Gallbladder Carcinoma in a Eurasian Otter (Lutra lutra)
Simple Summary
Abstract
1. Introduction
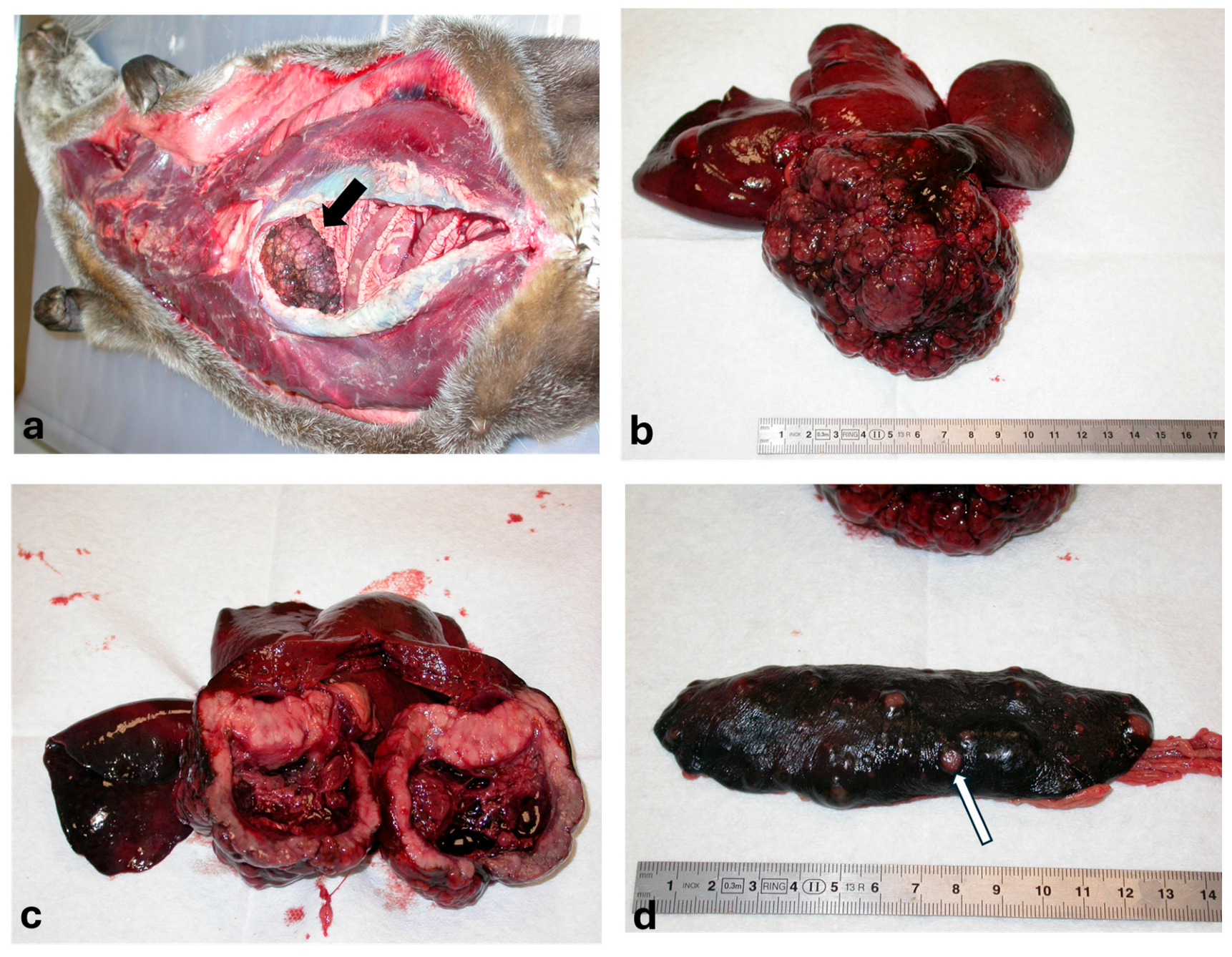
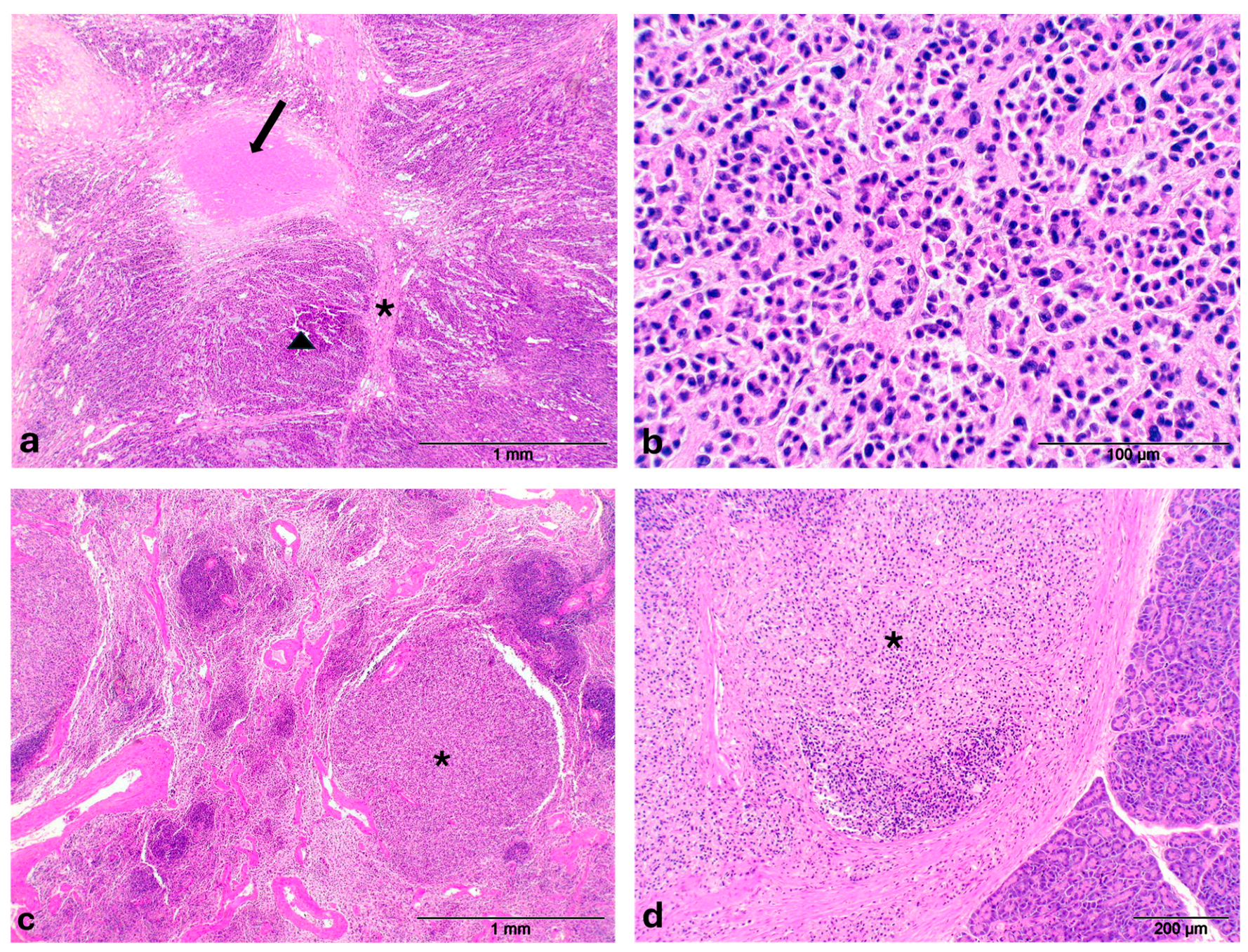
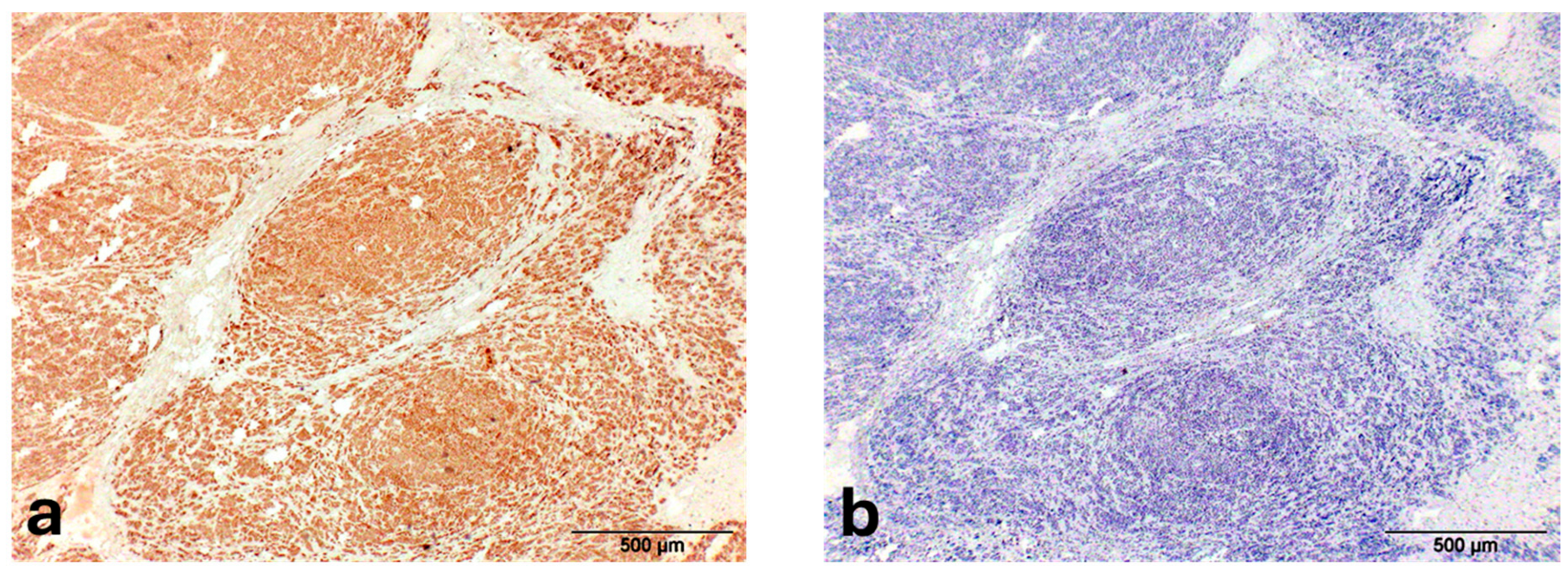
2. Case Description
3. Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Drake, L.E.; Cuff, J.P.; Bedmar, S.; McDonald, R.; Symondson, W.O.C.; Chadwick, E.A. Otterly delicious: Spatiotemporal variation in the diet of a recovering population of Eurasian otters (Lutra lutra) revealed through DNA metabarcoding and morphological analysis of prey remains. Ecol. Evol. 2023, 13, e10038. [Google Scholar] [CrossRef]
- Peracino, V. Progetto Lontra Gran Paradiso. IBEX J. Mt. Ecol. (Collana Sci. Parco Naz. Gran Parad.) 1996, 4, 96. [Google Scholar]
- Convention on the Conservation of European Wildlife and Natural Habitats (Bern Convention)—Decision 82/72/EEC. Available online: https://eur-lex.europa.eu/EN/legal-content/summary/bern-convention.html (accessed on 20 August 2025).
- Council Directive 92/43/EEC of 21 May 1992 on the Conservation of Natural Habitats and of Wild Fauna and Flora. Available online: https://eur-lex.europa.eu/eli/dir/1992/43/oj/eng (accessed on 26 July 2025).
- Red List of Threatened Species. Available online: https://www.iucnredlist.org (accessed on 26 July 2025).
- Rondinini, C.; Battistoni, A.; Peronace, V.; Teofili, C. (Eds.) Lista Rossa IUCN dei Vertebrati Italiani; Comitato Italiano IUCN e Ministero dell’Ambiente e della Tutela del Territorio e del Mare: Roma, Italy, 2013. [Google Scholar]
- Leoncini, F.; Semenzato, P.; Di Febbraro, M.; Loy, A.; Ferrari, C. Come back to stay: Landscape connectivity analysis for the Eurasian otter (Lutra lutra) in the western Alps. Biodivers. Conserv. 2023, 32, 653–669. [Google Scholar] [CrossRef]
- Bartlett, S.L.; Imai, D.M.; Trnpkiewicz, J.G.; Garner, M.M.; Ogasawara, S.; Stokol, T.; Kiupel, M.; Abou-Madi, N.; Kollias, G.V. Intestinal lymphoma of granular lymphocytes in a fisher (Martes pennanti) and a Eurasian otter (Lutra lutra). J. Zoo Wildl. Med. 2010, 41, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Fusillo, R.; Romanucci, M.; Marcelli, M.; Massimini, M.; Della Salda, L. Health and Mortality Monitoring in Threatened Mammals: A First Post Mortem Study of Otters (Lutra lutra L.) in Italy. Animals 2022, 12, 609. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Kim, B.H.; Kim, J.H.; Yoo, M.J.; Kim, D.Y. Lymphosarcoma in a sea otter (Enhydra lutris). J. Wildl. Dis. 2002, 38, 616–617. [Google Scholar] [CrossRef]
- Matt, C.L.; Mans, C.; Doss, G.; Pinkerton, M.; Elsmo, B. Splenic T-cell lymphoma in a North American River Otter (Lontra canadensis). Open Vet. J. 2020, 10, 272–275. [Google Scholar] [CrossRef] [PubMed]
- Stedman, N.L.; Mills, Z.V. Splenic marginal zone lymphoma in an Asian small-clawed otter (Aonyx cinerea). J. Zoo Wildl. Med. 2014, 45, 719–722. [Google Scholar] [CrossRef]
- Swenson, J.; Carpenter, J.W.; Janardhan, K.S.; Ketz-Riley, C.; Brinkman, E. Paresis in an Asian small clawed otter (Aonyx cinereus) associated with vertebral and ischial osteolysis caused by a malignant lymphangiosarcoma. J. Zoo Wildl. Med. 2008, 39, 236–243. [Google Scholar] [CrossRef]
- Tanaka, N.; Izawa, T.; Kashiwagi-Yamamoto, E.; Kuwamura, M.; Ozaki, M.; Nakao, T.; Yamate, J. Primary cerebral T-cell lymphoma in a sea otter (Enhydra lutris). J. Vet. Med. Sci. 2013, 75, 1667–1669. [Google Scholar] [CrossRef]
- Rebollada-Merino, A.; Canales, R.; Romani-Cremaschi, U.; Rodríguez-Bertos, A. Intestinal T-cell lymphoma in an Asian small-clawed otter: Case report and literature review of lymphoma in the subfamily Lutrinae. J. Vet. Diagn. Investig. 2023, 35, 72–75. [Google Scholar] [CrossRef]
- McEntire, M.S.; Tang, K.N.; O’Connor, M.R.; Haulena, M. The management of lymphoproliferative neoplasia in four Northern Sea Otters (Enhydra lutris kenyoni). J. Zoo Wildl. Med. 2024, 55, 511–520. [Google Scholar] [CrossRef] [PubMed]
- Burek-Huntington, K.A.; Mulcahy, D.M.; Doroff, A.M.; Johnson, T.O. Sarcomas in three tree-ranging northern sea otters (Enhydra lutris kenyoni) in Alaska. J. Wildl. Dis. 2012, 48, 483–487. [Google Scholar] [CrossRef]
- Armwood, A.R.; Anderson, C.E.; Clauss, T.; Camus, A.C. Primary intracranial fibrosarcoma in a southern sea otter Enhydra lutris nereis. Dis. Aquat. Organ. 2020, 138, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Stetzer, E.; Williams, T.D.; Nightingale, J.W. Cholangiocellular adenocarcinoma, leiomyoma, and pheochromocytoma in a sea otter. J. Am. Vet. Med. Assoc. 1981, 179, 1283–1284. [Google Scholar] [CrossRef] [PubMed]
- Williams, T.D.; Pulley, L.T. Leiomyomas in two sea otters, Enhydra lutris. J. Wildl. Dis. 1981, 17, 401–404. [Google Scholar] [CrossRef] [PubMed]
- Righton, A.L.; Jungers, H.T.; Mena, A.J.; Stedman, N.L.; St Leger, J.; Langan, J.N.; Rainwater, K.L.; Aziz, N.; Delaney, M.A. Morbidity and mortality of Asian small-clawed otters (Aonyx cinereus) and North American river otters (Lontra canadensis) in US zoos and aquaria from 2000 to 2020: A survey of lesions in 2 popular otter species. Vet. Pathol. 2025, 62, 3009858251345828. [Google Scholar] [CrossRef]
- Fuke, N.; Diep, N.V.; Hirai, T.; Suwanruengsri, M.; Izzati, U.Z.; Yamaguchi, R. Three neoplasms in a Eurasian otter (Lutra lutra): Malignant melanoma, trichoblastoma and mammary gland adenoma. J. Vet. Med. Sci. 2021, 83, 1661–1665. [Google Scholar] [CrossRef]
- Weber, H.; Mecklenburg, L. Malignant melanoma in a Eurasian otter (Lutra lutra). J. Zoo Wildl. Med. 2000, 31, 87–90. [Google Scholar] [CrossRef]
- van de Velde, N.; Demetrick, D.J.; Duignan, P.J. Primary pleural squamous cell carcinoma in a freeranging river otter (Lontra canadensis). J. Wildl. Dis. 2019, 55, 728–732. [Google Scholar] [CrossRef]
- Schlanser, J.R.; Patterson, J.S.; Kiupel, M.; Hencken, C.; Sikarskie, J.G.; Harrison, T.M. Disseminated pheochromocytoma in a North American river otter (Lontra canadensis). J. Zoo Wildl. Med. 2012, 43, 407–411. [Google Scholar] [CrossRef] [PubMed]
- Gaydos, J.K.; Delaney, M.A.; Raverty, S. Causes of Mortality in a Population of Marine-Foraging River Otters (Lontra canadensis). J. Wildl. Dis. 2020, 56, 941–946. [Google Scholar] [CrossRef]
- Pinto, M.H.B.; Braga, T.R.C.; Blume, G.R.; Oliveira, L.B.; Chagas, N.T.C.D.; Souza, F.R.; Cassali, G.D.; Sant’Ana, F.J.F. Metastatic exocrine pancreatic adenocarcinoma in a giant otter (Pteronura brasiliensis). Acta Vet. Hung. 2023, 71, 41–45. [Google Scholar] [CrossRef]
- Bae, I.H.; Pakhrin, B.; Jee, H.; Shin, N.S.; Kim, D.Y. Hepatocellular adenoma in a Eurasian otter (Lutra lutra). J. Vet. Sci. 2007, 8, 103–105. [Google Scholar] [CrossRef]
- Pye, G.W.; White, A.; Robbins, P.K.; Burns, R.E.; Rideout, B.A. Preventive medicine success: Thymoma removal in an African spot-necked otter (Lutra maculicollis). J. Zoo Wildl. Med. 2010, 41, 732–734. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.D.; Mathura, Y. Severe visceral pentastomiasis in an oriental small-clawed otter with functional thyroid carcinoma. J. Vet. Med. Sci. 2018, 80, 320–322. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Ramos Fernandez, J.; Thomas, N.; Dubielzig, R.; Drees, R. Osteosarcoma of the maxilla with concurrent osteoma in a southern sea otter (Enhydra lutris nereis). J. Comp. Pathol. 2012, 147, 391–396. [Google Scholar] [CrossRef]
- Nakamura, K.; Tanimura, H.; Katsuragi, K.; Shibahara, T.; Kadota, K. Differentiated basal cell carcinoma in a Cape clawless otter (Aonyx capensis). J. Comp. Pathol. 2002, 127, 223–227. [Google Scholar] [CrossRef]
- Zachary, J.F.; McDonald, S.C. Malignant ovarian teratoma in a spot-neck otter. J. Am. Vet. Med. Assoc. 1981, 179, 1279–1283. [Google Scholar] [CrossRef]
- Reimer, D.C.; Lipscomb, T.P. Malignant seminoma with metastasis and herpesvirus infection in a free-living sea otter (Enhydra lutris). J. Zoo Wildl. Med. 1998, 29, 35–39. [Google Scholar] [PubMed]
- Cullen, J.M. Tumors of the Liver and Gall Bladder. In Tumors in Domestic Animals, 5th ed.; Meuten, D.J., Ed.; John Wiley & Sons: Ames, IA, USA, 2017; pp. 622–623. [Google Scholar]
- Sreejit, G.; Flynn, M.C.; Patil, M.; Krishnamurthy, P.; Murphy, A.J.; Nagareddy, P.R. S100 family proteins in inflammation and beyond. Adv. Clin. Chem. 2020, 98, 173–231. [Google Scholar]
- Kanthan, R.; Senger, J.L.; Ahmed, S.; Kanthan, S.C. Gallbladder Cancer in the 21st Century. J. Oncol. 2015, 2015, 967472. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Ammapelem, S.; Karamveer, S.; Arunkumar, V.; Prasoon, S. Gallbladder Carcinoma: A Comprehensive Review and Recent Updates. In Hepatobiliary Medicine and Surgery—Gallbladder [Working Title]; Lunevicius, R., Ed.; IntechOpen: London, UK, 2025; Available online: https://www.intechopen.com/online-first/1219270 (accessed on 26 June 2025).
- Vitetta, L.; Sali, A.; Little, P.; Mrazek, L. Gallstones and gall bladder carcinoma. Aust. N. Z. J. Surg. 2000, 70, 667–673. [Google Scholar] [CrossRef]
- Mathur, S.K.; Duhan, A.; Singh, S.; Aggarwal, M.; Aggarwal, G.; Sen, R.; Singh, S.; Garg, S. Correlation of gallstone characteristics with mucosal changes in gallbladder. Trop. Gastroenterol. 2012, 33, 39–44. [Google Scholar] [CrossRef]
- Ushio, N.; Chambers, J.K.; Watanabe, K.I.; Kishimoto, T.E.; Shiga, T.; Li, J.Y.; Nakayama, H.; Uchida, K. Chronic Inflammatory and Proliferative Lesions of the Gallbladder in Aged Pigs. Vet. Pathol. 2020, 57, 122–131. [Google Scholar] [CrossRef]
- MacLeod, A.N.; Reichle, J.K.; Szabo, D.; Cohen, E.B.; Artiles, C.; Fulkerson, C.V.; Kurihara, M.; Mattoon, J. Ultrasonographic Appearance of Gallbladder Neoplasia in 14 Dogs and 1 Cat. Vet. Radiol. Ultrasound 2023, 64, 537–545. [Google Scholar] [CrossRef] [PubMed]
- Madsen, A.B.; Dietz, H.H.; Henriksen, P.; Clausen, B. Survey of Danish Free Living Otters Lutra lutra—A Consecutive Collection and Necroscopy of Dead Bodies. IUCN Otter Spec. Group Bull. 1999, 16, 65–76. [Google Scholar]
- Wells, G.A.; Keymer, I.F.; Barnett, K.C. Suspected Aleutian disease in a wild otter (Lutra lutra). Vet. Rec. 1989, 125, 232–235. [Google Scholar] [CrossRef]
- Simpson, V.R. Health status of otters (Lutra lutra) in south-west England based on postmortem findings. Vet. Rec. 1997, 141, 191–197. [Google Scholar] [CrossRef]
- Rohner, S.; Wohlsein, P.; Prenger-Berninghoff, E.; Ewers, C.; Waindok, P.; Strube, C.; Baechlein, C.; Becher, P.; Wilmes, D.; Rickerts, V.; et al. Pathological Findings in Eurasian Otters (Lutra lutra) Found Dead between 2015–2020 in Schleswig-Holstein, Germany. Animals 2021, 12, 59. [Google Scholar] [CrossRef] [PubMed]
- E’ Morta la Lontra del Centro Visite di Pescasseroli. Available online: https://www.neveappennino.it/news/lontra-pescasseroli-pnalm (accessed on 23 July 2025).
- Pesavento, P.A.; Agnew, D.; Keel, M.K.; Woolard, K.D. Cancer in wildlife: Patterns of emergence. Nat. Rev. Cancer 2018, 18, 646–661. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Domenis, L.; Pezzolato, M.; Biasibetti, E.; Spedicato, R.; Robetto, S. Gallbladder Carcinoma in a Eurasian Otter (Lutra lutra). Animals 2025, 15, 2484. https://doi.org/10.3390/ani15172484
Domenis L, Pezzolato M, Biasibetti E, Spedicato R, Robetto S. Gallbladder Carcinoma in a Eurasian Otter (Lutra lutra). Animals. 2025; 15(17):2484. https://doi.org/10.3390/ani15172484
Chicago/Turabian StyleDomenis, Lorenzo, Marzia Pezzolato, Elena Biasibetti, Raffaella Spedicato, and Serena Robetto. 2025. "Gallbladder Carcinoma in a Eurasian Otter (Lutra lutra)" Animals 15, no. 17: 2484. https://doi.org/10.3390/ani15172484
APA StyleDomenis, L., Pezzolato, M., Biasibetti, E., Spedicato, R., & Robetto, S. (2025). Gallbladder Carcinoma in a Eurasian Otter (Lutra lutra). Animals, 15(17), 2484. https://doi.org/10.3390/ani15172484





