Dietary Pyrroloquinoline Quinone Addition Alleviated Weaning Stress via Modulation of Gut Microbiota and Metabolic Profiles in Weaned Piglets
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animal Ethics
2.2. Animals and Experimental Design
2.3. Growth Performance
2.4. Sample Collection and Processing
2.5. Hematological Testing
2.6. Histopathological
2.7. Short-Chain Fatty Acid
2.8. Gut Microbiota
2.9. Metabolomics
2.10. Statistical Analysis
3. Results
3.1. Growth Performance
3.2. Hematology
3.3. Immunity and Antioxidation
3.4. Serum Inflammatory Cytokines
3.5. Intestinal Morphology
3.6. Short-Chain Fatty Acid
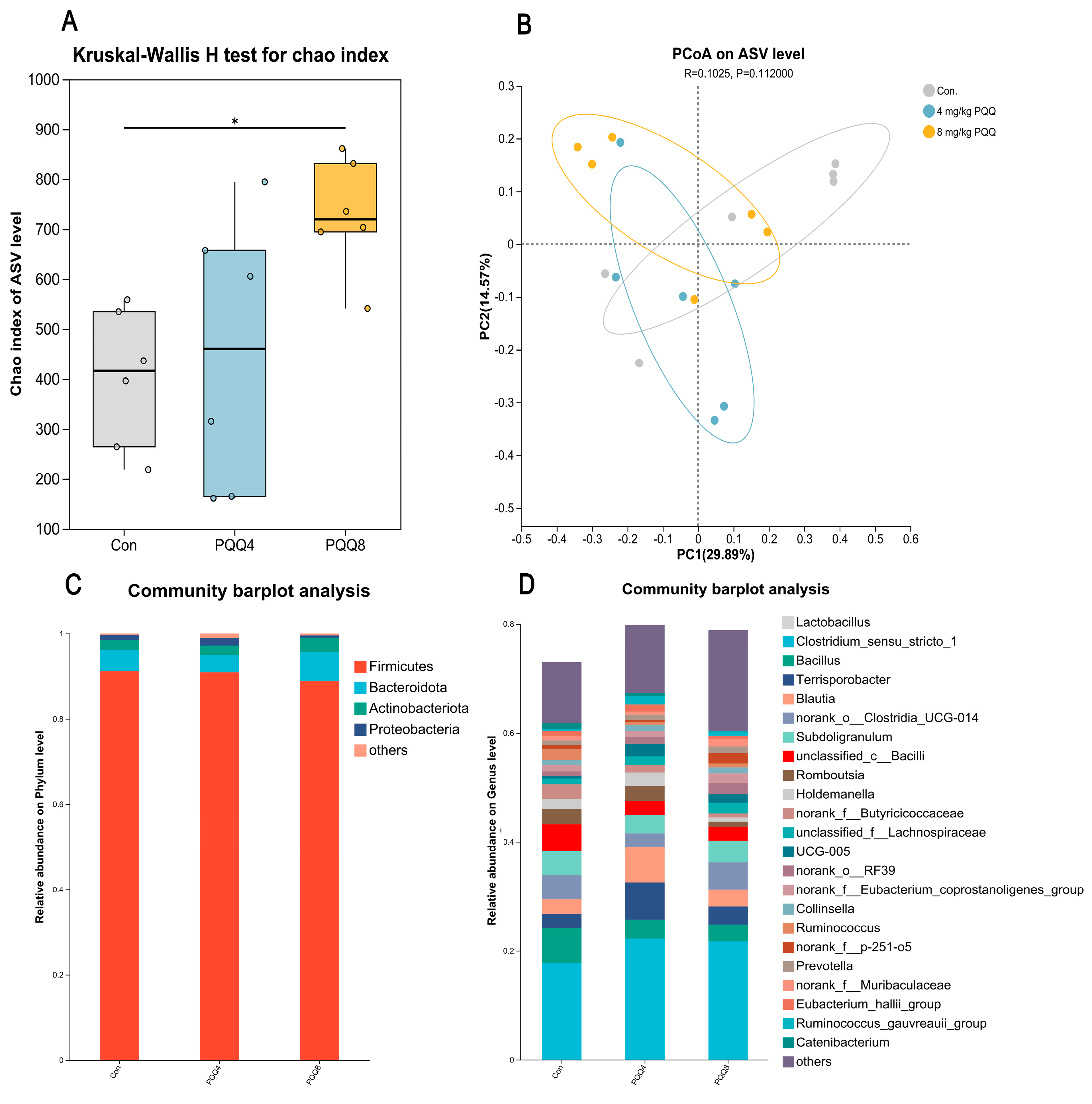
3.7. Gut Microbiota
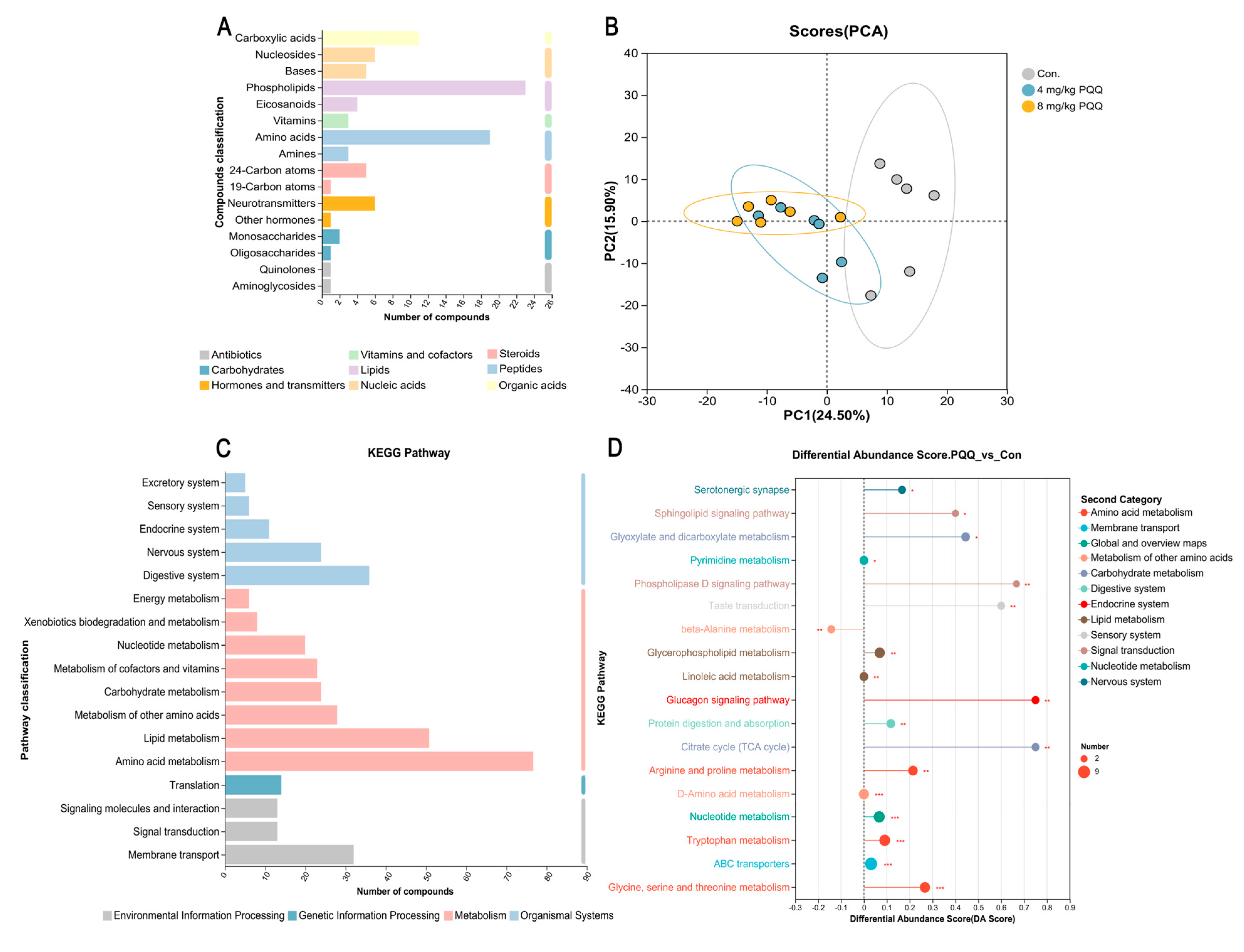
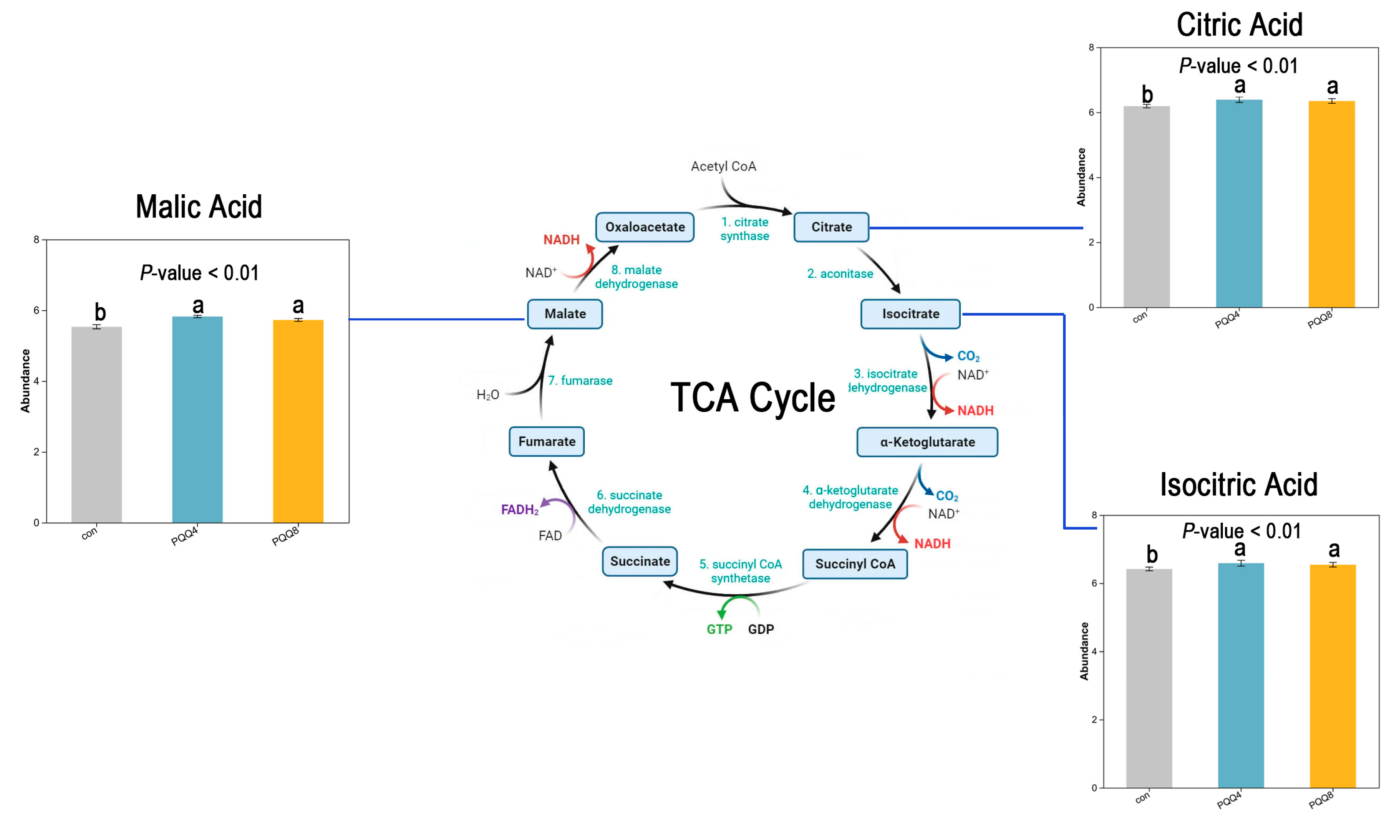
3.8. Metabolism
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ding, S.; Cheng, Y.; Azad, M.A.K.; Zhu, Q.; Huang, P.; Kong, X. Development of small intestinal barrier function and underlying mechanism in Chinese indigenous and Duroc piglets during suckling and weaning periods. Anim. Nutr. 2024, 16, 429–442. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.Y.; Yu, Z.R.; Wang, Z.J.; Ning, R.; Huang, C.Y.; Gao, Y.J.; Wang, F.L. Dietary supplementation with pyrroloquinoline quinone promotes growth, relieves weaning stress, and regulates metabolism of piglets compared with adding zinc oxide. Anim. Nutr. 2023, 15, 409–419. [Google Scholar] [CrossRef] [PubMed]
- He, K.; Nukada, H.; Urakami, T.; Murphy, M.P. Antioxidant and pro-oxidant properties of pyrroloquinoline quinone (PQQ): Implications for its function in biological systems. Biochem. Pharmacol. 2003, 65, 67–74. [Google Scholar] [CrossRef]
- Jonscher, K.R.; Chowanadisai, W.; Rucker, R.B. Pyrroloquinoline-Quinone Is More Than an Antioxidant: A Vitamin-like Accessory Factor Important in Health and Disease Prevention. Biomolecules 2021, 11, 1441. [Google Scholar] [CrossRef]
- Tao, R.; Karliner, J.S.; Simonis, U.; Zheng, J.; Zhang, J.; Honbo, N.; Alano, C.C. Pyrroloquinoline quinone preserves mitochondrial function and prevents oxidative injury in adult rat cardiac myocytes. Biochem. Biophys. Res. Commun. 2007, 363, 257–262. [Google Scholar] [CrossRef][Green Version]
- Huang, C.; Ming, D.; Wang, W.; Wang, Z.; Hu, Y.; Ma, X.; Wang, F. Pyrroloquinoline Quinone Alleviates Jejunal Mucosal Barrier Function Damage and Regulates Colonic Microbiota in Piglets Challenged with Enterotoxigenic Escherichia coli. Front. Microbiol. 2020, 11, 1754. [Google Scholar] [CrossRef]
- Huang, C.Y.; Fan, Z.J.; Han, D.D.; Johnston, L.J.; Ma, X.; Wang, F.L. Pyrroloquinoline quinone regulates the redox status in vitro and in vivo of weaned pigs via the Nrf2/HO-1 pathway. J. Anim. Sci. Biotechnol. 2021, 12, 77. [Google Scholar] [CrossRef]
- Cheng, Q.; Chen, J.; Guo, H.; Lu, J.L.; Zhou, J.; Guo, X.Y.; Shi, Y.; Zhang, Y.; Yu, S.; Zhang, Q.; et al. Pyrroloquinoline quinone promotes mitochondrial biogenesis in rotenone-induced Parkinson’s disease model via AMPK activation. Acta Pharmacol. Sin. 2021, 42, 665–678. [Google Scholar] [CrossRef]
- Jonscher, K.R.; Stewart, M.S.; Alfonso-Garcia, A.; DeFelice, B.C.; Wang, X.X.; Luo, Y.; Levi, M.; Heerwagen, M.J.R.; Janssen, R.C.; de la Houssaye, B.A.; et al. Early PQQ supplementation has persistent long-term protective effects on developmental programming of hepatic lipotoxicity and inflammation in obese mice. FASEB J. 2017, 31, 1434–1448. [Google Scholar] [CrossRef]
- Samuel, K.G.; Zhang, H.J.; Wang, J.; Wu, S.G.; Yue, H.Y.; Sun, L.L.; Qi, G.H. Effects of dietary pyrroloquinoline quinone disodium on growth performance, carcass yield and antioxidant status of broiler chicks. Animal 2015, 9, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Ming, D.; Huang, C.; Wang, W.; Wang, Z.; Shi, C.; Yin, X.; Sun, L.; Gao, Y.; Wang, F. Effects of Diet Supplemented with Excess Pyrroloquinoline Quinone Disodium on Growth Performance, Blood Parameters and Redox Status in Weaned Pigs. Animals 2021, 11, 359. [Google Scholar] [CrossRef]
- Yin, X.; Ming, D.; Bai, L.; Wu, F.; Liu, H.; Chen, Y.; Sun, L.; Wan, Y.; Thacker, P.A.; Wu, G.; et al. Effects of pyrroloquinoline quinone supplementation on growth performance and small intestine characteristics in weaned pigs. J. Anim. Sci. 2019, 97, 246–256. [Google Scholar] [CrossRef]
- Hu, J.; Zhang, S.; Li, M.; Zhao, G. Impact of dietary supplementation with β-alanine on the rumen microbial crude protein supply, nutrient digestibility and nitrogen retention in beef steers elucidated through sequencing the rumen bacterial community. Anim. Nutr. 2024, 17, 418–427. [Google Scholar] [CrossRef]
- ISO 5983-1; Animal Feeding Stuffs-Determination of Nitrogen Content and Calculation of Crude Protein Content-Part 1: Kjeldahl Method. ISO: Geneva, Switzerland, 2005.
- NY/T 65-2004; Feeding standard of swine. Standardization Administration of the People’s Republic of China: Beijing, China, 2004.
- Van Soest, P.J.; Robertson, J.B.; Lewis, B.A. Methods for Dietary Fiber, Neutral Detergent Fiber, and Nonstarch Polysaccharides in Relation to Animal Nutrition. J. Dairy Sci. 1991, 74, 3583–3597. [Google Scholar] [CrossRef]
- GB/T 18246-2019; Determination of Amino Acids in Feeds. Standardization Administration of the People’s Republic of China: Beijing, China, 2019.
- GB/T 6436-2018; Determination of Calcium in Feeds. Standardization Administration of the People’s Republic of China: Beijing, China, 2018.
- GB/T 6437-2018; Determination of Phosphorus in Feeds—Spectrophotometry. Standardization Administration of the People’s Republic of China: Beijing, China, 2018.
- National Research Council. Nutrient Requirements of Swine, 11th ed.; National Academies Press: Washington, DC, USA, 2012. [Google Scholar]
- Arnold, P.K.; Finley, L.W.S. Regulation and function of the mammalian tricarboxylic acid cycle. J. Biol. Chem. 2023, 299, 102838. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, H.J.; Xu, L.; Long, C.; Samuel, K.G.; Yue, H.Y.; Sung, L.L.; Wu, S.G.; Qi, G.H. Dietary supplementation of pyrroloquinoline quinone disodium protects against oxidative stress and liver damage in laying hens fed an oxidized sunflower oil-added diet. Animal 2016, 10, 1129–1136. [Google Scholar] [CrossRef]
- Novais, A.K.; Martel-Kennes, Y.; Roy, C.; Deschene, K.; Beaulieu, S.; Bergeron, N.; Laforest, J.P.; Lessard, M.; Matte, J.J.; Lapointe, J. Tissue-specific profiling reveals modulation of cellular and mitochondrial oxidative stress in normal- and low-birthweight piglets throughout the peri-weaning period. Animal 2020, 14, 1014–1024. [Google Scholar] [CrossRef] [PubMed]
- van Nieuwamerongen, S.E.; Bolhuis, J.E.; van der Peet-Schwering, C.M.C.; Kemp, B.; Soede, N.M. Effects of pre-weaning housing in a multi-suckling system on performance and carbohydrate absorption of relatively light and heavy piglets around weaning. Animal 2018, 12, 802–809. [Google Scholar] [CrossRef] [PubMed]
- Pu, Z.G.; Ge, F.; Zhou, Y.Q.; Liu, A.M.; Yang, C. Pyrroloquinoline quinone protects against murine hepatitis virus strain 3-induced fulminant hepatitis by inhibiting the Keap1/Nrf2 signaling. Cytotechnology 2024, 76, 441–452. [Google Scholar] [CrossRef] [PubMed]
- Hao, J.J.; Ni, X.S.; Giunta, S.; Wu, J.Z.; Shuang, X.P.; Xu, K.Q.; Li, R.; Zhang, W.; Xia, S.J. Pyrroloquinoline quinone delays inflammaging induced by TNF-α through the p16/p21 and Jagged1 signalling pathways. Clin. Exp. Pharmacol. Physiol. 2020, 47, 102–110. [Google Scholar] [CrossRef]
- Yang, C.; Yu, L.; Kong, L.; Ma, R.; Zhang, J.; Zhu, Q.; Zhu, J.; Hao, D. Pyrroloquinoline Quinone (PQQ) Inhibits Lipopolysaccharide Induced Inflammation in Part via Downregulated NF-κB and p38/JNK Activation in Microglial and Attenuates Microglia Activation in Lipopolysaccharide Treatment Mice. PLoS ONE 2014, 9, e109502. [Google Scholar] [CrossRef]
- Shao, D.; Liu, L.J.; Tong, H.B.; Shi, S.R. Dietary pyrroloquinoline quinone improvement of the antioxidant capacity of laying hens and eggs are linked to the alteration of Nrf2/HO-1 pathway and gut microbiota. Food Chem. X 2023, 20, 101021. [Google Scholar] [CrossRef]
- Wang, Z.; Li, Y.; Wang, Y.; Zhao, K.; Chi, Y.; Wang, B. Pyrroloquinoline quinine protects HK-2 cells against high glucose-induced oxidative stress and apoptosis through Sirt3 and PI3K/Akt/FoxO3a signaling pathway. Biochem. Biophys. Res. Commun. 2019, 508, 398–404. [Google Scholar] [CrossRef]
- Tang, X.; Xiong, K.; Fang, R.; Li, M. Weaning stress and intestinal health of piglets: A review. Front. Immunol. 2022, 13, 1042778. [Google Scholar] [CrossRef]
- Bergthaler, A.; Menche, J. The immune system as a social network. Nat. Immunol. 2017, 18, 481–482. [Google Scholar] [CrossRef]
- Zhang, H.Y.; Li, J.Z.; Cao, C.Y.; Zhang, B.R.; Yang, W.; Shi, B.M.; Shan, A.S. Pyrroloquinoline quinone inhibits the production of inflammatory cytokines via the SIRT1/NF-κB signal pathway in weaned piglet jejunum. Food Funct. 2020, 11, 2137–2153. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.Y.; Xu, S.; Huang, C.Y.; Wang, Z.J.; Wang, W.H.; Ming, D.X.; Yin, X.D.; Liu, H.; Wang, F.L. Pyrroloquinoline Quinone Regulates Enteric Neurochemical Plasticity of Weaned Rats Challenged with Lipopolysaccharide. Front. Neurosci. 2022, 16, 878541. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Luo, S.; Liu, S.; Jin, M.; Wang, Y.; Zong, X. Comparative multiomics analyses reveal the breed effect on the colonic host–microbe interactions in pig. iMetaOmics 2024, 1, e8. [Google Scholar] [CrossRef]
- Wang, H.Y.; Xu, R.Y.; Zhang, H.; Su, Y.; Zhu, W.Y. Swine gut microbiota and its interaction with host nutrient metabolism. Anim. Nutr. 2020, 6, 410–420. [Google Scholar] [CrossRef] [PubMed]
- Bell, A.; Brunt, J.; Crost, E.; Vaux, L.; Nepravishta, R.; Owen, C.D.; Latousakis, D.; Xiao, A.; Li, W.; Chen, X.; et al. Elucidation of a sialic acid metabolism pathway in mucus-foraging Ruminococcus gnavus unravels mechanisms of bacterial adaptation to the gut. Nat. Microbiol. 2019, 4, 2393–2404. [Google Scholar] [CrossRef]
- Laplanche, V.; Armiento, S.; Speciale, I.; Šuligoj, T.; Crost, E.H.; Lamprinaki, D.; Vaux, L.; Gotts, K.; De Castro, C.; Juge, N. The human gut symbiont Ruminococcus gnavus displays strain-specific exopolysaccharides modulating the host immune response. Carbohydr. Polym. 2025, 347, 122754. [Google Scholar] [CrossRef] [PubMed]
- Lu, Q.; Wei, Y.; Lin, L.; Liu, J.; Duan, Y.; Li, Y.; Zhai, W.; Liu, Y.; Ang, E.L.; Zhao, H.; et al. The Glycyl Radical Enzyme Arylacetate Decarboxylase from Olsenella scatoligenes. ACS Catal. 2021, 11, 5789–5794. [Google Scholar] [CrossRef]
- Ou, Y.; Guo, Y.; Chen, M.; Lu, X.; Guo, Z.; Zheng, B. Gut microbiome–serum metabolic profiles: Insight into the hypoglycemic effect of Porphyra haitanensis glycoprotein on hyperglycemic mice. Food Funct. 2023, 14, 7977–7991. [Google Scholar] [CrossRef]
- Wastyk, H.C.; Fragiadakis, G.K.; Perelman, D.; Dahan, D.; Merrill, B.D.; Yu, F.B.; Topf, M.; Gonzalez, C.G.; Robinson, J.L.; Elias, J.E.; et al. Gut Microbiota-Targeted Diets Modulate Human Immune Status. bioRxiv 2020. [Google Scholar] [CrossRef]
- Tan, J.K.; Macia, L.; Mackay, C.R. Dietary fiber and SCFAs in the regulation of mucosal immunity. J. Allergy Clin. Immunol. 2023, 151, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Sagar, S.; Faizan, M.I.; Chaudhary, N.; Singh, V.; Singh, P.; Gheware, A.; Sharma, K.; Azmi, I.; Singh, V.P.; Kharya, G.; et al. Obesity impairs cardiolipin-dependent mitophagy and therapeutic intercellular mitochondrial transfer ability of mesenchymal stem cells. Cell Death Dis. 2023, 14, 324. [Google Scholar] [CrossRef]
- Huang, C.; Shi, C.; Li, Z.; Wang, W.; Ming, D.; Gao, Y.; Liu, H.; Ma, X.; Wang, F. Pyrroloquinoline quinone regulates glycolipid metabolism in the jejunum via inhibiting AMPK phosphorylation of weaned pigs. Food Funct. 2022, 13, 9610–9621. [Google Scholar] [CrossRef]
- Hu, R.; Yang, X.; Gong, J.; Lv, J.; Yuan, X.; Shi, M.; Fu, C.; Tan, B.; Fan, Z.; Chen, L.; et al. Patterns of alteration in boar semen quality from 9 to 37 months old and improvement by protocatechuic acid. J. Anim. Sci. Biotechnol. 2024, 15, 78. [Google Scholar] [CrossRef]
- de Laval, B.; Sieweke, M.H. Trained macrophages support hygiene hypothesis. Nat. Immunol. 2017, 18, 1279–1280. [Google Scholar] [CrossRef]
- Urbanczyk, S.; Baris, O.R.; Hofmann, J.; Taudte, R.V.; Guegen, N.; Golombek, F.; Castiglione, K.; Meng, X.; Bozec, A.; Thomas, J. Mitochondrial respiration in B lymphocytes is essential for humoral immunity by controlling the flux of the TCA cycle. Cell Rep. 2022, 39, 110912. [Google Scholar] [CrossRef]
- Machi, K.; Ono, Y.; Iwahashi, H. Citrate, a TCA cycle metabolite, plays a role as a hydroxyl radical scavenger in vitro. J. Photochem. Photobiol. A Chem. 2024, 454, 115691. [Google Scholar] [CrossRef]
- Bauerly, K.; Harris, C.; Chowanadisai, W.; Graham, J.; Havel, P.J.; Tchaparian, E.; Satre, M.; Karliner, J.S.; Rucker, R.B. Altering Pyrroloquinoline Quinone Nutritional Status Modulates Mitochondrial, Lipid, and Energy Metabolism in Rats. PLoS ONE 2011, 6, e21779. [Google Scholar] [CrossRef]
- Sangha, V.; Aboulhassane, S.; Qu, Q.R.; Bendayan, R. Protective effects of pyrroloquinoline quinone in brain folate deficiency. Fluids Barriers CNS 2023, 20, 84. [Google Scholar] [CrossRef] [PubMed]
- Canovai, A.; Tribble, J.R.; Jöe, M.; Westerlund, D.Y.; Amato, R.; Trounce, I.A.; Dal Monte, M.; Williams, P.A. Pyrroloquinoline quinone drives ATP synthesis in vitro and in vivo and provides retinal ganglion cell neuroprotection. Acta Neuropathol. Commun. 2023, 11, 146. [Google Scholar] [CrossRef] [PubMed]



| Item | Content |
|---|---|
| Ingredients | |
| Corn | 67.2 |
| Expanded soybean | 7 |
| Expanded soybean meal (CP 43%) | 13.6 |
| Fermented soybean meal | 5.4 |
| Whey powder | 0.8 |
| Imported fish meal (CP 65%) | 1.2 |
| Rice husk powder | 0.25 |
| Dicalcium hydrogen phosphate | 1.12 |
| Limestone powder | 0.96 |
| Salt | 0.34 |
| Choline chloride | 0.1 |
| Lys (78%) | 0.56 |
| Met (98%) | 0.2 |
| Thr (98%) | 0.2 |
| Try | 0.07 |
| Trace element premix 1 | 0.5 |
| Vitamin premix 1 | 0.5 |
| Total | 100 |
| Nutritional components 2 | |
| OM (On an air-dry diet basis) | 93.68 |
| CP | 19.27 |
| DE (MJ/kg) | 14.1 |
| NDF | 8.32 |
| ADF | 3.81 |
| Lys | 1.3 |
| Met + Cys | 0.78 |
| Thr | 0.86 |
| Ca | 0.78 |
| TP | 0.61 |
| Items | PQQ, mg/kg | p-Value | |||||||
|---|---|---|---|---|---|---|---|---|---|
| 0 | 1 | 2 | 4 | 8 | 16 | T | L | Q | |
| Weight (kg) | |||||||||
| 0 d | 8.01 ± 0.034 | 7.97 ± 0.041 | 7.97 ± 0.027 | 7.98 ± 0.050 | 7.97 ± 0.054 | 7.99 ± 0.067 | 0.993 | 0.999 | 0.901 |
| 14 d | 11.07 ± 0.054 | 11.10 ± 0.084 | 11.24 ± 0.079 | 11.41 ± 0.103 | 11.37 ± 0.091 | 11.28 ± 0.083 | 0.182 | 0.185 | 0.039 |
| 28 d | 15.92 ± 0.271 b | 16.21 ± 0.343 b | 16.55 ± 0.098 ab | 17.39 ± 0.051 a | 17.38 ± 0.175 a | 16.93 ± 0.149 ab | 0.001 | 0.021 | 0.01 |
| ADG (g/d) | |||||||||
| 0–14 d | 219 ± 4.7 | 224 ± 4.6 | 233 ± 5.7 | 245 ± 4.4 | 243 ± 2.8 | 235 ± 1.3 | 0.191 | 0.212 | 0.037 |
| 15–28 d | 347 ± 5.3 b | 365 ± 7.4 ab | 380 ± 5.5 ab | 427 ± 4.9 a | 430 ± 4.2 a | 404 ± 7.9 ab | 0.002 | 0.022 | 0.001 |
| 0–28 d | 283 ± 4.7 b | 295 ± 4.1 ab | 306 ± 5.4 ab | 336 ± 5.8 a | 336 ± 5.7 a | 319 ± 4.2 ab | 0.001 | 0.024 | 0.003 |
| ADFI (g/d) | |||||||||
| 0–14 d | 453 ± 3.4 | 451 ± 5.6 | 452 ± 5.2 | 443 ± 3.1 | 441 ± 2.8 | 439 ± 3.9 | 0.884 | 0.235 | 0.428 |
| 15–28 d | 755 ± 12.5 | 801 ± 10.6 | 831 ± 10.2 | 781 ± 10.8 | 799 ± 12.6 | 777 ± 13.6 | 0.541 | 0.804 | 0.707 |
| 0–28 d | 604 ± 5.8 | 626 ± 6.7 | 642 ± 7.2 | 612 ± 4.3 | 620 ± 4.9 | 608 ± 5.3 | 0.593 | 0.55 | 0.721 |
| F: G | |||||||||
| 0–14 d | 2.07 ± 0.048 | 2.03 ± 0.051 | 1.94 ± 0.037 | 1.82 ± 0.029 | 1.83 ± 0.021 | 1.88 ± 0.051 | 0.051 | 0.054 | 0.006 |
| 15–28 d | 2.19 ± 0.021 a | 2.21 ± 0.030 ab | 2.20 ± 0.071 ab | 1.84 ± 0.063 b | 1.88 ± 0.027 b | 1.93 ± 0.034 ab | 0.02 | 0.023 | 0.006 |
| 0–28 d | 2.14 ± 0.042 a | 2.13 ± 0.031 a | 2.10 ± 0.029 ab | 1.83 ± 0.041 b | 1.86 ± 0.033 b | 1.91 ± 0.028 ab | 0.005 | 0.013 | 0.001 |
| Items | PQQ, mg/kg | p-Value | |||||||
|---|---|---|---|---|---|---|---|---|---|
| 0 | 1 | 2 | 4 | 8 | 16 | T | L | Q | |
| 14 d | |||||||||
| WBC (109/L) | 20.06 ± 0.521 | 20.55 ± 0.375 | 18.89 ± 0.322 | 20.93 ± 0.398 | 21.14 ± 0.541 | 22.32 ± 0.635 | 0.198 | 0.071 | 0.083 |
| RBC (1012/L) | 6.22 ± 0.042 | 6.39 ± 0.032 | 6.20 ± 0.037 | 6.12 ± 0.025 | 6.11 ± 0.020 | 6.36 ± 0.045 | 0.100 | 0.566 | 0.056 |
| HGB (g/L) | 95.42 ± 0.687 | 93.67 ± 0.591 | 96.17 ± 0.575 | 92.42 ± 0.513 | 92.75 ± 0.589 | 93.00 ± 0.879 | 0.436 | 0.228 | 0.3 |
| HCT (%) | 34.67 ± 0.442 | 32.97 ± 0.087 | 34.09 ± 0.413 | 33.12 ± 0.162 | 33.92 ± 0.362 | 34.11 ± 0.171 | 0.262 | 0.743 | 0.617 |
| MCV (fl) | 54.54 ± 0.379 | 55.68 ± 0.219 | 54.09 ± 0.232 | 55.30 ± 0.317 | 54.56 ± 0.198 | 54.28 ± 0.199 | 0.519 | 0.44 | 0.721 |
| MCH (pg) | 15.40 ± 0.050 | 14.90 ± 0.041 | 15.26 ± 0.047 | 15.44 ± 0.083 | 15.38 ± 0.054 | 15.21 ± 0.624 | 0.312 | 0.885 | 0.621 |
| MCHC (g/L) | 283.4 ± 0.85 | 284.3 ± 0.97 | 283.6 ± 0.62 | 281.3 ± 0.65 | 283.4 ± 0.62 | 282.8 ± 0.67 | 0.838 | 0.671 | 0.833 |
| RDW (%) | 20.64 ± 0.784 | 19.48 ± 0.079 | 20.14 ± 0.125 | 20.13 ± 0.176 | 20.00 ± 0.264 | 20.10 ± 0.146 | 0.083 | 0.856 | 0.804 |
| PLT (109/L) | 531.8 ± 9.89 | 547.7 ± 9.72 | 503.3 ± 8.54 | 564.8 ± 11.63 | 566.4 ± 11.97 | 490.0 ± 7.96 | 0.095 | 0.245 | 0.055 |
| 28 d | |||||||||
| WBC (109/L) | 21.10 ± 0.378 | 20.58 ± 0.746 | 19.70 ± 0.248 | 20.13 ± 0.211 | 19.03 ± 0.247 | 19.23 ± 0.263 | 0.310 | 0.059 | 0.078 |
| RBC (1012/L) | 6.09 ± 0.043 | 6.05 ± 0.041 | 5.93 ± 0.051 | 5.80 ± 0.058 | 5.87 ± 0.076 | 5.98 ± 0.062 | 0.106 | 0.45 | 0.071 |
| HGB (g/L) | 94.92 ± 0.468 | 93.00 ± 0.547 | 92.50 ± 0.613 | 92.17 ± 0.546 | 93.00 ± 0.945 | 92.50 ± 0.871 | 0.584 | 0.402 | 0.48 |
| HCT (%) | 32.47 ± 0.632 | 31.33 ± 0.431 | 31.68 ± 0.489 | 31.50 ± 0.178 | 31.64 ± 0.174 | 31.11 ± 0.368 | 0.225 | 0.104 | 0.251 |
| MCV (fl) | 52.45 ± 0.538 | 52.18 ± 0.274 | 52.59 ± 0.387 | 52.68 ± 0.346 | 53.92 ± 0.145 | 53.78 ± 0.201 | 0.112 | 0.071 | 0.063 |
| MCH (pg) | 15.53 ± 0.054 | 15.64 ± 0.046 | 15.47 ± 0.033 | 15.45 ± 0.065 | 15.93 ± 0.076 | 15.78 ± 0.054 | 0.163 | 0.076 | 0.164 |
| MCHC (g/L) | 295.1 ± 1.75 | 296.3 ± 0.98 | 293.0 ± 0.84 | 294.7 ± 1.26 | 292.1 ± 0.84 | 291.2 ± 0.96 | 0.260 | 0.032 | 0.091 |
| RDW (%) | 18.52 ± 0.221 | 18.61 ± 0.537 | 19.34 ± 0.487 | 18.48 ± 0.337 | 18.58 ± 0.429 | 18.95 ± 0.210 | 0.148 | 0.591 | 0.751 |
| PLT (109/L) | 441.7 ± 8.98 | 445.0 ± 6.87 | 431.7 ± 5.78 | 474.2 ± 5.02 | 407.8 ± 6.34 | 406.4 ± 8.52 | 0.204 | 0.081 | 0.217 |
| Items | PQQ, mg/kg | p-Value | ||||
|---|---|---|---|---|---|---|
| 0 | 4 | 8 | T | L | Q | |
| 14 d | ||||||
| IgA (g/L) | 2.18 ± 0.058 | 2.62 ± 0.075 | 2.28 ± 0.042 | 0.167 | 0.760 | 0.166 |
| IgG (g/L) | 17.05 ± 0.952 b | 19.06 ± 0.625 a | 19.35 ± 0.739 a | 0.002 | 0.010 | 0.010 |
| IgM (g/L) | 1.36 ± 0.051 | 1.47 ± 0.023 | 1.45 ± 0.043 | 0.624 | 0.536 | 0.177 |
| T-AOC (U/mL) | 7.61 ± 0.537 b | 8.50 ± 0.424 a | 8.33 ± 0.571 ab | 0.004 | 0.228 | 0.040 |
| SOD (U/mL) | 71.73 ± 1.462 b | 80.96 ± 1.205 a | 76.50 ± 1.168 ab | 0.003 | 0.274 | 0.115 |
| GSH-Px (μmol/L) | 127.61 ± 0.279 b | 140.74 ± 0.254 a | 134.00 ± 0.213 ab | 0.001 | 0.960 | 0.965 |
| TNF-α (ng/mL) | 51.72 ± 0.783 a | 44.35 ± 0.742 b | 43.75 ± 0.751 b | 0.001 | 0.026 | 0.150 |
| IL-1β (ng/mL) | 23.30 ± 0.328 a | 19.36 ± 0.375 b | 18.05 ± 0.312 b | 0.001 | 0.020 | 0.010 |
| IL-6 (ng/mL) | 145.26 ± 1.53 a | 121.10 ± 1.24 b | 115.83 ± 1.87 c | 0.001 | 0.002 | 0.020 |
| 28 d | ||||||
| IgA (g/L) | 2.22 ± 0.258 b | 2.80 ± 0.236 a | 2.60 ± 0.109 a | 0.001 | 0.023 | 0.010 |
| IgG (g/L) | 26.01 ± 0.533 | 27.24 ± 0.418 | 27.41 ± 0.454 | 0.332 | 0.172 | 0.332 |
| IgM (g/L) | 1.38 ± 0.037 | 1.50 ± 0.028 | 1.47 ± 0.030 | 0.624 | 0.555 | 0.627 |
| T-AOC (U/mL) | 10.21 ± 0.688 b | 12.39 ± 0.745 a | 11.12 ± 0.636 b | 0.001 | 0.111 | 0.010 |
| SOD (U/mL) | 90.53 ± 0.845 | 93.14 ± 0.746 | 90.53 ± 0.907 | 0.221 | 1.000 | 0.221 |
| GSH-Px (μmol/L) | 163.88 ± 0.754 | 166.76 ± 0.622 | 167.32 ± 0.619 | 0.480 | 0.256 | 0.479 |
| TNF-α (ng/mL) | 38.74 ± 0.385 | 40.10 ± 0.527 | 38.42 ± 0.372 | 0.187 | 0.296 | 0.157 |
| IL-1β (ng/mL) | 17.66 ± 0.276 | 17.08 ± 0.257 | 17.61 ± 0.293 | 0.159 | 0.718 | 0.294 |
| IL-6 (ng/mL) | 121.14 ± 0.951 a | 104.75 ± 0.924 b | 93.67 ± 0.964 c | 0.001 | 0.010 | 0.010 |
| Items | PQQ, mg/kg | p-Value | ||||
|---|---|---|---|---|---|---|
| 0 | 4 | 8 | T | L | Q | |
| Duodenum | ||||||
| Villus length (μm) | 443.08 ± 15.917 | 422.00 ± 6.949 | 449.94 ± 8.341 | 0.214 | 0.379 | 0.531 |
| Crypt depth (μm) | 467.12 ± 15.206 | 427.02 ± 15.715 | 453.18 ± 26.422 | 0.372 | 0.824 | 0.179 |
| V/C | 0.96 ± 0.061 | 1.00 ± 0.044 | 1.01 ± 0.061 | 0.794 | 0.635 | 0.249 |
| Jejunum | ||||||
| Villus length (μm) | 387.35 ± 10.931 | 363.52 ± 16.905 | 378.79 ± 13.042 | 0.485 | 0.571 | 0.397 |
| Crypt depth (μm) | 307.27 ± 20.922 | 278.60 ± 10.410 | 324.71 ± 9.247 | 0.109 | 0.847 | 0.361 |
| V/C | 1.28 ± 0.065 | 1.31 ± 0.075 | 1.17 ± 0.065 | 0.349 | 0.557 | 0.609 |
| Jejunum | ||||||
| Villus length (μm) | 313.60 ± 19.837 | 268.08 ± 16.218 | 347.75 ± 28.999 | 0.071 | 0.497 | 0.864 |
| Crypt depth (μm) | 328.23 ± 13.086 | 274.58 ± 21.909 | 305.92 ± 27.073 | 0.240 | 0.656 | 0.491 |
| V/C | 0.96 ± 0.067 | 0.98 ± 0.047 | 1.15 ± 0.081 | 0.118 | 0.315 | 0.530 |
| Items | PQQ, mg/kg | p-Value | ||||
|---|---|---|---|---|---|---|
| 0 | 4 | 8 | T | L | Q | |
| Acetic Acid (mg/g) | 16.64 ± 1.477 b | 50.95 ± 2.754 a | 94.56 ± 3.575 a | 0.001 | <0.01 | 0.020 |
| Propionic Acid (mg/g) | 9.16 ± 0.981 c | 40.25 ± 2.554 b | 117.32 ± 4.792 a | 0.001 | <0.01 | 0.347 |
| Isobutyric Acid (mg/g) | 0.027 ± 0.002 c | 0.82 ± 0.143 b | 0.89 ± 0.241 a | 0.007 | <0.01 | 0.882 |
| Butyrate (mg/g) | 5.15 ± 0.438 b | 29.64 ± 3.523 a | 36.51 ± 3.974 a | 0.001 | <0.01 | 0.074 |
| Isovaleric Acid (mg/g) | 0.048 ± 0.006 b | 0.72 ± 0.213 b | 0.83 ± 0.225 a | 0.001 | <0.01 | 0.015 |
| Valeric Acid (mg/g) | 0.22 ± 0.004 c | 0.24 ± 0.087 b | 2.97 ± 0.949 a | 0.001 | <0.01 | 0.040 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, H.; Wang, X.; Peng, W.; Hu, Y.; Xu, Y.; Xiao, X.; Dai, B.; Zhang, R.; Zhong, Y.; Yang, C. Dietary Pyrroloquinoline Quinone Addition Alleviated Weaning Stress via Modulation of Gut Microbiota and Metabolic Profiles in Weaned Piglets. Animals 2025, 15, 2543. https://doi.org/10.3390/ani15172543
Xu H, Wang X, Peng W, Hu Y, Xu Y, Xiao X, Dai B, Zhang R, Zhong Y, Yang C. Dietary Pyrroloquinoline Quinone Addition Alleviated Weaning Stress via Modulation of Gut Microbiota and Metabolic Profiles in Weaned Piglets. Animals. 2025; 15(17):2543. https://doi.org/10.3390/ani15172543
Chicago/Turabian StyleXu, Haocheng, Xiuxi Wang, Wenwen Peng, Yashi Hu, Yangyi Xu, Xiao Xiao, Bing Dai, Ruiqiang Zhang, Yifan Zhong, and Caimei Yang. 2025. "Dietary Pyrroloquinoline Quinone Addition Alleviated Weaning Stress via Modulation of Gut Microbiota and Metabolic Profiles in Weaned Piglets" Animals 15, no. 17: 2543. https://doi.org/10.3390/ani15172543
APA StyleXu, H., Wang, X., Peng, W., Hu, Y., Xu, Y., Xiao, X., Dai, B., Zhang, R., Zhong, Y., & Yang, C. (2025). Dietary Pyrroloquinoline Quinone Addition Alleviated Weaning Stress via Modulation of Gut Microbiota and Metabolic Profiles in Weaned Piglets. Animals, 15(17), 2543. https://doi.org/10.3390/ani15172543






