Apulo-Calabrese and Crossbreed Pigs Show Different Physiological Response and Meat Quality Traits after Short Distance Transport
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Pre-Slaughter and Slaughter
2.3. Behavioural Response
2.4. Blood Sampling and Analysis
2.5. Skin Bruises Measurement
2.6. Meat Quality Measurements
2.7. Genotyping
2.8. Statistical Analysis
3. Results and Discussion
3.1. Behavioural Recordings and Carcass Bruises
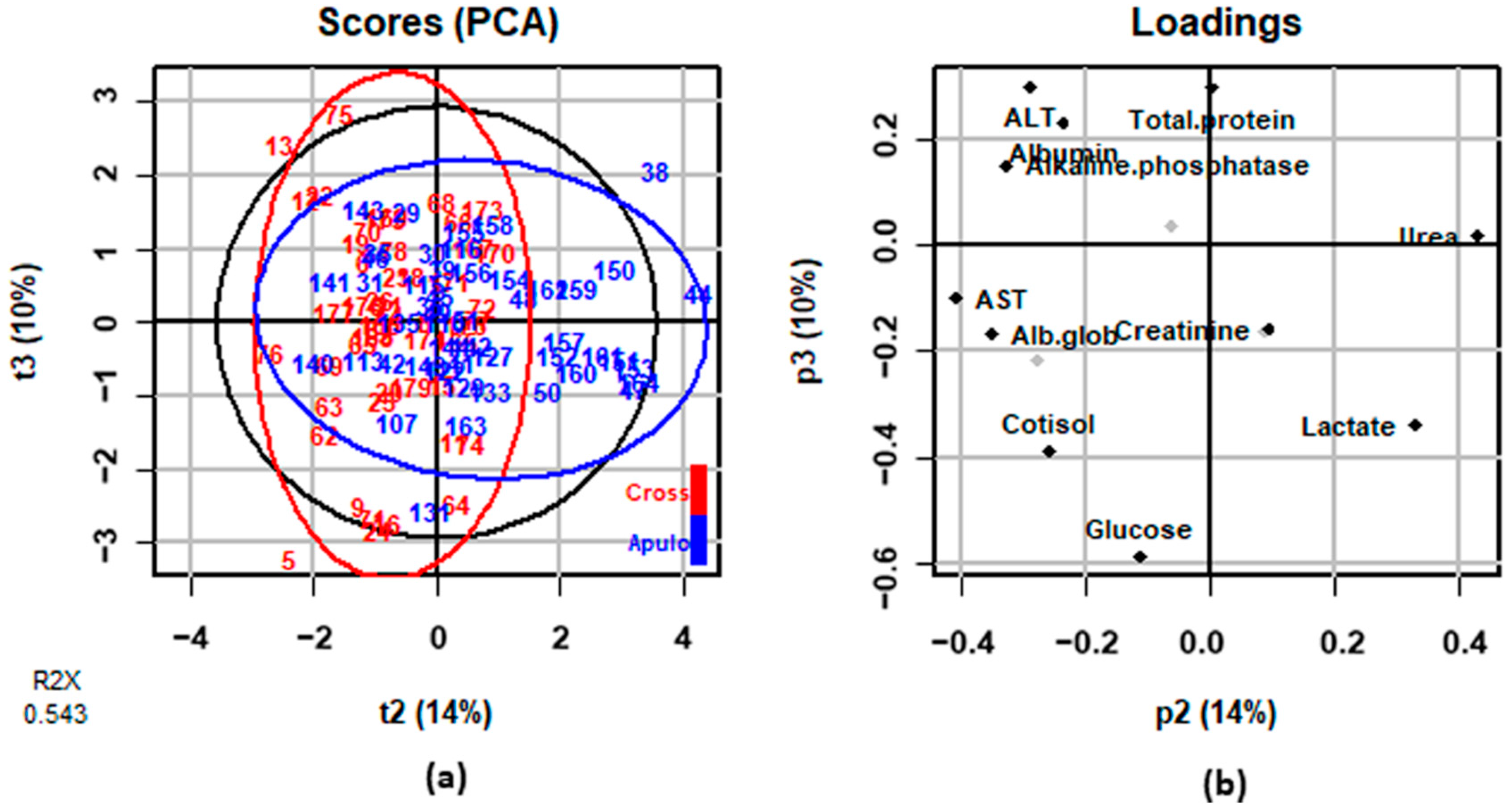
3.2. Blood Parameters
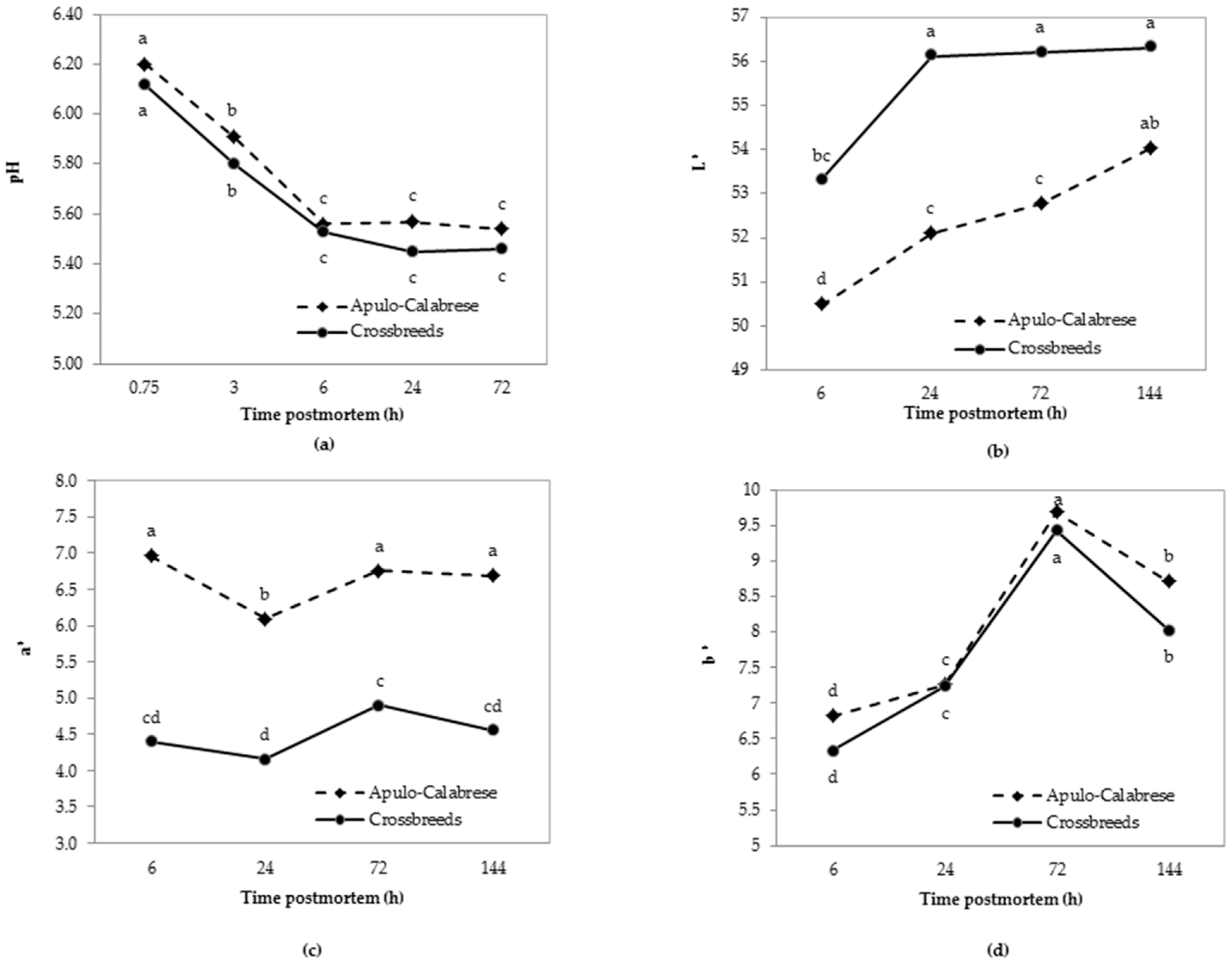
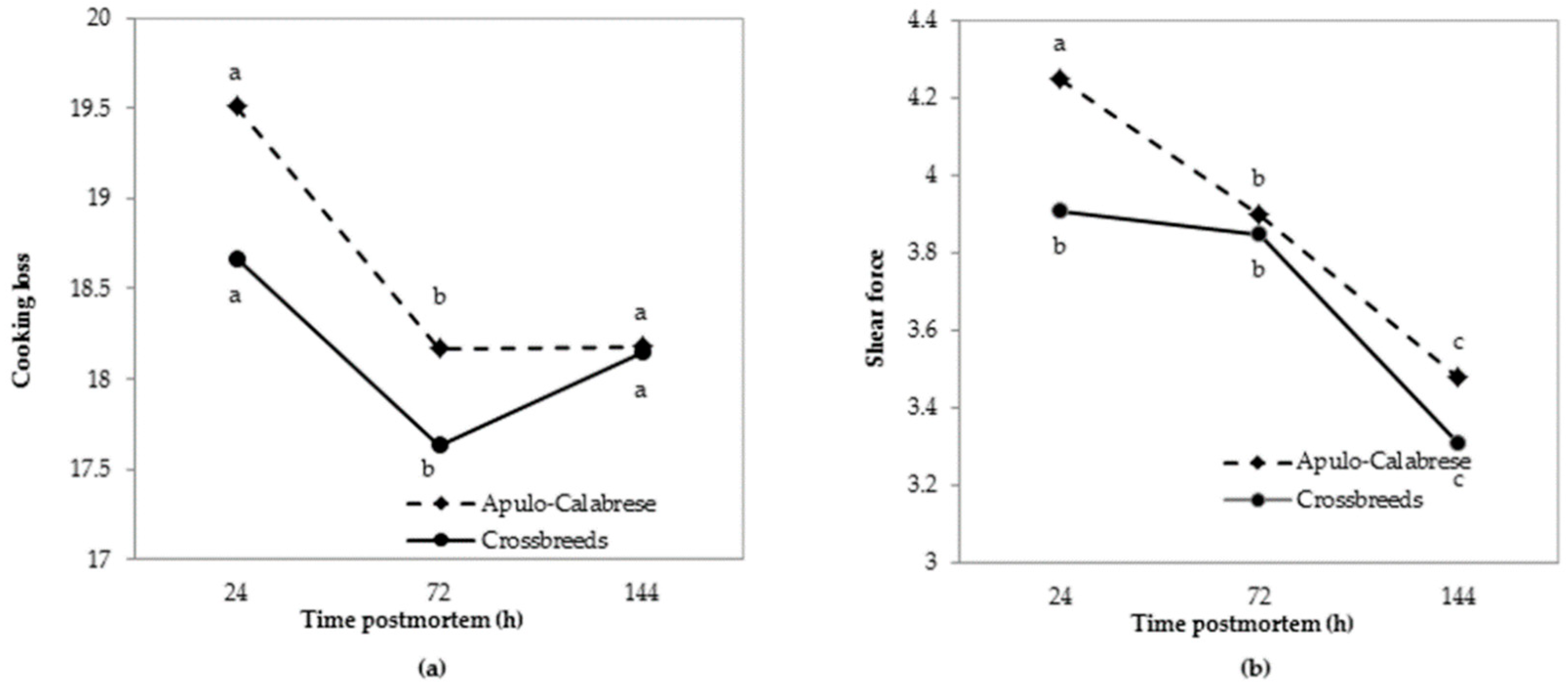
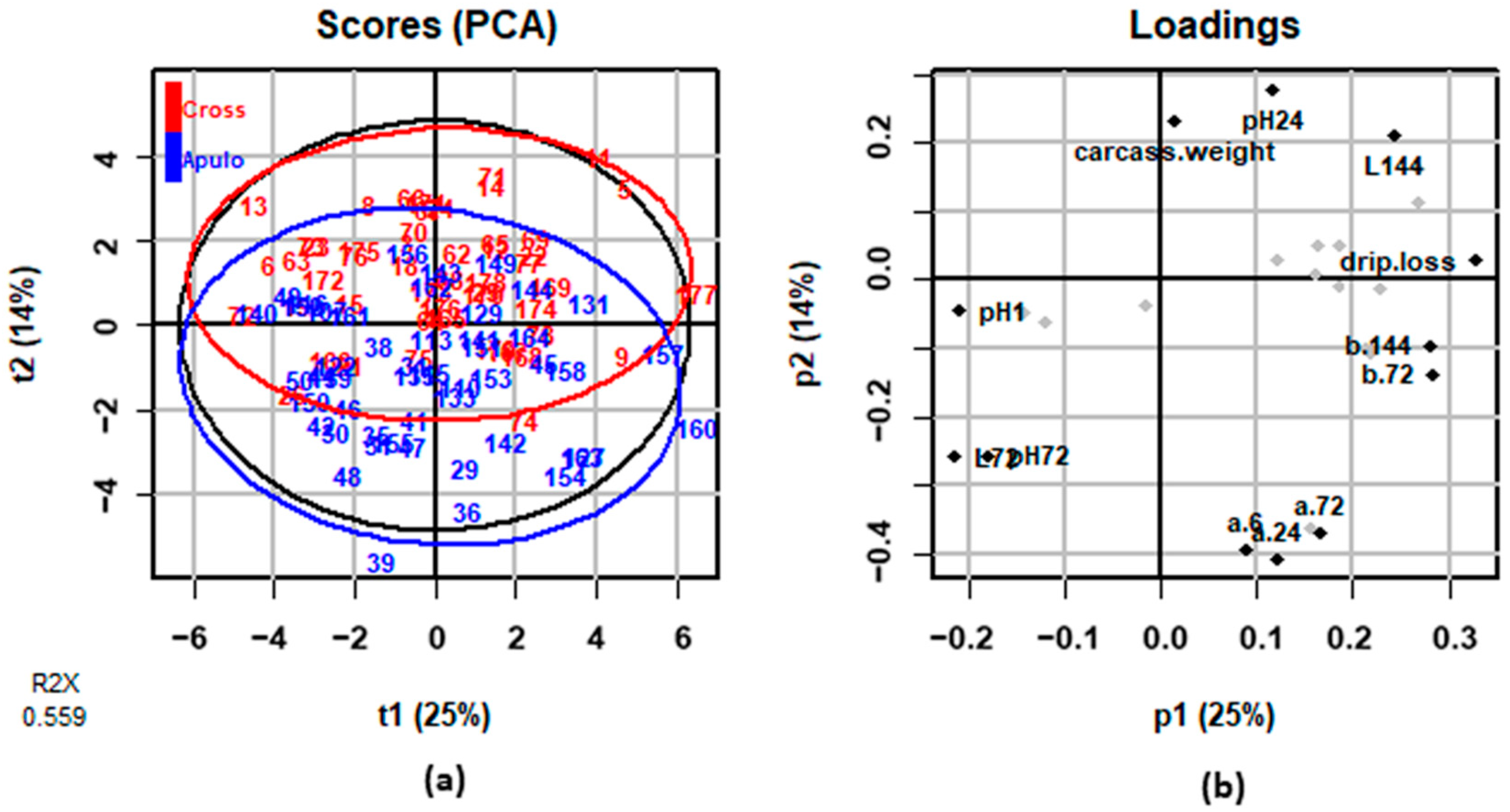
3.3. Meat Quality Traits
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Brandt, P.; Aaslyng, M.D. Welfare measurements of finishing pigs on the day of slaughter: A review. Meat Sci. 2015, 103, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Faucitano, L.; Goumon, S. Transport of Pigs to Slaughter and Associated Handling; Elsevier: Amsterdam, The Netherlands, 2017; pp. 261–293. ISBN 978-0-08-101012-9. [Google Scholar]
- Terlouw, E.M.C.; Arnould, C.; Auperin, B.; Berri, C.; Le Bihan-Duval, E.; Deiss, V.; Lefèvre, F.; Lensink, B.J.; Mounier, L. Pre-slaughter conditions, animal stress and welfare: Current status and possible future research. Animal 2008, 2, 1501–1517. [Google Scholar] [CrossRef] [PubMed]
- Brown, S.N.; Knowles, T.G.; Edwards, J.E.; Warriss, P.D. Behavioural and physiological responses of pigs to being transported for up to 24 h followed by six hours recovery in lairage. Vet. Rec. 1999, 145, 421–426. [Google Scholar] [CrossRef] [PubMed]
- Warriss, P.D. The Welfare of Slaughter Pigs During Transport. Anim. Welf. 1998, 7, 365–381. [Google Scholar]
- Gispert, M.; Faucitano, L.; Oliver, M.A.; Coll, C.; Siggens, K.; Harvey, K.; Diestre, A. A survey of pre-slaughter conditions, halothane gene frequency, and carcass and meat quality in five Spanish pig commercial abattoirs. Meat Sci. 2000, 10, 97–106. [Google Scholar] [CrossRef]
- Lambooy, E. Road transport of pigs over a long distance: Some aspects of behaviour, temperature and humidity during transport and some effects of the last two factors. Anim. Prod. 1988, 46, 257–263. [Google Scholar] [CrossRef]
- Sutherland, M.A.; McDonald, A.; McGlone, J.J. Effects of variations in the environment, length of journey and type of trailer on the mortality and morbidity of pigs being transported to slaughter. Vet. Rec. 2009, 165, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Dalla Costa, O.A.; Faucitano, L.; Coldebella, A.; Ludke, J.V.; Peloso, J.V.; dalla Roza, D.; Paranhos da Costa, M.J.R. Effects of the season of the year, truck type and location on truck on skin bruises and meat quality in pigs. Livest. Sci. 2007, 107, 29–36. [Google Scholar] [CrossRef]
- Torrey, S.; Bergeron, R.; Widowski, T.; Lewis, N.; Crowe, T.; Correa, J.A.; Brown, J.; Gonyou, H.W.; Faucitano, L. Transportation of market-weight pigs: I. Effect of season, truck type, and location within truck on behavior with a two-hour transport. J. Anim. Sci. 2013, 91, 2863–2871. [Google Scholar] [CrossRef] [PubMed]
- Gajana, C.S.; Nkukwana, T.T.; Marume, U.; Muchenje, V. Effects of transportation time, distance, stocking density, temperature and lairage time on incidences of pale soft exudative (PSE) and the physico-chemical characteristics of pork. Meat Sci. 2013, 95, 520–525. [Google Scholar] [CrossRef] [PubMed]
- Rocha, L.M.; Velarde, A.; Dalmau, A.; Saucier, L.; Faucitano, L. Can the monitoring of animal welfare parameters predict pork meat quality variation through the supply chain (from farm to slaughter)? J. Anim. Sci. 2016, 94, 359–376. [Google Scholar] [CrossRef] [PubMed]
- Mota-Rojas, D.; Herrera, M.B.; Trujillo-Ortega, M.E.; Alonso-Spilsbury, M.; Flores-Peinado, S.C.; Guerrero-Legarreta, I. Effects of Pre-Slaughter Transport, Lairage and Sex on Pig Chemical Serologic Profiles. J. Anim Vet. Adv. 2009, 6, 246–250. [Google Scholar]
- Probst, J.K.; Spengler Neff, A.; Hillmann, E.; Kreuzer, M.; Koch-Mathis, M.; Leiber, F. Relationship between stress-related exsanguination blood variables, vocalisation, and stressors imposed on cattle between lairage and stunning box under conventional abattoir conditions. Livest. Sci. 2014, 164, 154–158. [Google Scholar] [CrossRef]
- Werner, M.; Gallo, C. Effects of transport, lairage and stunning on the concentrations of some blood constituents in horses destined for slaughter. Livest. Sci. 2008, 115, 94–98. [Google Scholar] [CrossRef]
- Li, L.-A.; Xia, D.; Bao, E.-D.; Wei, S.; Xiao, J.-S.; Bao, J.-W.; Chen, W.-H.; Chen, J.; Hartung, J.; Zhao, R.-Q. Erhualian and Pietrain pigs exhibit distinct behavioral, endocrine and biochemical responses during transport. Livest. Sci. 2008, 113, 169–177. [Google Scholar] [CrossRef]
- Lebret, B.; Ecolan, P.; Bonhomme, N.; Méteau, K.; Prunier, A. Influence of production system in local and conventional pig breeds on stress indicators at slaughter, muscle and meat traits and pork eating quality. Animal 2015, 9, 1404–1413. [Google Scholar] [CrossRef] [PubMed]
- Bittante, G. Italian animal genetic resources in the Domestic Animal Diversity Information System of FAO. Ital. J. Anim. Sci. 2011, 10, e29. [Google Scholar] [CrossRef]
- ANAS. Available online: http//: www.anas.it (accessed on 7 June 2018).
- Micari, P.; Racinaro, L.; Sarullo, V.; Carpino, S.; Marzullo, A. Zoometric rates, reproductive and productive parameters of the Apulo-calabrian swine, obtained in breeding certified by ANAS Calabria. Ital. J. Anim. Sci. 2009, 8, 419–421. [Google Scholar] [CrossRef] [Green Version]
- Pugliese, C.; Sirtori, F. Quality of meat and meat products produced from southern European pig breeds. Meat Sci. 2012, 90, 511–518. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dalmau, A.; Temple, D.; Rodríguez, P.; Llonch, P.; Velarde, A. Application of the Welfare Quality® protocol at pig slaughterhouses. Anim. Welf. 2009, 18, 497–505. [Google Scholar]
- Correa, J.A.; Torrey, S.; Devillers, N.; Laforest, J.P.; Gonyou, H.W.; Faucitano, L. Effects of different moving devices at loading on stress response and meat quality in pigs. J. Anim. Sci. 2010, 88, 4086–4093. [Google Scholar] [CrossRef] [PubMed]
- Barton Gade, P.; Warriss, P.D.; Brown, S.N.; Lambooij, B. Methods of Improving Pig Welfare and Meat Quality by Reducing Stress and Discomfort Before Slaughter—Methods of Assessing Meat Quality. Available online: http://agris.fao.org/agris-search/search.do?recordID=DE97A3450 (accessed on 8 October 2018).
- Honikel, K.O. Reference methods for the assessment of physical characteristics of meat. Meat Sci. 1998, 49, 447–457. [Google Scholar] [CrossRef]
- Milan, D.; Jeon, J.T.; Looft, C.; Amarger, V.; Robic, A.; Thelander, M.; Rogel-Gaillard, C.; Paul, S.; Iannuccelli, N.; Rask, L.; et al. A mutation in PRKAG3 associated with excess glycogen content in pig skeletal muscle. Science 2000, 288, 1248–1251. [Google Scholar] [CrossRef] [PubMed]
- Fujii, J.; Otsu, K.; Zorzato, F.; de Leon, S.; Khanna, V.; Weiler, J.; O’Brien, P.; MacLennan, D. Identification of a mutation in porcine ryanodine receptor associated with malignant hyperthermia. Science 1991, 253, 448–451. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- Cherel, P.; Glénisson, J.; Figwer, P.; Pires, J.; Damon, M.; Franck, M.; Le Roy, P. Updated estimates of HAL n and RN−Effects on pork quality: Fresh and processed loin and ham. Meat Sci. 2010, 86, 949–954. [Google Scholar] [CrossRef] [PubMed]
- Otto, G.; Roehe, R.; Looft, H.; Thoelking, L.; Knap, P.W.; Rothschild, M.F.; Plastow, G.S.; Kalm, E. Associations of DNA markers with meat quality traits in pigs with emphasis on drip loss. Meat Sci. 2007, 75, 185–195. [Google Scholar] [CrossRef] [PubMed]
- Chai, J.; Xiong, Q.; Zhang, C.X.; Miao, W.; Li, F.E.; Zheng, R.; Peng, J.; Jiang, S.W. Effect of pre-slaughter transport plant on blood constituents and meat quality in halothane genotype of NN Large White×Landrace pigs. Livest. Sci. 2010, 127, 211–217. [Google Scholar] [CrossRef]
- Averos, X.; Herranz, A.; Sanchez, R.; Comella, J.X.; Gosalvez, L.F. Serum stress parameters in pigs transported to slaughter under commercial conditions in different seasons. Vet. Med. 2008, 52, 333–342. [Google Scholar] [CrossRef] [Green Version]
- Pérez, M.; Palacio, J.; Santolaria, M.; Aceña, M.; Chacón, G.; Gascón, M.; Calvo, J.; Zaragoza, P.; Beltran, J.; Garcı́a-Belenguer, S. Effect of transport time on welfare and meat quality in pigs. Meat Sci. 2002, 61, 425–433. [Google Scholar] [CrossRef]
- Broom, D.M. Causes of Poor Welfare in Large Animals During Transport. Vet. Res. Commun. 2003, 27, 515–518. [Google Scholar] [CrossRef] [PubMed]
- Swenson, M.J.; Recce, W.O. Physiological Properties and Cellular and Chemical constituents of blood. In Dukes’ Physiology of Domestic Animals, 11th ed.; Swenson, M.J., Recce, W.O., Eds.; Comstock Publishing: Ithaca, NY, USA, 1993; pp. 22–48. ISBN 0801428041. [Google Scholar]
- Bórnez, R.; Linares, M.B.; Vergara, H. Haematological, hormonal and biochemical blood parameters in lamb: Effect of age and blood sampling time. Livest. Sci. 2009, 121, 200–206. [Google Scholar] [CrossRef]
- Dikic, M.; Salajpal, K.; Djikic, D.; Karolyi, D.; Kosteliš, A.; Rupiš, V. Blood parameters in pigs of Turopolje breed and crossbreds TCSL. In Proceedings of the Hrvatski simpozij agronoma, Opatija, Hrvatska, 15–19 February 2010; pp. 1019–1022. [Google Scholar]
- Odink, J.; Smeets, J.F.; Visser, I.J.; Sandman, H.; Snijders, J.M. Hematological and clinicochemical profiles of healthy swine and swine with inflammatory processes. J. Anim. Sci. 1990, 68, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Mota-Rojas, D.; Becerril-Herrera, M.; Roldan-Santiago, P.; Alonso-Spilsbury, M.; Flores-Peinado, S.; Ramírez-Necoechea, R.; Ramírez-Telles, J.A.; Mora-Medina, P.; Pérez, M.; Molina, E.; et al. Effects of long distance transportation and CO2 stunning on critical blood values in pigs. Meat Sci. 2012, 90, 893–898. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.; Lei, H.; Zhang, S.; Li, X.; Li, M.; Jiang, X.; Zhu, K.; Zhu, L. The comparison of energy metabolism and meat quality among three pig breeds: Energy Metabolism Effect on Meat Quality of Pig. Anim. Sci. J. 2014, 85, 770–779. [Google Scholar] [CrossRef] [PubMed]
- Scheffler, T.L.; Gerrard, D.E. Mechanisms controlling pork quality development: The biochemistry controlling postmortem energy metabolism. Meat Sci. 2007, 77, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Franci, O.; Pugliese, C. Italian autochthonous pigs: Progress report and research perspectives. Ital. J. Anim. Sci. 2007, 6, 663–671. [Google Scholar] [CrossRef]
- Peinado, B.; Poto, A.; Gil, F.; López, G. Characteristics of the carcass and meat of the Chato Murciano pig. Livest. Prod. Sci. 2004, 90, 285–292. [Google Scholar] [CrossRef]
- Franci, O.; Bozzi, R.; Pugliese, C.; Acciaioli, A.; Campodoni, G.; Gandini, G. Performance of Cinta Senese pigs and their crosses with Large White. 1 Muscle and subcutaneous fat characteristics. Meat Sci. 2005, 69, 545–550. [Google Scholar] [CrossRef] [PubMed]
- Pugliese, C.; Madonia, G.; Chiofalo, V.; Margiotta, S.; Acciaioli, A.; Gandini, G. Comparison of the performances of Nero Siciliano pigs reared indoors and outdoors. 1. Growth and carcass composition. Meat Sci. 2003, 65, 825–831. [Google Scholar] [CrossRef] [Green Version]
| Delivery | Genetic Type | Number of Pigs | Loading | Transport | Unloading | ||||
|---|---|---|---|---|---|---|---|---|---|
| Duration 1 (min) | Temp (°C) | RH (%) | Duration (min) | Duration 2 (min) | Temp (°C) | RH (%) | |||
| 1 | Apulo-Calabrese | 19 | 8 | 10.3 | 79 | 63 | 2 | 11.2 | 88.4 |
| Crossbreed | 20 | 16 | 9 | ||||||
| 2 | Apulo-Calabrese | 17 | 10 | 18 | 72.3 | 67 | 7 | 19.5 | 65.8 |
| Crossbreed | 17 | 16 | 3 | ||||||
| 3 | Apulo-Calabrese | 15 | 6 | 21.5 | 59.6 | 60 | 5 | 19.7 | 65.5 |
| Crossbreed | 15 | 6 | 2 | ||||||
| Blood Parameters | Baseline | Exsanguination | ||||||
|---|---|---|---|---|---|---|---|---|
| Apulo-Calabrese | Crossbreed | Apulo-Calabrese | Crossbreed | p-Values | ||||
| L.S.M | S.E.M | T | GT | T × GT | ||||
| Creatine Kinase, CK (U/L) | 954.99 | 831.76 | 2089.30 | 2187.76 | 0.05 | <0.0001 | 0.7529 | 0.2130 |
| Cortisol (mg/dL) | 16.72 | 17.38 | 56.31 | 56.73 | 0.26 | <0.0001 | 0.8730 | 0.8690 |
| Glucose (mg/dL) | 71.23 | 78.13 | 100.57 | 114.59 | 0.00 | <0.0001 | 0.0004 | 0.9497 |
| Lactate (mg/dL) | 24.23 c | 33.85 b | 181.01 a | 143.81 a | 0.03 | <0.0001 | 0.4549 | <0.0001 |
| Albumin (g/dL) | 3.47 | 3.55 | 3.63 | 3.80 | 0.00 | <0.0001 | 0.1747 | 0.2862 |
| Albumin/globulin, Alb/glob | 0.93 a | 0.99 a | 0.82 b | 0.95 a | 0.01 | <0.0001 | 0.0031 | <0.0001 |
| Total protein (g/dL) | 7.30 | 7.20 | 8.15 | 7.84 | 0.10 | <0.0001 | 0.0686 | 0.0674 |
| Urea (mg/dL) | 37.71 a | 36.91 a | 37.55 a | 33.50 b | 0.96 | 0.0103 | 0.0404 | 0.0185 |
| Creatinine (mg/dL) | 1.65 c | 1.89 b | 2.08 a | 2.25 a | 0.00 | <0.0001 | <0.0001 | <0.0001 |
| Aspartate aminotransferase, AST (U/L) | 70.80 a | 33.25 c | 77.73 a | 54.29 b | 0.03 | <0.0001 | <0.0001 | <0.0001 |
| Alanine aminotranferase, ALT (U/L) | 54.83 a | 52.51 b | 58.52 ab | 59.41 ab | 0.11 | <0.0001 | 0.7309 | 0.0357 |
| Alkaline phosphatase, ALP (U/L) | 145.14 a | 118.12 b | 134.57 ab | 119.03 b | 5.69 | 0.1105 | 0.0527 | 0.0124 |
| Sodium, Na (mEq/L) | 139.03 b | 142.00 b | 147.97 a | 145.77 a | 1.18 | 0.7937 | 0.7937 | 0.0017 |
| Potassium, K (mEq/L) | 5.34 c | 5.74 b | 6.93 a | 6.20 ab | 0.00 | <0.0001 | 0.0017 | 0.0004 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aboagye, G.; Dall’Olio, S.; Tassone, F.; Zappaterra, M.; Carpino, S.; Nanni Costa, L. Apulo-Calabrese and Crossbreed Pigs Show Different Physiological Response and Meat Quality Traits after Short Distance Transport. Animals 2018, 8, 177. https://doi.org/10.3390/ani8100177
Aboagye G, Dall’Olio S, Tassone F, Zappaterra M, Carpino S, Nanni Costa L. Apulo-Calabrese and Crossbreed Pigs Show Different Physiological Response and Meat Quality Traits after Short Distance Transport. Animals. 2018; 8(10):177. https://doi.org/10.3390/ani8100177
Chicago/Turabian StyleAboagye, Gizella, Stefania Dall’Olio, Francesco Tassone, Martina Zappaterra, Salvatore Carpino, and Leonardo Nanni Costa. 2018. "Apulo-Calabrese and Crossbreed Pigs Show Different Physiological Response and Meat Quality Traits after Short Distance Transport" Animals 8, no. 10: 177. https://doi.org/10.3390/ani8100177
APA StyleAboagye, G., Dall’Olio, S., Tassone, F., Zappaterra, M., Carpino, S., & Nanni Costa, L. (2018). Apulo-Calabrese and Crossbreed Pigs Show Different Physiological Response and Meat Quality Traits after Short Distance Transport. Animals, 8(10), 177. https://doi.org/10.3390/ani8100177





