Uncovering Sexual Differences in the External Morphology, Appendicular Muscles, and Internal Organs of a Fossorial Narrow-Mouth Frog (Kaloula borealis)
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. 3D Reconstruction
2.3. External Morphological Characteristics
2.4. Appendicular Muscle Mass
2.5. Internal Organ Mass
2.6. Statistical Analyses
3. Results
3.1. Results of Statistical Analysis
3.1.1. External Morphological Comparisons (Table 2)
| External Morphological Characteristics | Mean ± Standard Deviation | R2 | F | p | |
|---|---|---|---|---|---|
| Males | Females | ||||
| Independent sample t-test | |||||
| 1. Snout-vent length | 1.61 ± 0.02 | 1.63 ± 0.04 | / | 9.266 | 0.03 |
| 2. Body mass | 0.80 ± 0.06 | 0.83 ± 0.11 | / | 7.039 | 0.12 |
| Covariance analysis | |||||
| 3. Head length | 0.95 ± 0.03 | 0.96 ± 0.04 | <0.001 | 0.189 | 0.67 |
| 4. Head width | 1.10 ± 0.03 | 1.09 ± 0.03 | 0.003 | 4.697 | 0.04 |
| 5. Eye diameter | 0.49 ± 0.05 | 0.53 ± 0.04 | 0.009 | 4.091 | 0.05 |
| 6. Upper arm length | 0.96 ± 0.03 | 0.97 ± 0.03 | <0.001 | 0.321 | 0.57 |
| 7. Upper arm width | 0.42 ± 0.06 | 0.41 ± 0.06 | 0.004 | 1.434 | 0.24 |
| 8. Lower arm width | 0.33 ± 0.05 | 0.32 ± 0.05 | 0.005 | 2.400 | 0.13 |
| 9. Lower arm and hand length | 1.27 ± 0.02 | 1.37 ± 0.39 | 0.174 | 2.211 | 0.14 |
| 10. Thigh width | 0.59 ± 0.04 | 0.58 ± 0.05 | 0.010 | 6.470 | 0.01 |
| 11. Tibia length | 1.08 ± 0.03 | 1.09 ± 0.03 | <0.001 | 0.731 | 0.40 |
| 12. Tibia width | 0.49 ± 0.05 | 0.50 ± 0.06 | <0.001 | 0.033 | 0.86 |
| 13. Tarsal length | 0.86 ± 0.03 | 0.87 ± 0.04 | <0.001 | 0.009 | 0.93 |
| 14. Tarsal width | 0.42 ± 0.03 | 0.44 ± 0.02 | <0.001 | 0.355 | 0.56 |
| 15. Foot length | 1.21 ± 0.02 | 1.20 ± 0.03 | 0.001 | 3.865 | 0.06 |
| 16. Thigh length | 1.18 ± 0.03 | 1.19 ± 0.03 | <0.001 | 0.249 | 0.62 |
3.1.2. Appendicular Muscle Mass Comparisons (Table 3)
| Appendicular Muscles | Mean ± Standard Deviation of Dry Mass (mg) | R2 | F | p | |
|---|---|---|---|---|---|
| Males | Females | ||||
| Pectoral girdle muscles | |||||
| 1. Coracobrachialis longus | 0.03 ± 0.21 | 0.06 ± 0.13 | 0.001 | 0.025 | 0.88 |
| 2. Coracobrachialis brevis | 0.20 ± 0.17 | 0.19 ± 0.13 | 0.009 | 0.441 | 0.51 |
| 3. Deltoideus scapularis | 0.51 ± 0.09 | 0.52 ± 0.12 | 0.016 | 3.316 | 0.08 |
| 4. Pectoralis | 0.37 ± 0.12 | 0.40 ± 0.14 | 0.010 | 0.089 | 0.77 |
| 5. Pectoralis abdominis, lateral portion | 0.39 ± 0.09 | 0.42 ± 0.11 | 0.002 | 0.384 | 0.54 |
| 6. Coracoradialis | 0.27 ± 0.09 | 0.26 ± 0.11 | 0.022 | 2.588 | 0.12 |
| Forelimb muscles | |||||
| 1. Flexor carpi radialis | 0.02 ± 0.22 | −0.03 ± 0.29 | 0.069 | 1.065 | 0.31 |
| 2. Flexor digitorum communis | 0.03 ± 0.13 | 0.14 ± 0.11 | 0.087 | 6.122 | 0.02 |
| 3. Anconaeus | 0.19 ± 0.15 | 0.06 ± 0.16 | 0.314 | 16.137 | 0.01 |
| 4. Extensor carpi radialis | 0.17 ± 0.17 | 0.11 ± 0.11 | 0.062 | 3.280 | 0.08 |
| 5. Extensor carpi ulnaris | 0.03 ± 0.15 | 0.12 ± 0.16 | 0.014 | 0.782 | 0.38 |
| 6. Triceps brachii | 0.76 ± 0.09 | 0.81 ± 0.12 | <0.001 | 0.005 | 0.95 |
| 7. Flexor carpi ulnaris | 0.00 ± 0.12 | 0.07 ± 0.16 | 0.007 | 0.401 | 0.53 |
| Abdominal muscles | |||||
| 1. Musculus rectus abdominis | 0.82 ± 0.13 | 0.69 ± 0.10 | 0.260 | 29.458 | 0.01 |
| 2. Obliquus abdominis externus | 1.59 ± 0.11 | 0.90 ± 0.16 | 5.170 | 676.250 | 0.01 |
| 3. Obliquus abdominis internus | 1.62 ± 0.24 | 1.02 ± 0.13 | 4.327 | 145.663 | 0.01 |
| Hindlimb muscles | |||||
| 1. Biceps femoris | 0.27 ± 0.12 | 0.21 ± 0.14 | 0.103 | 7.305 | 0.01 |
| 2. Triceps femoris | 1.33 ± 0.07 | 1.36 ± 0.11 | 0.002 | 0.661 | 0.42 |
| 3. Adductor longus | 0.46 ± 0.09 | 0.52 ± 0.15 | 0.005 | 0.396 | 0.53 |
| 4. Sartorius | 0.26 ± 0.14 | 0.27 ± 0.13 | 0.005 | 0.308 | 0.58 |
| 5. Adductor magnus | 0.98 ± 0.08 | 0.99 ± 0.13 | 0.011 | 2.288 | 0.14 |
| 6. Gracilis major | 0.84 ± 0.07 | 0.83 ± 0.09 | 0.029 | 8.396 | 0.01 |
| 7. Gracilis minor | 0.46 ± 0.11 | 0.44 ± 0.19 | 0.048 | 2.5475 | 0.12 |
| 8. Semimembranosus | 0.59 ± 0.09 | 0.56 ± 0.11 | 0.053 | 8.796 | 0.01 |
| 9. Semitendinosus | 0.64 ± 0.07 | 0.63 ± 0.11 | 0.021 | 4.943 | 0.03 |
| 10. Piriformis | 0.07 ± 0.11 | 0.09 ± 0.19 | <0.001 | 0.007 | 0.93 |
| 11. Iliopsoas | 0.30 ± 0.12 | 0.27 ± 0.19 | 0.075 | 4.008 | 0.05 |
| 12. Tibialis anticus longus | 0.79 ± 0.08 | 0.81 ± 0.12 | 0.007 | 1.384 | 0.25 |
| 13. Tastrocnemius | 0.87 ± 0.14 | 0.90 ± 0.09 | <0.001 | 0.022 | 0.88 |
| 14. Peroneus | 0.54 ± 0.09 | 0.58 ± 0.10 | <0.001 | 0.005 | 0.95 |
| 15. Extensor cruris | 0.03 ± 0.09 | 0.06 ± 0.11 | <0.001 | 0.008 | 0.93 |
| 16. Tibialis posticus | 0.05 ± 0.09 | 0.01 ± 0.16 | 0.079 | 7.399 | 0.01 |
3.1.3. Internal Organ Mass Comparisons (Table 4)
| Mean ± Standard Deviation of Dry Mass (mg) | R2 | F | p | ||
|---|---|---|---|---|---|
| Males | Females | ||||
| 1. Heart | 1.25 ± 0.15 | 1.22 ± 0.21 | 0.112 | 6.778 | 0.01 |
| 2. Liver | 1.58 ± 0.15 | 1.64 ± 0.29 | 0.015 | 0.534 | 0.47 |
| 3. Spleen | −0.61 ± 0.35 | −0.77 ± 0.29 | 0.386 | 3.934 | 0.06 |
| 4. Lung | 1.11 ± 0.09 | 1.04 ± 0.11 | 0.138 | 24.952 | 0.01 |
| 5. Kidney | 0.89 ± 0.06 | 0.94 ± 0.14 | 0.002 | 0.169 | 0.68 |
| 6. Digestive tract | 1.67 ± 0.10 | 1.71 ± 0.14 | 0.001 | 0.072 | 0.80 |
| 7. Corpus adiposum | 0.37 ± 0.65 | 0.24 ± 0.90 | 0.650 | 1.140 | 0.29 |
| 8. Vesica fellea | −0.39 ± 0.51 | −0.49 ± 0.34 | 0.154 | 0.780 | 0.38 |
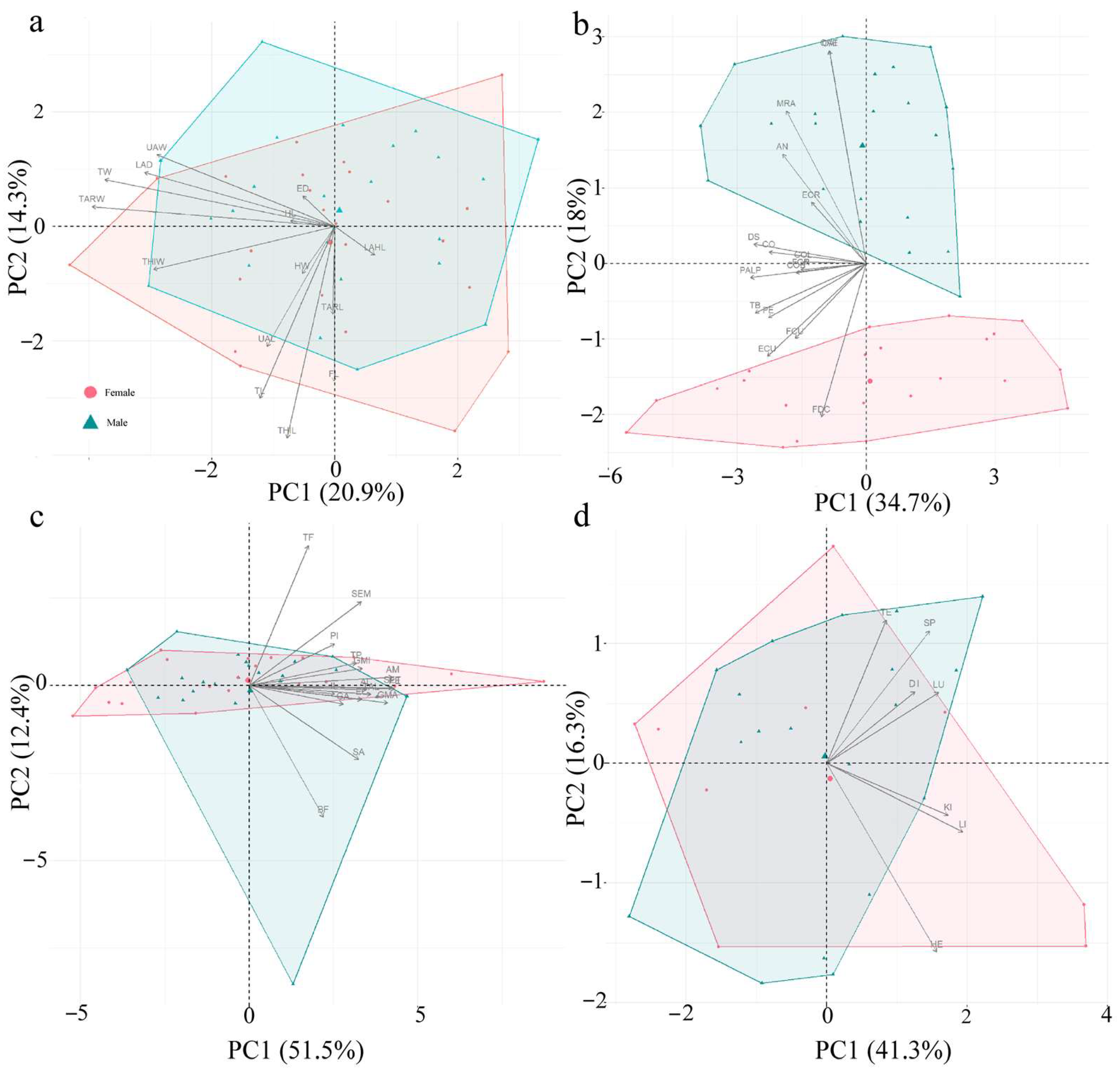
3.2. Results of Principal Component Analysis (Table 5; Figure 3)
| External Morphological Characteristics | PC1 | PC2 | PC3 | PC4 | PC5 | PC6 | Forelimb, Chest, and Abdominal Muscles | PC1 | PC2 | PC3 | PC4 | Hindlimb Muscles | PC1 | PC2 | PC3 | Internal Organs | PC1 | PC2 | PC3 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1. HL | 0.16 | −0.02 | 0.38 | 0.61 | 0.05 | −0.34 | 1. COL | 0.46 | −0.02 | 0.69 | −0.28 | 1.BF | 0.47 | −0.81 | 0.09 | 1.HE | 0.66 | −0.67 | −0.01 |
| 2. HW | 0.12 | 0.18 | −0.43 | 0.70 | −0.22 | −0.06 | 2. COB | 0.50 | −0.09 | −0.52 | −0.12 | 2.TF | 0.38 | 0.86 | 0.04 | 2.LI | 0.82 | −0.24 | 0.27 |
| 3. UAL | 0.24 | 0.46 | 0.37 | −0.27 | 0.17 | −0.52 | 3. DS | 0.81 | 0.06 | 0.31 | −0.16 | 3.AL | 0.74 | −0.03 | −0.07 | 3.SP | 0.62 | 0.48 | −0.07 |
| 4. UAW | 0.63 | −0.27 | 0.15 | 0.27 | 0.31 | 0.25 | 4. PE | 0.67 | −0.27 | −0.19 | −0.20 | 4.SA | 0.69 | −0.46 | −0.05 | 4.LU | 0.67 | 0.25 | −0.49 |
| 5. LAHL | −0.14 | 0.11 | 0.61 | 0.21 | 0.39 | 0.29 | 5. PALP | 0.82 | −0.13 | −0.01 | −0.07 | 5.AM | 0.91 | 0.05 | 0.03 | 5.KI | 0.73 | −0.18 | 0.18 |
| 6. LAD | 0.68 | −0.21 | 0.05 | −0.05 | 0.18 | −0.22 | 6. CO | 0.69 | 0.01 | −0.26 | 0.45 | 6.GMA | 0.88 | −0.11 | 0.23 | 6.DI | 0.36 | 0.51 | 0.72 |
| 7. THIL | 0.17 | 0.81 | −0.08 | −0.12 | 0.06 | 0.29 | 7. FCR | 0.47 | −0.08 | −0.38 | 0.20 | 7.GMI | 0.72 | 0.11 | −0.01 | 7.COAD | 0.53 | 0.25 | −0.45 |
| 8. THIW | 0.65 | 0.17 | −0.45 | 0.04 | −0.19 | 0.07 | 8. FCU | 0.49 | −0.36 | −0.27 | −0.36 | 8.SEM | 0.71 | 0.51 | 0.26 | ||||
| 9. TL | 0.27 | 0.66 | 0.17 | −0.31 | −0.17 | 0.14 | 9. FDC | 0.27 | −0.65 | 0.20 | 0.18 | 9.SET | 0.91 | −0.01 | −0.02 | ||||
| 10. TW | 0.82 | −0.18 | 0.00 | −0.11 | −0.16 | 0.00 | 10. AN | 0.64 | 0.39 | −0.20 | 0.21 | 10.PI | 0.54 | 0.26 | −0.53 | ||||
| 11. TARL | 0.01 | 0.34 | −0.41 | 0.22 | 0.65 | 0.21 | 11. ECR | 0.43 | 0.20 | 0.49 | 0.55 | 11.IL | 0.54 | −0.05 | 0.68 | ||||
| 12. TARW | 0.86 | −0.08 | 0.09 | −0.09 | 0.05 | 0.02 | 12. ECU | 0.68 | −0.47 | 0.01 | 0.19 | 12.TAL | 0.93 | −0.02 | −0.06 | ||||
| 13. FL | 0.00 | 0.59 | 0.21 | 0.44 | −0.28 | −0.19 | 13. MRA | 0.62 | 0.57 | 0.08 | −0.24 | 13.GA | 0.59 | −0.12 | −0.09 | ||||
| 14. ED | 0.11 | −0.12 | 0.45 | 0.15 | −0.44 | 0.54 | 14. OAE | 0.34 | 0.85 | −0.09 | 0.07 | 14.PE | 0.77 | −0.05 | −0.16 | ||||
| 15. OAI | 0.34 | 0.85 | 0.02 | −0.16 | 15.EC | 0.72 | −0.09 | −0.45 | |||||||||||
| 16. TB | 0.78 | −0.28 | 0.14 | −0.11 | 16.TP | 0.68 | 0.14 | 0.15 | |||||||||||
| Explanatory rate | 20.87 | 14.33 | 10.88 | 10.36 | 8.26 | 7.59 | 34.63 | 18.09 | 9.51 | 6.59 | 51.51 | 12.45 | 7.11 | 41.29 | 16.28 | 15.23 |
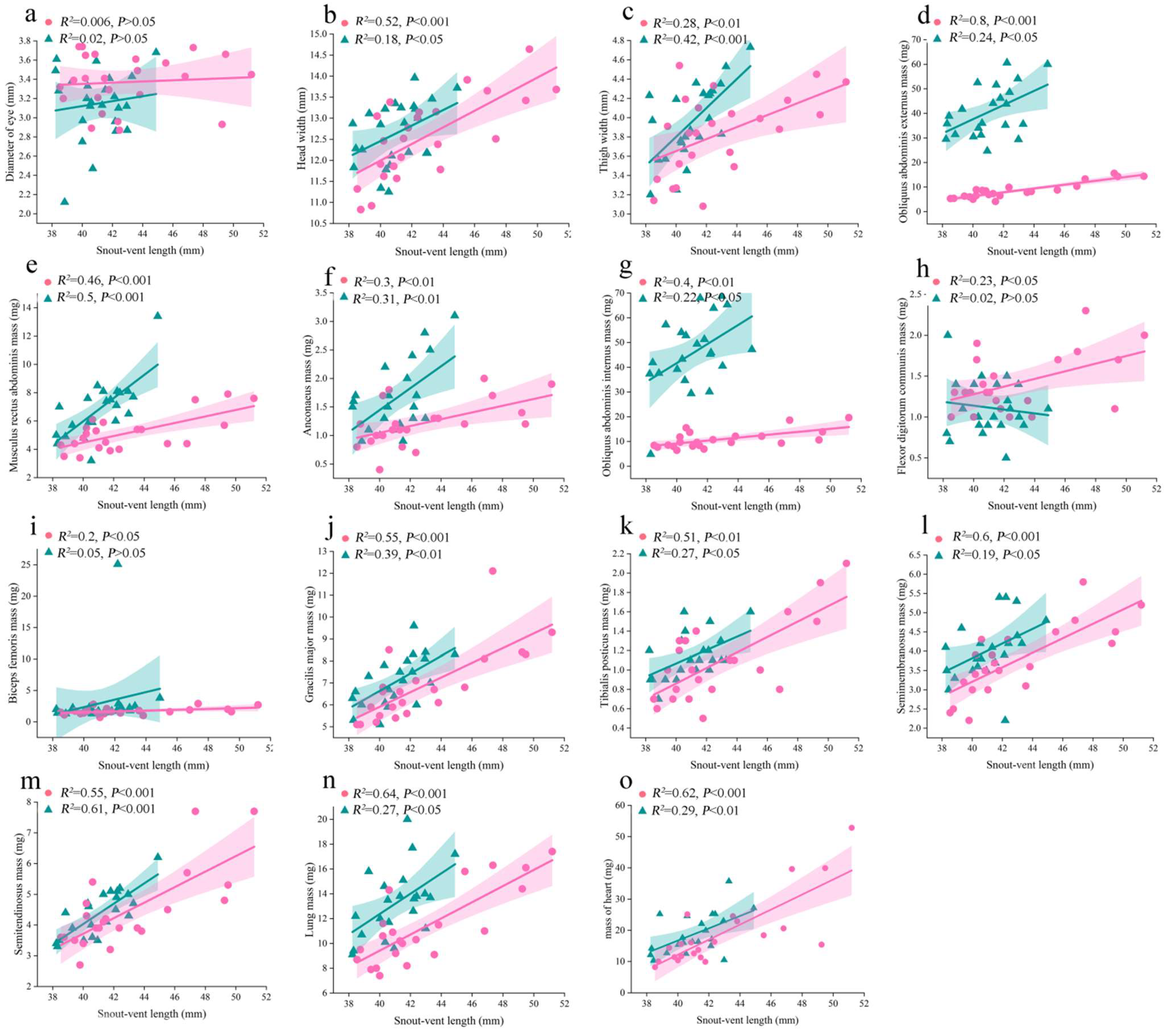
3.3. Results of Allometric Analysis for Significantly Sexual Different Traits (Figure 4)
4. Discussion
4.1. Sexual Dimorphism in the External Morphological Characteristics
4.2. Sexual Dimorphism in the Appendicular Muscle Mass
4.3. Sexual Differences in the Internal Organs
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cordeschi, G.; Canestrelli, D.; Porretta, D. Sex-biased phenotypic plasticity affects sexual dimorphism patterns under changing environmental conditions. Sci. Rep. 2024, 14, 892. [Google Scholar] [CrossRef] [PubMed]
- Dursun, C.; Gül, S.; Özdemir, N. Sexual size and shape dimorphism in Turkish common toads (Bufo bufo Linnaeus 1758). Anat. Rec. 2022, 305, 1548–1558. [Google Scholar] [CrossRef] [PubMed]
- Juarez, B.H.; Moen, D.S.; Adams, D.C. Ecology, sexual dimorphism, and jumping evolution in anurans. J. Evol. Biol. 2023, 36, 829–841. [Google Scholar] [CrossRef]
- Head, A.; Vaughn, P.L.; Livingston, E.H.; Colwell, C.; Muñoz, M.M.; Gangloff, E.J. Include the females: Morphology–performance relationships vary between sexes in lizards. J. Exp. Biol. 2024, 227, jeb248014. [Google Scholar] [CrossRef]
- Fairbairn, D.J.; Blanckenhorn, W.U.; Székely, T. Sex, Size and Gender Roles: Evolutionary Studies of Sexual Size Dimorphism; Oxford University Press: Oxford, UK, 2007. [Google Scholar]
- Kelley, D.B. Sexually dimorphic behaviors. Annu. Rev. Neurosci. 1988, 11, 225–251. [Google Scholar] [CrossRef]
- Andersson, M. Sexual Selection; Princeton University Press: Princeton, NJ, USA, 1994; Volume 72. [Google Scholar]
- Hosken, D.J.; House, C.M. Sexual selection. Curr. Biol. 2011, 21, R62–R65. [Google Scholar] [CrossRef]
- Shuker, D.M.; Kvarnemo, C. The definition of sexual selection. Behav. Ecol. 2021, 32, 781–794. [Google Scholar] [CrossRef]
- Pincheira-Donoso, D.; Hunt, J. Fecundity selection theory: Concepts and evidence. Biol. Rev. 2017, 92, 341–356. [Google Scholar] [CrossRef]
- De Lisle, S. Understanding the evolution of ecological sex differences: Integrating character displacement and the Darwin-Bateman paradigm. Evol. Lett. 2019, 3, 434–447. [Google Scholar] [CrossRef]
- Priambodo, B.; Hamidy, A.; Kurniawan, N. Morphological characters review on white-lipped frog (Chalcorana chalconota; Schelgel, 1837) based on morphometrical analysis, within the population of Java. In IOP Conference Series: Earth and Environmental Science; IOP Publishing: Bristol, UK, 2021; p. 012037. [Google Scholar]
- Englmaier, G.K.; Antonov, A.; Weiss, S.J. General patterns of sexual dimorphism in graylings (Thymallus), with a comparison to other salmonid species. Rev. Fish. Biol. Fish. 2022, 32, 645–667. [Google Scholar] [CrossRef]
- Shine, R. Sexual selection and sexual dimorphism in the Amphibia. Copeia 1979, 1979, 297–306. [Google Scholar] [CrossRef]
- Xu, M.; Wang, G.; Liu, P.; He, Z.; He, K.; Cheng, Z.; Wang, Z.; Chen, W.; Li, Z.; Zhang, L. Age structure and body size of two Tibetan toad (Bufo tibetanus) populations from different elevations in China. Ecol. Evol. 2024, 14, e11559. [Google Scholar] [CrossRef]
- Monnet, J.M.; Cherry, M.I. Sexual size dimorphism in anurans. Proc. R. Soc. Lond. Ser. B 2002, 269, 2301–2307. [Google Scholar] [CrossRef] [PubMed]
- Silva, N.R.; Berneck, B.V.; da Silva, H.R.; Haddad, C.F.; Zamudio, K.R.; Mott, T.; Nali, R.C.; Prado, C.P. Egg-laying site, fecundity and degree of sexual size dimorphism in frogs. Biol. J. Linn. Soc. 2020, 131, 600–610. [Google Scholar] [CrossRef]
- Zhang, L.; Yuan, X.; Sheng, Y.; Yu, F.; Zhong, X.; Chen, X.; Rao, D. Strong limb tactics of the Boulenger’s Lazy Toad, Scutiger boulengeri: Inferred from limb muscles. Asian Herpetol. Res. 2020, 11, 360–364. [Google Scholar] [CrossRef]
- Yekta, N.; Blackburn, D.G. Sexual dimorphism in mass and protein content of the forelimb muscles of the northern leopard frog, Rana pipiens. Can. J. Zool. 1992, 70, 670–674. [Google Scholar] [CrossRef]
- Chen, W.; Wu, Q.G.; Su, Z.X.; Lu, X. Age, body size and clutch size of Rana kunyuensis, a subtropical frog native to China. Herpetol. J. 2012, 22, 203–206. [Google Scholar]
- Liao, W.B.; Wu, Q.G.; Barrett, K. Evolution of sexual dimorphism in the forelimb muscles of Andrew’s toad (Bufo andrewsi) in response to putative sexual selection. Anim. Biol. 2012, 62, 83–93. [Google Scholar] [CrossRef]
- Lambert, M.R.; Stoler, A.B.; Smylie, M.S.; Relyea, R.A.; Skelly, D.K. Interactive effects of road salt and leaf litter on wood frog sex ratios and sexual size dimorphism. Can. J. Fish. Aquat. Sci. 2017, 74, 141–146. [Google Scholar] [CrossRef]
- Zhang, L.; Sheng, Y.; Yuan, X.; Yu, F.; Zhong, X.; Chen, X. Sexual dimorphism in Scutiger boulengeri, an endemic toad from the Tibetan Plateau. Anim. Biol. 2020, 70, 445–457. [Google Scholar] [CrossRef]
- Caldart, V.M.; Loebens, L.; Brum, A.J.C.; Bataioli, L.; Cechin, S.Z. Reproductive cycle, size and age at sexual maturity, and sexual dimorphism in the stream-breeding frog Crossodactylus schmidti (Hylodidae). S. Am. J. Herpetol. 2019, 14, 1–11. [Google Scholar] [CrossRef]
- Pacheco, E.O.; Ceron, K.; Akieda, P.S.; Santana, D.J. Diet and morphometry of two poison frog species (Anura, Dendrobatidae) from the plateaus surrounding the Pantanal of Mato Grosso do Sul state, Brazil. Stud. Neotrop. Fauna. Environ. 2021, 56, 99–107. [Google Scholar] [CrossRef]
- Araspin, L.; Measey, J.; Herrel, A. Does aquatic performance predict terrestrial performance: A case study with an aquatic frog, Xenopus laevis. J. Exp. Biol. 2023, 226, jeb246545. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Pelagio, K.P.; Gutiérrez, B.M.; Hernández, D.P.; Calva, N.P.; Segura-Alegria, B.; Ochoa-Ochoa, L.M. Locomotion and biomechanical adaptations in hind limb muscles of three Mexican anuran species. S. Am. J. Herpetol. 2023, 26, 37–43. [Google Scholar] [CrossRef]
- Casselman, S.; Schulte-Hostedde, A. Reproductive roles predict sexual dimorphism in internal and external morphology of lake whitefish, Coregonus clupeaformis. Ecol. Freshw. Fish. 2004, 13, 217–222. [Google Scholar] [CrossRef]
- Deng, W.; Jin, L.; Qiu, D.; Yan, C.; Liao, W. Geographic variation in organ size in a toad (Duttaphrynus melanostictus). Animals 2023, 13, 2645. [Google Scholar] [CrossRef]
- Gou, J.; Xiong, J.; Dong, B.; Liu, Z.; Hou, M. Sexual dimorphism of internal organ mass in Bufo gargarizans gargarizans from summer population. Liver 2017, 3, 1543–1546. [Google Scholar] [CrossRef]
- Roco, Á.S.; Ruiz-García, A.; Bullejos, M. Testis development and differentiation in amphibians. Genes 2021, 12, 578. [Google Scholar] [CrossRef]
- Fanali, L.Z. Alterações Morfofuncionais nos Melanomacrófagos Hepáticos de Peixes e Anfíbios Induzidas pelo Contaminante Benzo [a] Pireno. Ph.D. Thesis, Universidade Estadual Paulista, São Paulo, Brazil, 2020. [Google Scholar]
- Soares, E.T.T. Comparative Histological and Anatomical Biomarkers of the Spleen and Kidneys in Amphibians: A Special Focus on Rhinella icterica. Master’s Thesis, Universidade Federal de São Carlos, Sorocaba, Brazil, 2023. [Google Scholar]
- Barbour, T. Some New Reptiles and Amphibians. Bull. Mus. Comp. Zool. 1908, 51, 315–325. [Google Scholar]
- Ko, S.B.; Ko, Y.M.; Lee, J.H. Ecology, Body size and age structure of mating couples in boreal digging frog (Kaloula borealis). Korean J. Environ. 2014, 28, 281–286. [Google Scholar] [CrossRef]
- Zhang, M.H.; Chen, X.H.; Ye, C.Y.; Fei, L.; Li, P.P.; Jiang, J.P.; Wang, B. Osteology of the Asian narrow-mouth toad Kaloula borealis (Amphibia, Anura, Microhylidae) with comments on its osteological adaptation to fossorial life. Acta. Zool. 2020, 101, 366–383. [Google Scholar] [CrossRef]
- An, C.K.; Hong, S.G.; Na, S.; Doh, J.; Oh, K.C.; Yi, H. Comparison Study of the Snout-vent Length (SVL) and the Biomass for the Climate Change Sensitive Species, Narrow-mouthed Toads (Kaloula borealis, Endangered Species II), at the three different areas (Seoul, Nonsan, Busan) of South Korea. J. Wetlands Res. Vol. 2020, 22, 8–14. [Google Scholar] [CrossRef]
- Othman, S.N.; Putri, E.T.; Messenger, K.R.; Bae, Y.; Yang, Y.; Bova, T.; Reed, T.; Amin, H.; Chuang, M.F.; Jang, Y. Impact of the Miocene orogenesis on Kaloula spp. radiation and implication of local refugia on genetic diversification. Integr. Zool. 2022, 17, 261–284. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.M.; Lee, Y.Y.; Park, S.M.; Ham, C.H.; Sung, H.C. Confirmation of the applicability of skeletochronology and estimating the age Structure of Kaloula borealis (Microhylidae: Anura) at Lake Sihwa, South Korea. Biology 2022, 11, 898. [Google Scholar] [CrossRef]
- Choi, M.H.; Bae, Y.S. Monitoring the Alternative Habitat of Kaloula Borealis and Analysis on Meteorological Factors in Pyeongtaek. J. Life Sci. 2024, 34, 264–270. [Google Scholar] [CrossRef]
- Fei, L.; Hu, S.Q.; Ye, C.Y.; Huang, Y.Z. Amphibia, Anura Ranidae. In Fauna Sinica; Science Press: Beijing, China, 2009; Volume 3. [Google Scholar]
- Lanzetti, A.; Ekdale, E.G. Enhancing CT imaging: A safe protocol to stain and de-stain rare fetal museum specimens using diffusible iodine-based staining (diceCT). J. Anat. 2021, 239, 228–241. [Google Scholar] [CrossRef]
- Leavey, A.; Richards, C.T.; Porro, L.B. Comparative muscle anatomy of the anuran pelvis and hindlimb in relation to locomotor mode. J. Anat. 2024, 245, 751–774. [Google Scholar] [CrossRef]
- Meiri, S. Length–weight allometries in lizards. J. Zool. 2010, 281, 218–226. [Google Scholar] [CrossRef]
- Vukov, T.; Kuručki, M.; Anđelković, M.; Tomašević-Kolarov, N. Post-metamorphic ontogenetic changes in head size and shape of the pool frog (Pelophylax lessonae, Ranidae). Arch. Biol. Sci. 2018, 70, 307–312. [Google Scholar] [CrossRef]
- Thomas, K.N.; Gower, D.J.; Bell, R.C.; Fujita, M.K.; Schott, R.K.; Streicher, J.W. Eye size and investment in frogs and toads correlate with adult habitat, activity pattern and breeding ecology. P. Roy. Soc. B-Biol. Sci. 2020, 287, 20201393. [Google Scholar] [CrossRef]
- Fabrezi, M.; Goldberg, J.; Chuliver Pereyra, M. Morphological variation in anuran limbs: Constraints and novelties. J. Exp. Zool. Part B 2017, 328, 546–574. [Google Scholar] [CrossRef]
- Watters, J.L.; Cummings, S.T.; Flanagan, R.L.; Siler, C.D. Review of morphometric measurements used in anuran species descriptions and recommendations for a standardized approach. Zootaxa 2016, 4072, 477–495. [Google Scholar] [CrossRef]
- Liao, W.B.; Liao, Y.M.; Xiao, W.M.; Chen, W.; Mi, Z.P.; Li, C. Sexual dimorphism in hind limb muscle mass of the Andrew’s Toad (Bufo andrewsi) in relation to sexual selection. North-West J. Zool. 2012, 8, 252–256. [Google Scholar]
- Duellman, W.E.; Trueb, L. Biology of Amphibians; JHU Press: Baltimore, MD, USA, 1994. [Google Scholar]
- Myatt, J.P.; Crompton, R.H.; Thorpe, S.K. Hindlimb muscle architecture in non-human great apes and a comparison of methods for analysing inter-species variation. J. Anat. 2011, 219, 150–166. [Google Scholar] [CrossRef]
- Zhou, B.X. Frog Anatomy; Science Press: Beijing, China, 1956. [Google Scholar]
- Abdala, V.; Diogo, R. Comparative anatomy, homologies and evolution of the pectoral and forelimb musculature of tetrapods with special attention to extant limbed amphibians and reptiles. J. Anat. 2010, 217, 536–573. [Google Scholar] [CrossRef] [PubMed]
- Manzano, A.S.; Abdala, V.; Herrel, A. Morphology and function of the forelimb in arboreal frogs: Specializations for grasping ability? J. Anat. 2008, 213, 296–307. [Google Scholar] [CrossRef] [PubMed]
- Böhmer, C.; Fabre, A.C.; Taverne, M.; Herbin, M.; Peigné, S.; Herrel, A. Functional relationship between myology and ecology in carnivores: Do forelimb muscles reflect adaptations to prehension? Biol. J. Linn. Soc. 2019, 127, 661–680. [Google Scholar] [CrossRef]
- Taylor-Burt, K.R.; Biewener, A.A. Aquatic and terrestrial takeoffs require different hindlimb kinematics and muscle function in mallard ducks. J. Exp. Biol. 2020, 223, jeb223743. [Google Scholar] [CrossRef]
- Chin, B.Z.; Wee, I.J.Y.; Syn, N.L.X.; Krishna, L. Arthroscopic anterior cruciate ligament reconstruction: A meta-analysis comparing semitendinosus alone and semitendinosus with gracilis tendon autografts. J. Knee Surg. 2019, 32, 796–803. [Google Scholar] [CrossRef]
- Přikryl, T.; Aerts, P.; Havelková, P.; Herrel, A.; Roček, Z. Pelvic and thigh musculature in frogs (Anura) and origin of anuran jumping locomotion. J. Anat. 2009, 214, 100–139. [Google Scholar] [CrossRef]
- Padilla, P.; Courant, J.; Herrel, A. Allocation trade-offs impact organ size and muscle architecture in an invasive population of Xenopus laevis in Western France. J. Anat. 2019, 235, 1057–1064. [Google Scholar] [CrossRef]
- Feng, X.Y. Systematic Anatomy of Bufo Gargarizans; Higher Education Press: Beijing, China, 1990; Volume 3, pp. 12–58. [Google Scholar]
- Bell, M.B. Sex and age influence responses to changes in the cost of cooperative care in a social carnivore. Behav. Ecol. 2010, 21, 1118–1123. [Google Scholar] [CrossRef]
- Zamaletdinov, R.; Svinin, A.; Fayzulin, A.; Ermakov, O.; Mikhaylova, R.; Litvinchuk, S. Age Structure of Water Frogs of the Genus Pelophylax in the Middle Volga River Region (European Russia). Animals 2025, 15, 1273. [Google Scholar] [CrossRef] [PubMed]
- Rahman, A.; Muktadir, M.G. SPSS: An imperative quantitative data analysis tool for social science research. Int. J. Res. Innov. Soc. Sci. 2021, 5, 300–302. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2024; Available online: https://www.R-project.org/ (accessed on 19 March 2025).
- Wickham, H.; Chang, W. ggplot2: Create elegant data visualisations using the grammar of graphics. Version 2.1.0. Available online: https://cran.r-project.org/package=ggplot2 (accessed on 19 March 2025).
- Kim, E.B.; Kim, E.S.; Sung, H.C.; Lee, D.H.; Kim, G.J.; Nam, D.H. Comparison of the skeletal features of two sympatric tree frogs (Hylidae: Hyla)—Hyla japonica and Hyla suweonensis—Using three-dimensional micro-computed tomography. J. Asia Pac. Biodivers. 2021, 14, 147–153. [Google Scholar] [CrossRef]
- Liao, W.B.; Zeng, Y.; Zhou, C.Q.; Jehle, R. Sexual size dimorphism in anurans fails to obey Rensch’s rule. Front Zool. 2013, 10, 10. [Google Scholar] [CrossRef]
- Saulnier Masson, R.; Daoues, K.; Measey, J.; Herrel, A. The evolution of bite force and head morphology in scincid lizards: Diet and habitat use as possible drivers. Biol. J. Linn. Soc. 2023, 140, 58–73. [Google Scholar] [CrossRef]
- Xuereb, S. Evaluating Head Shape Morphology and Somatic Growth Rate Variation in Komodo dragons over Ontogeny; Deakin University: Geelong, VIC, Australia, 2021. [Google Scholar]
- Liang, T.; Wang, L.; Shi, L. Sexual and natural selection interplay in sexual head shape dimorphism of two sympatric racerunners (Squamata: Lacertidae). Front Ecol. Evol. 2022, 10, 1016885. [Google Scholar] [CrossRef]
- de Sousa Machado, H.T.; Ferreira da Silva, C.; Alves Benício, R.; Waldemar Ávila, R. Feeding ecology, reproductive biology, and sexual dimorphism of Boana raniceps (Anura: Hylidae) in an area of Caatinga, northeastern Brazil. Caldasia 2024, 46, 71–80. [Google Scholar] [CrossRef]
- Lappin, A.K.; Wilcox, S.C.; Moriarty, D.J.; Stoeppler, S.A.; Evans, S.E.; Jones, M.E. Bite force in the horned frog (Ceratophrys cranwelli) with implications for extinct giant frogs. Sci. Rep. 2017, 7, 759. [Google Scholar] [CrossRef]
- Butler, M.A.; Sawyer, S.A.; Losos, J.B. Sexual dimorphism and adaptive radiation in Anolis lizards. Nature 2007, 447, 202–205. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.H.; Zhong, M.J.; Liao, W.B.; Kotrschal, A. Investigating the role of body size, ecology, and behavior in anuran eye size evolution. Evol. Ecol. 2019, 33, 585–598. [Google Scholar] [CrossRef]
- Moo, E.K.; Peterson, D.R.; Leonard, T.R.; Kaya, M.; Herzog, W. In vivo muscle force and muscle power during near-maximal frog jumps. PLoS ONE 2017, 12, e0173415. [Google Scholar] [CrossRef] [PubMed]
- O’Reilly, J.C.; Summers, A.P.; Ritter, D.A. The evolution of the functional role of trunk muscles during locomotion in adult amphibians. Am. Zool. 2000, 40, 123–135. [Google Scholar] [CrossRef]
- Ma, H.; Jin, L.; Zhao, L.; Yan, C.; Mi, Z. Genetic and metabolic insights into sexual dimorphism in the flexor carpi radialis of Asiatic toads (Bufo gargarizans) associated with amplexus behavior. BMC Genom. 2025, 26, 192. [Google Scholar] [CrossRef]
- Navas, C.A.; James, R.S. Sexual dimorphism of extensor carpi radialis muscle size, isometric force, relaxation rate and stamina during the breeding season of the frog Rana temporaria Linnaeus 1758. J. Exp. Biol. 2007, 210, 715–721. [Google Scholar] [CrossRef]
- de Souza Junior, P.; de Souza Pahim, A.B.; Viotto-Souza, W.; Pellenz, J.; Bernardes, F.C.S.; Abidu-Figueiredo, M.; Santos, A.L.Q. Evolutionary history or function? Which preponderates in the expression of the muscle mass of the thoracic limb in wild carnivorans? Anat. Rec. 2021, 304, 1344–1356. [Google Scholar] [CrossRef]
- Yang, Y.J.; Jiang, Y.; Mi, Z.P.; Liao, W.B. Testing the role of environmental harshness and sexual selection in limb muscle mass in anurans. Front Ecol. Evol. 2022, 10, 879885. [Google Scholar] [CrossRef]
- Leavey, A.; Ruta, M.; Richards, C.T.; Porro, L.B. Locomotor, ecological and phylogenetic drivers of skeletal proportions in frogs. J. Anat. 2023, 243, 404–420. [Google Scholar] [CrossRef]
- Hasan, O.; Houlihan, M.; Yang, D.; Kohler, T. Testosterone as a Performance Enhancer. In Controversies in Testosterone Deficiency; Springer: Cham, Switzerland, 2021; pp. 123–138. [Google Scholar]
- Betancourth-Cundar, M.; Canoine, V.; Fusani, L.; Cadena, C.D. Does testosterone underly the interplay between male traits and territorial behavior in neotropical poison frogs? Horm. Behav. 2024, 162, 105547. [Google Scholar] [CrossRef]
- Patey, C.P.; Driedzic, W.R. Cold acclimation increases activities of mitochondrial long-chain acyl-CoA synthetase and carnitine acyl-CoA transferase I in heart of rainbow trout (Oncorhynchus mykiss). Can j. Zool. 1997, 75, 324–331. [Google Scholar] [CrossRef]
- Finkler, M.S.; Hayes, C.J.; Rifai, L. Sexual dimorphisms in metabolism, organ mass, and reproductive energetics in pre-breeding American toads (Anaxyrus americanus). Copeia 2014, 2014, 447–453. [Google Scholar] [CrossRef]
- Cotes, J.E.; Chinn, D.J.; Miller, M.R. Lung function: Physiology, Measurement and Application in Medicine; John Wiley and Sons: Hoboken, NJ, USA, 2009. [Google Scholar]
- Kooijman, S.A.L.M. Dynamic Energy Budget Theory for Metabolic Organisation; Cambridge University Press: Cambridge, UK, 2010. [Google Scholar]
- Crane, H.R.; Rollins, L.A.; Shine, R.; Brown, G. Effect of parasite infection and invasion history on feeding, growth, and energy allocation of cane toads. Biol. J. Linn. Soc. 2025, 144, blae054. [Google Scholar] [CrossRef]
- Li, H.; Chen, S.; Jiang, J.; He, B.; Zhang, M. Exploring sexual differences in external morphology and limb muscles of Hylarana guentheri (Anura: Ranidae) during non-breeding season. Acta Zool. 2023, 104, 647–656. [Google Scholar] [CrossRef]
- Zhelev, Z.; Mollov, I.; Tsonev, S. Application of fluctuating asymmetry values in Pelophylax ridibundus (Amphibia: Anura: Ranidae) meristic traits as a method for assessing environmental quality of areas with different degrees of urbanization. Divers. Distrib. 2023, 15, 118. [Google Scholar] [CrossRef]
- Salla, R.F.; Jones-Costa, M.; Abdalla, F.C.; Vidal, F.A.; Boeing, G.A.; Oliveira, C.R.; Silva-Zacarin, E.C.; Franco-Belussi, L.; Rizzi-Possignolo, G.M.; Lambertini, C. Differential liver histopathological responses to amphibian chytrid infection. Dis. Aquat. Organ. 2020, 142, 177–187. [Google Scholar] [CrossRef]

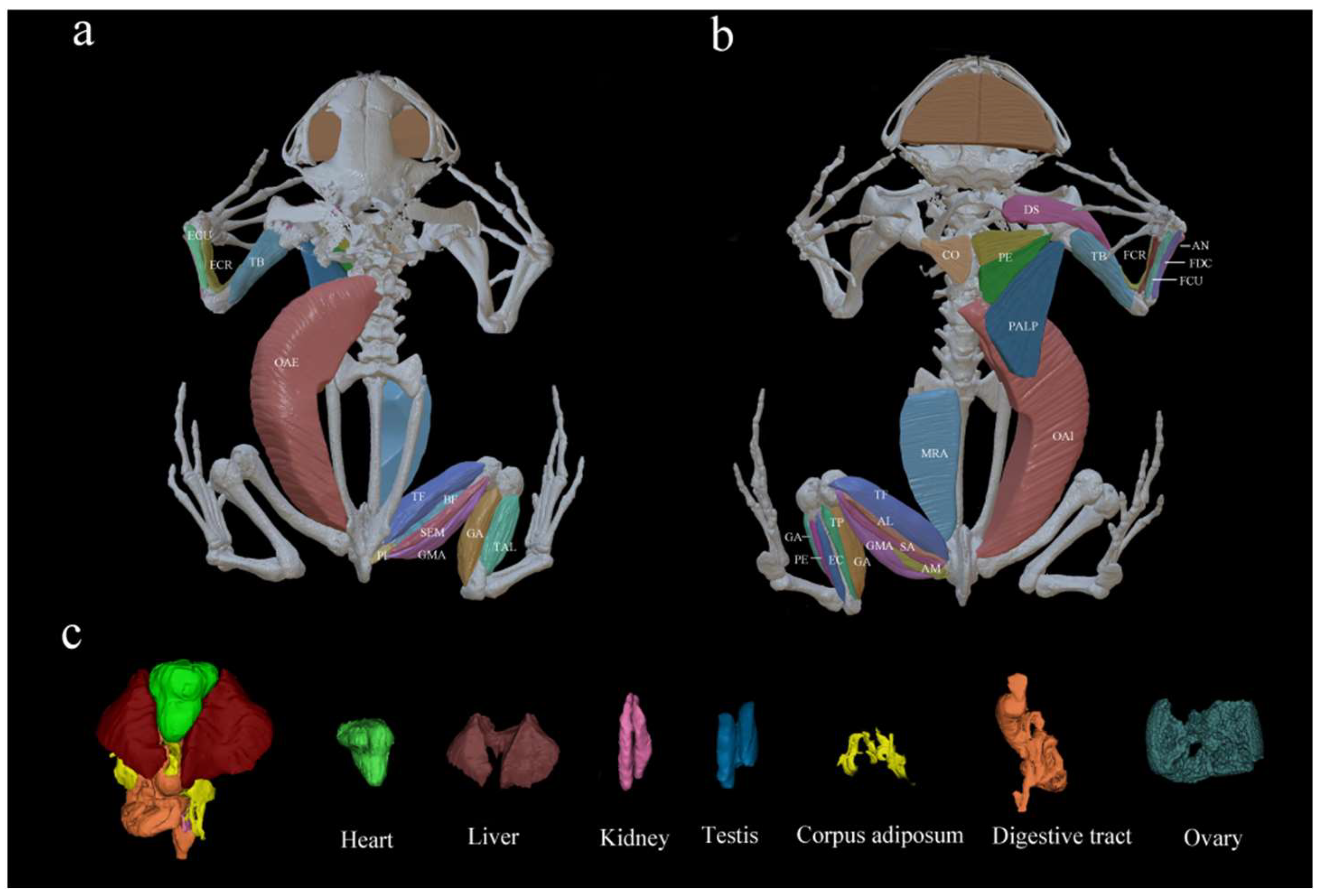
| Functional Groups | Muscles | References |
|---|---|---|
| 1. Pectoral girdle flexors | Deltoideus scapularis (DS); coracobrachialis longus (COL); coracobrachialis brevis (COB) | Duellman and Trueb [50]; Myatt, Crompton, and Thorpe [51]. |
| 2. Upper arm flexor | Pectoralis (PE); pectoralis abdominis, lateral portion (PALP); | Zhou [52]. |
| 3. Forearm flexor | Coracoradialis (CO) | Abdala and Diogo [53]. |
| 4. Carpus flexors | Flexor carpi ulnaris (FCU); flexor carpi radialis (FCR); flexor digitorum communis (FDC) | Manzano, Abdala, and Herrel [54]; Böhmer et al. [55]. |
| 5. Elbow extensors | Anconaeus (AN); triceps brachii (TB) | Böhmer et al. [55]. |
| 6. Carpus extensors | Extensor carpi ulnaris (ECU); extensor carpi radialis (ECR) | Böhmer et al. [55]. |
| 7. Abdominal muscle | Musculus rectus abdominis (MRA); obliquus abdominis internus (OAI), obliquus abdominis externus (OAE) | Zhou [52]. |
| 8. Knee flexors | Biceps femoris (BF); semimembranosus (SEM); sartorius (SA); gastrocnemius (GA) | Zhou [52]; Taylor-Burt and Biewener [56]. |
| 9. Knee flexors and internal tibial rotators | Gracilis major (GMA); gracilis minor (GMI); semitendinosus (SET) | Chin et al. [57]. |
| 10. Hip adductors | Adductor magnus (AM); adductor longus (AL) | Přikryl et al. [58]. |
| 11. Knee extensor | Triceps femoris (TF) | Padilla, Courant, and Herrel [59]. |
| 12. Long extensors | Peroneus (PE); tibialis anticus longus (TAL); tibialis posticus (TP); extensor cruris (EC) | Padilla, Courant, and Herrel [59]. |
| 13. Others | Piriformis (PI); iliopsoas (IL) | Zhou [52]. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, X.; Zhang, M.; Zhang, W.; Jiang, J.; Dong, B. Uncovering Sexual Differences in the External Morphology, Appendicular Muscles, and Internal Organs of a Fossorial Narrow-Mouth Frog (Kaloula borealis). Animals 2025, 15, 2118. https://doi.org/10.3390/ani15142118
Wang X, Zhang M, Zhang W, Jiang J, Dong B. Uncovering Sexual Differences in the External Morphology, Appendicular Muscles, and Internal Organs of a Fossorial Narrow-Mouth Frog (Kaloula borealis). Animals. 2025; 15(14):2118. https://doi.org/10.3390/ani15142118
Chicago/Turabian StyleWang, Xiuping, Meihua Zhang, Wenyi Zhang, Jianping Jiang, and Bingjun Dong. 2025. "Uncovering Sexual Differences in the External Morphology, Appendicular Muscles, and Internal Organs of a Fossorial Narrow-Mouth Frog (Kaloula borealis)" Animals 15, no. 14: 2118. https://doi.org/10.3390/ani15142118
APA StyleWang, X., Zhang, M., Zhang, W., Jiang, J., & Dong, B. (2025). Uncovering Sexual Differences in the External Morphology, Appendicular Muscles, and Internal Organs of a Fossorial Narrow-Mouth Frog (Kaloula borealis). Animals, 15(14), 2118. https://doi.org/10.3390/ani15142118






