A Good Death? Report of the Second Newcastle Meeting on Laboratory Animal Euthanasia
Abstract
:Simple Summary
Abstract
1. Introduction
- the recommendation that CO2 should not be used as a sole killing agent on conscious animals [4];
- addition of the use of inert gases as an “acceptable” technique for rodents [2];
- a presumption that decapitation is only acceptable when no other method is possible [2];
- the opposite presumption that the only welfare concern with decapitation and cervical dislocation is the skill of the person [3].
- review key areas of recent research into humane killing;
- consider how this could be interpreted and incorporated into practice;
- expand the remit beyond CO2, to include recent studies that have evaluated the humaneness of some physical methods for killing rodents and the use of anaesthetics and inert gases for killing rodents;
- include zebrafish as well as mice and rats, to encompass the three most commonly used laboratory species;
- discuss special considerations when killing neonatal animals;
- incorporate extended discussion sessions to explore ethical issues and criteria for judging the humaneness of different techniques and devices.
2. Summary of the Meeting
2.1. Inhaled Agents in Rodents
2.1.1. Aversion to CO2 and Inhaled Anaesthetics
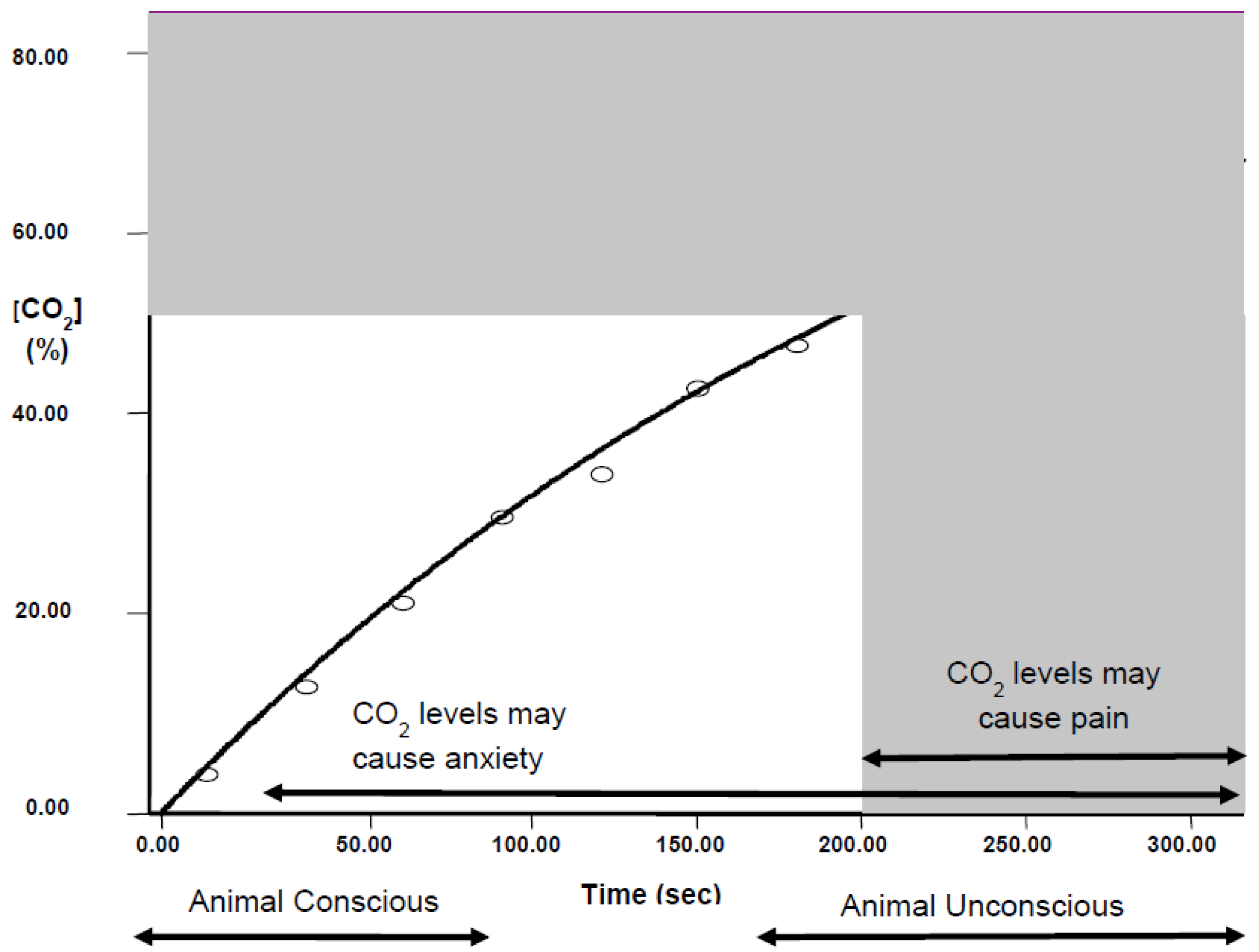
2.1.2. Pain and CO2
2.1.3. Fear and Anxiety with CO2
2.1.4. Aversion to Anoxia
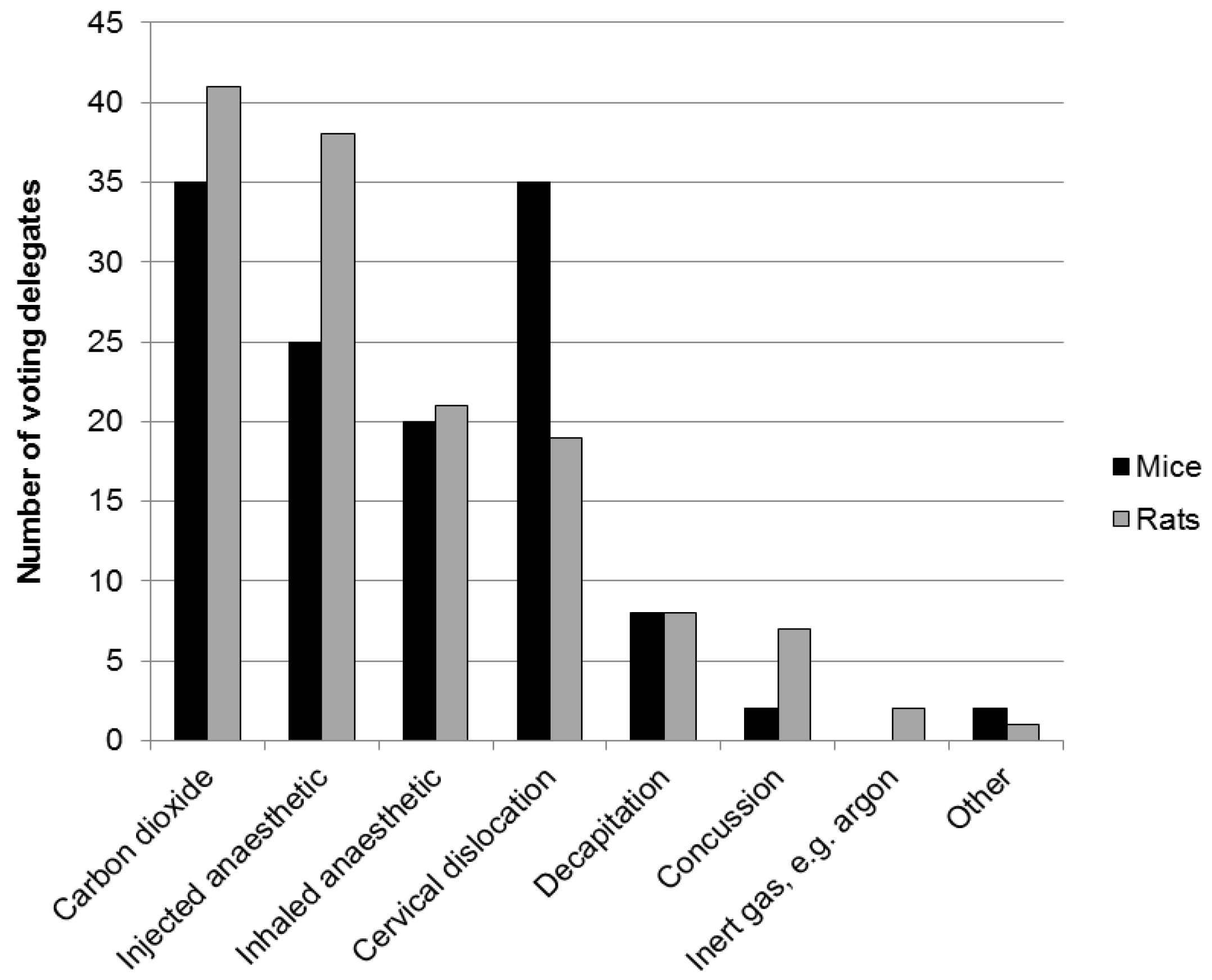
2.2. Physical Methods for Rodents—Decapitation and Cervical Dislocation
2.3. Euthanasia of Neonatal Rodents
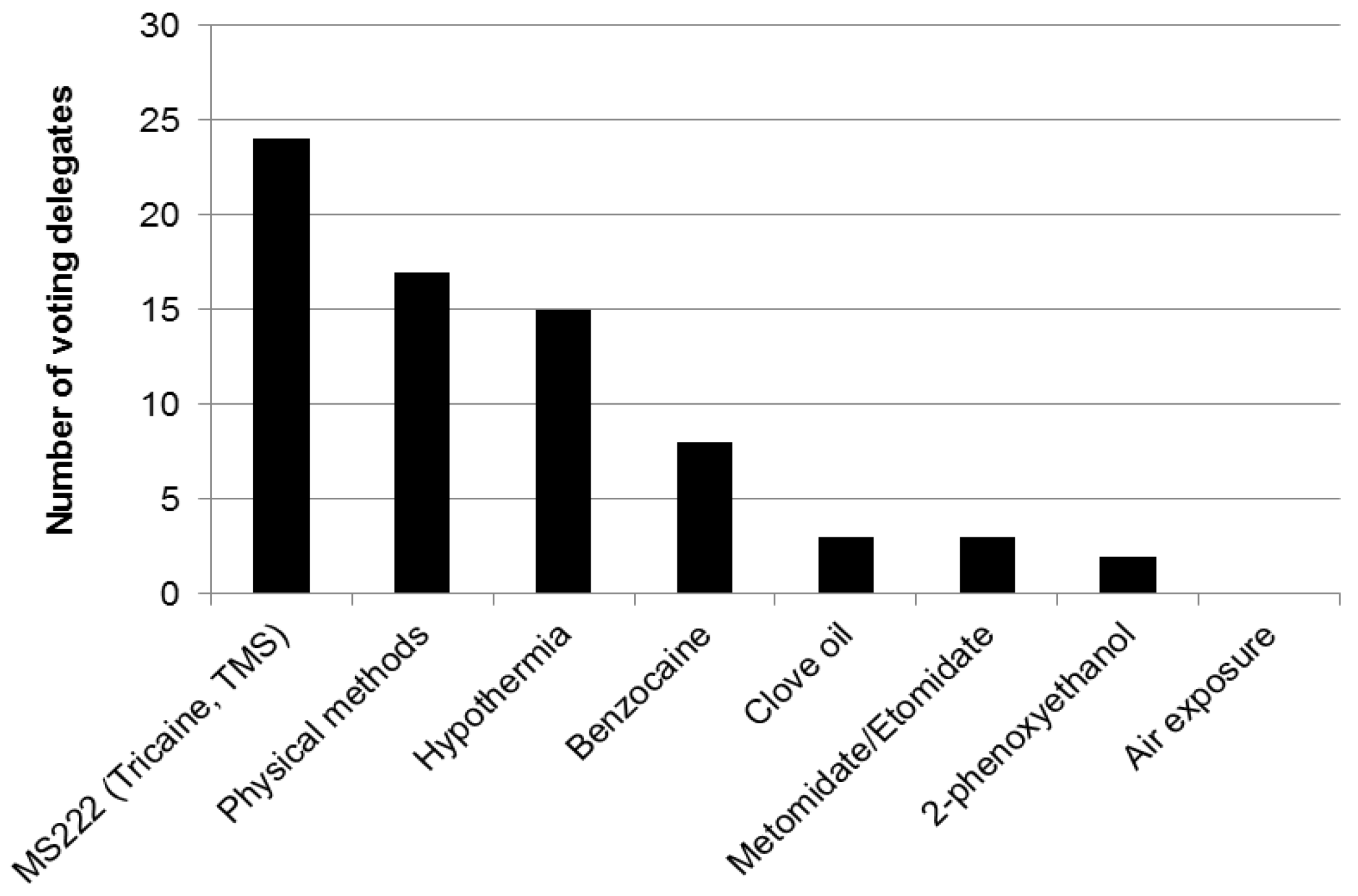
2.4. Euthanasia of Zebrafish
2.5. Standards of Evidence for Making Decisions about Euthanasia Techniques
Commercial Devices
2.6. Input from, and Dialogue with, Ethics or Animal Care and Use Committees
3. Main Points and Areas for Further Research
3.1. Inhaled Agents in Mice and Rats
- The majority of participants considered that all inhaled agents tested to date, including CO2 and inhaled anaesthetics, are aversive to some degree; animals will avoid exposure when provided with the opportunity.
- Approach-avoidance and aversion-avoidance tests have shown that, in general, isoflurane and sevoflurane are equally aversive to mice and rats, halothane is more aversive than either, and that CO2 is inherently more aversive than any of the three other anaesthetics (see Section 2.1.1).
- These tests have also shown that during first exposure to inhaled anaesthetics, more than 50% of rats and mice will choose exposure until they become recumbent rather than abandoning a preferred food reward or crossing over to a brightly lit chamber.
- Results from these tests have shown evidence of learned aversion to inhaled anaesthetics; these agents become more aversive after an initial exposure.
- Conditioned place aversion tests, which rely on an animal’s memory of exposure, confirm that repeat exposure to CO2, isoflurane and argon cause a negative affective response in rats—demonstrated as a learned aversion to the place where the substances were administered.
- Anoxia (as induced by argon or other gases used to displace air) is highly aversive to rats.
3.2. Carbon Dioxide in Mice and Rats
- Mice have specialised olfactory neurons that respond to CO2 and, unlike humans, can smell CO2 at near-atmospheric concentrations, actively avoiding levels of just 0.2%.
- Exposure to a rising concentration of CO2 (e.g., 20% chamber volume per minute) is unlikely to be painful for mice and rats because they lose consciousness before the chamber concentration reaches levels associated with nociceptor activation.
- However, animals exposed to gradual fill as above are still conscious when CO2 reaches levels associated with dyspnoea, and anxiety and fear-related behaviours.
- Mice and rats always leave test chambers when the concentration of CO2 rises to 12%–18%, even if the cost is to abandon a preferred food reward when they are food-deprived or if the cost is to enter a brightly lit chamber, which is known to be highly aversive.
- Humans breathing normoxic but hypercarbic gas report feelings of fear, anxiety and irritability. In rats, breathing a gas mix of the same composition leads to increased activity in areas of the brain involved in the initiation of fear and anxiety-associated behavioural responses.
- In an evaluation of exposure to CO2, plasma levels of the stress hormones adrenaline and noradrenaline (obtained immediately after loss of consciousness) were elevated in mice and rats. Levels were not elevated by the inhaled anaesthetic agents isoflurane or sevoflurane. CO2 can elevate catecholamine levels independent of conscious state; however, the agreement of this study with behavioural studies showing stronger aversion to CO2 than other tested agents suggests that the observed elevations may be related to consciously-experienced negative emotional states.
- Exposure to CO2 is thus likely to cause dyspnoea, anxiety and fear in mice, rats and possibly other rodents. These effects will be exacerbated if the killing process also involves capture, handling, transport to an unfamiliar environment, or mixing with unfamiliar individuals.
- At present, there do not appear to be welfare benefits associated with sedating mice with injectable agents before killing with CO2. A study aimed at identifying suitable injectable anxiolytics, such as midazolam, to pre-medicate mice before CO2 killing, found that the stress of capture, handling and administering the sedative negated the benefits.
- If mice and rats are anaesthetised using an inhaled agent before switching to CO2 to complete the killing process, the change in agent should not be made until they have been recumbent for a sufficiently long period to prevent rapid recovery of consciousness (for mice, evidence indicates waiting a minimum of 80 s after animals become recumbent—see Section 2.1.1).
- This evidence indicates that CO2 and agents used to induce anoxia cannot be considered humane methods of euthanasia for rodents, so developing replacements for these agents is an essential goal.
3.3. Inhaled Anaesthetic Agents in Mice and Rats
- The majority of evidence shows that the use of halogenated anaesthetics to induce unconsciousness in naïve animals is a refinement over the use of CO2 or any agent used to induce anoxia.
- When administered using a precision vaporiser, volatile anaesthetics like sevoflurane and isoflurane rapidly induce unconsciousness but may take a long time to cause death in rodents. This makes these agents suitable for inducing unconsciousness (therefore minimising distress during the period within which animals are able to experience poor welfare), but other methods, e.g., cervical dislocation or CO2 exposure, can be used to more rapidly kill animals once they have been anaesthetised.
- Rats and mice acquire learned aversion to volatile anaesthetic agents such as isoflurane. During repeat exposure to an inhaled anaesthetic, about half as many animals are willing to tolerate exposure until unconsciousness. This result indicates that any study requiring multiple exposures to anaesthesia (e.g., for surgery or imaging, and later for euthanasia) should be considered high risk.
- Rats “generalise” between different inhaled anaesthetics, so using a different agent for killing will not solve the problem of learned aversion.
- There is a clear need to better understand aversion to inhaled anaesthetics, and in particular the effects of repeated anaesthesia.
3.4. Physical Methods for Rodents—Decapitation and Cervical Dislocation
- When performed competently, physical methods may offer a rapid death, which many argue is humane. However, there is on-going debate about the humaneness of some physical methods, including decapitation and cervical dislocation.
- There is concern that a decapitated brain might be conscious and in pain. Both electroencephalogram (EEG) activity and visually evoked potentials (VEP) persist for some seconds in mice and rats, but the significance of this with respect to consciousness and suffering is not yet known. Brain activity also persists following cervical dislocation.
- “Cervical dislocation” can result in lower spinal dislocation and fracture without immediate respiratory arrest, such that animals continue to breathe and may still be conscious.
- In a study of operator success rate for cervical dislocation, the “anterograde” technique (see Section 2.2) proved most effective—but almost 10% of the (anaesthetised) mice continued to breathe for over 3 min, suggesting the technique was not effective.
- Good training, assessment of competence and benchmarking success rates in the use of physical techniques are all paramount with respect to minimising suffering.
3.5. Neonates
- The capacity for a neonate to suffer pain depends upon the species and the stage of neurological development at which individuals are born.
- There is evidence that rodents lack the electrophysiological signs believed to relate to conscious brain activity during the first few postnatal days, with consciousness appearing to emerge several days after birth. This suggests that neonatal rodents may lack the ability to suffer pain or distress during this period. However, research is on-going and there is still a case for giving developmental stages the “benefit of the doubt” and treating them as though they can suffer.
3.6. Zebrafish
- Avoidance responses and conditioned place-preference tests have been used to evaluate different agents used to kill zebrafish.
- Zebrafish show avoidance of propoxate, lidocaine, hydrochloric acid (HCl), 2-phenoxyethanol (2-PE), buffered MS-222 (also known as tricaine mesylate, tricane methanesulfonate, or TMS), benzocaine, isoeugenol and quinaldine sulphate. They show less evidence of avoidance of etomidate and tribromoethanol (TBE), but TBE is difficult to store and can decompose, forming bromide which is hazardous to humans.
- After just a single exposure to buffered MS-222, zebrafish will consistently avoid the chamber where they had encountered this agent, indicating that this experience was unpleasant for the fish and they remember this experience. Zebrafish show much less evidence of conditioned avoidance of either metomidate or clove oil.
- Together these results indicate that etomidate and metomidate could be humane alternatives for euthanizing zebrafish. Clove oil shows promise in conditioned place aversion testing but avoidance testing shows evidence of aversion to one component (isoeugenol), suggesting that this agent requires more study. The most commonly used agent, MS-222, cannot be considered a humane method for killing zebrafish.
3.7. Standards of Evidence for Making Decisions about Techniques for Humane Killing
- Sound scientific evidence is essential when deciding on the most appropriate technique for a particular species and stage of development. Usually this should be in the form of confirmatory publications from multiple laboratories; subjective judgements are not sufficient.
- It is preferable to use a combination of behavioural indicators and physiological data when assessing techniques; for example, a recumbent animal may have an active (high frequency, low amplitude) EEG indicating the possibility of consciousness, or an animal in a state of spasm may have an isoelectric EEG. Supplementary information such as success rates, physicochemical properties of agents or the potential to minimise stressful elements of the process may also be relevant.
- It is good practice to review the references associated with guidelines on humane killing to explore (and question if necessary) the basis for these; as with other areas of science, some guidelines may be based on a small number of studies using few animals or fail to acknowledge the potential implications for animal welfare of data outliers.
- Generalisation between species and strains should be avoided.
- Manufacturers of “euthanasia” devices often attempt to incorporate recent research findings and recommendations, but it is difficult to “test the humaneness” of one against another.
3.8. Areas for Further Research
- On-going research into mouse and rat euthanasia should focus on: identifying new techniques; refining current methods (e.g., administration of inhaled agents in the home cage); understanding the cause of learned aversion to inhaled anaesthetics; and assessing the level of suffering caused by different euthanasia agents.
- More studies are required to elucidate the magnitude of aversion in animals exposed to isoflurane more than twice, and the effect on the magnitude of aversion of longer or short intervals between anaesthesia.
- Research should be undertaken to evaluate when consciousness is lost after the various methods, including physical methods. If some level of consciousness persists even after recumbency (for example), there is a potential for severe suffering.
- More research is needed on types and rates of operator error when using various methods of killing such as cervical dislocation and decapitation, with priority given to the consequences for the animal. Protocols for audits of operator success rate should also be developed.
- The possibility that neonatal rodents have some form of consciousness needs to be further investigated.
- Research into fish euthanasia should focus on: (i) evaluations of the humaneness of some agents currently in use and (ii) identifying new agents and/or methods for commonly used species.
4. Conclusions
- Regularly review practice for humane killing, both at the establishment (e.g., via the ethics or animal care and use committee) and, as a researcher, within your own projects.
- Be aware that killing is a process, not an event. It is important to consider the whole experience of the animal, from capture to permanent loss of consciousness.
- When considering humane killing practice, critically review the basis for current guidelines, including the source literature.
- Ensure good communications between researchers and ethics or animal care and use committees, to help ensure that scientific justifications for particular methods are understood by all, and that causes of suffering are recognised and minimised.
- Critically consider device manufacturers’ claims regarding the humaneness of their products, reviewing the literature and seeking expert advice.
- Consider using isoflurane or sevoflurane as a refinement when killing naïve rodents by inducing anaesthesia prior to introducing CO2.
- If anaesthetising rodents with isoflurane or sevoflurane before changing to CO2, allow sufficient time post-recumbency before changing the agent.
- Do not use injectable anxiolytics before CO2 exposure, as this cannot be considered a refinement.
- If it is not possible to change from CO2 only in the short term, make sure that the administration protocol reflects current thinking regarding good practice for flow rates, use of diffusers, etc. (see also [1]). Consider administering CO2 in the home cage, which will at least eliminate the stress of being moved.
- Never use anoxia (e.g., as induced by argon) to kill rodents.
- Be aware of concerns about physical methods, including both animal and staff welfare. If physical methods are used, ensure that processes are in place to ensure that staff members are trained, competent and willing to use these physical techniques, and that benchmarks for success are set and monitored.
- Do not use decapitation to kill rodents routinely, as this may cause avoidable suffering.
- Follow the literature on sentience in neonatal rodents, giving them the benefit of the doubt with respect to the ability to suffer.
- Use etomidate or metomidate instead of MS-222 to kill zebrafish. Clove oil may also be a suitable refinement to the use of MS-222 but requires more research.
- Keep abreast of the published literature on humane killing techniques.
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Glossary
| Acidosis | an excessively acid condition of the body fluids or tissues. |
| Affective states | emotions and other feelings in animals that are experienced as pleasant or unpleasant. |
| Amygdala | a structure in the limbic system of the brain that is linked to emotions and aggression. |
| Anoxia | absence of oxygen. |
| Anxiogenic | anxiety causing. |
| Anxiolytic | anxiety inhibiting. |
| Approach-avoidance test | a test that uses a desirable reward (e.g., a food treat) to evaluate whether an inhaled agent is aversive and, if so, to what extent. Animals choose whether or not to access the reward in the presence of a range of concentrations of the agent, which provides a way of testing their motivation for the treat against their motivation to avoid the agent. For example, rats and mice always choose to avoid exposure to argon or CO2 even when they are food deprived and a preferred treat is present, indicating that these gases are highly aversive. |
| Aversion | avoidance of a stimulus, situation or behaviour. |
| Aversive | causing avoidance of a stimulus, situation or behaviour. |
| Aversion-avoidance test | a test that uses an animal’s motivation to avoid an unpleasant stimulus to evaluate its level of aversion to an inhaled agent. One commonly used paradigm is for rodents to choose between a preferred, dark chamber that is filling with a gas and an aversively brightly-lit chamber. Rats and mice always prefer the bright chamber over one filling with CO2, but some choose the dark chamber filling with anaesthetic until they lose consciousness. |
| Aversiveness | see aversion, aversive. |
| Chemotaxic | the movement of an organism in response to a chemical stimulus. |
| Conditioned place aversion | a commonly used technique to evaluate aversion to a specific environment that has been associated with a negative reward. |
| Conditioned place preference | a commonly used technique to evaluate preferences for stimuli that have been associated with a positive reward. In general, this procedure involves several trials where the animal is presented with the positive stimulus (e.g., food) paired with placement in a distinct environment containing various cues (e.g., tactile, visual, and/or olfactory). When later tested in the absence of the stimulus, approaches to and/or the amount of time spent in the compartments previously associated with the positive stimulus serve as indicators of preference. |
| Distress | a strongly negative emotional experience, associated with stress of such magnitude or duration that significant changes in biological function would be necessary for the animal to survive if it could not easily escape the stressor. |
| Dyspnoea | a subjective experience of breathing discomfort; “air hunger”. |
| Electroencephalogram (EEG) | a recording of the electrical activity of the brain, typically representing the activity of the cerebral cortex in mammals. |
| Electromyogram (EMG) | a recording of the electrical activity produced by skeletal muscles. |
| Eustress | “good” or “beneficial” stress, occurring in response to positive events, but which may give rise to the same physiological indicators of stress as negative events. |
| Euthanasia | frequently used to describe the process of killing laboratory animals, euthanasia means a “good death”, which the AVMA [47] defines as a death that occurs with “minimal pain and distress”. The term is also often applied to both humans and animals in the context of ending suffering [64]. Given this, the generic use of the term “euthanasia” for laboratory animals is questionable for two reasons. First, the humaneness of some methods used to kill laboratory animals remains in doubt, such that “killing” is often considered a better description of the processes used to end the lives of laboratory animals. Second, animals are sometimes killed for reasons other than ending their suffering (e.g., if an experiment requires their tissues or if they are surplus to requirements). The term “humane killing” is therefore generally more appropriate than euthanasia in relation to laboratory animals. |
| HPA axis | hypothalamic–pituitary–adrenal axis; the system of feedback interactions among the hypothalamus, pituitary gland, and adrenal glands that controls reactions to stress and regulates many body processes. |
| Humane endpoint | one or more predetermined physiological or behavioural signs that define the point at which an experimental animal’s pain and/or distress will be terminated, minimized or reduced by taking actions such as killing the animal, terminating a painful procedure or giving treatment to relieve pain. |
| Humaneness | characterised by concern or compassion for others; in the context of methods of humane killing, that which causes little or no pain or distress. |
| Hypercarbic | containing excess CO2. |
| Hypoxia | deficiency in the amount of oxygen reaching the tissues. |
| Inhaled agent | a substance (gas or vapour) which is administered via the inhalation route. |
| Isoelectric | having or involving no net electric charge or difference in electrical potential; in EEG a signal representing no on-going brain activity. |
| Monosynaptic connections | passing through a single synapse (junction between two nerve cells). |
| Neonatal | relating to the immediate period after birth. The age of neonatal rodents has been defined as up to and including day 10, where the day of birth is day 1 [56]. |
| Nociceptor | a sensory receptor (nerve) for painful stimuli. |
| Normoxic | containing the normal atmospheric concentration of oxygen (~21%). |
| Olfactory bulb | a neural structure of the vertebrate forebrain involved in olfaction or the sense of smell. |
| Pavlovian association | an association between a previously unrelated neutral stimulus (e.g., bell) and another stimulus (e.g., food) that reliably elicits a reaction (e.g., salivation). |
| REM | REM (rapid eye movement) sleep. |
| Visually evoked potential (VEP) | electrophysiological responses to stimulation by either patterned or unpatterned visual stimuli. |
References
- Hawkins, P.; Playle, L.; Golledge, H.; Leach, M.; Banzett, R.; Coenen, A.; Cooper, J.; Danneman, P.; Flecknell, P.; Kirkden, R.; et al. Newcastle Consensus Meeting on Carbon Dioxide Euthanasia of Laboratory Animals, Newcastle upon Tyne, UK, 27–28 February 2006; pp. 1–17. Available online: http://www.nc3rs.org.uk/sites/default/files/documents/Events/First%20Newcastle%20consensus%20meeting%20report.pdf (accessed on 1 December 2015).
- European Union. Directive 2010/63/EU of the European Parliament and of the Council of 22 September 2010 on the Protection of Animals Used for Scientific Purposes. Available online: http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2010:276:0033:0079:en:PDF (accessed on 1 December 2015).
- American Veterinary Medical Association (AVMA). 1986 Report of the AVMA Panel on Euthanasia. J. Am. Vet. Med. Assoc. 1986, 188, 252–286. [Google Scholar]
- Canadian Council on Animal Care (CCAC). CCAC Guidelines on Euthanasia of Animals Used in Science; CCAC: Ottawa, ON, Canada, 2010; pp. 1–32. [Google Scholar]
- Castelhano-Carlos, M.; Baumans, V. The impact of light, noise, cage cleaning and in-house transport on welfare and stress of laboratory rats. Lab. Anim. 2009, 43, 311–327. [Google Scholar] [CrossRef] [PubMed]
- Hurst, J.; West, C. Taming anxiety in laboratory mice. Nat. Methods 2010, 7, 825–826. [Google Scholar] [CrossRef] [PubMed]
- National Research Council. Recognition and Alleviation of Distress in Laboratory Animals; The National Academies Press: Washington, DC, USA, 2008; pp. 1–136. [Google Scholar]
- Makowska, I.J.; Niel, L.; Kirkden, R.D.; Weary, D.M. Rats show aversion to argon-induced hypoxia. Appl. Anim. Behav. Sci. 2008, 114, 572–581. [Google Scholar] [CrossRef]
- Niel, L.; Weary, D.M. Rats avoid exposure to carbon dioxide and argon. Appl. Anim. Behav. Sci. 2007, 107, 100–109. [Google Scholar] [CrossRef]
- Niel, L.; Stewart, S.A.; Weary, D.M. Effect of flow rate on aversion to gradual-fill carbon dioxide euthanasia in rats. Appl. Anim. Behav. Sci. 2008, 109, 77–84. [Google Scholar] [CrossRef]
- Kirkden, R.D.; Niel, L.; Stewart, S.A.; Weary, D.M. Gas killing of rats: The effect of supplemental oxygen on aversion to carbon dioxide. Anim. Welf. 2008, 17, 79–87. [Google Scholar]
- Kirkden, R.D.; Niel, L.; Lee, G.; Makowska, I.J.; Pfaffinger, M.J.; Weary, D.M. The validity of using an approach-avoidance test to measure the strength of aversion to carbon dioxide in rats. Appl. Anim. Behav. Sci. 2008, 114, 216–234. [Google Scholar] [CrossRef]
- Moody, C.M.; Weary, D.M. Mouse aversion to isoflurane versus carbon dioxide gas. Appl. Anim. Behav. Sci. 2014, 158, 95–101. [Google Scholar] [CrossRef]
- Wong, D.; Makowska, I.J.; Weary, D.M. Rat aversion to isoflurane versus carbon dioxide. Biol. Lett. 2013, 9, 20121000. [Google Scholar] [CrossRef] [PubMed]
- Boulanger-Bertolus, J.; Nemeth, G.; Makowska, I.J.; Weary, D.M. Rat aversion to sevoflurane and isoflurane. Appl. Anim. Behav. Sci. 2014, 164, 73–80. [Google Scholar] [CrossRef]
- Makowska, I.J.; Weary, D.M. Rat aversion to induction with inhalant anaesthetics. Appl. Anim. Behav. Sci. 2009, 119, 229–235. [Google Scholar] [CrossRef]
- Makowska, I.J.; Vickers, L.; Mancell, J.; Weary, D.M. Evaluating methods of gas euthanasia for laboratory mice. Appl. Anim. Behav. Sci. 2009, 121, 230–235. [Google Scholar] [CrossRef]
- Golledge, H. Response to Roustan et al. “Evaluating methods of mouse euthanasia on the oocyte quality: Cervical dislocation versus isoflurane inhalation”: Animal welfare concerns regarding the aversiveness of isoflurane and its inability to cause rapid death. Lab. Anim. 2012, 46, 358–359. [Google Scholar] [CrossRef] [PubMed]
- Moody, C.M.; Makowska, J.B.; Weary, D.M. Testing three measures of mouse insensibility following induction with isoflurane or carbon dioxide gas for a more humane euthanasia. Appl. Anim. Behav. Sci. 2015, 163, 183–187. [Google Scholar] [CrossRef]
- Golledge, H.; Lukic, J.; Flecknell, P.A. The effect of behavioural state and cage environment on responses to euthanasia with isoflurane or carbon dioxide in BALB/c mice. In Proceedings of the Altex: 8th World Congress, Montreal, QC, Canada, 21–28 August 2011.
- Pekow, C. Defining, measuring and interpreting stress in laboratory animals. Contemp. Top. Lab. Anim. Sci. 2005, 44, 41–51. [Google Scholar] [PubMed]
- Hallem, E.A.; Sternberg, P.W. Acute carbon dioxide avoidance in Caenorhabditis elegans. Proc. Natl. Acad. Sci. USA 2008, 105, 8038–8043. [Google Scholar] [CrossRef] [PubMed]
- Danneman, P.J.; Stein, S.; Walshaw, S.O. Humane and practical implications of using carbon dioxide mixed with oxygen for anesthesia or euthanasia of rats. Lab. Anim. Sci. 1997, 47, 376–385. [Google Scholar] [PubMed]
- Peppel, P.; Anton, F. Responses of rat medullary dorsal horn neurons following intranasal noxious chemical stimulation—Effects of stimulus intensity, duration and interstimulus interval. J. Neurophysiol. 1993, 70, 2260–2275. [Google Scholar] [PubMed]
- Conlee, K.M.; Stephens, M.L.; Rowan, A.N.; King, L.A. Carbon dioxide for euthanasia: Concerns regarding pain and distress, with special reference to mice and rats. Lab. Anim. 2005, 39, 137–161. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Zhong, C.; Ding, C.; Chi, Q.; Walz, A.; Mombaerts, P.; Matsunami, H.; Luo, M. Detection of near-atmospheric concentrations of CO2 by an olfactory subsystem in the mouse. Science 2007, 317, 953–957. [Google Scholar] [CrossRef] [PubMed]
- Shusterman, D.; Avila, P.C. Real-time monitoring of nasal mucosal pH during carbon dioxide stimulation: Implications for stimulus dynamics. Chem. Senses 2003, 28, 595–601. [Google Scholar] [CrossRef] [PubMed]
- Johnson, P.L.; Fitz, S.D.; Hollis, J.H.; Moratalla, R.; Lightman, S.L.; Shekhar, A.; Lowry, C.A. Induction of c-Fos in “panic/defence”-related brain circuits following brief hypercarbic gas exposure. J. Psychopharmacol. 2011, 25, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Bailey, J.E.; Argyropoulos, S.V.; Kendrick, A.H.; Nutt, D.J. The behavioural and cardiovascular effects of CO2 7.5% in human volunteers. Depress. Anxiety 2005, 21, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Garner, M.; Attwood, A.; Baldwin, D.S.; James, A.; Munafò, M.R. Inhalation of 7.5% carbon dioxide increases threat processing in humans. Neuropsychopharmacology 2011, 36, 1557–1562. [Google Scholar] [CrossRef] [PubMed]
- Bailey, J.E.; Kendrick, A.; Diaper Axotokar, J.P.; Nutt, D.J. A validation of the 7.5% CO2 model of GAD using paroxetine and lorazepam in healthy volunteers. J. Psychopharmacol. 2007, 21, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Ziemann, A.E.; Allen, J.E.; Dahdaleh, N.S.; Drebot, I.I.; Corvell, M.W.; Wunsch, A.M.; Lynch, C.M.; Faraci, F.M.; Howard, M.A.; Welsh, M.J.; et al. The amygdala is a chemosensor that detects carbon dioxide and acidosis to elicit fear behavior. Cell 2009, 139, 1012–1021. [Google Scholar] [CrossRef] [PubMed]
- Environmental Protection Agency. Carbon Dioxide as a Fire Suppressant: Examining the Risks; 430R00002; US Environmental Protection Agency, Office of Air and Radiation, Stratospheric Protection Division: Washington, DC, USA, 2000; pp. 1–54.
- Barklin, A.; Larsson, A.; Vestergaard, C.; Koefoed-Nielsen, J.; Bach, A.; Nyboe, R.; Wogensen, L.; Tønnesen, E. Does brain death induce a pro-inflammatory response at the organ level in a porcine model? Acta Anaesthesiol. Scand. 2008, 52, 621–627. [Google Scholar] [CrossRef] [PubMed]
- Chiari, P.; Hadour, G.; Michel, P.; Piriou, V.; Rodriguez, C.; Budat, C.; Ovize, M.; Jegaden, O.; Lehot, J.J.; Ferrera, R. Biphasic response after brain death induction: Prominent part of catecholamines release in this phenomenon. J. Heart Lung Transplant. 2000, 19, 675–682. [Google Scholar] [CrossRef]
- Valentine, H.; Williams, W.O.; Maurer, K.J. Sedation or inhalant anesthesia before euthanasia with CO2 does not reduce behavioral or physiologic signs of pain and stress in mice. J. Am. Assoc. Lab. Anim. Sci. 2012, 51, 50–57. [Google Scholar] [PubMed]
- Makowska, J.; Golledge, H.D.R.; Marquardt, N.; Weary, D.M. Letter to the editor: Sedation or inhalant anesthesia before euthanasia with CO2 does not reduce behavioral or physiologic signs of pain and stress in mice. J. Am. Assoc. Lab. Anim. Sci. 2012, 51, 396–397. [Google Scholar] [PubMed]
- Williams, W.O.; Riskin, D.K.; Mott, K.M. Ultrasonic sound as an indicator of acute pain in laboratory mice. J. Am. Assoc. Lab. Anim. Sci. 2008, 47, 8–10. [Google Scholar] [PubMed]
- Valentine, H.; Williams, W.O.; Maurer, K.J. Response to Makowska and colleagues’ letter to the editor. J. Am. Assoc. Lab. Anim. Sci. 2012, 51, 397–399. [Google Scholar]
- Arieli, R. Can the rat detect hypoxia in inspired air? Respir. Physiol. 1990, 79, 243–253. [Google Scholar] [CrossRef]
- Mikeska, J.S.; Klemm, W.R. EEG evaluation of humaneness of asphyxia and decapitation euthanasia of the laboratory rat. Lab. Anim. Sci. 1975, 25, 175–179. [Google Scholar] [PubMed]
- Vanderwolf, C.H.; Buzsaki, G.; Cain, D.P.; Cooley, R.K.; Robertson, B. Neocortical and hippocampal electrical activity following decapitation in the rat. Brain Res. 1988, 451, 340–344. [Google Scholar] [CrossRef]
- Van Rijn, C.M.; Krijnen, H.; Menting-Hermeling, S.; Coenen, A.M.L. Decapitation in rats: Latency to unconsciousness and the “wave of death”. PLoS ONE 2011, 6, e16514. [Google Scholar] [CrossRef] [PubMed]
- Kongara, K.; McIhone, A.; Kells, N.; Johnson, C.B. Electroencephalographic evaluation of decapitation of the anaesthetized rat. Lab. Anim. 2014, 48, 15–19. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority (EFSA). Opinion of the Scientific Panel on Animal Health and Welfare on a request from the Commission related to welfare aspects of the main systems of stunning and killing the main commercial species of animals. EFSA J. 2004, 45, 1–29. [Google Scholar]
- Cartner, S.C.; Barlow, S.C.; Ness, T.J. Loss of cortical function in mice after decapitation, cervical dislocation, potassium chloride injection, and CO2 inhalation. Comp. Med. 2007, 57, 570–573. [Google Scholar] [PubMed]
- American Veterinary Medical Association (AVMA). AVMA Guidelines for the Euthanasia of Animals: 2013 Edition; AVMA: Schaumberg, IL, USA, 2013; Available online: https://www.avma.org/KB/Policies/Documents/euthanasia.pdf (accessed on 1 December 2015).
- Derr, R.F. Pain perception in decapitated rat brain. Life Sci. 1991, 49, 1399–1402. [Google Scholar] [CrossRef]
- Carbone, L.; Carbone, E.T.; Yi, E.M.; Bauer, D.B.; Lindstrom, K.A.; Parker, J.M.; Austin, J.A.; Seo, Y.; Gandhi, A.D.; Wikerson, J.D. Assessing cervical dislocation as a humane euthanasia method in mice. J. Am. Assoc. Lab. Anim. Sci. 2012, 51, 352–356. [Google Scholar] [PubMed]
- Gibson, T.J.; Johnson, C.B.; Stafford, K.J.; Mitchinson, S.L.; Mellor, D.J. Validation of the acute electroencephalographic responses of calves to noxious stimulus with scoop dehorning. N. Z. Vet. J. 2007, 55, 152–157. [Google Scholar] [CrossRef] [PubMed]
- Murrell, J.C.; Johnson, C.B. Neurophysiological techniques to assess pain in animals. J. Vet. Pharmacol. Ther. 2006, 29, 325–335. [Google Scholar] [CrossRef] [PubMed]
- Diesch, T.J.; Mellor, D.J.; Johnson, C.B.; Lentle, R.G. Responsiveness to painful stimuli in anaesthetised newborn and young animals of varying neurological maturity (wallaby joeys, rat pups and lambs). AATEX 2008, 14, 549–552. [Google Scholar]
- Johnson, C.B.; Sylvester, S.P.; Stafford, K.J.; Mitchinson, S.L.; Ward, R.N.; Mellor, D.J. Effects of age on the electroencephalographic response to castration in lambs anaesthetized with halothane in oxygen from birth to 6 weeks old. Vet. Anaesth. Analg. 2009, 36, 273–279. [Google Scholar] [CrossRef] [PubMed]
- McCracken, L.; Waran, N.; Mitchinson, S.; Johnson, C.B. Effect of age at castration on behavioural response to subsequent tail docking in lambs. Vet. Anaesth. Analg. 2010, 37, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Diesch, T.J.; Mellor, D.J.; Johnson, C.B.; Lentle, R.G. Electroencephalographic responses to tail clamping in anaesthetised rat pups. Lab. Anim. 2009, 43, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Home Office. Guidance on the Operation of the Animals (Scientific Procedures) Act 1986; HMSO: London, UK, 2014; pp. 1–164. [Google Scholar]
- Zahl, I.H.; Samuelsen, O.; Kiessling, A. Anaesthesia of farmed fish: Implications for welfare. Fish Physiol. Biochem. 2012, 38, 201–218. [Google Scholar] [CrossRef] [PubMed]
- Readman, G.D.; Owen, S.F.; Murrell, J.C.; Knowles, T.G. Do fish perceive anaesthetics as aversive? PLoS ONE 2013, 8, e73773. [Google Scholar] [CrossRef] [PubMed]
- Wong, D.; von Keyserlingk, M.A.; Richards, J.G.; Weary, D.M. Conditioned place avoidance of zebrafish (Danio rerio) to three chemicals used for euthanasia and anaesthesia. PLoS ONE 2014, 9, e88030. [Google Scholar] [CrossRef] [PubMed]
- National Research Council. Guide for the Care and Use of Laboratory Animals, 8th ed.; The National Academies Press: Washington, DC, USA, 2011; pp. 1–220. [Google Scholar]
- Wallace, J. Attitudes to rodent euthanasia techniques: Views of trainee research workers. ALN Eur. 2008, Nov/Dec 2008, 14–18. [Google Scholar]
- Kilkenny, C.; Browne, W.J.; Cuthill, I.C.; Emerson, M.; Altman, D.G. Improving bioscience research reporting: The ARRIVE guidelines for reporting animal research. PLoS Biol. 2010, 8, e1000412. [Google Scholar] [CrossRef] [PubMed]
- European Commission. A Working Document on Animal Welfare Bodies and National Committees to Fulfil the Requirements under the Directive; European Commission: Brussels, Belgium, 2014; pp. 1–28. [Google Scholar]
- Royal College of Veterinary Surgeons (RCVS). Code of Professional Conduct for Veterinary Surgeons and Supporting Guidance; RCVS: London, UK, 2015; pp. 1–165. [Google Scholar]

| Adverse Effect for the Animal | Potential Sources of Suffering or Factors to Consider | Evidence |
|---|---|---|
| Pain | Injection Physicochemical properties of agent (e.g., unbuffered PBS, H2CO3 on mucous membranes) Muscle spasms/seizures Pain from decapitation | Behavioural; physical reactions, vocalisation, attention to site(s) of pain Active EEG Data on duration, incidence, severity of spasms/seizures, observations or electromyogram (EMG) |
| Aversion to inhaled agents | Molecular structure of agent Concentration Flow rate Whether other agents used to induce anaesthesia or as additives Highly species and strain specific | Behavioural; physical reactions (e.g., escape), conditioned place preference/avoidance Information on properties of agent, e.g., pH NOT time to recumbency or to cease movement |
| Suffering between administration and death | Dyspnoea Pain from injury with physical methods Anxiety, fear Inability to escape from aversive agent Other unpleasant effects of inhaled agents | Behavioural (physical reactions, vocalisation, defecation) Active EEG Respiration rate and depth Corticosterone NOT time to recumbency or to cease movement |
| Type of Evidence | Number of Voters |
|---|---|
| Behavioural indicators of stress, e.g., aversion | 47 |
| Behavioural indicators of pain, e.g., attention to eyes or injection sites | 47 |
| Positive indicators that suffering is minimal or absent | 41 |
| EEG data | 40 |
| Success rate | 40 |
| Known properties of agents, e.g., pH, mechanism of action | 35 |
| Experience of animal before euthanasia process | 34 |
| Time to cease moving | 28 |
| Time to death | 26 |
| EMG and other activity data | 19 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hawkins, P.; Prescott, M.J.; Carbone, L.; Dennison, N.; Johnson, C.; Makowska, I.J.; Marquardt, N.; Readman, G.; Weary, D.M.; Golledge, H.D.R. A Good Death? Report of the Second Newcastle Meeting on Laboratory Animal Euthanasia. Animals 2016, 6, 50. https://doi.org/10.3390/ani6090050
Hawkins P, Prescott MJ, Carbone L, Dennison N, Johnson C, Makowska IJ, Marquardt N, Readman G, Weary DM, Golledge HDR. A Good Death? Report of the Second Newcastle Meeting on Laboratory Animal Euthanasia. Animals. 2016; 6(9):50. https://doi.org/10.3390/ani6090050
Chicago/Turabian StyleHawkins, Penny, Mark J. Prescott, Larry Carbone, Ngaire Dennison, Craig Johnson, I. Joanna Makowska, Nicole Marquardt, Gareth Readman, Daniel M. Weary, and Huw D. R. Golledge. 2016. "A Good Death? Report of the Second Newcastle Meeting on Laboratory Animal Euthanasia" Animals 6, no. 9: 50. https://doi.org/10.3390/ani6090050
APA StyleHawkins, P., Prescott, M. J., Carbone, L., Dennison, N., Johnson, C., Makowska, I. J., Marquardt, N., Readman, G., Weary, D. M., & Golledge, H. D. R. (2016). A Good Death? Report of the Second Newcastle Meeting on Laboratory Animal Euthanasia. Animals, 6(9), 50. https://doi.org/10.3390/ani6090050






