleTrösch, M., Cuzol, F., Parias, C., Calandreau, L., Nowak, R., & Lansade, L. (2019). Horses Categorize Human Emotions Cross-Modally Based on Facial Expression and Non-Verbal Vocalizations. Animals, 9(11), 862. https://doi.org/10.3390/ani9110862



