The Global Epidemiology of Bovine Leukemia Virus: Current Trends and Future Implications
Abstract
Simple Summary
Abstract
1. Introduction
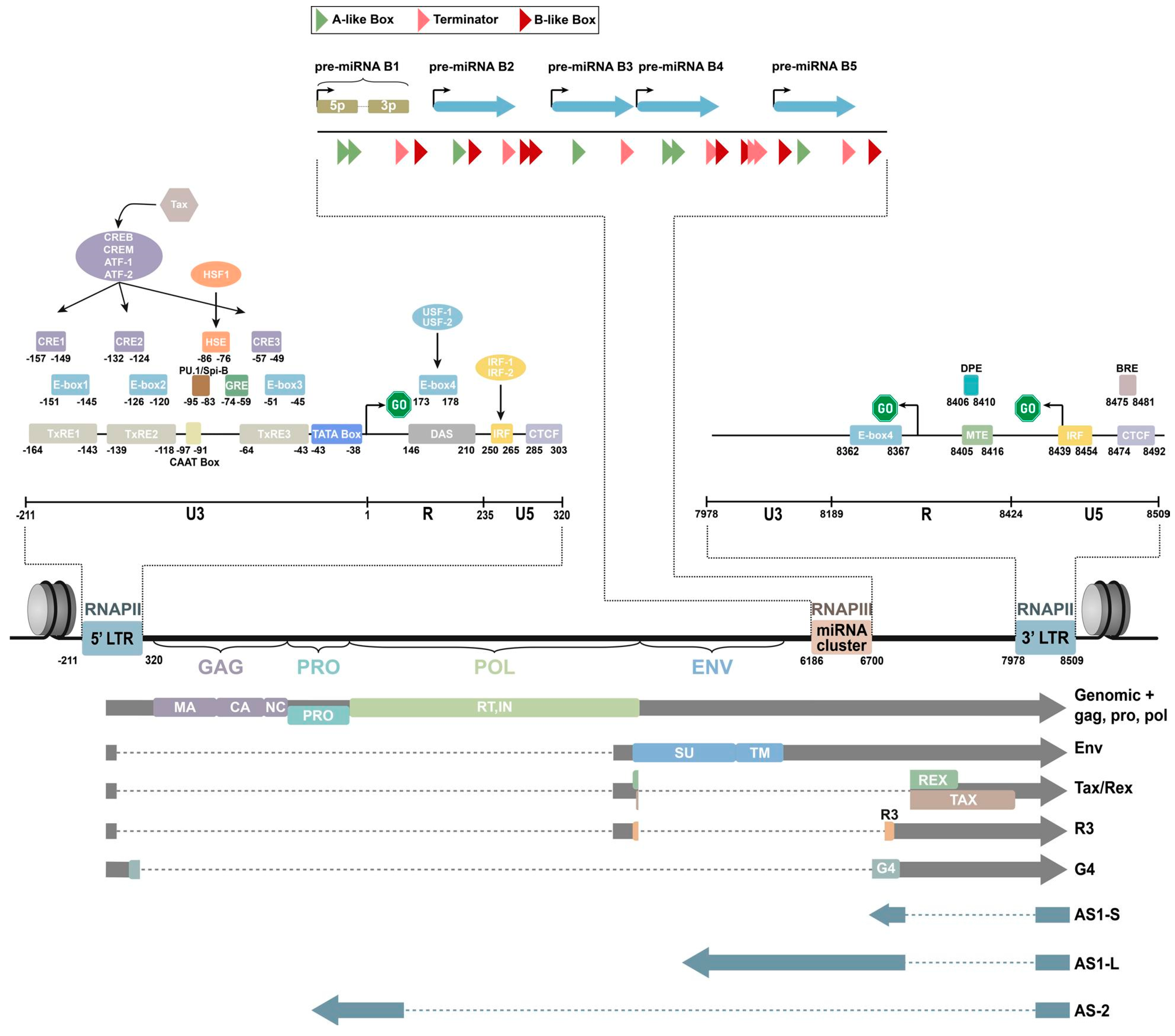
2. Genomic Composition of BLV
3. Current Epidemiological Distribution of BLV
4. Susceptibility and Transmission Pathways of BLV
5. Clinical Symptoms of EBL
6. Detection Methods of BLV
6.1. Serological Techniques Used for the Diagnosis of BLV
6.2. Molecular Techniques Used for the Diagnosis of BLV
| Test Assay | Sample Type | Advantages | Disadvantages | References |
|---|---|---|---|---|
| Conventional PCR (single, semi-nested, and nested PCR) | PBMC from blood, tumor sample, buffy coat, milk, somatic cells, semen, saliva, and nasal secretions. |
|
| [152,158,171] |
| Quantitative (real-time) |
|
| [123,153,162,163,164,169,172] | |
| Direct, blood-based PCR | Blood |
|
| [161] |
7. Hazards of BLV
7.1. Effects on the Function of the Immune System of Dairy Cows
7.2. Effects on the Milk Production and Milk Quality of Dairy Cows
8. Prevention and Control Strategies for BLV
9. Reducing the BLV PVL Contributes to BLV Prevention and Control
10. Conclusions
11. Future Prospective Studies
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Burny, A.; Bex, F.; Chantrenne, H.; Cleuter, Y.; Dekegel, D.; Ghysdael, J.; Kettmann, R.; Leclercq, M.; Leunen, J.; Mammerickx, M.; et al. Bovine leukemia virus involvement in enzootic bovine leukosis. Adv. Cancer Res. 1978, 28, 251–311. [Google Scholar] [CrossRef] [PubMed]
- Burny, A.; Bruck, C.; Cleuter, Y.; Couez, D.; Deschamps, J.; Gregoire, D.; Ghysdael, J.; Kettmann, R.; Mammerickx, M.; Marbaix, G.; et al. Bovine leukaemia virus and enzootic bovine leukosis. Onderstepoort J. Vet. Res. 1985, 52, 133–144. [Google Scholar] [PubMed]
- Kettmann, R.; Portetelle, D.; Mammerickx, M.; Cleuter, Y.; Dekegel, D.; Galoux, M.; Ghysdael, J.; Burny, A.; Chantrenne, H. Bovine leukemia virus: An exogenous RNA oncogenic virus. Proc. Natl. Acad. Sci. USA 1976, 73, 1014–1018. [Google Scholar] [CrossRef]
- Reed, V.I. Enzootic bovine leukosis. Can. Vet. J.-Rev. Vet. Can. 1981, 22, 95–102. [Google Scholar]
- Panel, E.A. Scientific opinion on enzootic bovine leukosis. EFSA J. 2015, 13, 4188. [Google Scholar]
- Nekouei, O.; Vanleeuwen, J.; Sanchez, J.; Kelton, D.; Tiwari, A.; Keefe, G. Herd-level risk factors for infection with bovine leukemia virus in Canadian dairy herds. Prev. Vet. Med. 2015, 119, 105–113. [Google Scholar] [CrossRef]
- Murakami, K.; Kobayashi, S.; Konishi, M.; Kameyama, K.; Tsutsui, T. Nationwide survey of bovine leukemia virus infection among dairy and beef breeding cattle in Japan from 2009–2011. J. Vet. Med. Sci. 2013, 75, 1123–1126. [Google Scholar] [CrossRef]
- Ma, B.; Gong, Q.; Sheng, C.; Liu, Y.; Ge, G.; Li, D.; Diao, N.; Shi, K.; Li, J.; Sun, Z.; et al. Prevalence of bovine leukemia in 1983–2019 in China: A systematic review and meta-analysis. Microb. Pathog. 2021, 150, 104681. [Google Scholar] [CrossRef]
- Ladronka, R.M.; Ainsworth, S.; Wilkins, M.J.; Norby, B.; Byrem, T.M.; Bartlett, P.C. Prevalence of bovine leukemia virus antibodies in US dairy cattle. Vet. Med. Int. 2018, 2018, 5831278. [Google Scholar] [CrossRef]
- Tsutsui, T.; Kobayashi, S.; Hayama, Y.; Yamamoto, T. Fraction of bovine leukemia virus-infected dairy cattle developing enzootic bovine leukosis. Prev. Vet. Med. 2016, 124, 96–101. [Google Scholar] [CrossRef]
- Gillet, N.; Florins, A.; Boxus, M.; Burteau, C.; Nigro, A.; Vandermeers, F.; Balon, H.; Bouzar, A.B.; Defoiche, J.; Burny, A.; et al. Mechanisms of leukemogenesis induced by bovine leukemia virus: Prospects for novel anti-retroviral therapies in human. Retrovirology 2007, 4, 18. [Google Scholar] [CrossRef]
- Norby, B.; Bartlett, P.C.; Byrem, T.M.; Erskine, R.J. Effect of infection with bovine leukemia virus on milk production in Michigan dairy cows. J. Dairy Sci. 2016, 99, 2043–2052. [Google Scholar] [CrossRef] [PubMed]
- Erskine, R.J.; Bartlett, P.C.; Byrem, T.M.; Render, C.L.; Febvay, C.; Houseman, J.T. Association between bovine leukemia virus, production, and population age in Michigan dairy herds. J. Dairy Sci. 2012, 95, 727–734. [Google Scholar] [CrossRef] [PubMed]
- Bartlett, P.C.; Norby, B.; Byrem, T.M.; Parmelee, A.; Ledergerber, J.T.; Erskine, R.J. Bovine leukemia virus and cow longevity in Michigan dairy herds. J. Dairy Sci. 2013, 96, 1591–1597. [Google Scholar] [CrossRef]
- Frie, M.C.; Coussens, P.M. Bovine leukemia virus: A major silent threat to proper immune responses in cattle. Vet. Immunol. Immunopathol. 2015, 163, 103–114. [Google Scholar] [CrossRef]
- Ruggiero, V.J.; Norby, B.; Benitez, O.J.; Hutchinson, H.; Sporer, K.; Droscha, C.; Swenson, C.L.; Bartlett, P.C. Controlling bovine leukemia virus in dairy herds by identifying and removing cows with the highest proviral load and lymphocyte counts. J. Dairy Sci. 2019, 102, 9165–9175. [Google Scholar] [CrossRef] [PubMed]
- Nuotio, L.; Rusanen, H.; Sihvonen, L.; Neuvonen, E. Eradication of enzootic bovine leukosis from Finland. Prev. Vet. Med. 2003, 59, 43–49. [Google Scholar] [CrossRef]
- Acaite, J.; Tamosiunas, V.; Lukauskas, K.; Milius, J.; Pieskus, J. The eradication experience of enzootic bovine leukosis from Lithuania. Prev. Vet. Med. 2007, 82, 83–89. [Google Scholar] [CrossRef]
- Enzootic Bovine Leukosis, Chapter 3.4.9. Available online: https://www.woah.org/fileadmin/Home/eng/Health_standards/tahm/3.04.09_EBL.pdf (accessed on 28 December 2023).
- Olaya-Galan, N.N.; Corredor-Figueroa, A.P.; Guzman-Garzon, T.C.; Rios-Hernandez, K.S.; Salas-Cardenas, S.P.; Patarroyo, M.A.; Gutierrez, M.F. Bovine leukaemia virus DNA in fresh milk and raw beef for human consumption. Epidemiol. Infect. 2017, 145, 3125–3130. [Google Scholar] [CrossRef]
- Khatami, A.; Pormohammad, A.; Farzi, R.; Saadati, H.; Mehrabi, M.; Kiani, S.J.; Ghorbani, S. Bovine Leukemia virus (BLV) and risk of breast cancer: A systematic review and meta-analysis of case-control studies. Infect. Agents Cancer 2020, 15, 48. [Google Scholar] [CrossRef]
- Khan, Z.; Abubakar, M.; Arshed, M.J.; Aslam, R.; Sattar, S.; Shah, N.A.; Javed, S.; Tariq, A.; Bostan, N.; Manzoor, S. Molecular investigation of possible relationships concerning bovine leukemia virus and breast cancer. Sci. Rep. 2022, 12, 4161. [Google Scholar] [CrossRef]
- Adekanmbi, F.; Mcneely, I.; Omeler, S.; Kalalah, A.; Poudel, A.; Merner, N.; Wang, C. Absence of bovine leukemia virus in the buffy coats of breast cancer cases from Alabama, USA. Microb. Pathog. 2021, 161, 105238. [Google Scholar] [CrossRef]
- Canova, R.; Weber, M.N.; Budaszewski, R.F.; Da, S.M.; Schwingel, D.; Canal, C.W.; Kreutz, L.C. Bovine leukemia viral DNA found on human breast tissue is genetically related to the cattle virus. One Health 2021, 13, 100252. [Google Scholar] [CrossRef] [PubMed]
- Buehring, G.C.; Shen, H.M.; Jensen, H.M.; Choi, K.Y.; Sun, D.; Nuovo, G. Bovine leukemia virus DNA in human breast tissue. Emerg. Infect. Dis. 2014, 20, 772–782. [Google Scholar] [CrossRef] [PubMed]
- Olaya-Galan, N.N.; Salas-Cardenas, S.P.; Rodriguez-Sarmiento, J.L.; Ibanez-Pinilla, M.; Monroy, R.; Corredor-Figueroa, A.P.; Rubiano, W.; de la Pena, J.; Shen, H.; Buehring, G.C.; et al. Risk factor for breast cancer development under exposure to bovine leukemia virus in Colombian women: A case-control study. PLoS ONE 2021, 16, e257492. [Google Scholar] [CrossRef]
- Afzal, S.; Fiaz, K.; Noor, A.; Sindhu, A.S.; Hanif, A.; Bibi, A.; Asad, M.; Nawaz, S.; Zafar, S.; Ayub, S.; et al. Interrelated Oncogenic Viruses and Breast Cancer. Front. Mol. Biosci. 2022, 9, 781111. [Google Scholar] [CrossRef]
- Lawson, J.S.; Salmons, B.; Glenn, W.K. Oncogenic Viruses and Breast Cancer: Mouse Mammary Tumor Virus (MMTV), Bovine Leukemia Virus (BLV), Human Papilloma Virus (HPV), and Epstein-Barr Virus (EBV). Front. Oncol. 2018, 8, 1. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Jiang, J.; Sun, W.; Zhang, J.; Huang, K.; Gu, X.; Yang, Y.; Xu, X.; Shi, Y.; Wang, C. Lack of association between bovine leukemia virus and breast cancer in Chinese patients. Breast Cancer Res. 2016, 18, 101. [Google Scholar] [CrossRef]
- Gillet, N.A.; Willems, L. Whole genome sequencing of 51 breast cancers reveals that tumors are devoid of bovine leukemia virus DNA. Retrovirology 2016, 13, 75. [Google Scholar] [CrossRef]
- Bender, A.P.; Robison, L.L.; Kashmiri, S.V.; Mcclain, K.L.; Woods, W.G.; Smithson, W.A.; Heyn, R.; Finlay, J.; Schuman, L.M.; Renier, C.; et al. No involvement of bovine leukemia virus in childhood acute lymphoblastic leukemia and non-Hodgkin’s lymphoma. Cancer Res. 1988, 48, 2919–2922. [Google Scholar] [PubMed]
- Zhao, X.; Buehring, G.C. Natural genetic variations in bovine leukemia virus envelope gene: Possible effects of selection and escape. Virology 2007, 366, 150–165. [Google Scholar] [CrossRef]
- Boris-Lawrie, K.; Altanerova, V.; Altaner, C.; Kucerova, L.; Temin, H.M. In vivo study of genetically simplified bovine leukemia virus derivatives that lack tax and rex. J. Virol. 1997, 71, 1514–1520. [Google Scholar] [CrossRef] [PubMed]
- Aida, Y.; Murakami, H.; Takahashi, M.; Takeshima, S.N. Mechanisms of pathogenesis induced by bovine leukemia virus as a model for human T-cell leukemia virus. Front. Microbiol. 2013, 4, 328. [Google Scholar] [CrossRef]
- Plant, E.; Bellefroid, M.; Van Lint, C. A complex network of transcription factors and epigenetic regulators involved in bovine leukemia virus transcriptional regulation. Retrovirology 2023, 20, 11. [Google Scholar] [CrossRef]
- Llames, L.; Goyache, J.; Domenech, A.; Montana, A.V.; Suarez, G.; Gomez-Lucia, E. Cellular distribution of bovine leukemia virus proteins gp51SU, Pr72(env), and Pr66(gag-pro) in persistently infected cells. Virus Res. 2001, 79, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Katoh, I.; Kyushiki, H.; Sakamoto, Y.; Ikawa, Y.; Yoshinaka, Y. Bovine leukemia virus matrix-associated protein MA(p15): Further processing and formation of a specific complex with the dimer of the 5′-terminal genomic RNA fragment. J. Virol. 1991, 65, 6845–6855. [Google Scholar] [CrossRef]
- Mamoun, R.Z.; Morisson, M.; Rebeyrotte, N.; Busetta, B.; Couez, D.; Kettmann, R.; Hospital, M.; Guillemain, B. Sequence variability of bovine leukemia virus env gene and its relevance to the structure and antigenicity of the glycoproteins. J. Virol. 1990, 64, 4180–4188. [Google Scholar] [CrossRef]
- Lairmore, M.D. Animal models of bovine leukemia virus and human T-lymphotrophic virus type-1: Insights in transmission and pathogenesis. Annu. Rev. Anim. Biosci. 2014, 2, 189–208. [Google Scholar] [CrossRef] [PubMed]
- Rola-Luszczak, M.; Sakhawat, A.; Pluta, A.; Rylo, A.; Bomba, A.; Bibi, N.; Kuzmak, J. Molecular Characterization of the env Gene of Bovine Leukemia Virus in Cattle from Pakistan with NGS-Based Evidence of Virus Heterogeneity. Pathogens 2021, 10, 910. [Google Scholar] [CrossRef] [PubMed]
- de Brogniez, A.; Bouzar, A.B.; Jacques, J.R.; Cosse, J.P.; Gillet, N.; Callebaut, I.; Reichert, M.; Willems, L. Mutation of a Single Envelope N-Linked Glycosylation Site Enhances the Pathogenicity of Bovine Leukemia Virus. J. Virol. 2015, 89, 8945–8956. [Google Scholar] [CrossRef]
- Gillet, N.A.; Gutierrez, G.; Rodriguez, S.M.; de Brogniez, A.; Renotte, N.; Alvarez, I.; Trono, K.; Willems, L. Massive depletion of bovine leukemia virus proviral clones located in genomic transcriptionally active sites during primary infection. PLoS Pathog. 2013, 9, e1003687. [Google Scholar] [CrossRef]
- Lefebvre, L.; Ciminale, V.; Vanderplasschen, A.; D’Agostino, D.; Burny, A.; Willems, L.; Kettmann, R. Subcellular localization of the bovine leukemia virus R3 and G4 accessory proteins. J. Virol. 2002, 76, 7843–7854. [Google Scholar] [CrossRef]
- Willems, L.; Kerkhofs, P.; Dequiedt, F.; Portetelle, D.; Mammerickx, M.; Burny, A.; Kettmann, R. Attenuation of bovine leukemia virus by deletion of R3 and G4 open reading frames. Proc. Natl. Acad. Sci. USA 1994, 91, 11532–11536. [Google Scholar] [CrossRef]
- Reichert, M.; Cantor, G.H.; Willems, L.; Kettmann, R. Protective effects of a live attenuated bovine leukaemia virus vaccine with deletion in the R3 and G4 genes. J. Gen. Virol. 2000, 81, 965–969. [Google Scholar] [CrossRef]
- Zyrianova, I.M.; Kovalchuk, S.N. Bovine leukemia virus tax gene/Tax protein polymorphism and its relation to Enzootic Bovine Leukosis. Virulence 2020, 11, 80–87. [Google Scholar] [CrossRef]
- Willems, L.; Grimonpont, C.; Kerkhofs, P.; Capiau, C.; Gheysen, D.; Conrath, K.; Roussef, R.; Mamoun, R.; Portetelle, D.; Burny, A.; et al. Phosphorylation of bovine leukemia virus Tax protein is required for in vitro transformation but not for transactivation. Oncogene 1998, 16, 2165–2176. [Google Scholar] [CrossRef]
- Pyeon, D.; Splitter, G.A. Regulation of bovine leukemia virus tax and pol mRNA levels by interleukin-2 and -10. J. Virol. 1999, 73, 8427–8434. [Google Scholar] [CrossRef]
- Kerkhofs, P.; Heremans, H.; Burny, A.; Kettmann, R.; Willems, L. In vitro and in vivo oncogenic potential of bovine leukemia virus G4 protein. J. Virol. 1998, 72, 2554–2559. [Google Scholar] [CrossRef] [PubMed]
- Licursi, M.; Inoshima, Y.; Wu, D.; Yokoyama, T.; Gonzalez, E.T.; Sentsui, H. Provirus variants of bovine leukemia virus in naturally infected cattle from Argentina and Japan. Vet. Microbiol. 2003, 96, 17–23. [Google Scholar] [CrossRef]
- Lee, E.; Kim, E.J.; Joung, H.K.; Kim, B.H.; Song, J.Y.; Cho, I.S.; Lee, K.K.; Shin, Y.K. Sequencing and phylogenetic analysis of the gp51 gene from Korean bovine leukemia virus isolates. Virol. J. 2015, 12, 64. [Google Scholar] [CrossRef] [PubMed]
- Corredor-Figueroa, A.P.; Salas, S.; Olaya-Galan, N.N.; Quintero, J.S.; Fajardo, A.; Sonora, M.; Moreno, P.; Cristina, J.; Sanchez, A.; Tobon, J.; et al. Prevalence and molecular epidemiology of bovine leukemia virus in Colombian cattle. Infect. Genet. Evol. 2020, 80, 104171. [Google Scholar] [CrossRef]
- Marawan, M.A.; Mekata, H.; Hayashi, T.; Sekiguchi, S.; Kirino, Y.; Horii, Y.; Moustafa, A.M.; Arnaout, F.K.; Galila, E.; Norimine, J. Phylogenetic analysis of env gene of bovine leukemia virus strains spread in Miyazaki prefecture, Japan. J. Vet. Med. Sci. 2017, 79, 912–916. [Google Scholar] [CrossRef]
- Pluta, A.; Rola-Luszczak, M.; Kubis, P.; Balov, S.; Moskalik, R.; Choudhury, B.; Kuzmak, J. Molecular characterization of bovine leukemia virus from Moldovan dairy cattle. Arch. Virol. 2017, 162, 1563–1576. [Google Scholar] [CrossRef]
- Le, D.T.; Yamashita-Kawanishi, N.; Okamoto, M.; Nguyen, S.V.; Nguyen, N.H.; Sugiura, K.; Miura, T.; Haga, T. Detection and genotyping of bovine leukemia virus (BLV) in Vietnamese cattle. J. Vet. Med. Sci. 2020, 82, 1042–1050. [Google Scholar] [CrossRef]
- Polat, M.; Takeshima, S.N.; Hosomichi, K.; Kim, J.; Miyasaka, T.; Yamada, K.; Arainga, M.; Murakami, T.; Matsumoto, Y.; de la Barra, D.V.; et al. A new genotype of bovine leukemia virus in South America identified by NGS-based whole genome sequencing and molecular evolutionary genetic analysis. Retrovirology 2016, 13, 4. [Google Scholar] [CrossRef]
- Hamada, R.; Metwally, S.; Polat, M.; Borjigin, L.; Ali, A.O.; Abdel-Hady, A.; Mohamed, A.; Wada, S.; Aida, Y. Detection and Molecular Characterization of Bovine Leukemia Virus in Egyptian Dairy Cattle. Front. Vet. Sci. 2020, 7, 608. [Google Scholar] [CrossRef]
- Rodriguez, S.M.; Golemba, M.D.; Campos, R.H.; Trono, K.; Jones, L.R. Bovine leukemia virus can be classified into seven genotypes: Evidence for the existence of two novel clades. J. Gen. Virol. 2009, 90, 2788–2797. [Google Scholar] [CrossRef]
- Gautam, S.; Mishra, N.; Kalaiyarasu, S.; Jhade, S.K.; Sood, R. Molecular Characterization of Bovine Leukaemia Virus (BLV) Strains Reveals Existence of Genotype 6 in Cattle in India with evidence of a new subgenotype. Transbound. Emerg. Dis. 2018, 65, 1968–1978. [Google Scholar] [CrossRef]
- Lee, E.; Kim, E.J.; Ratthanophart, J.; Vitoonpong, R.; Kim, B.H.; Cho, I.S.; Song, J.Y.; Lee, K.K.; Shin, Y.K. Molecular epidemiological and serological studies of bovine leukemia virus (BLV) infection in Thailand cattle. Infect. Genet. Evol. 2016, 41, 245–254. [Google Scholar] [CrossRef]
- Selim, A.; Manaa, E.A.; Alanazi, A.D.; Alyousif, M.S. Seroprevalence, Risk Factors and Molecular Identification of Bovine Leukemia Virus in Egyptian Cattle. Animals 2021, 11, 319. [Google Scholar] [CrossRef]
- Polat, M.; Takeshima, S.N.; Aida, Y. Epidemiology and genetic diversity of bovine leukemia virus. Virol. J. 2017, 14, 209. [Google Scholar] [CrossRef]
- Kincaid, R.P.; Burke, J.M.; Sullivan, C.S. RNA virus microRNA that mimics a B-cell oncomiR. Proc. Natl. Acad. Sci. USA 2012, 109, 3077–3082. [Google Scholar] [CrossRef]
- Rosewick, N.; Momont, M.; Durkin, K.; Takeda, H.; Caiment, F.; Cleuter, Y.; Vernin, C.; Mortreux, F.; Wattel, E.; Burny, A.; et al. Deep sequencing reveals abundant noncanonical retroviral microRNAs in B-cell leukemia/lymphoma. Proc. Natl. Acad. Sci. USA 2013, 110, 2306–2311. [Google Scholar] [CrossRef]
- Gillet, N.A.; Hamaidia, M.; de Brogniez, A.; Gutierrez, G.; Renotte, N.; Reichert, M.; Trono, K.; Willems, L. Bovine Leukemia Virus Small Noncoding RNAs Are Functional Elements That Regulate Replication and Contribute to Oncogenesis In Vivo. PLoS Pathog. 2016, 12, e1005588. [Google Scholar] [CrossRef]
- Santanam, U.; Zanesi, N.; Efanov, A.; Costinean, S.; Palamarchuk, A.; Hagan, J.P.; Volinia, S.; Alder, H.; Rassenti, L.; Kipps, T.; et al. Chronic lymphocytic leukemia modeled in mouse by targeted miR-29 expression. Proc. Natl. Acad. Sci. USA 2010, 107, 12210–12215. [Google Scholar] [CrossRef]
- Petersen, M.I.; Carignano, H.A.; Mongini, C.; Gonzalez, D.D.; Jaworski, J.P. Bovine leukemia virus encoded blv-miR-b4-3p microRNA is associated with reduced expression of anti-oncogenic gene in vivo. PLoS ONE 2023, 18, e281317. [Google Scholar] [CrossRef]
- Leisering, A. Hypertrophy der Malpigischen Korperchen der Milz. Ber. Uber Das Vet. Im Konigreich Sachs. 1871, 16, 15–16. [Google Scholar]
- Olson, C.; Miller, J. Enzootic Bovine Leukosis and Bovine Leukemia Virus; Martinus Nijhoff Publishing: Boston, MA, USA, 1987. [Google Scholar]
- Bollinger, O. ber Leukmie bei den Haustieren. Virchows Arch. 1874, 59, 4. [Google Scholar]
- Siedamgrotzky, O.; Hofmeister, V. Anleitung zur Mikroskopischen und Chemischen Diagnostik der Krankheiten der Hausthiere: Für Thierärzte und Landwirte; Schönfeld VIII: Dresden, Germany, 1876; p. 192. [Google Scholar]
- Miller, J.M.; Miller, L.D.; Olson, C.; Gillette, K.G. Virus-like particles in phytohemagglutinin-stimulated lymphocyte cultures with reference to bovine lymphosarcoma. JNCI-J. Natl. Cancer Inst. 1969, 43, 1297–1305. [Google Scholar]
- Bartlett, P.C.; Sordillo, L.M.; Byrem, T.M.; Norby, B.; Grooms, D.L.; Swenson, C.L.; Zalucha, J.; Erskine, R.J. Options for the control of bovine leukemia virus in dairy cattle. JAVMA-J. Am. Vet. Med. Assoc. 2014, 244, 914–922. [Google Scholar] [CrossRef]
- Whittington, R.; Donat, K.; Weber, M.F.; Kelton, D.; Nielsen, S.S.; Eisenberg, S.; Arrigoni, N.; Juste, R.; Saez, J.L.; Dhand, N.; et al. Control of paratuberculosis: Who, why and how. A review of 48 countries. BMC Vet. Res. 2019, 15, 198. [Google Scholar] [CrossRef]
- Ruggiero, V.J.; Bartlett, P.C. Control of Bovine Leukemia Virus in Three US Dairy Herds by Culling ELISA-Positive Cows. Vet. Med. Int. 2019, 2019, 3202184. [Google Scholar] [CrossRef]
- More, S.; Botner, A.; Butterworth, A.; Calistri, P.; Depner, K.; Edwards, S.; Garin-Bastuji, B.; Good, M.; Gortazar, S.C.; Michel, V.; et al. Assessment of listing and categorisation of animal diseases within the framework of the Animal Health Law (Regulation (EU) No 2016/429): Enzootic bovine leukosis (EBL). EFSA J. 2017, 15, e4956. [Google Scholar] [CrossRef][Green Version]
- WOAH. WOAH-WAHIS. Available online: https://wahis.woah.org/#/dashboards/country-or-disease-dashboard (accessed on 28 December 2023).
- OIE. World Animal Health Infromation Database-Version: 1.4. In World Animal Health Information Database; OIE: Paris, France, 2009. [Google Scholar]
- Commission Implementing Regulation (EU) 2021/620 of 15 April 2021 Laying Down Rules for the Application of Regulation (EU) 2016/429 of the European Parliament and of the Council as Regards the Approval of the Disease-Free and Non-Vaccination Status of Certain Member States or Zones or Compartments Thereof as Regards Certain Listed Diseases and the Approval of Eradication Programmes for Those Listed Diseases. Available online: http://data.europa.eu/eli/reg_impl/2021/620/oj (accessed on 28 December 2023).
- Nekouei, O.; Vanleeuwen, J.; Stryhn, H.; Kelton, D.; Keefe, G. Lifetime effects of infection with bovine leukemia virus on longevity and milk production of dairy cows. Prev. Vet. Med. 2016, 133, 1–9. [Google Scholar] [CrossRef]
- John, E.E.; Keefe, G.; Cameron, M.; Stryhn, H.; Mcclure, J.T. Development and implementation of a risk assessment and management program for enzootic bovine leukosis in Atlantic Canada. J. Dairy Sci. 2020, 103, 8398–8406. [Google Scholar] [CrossRef]
- Suzan, V.M.; Onuma, M.; Aguilar, R.E.; Murakami, Y. Prevalence of bovine herpesvirus-1, parainfluenza-3, bovine rotavirus, bovine viral diarrhea, bovine adenovirus-7, bovine leukemia virus and bluetongue virus antibodies in cattle in Mexico. JPN J. Vet. Res. 1983, 31, 125–132. [Google Scholar]
- Gonzalez, M.A.; Ceron-Tellez, F.; Sarmiento, S.R.; Tortora, P.J.; Rojas-Anaya, E.; Alvarez, H.R. Presence of co-infection between bovine leukemia virus and bovine herpesvirus 1 in herds vaccinated against bovine respiratory complex. Can. J. Vet. Res.-Rev. Can. Rech. Vet. 2023, 87, 105–109. [Google Scholar]
- Moratorio, G.; Obal, G.; Dubra, A.; Correa, A.; Bianchi, S.; Buschiazzo, A.; Cristina, J.; Pritsch, O. Phylogenetic analysis of bovine leukemia viruses isolated in South America reveals diversification in seven distinct genotypes. Arch. Virol. 2010, 155, 481–489. [Google Scholar] [CrossRef]
- Zinovieva, N.A.; Vinogradova, I.V.; Mikhailova, M.E.; Molofeeva, L.A.; Ernst, L.K. Prevalence of bovine leukemia virus in black and white cows with the different level of milk productive traits. Сельскoхoзяйственная биoлoгия 2012, 6, 49–55. [Google Scholar] [CrossRef]
- Trono, K.G.; Perez-Filgueira, D.M.; Duffy, S.; Borca, M.V.; Carrillo, C. Seroprevalence of bovine leukemia virus in dairy cattle in Argentina: Comparison of sensitivity and specificity of different detection methods. Vet. Microbiol. 2001, 83, 235–248. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, G.; Alvarez, I.; Politzki, R.; Lomonaco, M.; Dus, S.M.; Rondelli, F.; Fondevila, N.; Trono, K. Natural progression of Bovine Leukemia Virus infection in Argentinean dairy cattle. Vet. Microbiol. 2011, 151, 255–263. [Google Scholar] [CrossRef]
- Samara, S.I.; Lima, E.G.; Do Nascimento, A.A. Monitoring of enzootic bovine leukosis in dairy cattle from the Pitangueiras region in São Paulo, Brazil. Braz. J. Vet. Res. Anim. Sci. 1997, 34, 349–351. [Google Scholar] [CrossRef][Green Version]
- D’Angelino, J.L.; Garcia, M.; Birgel, E.H. Epidemiological study of enzootic bovine leukosis in Brazil. Trop. Anim. Health Prod. 1998, 30, 13–15. [Google Scholar] [CrossRef]
- Ramalho, G.C.; Silva, M.L.C.R.; Falcão, B.M.R.; Limeira, C.H.; Nogueira, D.B.; Dos Santos, A.M.; Martins, C.M.; Alves, C.J.; Clementino, I.J.; Santos, C.D.S.A. High herd-level seroprevalence and associated factors for bovine leukemia virus in the semi-arid Paraíba state, Northeast Region of Brazil. Prev. Vet. Med. 2021, 190, 105324. [Google Scholar] [CrossRef] [PubMed]
- Bonifaz, N.; Ulcuango, F. Prevalencia de leucosis bovina en la comunidad Santo Domingo n° 1, Cayambe-Ecuador 2012. La Granja Rev. De Cienc. De La Vida 2015, 22, 33–39. [Google Scholar]
- Ch, A.H. Bovine leukaemia virus infection in Peru. Trop. Anim. Health Prod. 1983, 15, 61. [Google Scholar] [CrossRef]
- Marin, C.; de Lopez, N.M.; Alvarez, L.; Lozano, O.; Espana, W.; Castanos, H.; Leon, A. Epidemiology of bovine leukemia in Venezuela. Ann. Rech. Vet. 1978, 9, 743–746. [Google Scholar]
- Nava, Z.; Obando, C.; Molina, M.; Bracamonte, M.; Tkachuk, O. Seroprevalence of enzootic bovine leukosis and its association with clinical signs and risk factors in dairy herds from Barinas State, Venezuela. Rev. De La Fac. De Cienc. Vet. 2011, 52, 13–23. [Google Scholar]
- Furtado, A.; Rosadilla, D.; Franco, G.; Piaggio, J.; Puentes, R. Leucosis Bovina Enzoótica en cuencas lecheras de productores familiares del Uruguay. Veterinaria 2013, 49, 29–37. [Google Scholar]
- Yang, Y.; Fan, W.; Mao, Y.; Yang, Z.; Lu, G.; Zhang, R.; Zhang, H.; Szeto, C.; Wang, C. Bovine leukemia virus infection in cattle of China: Association with reduced milk production and increased somatic cell score. J. Dairy Sci. 2016, 99, 3688–3697. [Google Scholar] [CrossRef]
- Meas, S.; Ohashi, K.; Tum, S.; Chhin, M.; Te, K.; Miura, K.; Sugimoto, C.; Onuma, M. Seroprevalence of bovine immunodeficiency virus and bovine leukemia virus in draught animals in Cambodia. J. Vet. Med. Sci. 2000, 62, 779–781. [Google Scholar] [CrossRef] [PubMed]
- Nekouei, O.; Stryhn, H.; Vanleeuwen, J.; Kelton, D.; Hanna, P.; Keefe, G. Predicting within-herd prevalence of infection with bovine leukemia virus using bulk-tank milk antibody levels. Prev. Vet. Med. 2015, 122, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, S.; Hidano, A.; Tsutsui, T.; Yamamoto, T.; Hayama, Y.; Nishida, T.; Muroga, N.; Konishi, M.; Kameyama, K.; Murakami, K. Analysis of risk factors associated with bovine leukemia virus seropositivity within dairy and beef breeding farms in Japan: A nationwide survey. Res. Vet. Sci. 2014, 96, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Ochirkhuu, N.; Konnai, S.; Odbileg, R.; Nishimori, A.; Okagawa, T.; Murata, S.; Ohashi, K. Detection of bovine leukemia virus and identification of its genotype in Mongolian cattle. Arch. Virol. 2016, 161, 985–991. [Google Scholar] [CrossRef]
- Polat, M.; Ohno, A.; Takeshima, S.N.; Kim, J.; Kikuya, M.; Matsumoto, Y.; Mingala, C.N.; Onuma, M.; Aida, Y. Detection and molecular characterization of bovine leukemia virus in Philippine cattle. Arch. Virol. 2015, 160, 285–296. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.F.; Siddique, U.; Shah, A.A.; Khan, I.; Anwar, F.; Ahmad, I.; Zeb, M.T.; Hassan, M.F.; Ali, T. Seroprevalence of bovine leukemia virus (BLV) in cattle from the North West of Pakistan. Pak. Vet. J 2019, 40, 127–129. [Google Scholar]
- Hsieh, J.C.; Li, C.Y.; Hsu, W.L.; Chuang, S.T. Molecular Epidemiological and Serological Studies of Bovine Leukemia Virus in Taiwan Dairy Cattle. Front. Vet. Sci. 2019, 6, 427. [Google Scholar] [CrossRef]
- Trainin, Z.; Brenner, J. The direct and indirect economic impacts of bovine leukemia virus infection on dairy cattle. Isr. J. Vet. Med. 2005, 60, 94. [Google Scholar]
- Hafez, S.M.; Sharif, M.; Al-Sukayran, A.; Dela-Cruz, D. Preliminary studies on enzootic bovine leukosis in Saudi dairy farms. DTW Dtsch. Tierarztl. Wochenschr. 1990, 97, 61–63. [Google Scholar]
- Burgu, I.; Alkan, F.; Karaoglu, T.; Bilge-Dagalp, S.; Can-Sahna, K.; Gungor, B.; Demir, B. Control and eradication programme of enzootic bovine leucosis (EBL) from selected dairy herds in Turkey. Dtsch. Tierarztl. Wochenschr. 2005, 112, 271–274. [Google Scholar]
- Khudhair, Y.I.; Hasso, S.A.; Yaseen, N.Y.; Al-Shammari, A.M. Serological and molecular detection of bovine leukemia virus in cattle in Iraq. Emerg. Microbes Infect. 2016, 5, e56. [Google Scholar] [CrossRef] [PubMed]
- Selim, A.; Megahed, A.A.; Kandeel, S.; Abdelhady, A. Risk factor analysis of bovine leukemia virus infection in dairy cattle in Egypt. Comp. Immunol. Microbiol. Infect. Dis. 2020, 72, 101517. [Google Scholar] [CrossRef] [PubMed]
- Lewin, H.A.; Bernoco, D. Evidence for BoLA-linked resistance and susceptibility to subclinical progression of bovine leukaemia virus infection. Anim. Genet. 1986, 17, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Eguchi, M.; Kawamura, H.; Wada, F.; Shimauchi, T.; Shiota, E.; Shibata, K.; Sugioka, Y. Promotion of calcification by imidazole and its suppression by diltiazem in the growth cartilage of rats with HEBP induced rickets. Int. Orthop. 1989, 13, 217–220. [Google Scholar] [CrossRef] [PubMed]
- Porta, N.G.; Alvarez, I.; Suarez, A.G.; Ruiz, V.; Abdala, A.; Trono, K. Experimental infection of sheep with Bovine leukemia virus (BLV): Minimum dose of BLV-FLK cells and cell-free BLV and neutralization activity of natural antibodies. Rev. Argent. Microbiol. 2019, 51, 316–323. [Google Scholar] [CrossRef]
- Abdalla, E.A.; Weigel, K.A.; Byrem, T.M.; Rosa, G. Genetic correlation of bovine leukosis incidence with somatic cell score and milk yield in a US Holstein population. J. Dairy Sci. 2016, 99, 2005–2009. [Google Scholar] [CrossRef] [PubMed]
- Lo, C.W.; Borjigin, L.; Saito, S.; Fukunaga, K.; Saitou, E.; Okazaki, K.; Mizutani, T.; Wada, S.; Takeshima, S.N.; Aida, Y. BoLA-DRB3 Polymorphism is Associated with Differential Susceptibility to Bovine Leukemia Virus-Induced Lymphoma and Proviral Load. Viruses 2020, 12, 352. [Google Scholar] [CrossRef] [PubMed]
- Le, T.D.; Nguyen, V.S.; Lo, C.W.; Dao, D.T.; Bui, N.V.; Ogawa, H.; Imai, K.; Sugiura, K.; Aida, Y.; Haga, T. Association between BoLA-DRB3 polymorphism and bovine leukemia virus proviral load in Vietnamese Holstein Friesian cattle. HLA 2022, 99, 105–112. [Google Scholar] [CrossRef]
- Maezawa, M.; Fujii, Y.; Akagami, M.; Kawakami, J.; Inokuma, H. BoLA-DRB3*15:01 allele is associated with susceptibility to early enzootic bovine leukosis onset in Holstein-Friesian and Japanese Black cattle. Vet. Microbiol. 2023, 284, 109829. [Google Scholar] [CrossRef]
- Daous, H.E.; Mitoma, S.; Elhanafy, E.; Thi, N.H.; Thi, M.N.; Notsu, K.; Kaneko, C.; Norimine, J.; Sekiguchi, S. Relationship between Allelic Heterozygosity in BoLA-DRB3 and Proviral Loads in Bovine Leukemia Virus-Infected Cattle. Animals 2021, 11, 647. [Google Scholar] [CrossRef]
- Bauermann, F.V.; Ridpath, J.F.; Dargatz, D.A. Bovine leukemia virus seroprevalence among cattle presented for slaughter in the United States. J. Vet. Diagn. Investig. 2017, 29, 704–706. [Google Scholar] [CrossRef] [PubMed]
- Murakami, K.; Kobayashi, S.; Konishi, M.; Kameyama, K.; Yamamoto, T.; Tsutsui, T. The recent prevalence of bovine leukemia virus (BLV) infection among Japanese cattle. Vet. Microbiol. 2011, 148, 84–88. [Google Scholar] [CrossRef]
- Oguma, K.; Suzuki, M.; Sentsui, H. Enzootic bovine leukosis in a two-month-old calf. Virus Res. 2017, 233, 120–124. [Google Scholar] [CrossRef]
- Shaghayegh, A. Detection and Identification of Enzootic Bovine Leukosis (EBL) in Calves in Iran. Arch. Razi Inst. 2019, 74, 321–325. [Google Scholar] [CrossRef] [PubMed]
- Watanuki, S.; Takeshima, S.N.; Borjigin, L.; Sato, H.; Bai, L.; Murakami, H.; Sato, R.; Ishizaki, H.; Matsumoto, Y.; Aida, Y. Visualizing bovine leukemia virus (BLV)-infected cells and measuring BLV proviral loads in the milk of BLV seropositive dams. Vet. Res. 2019, 50, 102. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, S.; Tsutsui, T.; Yamamoto, T.; Hayama, Y.; Muroga, N.; Konishi, M.; Kameyama, K.; Murakami, K. The role of neighboring infected cattle in bovine leukemia virus transmission risk. J. Vet. Med. Sci. 2015, 77, 861–863. [Google Scholar] [CrossRef]
- Yuan, Y.; Kitamura-Muramatsu, Y.; Saito, S.; Ishizaki, H.; Nakano, M.; Haga, S.; Matoba, K.; Ohno, A.; Murakami, H.; Takeshima, S.N.; et al. Detection of the BLV provirus from nasal secretion and saliva samples using BLV-CoCoMo-qPCR-2: Comparison with blood samples from the same cattle. Virus Res. 2015, 210, 248–254. [Google Scholar] [CrossRef]
- Haga, T. Enzootic Bovine Leukosis: How to prevent the disease and control the spread of BLV infection. In Proceedings of the 1st International Conference Postgraduate School Universitas Airlangga: “Implementation of Climate Change Agreement to Meet Sustainable Development Goals” (ICPSUAS 2017), Surabaya, Indonesia, 1–2 August 2017; pp. 13–14. [Google Scholar]
- Mekata, H.; Sekiguchi, S.; Konnai, S.; Kirino, Y.; Honkawa, K.; Nonaka, N.; Horii, Y.; Norimine, J. Evaluation of the natural perinatal transmission of bovine leukaemia virus. Vet. Rec. 2015, 176, 254. [Google Scholar] [CrossRef]
- Kohara, J.; Takeuchi, M.; Hirano, Y.; Sakurai, Y.; Takahashi, T. Vector control efficacy of fly nets on preventing bovine leukemia virus transmission. J. Vet. Med. Sci. 2018, 80, 1524–1527. [Google Scholar] [CrossRef]
- Selim, A.; Ali, A.F. Seroprevalence and risk factors for C. burentii infection in camels in Egypt. Comp. Immunol. Microbiol. Infect. Dis. 2020, 68, 101402. [Google Scholar] [CrossRef]
- Selim, A.; Radwan, A.; Arnaout, F. Seroprevalence and molecular characterization of West Nile Virus in Egypt. Comp. Immunol. Microbiol. Infect. Dis. 2020, 71, 101473. [Google Scholar] [CrossRef]
- Mekata, H.; Yamamoto, M.; Hayashi, T.; Kirino, Y.; Sekiguchi, S.; Konnai, S.; Horii, Y.; Norimine, J. Cattle with a low bovine leukemia virus proviral load are rarely an infectious source. JPN J. Vet. Res. 2018, 66, 157–163. [Google Scholar]
- Konishi, M.; Ishizaki, H.; Kameyama, K.I.; Murakami, K.; Yamamoto, T. The effectiveness of colostral antibodies for preventing bovine leukemia virus (BLV) infection in vitro. BMC Vet. Res. 2018, 14, 419. [Google Scholar] [CrossRef] [PubMed]
- Ott, S.L.; Johnson, R.; Wells, S.J. Association between bovine-leukosis virus seroprevalence and herd-level productivity on US dairy farms. Prev. Vet. Med. 2003, 61, 249–262. [Google Scholar] [CrossRef] [PubMed]
- Erskine, R.J.; Bartlett, P.C.; Byrem, T.M.; Render, C.L.; Febvay, C.; Houseman, J.T. Herd-level determinants of bovine leukaemia virus prevalence in dairy farms. J. Dairy Res. 2012, 79, 445–450. [Google Scholar] [CrossRef]
- Benitez, O.J.; Roberts, J.N.; Norby, B.; Bartlett, P.C.; Takeshima, S.N.; Watanuki, S.; Aida, Y.; Grooms, D.L. Breeding bulls as a potential source of bovine leukemia virus transmission in beef herds. JAVMA-J. Am. Vet. Med. Assoc. 2019, 254, 1335–1340. [Google Scholar] [CrossRef]
- Alvarez, I.; Gutierrez, G.; Gammella, M.; Martinez, C.; Politzki, R.; Gonzalez, C.; Caviglia, L.; Carignano, H.; Fondevila, N.; Poli, M.; et al. Evaluation of total white blood cell count as a marker for proviral load of bovine leukemia virus in dairy cattle from herds with a high seroprevalence of antibodies against bovine leukemia virus. Am. J. Vet. Res. 2013, 74, 744–749. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, I.; Levy, D. Pathobiology of bovine leukemia virus. Vet. Res. 1994, 25, 521–536. [Google Scholar]
- Willems, L.; Burny, A.; Collete, D.; Dangoisse, O.; Dequiedt, F.; Gatot, J.S.; Kerkhofs, P.; Lefebvre, L.; Merezak, C.; Peremans, T.; et al. Genetic determinants of bovine leukemia virus pathogenesis. Aids Res. Hum. Retrovir. 2000, 16, 1787–1795. [Google Scholar] [CrossRef]
- Zaghawa, A.; Beier, D.; Abd El Rahim, I.; El Ballal, S.; Karim, I.; Conraths, F.J.; Marquardt, O. An outbreak of enzootic bovine leukosis in upper egypt: Clinical, laboratory and molecular–epidemiological studies. J. Vet. Med. Ser. B 2002, 49, 123–129. [Google Scholar] [CrossRef]
- Zaher, K.S.; Ahmed, W.M. Bovine leukemia virus infection in dairy cows in Egypt. Acad. J. Cancer Res. 2014, 7, 126–130. [Google Scholar]
- Uchiyama, J.; Murakami, H.; Sato, R.; Mizukami, K.; Suzuki, T.; Shima, A.; Ishihara, G.; Sogawa, K.; Sakaguchi, M. Examination of the fecal microbiota in dairy cows infected with bovine leukemia virus. Vet. Microbiol. 2020, 240, 108547. [Google Scholar] [CrossRef] [PubMed]
- Blagitz, M.G.; Souza, F.N.; Batista, C.F.; Azevedo, L.; Sanchez, E.; Diniz, S.A.; Silva, M.X.; Haddad, J.P.; Della, L.A. Immunological implications of bovine leukemia virus infection. Res. Vet. Sci. 2017, 114, 109–116. [Google Scholar] [CrossRef]
- Patel, J.R.; Heldens, J.G.; Bakonyi, T.; Rusvai, M. Important mammalian veterinary viral immunodiseases and their control. Vaccine 2012, 30, 1767–1781. [Google Scholar] [CrossRef]
- Bai, L.; Yokoyama, K.; Watanuki, S.; Ishizaki, H.; Takeshima, S.N.; Aida, Y. Development of a new recombinant p24 ELISA system for diagnosis of bovine leukemia virus in serum and milk. Arch. Virol. 2019, 164, 201–211. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, G.; Alvarez, I.; Fondevila, N.; Politzki, R.; Lomonaco, M.; Rodriguez, S.; Dus, S.M.; Trono, K. Detection of bovine leukemia virus specific antibodies using recombinant p24-ELISA. Vet. Microbiol. 2009, 137, 224–234. [Google Scholar] [CrossRef]
- Kuczewski, A.; Orsel, K.; Barkema, H.W.; Kelton, D.F.; Hutchins, W.A.; van der Meer, F. Short communication: Evaluation of 5 different ELISA for the detection of bovine leukemia virus antibodies. J. Dairy Sci. 2018, 101, 2433–2437. [Google Scholar] [CrossRef]
- Endoh, D.; Mizutani, T.; Kirisawa, R.; Maki, Y.; Saito, H.; Kon, Y.; Morikawa, S.; Hayashi, M. Species-independent detection of RNA virus by representational difference analysis using non-ribosomal hexanucleotides for reverse transcription. Nucleic Acids Res. 2005, 33, e65. [Google Scholar] [CrossRef]
- Heinecke, N.; Tortora, J.; Martinez, H.A.; Gonzalez-Fernandez, V.D.; Ramirez, H. Detection and genotyping of bovine leukemia virus in Mexican cattle. Arch. Virol. 2017, 162, 3191–3196. [Google Scholar] [CrossRef]
- Bannenberg, T. Experiments using a modified disposition arrangement of the AGID test in the diagnosis of enzootic bovine leukosis. Zentralbl Vet. B 1982, 29, 676–680. [Google Scholar] [CrossRef]
- Martin, D.; Arjona, A.; Soto, I.; Barquero, N.; Viana, M.; Gomez-Lucia, E. Comparative study of PCR as a direct assay and ELISA and AGID as indirect assays for the detection of bovine leukaemia virus. J. Vet. Med. Ser. B 2001, 48, 97–106. [Google Scholar] [CrossRef]
- Buzala, E.; Deren, W. Comparison of PLA with AGID and ELISA results in serology diagnosis of bovine leukosis. Pol. J. Vet. Sci. 2003, 6, 9–11. [Google Scholar]
- Swart, K.; Hagemeijer, A.; Lowenberg, B. Density profiles and purification of chronic myeloid leukemia cells forming colonies in the PHA-leukocyte feeder assay. Exp. Hematol. 1981, 9, 588–594. [Google Scholar]
- World Organisation for Animal Health. Chapter 3.4.9 Enzootic bovine leukosis. In Manual of Diagnostic Tests and Vaccines for Terrestrial Animals; World Organisation for Animal Health: Paris, France, 2018; ISBN 978-92-95108-18-9. [Google Scholar]
- Monti, G.E.; Frankena, K.; Engel, B.; Buist, W.; Tarabla, H.D.; de Jong, M.C. Evaluation of a new antibody-based enzyme-linked immunosorbent assay for the detection of bovine leukemia virus infection in dairy cattle. J. Vet. Diagn. Investig. 2005, 17, 451–457. [Google Scholar] [CrossRef] [PubMed]
- Jimba, M.; Takeshima, S.; Murakami, H.; Kohara, J.; Kobayashi, N.; Matsuhashi, T.; Ohmori, T.; Nunoya, T.; Aida, Y. BLV-CoCoMo-qPCR: A useful tool for evaluating bovine leukemia virus infection status. BMC Vet. Res. 2012, 8, 167. [Google Scholar] [CrossRef] [PubMed]
- Levy, D.; Deshayes, L.; Parodi, A.L.; Levy, J.P.; Stephenson, J.R.; Devare, S.G.; Gilden, R.V. Bovine leukemia virus specific antibodies among French cattle. II. Radioimmunoassay with the major structural protein (BLV p24). Int. J. Cancer 1977, 20, 543–550. [Google Scholar] [CrossRef]
- Lew, A.E.; Bock, R.E.; Miles, J.; Cuttell, L.B.; Steer, P.; Nadin-Davis, S.A. Sensitive and specific detection of bovine immunodeficiency virus and bovine syncytial virus by 5′ Taq nuclease assays with fluorescent 3′ minor groove binder-DNA probes. J. Virol. Methods 2004, 116, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Juliarena, M.A.; Gutierrez, S.E.; Ceriani, C. Determination of proviral load in bovine leukemia virus-infected cattle with and without lymphocytosis. Am. J. Vet. Res. 2007, 68, 1220–1225. [Google Scholar] [CrossRef]
- Lew, A.E.; Bock, R.E.; Molloy, J.B.; Minchin, C.M.; Robinson, S.J.; Steer, P. Sensitive and specific detection of proviral bovine leukemia virus by 5′ Taq nuclease PCR using a 3′ minor groove binder fluorogenic probe. J. Virol. Methods 2004, 115, 167–175. [Google Scholar] [CrossRef]
- Evermann, J.F.; Jackson, M.K. Laboratory diagnostic tests for retroviral infections in dairy and beef cattle. Vet. Clin. N. Am. Food Anim. Pract. 1997, 13, 87–106. [Google Scholar] [CrossRef]
- Wu, X.; Notsu, K.; Matsuura, Y.; Mitoma, S.; El, D.H.; Norimine, J.; Sekiguchi, S. Development of droplet digital PCR for quantification of bovine leukemia virus proviral load using unpurified genomic DNA. J. Virol. Methods 2023, 315, 114706, Erratum in J. Virol. Methods 2023, 315, 114708. [Google Scholar] [CrossRef]
- De Brun, M.L.; Cosme, B.; Petersen, M.; Alvarez, I.; Folgueras-Flatschart, A.; Flatschart, R.; Panei, C.J.; Puentes, R. Development of a droplet digital PCR assay for quantification of the proviral load of bovine leukemia virus. J. Vet. Diagn. Investig. 2022, 34, 439–447. [Google Scholar] [CrossRef] [PubMed]
- Nishimori, A.; Konnai, S.; Ikebuchi, R.; Okagawa, T.; Nakahara, A.; Murata, S.; Ohashi, K. Direct polymerase chain reaction from blood and tissue samples for rapid diagnosis of bovine leukemia virus infection. J. Vet. Med. Sci. 2016, 78, 791–796. [Google Scholar] [CrossRef] [PubMed]
- Takeshima, S.; Kitamura-Muramatsu, Y.; Yuan, Y.; Polat, M.; Saito, S.; Aida, Y. BLV-CoCoMo-qPCR-2: Improvements to the BLV-CoCoMo-qPCR assay for bovine leukemia virus by reducing primer degeneracy and constructing an optimal standard curve. Arch. Virol. 2015, 160, 1325–1332. [Google Scholar] [CrossRef]
- Jimba, M.; Takeshima, S.N.; Matoba, K.; Endoh, D.; Aida, Y. BLV-CoCoMo-qPCR: Quantitation of bovine leukemia virus proviral load using the CoCoMo algorithm. Retrovirology 2010, 7, 91. [Google Scholar] [CrossRef] [PubMed]
- Panei, C.J.; Takeshima, S.; Omori, T.; Nunoya, T.; Davis, W.C.; Ishizaki, H.; Matoba, K.; Aida, Y. Estimation of bovine leukemia virus (BLV) proviral load harbored by lymphocyte subpopulations in BLV-infected cattle at the subclinical stage of enzootic bovine leucosis using BLV-CoCoMo-qPCR. BMC Vet. Res. 2013, 9, 95. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.; Rommel, F.; Mone, J. Development of a syncytia inhibition assay for the detection of antibodies to bovine leukemia virus in naturally infected cattle; comparison with Western blot and agar gel immunodiffusion. J. Virol. Methods 1998, 70, 177–182. [Google Scholar] [CrossRef]
- Trainin, Z.; Meirom, R.; Gluckmann, A. Comparison between the immunodiffusion and the immunofluorescence tests in the diagnosis of bovine leukemia virus (BLV). Ann. Rech. Vet. 1978, 9, 659–662. [Google Scholar]
- Sato, H.; Bai, L.; Borjigin, L.; Aida, Y. Overexpression of bovine leukemia virus receptor SLC7A1/CAT1 enhances cellular susceptibility to BLV infection on luminescence syncytium induction assay (LuSIA). Virol. J. 2020, 17, 57. [Google Scholar] [CrossRef]
- Sato, H.; Watanuki, S.; Murakami, H.; Sato, R.; Ishizaki, H.; Aida, Y. Development of a luminescence syncytium induction assay (LuSIA) for easily detecting and quantitatively measuring bovine leukemia virus infection. Arch. Virol. 2018, 163, 1519–1530. [Google Scholar] [CrossRef] [PubMed]
- Sato, H.; Watanuki, S.; Bai, L.; Borjigin, L.; Ishizaki, H.; Matsumoto, Y.; Hachiya, Y.; Sentsui, H.; Aida, Y. A sensitive luminescence syncytium induction assay (LuSIA) based on a reporter plasmid containing a mutation in the glucocorticoid response element in the long terminal repeat U3 region of bovine leukemia virus. Virol. J. 2019, 16, 66. [Google Scholar] [CrossRef]
- Sato, H.; Fukui, J.N.; Hirano, H.; Osada, H.; Arimura, Y.; Masuda, M.; Aida, Y. Application of the Luminescence Syncytium Induction Assay to Identify Chemical Compounds That Inhibit Bovine Leukemia Virus Replication. Viruses 2022, 15, 4. [Google Scholar] [CrossRef] [PubMed]
- Tajima, S.; Ikawa, Y.; Aida, Y. Complete bovine leukemia virus (BLV) provirus is conserved in BLV-infected cattle throughout the course of B-cell lymphosarcoma development. J. Virol. 1998, 72, 7569–7576. [Google Scholar] [CrossRef]
- Takeshima, S.; Watanuki, S.; Ishizaki, H.; Matoba, K.; Aida, Y. Development of a direct blood-based PCR system to detect BLV provirus using CoCoMo primers. Arch. Virol. 2016, 161, 1539–1546. [Google Scholar] [CrossRef]
- Casoli, C.; Pilotti, E.; Bertazzoni, U. Proviral load determination of HTLV-1 and HTLV-2 in patients’ peripheral blood mononuclear cells by real-time PCR. Methods Mol. Biol. 2014, 1087, 315–323. [Google Scholar] [CrossRef]
- Pineda, M.V.; Bouzas, M.B.; Remesar, M.; Fridman, A.; Remondegui, C.; Mammana, L.; Altamirano, N.; Paradiso, P.; Costantini, P.; Tadey, L.; et al. Relevance of HTLV-1 proviral load in asymptomatic and symptomatic patients living in endemic and non-endemic areas of Argentina. PLoS ONE 2019, 14, e225596. [Google Scholar] [CrossRef]
- Rodrigues, E.S.; Salustiano, S.; Santos, E.V.; Slavov, S.N.; Picanco-Castro, V.; Maconetto, J.M.; de Haes, T.M.; Takayanagui, O.M.; Covas, D.T.; Kashima, S. Monitoring of HTLV-1-associated diseases by proviral load quantification using multiplex real-time PCR. J. Neurovirol. 2022, 28, 27–34. [Google Scholar] [CrossRef]
- Torti, C.; Quiros-Roldan, M.E.; Cologni, G.; Nichelatti, M.; Ceresoli, F.; Pinti, M.; Nasi, M.; Cossarizza, A.; Lapadula, G.; Costarelli, S.; et al. Plasma HIV load and proviral DNA decreases after two standard antiretroviral regimens in HIV-positive patients naive to antiretrovirals. Curr. HIV Res. 2008, 6, 43–48. [Google Scholar] [CrossRef]
- Bruisten, S.M.; Reiss, P.; Loeliger, A.E.; van Swieten, P.; Schuurman, R.; Boucher, C.A.; Weverling, G.J.; Huisman, J.G. Cellular proviral HIV type 1 DNA load persists after long-term RT-inhibitor therapy in HIV type 1 infected persons. AIDS Res. Hum. Retrovir. 1998, 14, 1053–1058. [Google Scholar] [CrossRef]
- Watanabe, A.; Murakami, H.; Kakinuma, S.; Murao, K.; Ohmae, K.; Isobe, N.; Akamatsu, H.; Seto, T.; Hashimura, S.; Konda, K.; et al. Association between bovine leukemia virus proviral load and severity of clinical mastitis. J. Vet. Med. Sci. 2019, 81, 1431–1437. [Google Scholar] [CrossRef] [PubMed]
- Marin-Flamand, E.; Araiza-Hernandez, D.M.; Vargas-Ruiz, A.; Rangel-Rodriguez, I.C.; Gonzalez-Tapia, L.A.; Ramirez-Alvarez, H.; Hernandez-Balderas, R.J.; Garcia-Camacho, L.A. Relationship of persistent lymphocytosis, antibody titers, and proviral load with expression of interleukin-12, interferon-gamma, interleukin-2, interleukin-4, interleukin-10, and transforming growth factor-beta in cows infected with bovine leukemia virus from a high-prevalence dairy complex. Can. J. Vet. Res.-Rev. Can. Rech. Vet. 2022, 86, 269–285. [Google Scholar]
- Nieto, F.M.; Souza, F.N.; Lendez, P.A.; Martinez-Cuesta, L.; Santos, K.R.; Della, L.A.; Ceriani, M.C.; Dolcini, G.L. Lymphocyte proliferation and apoptosis of lymphocyte subpopulations in bovine leukemia virus-infected dairy cows with high and low proviral load. Vet. Immunol. Immunopathol. 2018, 206, 41–48. [Google Scholar] [CrossRef]
- Nakada, S.; Fujimoto, Y.; Kohara, J.; Makita, K. Economic losses associated with mastitis due to bovine leukemia virus infection. J. Dairy Sci. 2023, 106, 576–588. [Google Scholar] [CrossRef]
- Nishiike, M.; Haoka, M.; Doi, T.; Kohda, T.; Mukamoto, M. Development of a preliminary diagnostic measure for bovine leukosis in dairy cows using peripheral white blood cell and lymphocyte counts. J. Vet. Med. Sci. 2016, 78, 1145–1151. [Google Scholar] [CrossRef] [PubMed]
- Hutchinson, H.C.; Norby, B.; Droscha, C.J.; Sordillo, L.M.; Coussens, P.M.; Bartlett, P.C. Bovine leukemia virus detection and dynamics following experimental inoculation. Res. Vet. Sci. 2020, 133, 269–275. [Google Scholar] [CrossRef]
- Somura, Y.; Sugiyama, E.; Fujikawa, H.; Murakami, K. Comparison of the copy numbers of bovine leukemia virus in the lymph nodes of cattle with enzootic bovine leukosis and cattle with latent infection. Arch. Virol. 2014, 159, 2693–2697. [Google Scholar] [CrossRef]
- Jaworski, J.P.; Pluta, A.; Rola-Luszczak, M.; Mcgowan, S.L.; Finnegan, C.; Heenemann, K.; Carignano, H.A.; Alvarez, I.; Murakami, K.; Willems, L.; et al. Interlaboratory Comparison of Six Real-Time PCR Assays for Detection of Bovine Leukemia Virus Proviral DNA. J. Clin. Microbiol. 2018, 56, 10–1128. [Google Scholar] [CrossRef]
- Nakada, S.; Kohara, J.; Makita, K. Estimation of circulating bovine leukemia virus levels using conventional blood cell counts. J. Dairy Sci. 2018, 101, 11229–11236. [Google Scholar] [CrossRef]
- Kobayashi, T.; Inagaki, Y.; Ohnuki, N.; Sato, R.; Murakami, S.; Imakawa, K. Increasing Bovine leukemia virus (BLV) proviral load is a risk factor for progression of Enzootic bovine leucosis: A prospective study in Japan. Prev. Vet. Med. 2019, 178, 104680. [Google Scholar] [CrossRef]
- Yoneyama, S.; Kobayashi, S.; Matsunaga, T.; Tonosaki, K.; Leng, D.; Sakai, Y.; Yamada, S.; Kimura, A.; Ichijo, T.; Hikono, H.; et al. Comparative Evaluation of Three Commercial Quantitative Real-Time PCRs Used in Japan for Bovine Leukemia Virus. Viruses 2022, 14, 1182. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, G.; Carignano, H.; Alvarez, I.; Martinez, C.; Porta, N.; Politzki, R.; Gammella, M.; Lomonaco, M.; Fondevila, N.; Poli, M.; et al. Bovine leukemia virus p24 antibodies reflect blood proviral load. BMC Vet. Res. 2012, 8, 187. [Google Scholar] [CrossRef] [PubMed]
- Meirom, R.; Moss, S.; Brenner, J. Bovine leukemia virus-gp51 antigen expression is associated with CD5 and IgM markers on infected lymphocytes. Vet. Immunol. Immunopathol. 1997, 59, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Teutsch, M.R.; Lewin, H.A. Aberrant expression of immunoglobulin mRNA in bovine leukemia virus-infected cattle. Vet. Immunol. Immunopathol. 1996, 53, 87–94. [Google Scholar] [CrossRef]
- Meiron, R.; Brenner, J.; Gluckman, A.; Avraham, R.; Trainin, Z. Humoral and cellular responses in calves experimentally infected with bovine leukemia virus (BLV). Vet. Immunol. Immunopathol. 1985, 9, 105–114. [Google Scholar] [CrossRef] [PubMed]
- Trainin, Z.; Brenner, J.; Meirom, R.; Ungar-Waron, H. Detrimental effect of bovine leukemia virus (BLV) on the immunological state of cattle. Vet. Immunol. Immunopathol. 1996, 54, 293–302. [Google Scholar] [CrossRef] [PubMed]
- Amills, M.; Ramiya, V.; Norimine, J.; Olmstead, C.A.; Lewin, H.A. Reduced IL-2 and IL-4 mRNA expression in CD4+ T cells from bovine leukemia virus-infected cows with persistent lymphocytosis. Virology 2002, 304, 1–9. [Google Scholar] [CrossRef]
- Trueblood, E.S.; Brown, W.C.; Palmer, G.H.; Davis, W.C.; Stone, D.M.; Mcelwain, T.F. B-lymphocyte proliferation during bovine leukemia virus-induced persistent lymphocytosis is enhanced by T-lymphocyte-derived interleukin-2. J. Virol. 1998, 72, 3169–3177. [Google Scholar] [CrossRef]
- Usuga-Monroy, C.; Gonzalez, H.L.; Echeverri, Z.J.; Diaz, F.J.; Lopez-Herrera, A. IFN-gamma mRNA expression is lower in Holstein cows infected with bovine leukemia virus with high proviral load and persistent lymphocytosis. Acta Virol. 2020, 64, 451–456. [Google Scholar] [CrossRef]
- Farias, M.; Lendez, P.A.; Marin, M.; Quintana, S.; Martinez-Cuesta, L.; Ceriani, M.C.; Dolcini, G.L. Toll-like receptors, IFN-gamma and IL-12 expression in bovine leukemia virus-infected animals with low or high proviral load. Res. Vet. Sci. 2016, 107, 190–195. [Google Scholar] [CrossRef]
- Pyeon, D.; Splitter, G.A. Interleukin-12 p40 mRNA expression in bovine leukemia virus-infected animals: Increase in alymphocytosis but decrease in persistent lymphocytosis. J. Virol. 1998, 72, 6917–6921. [Google Scholar] [CrossRef] [PubMed]
- Iwan, E.; Szczotka, M.; Kocki, J.; Pluta, A. Determination of cytokine profiles in populations of dendritic cells from cattle infected with bovine leukaemia virus. Pol. J. Vet. Sci. 2018, 21, 681–690. [Google Scholar] [CrossRef] [PubMed]
- Iwan, E.; Szczotka, M.; Kocki, J. Cytokine profiles of dendritic cells (DCs) during infection with bovine leukaemia virus (BLV). Pol. J. Vet. Sci. 2017, 20, 221–231. [Google Scholar] [CrossRef]
- Konnai, S.; Suzuki, S.; Shirai, T.; Ikebuchi, R.; Okagawa, T.; Sunden, Y.; Mingala, C.N.; Onuma, M.; Murata, S.; Ohashi, K. Enhanced expression of LAG-3 on lymphocyte subpopulations from persistently lymphocytotic cattle infected with bovine leukemia virus. Comp. Immunol. Microbiol. Infect. Dis. 2013, 36, 63–69. [Google Scholar] [CrossRef]
- Shirai, T.; Konnai, S.; Ikebuchi, R.; Okagawa, T.; Suzuki, S.; Sunden, Y.; Onuma, M.; Murata, S.; Ohashi, K. Molecular cloning of bovine lymphocyte activation gene-3 and its expression characteristics in bovine leukemia virus-infected cattle. Vet. Immunol. Immunopathol. 2011, 144, 462–467. [Google Scholar] [CrossRef]
- Okagawa, T.; Konnai, S.; Ikebuchi, R.; Suzuki, S.; Shirai, T.; Sunden, Y.; Onuma, M.; Murata, S.; Ohashi, K. Increased bovine Tim-3 and its ligand expressions during bovine leukemia virus infection. Vet. Res. 2012, 43, 45. [Google Scholar] [CrossRef]
- Suzuki, S.; Konnai, S.; Okagawa, T.; Ikebuchi, R.; Nishimori, A.; Kohara, J.; Mingala, C.N.; Murata, S.; Ohashi, K. Increased expression of the regulatory T cell-associated marker CTLA-4 in bovine leukemia virus infection. Vet. Immunol. Immunopathol. 2015, 163, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Pollari, F.L.; Wangsuphachart, V.L.; Digiacomo, R.F.; Evermann, J.F. Effects of bovine leukemia virus infection on production and reproduction in dairy cattle. Can. J. Vet. Res.-Rev. Can. Rech. Vet. 1992, 56, 289–295. [Google Scholar]
- Yang, Y.; Gong, Z.; Lu, Y.; Lu, X.; Zhang, J.; Meng, Y.; Peng, Y.; Chu, S.; Cao, W.; Hao, X.; et al. Dairy Cows Experimentally Infected With Bovine Leukemia Virus Showed an Increased Milk Production in Lactation Numbers 3–4: A 4-Year Longitudinal Study. Front. Microbiol. 2022, 13, 946463. [Google Scholar] [CrossRef]
- Jacobs, R.M.; Heeney, J.L.; Godkin, M.A.; Leslie, K.E.; Taylor, J.A.; Davies, C.; Valli, V.E. Production and related variables in bovine leukaemia virus-infected cows. Vet. Res. Commun. 1991, 15, 463–474. [Google Scholar] [CrossRef]
- Ruiz, V.; Porta, N.G.; Lomonaco, M.; Trono, K.; Alvarez, I. Bovine Leukemia Virus Infection in Neonatal Calves. Risk Factors and Control Measures. Front. Vet. Sci. 2018, 5, 267. [Google Scholar] [CrossRef]
- Esteban, E.N.; Poli, M.; Poiesz, B.; Ceriani, C.; Dube, S.; Gutierrez, S.; Dolcini, G.; Gagliardi, R.; Perez, S.; Lützelschwab, C. Bovine leukemia virus (BLV), proposed control and eradication programs by marker assisted breeding of genetically resistant cattle. Anim. Genet. 2009, 2009, 107–130. [Google Scholar]
- Rodriguez, S.M.; Florins, A.; Gillet, N.; de Brogniez, A.; Sanchez-Alcaraz, M.T.; Boxus, M.; Boulanger, F.; Gutierrez, G.; Trono, K.; Alvarez, I.; et al. Preventive and therapeutic strategies for bovine leukemia virus: Lessons for HTLV. Viruses 2011, 3, 1210–1248. [Google Scholar] [CrossRef] [PubMed]
- Balic, D.; Lojkic, I.; Periskic, M.; Bedekovic, T.; Jungic, A.; Lemo, N.; Roic, B.; Cac, Z.; Barbic, L.; Madic, J. Identification of a new genotype of bovine leukemia virus. Arch. Virol. 2012, 157, 1281–1290. [Google Scholar] [CrossRef]
- Bartlett, P.C.; Ruggiero, V.J.; Hutchinson, H.C.; Droscha, C.J.; Norby, B.; Sporer, K.; Taxis, T.M. Current Developments in the Epidemiology and Control of Enzootic Bovine Leukosis as Caused by Bovine Leukemia Virus. Pathogens 2020, 9, 58. [Google Scholar] [CrossRef]
- Hayashi, T.; Mekata, H.; Sekiguchi, S.; Kirino, Y.; Mitoma, S.; Honkawa, K.; Horii, Y.; Norimine, J. Cattle with the BoLA class II DRB3*0902 allele have significantly lower bovine leukemia proviral loads. J. Vet. Med. Sci. 2017, 79, 1552–1555. [Google Scholar] [CrossRef]
- Nikbakht, B.G.; Ghorbanpour, R.; Esmailnejad, A. Association of BoLA-DRB3.2 Alleles with BLV Infection Profiles (Persistent Lymphocytosis/Lymphosarcoma) and Lymphocyte Subsets in Iranian Holstein Cattle. Biochem. Genet. 2016, 54, 194–207. [Google Scholar] [CrossRef] [PubMed]
- Nakatsuchi, A.; Watanuki, S.; Borjigin, L.; Sato, H.; Bai, L.; Matsuura, R.; Kuroda, M.; Murakami, H.; Sato, R.; Asaji, S.; et al. BoLA-DRB3 Polymorphism Controls Proviral Load and Infectivity of Bovine Leukemia Virus (BLV) in Milk. Pathogens 2022, 11, 210. [Google Scholar] [CrossRef]
- Borjigin, L.; Lo, C.W.; Bai, L.; Hamada, R.; Sato, H.; Yoneyama, S.; Yasui, A.; Yasuda, S.; Yamanaka, R.; Mimura, M.; et al. Risk Assessment of Bovine Major Histocompatibility Complex Class II DRB3 Alleles for Perinatal Transmission of Bovine Leukemia Virus. Pathogens 2021, 10, 502. [Google Scholar] [CrossRef]
- Glass, E.J.; Baxter, R.; Leach, R.; Taylor, G. Breeding for Disease Resistance in Farm Animals; CAB International: Wallingford, UK, 2010. [Google Scholar]
- Basrur, P.K.; King, W.A. Genetics then and now: Breeding the best and biotechnology. Rev. Sci. Tech. Off. Int. Epizoot. 2005, 24, 31–49. [Google Scholar] [CrossRef]
- Williams, J.L. The use of marker-assisted selection in animal breeding and biotechnology. Rev. Sci. Tech. Off. Int. Epizoot. 2005, 24, 379–391. [Google Scholar] [CrossRef]
- Maradei, E.; Perez, B.C.; Malirat, V.; Salgado, G.; Seki, C.; Pedemonte, A.; Bonastre, P.; D’Aloia, R.; La Torre, J.L.; Mattion, N.; et al. Characterization of foot-and-mouth disease virus from outbreaks in Ecuador during 2009–2010 and cross-protection studies with the vaccine strain in use in the region. Vaccine 2011, 29, 8230–8240. [Google Scholar] [CrossRef]
- Miller, J.M.; Van Der Maaten, M.J. Evaluation of an inactivated bovine leukemia virus preparation as an immunogen in cattle. Ann. Rech. Vet. 1978, 9, 871–877. [Google Scholar] [PubMed]
- Patrascu, I.V.; Coman, S.; Sandu, I.; Stiube, P.; Munteanu, I.; Coman, T.; Ionescu, M.; Popescu, D.; Mihailescu, D. Specific protection against bovine leukemia virus infection conferred on cattle by the Romanian inactivated vaccine BL-VACC-RO. Virologie 1980, 31, 95–102. [Google Scholar]
- Parfanovich, M.I.; Zhdanov, V.M.; Lazarenko, A.A.; Nomm, E.M.; Simovart, Y.; Parakin, V.K.; Lemesh, V.M. The possibility of specific protection against bovine leukaemia virus infection and bovine leukaemia with inactivated BLV. Br. Vet. J. 1983, 139, 137–146. [Google Scholar] [CrossRef]
- Kabeya, H.; Ohashi, K.; Ohishi, K.; Sugimoto, C.; Amanuma, H.; Onuma, M. An effective peptide vaccine to eliminate bovine leukaemia virus (BLV) infected cells in carrier sheep. Vaccine 1996, 14, 1118–1122. [Google Scholar] [CrossRef]
- Miller, J.M.; Van der Maaten, M.J.; Schmerr, M.J. Vaccination of cattle with binary ethylenimine-treated bovine leukemia virus. Am. J. Vet. Res. 1983, 44, 64–67. [Google Scholar] [PubMed]
- Fukuyama, S.; Kodama, K.; Hirahara, T.; Nakajima, N.; Takamura, K.; Sasaki, O.; Imanishi, J. Protection against bovine leukemia virus infection by use of inactivated vaccines in cattle. J. Vet. Med. Sci. 1993, 55, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Onuma, M.; Hodatsu, T.; Yamamoto, S.; Higashihara, M.; Masu, S.; Mikami, T.; Izawa, H. Protection by vaccination against bovine leukemia virus infection in sheep. Am. J. Vet. Res. 1984, 45, 1212–1215. [Google Scholar]
- Burkhardt, H.; Rosenthal, S.; Wittmann, W.; Starick, E.; Scholz, D.; Rosenthal, H.A.; Kluge, K.H. Immunization of young cattle with gp51 of the bovine leukosis virus and the subsequent experimental infection. Arch. Exp. Veterinarmed. 1989, 43, 933–942. [Google Scholar] [PubMed]
- Merza, M.; Sober, J.; Sundquist, B.; Toots, I.; Morein, B. Characterization of purified gp 51 from bovine leukemia virus integrated into iscom. Physicochemical properties and serum antibody response to the integrated gp51. Arch. Virol. 1991, 120, 219–231. [Google Scholar] [CrossRef]
- Bruck, C.; Mathot, S.; Portetelle, D.; Berte, C.; Franssen, J.D.; Herion, P.; Burny, A. Monoclonal antibodies define eight independent antigenic regions on the bovine leukemia virus (BLV) envelope glycoprotein gp51. Virology 1982, 122, 342–352. [Google Scholar] [CrossRef]
- Gatei, M.H.; Naif, H.M.; Kumar, S.; Boyle, D.B.; Daniel, R.C.; Good, M.F.; Lavin, M.F. Protection of sheep against bovine leukemia virus (BLV) infection by vaccination with recombinant vaccinia viruses expressing BLV envelope glycoproteins: Correlation of protection with CD4 T-cell response to gp51 peptide 51–70. J. Virol. 1993, 67, 1803–1810. [Google Scholar] [CrossRef]
- Callebaut, I.; Mornon, J.P.; Burny, A.; Portetelle, D. The bovine leukemia virus (BLV) envelope glycoprotein gp51 as a general model for the design of a subunit vaccine against retroviral infection: Mapping of functional sites through immunological and structural data. Leukemia 1994, 8 (Suppl. S1), S218–S221. [Google Scholar]
- Okada, K.; Sonoda, K.; Koyama, M.; Yin, S.; Ikeda, M.; Goryo, M.; Chen, S.L.; Kabeya, H.; Ohishi, K.; Onuma, M. Delayed-type hypersensitivity in sheep induced by synthetic peptides of bovine leukemia virus encapsulated in mannan-coated liposome. J. Vet. Med. Sci. 2003, 65, 515–518. [Google Scholar] [CrossRef]
- Ohishi, K.; Kabeya, H.; Amanuma, H.; Onuma, M. Peptide-based bovine leukemia virus (BLV) vaccine that induces BLV-Env specific Th-1 type immunity. Leukemia 1997, 11 (Suppl. S3), 223–226. [Google Scholar]
- Portetelle, D.; Limbach, K.; Burny, A.; Mammerickx, M.; Desmettre, P.; Riviere, M.; Zavada, J.; Paoletti, E. Recombinant vaccinia virus expression of the bovine leukaemia virus envelope gene and protection of immunized sheep against infection. Vaccine 1991, 9, 194–200. [Google Scholar] [CrossRef]
- Kumar, S.; Andrew, M.E.; Boyle, D.B.; Brandon, R.B.; Lavin, M.F.; Daniel, R.C. Expression of bovine leukaemia virus envelope gene by recombinant vaccinia viruses. Virus Res. 1990, 17, 131–142. [Google Scholar] [CrossRef]
- Ohishi, K.; Ikawa, Y. T cell-mediated destruction of bovine leukemia virus-infected peripheral lymphocytes by bovine leukemia virus env-vaccinia recombinant vaccine. Aids Res. Hum. Retrovir. 1996, 12, 393–398. [Google Scholar] [CrossRef]
- Cherney, T.M.; Schultz, R.D. Viral status and antibody response in cattle inoculated with recombinant bovine leukemia virus-vaccinia virus vaccines after challenge exposure with bovine leukemia virus-infected lymphocytes. Am. J. Vet. Res. 1996, 57, 812–818. [Google Scholar]
- Brillowska, A.; Dabrowski, S.; Rulka, J.; Kubis, P.; Buzala, E.; Kur, J. Protection of cattle against bovine leukemia virus (BLV) infection could be attained by DNA vaccination. Acta Biochim. Pol. 1999, 46, 971–976. [Google Scholar] [CrossRef] [PubMed]
- Van den Broeke, A.; Oumouna, M.; Beskorwayne, T.; Szynal, M.; Cleuter, Y.; Babiuk, S.; Bagnis, C.; Martiat, P.; Burny, A.; Griebel, P. Cytotoxic responses to BLV tax oncoprotein do not prevent leukemogenesis in sheep. Leuk. Res. 2010, 34, 1663–1669. [Google Scholar] [CrossRef] [PubMed]
- Suarez, A.G.; Gutierrez, G.; Camussone, C.; Calvinho, L.; Abdala, A.; Alvarez, I.; Petersen, M.; Franco, L.; Destefano, G.; Monti, G.; et al. A safe and effective vaccine against bovine leukemia virus. Front. Immunol. 2022, 13, 980514. [Google Scholar] [CrossRef] [PubMed]
- Kechine, T.; Ali, T.; Worku, T.; Abdisa, L.; Assebe, Y.T. Anxiety and Associated Factors Among Clients on Highly Active Antiretroviral Therapy (HAART) in Public Hospitals of Southern Ethiopia: A Multi-Center Cross-Sectional Study. Psychol. Res. Behav. Manag. 2022, 15, 3889–3900. [Google Scholar] [CrossRef]
- Ntolou, P.; Pani, P.; Panis, V.; Madianos, P.; Vassilopoulos, S. The effect of antiretroviral therapyon the periodontal conditions of patients with HIV infection: A systematic review and meta-analysis. J. Clin. Periodontol. 2023, 50, 170–182. [Google Scholar] [CrossRef]
- Murakami, H.; Murakami-Kawai, M.; Kamisuki, S.; Hisanobu, S.; Tsurukawa, Y.; Uchiyama, J.; Sakaguchi, M.; Tsukamoto, K. Specific antiviral effect of violaceoid E on bovine leukemia virus. Virology 2021, 562, 1–8. [Google Scholar] [CrossRef]
- Nishimori, A.; Konnai, S.; Okagawa, T.; Maekawa, N.; Ikebuchi, R.; Goto, S.; Sajiki, Y.; Suzuki, Y.; Kohara, J.; Ogasawara, S.; et al. In vitro and in vivo antivirus activity of an anti-programmed death-ligand 1 (PD-L1) rat-bovine chimeric antibody against bovine leukemia virus infection. PLoS ONE 2017, 12, e174916. [Google Scholar] [CrossRef]

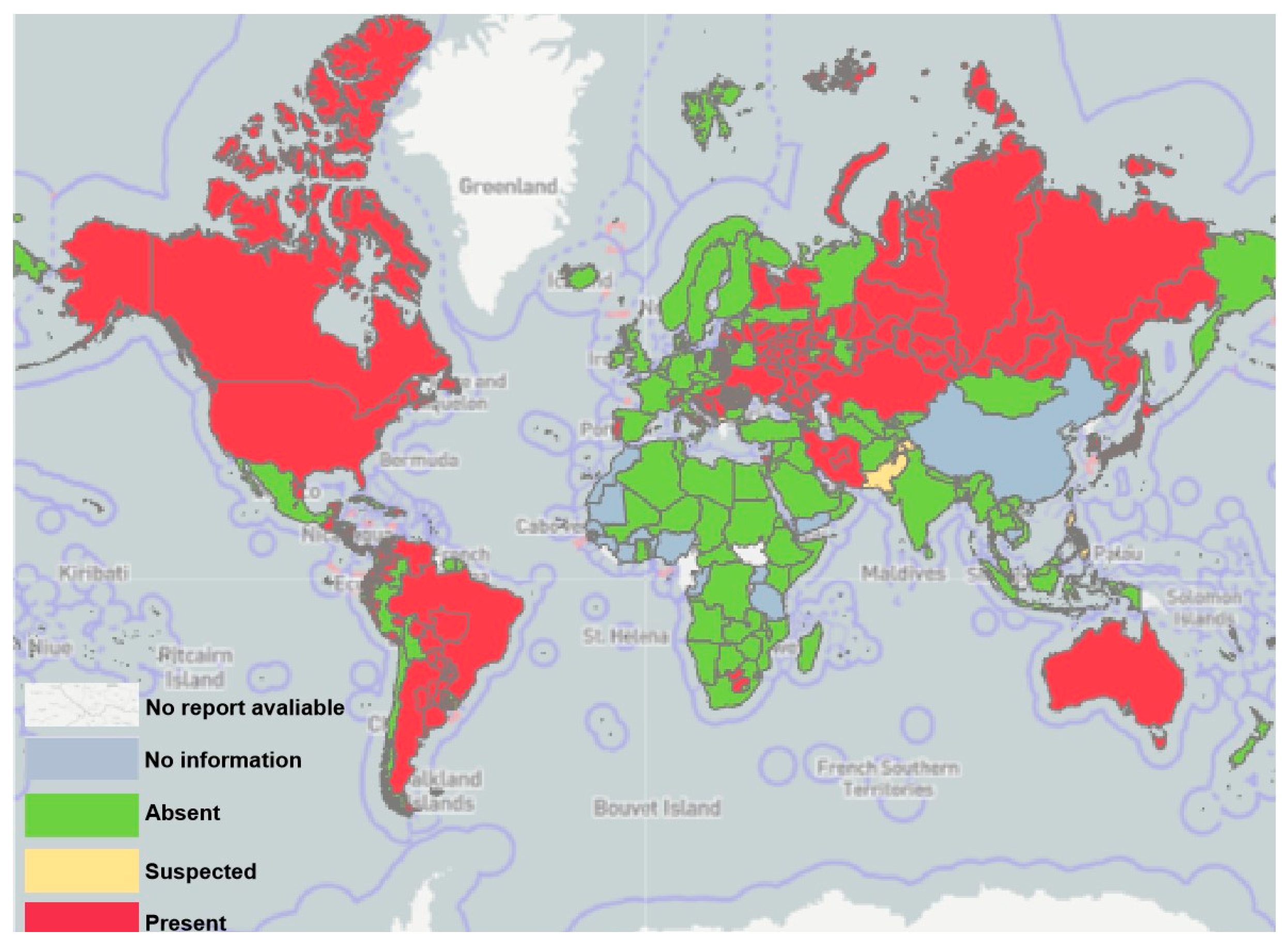
| Status | Continent | Countries | Year | References |
|---|---|---|---|---|
| BLV-free | Europe | Belgium | 2016 2021 | [5,78,79] |
| Czech Republic | ||||
| Denmark | ||||
| Germany | ||||
| Estonia | ||||
| Ireland | ||||
| Spain | ||||
| France | ||||
| Italy | ||||
| Cyprus | ||||
| Latvia | ||||
| Lithuania | ||||
| Luxembourg | ||||
| The Netherlands | ||||
| Austria | ||||
| Poland | ||||
| Portugal | ||||
| Romania | ||||
| Slovenia | ||||
| Finland | ||||
| Sweden | ||||
| United Kingdom (Northern Ireland) | ||||
| Oceania | Australia | 2013 | ||
| New Zealand | 2008 | |||
| Tunisia | 2005 | |||
| Asia | Kyrgyzstan | 2008 | ||
| Kazakhstan | 2007 | |||
| BLV existing in countries with unknown prevalence | Europe | Belarus | Present | |
| Bulgaria | ||||
| Croatia | ||||
| Greece | ||||
| Ukraine | ||||
| BLV existing in countries with variable prevalence | North America | Canada 78% at herd level 88.39% at herd level 89.30% at herd level | 1998–2003 2016 2018 | [80,81] |
| Mexico Dairy 36.1%, beef 4% Dairy 50.6% | 1983 2023 | [82,83] | ||
| USA Dairy 83.9%, beef 39% | 2007 | [84] | ||
| Europe | Russia 22.1% to 25.4% | 2012 | [85] | |
| South America | Argentina (Buenos Aires) 90.9% at herd level Argentina (multiple regions) 84% at herd level 90.16% | 1998–1999 2007 2011 | [56,86,87] | |
| Bolivia (multiple regions) 30.7% at individual level | 2008 | [56] | ||
| Brazil 17.1% at herd level 60.8% at herd level 23.4% at herd level | 1980–1989 1992–1995 2021 | [88,89,90] | ||
| Chile (southern regions) 27.9% at individual level | 2009 | [56] | ||
| Colombia 62% at individual level | 2020 | [52] | ||
| Ecuador 5.6% at individual level | 2012 | [91] | ||
| Paraguay (Asuncion) 54.7% at individual level | 2008 | [56] | ||
| Peru (multiple regions) 31% at individual level, 42.3% at individual level | 1983 2008 | [56,92] | ||
| Venezuela 33.3% at individual level 60.83% at individual level | 1978 2011 | [93,94] | ||
| Uruguay 10.4% at individual level | 2013 | [95] | ||
| Asia | China Dairy 49.1%, beef 1.6% | 2013–2014 | [96] | |
| Cambodia Draught cattle 5.3% | 2000 | [97] | ||
| Iran (nationwide) 22.1% to 25.4% | 2012–2014 | [98] | ||
| Myanmar 9.1% at individual level | 2016 | [56] | ||
| Japan (nationwide) Dairy 49.1%, beef 1.6% 79.1% at dairy herd level 73.3% at individual level | 2009–2011 2007 2012–2014 | [6,99] | ||
| Mongolia Dairy 3.9% | 2014 | [100] | ||
| The Philippines 4.8% to 9.7% | 2010–2012 | [101] | ||
| Pakistan Dairy 20% | 2019 | [102] | ||
| Taiwan 81.8% at animal level, 99.1% at herd level | 2019 | [103] | ||
| Thailand 58.7% at individual level | 2013–2014 | [60] | ||
| Middle East | Israel 5% at individual level | 2005 | [104] | |
| Saudi Arabia Dairy 20.2% | 1990 | [105] | ||
| Turkey Dairy 48.3% | 2005 | [106] | ||
| Iraq Dairy 7% | 2015 | [107] | ||
| Egypt Dairy 17.7% | 2020 | [108] |
| Test Assay | Sample Type | Target | Advantages | Disadvantages | References |
|---|---|---|---|---|---|
| AGID | Serum | Antibodies (p24, gp51) | Specific, simple, rapid, and low screening cost | Less sensitive, inconclusive, and fails to evaluate disease states | [137,147,148,149,152] |
| ELISA | Serum, milk, bulk milk | Antibodies (p24, gp51) | Sensitive, specific, large-scale screening, and rapid | False negatives (particularly in cattle during the early stages of infection) False positives (maternally derived antibodies) Cannot evaluate disease states of infected cattle | [142,143,148,149,153] |
| RIA | Serum | Antibodies (p24) | Sensitive in detecting BLV at an early stage of infection | Cannot be used for mass screening | [154] |
| PHA | Virus particle | BLV glycoprotein | Sensitive, specific, rapid, and low screening cost | Affected by pH and temperature Hemagglutination activity reduced by trypsin and neuraminidase | [150] |
| Goal/Method | Tool/Components | Applicability |
|---|---|---|
| Eradication (elimination of infected animals) | Test and slaughter
| Small heard, with low herd BLV prevalence, supported for restocking
|
Test and separate
| ||
| Control (reduction of the rate of effective contacts) | Safe herd management practices
| All herds |
| Prevention (avoiding introduction) | Biosecurity measures
| All herds |
| Surveillance (maintaining disease-/infection free-status) |
| Free herds/territories |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lv, G.; Wang, J.; Lian, S.; Wang, H.; Wu, R. The Global Epidemiology of Bovine Leukemia Virus: Current Trends and Future Implications. Animals 2024, 14, 297. https://doi.org/10.3390/ani14020297
Lv G, Wang J, Lian S, Wang H, Wu R. The Global Epidemiology of Bovine Leukemia Virus: Current Trends and Future Implications. Animals. 2024; 14(2):297. https://doi.org/10.3390/ani14020297
Chicago/Turabian StyleLv, Guanxin, Jianfa Wang, Shuai Lian, Hai Wang, and Rui Wu. 2024. "The Global Epidemiology of Bovine Leukemia Virus: Current Trends and Future Implications" Animals 14, no. 2: 297. https://doi.org/10.3390/ani14020297
APA StyleLv, G., Wang, J., Lian, S., Wang, H., & Wu, R. (2024). The Global Epidemiology of Bovine Leukemia Virus: Current Trends and Future Implications. Animals, 14(2), 297. https://doi.org/10.3390/ani14020297





