Assessing the Role of Lizards as Potential Pollinators of an Insular Plant Community and Its Intraspecific Variation
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Site and Species
2.2. Plant–Lizard Interactions
2.3. Representation of Species Interactions and Statistical Analyses
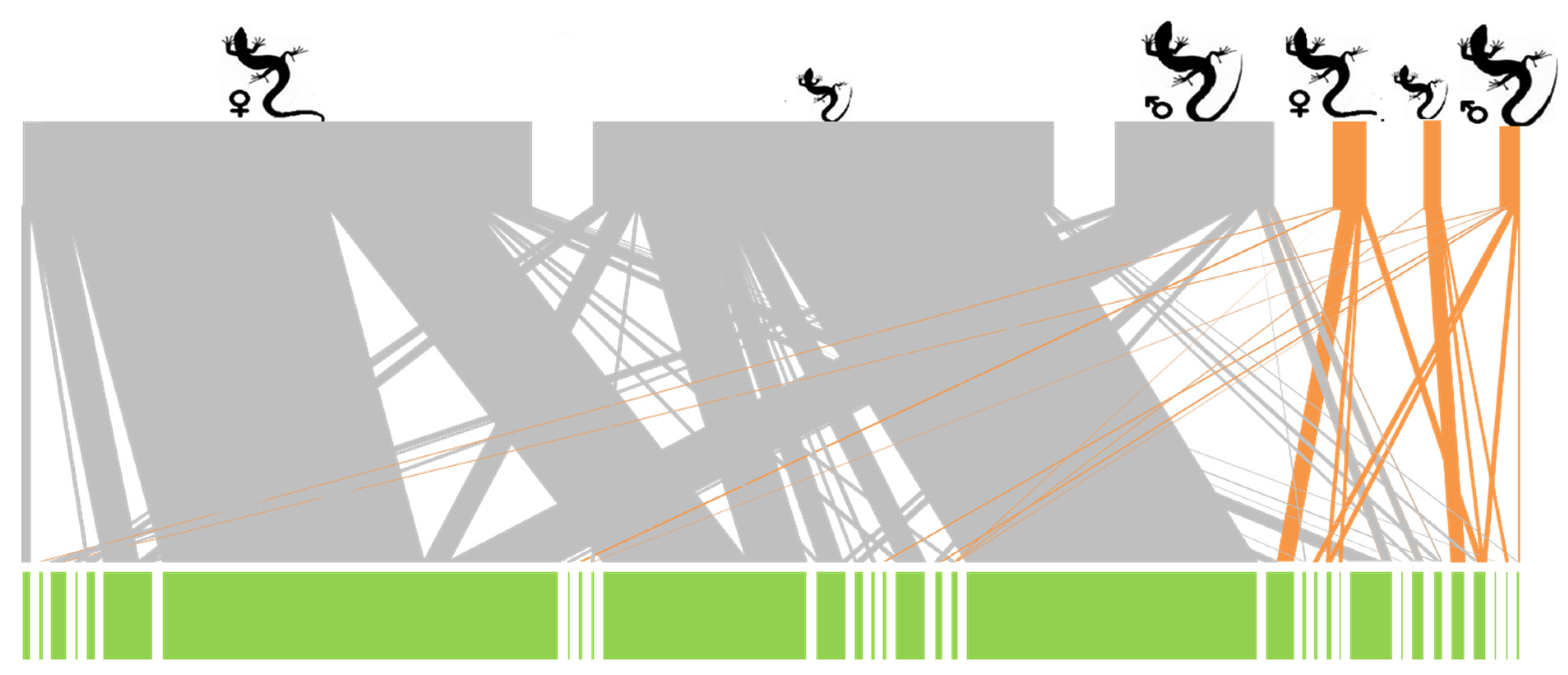
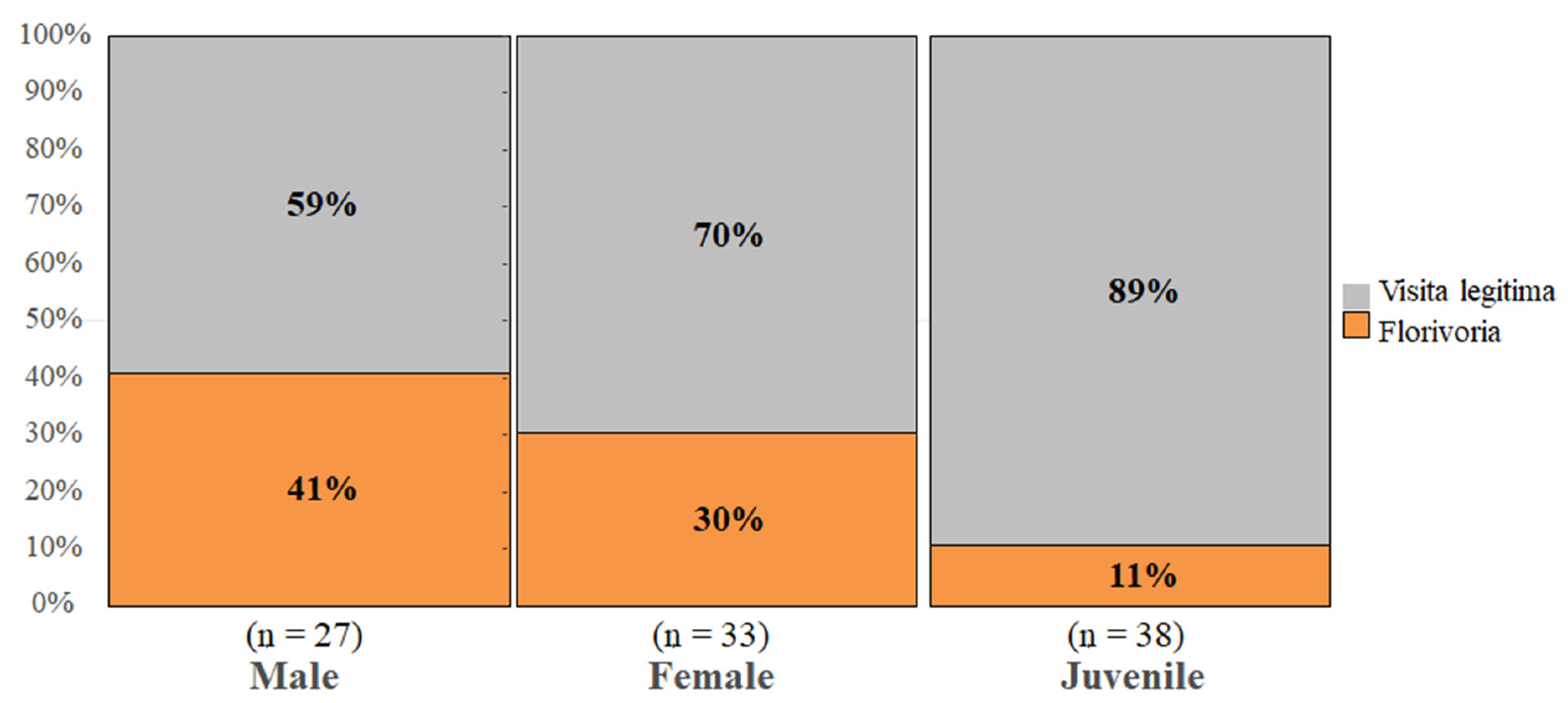
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Olesen, J.M.; Valido, A. Lizards as pollinators and seed dispersers: An island phenomenon. Trends Ecol. Evol. 2003, 18, 177–181. [Google Scholar] [CrossRef]
- Valido, A.; Nogales, M.; Medina, F.M. Fleshy fruits in the diet of canarian lizards Gallotia galloti (Lacertidae) in a xeric habitat of the island of Tenerife. J. Herpetol. 2003, 37, 741–747. [Google Scholar] [CrossRef] [Green Version]
- Galindo-Uribe, D.; Hoyos-Hoyos, J.M. Relaciones planta-herpetofauna: Nuevas perspectivas para la investigación en Colombia. Univ. Sci. 2007, 12, 9–34. [Google Scholar]
- Cooper, W.E.; Vitt, L.J. Distribution, extent, and evolution of plant consumption by lizards. J. Zool. 2002, 257, 487–517. [Google Scholar] [CrossRef] [Green Version]
- Traveset, A.; Sáez, E. Pollination of Euphorbia dendroides by lizards and insects: Spatio-temporal variation in patterns of flower visitation. Oecologia 1997, 111, 241–248. [Google Scholar] [CrossRef]
- Rodrigo, E.; Gallettu, M. Frugivoria e dispersão de sementes pelo lagarto teiú Tupinambis merianae (Reptilia: Teiidae). Pap. Avulsos Zool. 2004, 44, 91–97. [Google Scholar]
- Nogales, M.; Padilla, D.P.; Nieves, C.; Illera, J.C.; Traveset, A. Secondary seed dispersal systems, frugivorous lizards and predatory birds in insular volcanic badlands. J. Ecol. 2007, 95, 1394–1403. [Google Scholar] [CrossRef] [Green Version]
- Valido, A.; Olesen, J.M. Frugivory and seed dispersal by lizards: A global review. Front. Ecol. Evol. 2019, 7, 1394–1403. [Google Scholar] [CrossRef] [Green Version]
- Hervías-Parejo, S.; Nogales, M.; Guzmán, B.; Trigo, M.D.M.; Olesen, J.M.; Vargas, P.; Heleno, R.; Traveset, A. Potential role of lava lizards as pollinators across the Galápagos Islands. Integr. Zool. 2020, 15, 144–148. [Google Scholar] [CrossRef]
- Hervías-Parejo, S.; Heleno, R.; Rumeu, B.; Guzmán, B.; Vargas, P.; Olesen, J.M.; Traveset, A.; Vera, C.; Benavides, E.; Nogales, M. Small size does not restrain frugivory and seed dispersal across the evolutionary radiation of Galápagos lava lizards. Curr. Zool. 2019, 65, 353–361. [Google Scholar] [CrossRef] [Green Version]
- Cozien, R.J.; Niet, T.; Johnson, S.D.; Steenhuisen, S. Saurian surprise: Lizards pollinate south Africa’s enigmatic hidden flower. Bull. Ecol. Soc. Am. 2019, 100, 1–3. [Google Scholar] [CrossRef] [Green Version]
- Wester, P. First observations of nectar-drinking lizards on the African mainland. Plant Ecol. Evol. 2019, 152, 78–83. [Google Scholar] [CrossRef]
- Morando, M.; Avila, L.J. Lizards of Patagonia. Diversity, Systematics, Biogeography and Biology of the Reptiles at the End of the World, 1st ed.; Springer: Charm, Switzerland, 2020. [Google Scholar]
- Rodríguez-Rodríguez, M.C.; Siverio, F. Gallotia galloti (Canary Lizard). Nectarivory Hardly Nat. Hist. Notes 2012, 43, 333–334. [Google Scholar]
- Bègue, J.F.; Sanchez, M.; Micheneau, C.; Fournel, J. New record of day geckos feeding on orchid nectar in Reunion Island: Can lizards pollinate orchid species? Herpetol. Notes 2014, 7, 689–692. [Google Scholar]
- Jaca, J.; Nogales, M.; Traveset, A. Reproductive success of the Canarian Echium simplex (Boraginaceae) mediated by vertebrates and insects. Plant Biol. 2019, 21, 216–226. [Google Scholar] [CrossRef] [PubMed]
- MacArthur, R.H.; Diamond, J.M.; Karr, J.R. Density compensation in island faunas. Ecology 1972, 53, 330–342. [Google Scholar] [CrossRef]
- Cox, G.W.; Ricklefs, R.E. Species diversity and ecological release in Caribbean land bird faunas. Oikos 1977, 28, 113. [Google Scholar] [CrossRef]
- Justicia Correcher, E.; Hervías-Parejo, S.; Ruíz de Ybáñez Carnero, R.; Sauroy-Toucouère, S.; Traveset, A. Environmental and morphological drivers of mutualistic plant–lizard interactions: A global review. Ecography 2023, 2, 1–11. [Google Scholar] [CrossRef]
- Best, T.L.; Pfaffenberger, G.S. Age and sexual variation in the diet of collared lizards (Crotaphytus collaris). Southwest. Assoc. Nat. 1987, 32, 415. [Google Scholar] [CrossRef]
- Pérez-Quintero, J.C.; Rubio-Garcia, J.C. Alimentación de la lagartija colilarga, Psammodromus algirus (sauria, lacertidae), en el litoral de Huelva (so España). Doñana Acta Vert. 1997, 24, 3–26. [Google Scholar]
- Mateo, J.A.; Geniez, P.; Hernández-Acosta, C.N.; López-Jurado, L.F. ¿Realmente importa tanto el tamaño? I: La dieta de las dos especies del género Tarentola de la Isla de Raso (Cabo Verde). Boletín Asoc. Herpetológica Española 2016, 27, 47–51. [Google Scholar]
- Jonathan, B.; Katleen, H.; Bieke, V.; Herrel, A.; Grbac, I.; Van Damme, R. Functional and ecological relevance of intraspecific variation in body size and shape in the lizard Podarcis melisellensis (Lacertidae). Biol. J. Linn. Soc. 2008, 94, 251–264. [Google Scholar]
- Butler, M.A.; Sawyer, S.A.; Losos, J.B. Sexual dimorphism and adaptive radiation in Anolis lizards. Nature 2007, 447, 202–205. [Google Scholar] [CrossRef]
- Abraham, L.; Aurelio, R.B.; Raciel, C.E. Intraspecific variation in a lizard from the Central Mexican Plateau: Intersexual differences in size and shape explored. Biol. J. Linn. Soc. 2020, 131, 656–666. [Google Scholar]
- Pough, F.H. Lizard energetics and diet. Ecology 1973, 54, 837–844. [Google Scholar] [CrossRef] [Green Version]
- Garrison, G.; Phillips, M.; Eifler, M.; Eifler, D. Intraspecific variation in opportunistic use of trophic resources by the lizard Ameiva corax (Squamata: Teiidae). Amphib. Reptil. 2016, 37, 331–334. [Google Scholar] [CrossRef]
- Durtsche, R.D. Ontogenetic plasticity of food habits in the Mexican spiny-tailed iguana, Ctenosaura pectinata. Oecologia 2000, 124, 185–195. [Google Scholar] [CrossRef]
- Fialho, R.F.; Rocha, C.F.D.; Vrcibradic, D. Feeding ecology of Tropidurus torquatus: Ontogenetic shift in plant consumption and seasonal trends in diet. J. Herpetol. 2000, 34, 325. [Google Scholar] [CrossRef]
- Fuster, F.; Traveset, A. Importance of intraspecific variation in the pollination and seed dispersal functions of a double mutualist animal species. Oikos 2020, 129, 106–116. [Google Scholar] [CrossRef]
- Whitaker, A.H. The roles of lizards in New Zealand plant reproductive strategies. N. Z. J. Bot. 1987, 25, 315–328. [Google Scholar] [CrossRef]
- Hansen, D.M.; Kiesbüy, H.C.; Jones, C.G.; Müller, C.B. Positive indirect interactions between neighboring plant species via a lizard pollinator. Am. Nat. 2007, 169, 534–542. [Google Scholar] [CrossRef] [PubMed]
- Gomes, V.G.N.; Meiado, M.V.; Quirino, Z.G.M.; de Araujo, H.F.P.; Machado, I.C. Synchronous fruiting and common seed dispersers of two endemic columnar cacti in the Caatinga, a dry forest in Brazil. Plant Ecol. 2017, 218, 1325–1338. [Google Scholar] [CrossRef]
- Fuster, F.; Traveset, A. Evidence for a double mutualistic interaction between a lizard and a Mediterranean gymnosperm, Ephedra fragilis. AoB Plants 2019, 11, plz001. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Traveset, A. Consecuencias de la ruptura de mutualismos planta-animal para la distribución de especies vegetales en las Islas Baleares. Rev. Chil. De Hist. Nat. 2002, 75, 117–126. [Google Scholar] [CrossRef] [Green Version]
- Salvador, A. Lagartija balear—Podarcis lilfordi. In Enciclopedia Virtual de los Vertebrados Españoles; Salvador, A., Marco, A., Eds.; Museo Nacional de Ciencias Naturales: Madrid, Spain, 2015. [Google Scholar]
- Eisentraut, M. Die Eidechsen der Spanischen Mittelmeerinseln und ihre Rassenaufspaltung im Lichte der Evolution. Mitt. Zool. Mus. Berl. 1949, 26, 1–225. [Google Scholar]
- Pérez-Mellado, V.; Corti, C. Dietary adaptations and herbivory in lacertid lizards of the genus Podarcis from western Mediterranean islands (Reptilia. Sauria). Bonn. Zool. Beiträge 1993, 44, 193–220. [Google Scholar]
- Saez, E.; Traveset, A. Fruit and nectar feeding by Podarcis lilfordi (Lacertidae) on Cabrera Archipelago (Balearic Islands). Herpetol. Rev. 1995, 26, 121–123. [Google Scholar]
- Traveset, A. La lagartija balear, una eficaz polinizadora y dispersante de plantas. Quercus 1997, 139, 20–22. [Google Scholar]
- Pérez-Mellado, V.; Traveset, A. Relationships between plants and Mediterranean lizards. Nat. Croat. Period. Musei Hist. Nat. Croat. 1999, 8, 275–285. [Google Scholar]
- Pérez-Mellado, V.; Cortázar, G.; López-Vicente, M.; Perera, A.; Sillero, N. Interactions between the Balearic lizard Podarcis lilfordi and the plant Dracunculus muscivorus. Amphib. Reptil. 2000, 21, 223–226. [Google Scholar]
- Pérez-Mellado, V.; Perera, A.; Cortázar, G. La lagartija balear, Podarcis lilfordi (Günther, 1874) de l’illa d’en Colom, Parc Natural de s’albufera des Grau (Menorca). Situación actual y estado de conservación. Butll. Cient. Espais Prot. Balears 2003, 1, 23–34. [Google Scholar]
- Traveset, A.; Riera, N. Disruption of a plant-lizard seed dispersal system and its ecological effects on a threatened endemic plant in the Balearic Islands. Conserv. Biol. 2005, 19, 421–431. [Google Scholar] [CrossRef] [Green Version]
- Salvador, A. Materiales para una “Herpetofauna Balearica”. 5. Las salamanquesas y tortugas del archipiélago de Cabrera. Doñana Acta Vert. 1978, 5, 5–17. [Google Scholar]
- Pérez-Mellado, V. Estudio ecológico de la Lagartija Balear Podarcis lilfordi (Günther, 1874) en Menorca. Rev. De Menorca 1989, 80, 455–511. [Google Scholar]
- Pérez-Mellado, V.; Ortega, F.; Martín-García, S.; Perera, A.; Cortázar, G. Pollen load and transport by the insular lizard, Podarcis lilfordi (Squamata, Lacertidae) in coastal islets of Menorca (Balearic Islands, Spain). Isr. J. Ecol. Evol. 2000, 46, 193–200. [Google Scholar]
- Gil, M.J.; Guerrero, F.; Pérez-Mellado, V. Seasonal variation in diet composition and prey selection in the Mediterranean gecko Tarentola mauritanica. Isr. J. Ecol. Evol. 1994, 40, 61–74. [Google Scholar]
- Hódar, J.A.; Pleguezuelos, J.M. Diet of the moorish gecko Tarentola mauritanica in an arid zone of south-eastern Spain. Herpetol. J. 1999, 9, 29–32. [Google Scholar]
- Dormann, C.; Fründ, J.; Bluthgen, N.; Gruber, B. Indices, graphs and null models: Analyzing bipartite ecological networks. Open Ecol. J. 2009, 2, 7–24. [Google Scholar] [CrossRef]
- Warnes, G.R.; Bolker, B.; Lumley, T.; Johnson, R.C. Contributions from R.C. Johnson are Copyright SAIC-Frederick, Inc. Funded by the Intramural Research Program, of the NIH, National Cancer Institute and Center for Cancer Research under NCI Contract NO1-CO-12400. gmodels: Various R Programming Tools for Model Fitting. R Package Version 2.18.1.1. 2022. Available online: https://CRAN.R-project.org/package=gmodels (accessed on 13 October 2022).
- Cardona, C.; Capó, M. First insular population of the Critically Endangered Cistus heterophyllus ssp. carthaginensis on Cabrera Archipelago National Park (Balearic Islands, Spain). Biodivers. Conserv. 2023, 32, 811–820. [Google Scholar]
- Moreno, J.C. Lista Roja 2008 de la Flora Vascular Española; Dirección General de Medio Natural y Política Forestal: Madrid, Spain, 2008; p. 86. [Google Scholar]
- Vicente, M.J.; Martínez-Sánchez, J.J. La jara de Cartagena (Cistus heterophyllus), una especie en peligro. In Estado Actual de Conocimientos, 1st ed.; Universidad Politécnica de Cartagena: Cartagena, Spain, 2018. [Google Scholar]
- Cooper, W.E.; Pérez-Mellado, V. Location of fruit using only airborne odor cues by a lizard. Physiol. Behav. 2001, 74, 339–342. [Google Scholar] [CrossRef]
- Cooper, W.E.; Pérez-Mellado, V. Food chemical cues elicit general and population-specific effects on lingual and biting behaviors in the lacertid lizard Podarcis lilfordi. J. Exp. Zool. 2001, 290, 207–217. [Google Scholar] [CrossRef] [PubMed]
- Cooper, W.E.; Pérez-Mellado, V.; Vitt, L.; Budzinsky, B. Behavioral responses to plant toxins by two omnivorous lizard species. Physiol. Behav. 2002, 76, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Cooper, W.E.; Pérez-Mellado, V.; Vitt, L.J. Lingual and biting responses to selected lipids by the lizard Podarcis lilfordi. Physiol. Behav. 2002, 75, 237–241. [Google Scholar] [CrossRef]
- Cooper, J.W.E.; Pérez-Mellado, V.; Vitt, L.J. Responses to Major Categories of Food Chemicals by the Lizard Podarcis lilfordi. J. Chem. Ecol. 2002, 28, 709–720. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Rica, J.P. Contribución al estudio de la biología de los gecónidos ibéricos (Rept., Sauria). Publ. Centro Piren. Biol. Exp. 1974, 5, 1–291. [Google Scholar]
- Valverde, J.A. Estructura de una comunidad mediterránea de vertebrados terrestres. In Monografías de Ciencias Moderna 76, 1st ed.; CSIC: Madrid, Spain, 1967. [Google Scholar]
- Pérez-Mellado, V. Tarentola mauritanica (Moorish Gecko) behavior. Herpetol. Rev. 1994, 25, 68–69. [Google Scholar]
- Hódar, J.; Pleguezuelos, J.; Villafranca, C.; Fernández-Cardenete, J. Foraging mode of the Moorish gecko Tarentola mauritanica in an arid environment: Inferences from abiotic setting, prey availability and dietary composition. J. Arid. Environ. 2006, 65, 83–93. [Google Scholar] [CrossRef]
- Gil, M.J.; Pérez-Mellado, V.; Guerrero, F. Eine vergleichende studie des nahrungswerb von Tarentola mauritanica (Reptilia: Gekkonidae) in habitaten auf dem festland und auf inseln. Sauria 1993, 15, 9–17. [Google Scholar]
- Cooper, W.E., Jr.; Pérez-Mellado, V.; Hawlena, D. Foraging by the omnivorous lizard Podarcis lilfordi: Effects of nectivory in an ancestrally insectivorous active forager. J. Herpetol. 2014, 48, 203–209. [Google Scholar] [CrossRef]
- Hernández-Teixidor, D.; Díaz-Luis, N.; Medina, F.M.; Nogales, M. First record of geckos visiting flowers in the Palaearctic Ecozone. Curr. Zool. 2020, 66, 447–448. [Google Scholar] [CrossRef] [Green Version]
- Hansen, D.M.; Müller, C.B. Reproductive ecology of the endangered enigmatic Mauritian endemic Roussea simplex (Rousseaceae). Int. J. Plant Sci. 2009, 170, 42–52. [Google Scholar] [CrossRef]
- Kaiser-Bunbury, C.N.; Memmott, J.; Müller, C.B. Community structure of pollination webs of Mauritian heathland habitats. Perspect. Plant Ecol. Evol. Syst. 2009, 11, 241–254. [Google Scholar] [CrossRef] [Green Version]
- Bissessur, P.; Bunsy, Y.; Baiderd, C.; Florensa, F.B.V. Non-intrusive systematic study reveals mutualistic interactions between threatened island endemic species and points to more impactful conservation. J. Nat. Conserv. 2019, 49, 108–117. [Google Scholar] [CrossRef]

| Plant Species | Interaction Type | Known on Cabrera Gran | Known Anywhere |
|---|---|---|---|
| Apiaceae | Unknown (pollen samples) | No | No |
| Asparagus horridus | Legitimate visit | No | No |
| Asphodelus fistulosus | Florivory | No | No |
| Astragalus balearicus | Florivory | No | No |
| Calendula arvensis | Legitimate visit | No | Aire, Rei, Sanitja [47] |
| Cakile maritima | Florivory | No | No |
| Centrathus calcitrapae | Legitimate visit | No | Na Redona [Islet food-web team; pers. observ.] |
| Cistus heterophyllus | Legitimate visit | No | No |
| Cistus monspeliensis | Legitimate visit | No | No |
| Daucus carota | Legitimate visit | No | No |
| Echium parviflorum | Legitimate visit | No | No |
| Erodium malacoides | Florivory | No | No |
| Ephedra fragilis | Legitimate visit | Yes [34] | Na Redona, Dragonera [Islet food-web team; pers. observ.]; [34] |
| Euphorbia dendroides | Legitimate visit | Yes [5] | Na Redona [Islet food-web team; pers. observ.] |
| Euphorbia segetalis | Legitimate visit | No | No |
| Fumana ericoides | Legitimate visit | Yes [41] | No |
| Geranium molle | Legitimate visit | No | Na Redona [Islet food-web team; pers. observ.] |
| Helichrysum stoechas | Florivory | No | Estell de dos Colls [41] |
| Lavatera arborea | Legitimate visit | No | Xes Rates, Xapat Gros [41] |
| Lavatera cretica | Florivory | No | No |
| Linum sp. | Florivory | No | No |
| Lobularia maritima | Legitimate visit | No | No |
| Lomelosia cretica | Legitimate visit | No | No |
| Lysimachia arvensis | Legitimate visit | No | No |
| Medicago arborea | Legitimate visit | No | Estell de dos Colls, Na Redona [41]; [Islet food-web team; pers. observ.] |
| Medicago littoralis | Florivory | No | Aire, Rei, Sanitja [47] |
| Melilotus sp. | Florivory | No | No |
| Narcissus tazetta | Legitimate visit | No | Rei [47] |
| Papaver somniferum | Florivory | No | No |
| Paronychia capitata | Legitimate visit | No | No |
| Plumbago sp. | Legitimate visit | No | No |
| Rubia peregrina | Legitimate visit | No | No |
| Ruta graveolens | Legitimate visit | No | No |
| Rhamnus ludovici-salvatoris | Legitimate visit | No | No |
| Salvia rosmarinus | Legitimate visit | No | Menorca [45] |
| Scorpiurus sulcatus | Florivory | No | No |
| Silene vulgaris | Legitimate visit | No | No |
| Sinapis arvensis | Florivory | No | No |
| Sonchus tenerrimus | Florivory | No | No |
| Teucrium capitatum | Legitimate visit | No | No |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Romero-Egea, V.; Robles, C.; Traveset, A.; Del Rio, L.; Hervías-Parejo, S. Assessing the Role of Lizards as Potential Pollinators of an Insular Plant Community and Its Intraspecific Variation. Animals 2023, 13, 1122. https://doi.org/10.3390/ani13061122
Romero-Egea V, Robles C, Traveset A, Del Rio L, Hervías-Parejo S. Assessing the Role of Lizards as Potential Pollinators of an Insular Plant Community and Its Intraspecific Variation. Animals. 2023; 13(6):1122. https://doi.org/10.3390/ani13061122
Chicago/Turabian StyleRomero-Egea, Víctor, Cristina Robles, Anna Traveset, Laura Del Rio, and Sandra Hervías-Parejo. 2023. "Assessing the Role of Lizards as Potential Pollinators of an Insular Plant Community and Its Intraspecific Variation" Animals 13, no. 6: 1122. https://doi.org/10.3390/ani13061122






