Tolerance of Broilers to Dietary Supplementation with High Levels of the DHA-Rich Microalga, Aurantiochytrium Limacinum: Effects on Health and Productivity
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Ingredient and Diet Analysis
3.2. Performance and Bird Health
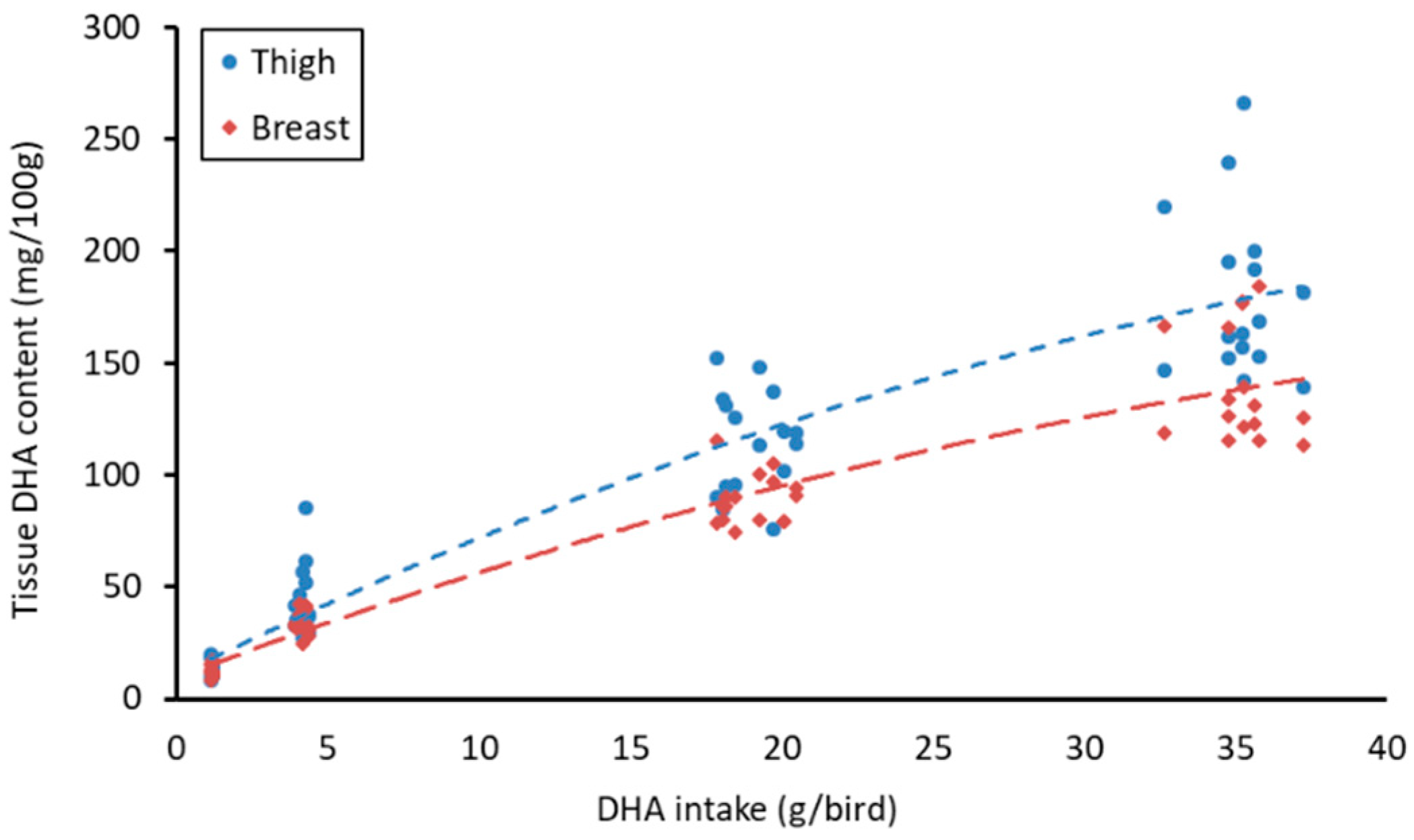
3.3. Meat Fatty Acid Content
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Calder, P.C. Very long chain omega-3 (n-3) fatty acids and human health. Eur. J. Lipid Sci. Technol. 2014, 116, 1280–1300. [Google Scholar] [CrossRef]
- Nestel, P.; Clifton, P.; Colquhoun, D.; Noakes, M.; Mori, T.A.; Sullivan, D.; Thomas, B. Indications for omega-3 long chain polyunsaturated fatty acid in the prevention and treatment of cardiovascular disease. Hear Lung Circ. 2015, 24, 769–779. [Google Scholar] [CrossRef] [PubMed]
- Vaughan, V.C.; Hassing, M.R.; Lewandowski, P.A. Marine polyunsaturated fatty acids and cancer therapy. Br. J. Cancer 2013, 108, 486–492. [Google Scholar] [CrossRef] [PubMed]
- Willatts, P. Effects of Nutrition on the Development of Higher-Order Cognition. In Recent Research in Nutrition and Growth; Colombo, J., Koletzko, B., Lampl, M., Eds.; Karger: Basel, Switzerland, 2018; Volume 89, pp. 175–184. [Google Scholar]
- Derbyshire, E. Brain Health across the lifespan: A systematic review on the role of omega-3 fatty acid supplements. Nutrients 2018, 10, 1094. [Google Scholar] [CrossRef] [PubMed]
- Howe, P.; Meyer, B.; Record, S.; Baghurst, K. Dietary intake of long-chain ω-3 polyunsaturated fatty acids: Contribution of meat sources. Nutrition 2006, 22, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Marine omega-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance. Biochim. Biophys. Acta 2015, 1851, 469–484. [Google Scholar] [CrossRef] [PubMed]
- Givens, D.I.; Gibbs, R.A. Current intakes of EPA and DHA in European populations and the potential of animal-derived foods to increase them. Proc. Nutr. Soc. 2008, 67, 273–280. [Google Scholar] [CrossRef] [PubMed]
- Rymer, C.; Givens, D.I. n-3 fatty acid enrichment of edible tissue of poultry: A review. Lipids 2005, 40, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Rymer, C.; Gibbs, R.A.; Givens, D.I. Comparison of algal and fish sources on the oxidative stability of poultry meat and its enrichment with omega-3 polyunsaturated fatty acids. Poult. Sci. 2010, 89, 150–159. [Google Scholar] [CrossRef] [PubMed]
- Salem, N.; Eggersdorfer, M. Is the world supply of omega-3 fatty acids adequate for optimal human nutrition? Curr. Opin. Clin. Nutr. Metab. Care 2015, 18, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Finco, A.M.D.O.; Mamani, L.D.G.; Carvalho, J.C.D.; de Melo Pereira, G.V.; Thomaz-Soccol, V.; Soccol, C.R. Technological trends and market perspectives for production of microbial oils rich in omega-3. Crit. Rev. Biotechnol. 2016, 8551, 656–671. [Google Scholar] [CrossRef] [PubMed]
- Abril, R.; Barclay, W. Production of docosahexaenoic acid-enriched poultry eggs and meat using an algae-based feed ingredient. World Rev. Nutr. Diet. 1998, 83, 77–88. [Google Scholar] [PubMed]
- Mooney, J.W.; Hirschler, E.M.; Kennedy, A.K.; Sams, A.R.; Van Elswyk, M.E. Lipid and flavour quality of stored breast meat from broilers fed marine algae. J. Sci. Food Agric. 1998, 78, 134–140. [Google Scholar] [CrossRef]
- Moran, C.A.; Morlacchini, M.; Fusconi, G. Enhancing the DHA content in milk from dairy cows by feeding ALL-G-RICHTM. J. Appl. Anim. Nutr. 2017, 5, 1–9. [Google Scholar] [CrossRef]
- Moran, C.A.; Morlacchini, M.; Keegan, J.D.; Fusconi, G. The effect of dietary supplementation with Aurantiochytrium limacinum on lactating dairy cows in terms of animal health, productivity and milk composition. J. Anim. Physiol. Anim. Nutr. (Berl.) 2018, 102, 576–590. [Google Scholar] [CrossRef] [PubMed]
- Moran, C.A.; Morlacchini, M.; Keegan, J.D.; Delles, R.; Fusconi, G. Effects of a DHA-rich unextracted microalgae as a dietary supplement on performance, carcass traits and meat fatty acid profile in growing-finishing pigs. J. Anim. Physiol. Anim. Nutr. (Berl.) 2018, 102, 1026–1038. [Google Scholar] [CrossRef] [PubMed]
- Moran, C.A.; Morlacchini, M.; Keegan, J.D.; Fusconi, G. Dietary supplementation of finishing pigs with the docosahexaenoic acid-rich microalgae, Aurantiochytrium limacinum: Effects on performance, carcass characteristics and tissue fatty acid profile. Asian-Australas. J. Anim. Sci. 2018, 31, 712–720. [Google Scholar] [CrossRef] [PubMed]
- Ao, T.; Macalintal, L.M.; Paul, M.A.; Pescatore, A.J.; Cantor, A.H.; Ford, M.J.; Timmons, B.; Dawson, K.A. Effects of supplementing microalgae in laying hen diets on productive performance, fatty-acid profile, and oxidative stability of eggs. J. Appl. Poult. Res. 2015, 24, 394–400. [Google Scholar] [CrossRef]
- European Food Safety Authority. Technical guidance: Tolerance and efficacy studies in target animals. EFSA J. 2011, 9, 2175. [Google Scholar] [CrossRef]
- Aviagen Ross 308 Broiler: Nutrition Specifications. Available online: http://en.aviagen.com/assets/Tech_Center/Ross_Broiler/Ross308BroilerNutritionSpecs2014-EN.pdf (accessed on 10 October 2018).
- Yan, L.; Kim, I.H. Effects of dietary ω-3 fatty acid-enriched microalgae supplementation on growth performance, blood profiles, meat quality, and fatty acid composition of meat in broilers. J. Appl. Anim. Res. 2013, 41, 392–397. [Google Scholar] [CrossRef]
- Pappas, A.C.; Acamovic, T.; Sparks, N.H.C.; Surai, P.F.; McDevitt, R.M. Effects of supplementing broiler breeder diets with organoselenium compounds and polyunsaturated fatty acids on hatchability. Poult. Sci. 2006, 85, 1584–1593. [Google Scholar] [CrossRef] [PubMed]
- Haug, A.; Eich-Greatorex, S.; Bernhoft, A.; Wold, J.P.; Hetland, H.; Christophersen, O.A.; Sogn, T. Effect of dietary selenium and omega-3 fatty acids on muscle composition and quality in broilers. Lipids Health Dis. 2007, 6, 29. [Google Scholar] [CrossRef] [PubMed]
- Fébel, H.; Mézes, M.; Pálfy, T.; Hermán, A.; Gundel, J.; Lugasi, A.; Balogh, K.; Kocsis, I.; Blázovics, A. Effect of dietary fatty acid pattern on growth, body fat composition and antioxidant parameters in broilers. J. Anim. Physiol. Anim. Nutr. (Berl.) 2008, 92, 369–376. [Google Scholar] [CrossRef] [PubMed]
- European Commission Nutrition Claims. Available online: https://ec.europa.eu/food/safety/labelling_nutrition/claims/nutrition_claims_en (accessed on 12 September 2018).
- European Food Safety Authority. Scientific opinion on dietary reference values for fats, including saturated fatty acids, polyunsaturated fatty acids, monounsaturated fatty acids, trans fatty acids, and cholesterol. EFSA J. 2010, 8, 1461. [Google Scholar] [CrossRef]
- Ribeiro, T.; Lordelo, M.M.; Alves, S.P.; Bessa, R.J.B.; Costa, P.; Lemos, J.P.C.; Ferreira, L.M.A.; Fontes, C.M.G.A.; Prates, J.A.M. Direct supplementation of diet is the most efficient way of enriching broiler meat with n-3 long-chain polyunsaturated fatty acids. Br. Poult. Sci. 2013, 54, 753–765. [Google Scholar] [CrossRef] [PubMed]
- Sprecher, H.; Luthria, D.L.; Mohammed, B.S.; Baykousheva, S.P. Reevaluation of the pathways for the biosynthesis of polyunsaturated fatty acids. J. Lipid Res. 1995, 36, 2471–2477. [Google Scholar] [PubMed]
- Simopoulos, A.P. The importance of the omega-6/omega-3 fatty acid ratio in cardiovascular disease and other chronic diseases. Exp. Biol. Med. 2008, 233, 674–688. [Google Scholar] [CrossRef] [PubMed]
| Diet Ingredients | Starter | Grower |
|---|---|---|
| Maize | 64.042 | 66.902 |
| Soya oil | 1.637 | 2.501 |
| DL-Methionine | 0.267 | 0.225 |
| L-Threonine | 0.072 | 0.055 |
| L-Lysine HCI | 0.272 | 0.248 |
| Limestone | 0.528 | 0.427 |
| Dicalcium phosphate | 1.827 | 1.523 |
| Sodium bicarbonate | 0.341 | 0.344 |
| Salt | 0.150 | 0.150 |
| Hipro soya | 30.364 | 27.125 |
| RNL vit/min 1 | 0.500 | 0.500 |
| Diet nutrients | ||
| Dry matter | 87.8 | 87.864 |
| Crude protein | 20.25 | 18.854 |
| Crude fat | 4.47 | 5.360 |
| Crude fiber | 2.31 | 2.260 |
| Ash | 6 | 5.429 |
| Starch (ewers) | 41.2 | 43.034 |
| Sugar | 4.408 | 4.132 |
| Calcium | 0.9 | 0.780 |
| Phosphorous P | 0.667 | 0.598 |
| Sodium | 0.16 | 0.160 |
| Potassium | 0.868 | 0.804 |
| Chloride | 0.19 | 0.185 |
| Lysine | 1.268 | 1.159 |
| Methionine | 0.577 | 0.518 |
| Methionine + cysteine | 0.909 | 0.832 |
| Threonine | 0.826 | 0.756 |
| Tryptophan | 0.226 | 0.208 |
| ME 2 broiler | 2800 | 2900 |
| Starter D0–21 | Grower D22–42 | |||||||
|---|---|---|---|---|---|---|---|---|
| Nutrient Values | 0.0% | 0.5% | 2.5% | 5.0% | 0.0% | 0.5% | 2.5% | 5.0% |
| Dry matter | 87.0 | 87.1 | 87.2 | 86.8 | 87.8 | 87.6 | 87.6 | 87.9 |
| Crude fat (Oil A) | 5.6 | 5.5 | 5.7 | 5.6 | 5.0 | 5.4 | 5.5 | 5.4 |
| Crude protein | 20.0 | 20.2 | 20.4 | 20.4 | 18.7 | 18.6 | 18.6 | 18.4 |
| Ash | 5.8 | 5.8 | 5.6 | 5.3 | 4.9 | 4.8 | 4.8 | 4.8 |
| Fatty acid 1 content (mg/g) | ||||||||
| C14:0 Myristic acid | 0.1 | 0.2 | 0.7 | 1.3 | 0.1 | 0.2 | 0.7 | 1.3 |
| C16:0 Palmitic acid | 6.8 | 7.9 | 13.7 | 20.7 | 7.2 | 8.4 | 14.8 | 20.9 |
| C18:2 n-6 (LA) | 23.0 | 22.7 | 22.9 | 23.0 | 27.9 | 27.5 | 28.2 | 25.9 |
| C18:3 n-3 (ALA) | 1.8 | 1.7 | 1.8 | 1.8 | 2.4 | 2.3 | 2.4 | 2.3 |
| C20:5 n-3 (EPA) | 0.0 | 0.0 | 0.0 | 0.1 | 0.0 | 0.0 | 0.0 | 0.1 |
| C22:5 n-6 (DPA) | 0.1 | 0.2 | 0.7 | 1.4 | 0.0 | 0.1 | 0.7 | 1.4 |
| C22:6 n-3 (DHA) | 0.3 | 0.9 | 3.7 | 7.1 | 0.0 | 0.6 | 3.7 | 6.9 |
| Trait | Age (day) | 0% | 0.5% | 2.5% | 5% | SE | p-Value |
|---|---|---|---|---|---|---|---|
| Weight gain (g) | 0–21 | 843 b | 868 ab | 902 a | 870 ab | 21.3 | 0.033 |
| Weight gain (g) | 21–42 | 2234 | 2192 | 2250 | 2198 | 71.1 | 0.736 |
| Weight gain (g) | 0–42 | 3077 | 3060 | 3151 | 3069 | 83.3 | 0.562 |
| Feed intake (g) | 0–21 | 1107 b | 1159 ab | 1179 a | 1122 b | 24.8 | 0.010 |
| Feed intake (g) | 21–42 | 3904 | 3866 | 3961 | 3864 | 120.0 | 0.762 |
| Feed intake (g) | 0–42 | 5012 | 5025 | 5140 | 4986 | 133 | 0.553 |
| 1 FCR | 0–21 | 1.32 ab | 1.34 a | 1.31 ab | 1.29 b | 0.01 | 0.002 |
| FCR | 21–42 | 1.75 | 1.76 | 1.76 | 1.76 | 0.02 | 0.579 |
| FCR D0–42 | 0–42 | 1.63 | 1.64 | 1.63 | 1.62 | 0.001 | 0.086 |
| Parameter | 0% | 0.5% | 2.5% | 5% | SEM 1 | p-Value |
|---|---|---|---|---|---|---|
| Blood Biochemistry | ||||||
| Alkaline phosphatase (IU/L) | 13,663 | 13,298 | 11,919 | 13,877 | 1186 | 0.937 |
| Aspartate amino transferase (IU/L) | 618 | 630 | 616 | 415 | 31.5 | 0.067 |
| Calcium (mmol/L) | 2.34 | 2.33 | 2.39 | 2.31 | 0.03 | 0.861 |
| Cholesterol (mmol/L) | 3.69 b | 3.39 ab | 3.35 ab | 2.93 a | 0.08 | 0.019 |
| Creatinine (µmol/L) | 29.63 | 29.50 | 27.50 | 26.63 | 0.46 | 0.067 |
| Glucose (mmol/L) | 14.15 | 13.70 | 13.69 | 13.74 | 0.12 | 0.503 |
| Glutathione peroxidase (u/mL RBC) | 95.33 a | 112.16 ab | 112.20 ab | 119.96 b | 2.66 | 0.019 |
| Lactose dehydrogenase (IU/L) | 5161 | 4644 | 4153 | 2628 | 381 | 0.128 |
| Magnesium (mmol/L) | 0.963 | 0.963 | 0.988 | 0.988 | 0.01 | 0.607 |
| Phosphate (mmol/L) | 2.14 y | 2.14 y | 2.40 x | 2.19 xy | 0.03 | 0.035 |
| Triglycerides (mmol/L) | 0.726 | 0.726 | 0.818 | 0.731 | 0.05 | 0.868 |
| Urea (mmol/L) | 0.638 | 0.888 | 0.650 | 0.563 | 0.05 | 0.189 |
| Uric acid (µmol/L) | 357 | 296 | 380 | 362 | 22.08 | 0.571 |
| Blood hematology | ||||||
| Basophil (×103/µL) | 0.18 | 0.37 | 0.13 | 0.39 | 0.06 | 0.375 |
| Eosinophil (×103/µL) | 0.16 | 0.44 | 0.60 | 0.60 | 0.08 | 0.191 |
| Hemoglobin (g/L) | 96.13 | 94.14 | 91.38 | 98.25 | 1.81 | 0.575 |
| Heterophil (×103/µL) | 8.39 | 6.10 | 7.64 | 8.93 | 0.62 | 0.433 |
| Lymphocyte (×103/µL) | 2.51 | 2.84 | 2.76 | 2.89 | 0.26 | 0.956 |
| Mean corpuscular hemoglobin (g/L) | 277 | 302 | 270 | 274 | 5.83 | 0.241 |
| Mean corpuscular volume (fL) | 181 | 164 | 174 | 181 | 4.12 | 0.440 |
| Monocyte (×103/µL) | 0.20 | 0.40 | 0.60 | 0.40 | 0.07 | 0.301 |
| Packed cell volume (l/L) | 0.35 | 0.32 | 0.34 | 0.36 | 0.01 | 0.258 |
| Red blood cell count (×106/µL) | 1.98 | 1.96 | 1.95 | 2.00 | 0.05 | 0.984 |
| White blood cell count (×103/µL) | 11.48 | 10.16 | 11.81 | 13.19 | 0.70 | 0.526 |
| Fatty Acid (mg/100 g) | 0% | 0.5% | 2.5% | 5% | SEM | p-Value |
|---|---|---|---|---|---|---|
| C14:0 | 7.12 b | 6.42 b | 8.76 b | 14.34 a | 2.18 | <0.001 |
| C16:0 | 365.61 | 300.06 | 283.62 | 337.15 | 66.1 | 0.469 |
| C18:2 n-6 (LA) | 401.10 | 319.93 | 265.28 | 295.18 | 75.0 | 0.187 |
| C18:3 n-6 (GLA) | 3.76 a | 2.38 ab | 1.54 b | 1.37 b | 0.75 | 0.001 |
| C18:3 n-3 (ALA) | 26.32 | 19.81 | 17.50 | 21.07 | 5.91 | 0.359 |
| C18:4 n-3 (SDA) | 0.93 | 0.48 | 0.39 | 0.77 | 0.30 | 0.125 |
| C20:2 n-6 | 8.15 | 8.11 | 7.18 | 6.83 | 0.62 | 0.032 |
| C20:3 n-6 | 12.20 a | 11.21 a | 8.73 b | 7.19 c | 0.64 | <0.001 |
| C20:4 n-6 (AA) | 57.80 a | 51.32 b | 36.58 c | 32.45 c | 2.16 | <0.001 |
| C20:3 n-3 | 0.65 b | 1.05 a | 0.89 ab | 0.94 ab | 0.15 | 0.026 |
| C20:4 n-3 | 0.14 b | 0.15 b | 0.31 b | 1.08 a | 0.19 | <0.001 |
| C20:5 n-3 (EPA) | 3.28 c | 3.55 c | 6.59 b | 10.14 a | 0.71 | <0.001 |
| C22:4 n-6 | 13.80 a | 10.42 b | 4.16 c | 2.61 d | 0.50 | <0.001 |
| C22:5 n-6 (n-6 DPA) | 3.11 c | 4.85 c | 9.25 b | 14.75 a | 0.99 | <0.001 |
| C22:5 n-3 (n-3 DPA) | 9.02 b | 9.02 b | 10.19 ab | 10.52 a | 0.60 | 0.005 |
| C22:6 n-3 (DHA) | 12.20 d | 34.49 c | 89.06 b | 139.50 a | 6.03 | <0.001 |
| Total n-3 FA | 52.54 c | 68.55 c | 124.92 b | 184.02 a | 11.7 | <0.001 |
| Total n-6 FA | 499.92 | 408.21 | 332.72 | 360.39 | 78.5 | 0.078 |
| n-6/n-3 ratio | 9.33 a | 5.78 b | 2.57 c | 1.89 c | 0.37 | <0.001 |
| Fatty Acid (mg/100 g) | 0% | 0.5% | 2.5% | 5% | SEM | p-Value |
|---|---|---|---|---|---|---|
| C14:0 | 8.75 b | 11.94 b | 15.92 ab | 22.32 a | 3.45 | <0.001 |
| C16:0 | 442.28 | 512.1 | 465.95 | 489.95 | 100 | 0.861 |
| C18:2 n-6 (LA) | 504.1 | 575.7 | 465.0 | 443.1 | 115 | 0.552 |
| C18:3 n-6 (GLA) | 3.77 ab | 3.95 a | 2.43 ab | 1.94 b | 0.82 | 0.012 |
| C18:3 n-3 (ALA) | 32.40 | 38.07 | 32.87 | 32.75 | 9.15 | 0.868 |
| C18:4 n-3 (SDA) | 0.95 | 1.13 | 1.01 | 1.26 | 0.35 | 0.735 |
| C20:2 n-6 | 8.55 | 9.43 | 8.63 | 8.24 | 1.04 | 0.579 |
| C20:3 n-6 | 13.41 a | 14.05 a | 11.38 ab | 9.38 b | 1.31 | <0.001 |
| C20:4 n-6 (AA) | 67.51 a | 65.53 a | 46.95 b | 42.67 b | 6.97 | <0.001 |
| C20:3 n-3 | 1.03 | 1.26 | 1.26 | 1.22 | 0.15 | 0.230 |
| C20:4 n-3 | 0.28 c | 0.52 bc | 0.97 b | 1.76 a | 0.27 | <0.001 |
| C20:5 n-3 (EPA) | 3.16 c | 4.71 c | 10.12 b | 15.66 a | 1.65 | <0.001 |
| C22:4 n-6 | 15.26 a | 12.12b | 5.19 c | 3.50 c | 1.20 | <0.001 |
| C22:5 n-6 (n-6 DPA) | 3.75 c | 6.30 c | 12.83 b | 20.16 a | 1.49 | <0.001 |
| C22:5 n-3 (n-3 DPA) | 10.63 | 10.73 | 12.21 | 12.94 | 1.19 | 0.069 |
| C22:6 n-3 (DHA) | 14.26 d | 42.55c | 114.77 b | 179.84 a | 9.77 | <0.001 |
| Total n-3 FA | 62.69 c | 98.97 c | 173.21 b | 245.43 a | 20.8 | <0.001 |
| Total n-6 FA | 616.39 | 687.08 | 552.41 | 529.02 | 126 | 0.463 |
| n-6/n-3 ratio | 9.74 a | 6.68 b | 3.07 c | 2.10 d | 0.12 | <0.001 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moran, C.A.; Currie, D.; Keegan, J.D.; Knox, A. Tolerance of Broilers to Dietary Supplementation with High Levels of the DHA-Rich Microalga, Aurantiochytrium Limacinum: Effects on Health and Productivity. Animals 2018, 8, 180. https://doi.org/10.3390/ani8100180
Moran CA, Currie D, Keegan JD, Knox A. Tolerance of Broilers to Dietary Supplementation with High Levels of the DHA-Rich Microalga, Aurantiochytrium Limacinum: Effects on Health and Productivity. Animals. 2018; 8(10):180. https://doi.org/10.3390/ani8100180
Chicago/Turabian StyleMoran, Colm A., Douglas Currie, Jason D. Keegan, and Anne Knox. 2018. "Tolerance of Broilers to Dietary Supplementation with High Levels of the DHA-Rich Microalga, Aurantiochytrium Limacinum: Effects on Health and Productivity" Animals 8, no. 10: 180. https://doi.org/10.3390/ani8100180
APA StyleMoran, C. A., Currie, D., Keegan, J. D., & Knox, A. (2018). Tolerance of Broilers to Dietary Supplementation with High Levels of the DHA-Rich Microalga, Aurantiochytrium Limacinum: Effects on Health and Productivity. Animals, 8(10), 180. https://doi.org/10.3390/ani8100180





