1. Buffalo Introduction
Bovidae is a family rank that contains nine genera, among which there is the genus
Bubalus. This genus, in turn, includes five species:
B. arnee also known as wild water buffalo which lives in Asia;
B. depressicornis and
B. quarlesi known as lowland anoa and mountain anoa, respectively, which live only in Indonesia;
B. mindorensis known as Tamarraw or Mindoro which lives only on Mindoro Island in the Philippines; and
B. bubalis known as water buffalo [
1]. This last species includes two different subspecies:
B. bubalis bubalis known as river buffalo and
B. bubalis kerebau known as swamp buffalo. The first one is bigger than the last one and is raised especially for milk production. A river buffalo’s body weight is between 450 and 1000 kg compared to 320–450 kg of swamp buffalo which is raised mainly for work purposes. In addition, these two subspecies are interfertile with their progeny containing 49 chromosomes given that the river buffalo is 2
n = 50 chromosomes and the swamp buffalo is 2
n = 48 chromosomes [
2]. Globally, there are 123 breeds of water buffalo [
3], but only three breeds are widely distributed worldwide: Murrah, Nili-Ravi, and Mediterranean Italian thanks to their high milk yield [
1]. FAO [
4] reported that the population of buffalo at the worldwide level amounted to 203,939,158 heads in 2021.
The introduction of this animals in Italy is not yet well known, but year by year, it has begun to become more important to the economy of the State, so much that in 2000, the “Mediterranean Italian” buffalo breed was officially recognized by the Ministry of Agriculture [
5]. This breed has a thick skin, is black to reddish or slate gray in color, and becomes lighter in the belly area. The horns are brown, symmetrical, 50–60 cm long, and directed sideways and backwards. The average weight of the female is around 650 kg despite the weight of the male being between 700–800 kg [
6]. Mediterranean Italian buffalo breed is raised almost exclusively for milk production, which is mainly used to produce mozzarella cheese, and in central-south Italy, milk is used to produce a specific mozzarella cheese called “Mozzarella di Bufala Campana PDO”. Productivity is about 1800–3500 kg per lactation [
7] and milk production at a global level is from 600 to 4500 kg per lactation [
8]. FAO [
4] reports worldwide milk production amount to have been 137,761,642.79 L in 2021, which corresponds to 15% of the total milk yield. This result puts buffalo milk in second place to milk’s ranking around the world, it is preceded only by bovine milk. The nutritional quality of buffalo milk is better than cow’s milk; in fact, buffalo milk contains about 8.2% fat and 4.3% protein with respect to the 3.8%fat and 3.4%protein of cow’s milk. Moreover, buffalo milk also has a higher content of minerals compared to cow’s milk [
9].
Today, Mediterranean Italian buffalo management is mainly intensive. Dairy buffalos are kept loose in paddocks close to the milking room, where the animals are submitted to udder control and mechanically milked twice a day. Some females are subjected to estrus induction to obtain calving before spring (about 50% fertility) as the milk pays out more in spring and summer according to consumer demand. In intensive systems, dairy buffalos normally receive a total mixed ration (TMR) composed of maize silage, concentrates, hay, straw, and sometimes by-products [
10].
In Asia, the main production systems are extensive, where the animals live in pastures and grazing provides 50–60% of the feed resources, or semi-intensive, where the feedstuff used is wheat straw, chopped green roughages, concentrates, cotton seed cakes, and rice by-products, and they are given in the open air along feeding ways [
10,
11].
The production of milk and its chemical composition are affected by several factors such as breed, genetics, the number of lactations, the days of lactation, the lactation phase, feeding, season, health status, etc. [
12,
13].
The objective of this review is to analyze how the season and, in particular, the hot season (namely heat stress, HS) interfere with buffalo livestock and especially the effect that HS has on the dairy buffalo performances.
2. Heat Stress Introduction
The demand for livestock products is growing rapidly, driven by population and income growth plus urbanization. Simultaneously, livestock production is facing increasing pressure from climate change effects, such as increasing temperatures, more variable precipitation patterns, more frequent extreme events, and increasing carbon dioxide concentration [
14]. The global temperature has increased by 1.5 °C above pre-industrial levels [
15]. Moreover, the increasing temperature is accompanied by water scarcity, and this condition leads to a more severe situation of alert [
16]. Given the current climate scenario and not very encouraging future estimates [
17,
18], we must expect farming to be increasingly adversely affected by the effects of HS. Heat stress can be simply defined as a condition that occurs when an animal cannot dissipate an adequate quantity of endogenous heat, whether it is produced or absorbed by the body, to maintain body thermal balance [
19]. This can happen when the ambient temperature and humidity are high and can cause a range of negative effects on the animal’s health and productivity. Thermal stress is triggered when environmental conditions exceed the upper or lower critical temperature of domestic animals requiring an increase in basal metabolism to deal with the stress [
20]. Heat stress occurs when the ambient temperature is above the thermoneutral zone.
It is well known that HS can cause a range of negative effects on dairy cattle, including decreased milk production, reduced feed intake, reduced reproductive performance, and increased susceptibility to diseases [
21]. In addition, HS can cause physiological changes such as an increased heart and respiration rate, increased blood flow to the skin, and increased sweating which can lead to a loss of electrolytes, dehydration, and inflammation. For these reasons, HS has a huge economic impact on the global dairy industry.
Little information is available on the effect of HS in buffalo reared under intensive conditions. Buffalos are widespread in many areas of the world, mainly in Asia, some Mediterranean countries, and some Eastern European and Latin American countries [
22]. Therefore, buffalos are adapted to different climates. In particular, due to their morphological, anatomical, and behavioral characteristics, buffalos are better adapted to hot and humid climates [
22]. Compared to cows, it appears that buffalos are more tolerant to HS and can better handle tropical climates, with fewer negative effects on production and physiology. However, they exhibit signs of greater distress when exposed to direct solar radiation [
22,
23]. The body temperature of buffalo is slightly lower than that of cattle, but buffalo skin is usually black, so their body absorbs a large amount of solar radiation due to their dark skin and sparse hair, and, in addition, buffalo have a less efficient evaporative cooling system due to their rather poor sweating capacity compared with cattle [
22]. In fact, buffalo skin has one-sixth of the density of sweat glands compared to cows; thus, buffalo dissipate heat poorly by sweating [
22]. The maximum number of sweat glands/mm
2 on buffalo skin was observed the in head dorsal followed by the abdomen dorsal, tail dorsal, neck dorsal, and thorax dorsal areas [
24]. The distribution of sweat glands, the dark color of the skin, and the sparse body hair adversely affect heat tolerance in these animals.
The greater presence of buffalo is mostly found at latitudes characterized by a hot–humid climate (tropical and subtropical areas) and for this reason, they have developed defensive mechanisms over time such as a large quantity of melanin and sebaceous glands which, respectively, prevent ultraviolet rays from being able to pass through the dermis [
25] and, through the sebum secreted and dissolved by high temperatures, reflect the sun’s rays [
26]. These glands secrete sebum, a fatty substance that covers the surface of the skin and coats it with a lubricant, making it slippery to water and mud. This fatty sebum, together with the thick upper horny layer of the skin, prevents the water and solutes it contains from being absorbed by the skin. In this way, the animal is protected from the harmful effects of any deleterious chemical compounds present in the water. Furthermore, the sebum layer melts during the hot season and becomes more shiny reflecting many of the Sun’s rays, thus relieving the animal from excessive external thermal load.
Buffalos are more heat-stressed (HSed) when they are prevented from displaying their adaptive behavioral traits such as seeking shelter, wallowing, and/or diving into water [
27]. It has been shown that buffalos are better able to regulate their body temperature if left to splash around in pools of water rather than using showers as a cooling system [
28]. It is not always possible to have pools; however, like any other animal, they still need to have appropriate measures for HS management to maintain their productivity and health.
The exposure of buffalo to hot conditions causes a series of changes in their biological functions which includes a decrease in feed intake, the efficiency and utilization of their diet, disturbances in the metabolism of water, proteins, energy and mineral balances, enzymatic reactions, hormonal secretions, and blood metabolites [
29], a reduction in milk yield and quality [
30], and a reduction in the manifestation of heat and therefore fertility [
31]. The effects of HS on the quality of production are less evident than in cattle [
30]; the negative impacts of HS on various biological functions are discussed in more detail later in the paper.
High air temperature and humidity are the main factors responsible for variations in the physiological reactions of animals, and these variations can be different depending on the species and breed [
21,
32]. As reported in the literature [
33], thermal parameters are the key factors used to calculate heat transfer: these include temperature, humidity, wind speed, and solar radiation. The combination of those parameters results in several indicators that are useful to determine the stress condition to which animals are exposed. The most widely used is the temperature–humidity index (THI), which combines air temperature with humidity providing a single index that gives information on animal comfort. The temperature–humidity index is mostly used for the measurement of HS condition because data relating to other parameters that could affect HS, such as solar radiation, wind speed, and precipitation, are not always available.
Numerous studies have established THI thresholds for HS in cattle but there are few studies that indicate the optimal values of THI for buffalo. A value of THI < 72 is considered optimal, THI between 72 to 79 is considered as mild stress, 80–89 is considered as moderate stress, and ≥90 is considered as severe stress in buffalo [
34]. When the maximum THI increases above 72, mild stress conditions in buffalo are reflected in a slight decline in milk yield [
34].
Considering the effects of THI on the reproductive performance of buffalo, Dash et al. [
35] classified the months of the year into two categories based on the THI values: a non-heat stress zone (NHSZ) and a heat stress zone (HSZ). The months from October to March were included in the NHSZ with the THI ranging from 56.71 to 73.21, and the months from April to September were considered in the HSZ with the THI ranging from 75.39 to 81.60. Within the HSZ, a critical heat stress zone (CHSZ) was also characterized. The CHSZ corresponded to the months of May and June and the THI ranged from 80.27 to 81.60. Choudhary and Sirohi [
34], based on an analysis of their data set, reported the maximum, minimum, and average THI threshold levels for buffalo: the cut-off values were 74.37, 61.73, and 68.15, respectively. The threshold level indicates the critical level of THI up to which the animal can tolerate HS and after which there is a significant productivity decline. Thus, in buffalo, although a decrease in milk production was perceptible after the maximum THI exceeded 72, the decrease was significant only when the THI exceeded 74.
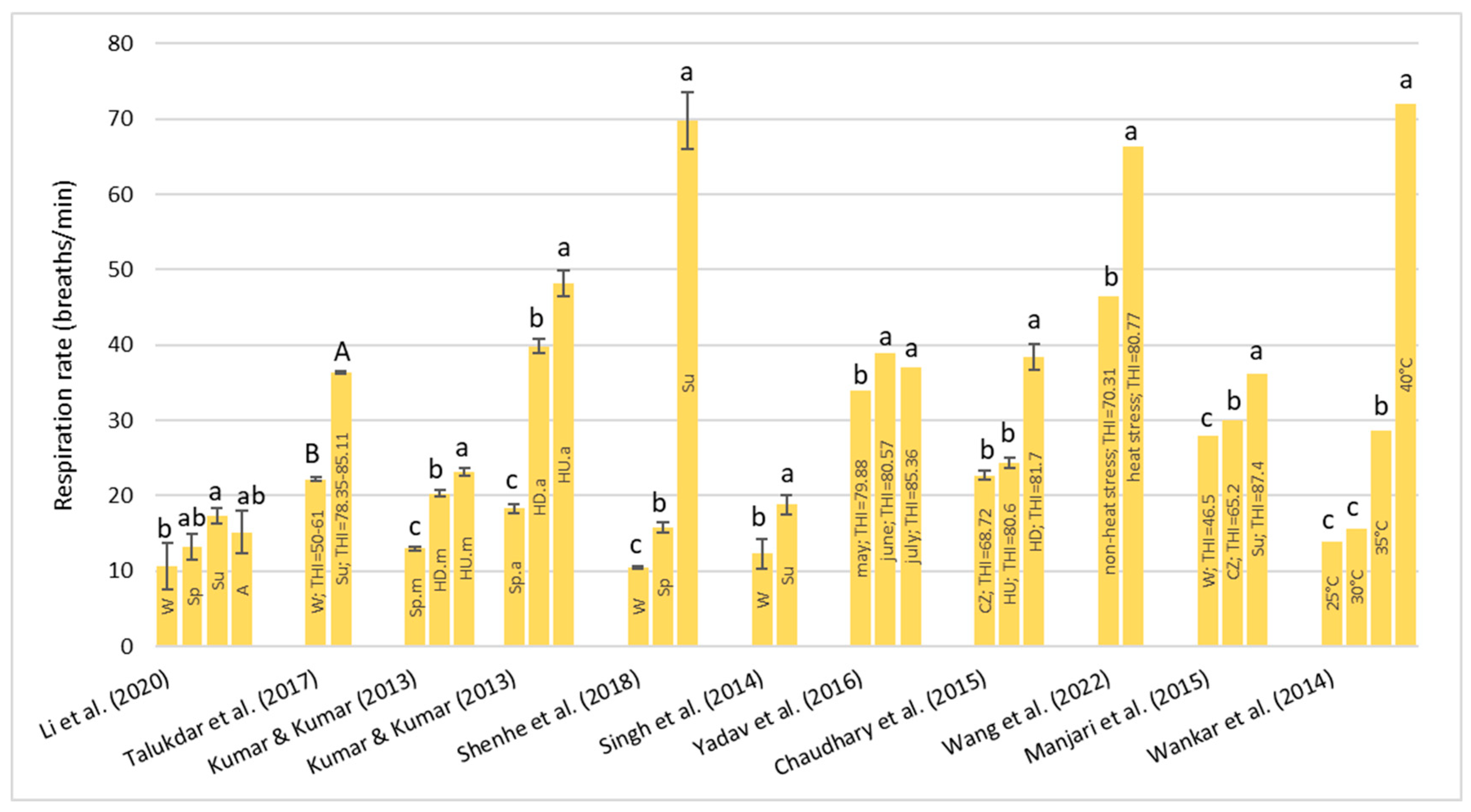
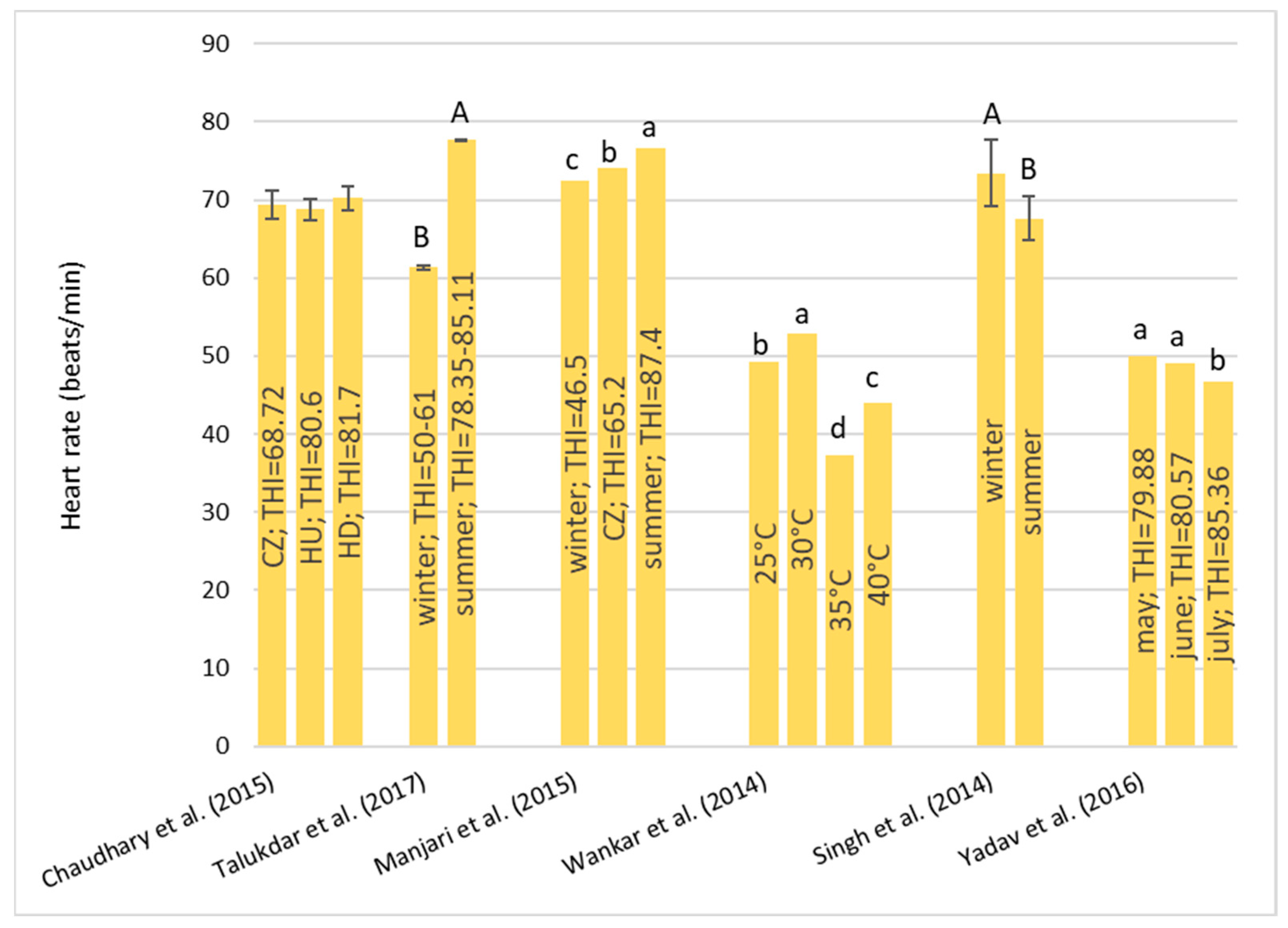
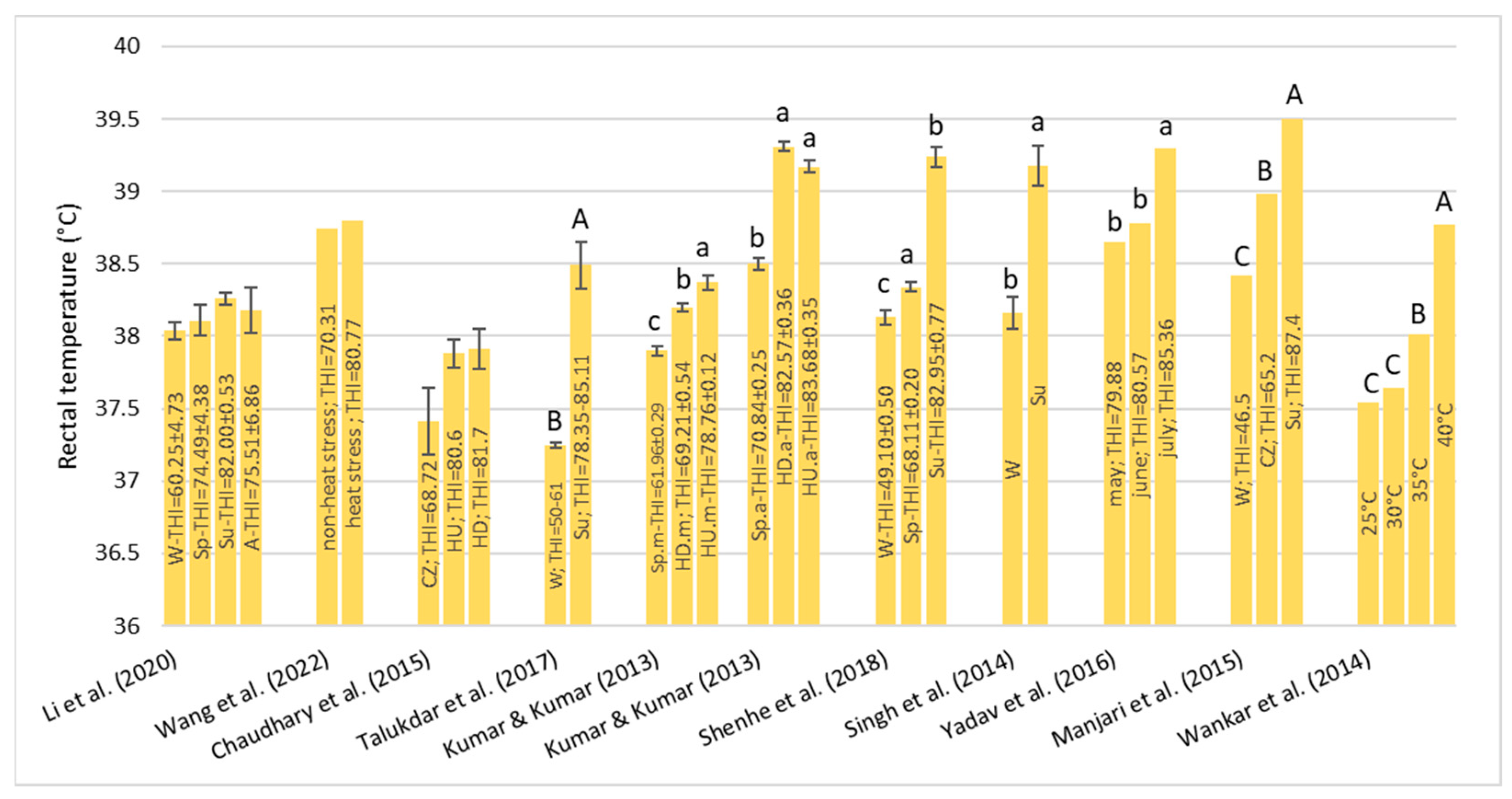
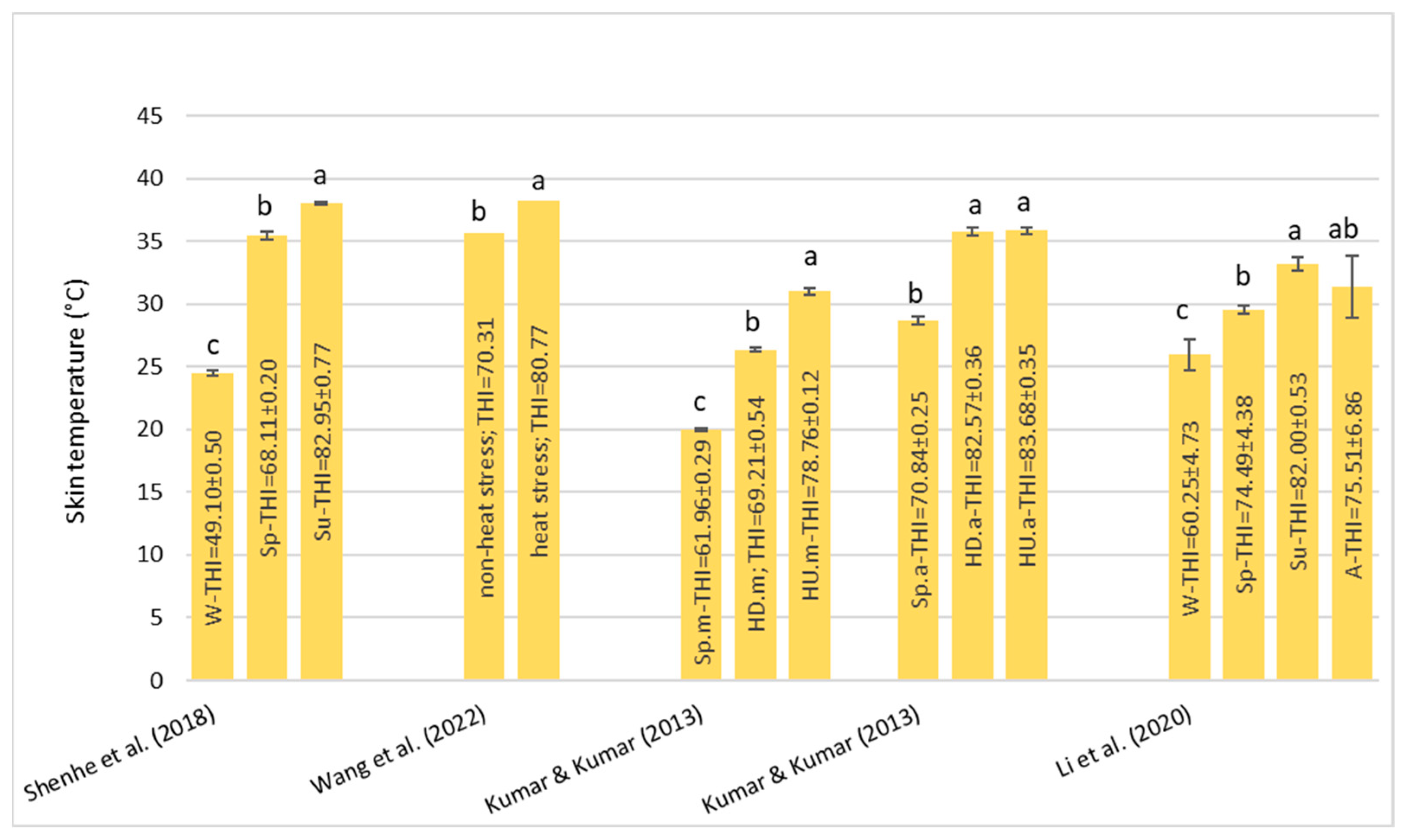
Under HS conditions, buffalos modify various physiological parameters, such as their rectal temperature (RT), respiratory rate (RR), heart rate (HR), skin temperature (ST), and body temperature (BT) [
36]; these are considered the main physiological parameters that can be quantified to evaluate the presence of stressful conditions, such as HS, in animals [
22]. It has been observed that buffalo did not modify their BT, HR, and RR compared to cattle in barns when the air temperature ranged from 30–33 °C with low (33–38%) and high (82–88%) humidity [
32]. However, if buffalos were in the scorching sun for a long time, the BT, HR, RR, and general discomfort of buffalo increased more rapidly than those of cows [
32]. Gudev et al. [
37] reported that the exposure of lactating buffalo to direct solar radiation (THI = 77.83) causes a significant increase in their RT and RR, showing that the heat load is greater than the body’s heat dissipation capacity. The same THI value did not induce significant changes in RT when buffalos were placed in the shade; although, maintenance of RT within the thermoneutral zone was achieved at the expense of a greater RR. A study carried out in Egypt [
38] on buffalos and cows reported that under the same rearing conditions (the air temperature was 30–33 °C), the mean BT of both species was the same (38.8 °C), while the mean RR was widely different (27.19 for the buffalo and the 46.69 for cows for respiratory acts for minute). Those authors reported that between 5 and 7 years of age, the RT significantly decreases by 0.90 °C in buffalo and 0.25 °C in cows and remains constant thereafter. The respiration rate also follows the same pattern as BT, with it tending to decrease with advancing age.
The symptoms of the buffalo at different THI ranges were reported by Dash et al. [
35]: with THI values below 72, there was no stress condition and the buffalos were in an optimal condition for reproduction and production; for the THI range between 72–78, there was a mild stress condition and there was an increase in RT and RR; for the THI range between 79–88, there was a moderate stress condition, RR and water intake were significantly increased, dry matter intake was decreased, and the ratio of forage to concentrate intake was decreased; and for the THI range between 89–98, there was a severe stress condition. In this case, excessive panting and restlessness were observed. Rumination and urination were lowered along with a negative impact on reproductive performance in the buffalo; a value of THI above 98 is an extreme stress condition and buffalo may also die.
From the studies examined, due to their peculiar physical characteristics, buffalo suffers from HS, especially in tropical areas. The threshold levels of the THI are higher than those reported for cows. Once these levels are exceeded, they react by modifying some clinical, reproductive, and production parameters.
5. Conclusions
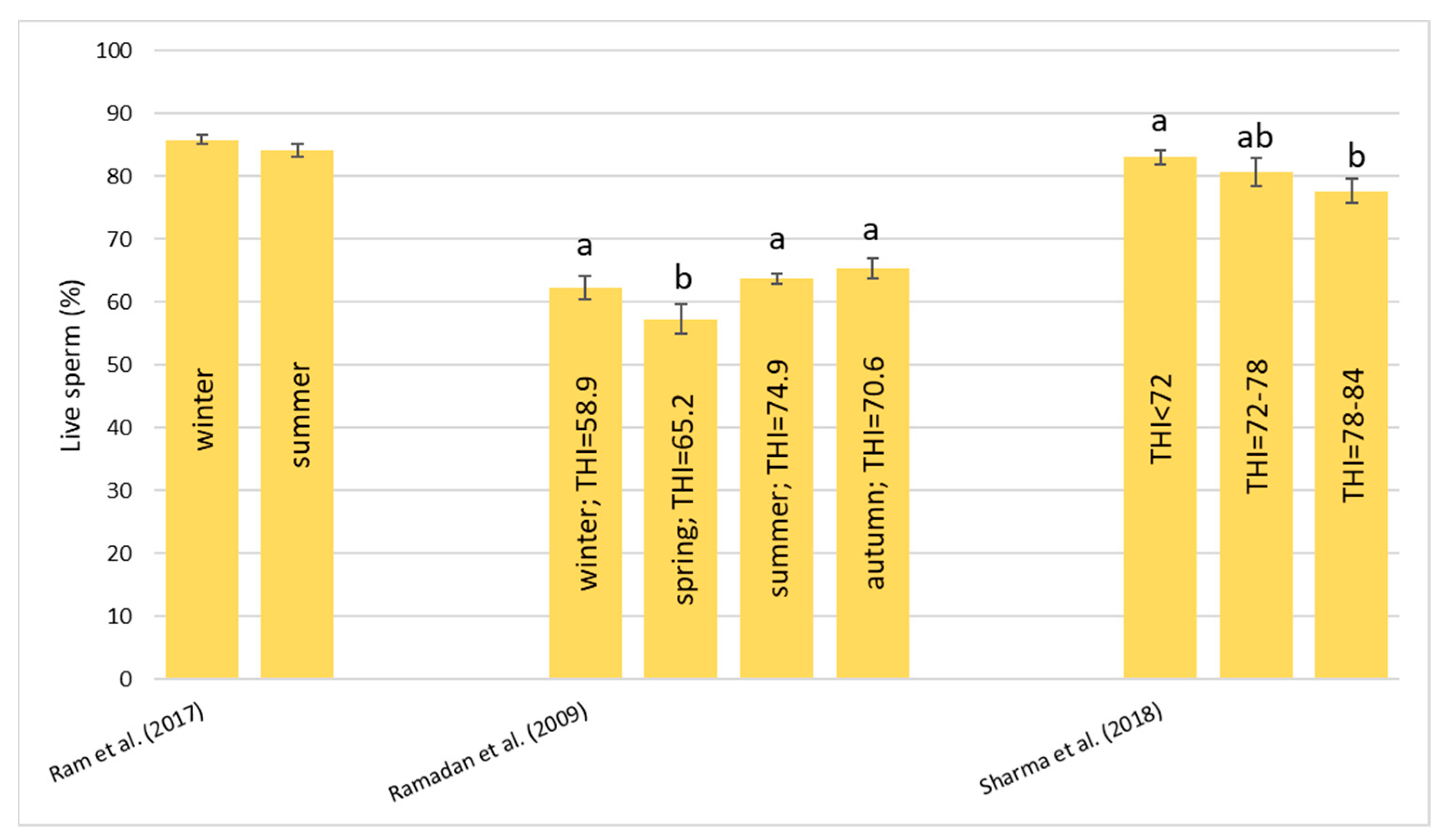
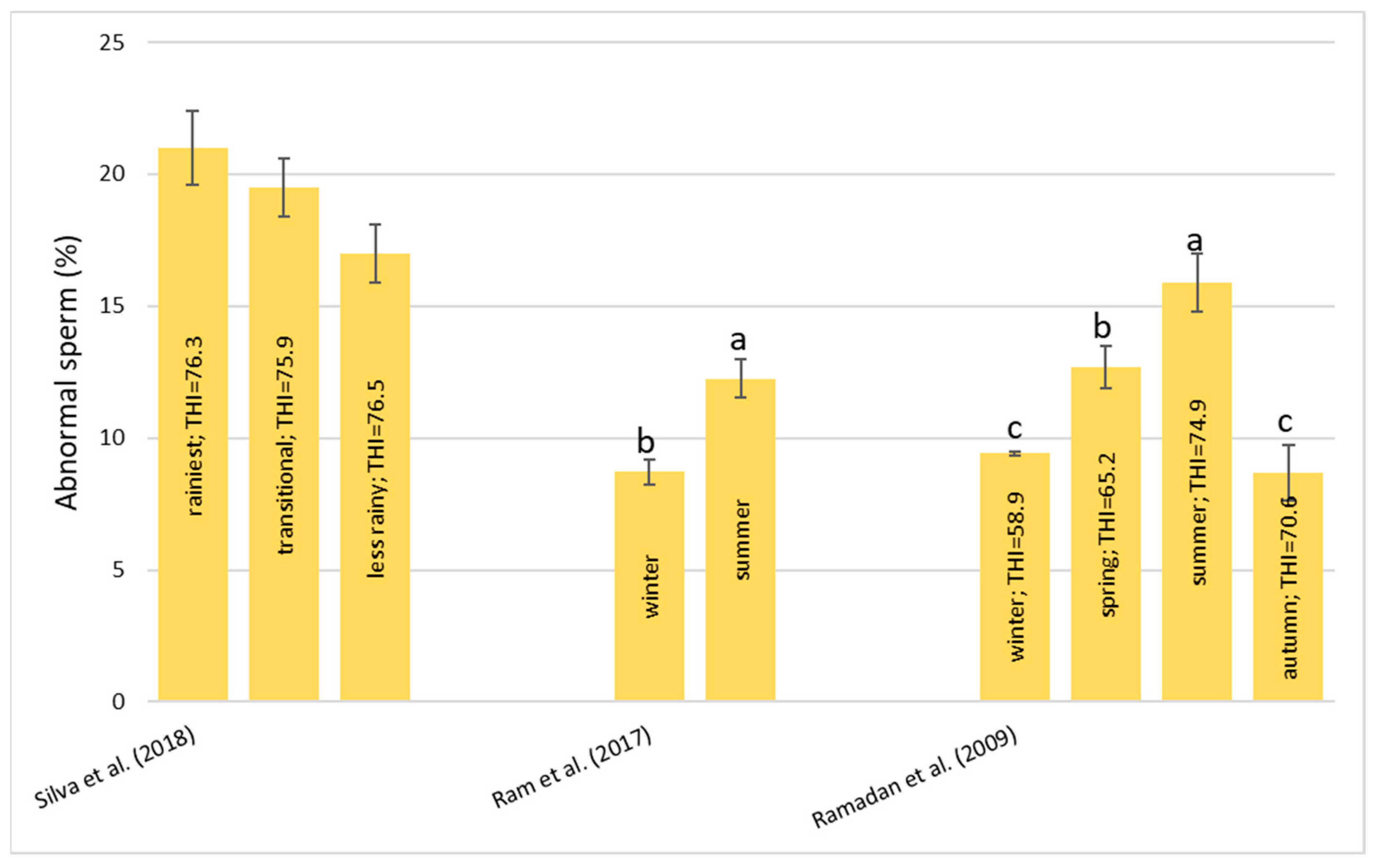
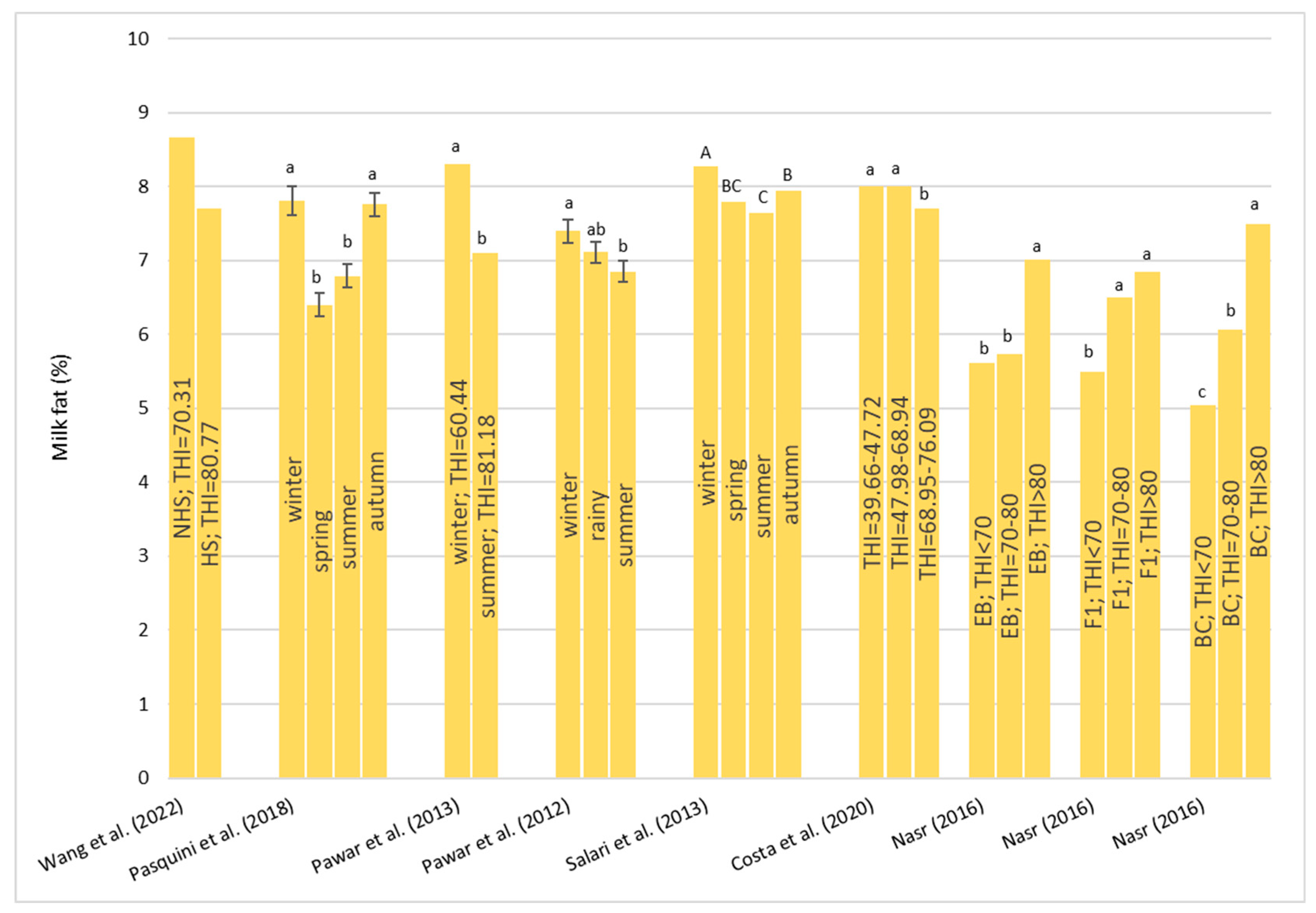
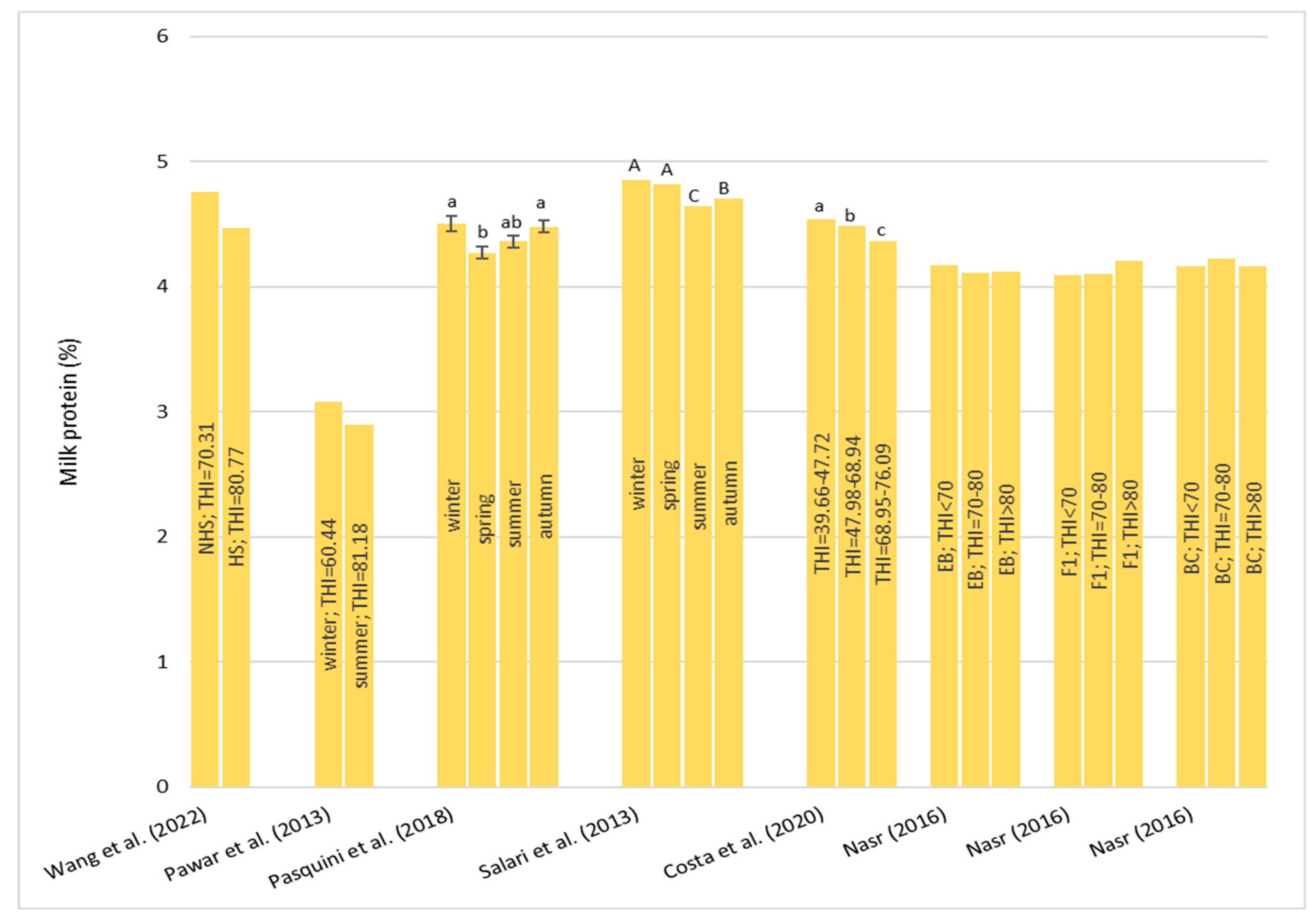
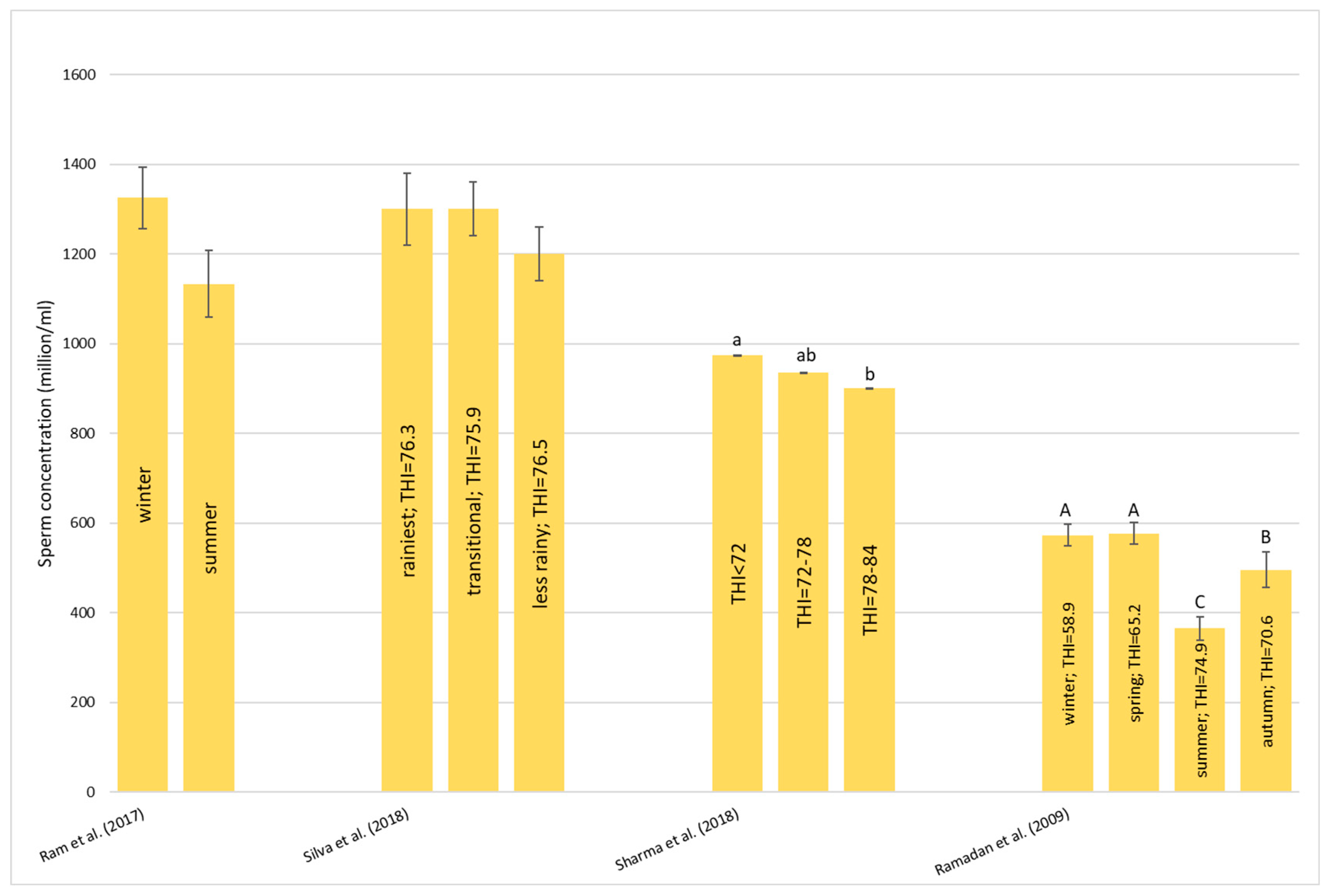
Heat stress causes a huge loss in MY and alters milk characteristics in dairy buffalo. HS also reduces the DMI which is directly co-responsible for the lower MY. The reduction in the DMI due to the increase in the THI is a known strategy to reduce the heat load that reduces the production of endogenous heat in an animal’s body which is produced by digestive and metabolic processes. Moreover, HS, in most cases, has an impact on milk quality through a reduction in fat, protein, and lactose and an increase in MUN, TBC, and SCC. Heat stress negatively affects physiological condition by increasing RR, HR, RT, and ST and reduces reproduction efficiency in both males and females by altering the quality of semen and reducing conception rates and the quality of oocytes, respectively. In males, even if HS does not affect ejaculate volumes, it causes a decrease in fertility due to its negative effect on sperm concentration, motility, viability, and defected spermatozoa. In females, HS causes difficulties in the induction of heat, a reduced lactation length, lower pregnancy rates, bad-quality oocytes, and an increase in the service period and calving interval.
Different approaches are used to mitigate the negative effects of HS on buffalo. Feeding management and the integration of diet with antioxidant substances can help the animal in fighting HS. In addition, the modification of their diet, with particular attention to fiber concentration and quality and protein quality, can be a useful strategy to mitigate the negative effects of HS.
Cooling is the first thing to carry out to keep buffalo cool. Between the cooling systems, the use of pools is the most effective. Wallowing can prevent increases in RT, RR, and HR by decreasing the heat load of the body by enforcing additional physical heat loss from the body. Wallowing also improves DMI, MY, and milk quality and the eating and social behavior of buffalo which, through wallowing, can demonstrate its typical species-specific behavior. Wallowing is more effective because besides evaporative heat loss, conductive and convective heat loss also prevail during wallowing. This is very important in buffalo because the low hair density on their skin helps in readily exchanging heat from their skin to water. In addition, the use of fans and sprinklers is effective in reducing the negative impact of HS on the physiology and MY of buffalo. This approach is increasingly being used as a replacement for pools, as pool management is much more complicated.
The data from the literature are almost always discordant due to the different sensitivities of the different breeds tested to the different environmental climatic characteristics and different levels of milk production. Most of the data come from studies carried out on buffalo reared under tropical and subtropical conditions. More studies to better understand physiological and metabolic responses to HS and the best mitigation strategies to be adopted for improving welfare and productivity are needed in buffalo breeds reared under temperate climate.