Reducing Hormonal Inputs in Rabbit Reproduction: Physical Ovulation Induction with a 3D-Printed Cannula
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Vaginal Tissue Collection and Histological Processing
2.3. Semen Collection and Evaluation
2.4. Canula Design
2.5. Synchronization Receptivity Method
2.6. Insemination Procedure
2.7. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AI | Artificial insemination |
| GnRH | Gonadotropin-releasing hormone |
| eCG | Equine chorionic gonadotropin |
| LH | Luteinizing hormone |
| β-NGF | β-Nerve growth factor |
| TCG | Tris-citric acid-glucose |
| FITC-PNA | FITC-labelled peanut agglutinin |
| PI | Propidium iodide |
| LSM | Least square mean |
| SEM | Standard error of the mean |
| GLM | General linear model |
References
- Bakker, J.; Baum, M.J. Neuroendocrine regulation of GnRH release in induced ovulators. Front. Neuroendocrinol. 2000, 21, 220–262. [Google Scholar] [CrossRef] [PubMed]
- Sundquist, C.; Ellis, L.C.; Bartke, A. Reproductive endocrinology of the mink (Mustela vison). Endocr. Rev. 1988, 9, 247–266. [Google Scholar] [CrossRef]
- Ramirez, V.D.; Soufi, W.L. The neuroendocrine control of the rabbit ovarian cycle. In Physiology of Reproduction; Knobil, E., Neill, J.D., Eds.; Raven Press: New York, NY, USA, 1994; pp. 581–611. [Google Scholar]
- Kauffman, A.S.; Rissman, E.F. Neuroendocrine control of mating-induced ovulation. In Knobil and Neill’s Physiology of Reproduction; Neill, J.D., Ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2006; pp. 2283–2326. [Google Scholar]
- Ratto, M.H.; Berland, M.; Silva, M.E.; Adams, G.P. New insights of the role of β-NGF in the ovulation mechanism of induced ovulating species. Reproduction 2019, 157, 199–207. [Google Scholar] [CrossRef]
- Carrasco, R.A.; Singh, J.; Ratto, M.H.; Adams, G.P. Neuroanatomical basis of the nerve growth factor ovulation-induction pathway in llamas. Biol. Reprod. 2021, 104, 578–588. [Google Scholar] [CrossRef]
- Maranesi, M.; Zerani, M.; Leonardi, L.; Pistilli, A.; Arruda-Alencar, J.; Stabile, A.; Rende, M.; Castellini, C.; Petrucci, L.; Parillo, F.; et al. Gene expression and localization of β-NGF and its cognate receptors NTRK1 and NGFR in the sex organs of male rabbits. Reprod. Domest. Anim. 2015, 50, 918–925. [Google Scholar] [CrossRef]
- Casares-Crespo, L.; Vicente, J.S.; Talaván, A.M.; Viudes-de-Castro, M.P. Does the inclusion of protease inhibitors in the insemination extender affect rabbit reproductive performance? Theriogenology 2016, 85, 928–932. [Google Scholar] [CrossRef]
- Casares-Crespo, L.; Fernández-Serrano, P.; Viudes-de-Castro, M.P. Protection of GnRH analogue by chitosan-dextran sulfate nanoparticles for intravaginal application in rabbit artificial insemination. Theriogenology 2018, 116, 49–52. [Google Scholar] [CrossRef] [PubMed]
- García-García, R.M.; Masdeu, M.M.; Sanchez-Rodriguez, A.; Millan, P.; Arias-Alvarez, M.; Sakr, O.G.; Bautista, J.M.; Castellini, C.; Lorenzo, P.L.; Rebollar, P.G. B-nerve growth factor identification in male rabbit genital tract and seminal plasma and its role in ovulation induction in rabbit does. Ital. J. Anim. Sci. 2017, 17, 442–453. [Google Scholar] [CrossRef]
- Castellini, C.; Mattioli, S.; Cotozzolo, E.; Pistilli, A.; Rende, M.; Bartolini, D.; Di Sante, G.; Menchetti, L.; Dal Bosco, A.; Stabile, A.M. The Effect of Interaction NGF/p75NTR in Sperm Cells: A Rabbit Model. Cells 2022, 11, 1035. [Google Scholar] [CrossRef]
- Maranesi, M.; Parillo, F.; Leonardi, L.; Rebollar, P.G.; Alonso, B.; Petrucci, L.; Gobbetti, A.; Boiti, C.; Arruda-Alencar, J.; Moura, A.; et al. Expression of nerve growth factor and its receptors in the uterus of rabbits: Functional involvement in prostaglandin synthesis. Domest. Anim. Endocrinol. 2016, 56, 20–28. [Google Scholar] [CrossRef]
- Maranesi, M.; Petrucci, L.; Leonardi, L.; Piro, F.; Rebollar, P.G.; Millán, P.; Cocci, P.; Vullo, C.; Parillo, F.; Moura, A.; et al. New insights on a NGF-mediated pathway to induce ovulation in rabbits (Oryctolagus cuniculus). Biol. Reprod. 2018, 98, 634–643. [Google Scholar] [CrossRef]
- Silva, M.; Nino, A.; Guerra, M.; Letelier, C.; Valderrama, X.P.; Adams, G.P.; Ratto, M.H. Is an ovulation-inducing factor (OIF) present in the seminal plasma of rabbits? Anim. Reprod. Sci. 2011, 127, 213–221. [Google Scholar] [CrossRef]
- Cervantes, M.P.; Palomino, J.M.; Adams, G.P. In vivo imaging in the rabbit as a model for the study of ovulation-inducing factors. Lab. Anim. 2015, 49, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Rodríguez, A.; Abad, P.; Arias-Alvarez, M.; Millán, P.; Rebollar, P.G.; Bautista, J.M.; Lorenzo, P.L.; García-García, R.M. Recombinant rabbit beta nerve growth factor production and its biological effects on sperm and ovulation in rabbits. PLoS ONE 2019, 14, e0223091. [Google Scholar] [CrossRef]
- Sánchez-Rodríguez, A.; Arias-Alvarez, M.; Millán, P.; Lorenzo, P.L.; García-García, R.M.; Rebollar, P.G. Physiological effects on rabbit sperm and reproductive response to recombinant rabbit beta nerve growth factor administered by intravaginal route in rabbit does. Theriogenology 2020, 157, 327–334. [Google Scholar] [CrossRef]
- Padula, A.M. GnRH analogues-agonists and antagonists. Anim. Reprod. Sci. 2005, 88, 115–126. [Google Scholar] [CrossRef]
- Quintela, L.A.; Pena, A.I.; Vega, M.D.; Gullon, J.; Prieto, M.C.; Barrio, M.; Becerra, J.J.; Maseda, F.; Herradón, P.G. Ovulation induction in rabbit does submitted to artificial insemination by adding buserelin to the seminal dose. Reprod. Nutr. Dev. 2004, 44, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Quintela, L.A.; Peña, A.I.; Vega, M.D.; Gullon, J.; Prieto, C.; Barrio, M.; Becerra, J.J.; Herradón, P.G. Reproductive performance of rabbit does artificially inseminated via intravaginal administration of [des-gly10, D-Ala6]-LHRH ethylamide as ovulation inductor. Reprod. Domest. Anim. 2009, 44, 829–833. [Google Scholar] [CrossRef] [PubMed]
- Dal Bosco, A.; Rebollar, P.G.; Boiti, C.; Zerani, M.; Castellini, C. Ovulation induction in rabbit does: Current knowledge and perspectives. Anim. Reprod. Sci. 2011, 129, 106–117. [Google Scholar] [CrossRef]
- Vicente, J.S.; Lavara, R.; Marco-Jiménez, F.; Viudes-de-Castro, M.P. Detrimental effect on availability of buserelin acetate administered in seminal doses in rabbits. Theriogenology 2011, 76, 1120–1125. [Google Scholar] [CrossRef]
- Viudes-de-Castro, M.P.; Mocé, E.; Lavara, R.; Marco-Jiménez, F.; Vicente, J.S. Aminopeptidase activity in seminal plasma and effect of dilution rate on rabbit reproductive performance after insemination with an extender supplemented with buserelin acetate. Theriogenology 2014, 81, 1223–1228. [Google Scholar] [CrossRef]
- Viudes-de-Castro, M.P.; Marco-Jimenez, F.; Vicente, J.S. Reproductive performance of female rabbits inseminated with extenders supplemented with GnRH analogue entrapped in chitosan-based nanoparticles. Animals 2023, 13, 1628. [Google Scholar] [CrossRef]
- Fernández-Serrano, P.; Casares-Crespo, L.; Viudes-de-Castro, M.P. Efecto de potenciadores de permeabilidad sobre la calidad seminal de conejo. In Proceedings of the XVI Jornadas sobre Producción Animal, Zaragoza, Spain, 19–20 May 2017; pp. 386–388. [Google Scholar]
- Sawyer, C.H.; Markee, J.E.; Hollinshead, W.H. Inhibition of ovulation in the rabbit by the adrenergic-blocking agent dibenamine. Endocrinology 1947, 41, 395–402. [Google Scholar] [CrossRef]
- Sawyer, C.H.; Everett, J.W.; Markee, J.E. A neural factor in the mechanism by which estrogen induces the release of luteinizing hormone in the rat. Endocrinology 1949, 44, 218–233. [Google Scholar] [CrossRef]
- Kim, N.N.; Min, K.; Huang, Y.H.; Goldstein, I.; Traish, A.M. Biochemical and functional characterization of alpha-adrenergic receptors in the rabbit vagina. Life Sci. 2002, 71, 2909–2920. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.J.; Hong, S.K.; Kim, S.W.; Paick, J.S. Histological and functional aspects of different regions of the rabbit vagina. Int. J. Impot. Res. 2003, 15, 142–150. [Google Scholar] [CrossRef]
- Owman, C.; Sjoberg, N.O. Adrenergic nerves in the female genital tract of the rabbit. With remarks on cholinesterase-containing structures. Z. Zellforsch. 1966, 74, 182–197. [Google Scholar] [CrossRef] [PubMed]
- Castellini, C. Recent advances in rabbit artificial insemination. In Proceedings of the 6th World Rabbit Congress, Toulouse, France, 9–12 July 1996; Volume 2, pp. 13–26. [Google Scholar]
- Viudes-de-Castro, M.P.; Casares-Crespo, L.; Marco-Jiménez, F.; Vicente, J.S. Efecto del estímulo físico de la cánula de inseminación sobre la inducción de la ovulación en coneja. In Proceedings of the XVI Jornadas Sobre Producción Animal, AIDA, Zaragoza, Spain, 19–20 May 2017; pp. 380–382. [Google Scholar]
- Viudes-de-Castro, M.P.; Marco-Jiménez, F.; Vicente, J.S. Differential ovulation responses in nulliparous and multiparous rabbit females: A cannula-mediated study. Livest. Sci. 2024, 283, 105475. [Google Scholar] [CrossRef]
- Castillo, J.J.; Viudes-de-Castro, M.P.; Marco-Jimenez, F.; Vicente, J.S. Efecto de la profundidad de inserción de la cánula de inseminación en la fertilidad y prolificidad de conejas no lactantes sin el uso de análogos GnRH. In Proceedings of the XLVIII Symposium de Cunicultura, Córdoba, Spain, 23–24 April 2024; pp. 109–112. [Google Scholar]
- Castillo, J.J.; Vicente, J.S.; Marco-Jiménez, F.; Viudes-de-Castro, M.P. Enhancing ovulation response with cannula use in rabbit artificial insemination. In Proceedings of the 13th World Rabbit Congress, Tarragona, Spain, 2–4 October 2024; pp. 806–809. [Google Scholar]
- Viudes de Castro, M.P.; Vicente, J.S. Effect of sperm count on the fertility and prolificity of meat rabbits. Anim. Reprod. Sci. 1997, 46, 313–319. [Google Scholar] [CrossRef]
- Ratto, M.H.; Wanca, W.; Adams, G.P. Ovulation-inducing factor: A protein component of llama seminal plasma. Reprod. Biol. Endocrinol. 2010, 8, 44. [Google Scholar] [CrossRef] [PubMed]
- Martín-Alguacil, N.; Aardsma, N.; Litvin, Y.; Mayoglou, L.; Dupré, C.; Pfaff, D.W.; Schober, J.M. Immunocytochemical characterization of pacinian-like corpuscles in the labia minora of prepubertal girls. J. Pediatr. Adolesc. Gynecol. 2011, 24, 353–358. [Google Scholar] [CrossRef] [PubMed]
- Baudino, G.F. Anatomy and physiology of the pelvic floor. In Childbirth-Related Pelvic Floor Dysfunction: Risk Factors, Prevention, Evaluation, and Treatment; Springer International Publishing: Cham, Switzerland, 2016; pp. 1–24. [Google Scholar]
- Cruz, Y.; Hudson, R.; Pacheco, P.; Lucio, R.A.; Martínez-Gómez, M. Anatomical and physiological characteristics of perineal muscles in the female rabbit. Physiol. Behav. 2002, 75, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Gómez, M.; Corona-Quintanilla, D.L.; Cruz-Gómez, Y.; Zempoalteca, R.; Rodríguez-Antolín, J.; Castelán, F. The role of pelvic and perineal muscles in reproductive and excretory functions. In Applications of EMG in clinical and Sports Medicine; IntechOpen: London, UK, 2012. [Google Scholar] [CrossRef][Green Version]
- Rocha, M.A.; Sartori, M.G.; De Jesus Simões, M.; Herrmann, V.; Baracat, E.C.; Rodrigues de Lima, G.; Girão, M.J. Impact of pregnancy and childbirth on female rats’ urethral nerve fibers. Int. Urogynecol. J. Pelvic Floor Dysfunct. 2007, 18, 1453–1458. [Google Scholar] [CrossRef]
- Lin, Y.H.; Liu, G.; Li, M.; Xiao, N.; Daneshgari, F. Recovery of continence function following simulated birth trauma involves repair of muscle and nerves in the urethra in the female mouse. Eur. Urol. 2010, 57, 506–512. [Google Scholar] [CrossRef]
- Martínez-Gómez, M.; Mendoza-Martínez, G.; Corona-Quintanilla, D.L.; Fajardo, V.; Rodríguez-Antolín, J.; Castelán, F. Multiparity causes uncoordinated activity of pelvic- and perineal-striated muscles and urodynamic changes in rabbits. Reprod. Sci. 2011, 18, 1246–1252. [Google Scholar] [CrossRef]
- Castelán, F.; Xelhuantzi, N.; Hernández-Aragón, L.G.; Rodríguez-Antolín, J.; Cuevas, E.; Martínez-Gómez, M. Morphometry of paravaginal ganglia from the pelvic plexus: Impact of multiparity, primiparity, and pregnancy. Eur. J. Obstet. Gynecol. Reprod. Biol. 2013, 170, 286–292. [Google Scholar] [CrossRef]
- López-Juárez, R.; Zempoalteca, R.; Corona-Quintanilla, D.L.; Jiménez-Estrada, I.; Castelán, F.; Martínez-Gómez, M. Multiparity modifies contractile properties of pelvic muscles affecting the genesis of vaginal pressure in rabbits. Neurourol. Urodyn. 2018, 37, 106–114. [Google Scholar] [CrossRef]
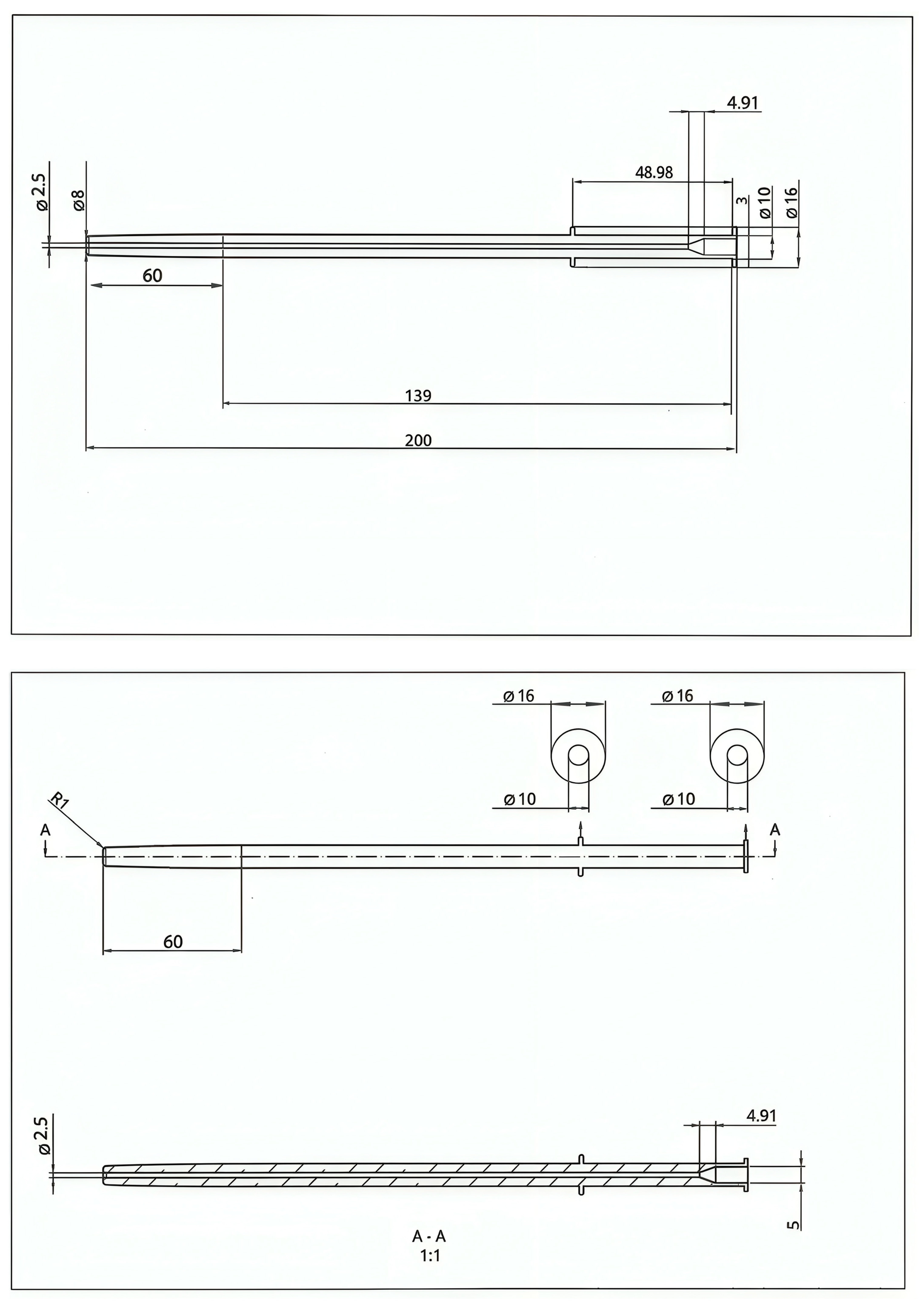

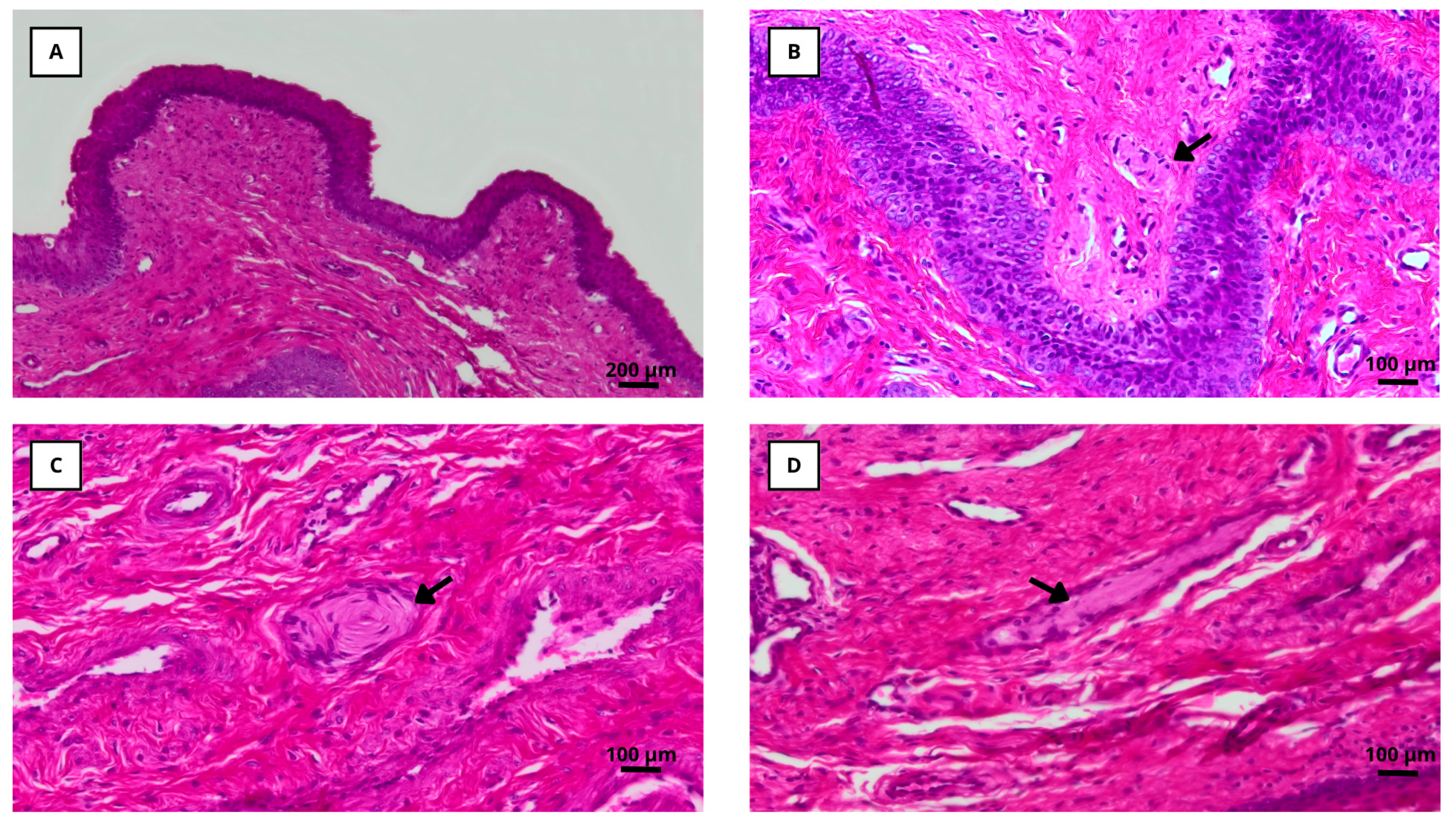
| Meissner Corpuscles LSM ± SEM | Pacinian Corpuscles LSM ± SEM | Ruffini Corpuscles LSM ± SEM | Total LSM ± SEM (n) | |
|---|---|---|---|---|
| Multiparous | 1.0 ± 0.16 | 1.2 ± 0.38 a | 4.4 ± 0.44 | 6.6 ± 0.68 a (30) |
| Nulliparous | 0.6 ± 0.16 | 4.0 ± 0.38 b | 4.3 ± 0.44 | 8.9 ± 0.68 b (30) |
| p-Value | 0.078 | <0.001 | 0.929 | 0.046 |
| Total | 0.8 ± 0.11 | 2.6 ± 0.26 | 4.3 ± 0.31 | 7.7 ± 0.48 |
| Factors | Delivery Rate LSM ± SEM | Total Litter Size LSM ± SEM | Live Litter Size LSM ± SEM | |
|---|---|---|---|---|
| Insemination procedure | Control (n) | 79 ± 4.2 a (105) | 10.3 ± 0.30 (80) | 10.1 ± 0.34 (80) |
| Cannula (n) | 65 ± 3.3 b (220) | 10.5 ± 0.23 (145) | 9.9 ± 0.26 (145) | |
| p-Value | 0.011 | 0.723 | 0.562 | |
| Physiological status | Nulliparous (n) | 76 ± 5.2 (97) | 10.4 ± 0.34 (68) | 9.9 ± 0.38 (68) |
| Multiparous lactating (n) | 66 ± 4.8 (116) | 10.8 ± 0.33 (76) | 10.7 ± 0.37 (76) | |
| Multiparous non-lactating (n) | 75 ± 4.6 (112) | 9.9 ± 0.32 (78) | 9.5 ± 0.36 (78) | |
| p-Value | 0.294 | 0.152 | 0.071 | |
| Synchronization | eCG (n) | 72 ± 3.7 (190) | 10.3 ± 0.25 (134) | 10.0 ± 0.29 (134) |
| Bio (n) | 73 ± 4.3 (135) | 10.5 ± 0.29 (91) | 10.0 ± 0.32 (91) | |
| p-Value | 0.988 | 0.662 | 0.970 | |
| Total (n) | 73 ± 2.9 (325) | 10.4 ± 0.19 (225) | 10.0 ± 0.21 (225) |
| Physiological Status by Insemination Procedure | Delivery Rate LSM ± SEM | Total Litter Size LSM ± SEM | Live Litter Size LSM ± SEM | ||
|---|---|---|---|---|---|
| Nulliparous | Control (n) | 86 ± 6.6 (28) | 10.0 ± 0.55 (24) | 9.5 ± 0.62 (24) | |
| Cannula (n) | 64 ± 5.8 (69) | 10.8 ± 0.41 (44) | 10.3 ± 0.45 (44) | ||
| Multiparous lactating | Control (n) | 65 ± 7.7 (39) | 11.2 ± 0.54 (25) | 11.2 ± 0.60 (25) | |
| Cannula (n) | 68 ± 5.6 (77) | 10.5 ± 0.39 (53) | 10.2 ± 0.43 (53) | ||
| Multiparous non-lactating | Control (n) | 84 ± 5.9 (38) | 9.8 ± 0.49 (31) | 9.7 ± 0.55 (31) | |
| Cannula (n) | 64 ± 5.8 (74) | 10.1 ± 0.41 (48) | 9.3 ± 0.45 (48) | ||
| p-Value | 0.070 | 0.266 | 0.199 | ||
| Synchronization treatment by insemination procedure | |||||
| eCG | Control (n) | 77 ± 5.8 (58) | 10.3 ± 0.42 (43) | 10.3 ± 0.47 (43) | |
| Cannula (n) | 68 ± 4.1 (132) | 10.3 ± 0.29 (91) | 9.8 ± 0.32 (91) | ||
| Bio | Control (n) | 82 ± 5.7 (47) | 10.4 ± 0.44 (37) | 10.0 ± 0.49 (37) | |
| Cannula (n) | 61 ± 5.2 (88) | 10.6 ± 0.37 (54) | 10.1 ± 0.41 (54) | ||
| p-Value | 0.241 | 0.710 | 0.577 | ||
| Physiological status by synchronization treatment | |||||
| Nulliparous | eCG (n) | 75 ± 7.0 (47) | 10.8 ± 0.50 (33) | 10.4 ± 0.55 (33) | |
| Bio (n) | 78 ± 6.7 (50) | 10.0 ± 0.46 (35) | 9.4 ± 0.51 (35) | ||
| Multiparous lactating | eCG (n) | 66 ± 5.9 (74) | 10.2 ± 0.41 (50) | 10.2 ± 0.46 (50) | |
| Bio (n) | 67 ± 7.4 (42) | 11.5 ± 0.51 (28) | 11.1 ± 0.57 (28) | ||
| Multiparous non-lactating | eCG (n) | 77 ± 5.5 (69) | 9.9 ± 0.39 (51) | 9.5 ± 0.43 (51) | |
| Bio (n) | 73 ± 7.2 (43) | 9.9 ± 0.51 (28) | 9.5 ± 0.57 (28) | ||
| p-Value | 0.787 | 0.070 | 0.176 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Castillo, J.J.; Vicente, J.S.; Marco-Jiménez, F.; Aguilar, E.; Viudes-de-Castro, M.P. Reducing Hormonal Inputs in Rabbit Reproduction: Physical Ovulation Induction with a 3D-Printed Cannula. Animals 2025, 15, 2544. https://doi.org/10.3390/ani15172544
Castillo JJ, Vicente JS, Marco-Jiménez F, Aguilar E, Viudes-de-Castro MP. Reducing Hormonal Inputs in Rabbit Reproduction: Physical Ovulation Induction with a 3D-Printed Cannula. Animals. 2025; 15(17):2544. https://doi.org/10.3390/ani15172544
Chicago/Turabian StyleCastillo, Juan José, José Salvador Vicente, Francisco Marco-Jiménez, Enrique Aguilar, and María Pilar Viudes-de-Castro. 2025. "Reducing Hormonal Inputs in Rabbit Reproduction: Physical Ovulation Induction with a 3D-Printed Cannula" Animals 15, no. 17: 2544. https://doi.org/10.3390/ani15172544
APA StyleCastillo, J. J., Vicente, J. S., Marco-Jiménez, F., Aguilar, E., & Viudes-de-Castro, M. P. (2025). Reducing Hormonal Inputs in Rabbit Reproduction: Physical Ovulation Induction with a 3D-Printed Cannula. Animals, 15(17), 2544. https://doi.org/10.3390/ani15172544






