Pup Recruitment in a Eusocial Mammal—Which Factors Influence Early Pup Survival in Naked Mole-Rats?
Simple Summary
Abstract
1. Introduction
| Effect | Species | Condition | Sources | |
|---|---|---|---|---|
| Pup birth body mass | + | Fukomys mechowii | captive | [1] |
| + | Ctenomys talarum | captive | [2] | |
| + | Peromyscus californicus | captive | [38] | |
| + | Oryctolagus cuniculus | semi-captive | [6] | |
| Pup age | + | Leptonychotes weddellii | wild | [39] |
| + | Fukomys mechowii | captive | [1] | |
| + | Meriones unguiculatus | captive | [31] | |
| + | Mus musculus L. | wild | [40] | |
| + | Oryctolagus cuniculus | semi-captive | [6] | |
| + | Heterocephalus glaber | captive | [41,42] | |
| Maternal body mass | + | Clethrionomys glareolus | captive | [4] |
| + | Oryctolagus cuniculus | semi-captive | [6] | |
| n.e. | Octodon degus | captive | [43] | |
| n.e. | Sigmodon hispidus | wild/captive | [44] | |
| n.e. | Peromyscus polionotus | captive | [10] | |
| Maternal age | + | Leptonychotes weddellii | wild | [39,45] |
| − | Heterocephalus glaber | captive | [41] | |
| Maternal behavior | + | Peromyscus polionotus | captive | [10] |
| + | Crocuta crocuta | wild | [3] | |
| Colony size | + | Marmota marmota | wild | [46] |
| + | Lycaon pictus | wild | [5] | |
| + | Suricata suricatta | wild | [47] | |
| − | Meriones unguiculatus | captive | [7] | |
| − | Arctocephalus gazella | wild | [48] | |
| − | Suricata suricatta | wild | [47] | |
| n.e. | Fukomys mechowii | wild | [1] | |
| n.e. | Meriones unguiculatus | wild | [49] | |
| n.e. | Canis lycaon | wild | [50] | |
| n.e. | Meles meles | wild | [51] | |
| n.e. | Myotis myotis | wild | [52] | |
| n.e. | Mirounga leonina | wild | [53] | |
| Litter size | + | Rattus norvegicus | captive | [54] |
| − | Meriones unguiculatus | captive | [31] | |
| n.e. | Fukomys mechowii | wild | [1] | |
| n.e. | Clethrionomys glareolus | captive | [4] | |
| n.e. | Octodon degus | captive | [43] | |
| n.e. | Mus musculus | captive | [55] | |
| n.e. | Peromyscus leucopus | wild | [56] | |
| n.e. | Marmota marmota | wild | [46] | |
| n.e. | Sus scrofa domesticus | captive | [57] | |
| n.e. | Canis lycaon | wild | [50] | |
| Ambient temperature | + | Meriones unguiculatus | captive | [49] |
| n.e. | Heterocephalus glaber | captive | [58] | |
| n.e. | Rhabdomys pumilio | captive | [59] | |
| Handling/disturbance | − | Cryptomys hottentotus | captive | [60] |
| n.e. | Heterocephalus glaber | captive | [37] | |
| n.e. | Rattus norvegicus | captive | [61] | |
| n.e. | Mus musculus | captive | [55] |
2. Materials and Methods
2.1. Study Species
2.2. Housing Conditions
2.3. Data Collection
2.4. Statistical Analysis
3. Results
3.1. Description of Early Pup Survival
3.2. Predictors of Early Pup Survival
3.2.1. Effect of Individual Characteristics
3.2.2. Effect of Maternal Characteristics
3.2.3. Effect of Social Characteristics
3.2.4. Effect of Environmental Characteristics
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Scharff, A.; Begall, S.; Grütjen, O.; Burda, H. Reproductive characteristics and growth of Zambian giant mole-rats, Cryptomys mechowi (Rodentia: Bathyergidae). Mammalia 1999, 63, 217–230. [Google Scholar] [CrossRef]
- Zenuto, R.R.; Antinuchi, C.D.; Busch, C. Bioenergetics of Reproduction and Pup Development in a Subterranean Rodent (Ctenomys talarum). Physiol. Biochem. Zool. 2002, 75, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Hofer, H.; East, M.L. Behavioral processes and costs of co-existence in female spotted hyenas: A life history perspective. Evol. Ecol. 2003, 17, 315–331. [Google Scholar] [CrossRef]
- Jonsson, P.; Agrell, J.; Koskela, E.; Mappes, T. Effects of litter size on pup defence and weaning success of neighbouring bank vole females. Can. J. Zool. 2002, 80, 1–5. [Google Scholar] [CrossRef]
- Creel, S.; Creel, N.M. Opposing effects of group size on reproduction and survival in African wild dogs. Behav. Ecol. 2015, 26, 1414–1422. [Google Scholar] [CrossRef]
- Rödel, H.; Starkloff, A.; Seltmann, M.W.; Prager, G.; Von Holst, D. Causes and predictors of nest mortality in a European rabbit population. Mamm. Biol. 2009, 74, 198–209. [Google Scholar] [CrossRef]
- Scheibler, E.; Weinandy, R.; Gattermann, R. Social factors affecting litters in families of Mongolian gerbils, Meriones unguiculatus. Folia Zool. 2005, 54, 61. [Google Scholar]
- Hofer, H.; East, M. Population dynamics, population size, and the commuting system of Serengeti spotted hyenas. In Serengeti II: Dynamics, Management, and Conservation of an Ecosystem; Sinclaire, A.R.E., Arcese, P., Eds.; University of Chicago Press: Chicago, IL, USA, 1995; p. 332. [Google Scholar]
- Maestripieri, D.; Mateo, J.M. Maternal Effects in Mammals; University of Chicago Press: Chicago, IL, USA, 2009. [Google Scholar]
- Margulis, S.W.; Nabong, M.; Alaks, G.; Walsh, A.; Lacy, R.C. Effects of early experience on subsequent parental behaviour and reproductive success in oldfield mice, Peromyscus polionotus. Anim. Behav. 2005, 69, 627–634. [Google Scholar] [CrossRef]
- Hofer, H.; Benhaiem, S.; Golla, W.; East, M.L. Trade-offs in lactation and milk intake by competing siblings in a fluctuating environment. Behav. Ecol. 2016, 27, 1567–1578. [Google Scholar] [CrossRef]
- Hood, W.R.; Kessler, D.S.; Oftedal, O.T. Milk composition and lactation strategy of a eusocial mammal, the naked mole-rat. J. Zool. 2014, 293, 108–118. [Google Scholar] [CrossRef]
- Jennions, M.D. Cooperative breeding in mammals. Trends Ecol. Evol. 1994, 9, 89–93. [Google Scholar] [CrossRef] [PubMed]
- Ciszek, D. Behavior of the Naked Mole-Rat (Heterocephalus glaber), with Emphasis on Factors Related to the Attainment of Breeding Status. Ph.D. Thesis, University of Michigan, Ann Arbor, MI, USA, 2001. [Google Scholar]
- Tardif, S.D.; Moehlman, P.D.; Hofer, H.; Asa, C.S.; Creel, S.R.; Waser, P.M.; Lucas, J.R.; Getz, L.L.; Carter, C.S.; Roberts, R.L.; et al. Cooperative Breeding in Mammals; Solomon, N.G., French, J.A., Eds.; Cambridge University Press: Cambridge, UK, 1997. [Google Scholar] [CrossRef]
- Crespi, B.J.; Yanega, D. The definition of eusociality. Behav. Ecol. 1995, 6, 109–115. [Google Scholar] [CrossRef]
- Bennett, N.C.; Faulkes, C.G. African Mole-Rats: Ecology and Eusociality; Cambridge University Press: Cambridge, UK, 2000. [Google Scholar]
- Burda, H.; Honeycutt, R.L.; Begall, S.; Locker-Grütjen, O.; Scharff, A. Are naked and common mole-rats eusocial and if so, why? Behav. Ecol. Sociobiol. 2000, 47, 293–303. [Google Scholar] [CrossRef]
- Jarvis, J.U.M. Eusociality in a Mammal: Cooperative Breeding in Naked Mole-Rat Colonies. Science 1981, 212, 571–573. [Google Scholar] [CrossRef]
- Jarvis, J.U.M.; Bennett, N. Eusociality has evolved independently in two genera of bathyergid mole-rats—But occurs in no other subterranean mammal. Behav. Ecol. Sociobiol. 1993, 33, 253–260. [Google Scholar] [CrossRef]
- Batra, S.W. Nests and social behavior of halictine bees of India (Hymenoptera: Halictidae). Indian J. Entomol. 1966, 28, 375. [Google Scholar]
- Michener, C.D. Comparative Social Behavior of Bees. Annu. Rev. Entomol. 1969, 14, 299–342. [Google Scholar] [CrossRef]
- Wilson, E.O. The Insect Societies; Harvard University Press: Cambridge, MA, USA, 1971. [Google Scholar]
- Dengler-Crish, C.M.; Catania, K.C. Phenotypic plasticity in female naked mole-rats after removal from reproductive suppression. J. Exp. Biol. 2007, 210, 4351–4358. [Google Scholar] [CrossRef]
- Henry, E.C.; Crish, C.; Catania, K.C. Growing out of a caste—Reproduction and the making of the queen mole-rat. J. Exp. Biol. 2007, 210, 261–268. [Google Scholar] [CrossRef]
- O’Riain, M.J.; Jarvis, J.U.M.; Alexander, R.; Buffenstein, R.; Peeters, C. Morphological castes in a vertebrate. Proc. Natl. Acad. Sci. USA 2000, 97, 13194–13197. [Google Scholar] [CrossRef]
- Braude, S. Dispersal and new colony formation in wild naked mole-rats: Evidence against inbreeding as the system of mating. Behav. Ecol. 2000, 11, 7–12. [Google Scholar] [CrossRef]
- Jarvis, J.U.M. Reproduction of Naked Mole-Rats. In The Biology of the Naked Mole-Rat; Sherman, P.W., Jarvis, J.U.M., Alexander, R.D., Eds.; Princeton University Press: Princeton, NJ, USA, 1991; pp. 384–425. [Google Scholar] [CrossRef]
- Buffenstein, R. Negligible senescence in the longest living rodent, the naked mole-rat: Insights from a successfully aging species. J. Comp. Physiol. B 2008, 178, 439–445. [Google Scholar] [CrossRef]
- Hallett, A.F.; Meester, J. Early Postnatal Development of the South African Hamster Mystromys Albicaudatus. Zool. Afr. 1971, 6, 221–228. [Google Scholar] [CrossRef]
- Norris, M.L.; Adams, C.E. Mortality from birth to weaning in the mongolian gerbil, Meriones unguiculatus. Lab. Anim. 1972, 6, 49–53. [Google Scholar] [CrossRef]
- Rödel, H.G.; Starkloff, A.; Bautista, A.; Friedrich, A.-C.; Von Holst, D. Infanticide and Maternal Offspring Defence in European Rabbits under Natural Breeding Conditions. Ethology 2008, 114, 22–31. [Google Scholar] [CrossRef]
- Edrey, Y.H.; Hanes, M.; Pinto, M.; Mele, J.; Buffenstein, R. Successful Aging and Sustained Good Health in the Naked Mole Rat: A Long-Lived Mammalian Model for Biogerontology and Biomedical Research. ILAR J. 2011, 52, 41–53. [Google Scholar] [CrossRef] [PubMed]
- O’Riain, J. Pup Ontogeny and Factors Influencing Behavioural and Morphological Variation in Naked Mole-Rats, Heterocephalus glaber (Rodentia, Bathyergidae). Ph.D. Thesis, University of Cape Town, Cape Town, South Africa, March 1996. [Google Scholar]
- Lacey, E.A.; Sherman, P.W. Social Organization of Naked Mole-Rat Colonies: Evidence for Divisions of Labor. In The Biology of the Naked Mole-Rat; Sherman, P.W., Jarvis, J.U.M., Alexander, R.D., Eds.; Princeton University Press: Princeton, NJ, USA, 1991; pp. 275–336. [Google Scholar] [CrossRef]
- Ke, Z.; Vaidya, A.; Ascher, J.; Seluanov, A.; Gorbunova, V. Novel husbandry techniques support survival of naked mole rat (Heterocephalus glaber) pups. J. Am. Assoc. Lab. Anim. Sci. 2014, 53, 89–91. [Google Scholar] [PubMed]
- Payne, S.F. Social Organization of the Naked Mole-Rat (Heterocephalus Glaber): Cooperation in Colony Labor and Reproduction. Master Thesis, University of California, Santa Cruz, CA, USA, December 1982. [Google Scholar]
- Dudley, D. Contributions of paternal care to the growth and development of the young in Peromyscus californicus. Behav. Biol. 1974, 11, 155–166. [Google Scholar] [CrossRef]
- Proffitt, K.M.; Rotella, J.J.; Garrott, R.A. Effects of pup age, maternal age, and birth date on pre-weaning survival rates of Weddell seals in Erebus Bay, Antarctica. Oikos 2010, 119, 1255–1264. [Google Scholar] [CrossRef]
- Brown, R.Z. Social Behavior, Reproduction, and Population Changes in the House Mouse (Mus musculus L.). Ecol. Monogr. 1953, 23, 217–240. [Google Scholar] [CrossRef]
- Buffenstein, R. The Naked Mole-Rat: A New Long-Living Model for Human Aging Research. J. Gerontol. Ser. A 2005, 60, 1369–1377. [Google Scholar] [CrossRef]
- O’Riain, M.J.; Jarvis, J.U.M.; Faulkes, C.G. A dispersive morph in the naked mole-rat. Nature 1996, 380, 619–621. [Google Scholar] [CrossRef]
- Ebensperger, L.A.; Hurtado, M.J.; León, C. An experimental examination of the consequences of communal versus solitary breeding on maternal condition and the early postnatal growth and survival of degu, Octodon degus, pups. Anim. Behav. 2007, 73, 185–194. [Google Scholar] [CrossRef]
- Campbell, M.T.; Slade, N.A. The effect of maternal mass on litter size and offspring survival in the hispid cotton rat (Sigmodon hispidus). Can. J. Zool. 1995, 73, 133–140. [Google Scholar] [CrossRef]
- Hastings, K.K.; Testa, J.W. Maternal and birth colony effects on survival of Weddell seal offspring from McMurdo Sound, Antarctica. J. Anim. Ecol. 1998, 67, 722–740. [Google Scholar] [CrossRef]
- Allainé, D.; Brondex, F.; Graziani, L.; Coulon, J.; Till-Bottraud, I. Male-biased sex ratio in litters of Alpine marmots supports the helper repayment hypothesis. Behav. Ecol. 2000, 11, 507–514. [Google Scholar] [CrossRef][Green Version]
- Clutton-Brock, T.H.; Gaynor, D.; McIlrath, G.M.; Maccoll, A.D.C.; Kansky, R.; Chadwick, P.; Manser, M.; Skinner, J.D.; Brotherton, P.N.M. Predation, group size and mortality in a cooperative mongoose, Suricata suricatta. J. Anim. Ecol. 1999, 68, 672–683. [Google Scholar] [CrossRef]
- Doidge, D.W.; Croxall, J.P.; Baker, J.R. Density-dependent pup mortality in the Antarctic fur seal Arctocephalus gazella at South Georgia. J. Zool. 1984, 202, 449–460. [Google Scholar] [CrossRef]
- French, J.A. Alloparents in the Mongolian gerbil: Impact on long-term reproductive performance of breeders and opportunities for independent reproduction. Behav. Ecol. 1994, 5, 273–279. [Google Scholar] [CrossRef]
- Mills, K.J.; Patterson, B.R.; Murray, D.L. Direct Estimation of Early Survival and Movements in Eastern Wolf Pups. J. Wildl. Manag. 2008, 72, 949–954. [Google Scholar] [CrossRef]
- Woodroffe, R.; Macdonald, D.W. Helpers provide no detectable benefits in the European badger (Meles meles). J. Zool. 2000, 250, 113–119. [Google Scholar] [CrossRef]
- Zahn, A. Reproductive success, colony size and roost temperature in attic-dwelling bat Myotis myotis. J. Zool. 1999, 247, 275–280. [Google Scholar] [CrossRef]
- Pistorius, P.A.; Bester, M.N.; Kirkman, S.P.; Taylor, F.E. Pup mortality in southern elephant seals at Marion Island. Polar Biol. 2001, 24, 828–831. [Google Scholar] [CrossRef]
- Kahan, E.; Rosen, M. The influence of litter size and favourable conditions on mortality and weaning weights in rats. Lab. Anim. 1984, 18, 247–251. [Google Scholar] [CrossRef] [PubMed]
- Gates, W.H. Litter size, birth weight, and early growth rate of mice (Mus musculus). Anat. Rec. 1925, 29, 183–193. [Google Scholar] [CrossRef]
- Goundie, T.R.; Vessey, S.H. Survival and Dispersal of Young White-Footed Mice Born in Nest Boxes. J. Mammal. 1986, 67, 53–60. [Google Scholar] [CrossRef]
- Milligan, B.N.; Fraser, D.; Kramer, D.L. Birth weight variation in the domestic pig: Effects on offspring survival, weight gain and suckling behaviour. Appl. Anim. Behav. Sci. 2001, 73, 179–191. [Google Scholar] [CrossRef]
- Woodley, R.; Buffenstein, R. Thermogenic changes with chronic cold exposure in the naked mole-rat (Heterocephalus glaber). Comp. Biochem. Physiol. Part A: Mol. Integr. Physiol. 2002, 133, 827–834. [Google Scholar] [CrossRef]
- Schradin, C.; Pillay, N. The influence of the father on offspring development in the striped mouse. Behav. Ecol. 2004, 16, 450–455. [Google Scholar] [CrossRef]
- Malherbe, G.; Bennett, N.; Schoeman, A. The reproductive biology and postnatal development of the highveld mole-rat, Cryptomys hottentotus pretoriae (Rodentia: Bathyergidae). Afr. Zool. 2003, 38, 161–167. [Google Scholar] [CrossRef]
- Burn, C.C.; Mason, G.J. Effects of cage-cleaning frequency on laboratory rat reproduction, cannibalism, and welfare. Appl. Anim. Behav. Sci. 2008, 114, 235–247. [Google Scholar] [CrossRef][Green Version]
- Braude, S. The Behavior and Demographics of the Naked Mole-Rat, Heterocephalus glaber. Ph.D. Thesis, University of Michigan, Ann Arbor, MI, USA, 1991. [Google Scholar]
- Edwards, P.D. For the physiology of cooperative breeding, it’s time to move beyond stress: A Comment on: ‘Stress in an underground empire’ by Medger. Biol. Lett. 2022, 18, 20220375. [Google Scholar] [CrossRef] [PubMed]
- Faulkes, C.G.; Abbott, D.H. Evidence that primer pheromones do not cause social suppression of reproduction in male and female naked mole-rats (Heterocephalus glaber). Reproduction 1993, 99, 225–230. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.P.; Smith, M.; Buffenstein, R.; Harries, L.W. Negligible senescence in naked mole rats may be a consequence of well-maintained splicing regulation. Geroscience 2020, 42, 633–651. [Google Scholar] [CrossRef] [PubMed]
- O’Riain, M.J.; Faulkes, C.G. African Mole-Rats: Eusociality, Relatedness and Ecological Constraints. In Ecology of Social Evolution; Korb, J., Heinze, J., Eds.; Springer: Berlin/Heidelberg, Germany, 2008; pp. 207–223. [Google Scholar] [CrossRef]
- Brett, R.A. The Population Structure of Naked Mole-Rat Colonies. In The Biology of the Naked Mole-Rat; Sherman, P.W., Jarvis, J.U.M., Alexander, R.D., Eds.; Princeton University Press: Princeton, NJ, USA, 1991; pp. 97–136. [Google Scholar] [CrossRef]
- Yahav, S.; Buffenstein, R. Huddling Behavior Facilitates Homeothermy in the Naked Mole Rat Heterocephalus glaber. Physiol. Zool. 1991, 64, 871–884. [Google Scholar] [CrossRef]
- Ingram, C.M.; Troendle, N.J.; Gill, C.A.; Braude, S.; Honeycutt, R.L. Challenging the inbreeding hypothesis in a eusocial mammal: Population genetics of the naked mole-rat, Heterocephalus glaber. Mol. Ecol. 2015, 24, 4848–4865. [Google Scholar] [CrossRef]
- Reeve, H.K.; Westneat, D.F.; Noon, W.A.; Sherman, P.W.; Aquadro, C.F. DNA “fingerprinting” reveals high levels of inbreeding in colonies of the eusocial naked mole-rat. Proc. Natl. Acad. Sci. USA 1990, 87, 2496–2500. [Google Scholar] [CrossRef]
- Clarke, F.M.; Faulkes, C.G. Kin discrimination and female mate choice in the naked mole-rat Heterocephalus glaber. Proc. R. Soc. B Boil. Sci. 1999, 266, 1995–2002. [Google Scholar] [CrossRef]
- Hamilton, W. The genetical evolution of social behaviour. II. J. Theor. Biol. 1964, 7, 17–52. [Google Scholar] [CrossRef]
- McNab, B.K. The Metabolism of Fossorial Rodents: A Study of Convergence. Ecology 1966, 47, 712–733. [Google Scholar] [CrossRef]
- Withers, P.; Jarvis, J. The effect of huddling on thermoregulation and oxygen consumption for the naked mole-rat. Comp. Biochem. Physiol. Part A Physiol. 1980, 66, 215–219. [Google Scholar] [CrossRef]
- Röllig, K.; Drews, B.; Göritz, F.; Hildebrandt, T.B. The Long Gestation of the Small Naked Mole-Rat (Heterocephalus glaber RÜPPELL, 1842) Studied with Ultrasound Biomicroscopy and 3D-Ultrasonography. PLoS ONE 2011, 6, e17744. [Google Scholar] [CrossRef] [PubMed]
- Katsushima, K.; Nishida, C.; Yosida, S.; Kato, M.; Okanoya, K.; Matsuda, Y. A multiplex PCR assay for molecular sexing of the naked mole-rat (Heterocephalus glaber). Mol. Ecol. Resour. 2010, 10, 222–224. [Google Scholar] [CrossRef] [PubMed]
- Szafranski, K.; Wetzel, M.; Holtze, S.; Büntjen, I.; Lieckfeldt, D.; Ludwig, A.; Huse, K.; Platzer, M.; Hildebrandt, T. The Mating Pattern of Captive Naked Mole-Rats Is Best Described by a Monogamy Model. Front. Ecol. Evol. 2022, 10, 855688. [Google Scholar] [CrossRef]
- Rousset, F.; Ferdy, J.-B. Testing environmental and genetic effects in the presence of spatial autocorrelation. Ecography 2014, 37, 781–790. [Google Scholar] [CrossRef]
- R-Core-Team. R: A Language and Environment for Statistical Computing, 2014; R Foundation for Statistical Computing: Vienna, Austria, 2016. [Google Scholar]
- Burda, H. Constraints of pregnancy and evolution of sociality in mole-rats with special reference to reproductive and social patterns in Cryptomys hottentotus (Bathyergidae, Rodentia). J. Zool. Syst. Evol. Res. 1990, 28, 26–39. [Google Scholar] [CrossRef]
- Šumbera, R.; Burda, H.; Chitaukali, W.N. Reproductive Biology of a Solitary Subterranean Bathyergid Rodent, the Silvery Mole-Rat (Heliophobius Argenteocinereus). J. Mammal. 2003, 84, 278–287. [Google Scholar] [CrossRef]
- Yu, C.; Wang, S.; Yang, G.; Zhao, S.; Lin, L.; Yang, W.; Tang, Q.; Sun, W.; Cui, S. Breeding and Rearing Naked Mole-Rats (Heterocephalus glaber) under Laboratory Conditions. J. Am. Assoc. Lab. Anim. Sci. 2017, 56, 98–101. [Google Scholar]
- Sherman, P.W.; Braude, S.; Jarvis, J.U.M. Litter Sizes and Mammary Numbers of Naked Mole-Rats: Breaking the One-Half Rule. J. Mammal. 1999, 80, 720–733. [Google Scholar] [CrossRef][Green Version]
- Knight, C.H.; Maltz, E.; Docherty, A.H. Milk yield and composition in mice: Effects of litter size and lactation number. Comp. Biochem. Physiol. Part A Physiol. 1986, 84, 127–133. [Google Scholar] [CrossRef]
- Stockley, P.; Parker, G.A. Life history consequences of mammal sibling rivalry. Proc. Natl. Acad. Sci. USA 2002, 99, 12932–12937. [Google Scholar] [CrossRef]
- Braude, S.; Ciszek, D.; Berg, N.E.; Shefferly, N. The ontogeny and distribution of countershading in colonies of the naked mole-rat (Heterocephalus glaber). J. Zool. 2001, 253, 351–357. [Google Scholar] [CrossRef]
- Hilakivi-Clarke, L.A.; Lister, R.G. The role of body weight in resident-intruder aggression. Aggress. Behav. 1992, 18, 281–287. [Google Scholar] [CrossRef]
- Heymann, E.W.; Soini, P. Offspring number in pygmy marmosets, Cebuella pygmaea *, in relation to group size and the number of adult males. Behav. Ecol. Sociobiol. 1999, 46, 400–404. [Google Scholar] [CrossRef]
- Harbo, J.R. Effect of Population Size on Brood Production, Worker Survival and Honey Gain in Colonies of Honeybees. J. Apic. Res. 1986, 25, 22–29. [Google Scholar] [CrossRef]
- Brett, R.A. The Ecology and Behaviour of the Naked Mole-Rat, Heterocephalus glaber Rüppell (Rodenti: Bathyergidae). Ph.D. Thesis, University College London (University of London), London, UK, 1986. [Google Scholar]
- Smith, M.; Buffenstein, R. Managed Care of Naked Mole-Rats. Adv. Exp. Med. Biol. 2021, 1319, 381–407. [Google Scholar] [CrossRef] [PubMed]

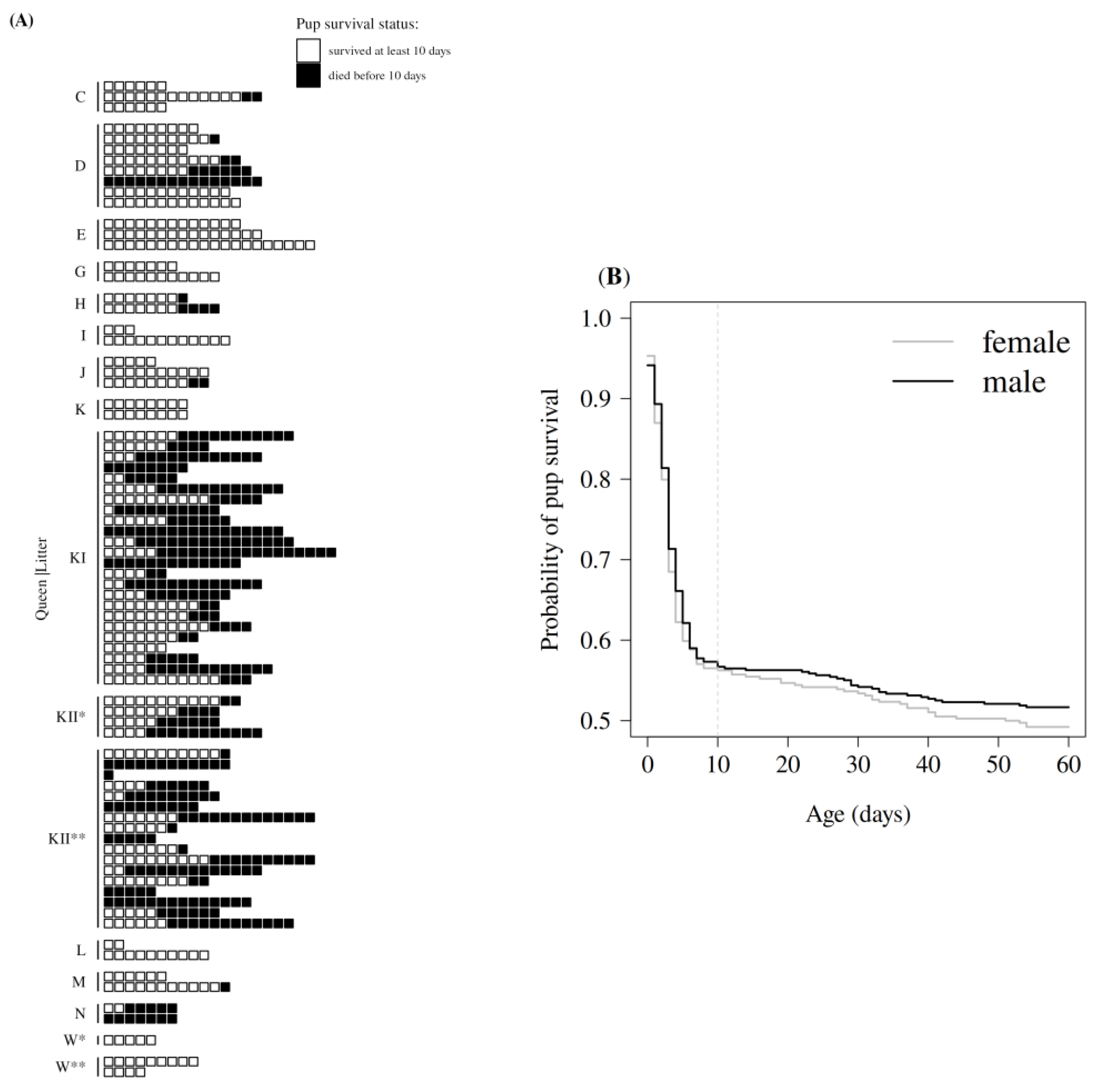
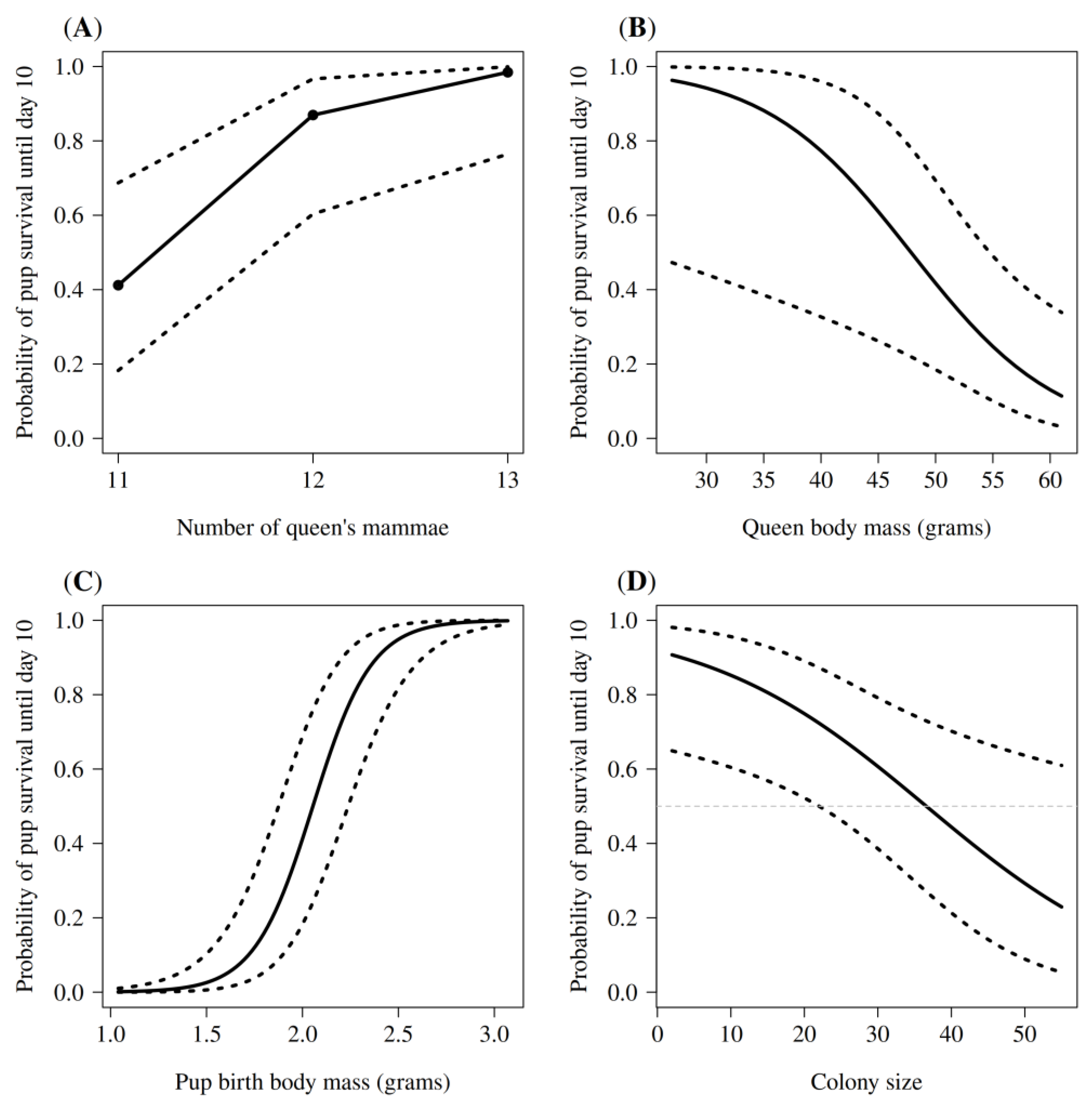
| Predictor Variables | Beta | SE | χ2 | df | p-Value |
|---|---|---|---|---|---|
| Individual characteristics | 88.02 | 2 | 0.001 | ||
| Pup birth body mass | 6.55 | 0.90 | 88.01 | 1 | 0.001 |
| Sex (male) | −0.22 | 0.33 | 0.53 | 1 | 0.453 |
| Maternal characteristics | 16.66 | 2 | 0.015 | ||
| Queen body mass | −0.16 | 0.06 | 7.96 | 1 | 0.015 |
| Queen mammae | 2.25 | 0.87 | 6.31 | 1 | 0.048 |
| Social characteristics | 6.17 | 2 | 0.090 | ||
| Colony size | −0.07 | 0.03 | 6.17 | 1 | 0.039 |
| Litter size | 0.09 | 0.09 | 0.91 | 1 | 0.423 |
| Environmental characteristics | 4.31 | 3 | 0.316 | ||
| Temperature | 0.26 | 0.58 | 0.25 | 1 | 0.653 |
| Humidity | 0.04 | 0.04 | 0.94 | 1 | 0.404 |
| Nestbox change | −0.32 | 0.23 | 2.53 | 1 | 0.182 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wetzel, M.; Courtiol, A.; Hofer, H.; Holtze, S.; Hildebrandt, T.B. Pup Recruitment in a Eusocial Mammal—Which Factors Influence Early Pup Survival in Naked Mole-Rats? Animals 2023, 13, 630. https://doi.org/10.3390/ani13040630
Wetzel M, Courtiol A, Hofer H, Holtze S, Hildebrandt TB. Pup Recruitment in a Eusocial Mammal—Which Factors Influence Early Pup Survival in Naked Mole-Rats? Animals. 2023; 13(4):630. https://doi.org/10.3390/ani13040630
Chicago/Turabian StyleWetzel, Michaela, Alexandre Courtiol, Heribert Hofer, Susanne Holtze, and Thomas B. Hildebrandt. 2023. "Pup Recruitment in a Eusocial Mammal—Which Factors Influence Early Pup Survival in Naked Mole-Rats?" Animals 13, no. 4: 630. https://doi.org/10.3390/ani13040630
APA StyleWetzel, M., Courtiol, A., Hofer, H., Holtze, S., & Hildebrandt, T. B. (2023). Pup Recruitment in a Eusocial Mammal—Which Factors Influence Early Pup Survival in Naked Mole-Rats? Animals, 13(4), 630. https://doi.org/10.3390/ani13040630






