Bta-miR-24-3p Controls the Myogenic Differentiation and Proliferation of Fetal Bovine Skeletal Muscle-Derived Progenitor Cells by Targeting ACVR1B
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Primary Cell Isolation and Cell Culture
2.3. RNA Isolation, Reverse Transcription (RT), and Quantitative Real-Time PCR (qRT-PCR)
2.4. Immunofluorescence
2.5. Western Blot
2.6. 5-Ethynyl-2′-deoxyuridine (EdU) Assay
2.7. Cell Counting Kit-8 (CCK8) Assay
2.8. RNA Oligonucleotide Preparation and Cell Transfection
2.9. Prediction of miRNA Target Genes
2.10. Dual-Luciferase Reporter Assay
2.11. Statistical Analysis
3. Results
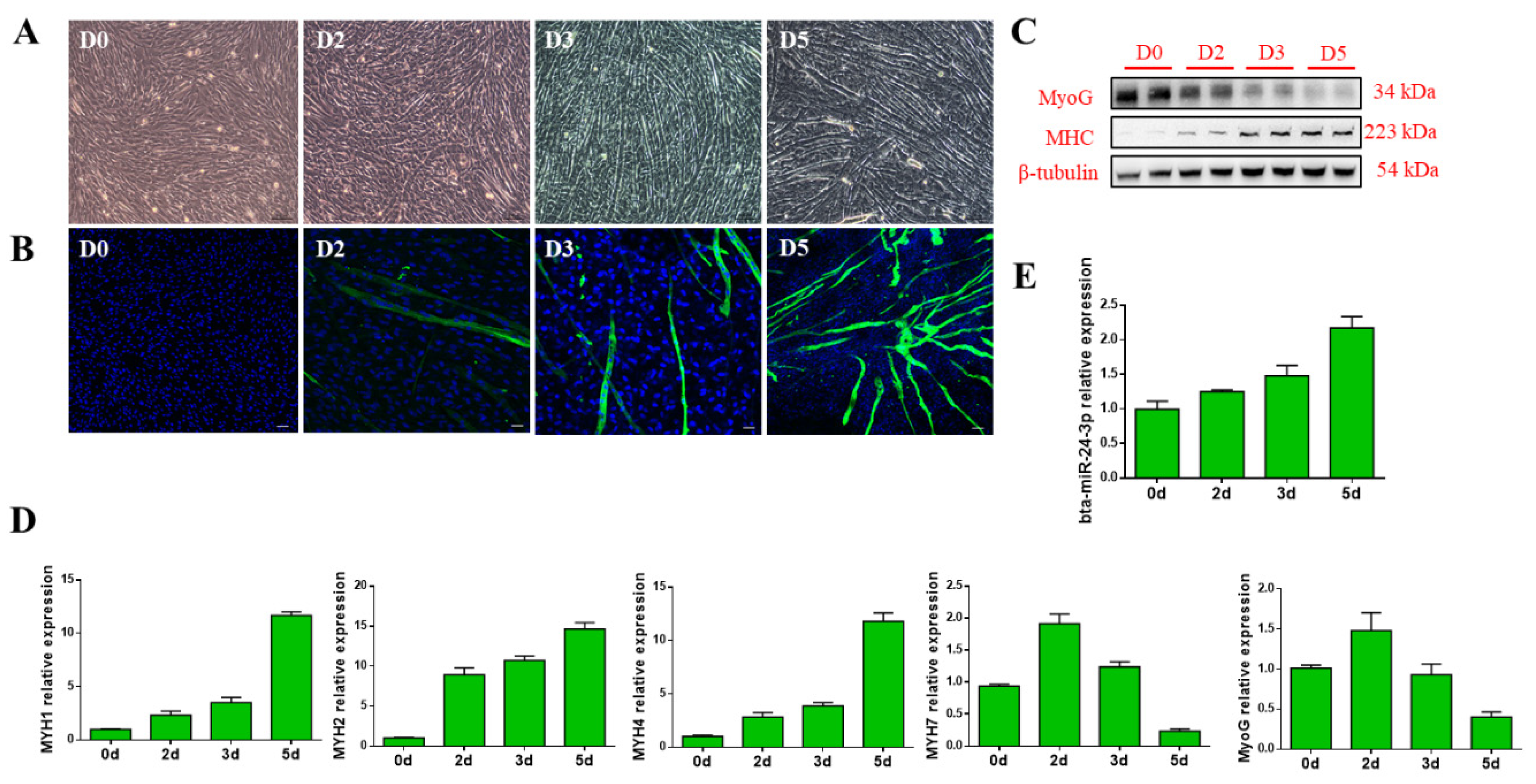
3.1. Bta-miR-24-3p Is Up-Regulated During the Myogenic Differentiation of PDGFRα- Progenitor Cells
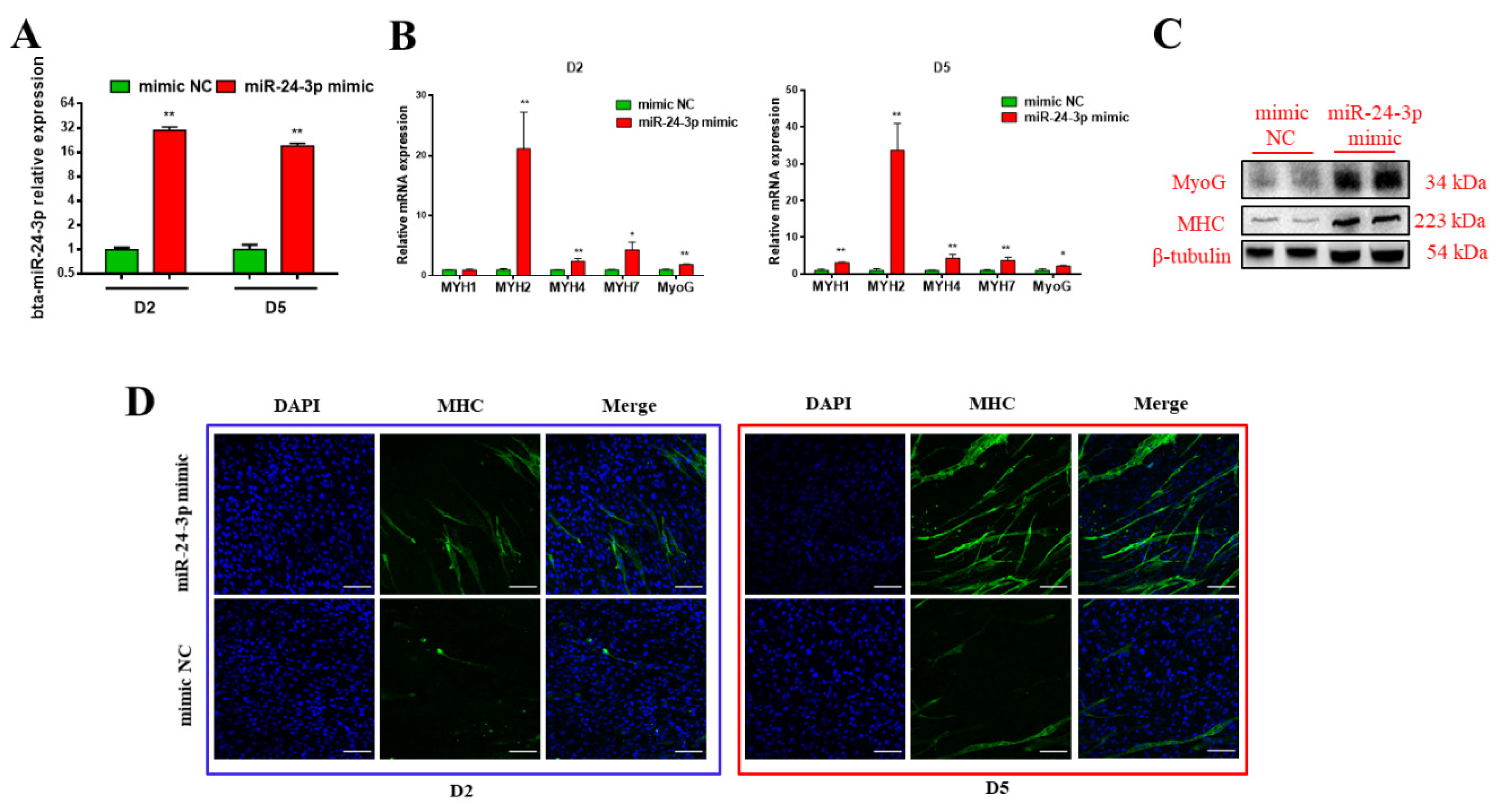
3.2. Bta-miR-24-3p Promotes the Myogenic Differentiation of Bovine PDGFRα- Progenitor Cells
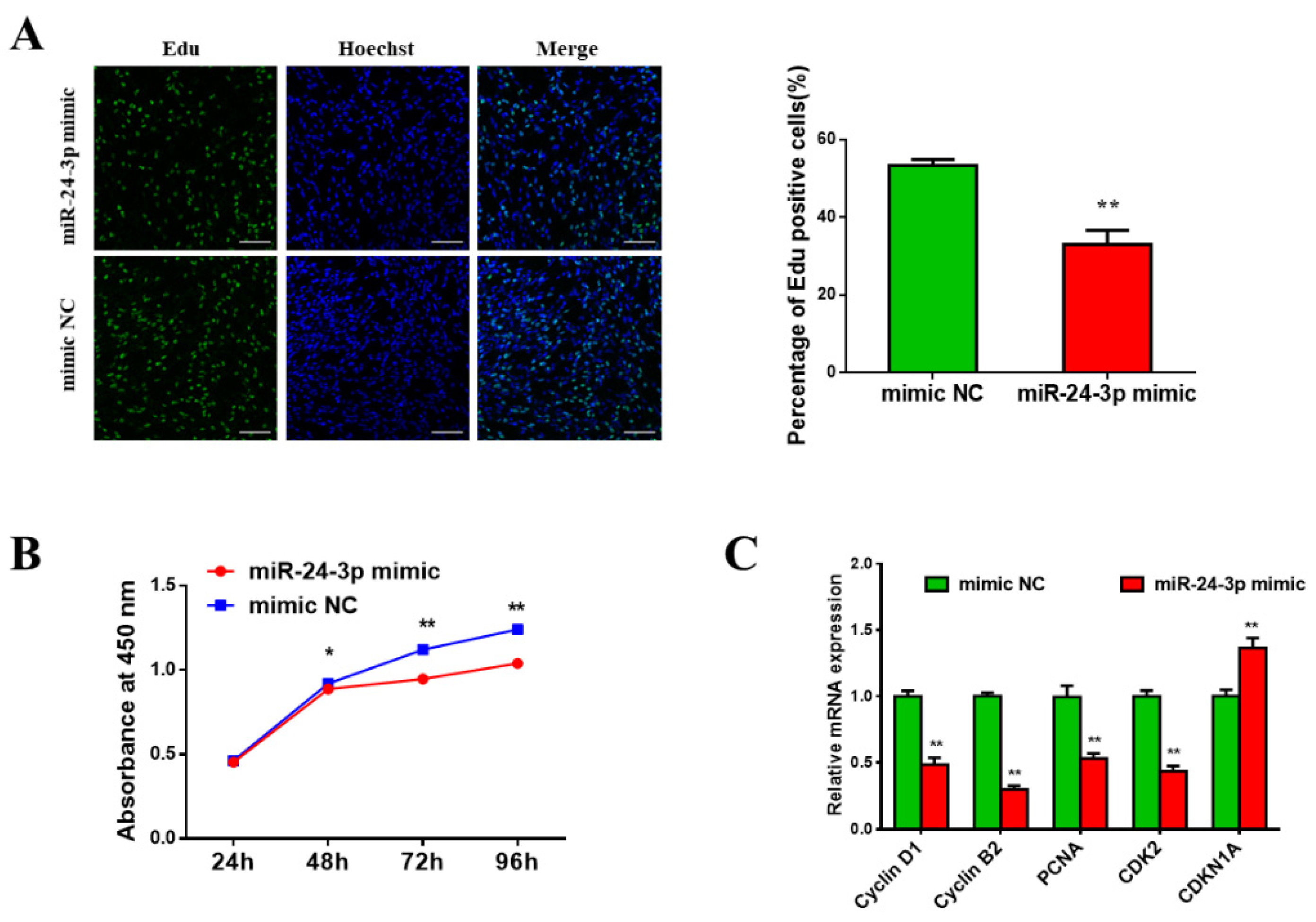
3.3. Bta-miR-24-3p Inhibits Bovine PDGFRα- Progenitor Cell Proliferation
3.4. ACVR1B Is a Direct Target of Bta-miR-24-3p
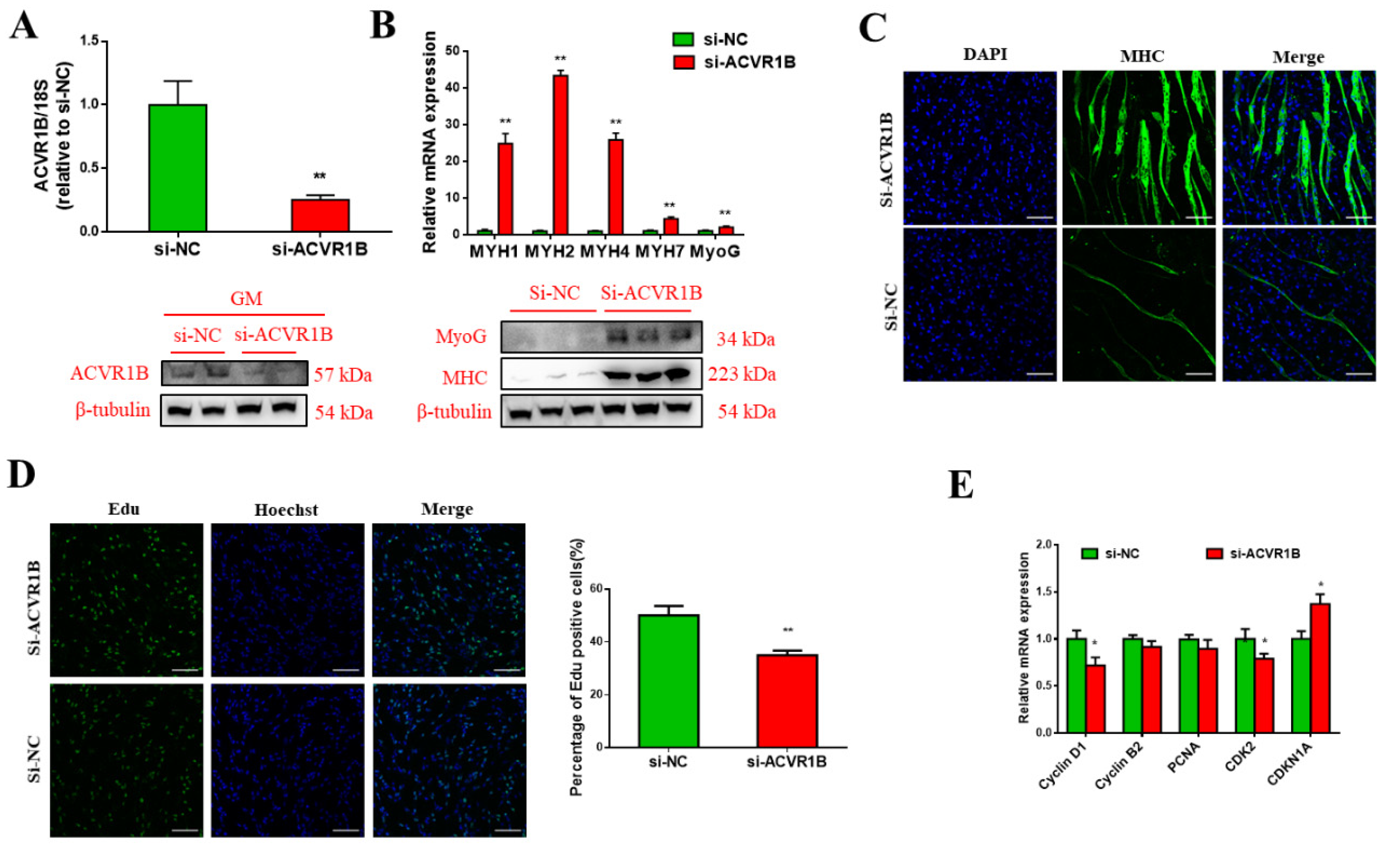
3.5. ACVR1B Silencing Promotes Myogenic Differentiation But Inhibits Proliferation
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Du, M.; Tong, J.; Zhao, J.; Underwood, K.R.; Zhu, M.; Ford, S.P.; Nathanielsz, P.W. Fetal programming of skeletal muscle development in ruminant animals. J. Anim. Sci. 2010, 88, E51–E60. [Google Scholar] [CrossRef] [PubMed]
- Sabourin, L.A.; Rudnicki, M.A. The molecular regulation of myogenesis. Clin. Genet. 2000, 57, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Lassar, A.B.; Skapek, S.X.; Novitch, B. Regulatory mechanisms that coordinate skeletal muscle differentiation and cell cycle withdrawal. Curr. Opin. Cell Biol. 1994, 6, 788–794. [Google Scholar] [CrossRef]
- Pownall, M.E.; Gustafsson, M.K.; Emerson, C.P., Jr. Myogenic regulatory factors and the specification of muscle progenitors in vertebrate embryos. Annu. Rev. Cell Dev. Biol. 2002, 18, 747–783. [Google Scholar] [CrossRef]
- Yun, K.; Wold, B. Skeletal muscle determination and differentiation: Story of a core regulatory network and its context. Curr. Opin. Cell Biol. 1996, 8, 877–889. [Google Scholar] [CrossRef]
- Bartel, D.P. MicroRNAs: Genomics, biogenesis, mechanism, and function. Cell 2004, 116, 281–297. [Google Scholar] [CrossRef]
- Wahid, F.; Shehzad, A.; Khan, T.; Kim, Y.Y. MicroRNAs: Synthesis, mechanism, function, and recent clinical trials. BBA Mol. Cell Res. 2010, 1803, 1231–1243. [Google Scholar] [CrossRef]
- Xie, S.J.; Li, J.H.; Chen, H.F.; Tan, Y.Y.; Liu, S.R.; Zhang, Y.; Xu, H.; Yang, J.H.; Liu, S.; Zheng, L.L.; et al. Inhibition of the JNK/MAPK signaling pathway by myogenesis-associated miRNAs is required for skeletal muscle development. Cell Death Differ. 2018, 25, 1581–1597. [Google Scholar] [CrossRef]
- Zhang, Y.; Yan, H.; Zhou, P.; Zhang, Z.; Liu, J.; Zhang, H. MicroRNA-152 Promotes Slow-Twitch Myofiber Formation via Targeting Uncoupling Protein-3 Gene. Animals 2019, 9. [Google Scholar] [CrossRef]
- Zhang, W.R.; Zhang, H.N.; Wang, Y.M.; Dai, Y.; Liu, X.F.; Li, X.; Ding, X.B.; Guo, H. miR-143 regulates proliferation and differentiation of bovine skeletal muscle satellite cells by targeting IGFBP5. In Vitro Cell Dev. Biol. Anim. 2017, 53, 265–271. [Google Scholar] [CrossRef]
- Song, C.; Yang, J.; Jiang, R.; Yang, Z.; Li, H.; Huang, Y.; Lan, X.; Lei, C.; Ma, Y.; Qi, X.; et al. miR-148a-3p regulates proliferation and apoptosis of bovine muscle cells by targeting KLF6. J. Cell. Physiol. 2019, 234. [Google Scholar] [CrossRef] [PubMed]
- Tong, H.; Jiang, R.; Liu, T.; Wei, Y.; Li, S.; Yan, Y. bta-miR-378 promote the differentiation of bovine skeletal muscle-derived satellite cells. Gene 2018, 668, 246–251. [Google Scholar] [CrossRef] [PubMed]
- Sun, Q.; Zhang, Y.; Yang, G.; Chen, X.; Zhang, Y.; Cao, G.; Wang, J.; Sun, Y.; Zhang, P.; Fan, M.; et al. Transforming growth factor-beta-regulated miR-24 promotes skeletal muscle differentiation. Nucleic Acids Res. 2008, 36, 2690–2699. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Wang, H.; Li, Y.; Liu, S.; Chen, J.; Ying, H. miR-24 and miR-122 Negatively Regulate the Transforming Growth Factor-beta/Smad Signaling Pathway in Skeletal Muscle Fibrosis. Mol. Nucleic Acids 2018, 11, 528–537. [Google Scholar] [CrossRef]
- Zimmerman, C.M.; Mathews, L.S. Activin receptors: Cellular signalling by receptor serine kinases. Biochem. Soc. Symp. 1996, 62, 25–38. [Google Scholar]
- Reissmann, E.; Jornvall, H.; Blokzijl, A.; Andersson, O.; Chang, C.; Minchiotti, G.; Persico, M.G.; Ibanez, C.F.; Brivanlou, A.H. The orphan receptor ALK7 and the Activin receptor ALK4 mediate signaling by Nodal proteins during vertebrate development. Genes Dev. 2001, 15, 2010–2022. [Google Scholar] [CrossRef]
- Ramsdell, A.F.; Bernanke, J.M.; Trusk, T.C. Left-right lineage analysis of the embryonic Xenopus heart reveals a novel framework linking congenital cardiac defects and laterality disease. Development 2006, 133, 1399–1410. [Google Scholar] [CrossRef]
- Pasteuning-Vuhman, S.; Boertje-van der Meulen, J.W.; van Putten, M.; Overzier, M.; Ten Dijke, P.; Kielbasa, S.M.; Arindrarto, W.; Wolterbeek, R.; Lezhnina, K.V.; Ozerov, I.V.; et al. New function of the myostatin/activin type I receptor (ALK4) as a mediator of muscle atrophy and muscle regeneration. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2017, 31, 238–255. [Google Scholar] [CrossRef]
- Mizuno, Y.; Tokuzawa, Y.; Ninomiya, Y.; Yagi, K.; Yatsuka-Kanesaki, Y.; Suda, T.; Fukuda, T.; Katagiri, T.; Kondoh, Y.; Amemiya, T.; et al. miR-210 promotes osteoblastic differentiation through inhibition of AcvR1b. FEBS Lett. 2009, 583, 2263–2268. [Google Scholar] [CrossRef]
- Qiu, W.; Li, X.; Tang, H.; Huang, A.S.; Panteleyev, A.A.; Owens, D.M.; Su, G.H. Conditional activin receptor type 1B (Acvr1b) knockout mice reveal hair loss abnormality. J. Investig. Dermatol. 2011, 131, 1067–1076. [Google Scholar] [CrossRef]
- Uezumi, A.; Fukada, S.; Yamamoto, N.; Takeda, S.; Tsuchida, K. Mesenchymal progenitors distinct from satellite cells contribute to ectopic fat cell formation in skeletal muscle. Nat. Cell Biol. 2010, 12, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Guan, L.; Hu, X.; Liu, L.; Xing, Y.S.; Zhou, Z.K.; Liang, X.W.; Yang, Q.Y.; Jin, S.Y.; Bao, J.S.; Gao, H.J.; et al. bta-miR-23a involves in adipogenesis of progenitor cells derived from fetal bovine skeletal muscle. Sci. Rep. UK 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Yu, F.; Pang, G.; Zhao, G. ANRIL acts as onco-lncRNA by regulation of microRNA-24/c-Myc, MEK/ERK and Wnt/beta-catenin pathway in retinoblastoma. Int. J. Biol. Macromol. 2019, 128, 583–592. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Sun, Y.; Zhou, Y.; Zhang, Y.; Zhang, T.; Li, Y.; You, W.; Chang, X.; Yuan, L.; Han, X. MicroRNA-24 promotes pancreatic beta cells toward dedifferentiation to avoid endoplasmic reticulum stress-induced apoptosis. J. Mol. Cell Biol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Yin, K.; Lv, X.; Yang, Q.; Shao, M.; Liu, X.; Sun, H. MicroRNA-24-3p regulates Hodgkin’s lymphoma cell proliferation, migration and invasion by targeting DEDD. Oncol. Lett. 2019, 17, 365–371. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Yang, Y. MiR-24 inhibits inflammatory responses in LPS-induced acute lung injury of neonatal rats through targeting NLRP3. Pathol. Res. Pract. 2019, 215, 683–688. [Google Scholar] [CrossRef] [PubMed]
- Xiao, X.; Lu, Z.; Lin, V.; May, A.; Shaw, D.H.; Wang, Z.; Che, B.; Tran, K.; Du, H.; Shaw, P.X. MicroRNA miR-24-3p Reduces Apoptosis and Regulates Keap1-Nrf2 Pathway in Mouse Cardiomyocytes Responding to Ischemia/Reperfusion Injury. Oxidative Med. Cell. Longev. 2018, 2018, 7042105. [Google Scholar] [CrossRef]
- Yan, L.; Ma, J.; Zhu, Y.; Zan, J.; Wang, Z.; Ling, L.; Li, Q.; Lv, J.; Qi, S.; Cao, Y.; et al. miR-24-3p promotes cell migration and proliferation in lung cancer by targeting SOX7. J. Cell. Biochem. 2018, 119, 3989–3998. [Google Scholar] [CrossRef]
- Wang, Q.; Huang, Z.; Xue, H.; Jin, C.; Ju, X.L.; Han, J.D.; Chen, Y.G. MicroRNA miR-24 inhibits erythropoiesis by targeting activin type I receptor ALK4. Blood 2008, 111, 588–595. [Google Scholar] [CrossRef]
- Li, Z.; Sun, Y.; Cao, S.; Zhang, J.; Wei, J. Downregulation of miR-24-3p promotes osteogenic differentiation of human periodontal ligament stem cells by targeting SMAD family member 5. J. Cell. Physiol. 2019, 234, 7411–7419. [Google Scholar] [CrossRef]
- Jin, L.; Li, Y.; Nie, L.; He, T.; Hu, J.; Liu, J.; Chen, M.; Shi, M.; Jiang, Z.; Gui, Y.; et al. MicroRNA242 is associated with cell proliferation, invasion, migration and apoptosis in renal cell carcinoma. Mol. Med. Rep. 2017, 16, 9157–9164. [Google Scholar] [CrossRef] [PubMed]
- Hua, K.; Chen, Y.T.; Chen, C.F.; Tang, Y.S.; Huang, T.T.; Lin, Y.C.; Yeh, T.S.; Huang, K.H.; Lee, H.C.; Hsu, M.T.; et al. MicroRNA-23a/27a/24-2 cluster promotes gastric cancer cell proliferation synergistically. Oncol. Lett. 2018, 16, 2319–2325. [Google Scholar] [CrossRef] [PubMed]
- Zhou, N.; Yan, H.L. MiR-24 promotes the proliferation and apoptosis of lung carcinoma via targeting MAPK7. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 6845–6852. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Pan, J.; Wang, H.; Ma, Z.; Yin, J.; Yuan, F.; Yuan, Q.; Zhou, L.; Liu, X.; Zhang, Y.; et al. von Willebrand factor rescued by miR-24 inhibition facilitates the proliferation and migration of osteosarcoma cells in vitro. Biosci. Rep. 2018, 38. [Google Scholar] [CrossRef] [PubMed]
- Yu, G.; Jia, Z.; Dou, Z. miR-24-3p regulates bladder cancer cell proliferation, migration, invasion and autophagy by targeting DEDD. Oncol. Rep. 2017, 37, 1123–1131. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Chen, L.; Ding, J.; Fan, Z.; Li, S.; Wu, H.; Zhang, J.; Yang, C.; Wang, H.; Zeng, P.; et al. MicroRNA-24 inhibits high glucose-induced vascular smooth muscle cell proliferation and migration by targeting HMGB1. Gene 2016, 586, 268–273. [Google Scholar] [CrossRef]
- Lal, A.; Navarro, F.; Maher, C.A.; Maliszewski, L.E.; Yan, N.; O’Day, E.; Chowdhury, D.; Dykxhoorn, D.M.; Tsai, P.; Hofmann, O.; et al. miR-24 Inhibits cell proliferation by targeting E2F2, MYC, and other cell-cycle genes via binding to “seedless” 3’UTR microRNA recognition elements. Mol. Cell 2009, 35, 610–625. [Google Scholar] [CrossRef]
- Feng, X.H.; Derynck, R. Specificity and versatility in tgf-beta signaling through Smads. Annu. Rev. Cell Dev. Biol. 2005, 21, 659–693. [Google Scholar] [CrossRef]
- Song, C.; Fan, B.; Xiao, Z. Overexpression of ALK4 inhibits cell proliferation and migration through the inactivation of JAK/STAT3 signaling pathway in glioma. Biomed. Pharmacother. 2018, 98, 440–445. [Google Scholar] [CrossRef]
- Zhu, S.; Goldschmidt-Clermont, P.J.; Dong, C. Transforming growth factor-beta-induced inhibition of myogenesis is mediated through Smad pathway and is modulated by microtubule dynamic stability. Circ. Res. 2004, 94, 617–625. [Google Scholar] [CrossRef]
- De Angelis, L.; Borghi, S.; Melchionna, R.; Berghella, L.; Baccarani-Contri, M.; Parise, F.; Ferrari, S.; Cossu, G. Inhibition of myogenesis by transforming growth factor beta is density-dependent and related to the translocation of transcription factor MEF2 to the cytoplasm. Proc. Natl. Acad. Sci. USA 1998, 95, 12358–12363. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Black, B.L.; Derynck, R. TGF-beta inhibits muscle differentiation through functional repression of myogenic transcription factors by Smad3. Genes Dev. 2001, 15, 2950–2966. [Google Scholar] [CrossRef] [PubMed]
- Murakami, M.; Ohkuma, M.; Nakamura, M. Molecular mechanism of transforming growth factor-beta-mediated inhibition of growth arrest and differentiation in a myoblast cell line. Dev. Growth Differ. 2008, 50, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, Y.; Otsuka, F.; Hino, J.; Miyoshi, T.; Takano, M.; Miyazato, M.; Makino, H.; Kangawa, K. Bone morphogenetic protein-3b (BMP-3b) inhibits osteoblast differentiation via Smad2/3 pathway by counteracting Smad1/5/8 signaling. Mol. Cell. Endocrinol. 2012, 350, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Rebbapragada, A.; Benchabane, H.; Wrana, J.L.; Celeste, A.J.; Attisano, L. Myostatin Signals through a Transforming Growth Factor β-Like Signaling Pathway To Block Adipogenesis. Mol. Cell. Biol. 2003, 23, 7230–7242. [Google Scholar] [CrossRef] [PubMed]
- Levine, A.J.; Brivanlou, A.H. GDF3 at the crossroads of TGF-beta signaling. Cell Cycle 2006, 5, 1069–1073. [Google Scholar] [CrossRef]
- Hata, A.; Chen, Y.G. TGF-beta Signaling from Receptors to Smads. Cold Spring Harb. Perspect. Biol. 2016, 8. [Google Scholar] [CrossRef]
- Hill, C.S. Transcriptional Control by the SMADs. Cold Spring Harb. Perspect. Biol. 2016, 8. [Google Scholar] [CrossRef]
- Hinck, A.P. Structural studies of the TGF-betas and their receptors - insights into evolution of the TGF-beta superfamily. FEBS Lett. 2012, 586, 1860–1870. [Google Scholar] [CrossRef]
- Clop, A.; Marcq, F.; Takeda, H.; Pirottin, D.; Tordoir, X.; Bibe, B.; Bouix, J.; Caiment, F.; Elsen, J.M.; Eychenne, F.; et al. A mutation creating a potential illegitimate microRNA target site in the myostatin gene affects muscularity in sheep. Nat. Genet. 2006, 38, 813–818. [Google Scholar] [CrossRef]
- McPherron, A.C.; Lawler, A.M.; Lee, S.J. Regulation of skeletal muscle mass in mice by a new TGF-beta superfamily member. Nature 1997, 387, 83–90. [Google Scholar] [CrossRef] [PubMed]
- McPherron, A.C.; Lee, S.J. Double muscling in cattle due to mutations in the myostatin gene. Proc. Natl. Acad. Sci. USA 1997, 94, 12457–12461. [Google Scholar] [CrossRef] [PubMed]
- Kemaladewi, D.U.; de Gorter, D.J.; Aartsma-Rus, A.; van Ommen, G.J.; ten Dijke, P.; ’t Hoen, P.A.; Hoogaars, W.M. Cell-type specific regulation of myostatin signaling. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2012, 26, 1462–1472. [Google Scholar] [CrossRef] [PubMed]
- Langley, B.; Thomas, M.; Bishop, A.; Sharma, M.; Gilmour, S.; Kambadur, R. Myostatin inhibits myoblast differentiation by down-regulating MyoD expression. J. Biol. Chem. 2002, 277, 49831–49840. [Google Scholar] [CrossRef] [PubMed]
- Joulia, D.; Bernardi, H.; Garandel, V.; Rabenoelina, F.; Vernus, B.; Cabello, G. Mechanisms involved in the inhibition of myoblast proliferation and differentiation by myostatin. Exp. Cell Res. 2003, 286, 263–275. [Google Scholar] [CrossRef]
- Taylor, W.E.; Bhasin, S.; Artaza, J.; Byhower, F.; Azam, M.; Willard, D.H., Jr.; Kull, F.C., Jr.; Gonzalez-Cadavid, N. Myostatin inhibits cell proliferation and protein synthesis in C2C12 muscle cells. Am. J. Physiol. Endocrinol. Metab. 2001, 280, E221–E228. [Google Scholar] [CrossRef]
- Thomas, M.; Langley, B.; Berry, C.; Sharma, M.; Kirk, S.; Bass, J.; Kambadur, R. Myostatin, a negative regulator of muscle growth, functions by inhibiting myoblast proliferation. J. Biol. Chem. 2000, 275, 40235–40243. [Google Scholar] [CrossRef]
- Garikipati, D.K.; Rodgers, B.D. Myostatin inhibits myosatellite cell proliferation and consequently activates differentiation: Evidence for endocrine-regulated transcript processing. J. Endocrinol. 2012, 215, 177–187. [Google Scholar] [CrossRef]
- Rodgers, B.D.; Wiedeback, B.D.; Hoversten, K.E.; Jackson, M.F.; Walker, R.G.; Thompson, T.B. Myostatin stimulates, not inihibits, C2C12 myoblast proliferation. Endocrinology 2014, 155, 670–675. [Google Scholar] [CrossRef]
- Manceau, M.; Gros, J.; Savage, K.; Thome, V.; McPherron, A.; Paterson, B.; Marcelle, C. Myostatin promotes the terminal differentiation of embryonic muscle progenitors. Genes Dev. 2008, 22, 668–681. [Google Scholar] [CrossRef]
- Chen, X.; Huang, Z.; Chen, D.; Yang, T.; Liu, G. Role of microRNA-27a in myoblast differentiation. Cell Biol. Int. 2014, 38, 266–271. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.; Li, M.; Wang, B.; Klein, J.D.; Price, S.R.; Wang, X.H. miRNA-23a/27a attenuates muscle atrophy and renal fibrosis through muscle-kidney crosstalk. J. Cachexia Sarcopenia Muscle 2018, 9, 755–770. [Google Scholar] [CrossRef]
- Wang, B.; Zhang, C.; Zhang, A.; Cai, H.; Price, S.R.; Wang, X.H. MicroRNA-23a and MicroRNA-27a Mimic Exercise by Ameliorating CKD-Induced Muscle Atrophy. J. Am. Soc. Nephrol. JASN 2017, 28, 2631–2640. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Luo, J.; Zhang, H.; Lu, J. microRNAs in the Same Clusters Evolve to Coordinately Regulate Functionally Related Genes. Mol. Biol. Evol. 2016, 33, 2232–2247. [Google Scholar] [CrossRef] [PubMed]

| Name | Forward (5’–3’) | Reverse (5’–3’) |
|---|---|---|
| MyoG | CAAATCCACTCCCTGAAA | GCATAGGAAGAGATGAACA |
| MYH1 | GGGAAACTGGCTTCTGCTGAT | TGGGTTGGTGGTGATTAGGAG |
| MYH2 | GTCAAAGGGACTATCCAGAGCAG | AGAAGAGGCCCGAGTAGGTGT |
| MYH4 | CTCCTAATCACCACCAACCCATA | TGTCAGCAACTTCAGTGCCATC |
| MYH7 | AAGACAGTGACCGTGAAGGAGG | GGTTGATGGTGACGCAGAAGA |
| ACVR1B | TGCCCTCTGACCCTTCCATC | CACTCCCGCATCATCTTCCC |
| bta-miR-24-3p | CAGTGGCTCAGTTCAGCAGGA | |
| 18s | GTAACCCGTTGAACCCCATT | CCATCCAATCGGTAGTAGCG |
| Cyclin D1 | GACGAGCTGCTGCACATGGA | TGCTTGTTCTCCTCGGCCAC |
| Cyclin B1 | TGGGAGAGACATAAACGG | TGGAAGCCAAGAGCAGTG |
| PCNA | GCTGTGTAGTAAAGATGCCT | ATCTCTATGGCAACAGCTTC |
| CDK2 | TCTTTGCTGAGATGGTGACCC | TAACTCCTGGCCAAACCACC |
| CDKN1A | AAACGGCGGCAGACC | GCCCAAGGCAAAAGG |
| Name | Forward (5’–3’) | Reverse (5’–3’) |
|---|---|---|
| Psi-CHECK-ACVR1B-W | ACTCTCGAGAACAACCACACACACAAAA | ATAGCGGCCGCGGATGAGAAGGTGCAACTT |
| Psi-CHECK-ACVR1B-Mut | TATCTGTCCTTGTACTGCTCCCCCCCGCCC | GGGCGGGGGGGAGCAGTACAAGGACAGATA |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, X.; Xing, Y.; Ren, L.; Wang, Y.; Li, Q.; Fu, X.; Yang, Q.; Xu, L.; Willems, L.; Li, J.; et al. Bta-miR-24-3p Controls the Myogenic Differentiation and Proliferation of Fetal Bovine Skeletal Muscle-Derived Progenitor Cells by Targeting ACVR1B. Animals 2019, 9, 859. https://doi.org/10.3390/ani9110859
Hu X, Xing Y, Ren L, Wang Y, Li Q, Fu X, Yang Q, Xu L, Willems L, Li J, et al. Bta-miR-24-3p Controls the Myogenic Differentiation and Proliferation of Fetal Bovine Skeletal Muscle-Derived Progenitor Cells by Targeting ACVR1B. Animals. 2019; 9(11):859. https://doi.org/10.3390/ani9110859
Chicago/Turabian StyleHu, Xin, Yishen Xing, Ling Ren, Yahui Wang, Qian Li, Xing Fu, Qiyuan Yang, Lingyang Xu, Luc Willems, Junya Li, and et al. 2019. "Bta-miR-24-3p Controls the Myogenic Differentiation and Proliferation of Fetal Bovine Skeletal Muscle-Derived Progenitor Cells by Targeting ACVR1B" Animals 9, no. 11: 859. https://doi.org/10.3390/ani9110859
APA StyleHu, X., Xing, Y., Ren, L., Wang, Y., Li, Q., Fu, X., Yang, Q., Xu, L., Willems, L., Li, J., & Zhang, L. (2019). Bta-miR-24-3p Controls the Myogenic Differentiation and Proliferation of Fetal Bovine Skeletal Muscle-Derived Progenitor Cells by Targeting ACVR1B. Animals, 9(11), 859. https://doi.org/10.3390/ani9110859






