N-3 Polyunsaturated Fatty Acids as a Nutritional Support of the Reproductive and Immune System of Cattle—A Review
Abstract
Simple Summary
Abstract
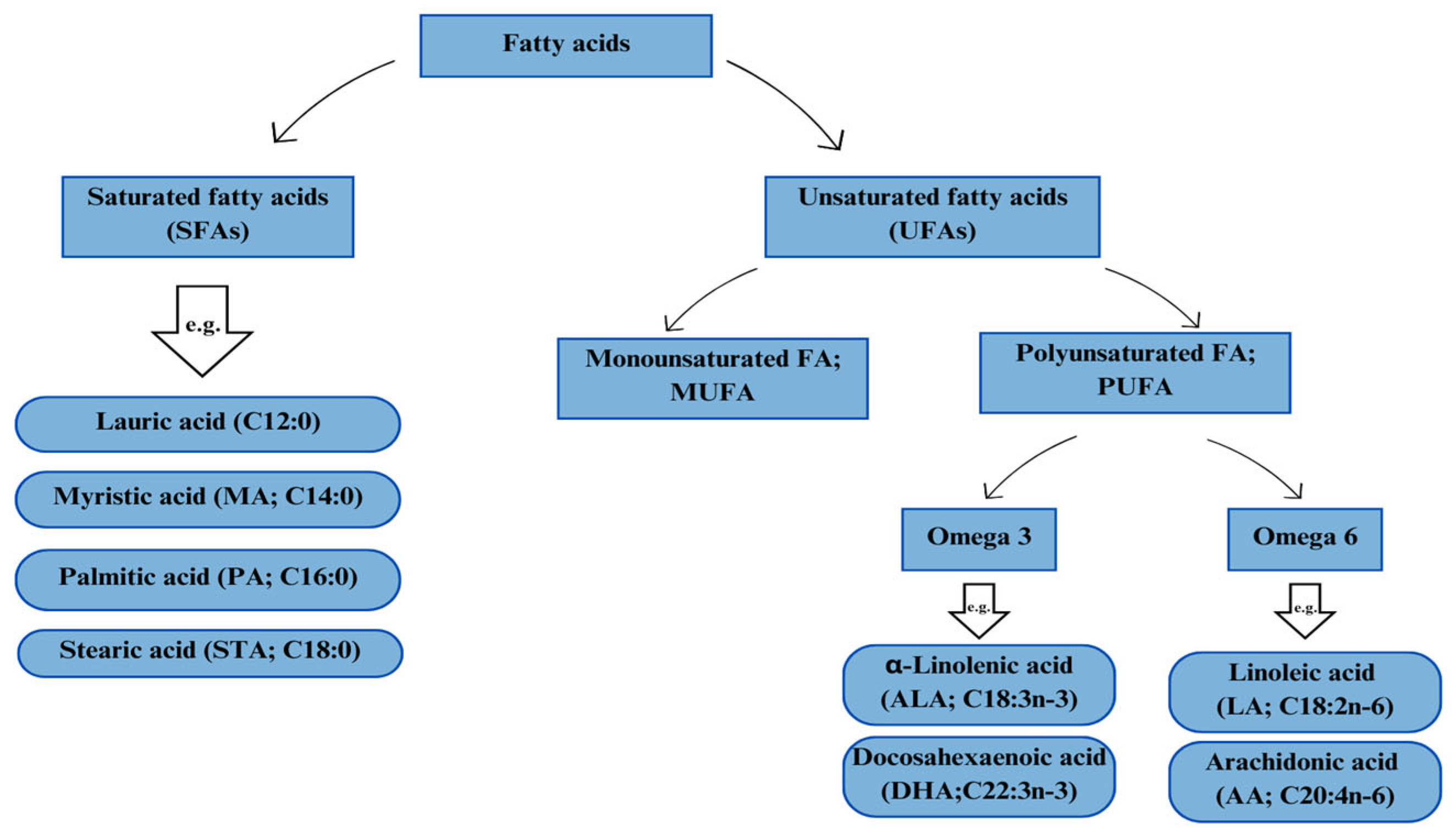
1. Introduction
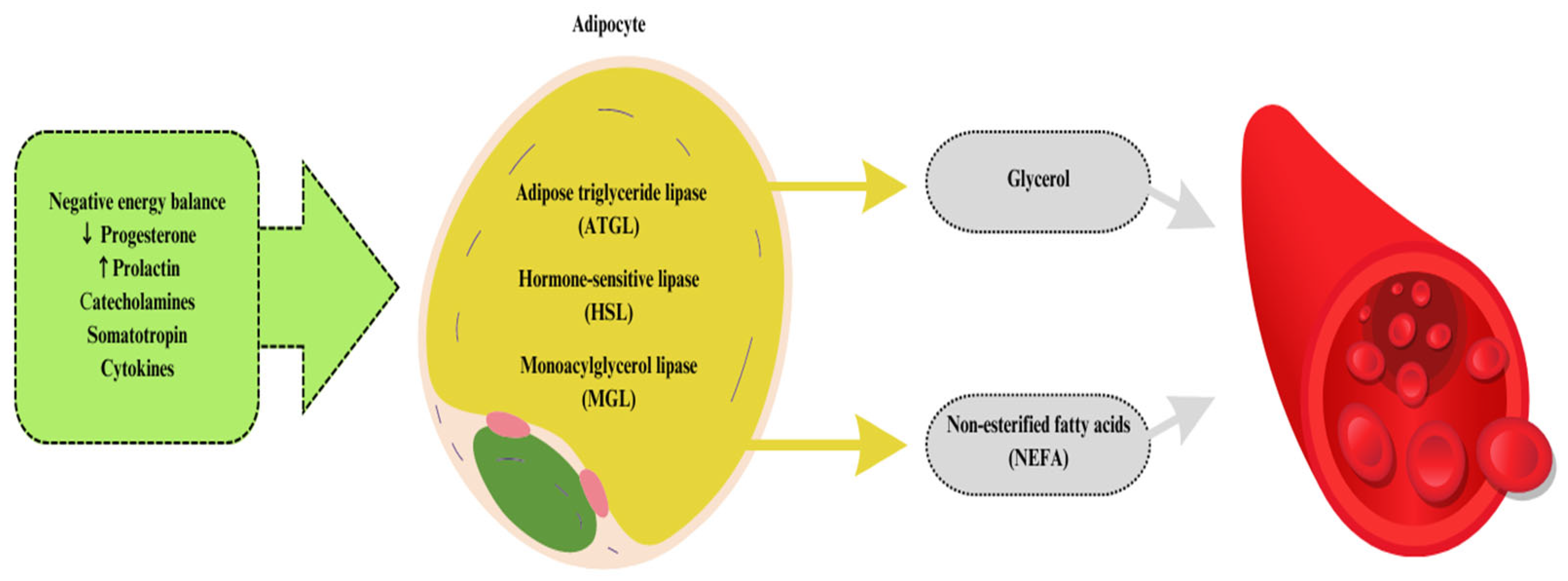
2. The Energetic Importance of Fat in Ruminant Nutrition
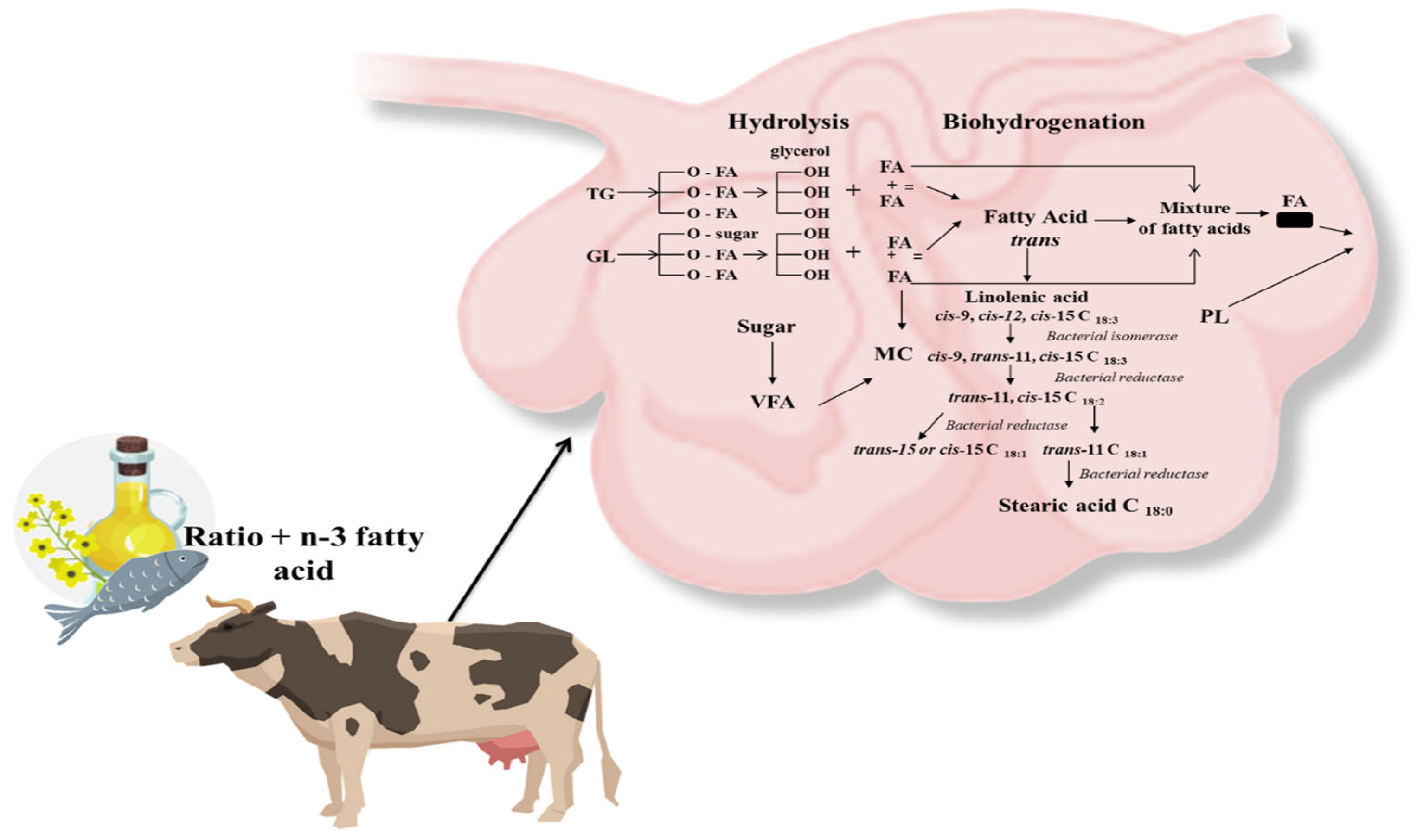
3. Lipid Metabolism in Rumen—Impact of Rumen Microbiota on FA Biohydrogenation and Transformation and the Role of By-Pass Fat
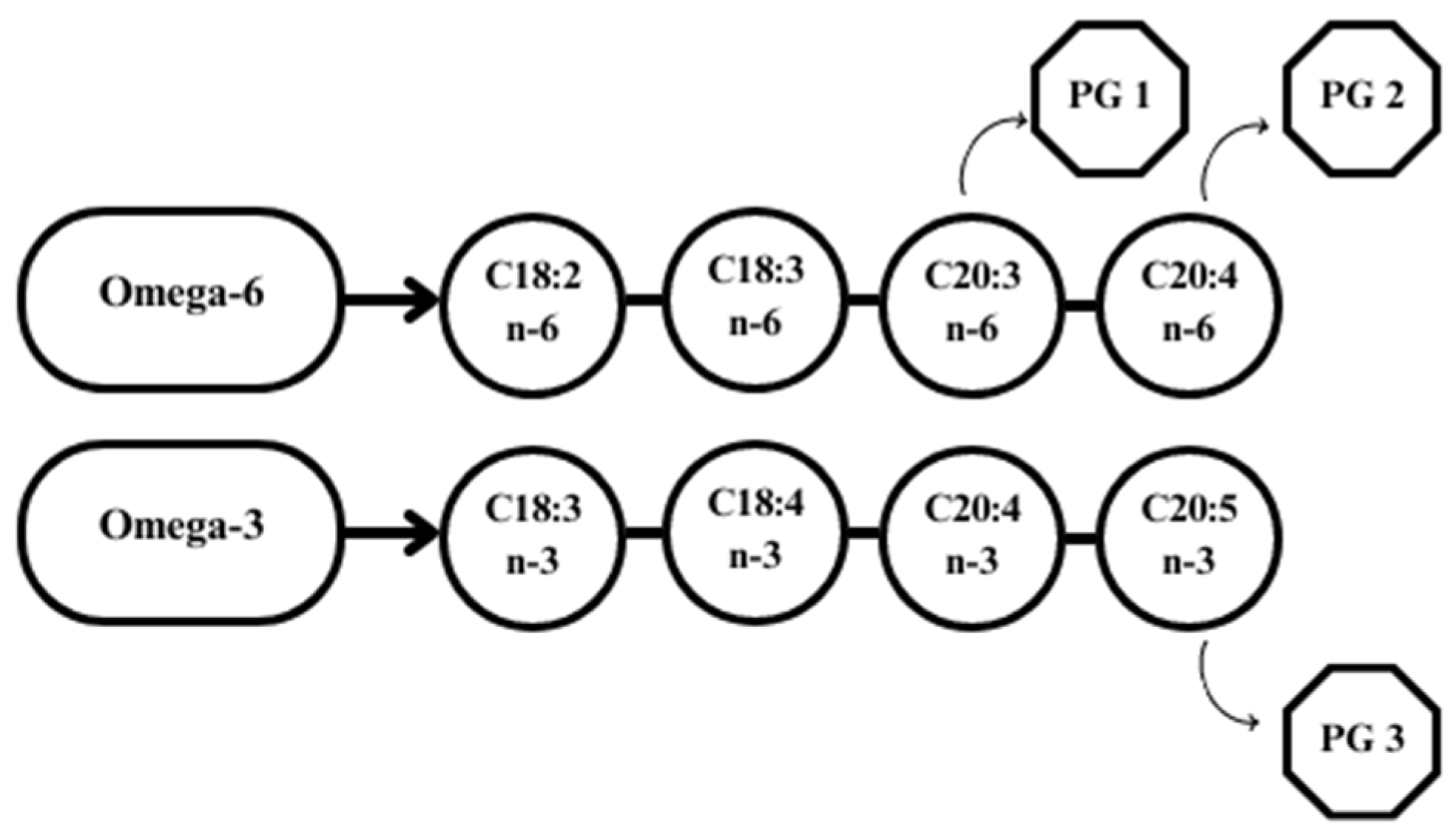
4. Regulatory Role of N-3 Fatty Acids in Reproduction of Ruminants
| Experimental Animals | Experiment Design | Number of Fatty Acids in the Ration/Fat Supplement | The Observed Results | Source |
|---|---|---|---|---|
| 739 primiparous and multiparous Holstein cows | Cows were assigned randomly to either a control or the same diet supplemented daily with 100 g/cow of an algae product containing 10% DHA Experiment duration: 27 to 147 days postpartum | Control diet: n-3—3.41% n-6—40.66% Algae supplement: n-3—30.50% n-6—6.56% | ↑ Resumption of estrous cyclicity ↑ Pregnancy first in primiparous cows ↑ Number of pregnancy primiparous and multiparous cows ↑ Expression of RTP4 in peripheral blood leukocytes ↑ Increased the incorporation of DHA, EPA, conjugated linoleic acid isomers cis-9 trans-11, trans-10, cis-12, and total n-3 FA in plasma phospholipids | [105] |
| 42,256-day pregnant Israeli Holstein dry cows | Cows were supplemented with encapsulated fats in treatments designated as an SFA—saturated fat at 240 and 560 g/day per cow, prepartum and postpartum (PP), respectively; FLX—flaxseed oil at 300 and 700 g/day per cow prepartum and PP, respectively; FO—fish oil at 300 and 700 g/day per cow prepartum and PP, respectively | FLX: SFA—62.41% MUFA—8.10% PUFA—26.49% n-3—23.42% FO: SFA—71.73% MUFA—16.66% PUFA—11.60% n-3—7.68% | FO: ↑ Proportion of docosahexaenoic acid (DHA) ↑ The follicle number during ovum pickup ↑ Percentage of oocytes that developed to blastocysts Oocyte cleavage FLX: ↑ The follicle number during ovum pickup ↑ The proportion of a-linolenic acid (ALA) in follicular fluid, granulosa cells, and cumulus–oocyte complexes ↑ Oocyte cleavage | [88] |
| 37 Angus heifers (Bos taurus) aged between 10 and 27 months | Angus heifers were supplemented with either 450 g of rumen-protected fish oil (omega 3 FA) or sunflower oil (omega 6 FA). Experiment duration: 56 days | A diet with fish oil: SFA—72.8% MUFA—8.04% PUFA—19.1% n-3—13% n-6—5.7% n-6/n-3—0.46 A diet with fish oil: SFA—70.8% MUFA—10.1% PUFA—19% n-3—4.1% n-6—15.1% n-6/n-3—3.91% | A diet with fish oil: ↑ Embryo elongation ↑ Concentration of plasma progesterone during luteal growth ↑ Increased plasma P4 concentration ↓ Endometrial concentration—precursor of arachidonic acid | [91] |
| 315 early lactation Holstein cows | Cows were offered rations formulated to contain 0 g/kg (No-Lin), 25 g/kg (LoLin), and 50 g/kg (HiLin) dry matter (DM) of LinPro—products based on flax seeds | NoLin diet: * SFA—24.76% MUFA—26.46% PUFA—48.47% n-3—4.42% LoLin diet: * SFA—22.85% MUFA—26.12% PUFA—50.73% n-3—10.23% HiLin: * SFA—21.47% MUFA—26.43% PUFA—51.88% n-3—15.53% | LoLin, HiLin diet: ↓ Fertilization frequency ↓ The reproductive performance ↓ The body condition score (BCS) in early lactation with LinPro feeding ↑ Increased plasma P4 concentration or HiLin cows ↑ The health status of cows | [111] |
| 120 nonlactating pregnant Holstein cattle | Prepartum cattle were fed 1 of the following 3 diets: (1) no fat supplement (CON); (2) 1.15% of dietary DM as Ca-salts of soybean oil (CSO, 140 g/cow/daily) supplement; (3) 1.15% of dietary DM as Ca salts of fish oil (CFO, 140 g/cow/daily) supplement | CSO: SFA—20.4% UFA—76.8% PUFA—60% n-3—13.6% n-6—46.4% CFO: SFA—26.1% UFA—48.2% PUFA—51.2% n-3—14.7% n-6—31.1% | CFO: ↓ The period between first estrus and first insemination ↑ The health status of cows ↓ Total reproductive disorders ↑ Improved productive and reproductive performance in the subsequent lactation | [114] |
5. Role of N-3 Fatty Acids in the Cows’ Nutrition during Periparturient Period
6. Importance of FA N-3 in Fetus Developmental Programming and Embryo Development
7. N-3 Fatty Acids Are a Key Component in Building Immunity
8. N-3 Fatty Acids as Immune Support for Newborn Calves
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ALT | alanine aminotransferase |
| AST | aspartate aminotransferase |
| ATP | adenosine triphosphate |
| BHB | beta-hydroxybutyrate |
| C12:0 | lauric acid |
| C14:0; MA | myristic acid |
| C16:0; PA | palmitic acid |
| C18:0; STA | stearic acid |
| C18:1 | oleic acid |
| C18:1; TVA | vaccenic acid |
| C18:2; LA | linoleic acid |
| C18:3 n-3; ALA | alpha-lipoic acid |
| C18:3; LNA | linolenic acid |
| C20:4; AA | arachidonic acid |
| C20:5 n-3; EPA | eicosapentaenoic acid |
| C22:6 n-3; DHA | docosahexaenoic acid |
| CAT | catalase |
| CD62 | selectin P |
| CL | corpus luteum |
| CNS | central nervous system |
| COX-2 | enzyme cyclooxygenase-2 |
| CPT-I | carnitine palmitoyltransferase-I |
| DNMT | DNA methyltransferase |
| EFA | essential fatty acids |
| FA | fatty acids |
| FABP | intracellular FA-binding proteins |
| FATP | fatty acid transporter proteins |
| FFAR | free fatty acid receptor |
| FFAs | free fatty acids |
| FLX | flaxseed |
| FO | fish oils |
| GPx (GSH-Px) | glutathione peroxidase |
| IFN | interferon |
| IFN-τ | interferon-tau |
| Ig | immunoglobulins |
| IgA | immunoglobulin A |
| IgE | immunoglobulin E |
| IGF-1 | insulin-like growth factor 1 |
| IgG | immunoglobulin G |
| IgM | immunoglobulin M |
| IL | interleukin |
| IL-1β | pro-inflammatory interleukin |
| IL-4 | interleukin-4 |
| IL-8 | interleukin-8 |
| ISGs | interferon-induced genes |
| IVM | in vitro maturation medium |
| LC-PUFA | long-chain polyunsaturated fatty acids |
| LF | lactoferrin |
| LH | luteinizing hormone |
| LPS | lipopolysaccharides |
| Lz | lysozyme |
| MDA | lipid peroxide |
| MII | oocyte into metaphase II |
| MONO | monocytes |
| MR | milk replacer |
| mRNA | messenger RNA |
| MUFA | monounsaturated fatty acid |
| N-3 PUFA | omega-3 fatty acids |
| NEB | negative energy balance |
| NEFA | non-esterified fatty acids |
| NFκB | nuclear factor kappa B |
| NK | natural killer cells |
| NL | neutral lipids |
| OCC | oocyte–cell complex |
| OSi | oxidative status index |
| PBL | blood leukocytes |
| PBMC | peripheral blood mononuclear cells |
| PG | prostaglandin |
| PGFM | prostaglandin F2 alpha metabolite |
| PGHS | prostaglandin endonadoxide synthase |
| PL | phospholipids |
| PPARs | peroxisome proliferator-activated receptors |
| PTGS2 | prostaglandin-endoperoxidase 2 |
| PUFA | polyunsaturated fatty acid |
| RELA | v-rel avian reticuloendotheliosis viral oncogene homolog A |
| RONS | reactive oxygen and nitrogen species |
| SFA | saturated fatty acid |
| SOD | superoxide dismutase |
| TAG | triacylglycerol |
| TAS | total antioxidant capacity |
| TG | triglycerides |
| Th1 | T helper 1 |
| TNF-α | tumor necrosis factor-α |
| TXA2 | thromboxane A2 |
| UFA | unsaturated fatty acids |
| VLDL | very low-density lipoproteins |
| WBC | leukocytes |
| α-LG | α-lactalbumin |
| β-LG | β-lactoglobulin |
References
- Bordón, M.G.; Meriles, S.P.; Ribotta, P.D.; Martinez, M.L. Enhancement of Composition and Oxidative Stability of Chia (Salvia hispanica L.) Seed Oil by Blending with Specialty Oils. J. Food Sci. 2019, 84, 1035–1044. [Google Scholar] [CrossRef]
- Das, R.; Sailo, L.; Verma, N.; Bharti, P.; Saikia, J.; Kumar, R. Impact of Heat Stress on Health and Performance of Dairy Animals: A Review. Vet. World 2016, 9, 260. [Google Scholar] [CrossRef] [PubMed]
- Chitra, P. Potential and Utilization of By-Products of Oilseeds in Animal Feed Industry. Biot. Res. Today 2021, 3, 655–657. [Google Scholar]
- Scholljegerdes, E.J.; Lekatz, L.A.; Vonnahme, K.A. Effects of Short-Term Oilseed Supplementation on Plasma Fatty Acid Composition, Progesterone and Prostaglandin F Metabolite in Lactating Beef Cows. Animal 2014, 8, 777–785. [Google Scholar] [CrossRef]
- Dewanckele, L.; Toral, P.G.; Vlaeminck, B.; Fievez, V. Invited Review: Role of Rumen Biohydrogenation Intermediates and Rumen Microbes in Diet-Induced Milk Fat Depression: An Update. J. Dairy Sci. 2020, 103, 7655–7681. [Google Scholar] [CrossRef] [PubMed]
- Busato, S.; Bionaz, M. When Two plus Two Is More than Four: Evidence for a Synergistic Effect of Fatty Acids on Peroxisome Proliferator—Activated Receptor Activity in a Bovine Hepatic Model. Genes 2021, 12, 1283. [Google Scholar] [CrossRef]
- He, M.; Armentano, L.E. Effect of Fatty Acid Profile in Vegetable Oils and Antioxidant Supplementation on Dairy Cattle Performance and Milk Fat Depression. J. Dairy Sci. 2011, 94, 2481–2491. [Google Scholar] [CrossRef]
- Ganesan, B.; Brothersen, C.; McMahon, D.J. Fortification of Foods with Omega-3 Polyunsaturated Fatty Acids. Crit. Rev. Food Sci. Nutr. 2014, 54, 98–114. [Google Scholar] [CrossRef]
- De Carvalho, C.; Caramujo, M. The Various Roles of Fatty Acids. Molecules 2018, 23, 2583. [Google Scholar] [CrossRef]
- Sassa, T.; Kihara, A. Metabolism of Very Long-Chain Fatty Acids: Genes and Pathophysiology. Biomol. Ther. 2014, 22, 83–92. [Google Scholar] [CrossRef]
- Dyall, S.C.; Balas, L.; Bazan, N.G.; Brenna, J.T.; Chiang, N.; da Costa Souza, F.; Dalli, J.; Durand, T.; Galano, J.-M.; Lein, P.J.; et al. Polyunsaturated Fatty Acids and Fatty Acid-Derived Lipid Mediators: Recent Advances in the Understanding of Their Biosynthesis, Structures, and Functions. Prog. Lipid Res. 2022, 86, 101165. [Google Scholar] [CrossRef] [PubMed]
- Cholewski, M.; Tomczykowa, M.; Tomczyk, M. A Comprehensive Review of Chemistry, Sources and Bioavailability of Omega-3 Fatty Acids. Nutrients 2018, 10, 1662. [Google Scholar] [CrossRef] [PubMed]
- Gammone, M.; Riccioni, G.; Parrinello, G.; D’Orazio, N. Omega-3 Polyunsaturated Fatty Acids: Benefits and Endpoints in Sport. Nutrients 2018, 11, 46. [Google Scholar] [CrossRef] [PubMed]
- Raphael, W.; Halbert, L.; Contreras, G.A.; Sordillo, L.M. Association between Polyunsaturated Fatty Acid-Derived Oxylipid Biosynthesis and Leukocyte Inflammatory Marker Expression in Periparturient Dairy Cows. J. Dairy Sci. 2014, 97, 3615–3625. [Google Scholar] [CrossRef]
- Kazemi-Bonchenari, M.; Dehghan-Banadaky, M.; Fattahnia, F.; Saleh-Bahmanpour, A.; Jahani-Moghadam, M.; Mirzaei, M. Effects of Linseed Oil and Rumen Undegradable Protein: Rumen Degradable Protein Ratio on Performance of Holstein Dairy Calves. Br. J. Nutr. 2020, 123, 1247–1257. [Google Scholar] [CrossRef]
- Moallem, U.; Lehrer, H.; Livshits, L.; Zachut, M. The Effects of Omega-3 α-Linolenic Acid from Flaxseed Oil Supplemented to High-Yielding Dairy Cows on Production, Health, and Fertility. Livest. Sci. 2020, 242, 104302. [Google Scholar] [CrossRef]
- Hansen, T.V.; Dalli, J.; Serhan, C.N. The Novel Lipid Mediator PD1n-3 DPA: An Overview of the Structural Elucidation, Synthesis, Biosynthesis and Bioactions. Prostaglandins Other Lipid Mediat. 2017, 133, 103–110. [Google Scholar] [CrossRef]
- Calder, P.C. Omega-3 Fatty Acids and Inflammatory Processes: From Molecules to Man. Biochem. Soc. Trans. 2017, 45, 1105–1115. [Google Scholar] [CrossRef]
- Cherian, G. Hatching Egg Polyunsaturated Fatty Acids and the Broiler Chick. J. Anim. Sci. Biotechnol. 2022, 13, 98. [Google Scholar] [CrossRef]
- Gladine, C.; Mazur, A. Nutrigenomic Effects of Omega-3 Fatty Acids: Nutrigenomic Effects of Omega-3 Fatty Acids. Lipid Technol. 2014, 26, 227–229. [Google Scholar] [CrossRef]
- Libera, K.; Cieslak, A.; Szumacher, M.; Cieslak, D. Dodatek Nienasyconych Kwasów Tłuszczowych Do Dawki Pokarmowej a Wyniki Rozrodu Bydła. Przegląd Hod. 2019, 87, 22–25. [Google Scholar]
- Bionaz, M.; Vargas-Bello-Pérez, E.; Busato, S. Advances in Fatty Acids Nutrition in Dairy Cows: From Gut to Cells and Effects on Performance. J. Anim. Sci. Biotechnol. 2020, 11, 110. [Google Scholar] [CrossRef]
- VandeHaar, M.J.; Armentano, L.E.; Weigel, K.; Spurlock, D.M.; Tempelman, R.J.; Veerkamp, R. Harnessing the Genetics of the Modern Dairy Cow to Continue Improvements in Feed Efficiency. J. Dairy Sci. 2016, 99, 4941–4954. [Google Scholar] [CrossRef] [PubMed]
- Knob, D.A.; Thaler Neto, A.; Schweizer, H.; Weigand, A.C.; Kappes, R.; Scholz, A.M. Energy Balance Indicators during the Transition Period and Early Lactation of Purebred Holstein and Simmental Cows and Their Crosses. Animals 2021, 11, 309. [Google Scholar] [CrossRef]
- Tessari, R.; Berlanda, M.; Morgante, M.; Badon, T.; Gianesella, M.; Mazzotta, E.; Contiero, B.; Fiore, E. Changes of Plasma Fatty Acids in Four Lipid Classes to Understand Energy Metabolism at Different Levels of Non-Esterified Fatty Acid (NEFA) in Dairy Cows. Animals 2020, 10, 1410. [Google Scholar] [CrossRef]
- Puppel, K.; Kuczyńska, B. Metabolic Profiles of Cow’s Blood; a Review. J. Sci. Food Agric. 2016, 96, 4321–4328. [Google Scholar] [CrossRef]
- Kuhla, B.; Metges, C.C.; Hammon, H.M. Endogenous and Dietary Lipids Influencing Feed Intake and Energy Metabolism of Periparturient Dairy Cows. Domest. Anim. Endocrinol. 2016, 56, S2–S10. [Google Scholar] [CrossRef]
- McNamara, J.P.; Huber, K. Metabolic and Endocrine Role of Adipose Tissue During Lactation. Annu. Rev. Anim. Biosci. 2018, 6, 177–195. [Google Scholar] [CrossRef]
- Grabner, G.F.; Xie, H.; Schweiger, M.; Zechner, R. Lipolysis: Cellular Mechanisms for Lipid Mobilization from Fat Stores. Nat. Metab. 2021, 3, 1445–1465. [Google Scholar] [CrossRef]
- Ingvartsen, K.L.; Moyes, K.M. Factors Contributing to Immunosuppression in the Dairy Cow during the Periparturient Period. Jpn. J. Vet. Res. 2015, 63 (Suppl. 1), S15–S24. [Google Scholar]
- Abo Alrob, O.; Lopaschuk, G.D. Role of CoA and Acetyl-CoA in Regulating Cardiac Fatty Acid and Glucose Oxidation. Biochem. Soc. Trans. 2014, 42, 1043–1051. [Google Scholar] [CrossRef]
- Tahri-Joutey, M.; Andreoletti, P.; Surapureddi, S.; Nasser, B.; Cherkaoui-Malki, M.; Latruffe, N. Mechanisms Mediating the Regulation of Peroxisomal Fatty Acid Beta-Oxidation by PPARα. Int. J. Mol. Sci. 2021, 22, 8969. [Google Scholar] [CrossRef]
- Schlaepfer, I.R.; Joshi, M. CPT1A-Mediated Fat Oxidation, Mechanisms, and Therapeutic Potential. Endocrinology 2020, 161, bqz046. [Google Scholar] [CrossRef] [PubMed]
- Soares, R.A.N.; Vargas, G.; Muniz, M.M.M.; Soares, M.A.M.; Cánovas, A.; Schenkel, F.; Squires, E.J. Differential Gene Expression in Dairy Cows under Negative Energy Balance and Ketosis: A Systematic Review and Meta-Analysis. J. Dairy Sci. 2021, 104, 602–615. [Google Scholar] [CrossRef] [PubMed]
- Van Der Kolk, J.H.; Gross, J.J.; Gerber, V.; Bruckmaier, R.M. Disturbed Bovine Mitochondrial Lipid Metabolism: A Review. Vet. Q. 2017, 37, 262–273. [Google Scholar] [CrossRef] [PubMed]
- Zhao, F.-Q. Biology of Glucose Transport in the Mammary Gland. J. Mammary Gland Biol. Neoplasia 2014, 19, 3–17. [Google Scholar] [CrossRef] [PubMed]
- Mellado, M.; Dávila, A.; Gaytán, L.; Macías-Cruz, U.; Avendaño-Reyes, L.; García, E. Risk Factors for Clinical Ketosis and Association with Milk Production and Reproduction Variables in Dairy Cows in a Hot Environment. Trop. Anim. Health Prod. 2018, 50, 1611–1616. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Li, X.; Li, Y.; Guan, Y.; Song, Y.; Yin, L.; Chen, H.; Lei, L.; Liu, J.; Li, X.; et al. Effects of Nonesterified Fatty Acids on the Synthesis and Assembly of Very Low Density Lipoprotein in Bovine Hepatocytes in Vitro. J. Dairy Sci. 2014, 97, 1328–1335. [Google Scholar] [CrossRef]
- Lanier, J.S.; Corl, B.A. Challenges in Enriching Milk Fat with Polyunsaturated Fatty Acids. J. Anim. Sci. Biotechnol. 2015, 6, 26. [Google Scholar] [CrossRef]
- Pascottini, O.B.; Leroy, J.L.M.R.; Opsomer, G. Metabolic Stress in the Transition Period of Dairy Cows: Focusing on the Prepartum Period. Animals 2020, 10, 1419. [Google Scholar] [CrossRef]
- Contreras, G.A.; Raphael, W.; Mattmiller, S.A.; Gandy, J.; Sordillo, L.M. Nonesterified Fatty Acids Modify Inflammatory Response and Eicosanoid Biosynthesis in Bovine Endothelial Cells. J. Dairy Sci. 2012, 95, 5011–5023. [Google Scholar] [CrossRef] [PubMed]
- Mylostyvyi, R.; Sejian, V.; Izhboldina, O.; Kalinichenko, O.; Karlova, L.; Lesnovskay, O.; Begma, N.; Marenkov, O.; Lykhach, V.; Midyk, S.; et al. Changes in the Spectrum of Free Fatty Acids in Blood Serum of Dairy Cows during a Prolonged Summer Heat Wave. Animals 2021, 11, 3391. [Google Scholar] [CrossRef] [PubMed]
- Ognik, K.; Patkowski, K.; Gruszecki, T.; Kostro, K. Redox Status in the Blood of Ewes in the Perinatal Period and during Lactation. Bull. Vet. Inst. Pulawy 2015, 59, 557–562. [Google Scholar] [CrossRef]
- Greco, L.F.; Neto, J.T.N.; Pedrico, A.; Ferrazza, R.A.; Lima, F.S.; Bisinotto, R.S.; Martinez, N.; Garcia, M.; Ribeiro, E.S.; Gomes, G.C.; et al. Effects of Altering the Ratio of Dietary N-6 to n-3 Fatty Acids on Performance and Inflammatory Responses to a Lipopolysaccharide Challenge in Lactating Holstein Cows. J. Dairy Sci. 2015, 98, 602–617. [Google Scholar] [CrossRef]
- Mezzetti, M.; Bionaz, M.; Trevisi, E. Interaction between Inflammation and Metabolism in Periparturient Dairy Cows. J. Anim. Sci. 2020, 98 (Suppl. 1), S155–S174. [Google Scholar] [CrossRef]
- Ling, T.; Hernandez-Jover, M.; Sordillo, L.M.; Abuelo, A. Maternal Late-Gestation Metabolic Stress Is Associated with Changes in Immune and Metabolic Responses of Dairy Calves. J. Dairy Sci. 2018, 101, 6568–6580. [Google Scholar] [CrossRef]
- Ohtsu, A.; Tanaka, H.; Seno, K.; Iwata, H.; Kuwayama, T.; Shirasuna, K. Palmitic Acid Stimulates Interleukin-8 via the TLR4/NF-κB/ROS Pathway and Induces Mitochondrial Dysfunction in Bovine Oviduct Epithelial Cells. Am. J. Reprod. Immunol. 2017, 77, e12642. [Google Scholar] [CrossRef]
- Meignan, T.; Lechartier, C.; Chesneau, G.; Bareille, N. Effects of Feeding Extruded Linseed on Production Performance and Milk Fatty Acid Profile in Dairy Cows: A Meta-Analysis. J. Dairy Sci. 2017, 100, 4394–4408. [Google Scholar] [CrossRef]
- Beauchemin, K.A. Invited Review: Current Perspectives on Eating and Rumination Activity in Dairy Cows. J. Dairy Sci. 2018, 101, 4762–4784. [Google Scholar] [CrossRef]
- Bauman, D.E.; Perfield, J.W.; De Veth, M.J.; Lock, A.L. New Perspectives on Lipid Digestion and Metabolism in Ruminants. In Proceedings Cornell Nutrition Conference; Cornell University: Ithaca, NY, USA, 2003; Volume 65, pp. 175–189. [Google Scholar]
- Abdelmegeid, M.K.; Elolimy, A.A.; Zhou, Z.; Lopreiato, V.; McCann, J.C.; Loor, J.J. Rumen-Protected Methionine during the Peripartal Period in Dairy Cows and Its Effects on Abundance of Major Species of Ruminal Bacteria. J. Anim. Sci. Biotechnol. 2018, 9, 17. [Google Scholar] [CrossRef]
- Kuczaj, M.; Preś, J.; Kinal, S.; Szulc, T.; Twardoń, J.; Łuczak, W. Przemiany w żwaczu kwasów tłuszczowych paszy i ich wpływ na skład chemiczny tłuszczu mleka. Vet. Med.-Sci. Pract. 2011, 67, 818–823. [Google Scholar]
- Buccioni, A.; Decandia, M.; Minieri, S.; Molle, G.; Cabiddu, A. Lipid Metabolism in the Rumen: New Insights on Lipolysis and Biohydrogenation with an Emphasis on the Role of Endogenous Plant Factors. Anim. Feed Sci. Technol. 2012, 174, 1–25. [Google Scholar] [CrossRef]
- McFadden, J.W. Review: Lipid Biology in the Periparturient Dairy Cow: Contemporary Perspectives. Animal 2020, 14, s165–s175. [Google Scholar] [CrossRef]
- Sun, X.; Hou, Y.; Wang, Y.; Guo, C.; Wang, Q.; Zhang, Y.; Yang, Z.; Wang, Z.; Cao, Z.; Wang, W.; et al. The Blood Immune Cell Count, Immunoglobulin, Inflammatory Factor, and Milk Trace Element in Transition Cows and Calves Were Altered by Increasing the Dietary n-3 or n-6 Polyunsaturated Fatty Acid Levels. Front. Immunol. 2022, 13, 897660. [Google Scholar] [CrossRef] [PubMed]
- Caldari-Torres, C.; McGilliard, M.L.; Corl, B.A. Esterification of Essential and Non-Essential Fatty Acids into Distinct Lipid Classes in Ruminant and Non-Ruminant Tissues. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2016, 200, 1–5. [Google Scholar] [CrossRef]
- Elgersma, A. Grazing Increases the Unsaturated Fatty Acid Concentration of Milk from Grass-fed Cows: A Review of the Contributing Factors, Challenges and Future Perspectives. Eur. J. Lipid Sci. Technol. 2015, 117, 1345–1369. [Google Scholar] [CrossRef]
- Phuoc Thanh, L.; Suksombat, W.; Loor, J.J.; Thi Thuy Hang, T. Polyunsaturated Fatty Acids and Rumen Undegradable Protein Alter Ruminal Fermentation and Milk Fatty Acid Profiles in Dairy Cows. Arch. Anim. Nutr. 2023, 77, 58–76. [Google Scholar] [CrossRef]
- Toral, P.G.; Monahan, F.J.; Hervás, G.; Frutos, P.; Moloney, A.P. Modulating Ruminal Lipid Metabolism to Improve the Fatty Acid Composition of Meat and Milk. Challenges and Opportunities. Animal 2018, 12, s272–s281. [Google Scholar] [CrossRef]
- Castillo-Lopez, E.; Jenkins, C.J.R.; Aluthge, N.D.; Tom, W.; Kononoff, P.J.; Fernando, S.C. The Effect of Regular or Reduced-fat Distillers Grains with Solubles on Rumen Methanogenesis and the Rumen Bacterial Community. J. Appl. Microbiol. 2017, 123, 1381–1395. [Google Scholar] [CrossRef]
- Neveu, C.; Baurhoo, B.; Mustafa, A. Effect of Feeding Extruded Flaxseed with Different Grains on the Performance of Dairy Cows and Milk Fatty Acid Profile. J. Dairy Sci. 2014, 97, 1543–1551. [Google Scholar] [CrossRef]
- Erickson, P.S.; Kalscheur, K.F. Nutrition and Feeding of Dairy Cattle. In Animal Agriculture; Elsevier: Amsterdam, The Netherlands, 2020; pp. 157–180. [Google Scholar] [CrossRef]
- Alves, S.P.; Mendonça, S.H.; Silva, J.L.; Bessa, R.J.B. Nannochloropsis Oceanica, a Novel Natural Source of Rumen-Protected Eicosapentaenoic Acid (EPA) for Ruminants. Sci. Rep. 2018, 8, 10269. [Google Scholar] [CrossRef] [PubMed]
- Anjum, F.M.; Haider, M.F.; Khan, M.I.; Sohaib, M.; Arshad, M.S. Impact of Extruded Flaxseed Meal Supplemented Diet on Growth Performance, Oxidative Stability and Quality of Broiler Meat and Meat Products. Lipids Health Dis. 2013, 12, 13. [Google Scholar] [CrossRef] [PubMed]
- Dawod, A.; Ahmed, H.; Abou-Elkhair, R.; Elbaz, H.T.; Taha, A.E.; Swelum, A.A.; Alhidary, I.A.; Saadeldin, I.M.; Al-Ghadi, M.Q.; Ba-Awadh, H.A.; et al. Effects of Extruded Linseed and Soybean Dietary Supplementation on Lactation Performance, First-Service Conception Rate, and Mastitis Incidence in Holstein Dairy Cows. Animals 2020, 10, 436. [Google Scholar] [CrossRef] [PubMed]
- Ababakri, R.; Dayani, O.; Khezri, A.; Naserian, A.-A. Influence of Flaxseed with Rumen Undegradable Protein Level on Milk Yield, Milk Fatty Acids and Blood Metabolites in Transition Ewes. J. Anim. Sci. Technol. 2021, 63, 475–490. [Google Scholar] [CrossRef] [PubMed]
- Bennato, F.; Ianni, A.; Innosa, D.; Grotta, L.; D’Onofrio, A.; Martino, G. Chemical-Nutritional Characteristics and Aromatic Profile of Milk and Related Dairy Products Obtained from Goats Fed with Extruded Linseed. Asian-Australas. J. Anim. Sci. 2020, 33, 148–156. [Google Scholar] [CrossRef]
- Singh, M.; Roy, A.K.; Sharma, S. Augmentation of Milk Production by Supplementing Bypass Fat in Dairy Animals. Asian J. Anim. Vet. Adv. 2015, 10, 476–488. [Google Scholar] [CrossRef][Green Version]
- Behan, A.A.; Loh, T.C.; Fakurazi, S.; Kaka, U.; Kaka, A.; Samsudin, A.A. Effects of Supplementation of Rumen Protected Fats on Rumen Ecology and Digestibility of Nutrients in Sheep. Animals 2019, 9, 400. [Google Scholar] [CrossRef]
- Mahé, C.; Lavigne, R.; Com, E.; Pineau, C.; Locatelli, Y.; Zlotkowska, A.M.; Almiñana, C.; Tsikis, G.; Mermillod, P.; Schoen, J.; et al. Spatiotemporal Profiling of the Bovine Oviduct Fluid Proteome around the Time of Ovulation. Sci. Rep. 2022, 12, 4135. [Google Scholar] [CrossRef]
- O’Doherty, A.M.; O’Gorman, A.; Al Naib, A.; Brennan, L.; Daly, E.; Duffy, P.; Fair, T. Negative Energy Balance Affects Imprint Stability in Oocytes Recovered from Postpartum Dairy Cows. Genomics 2014, 104, 177–185. [Google Scholar] [CrossRef]
- Leroy, J.; Sturmey, R.; Van Hoeck, V.; De Bie, J.; McKeegan, P.; Bols, P. Dietary Fat Supplementation and the Consequences for Oocyte and Embryo Quality: Hype or Significant Benefit for Dairy Cow Reproduction? Reprod. Domest. Anim. 2014, 49, 353–361. [Google Scholar] [CrossRef]
- Roque-Jiménez, J.A.; Rosa-Velázquez, M.; Pinos-Rodríguez, J.M.; Vicente-Martínez, J.G.; Mendoza-Cervantes, G.; Flores-Primo, A.; Lee-Rangel, H.A.; Relling, A.E. Role of Long Chain Fatty Acids in Developmental Programming in Ruminants. Animals 2021, 11, 762. [Google Scholar] [CrossRef] [PubMed]
- Oseikria, M.; Elis, S.; Maillard, V.; Corbin, E.; Uzbekova, S. N-3 Polyunsaturated Fatty Acid DHA during IVM Affected Oocyte Developmental Competence in Cattle. Theriogenology 2016, 85, 1625–1634.e2. [Google Scholar] [CrossRef]
- D’Occhio, M.J.; Baruselli, P.S.; Campanile, G. Influence of Nutrition, Body Condition, and Metabolic Status on Reproduction in Female Beef Cattle: A Review. Theriogenology 2019, 125, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Baddela, V.S.; Roettgen, V.; Vernunft, A.; Viergutz, T.; Dannenberger, D.; Hammon, H.M.; Schoen, J.; Vanselow, J. Effects of Dietary Fatty Acids on Bovine Oocyte Competence and Granulosa Cells. Front. Endocrinol. 2020, 11, 87. [Google Scholar] [CrossRef] [PubMed]
- Dirandeh, E.; Towhidi, A.; Ansari Pirsaraei, Z.; Saberifar, T.; Akhlaghi, A.; Rezaei Roodbari, A. The Endometrial Expression of Prostaglandin Cascade Components in Lactating Dairy Cows Fed Different Polyunsaturated Fatty Acids. Theriogenology 2015, 83, 206–212. [Google Scholar] [CrossRef]
- Hatzirodos, N.; Irving-Rodgers, H.F.; Hummitzsch, K.; Harland, M.L.; Morris, S.E.; Rodgers, R.J. Transcriptome Profiling of Granulosa Cells of Bovine Ovarian Follicles during Growth from Small to Large Antral Sizes. BMC Genom. 2014, 15, 24. [Google Scholar] [CrossRef]
- Gu, L.; Liu, H.; Gu, X.; Boots, C.; Moley, K.H.; Wang, Q. Metabolic Control of Oocyte Development: Linking Maternal Nutrition and Reproductive Outcomes. Cell. Mol. Life Sci. 2015, 72, 251–271. [Google Scholar] [CrossRef]
- Rybska, M.; Knap, S.; Jankowski, M.; Jeseta, M.; Bukowska, D.; Antosik, P.; Nowicki, M.; Zabel, M.; Kempisty, B.; Jaśkowski, J.M. Cytoplasmic and Nuclear Maturation of Oocytes in Mammals—Living in the Shadow of Cells Developmental Capability. Med. J. Cell Biol. 2018, 6, 13–17. [Google Scholar] [CrossRef]
- Gutnisky, C.; Morado, S.; Gadze, T.; Donato, A.; Alvarez, G.; Dalvit, G.; Cetica, P. Morphological, Biochemical and Functional Studies to Evaluate Bovine Oocyte Vitrification. Theriogenology 2020, 143, 18–26. [Google Scholar] [CrossRef]
- Dunning, K.R.; Russell, D.L.; Robker, R.L. Lipids and Oocyte Developmental Competence: The Role of Fatty Acids and β-Oxidation. Reproduction 2014, 148, R15–R27. [Google Scholar] [CrossRef]
- Nikoloff, N.; Campagna, A.; Luchetti, C.; Carranza-Martín, A.C.; Pascua, A.M.; Anchordoquy, J.M.; Anchordoquy, J.P.; Lombardo, D.M.; Seoane, A.; Furnus, C.C. Effects of EPA on Bovine Oocytes Matured in Vitro with Antioxidants: Impact on the Lipid Content of Oocytes and Early Embryo Development. Theriogenology 2020, 146, 152–161. [Google Scholar] [CrossRef] [PubMed]
- Prates, E.G.; Nunes, J.T.; Pereira, R.M. A Role of Lipid Metabolism during Cumulus-Oocyte Complex Maturation: Impact of Lipid Modulators to Improve Embryo Production. Mediat. Inflamm. 2014, 2014, 692067. [Google Scholar] [CrossRef] [PubMed]
- Plante-Dubé, M.; Picard, C.; Gilbert, I.; Robert, C.; Fievez, V.; Vlaeminck, B.; Belleannée, C.; Gervais, R.; Chouinard, P.Y. Effects of a Dietary Supplement Enriched in Palmitoleic Acid on Fatty Acid Composition of Follicular Fluid, Granulosa Cell Metabolism, and Oocyte Developmental Capacity in Early Lactation Dairy Cows. J. Dairy Sci. 2021, 104, 3693–3706. [Google Scholar] [CrossRef]
- Zarezadeh, R.; Mehdizadeh, A.; Leroy, J.L.M.R.; Nouri, M.; Fayezi, S.; Darabi, M. Action Mechanisms of N-3 Polyunsaturated Fatty Acids on the Oocyte Maturation and Developmental Competence: Potential Advantages and Disadvantages. J. Cell. Physiol. 2019, 234, 1016–1029. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, C.J.T.; Lima, A.C.B.D.; Silva, W.A.L.D.; Souza-Cáceres, M.B.; Pereira, D.M.; Oliveira, M.V.M.D.; Nogueira, É.; Melo-Sterza, F.D.A. Supplementation of Cows with Linseed: Effects on Antral Follicle Count, Oocyte Quality, and Nutritional and Plasma Metabolite Parameters. Semin. Ciênc. Agrár. 2019, 40, 2637. [Google Scholar] [CrossRef]
- Moallem, U.; Shafran, A.; Zachut, M.; Dekel, I.; Portnick, Y.; Arieli, A. Dietary α-Linolenic Acid from Flaxseed Oil Improved Folliculogenesis and IVF Performance in Dairy Cows, Similar to Eicosapentaenoic and Docosahexaenoic Acids from Fish Oil. Reproduction 2013, 146, 603–614. [Google Scholar] [CrossRef] [PubMed]
- Doyle, D.N.; Lonergan, P.; Diskin, M.G.; Pierce, K.M.; Kelly, A.K.; Stanton, C.; Waters, S.M.; Parr, M.H.; Kenny, D.A. Effect of Dietary N-3 Polyunsaturated Fatty Acid Supplementation and Post-Insemination Plane of Nutrition on Systemic Concentrations of Metabolic Analytes, Progesterone, Hepatic Gene Expression and Embryo Development and Survival in Beef Heifers. Theriogenology 2019, 127, 102–113. [Google Scholar] [CrossRef]
- Mahla, A.S.; Chaudhari, R.K.; Verma, A.K.; Singh, A.K.; Singh, S.K.; Singh, G.; Sarkar, M.; Dutta, N.; Kumar, H.; Krishnaswamy, N. Effect of Dietary Supplementation of Omega-3 Polyunsaturated Fatty Acid (PUFA) Rich Fish Oil on Reproductive Performance of the Goat (Capra hircus). Theriogenology 2017, 99, 79–89. [Google Scholar] [CrossRef]
- Giller, K.; Drews, B.; Berard, J.; Kienberger, H.; Schmicke, M.; Frank, J.; Spanier, B.; Daniel, H.; Geisslinger, G.; Ulbrich, S.E. Bovine Embryo Elongation Is Altered Due to Maternal Fatty Acid Supplementation. Biol. Reprod. 2018, 99, 600–610. [Google Scholar] [CrossRef]
- Leng, X.; Jiang, H. Effects of Arachidonic Acid and Its Major Prostaglandin Derivatives on Bovine Myoblast Proliferation, Differentiation, and Fusion. Domest. Anim. Endocrinol. 2019, 67, 28–36. [Google Scholar] [CrossRef]
- Szóstek-Mioduchowska, A.Z.; Shiotani, H.; Yamamoto, Y.; Sadowska, A.; Wójtowicz, A.; Kozai, K.; Hojo, T.; Kimura, K.; Skarzynski, D.J.; Okuda, K. Effects of Cortisol on Prostaglandin F2α Secretion and Expression of Genes Involved in the Arachidonic Acid Metabolic Pathway in Equine Endometrium—In Vitro Study. Theriogenology 2021, 173, 221–229. [Google Scholar] [CrossRef]
- Anamthathmakula, P.; Winuthayanon, W. Prostaglandin-Endoperoxide Synthase 2 (PTGS2) in the Oviduct: Roles in Fertilization and Early Embryo Development. Endocrinology 2021, 162, bqab025. [Google Scholar] [CrossRef]
- Rahbar, B.; Safdar, A.H.A.; Kor, N.M. Mechanisms through Which Fat Supplementation Could Enhance Reproduction in Farm Animal. Eur. J. Exp. Biol. 2014, 4, 340–348. [Google Scholar]
- Socha, B.M.; Łada, P.; Jończyk, A.W.; Korzekwa, A.J.; Skarżyński, D.J. The Role of Peroxisome Proliferator-Activated Receptors in PGF2α-Induced Luteolysis in the Bovine Corpus Luteum. Animals 2022, 12, 1542. [Google Scholar] [CrossRef]
- Sakai, S.; Hagihara, N.; Kuse, M.; Kimura, K.; Okuda, K. Heat Stress Affects Prostaglandin Synthesis in Bovine Endometrial Cells. J. Reprod. Dev. 2018, 64, 311–317. [Google Scholar] [CrossRef]
- Plewes, M.R.; Burns, P.D.; Hyslop, R.M.; George Barisas, B. Influence of Omega-3 Fatty Acids on Bovine Luteal Cell Plasma Membrane Dynamics. Biochim. Biophys. Acta BBA-Biomembr. 2017, 1859, 2413–2419. [Google Scholar] [CrossRef]
- Soydan, E.; Şen, U.; Şirin, E. Relationship Between Dietary Fatty Acids and Reproductive Functions in Dairy Cattle. Turk. J. Agric.-Food Sci. Technol. 2017, 5, 1575. [Google Scholar] [CrossRef]
- Silvestre, F.T.; Carvalho, T.S.M.; Francisco, N.; Santos, J.E.P.; Staples, C.R.; Jenkins, T.C.; Thatcher, W.W. Effects of differential supplementation of fatty acids during the peripartum and breeding periods of Holstein cows: I. Uterine and metabolic responses, reproduction, and lactation. J. Dairy Sci. 2011, 94, 189–204. [Google Scholar] [CrossRef]
- Park, H.G.; Kothapalli, K.S.D.; Park, W.J.; DeAllie, C.; Liu, L.; Liang, A.; Lawrence, P.; Brenna, J.T. Palmitic Acid (16:0) Competes with Omega-6 Linoleic and Omega-3 ɑ-Linolenic Acids for FADS2 Mediated Δ6-Desaturation. Biochim. Biophys. Acta BBA-Mol. Cell Biol. Lipids 2016, 1861, 91–97. [Google Scholar] [CrossRef]
- Elis, S.; Freret, S.; Desmarchais, A.; Maillard, V.; Cognié, J.; Briant, E.; Touzé, J.-L.; Dupont, M.; Faverdin, P.; Chajès, V.; et al. Effect of a Long Chain N-3 PUFA-Enriched Diet on Production and Reproduction Variables in Holstein Dairy Cows. Anim. Reprod. Sci. 2016, 164, 121–132. [Google Scholar] [CrossRef]
- Freret, S.; Oseikria, M.; Bourhis, D.L.; Desmarchais, A.; Briant, E.; Desnoes, O.; Dupont, M.; Le Berre, L.; Ghazouani, O.; Bertevello, P.S.; et al. Effects of a N-3 Polyunsaturated Fatty Acid-Enriched Diet on Embryo Production in Dairy Cows. Reproduction 2019, 158, 71–83. [Google Scholar] [CrossRef] [PubMed]
- Ulfina, G.G.; Kimothi, S.P.; Oberoi, P.S.; Baithalu, R.K.; Kumaresan, A.; Mohanty, T.K.; Imtiwati, P.; Dang, A.K. Modulation of Post-Partum Reproductive Performance in Dairy Cows through Supplementation of Long- or Short-Chain Fatty Acids during Transition Period. J. Anim. Physiol. Anim. Nutr. 2015, 99, 1056–1064. [Google Scholar] [CrossRef] [PubMed]
- Sinedino, L.D.P.; Honda, P.M.; Souza, L.R.L.; Lock, A.L.; Boland, M.P.; Staples, C.R.; Thatcher, W.W.; Santos, J.E.P. Effects of Supplementation with Docosahexaenoic Acid on Reproduction of Dairy Cows. Reproduction 2017, 153, 707–723. [Google Scholar] [CrossRef] [PubMed]
- Winders, T.M.; Holman, D.B.; Schmidt, K.N.; Luecke, S.M.; Smith, D.J.; Neville, B.W.; Dahlen, C.R.; Swanson, K.C.; Amat, S. Feeding Hempseed Cake Alters the Bovine Gut, Respiratory and Reproductive Microbiota. Sci. Rep. 2023, 13, 8121. [Google Scholar] [CrossRef]
- Karas, J.A.; Wong, L.J.M.; Paulin, O.K.A.; Mazeh, A.C.; Hussein, M.H.; Li, J.; Velkov, T. The Antimicrobial Activity of Cannabinoids. Antibiotics 2020, 9, 406. [Google Scholar] [CrossRef]
- Gábor, G.; Balogh, O.G.; Kern, L.; Gábor, P.R.; Fébel, H. Nutrition, Metabolic Status and Reproductive Efficiency in Dairy Herds. Open, J. Anim. Sci. 2016, 6, 75–84. [Google Scholar] [CrossRef]
- Dash, S.; Chakravarty, A.K.; Singh, A.; Upadhyay, A.; Singh, M.; Yousuf, S. Effect of Heat Stress on Reproductive Performances of Dairy Cattle and Buffaloes: A Review. Vet. World 2016, 9, 235–244. [Google Scholar] [CrossRef]
- Kumar, S.; Sihag, S.; Sihag, Z.S.; Patil, C.S.; Dhaka, S.S.; Pandey, A.K. Effect of Dietary Enrichment with Omega 3 and 6 Fatty Acids on Blood Metabolites, Hormone Concentration and Ovarian Function in Sahiwal Heifers. Indian J. Anim. Res. 2021, 56, 1084–1089. [Google Scholar] [CrossRef]
- Swanepoel, N.; Robinson, P.H. Impacts of Feeding a Flax-Seed Based Feed Supplement on Productive and Reproductive Performance of Early Lactation Multiparous Holstein Cows. Anim. Feed Sci. Technol. 2019, 251, 134–152. [Google Scholar] [CrossRef]
- González, L.; Moreno, T.; Bispo, E.; Dugan, M.E.R.; Franco, D. Effect of Supplementing Different Oils: Linseed, Sunflower and Soybean, on Animal Performance, Carcass Characteristics, Meat Quality and Fatty Acid Profile of Veal from “Rubia Gallega” Calves. Meat Sci. 2014, 96, 829–836. [Google Scholar] [CrossRef]
- Rosa Velazquez, M.; Batistel, F.; Pinos Rodriguez, J.M.; Relling, A.E. Effects of Maternal Dietary Omega-3 Polyunsaturated Fatty Acids and Methionine during Late Gestation on Fetal Growth, DNA Methylation, and mRNA Relative Expression of Genes Associated with the Inflammatory Response, Lipid Metabolism and DNA Methylation in Placenta and Offspring’s Liver in Sheep. J. Anim. Sci. Biotechnol. 2020, 11, 111. [Google Scholar] [CrossRef]
- Jolazadeh, A.R.; Mohammadabadi, T.; Dehghan-banadaky, M.; Chaji, M.; Garcia, M. Effect of Supplementation Fat during the Last 3 Weeks of Uterine Life and the Preweaning Period on Performance, Ruminal Fermentation, Blood Metabolites, Passive Immunity and Health of the Newborn Calf. Br. J. Nutr. 2019, 122, 1346–1358. [Google Scholar] [CrossRef] [PubMed]
- Liermann, W.; Uken, K.L.; Schäff, C.; Vogel, L.; Gnott, M.; Tuchscherer, A.; Trevisi, E.; Stefaniak, T.; Sauerwein, H.; Tröscher, A.; et al. Effects of a Maternal Essential Fatty Acid and Conjugated Linoleic Acid Supplementation during Late Pregnancy and Early Lactation on Hematologic and Immunological Traits and the Oxidative and Anti-Oxidative Status in Blood Plasma of Neonatal Calves. Animals 2021, 11, 2168. [Google Scholar] [CrossRef] [PubMed]
- Pi, Y.; Gao, S.T.; Ma, L.; Zhu, Y.X.; Wang, J.Q.; Zhang, J.M.; Xu, J.C.; Bu, D.P. Effectiveness of rubber seed oil and flaxseed oil to enhance the α-linolenic acid content in milk from dairy cows. J. Dairy Sci. 2016, 99, 5719–5730. [Google Scholar] [CrossRef] [PubMed]
- Lopreiato, V.; Mezzetti, M.; Cattaneo, L.; Ferronato, G.; Minuti, A.; Trevisi, E. Role of Nutraceuticals during the Transition Period of Dairy Cows: A Review. J. Anim. Sci. Biotechnol. 2020, 11, 96. [Google Scholar] [CrossRef]
- Ighodaro, O.M.; Akinloye, O.A. First Line Defence Antioxidants-Superoxide Dismutase (SOD), Catalase (CAT) and Glutathione Peroxidase (GPX): Their Fundamental Role in the Entire Antioxidant Defence Grid. Alex. J. Med. 2018, 54, 287–293. [Google Scholar] [CrossRef]
- Brito, A.; Zang, Y. A Review of Lignan Metabolism, Milk Enterolactone Concentration, and Antioxidant Status of Dairy Cows Fed Flaxseed. Molecules 2018, 24, 41. [Google Scholar] [CrossRef] [PubMed]
- Santos, N.W.; Yoshimura, E.H.; Machado, E.; Matumoto-Pintro, P.T.; Montanher, P.F.; Visentainer, J.V.; Dos Santos, G.T.; Zeoula, L.M. Antioxidant Effects of a Propolis Extract and Vitamin E in Blood and Milk of Dairy Cows Fed Diet Containing Flaxseed Oil. Livest. Sci. 2016, 191, 132–138. [Google Scholar] [CrossRef]
- Huang, G.; Wang, J.; Liu, K.; Wang, F.; Zheng, N.; Zhao, S.; Qu, X.; Yu, J.; Zhang, Y.; Wang, J. Effect of Flaxseed Supplementation on Milk and Plasma Fatty Acid Composition and Plasma Parameters of Holstein Dairy Cows. Animals 2022, 12, 1898. [Google Scholar] [CrossRef]
- Chhillar, H.; Chopra, P.; Ashfaq, M.A. Lignans from Linseed (Linum usitatissimum, L.) and Its Allied Species: Retrospect, Introspect and Prospect. Crit. Rev. Food Sci. Nutr. 2021, 61, 2719–2741. [Google Scholar] [CrossRef]
- Imran, M.; Ahmad, N.; Anjum, F.M.; Khan, M.K.; Mushtaq, Z.; Nadeem, M.; Hussain, S. Potential Protective Properties of Flax Lignan Secoisolariciresinol Diglucoside. Nutr. J. 2015, 14, 71. [Google Scholar] [CrossRef]
- Gandra, J.R.; Barletta, R.V.; Mingoti, R.D.; Verdurico, L.C.; Freitas, J.E., Jr.; Oliveira, L.J.; Takiya, C.S.; Kfoury, J.R., Jr.; Wiltbank, M.C.; Renno, F.P. Effects of Whole Flaxseed, Raw Soybeans, and Calcium Salts of Fatty Acids on Measures of Cellular Immune Function of Transition Dairy Cows. J. Dairy Sci. 2016, 99, 4590–4606. [Google Scholar] [CrossRef] [PubMed]
- Xia, W.; Xie, W. Rebooting the Epigenomes during Mammalian Early Embryogenesis. Stem Cell Rep. 2020, 15, 1158–1175. [Google Scholar] [CrossRef] [PubMed]
- Kra, G.; Nemes-Navon, N.; Daddam, J.R.; Livshits, L.; Jacoby, S.; Levin, Y.; Zachut, M.; Moallem, U. Proteomic Analysis of Peripheral Blood Mononuclear Cells and Inflammatory Status in Postpartum Dairy Cows Supplemented with Different Sources of Omega-3 Fatty Acids. J. Proteom. 2021, 246, 104313. [Google Scholar] [CrossRef] [PubMed]
- Haudek-Prinz, V.J.; Klepeisz, P.; Slany, A.; Griss, J.; Meshcheryakova, A.; Paulitschke, V.; Mitulovic, G.; Stöckl, J.; Gerner, C. Proteome Signatures of Inflammatory Activated Primary Human Peripheral Blood Mononuclear Cells. J. Proteom. 2012, 76, 150–162. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, D.M.; Salama, A.A.K.; Vitor, A.C.M.; Argüello, A.; Moncau, C.T.; Santos, E.M.; Caja, G.; De Oliveira, J.S.; Balieiro, J.C.C.; Hernández-Castellano, L.E.; et al. The Application of Omics in Ruminant Production: A Review in the Tropical and Sub-Tropical Animal Production Context. J. Proteomics 2020, 227, 103905. [Google Scholar] [CrossRef] [PubMed]
- Bragaglio, A.; Braghieri, A.; Napolitano, F.; De Rosa, G.; Riviezzi, A.M.; Surianello, F.; Pacelli, C. Omega-3 Supplementation, Milk Quality and Cow Immune-Competence. Ital. J. Agron. 2015, 10, 9–14. [Google Scholar] [CrossRef]
- Brett, K.; Ferraro, Z.; Yockell-Lelievre, J.; Gruslin, A.; Adamo, K. Maternal–Fetal Nutrient Transport in Pregnancy Pathologies: The Role of the Placenta. Int. J. Mol. Sci. 2014, 15, 16153–16185. [Google Scholar] [CrossRef]
- Alagawany, M.; Elnesr, S.S.; Farag, M.R.; El-Sabrout, K.; Alqaisi, O.; Dawood, M.A.O.; Soomro, H.; Abdelnour, S.A. Nutritional Significance and Health Benefits of Omega-3, -6 and -9 Fatty Acids in Animals. Anim. Biotechnol. 2022, 33, 1678–1690. [Google Scholar] [CrossRef]
- Lonergan, P.; Fair, T.; Forde, N.; Rizos, D. Embryo Development in Dairy Cattle. Theriogenology 2016, 86, 270–277. [Google Scholar] [CrossRef]
- Marques, R.S.; Cooke, R.F.; Rodrigues, M.C.; Brandão, A.P.; Schubach, K.M.; Lippolis, K.D.; Moriel, P.; Perry, G.A.; Lock, A.; Bohnert, D.W. Effects of Supplementing Calcium Salts of Polyunsaturated Fatty Acids to Late-Gestating Beef Cows on Performance and Physiological Responses of the Offspring1. J. Anim. Sci. 2017, 95, 5347–5357. [Google Scholar] [CrossRef] [PubMed]
- Meyer, A.M.; Caton, J.S. Role of the Small Intestine in Developmental Programming: Impact of Maternal Nutrition on the Dam and Offspring. Adv. Nutr. 2016, 7, 169–178. [Google Scholar] [CrossRef] [PubMed]
- Opgenorth, J.; Sordillo, L.M.; Lock, A.L.; Gandy, J.C.; VandeHaar, M.J. Colostrum Supplementation with N-3 Fatty Acids Alters Plasma Polyunsaturated Fatty Acids and Inflammatory Mediators in Newborn Calves. J. Dairy Sci. 2020, 103, 11676–11688. [Google Scholar] [CrossRef] [PubMed]
- Khanal, P.; Nielsen, M.O. Impacts of Prenatal Nutrition on Animal Production and Performance: A Focus on Growth and Metabolic and Endocrine Function in Sheep. J. Anim. Sci. Biotechnol. 2017, 8, 75. [Google Scholar] [CrossRef] [PubMed]
- Kabaran, S.; Besler, H.T. Do Fatty Acids Affect Fetal Programming? J. Health Popul. Nutr. 2015, 33, 14. [Google Scholar] [CrossRef]
- Chankeaw, W.; Lignier, S.; Richard, C.; Ntallaris, T.; Raliou, M.; Guo, Y.; Plassard, D.; Bevilacqua, C.; Sandra, O.; Andersson, G.; et al. Analysis of the Transcriptome of Bovine Endometrial Cells Isolated by Laser Micro-Dissection (2): Impacts of Post-Partum Negative Energy Balance on Stromal, Glandular and Luminal Epithelial Cells. BMC Genom. 2021, 22, 450. [Google Scholar] [CrossRef]
- Staud, F.; Karahoda, R. Trophoblast: The Central Unit of Fetal Growth, Protection and Programming. Int. J. Biochem. Cell Biol. 2018, 105, 35–40. [Google Scholar] [CrossRef]
- Madoz, L.V.; Rabaglino, M.B.; Migliorisi, A.L.; Jaureguiberry, M.; Perez Wallace, S.; Lorenti, N.; Domínguez, G.; Giuliodori, M.J.; De La Sota, R.L. Association between Progesterone Concentration and Endometrial Gene Expression in Dairy Cows. Domest. Anim. Endocrinol. 2021, 74, 106481. [Google Scholar] [CrossRef]
- Jaśkowski, B.M.; Opałka, A.; Gehrke, M.; Herudzińska, M.; Czeladko, J.; Baumgartner, W.; Jaśkowski, J.M. A Critical Overview on Prostaglandin Inhibitors and Their Influence on Pregnancy Results after Insemination and Embryo Transfer in Cows. Animals 2021, 11, 3368. [Google Scholar] [CrossRef]
- Carneiro, L.C.; Williams, E.J.; Saut, J.P.E.; Santos, R.M.D.; Celeghini, E.C.C. The Effect of N- 3 Polyunsaturated Fatty Acid Supplementation on Immune and Reproductive Parameters in Dairy Cows: A Review. Braz. J. Vet. Res. Anim. Sci. 2021, 58, e175224. [Google Scholar] [CrossRef]
- Brooks, K.; Burns, G.; Spencer, T.E. Conceptus Elongation in Ruminants: Roles of Progesterone, Prostaglandin, Interferon Tau and Cortisol. J. Anim. Sci. Biotechnol. 2014, 5, 53. [Google Scholar] [CrossRef] [PubMed]
- Kowalczyk, A.; Czerniawska-Piątkowska, E.; Wrzecińska, M. The Importance of Interferon-Tau in the Diagnosis of Pregnancy. BioMed Res. Int. 2021, 2021, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Chaney, H.L.; Grose, L.F.; Charpigny, G.; Behura, S.K.; Sheldon, I.M.; Cronin, J.G.; Lonergan, P.; Spencer, T.E.; Mathew, D.J. Conceptus-Induced, Interferon Tau-Dependent Gene Expression in Bovine Endometrial Epithelial and Stromal Cells. Biol. Reprod. 2021, 104, 669–683. [Google Scholar] [CrossRef]
- Sponchiado, M.; Gomes, N.S.; Fontes, P.K.; Martins, T.; Del Collado, M.; Pastore, A.D.A.; Pugliesi, G.; Nogueira, M.F.G.; Binelli, M. Pre-Hatching Embryo-Dependent and -Independent Programming of Endometrial Function in Cattle. PLoS ONE 2017, 12, e0175954. [Google Scholar] [CrossRef] [PubMed]
- Matras, J.; Kowalczuk-Vasilev, E.; Klebaniuk, R.; Grela, E. Influence of Two Flaxseed Varieties, Differing in Fatty Acid Profile, in Dairy Cow Diets on Selected Blood Indices and Reproduction. Vet. Med.-Sci. Pract. 2014, 70, 422–427. [Google Scholar]
- Azam, A.; Shahzad, Q.; Ul-Husna, A.; Qadeer, S.; Ejaz, R.; Fouladi-Nashta, A.A.; Khalid, M.; Ullah, N.; Akhtar, T.; Akhter, S. Supplementing ±-Linolenic Acid in the in Vitro Maturation Media Improves Nuclear Maturation Rate of Oocytes and Early Embryonic Development in the Nili Ravi Buffalo. Anim. Reprod. 2017, 14, 1161–1169. [Google Scholar] [CrossRef]
- Shrestha, N.; Sleep, S.L.; Cuffe, J.S.M.; Holland, O.J.; Perkins, A.V.; Yau, S.Y.; McAinch, A.J.; Hryciw, D.H. Role of Omega-6 and Omega-3 Fatty Acids in Fetal Programming. Clin. Exp. Pharmacol. Physiol. 2020, 47, 907–915. [Google Scholar] [CrossRef]
- Uken, K.L.; Schäff, C.T.; Vogel, L.; Gnott, M.; Dannenberger, D.; Görs, S.; Tuchscherer, A.; Tröscher, A.; Liermann, W.; Hammon, H.M. Modulation of Colostrum Composition and Fatty Acid Status in Neonatal Calves by Maternal Supplementation with Essential Fatty Acids and Conjugated Linoleic Acid Starting in Late Lactation. J. Dairy Sci. 2021, 104, 4950–4969. [Google Scholar] [CrossRef]
- Zhang, S.; Regnault, T.; Barker, P.; Botting, K.; McMillen, I.; McMillan, C.; Roberts, C.; Morrison, J. Placental Adaptations in Growth Restriction. Nutrients 2015, 7, 360–389. [Google Scholar] [CrossRef]
- Zhang, T.; Xu, Z.; Wen, L.; Lei, D.; Li, S.; Wang, J.; Huang, J.; Wang, N.; Durkan, C.; Liao, X.; et al. Cadmium-Induced Dysfunction of the Blood-Brain Barrier Depends on ROS-Mediated Inhibition of PTPase Activity in Zebrafish. J. Hazard. Mater. 2021, 412, 125198. [Google Scholar] [CrossRef]
- Larqué, E.; Pagán, A.; Prieto, M.T.; Blanco, J.E.; Gil-Sánchez, A.; Zornoza-Moreno, M.; Ruiz-Palacios, M.; Gázquez, A.; Demmelmair, H.; Parrilla, J.J.; et al. Placental Fatty Acid Transfer: A Key Factor in Fetal Growth. Ann. Nutr. Metab. 2014, 64, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Desantadina, R.; Quintana, S.; Recavarren, M.I.; Relling, A.E. Effect of Time of Gestation on Fatty Acid Transporters Mrna Expression in Bovine Placenta. Biosci. J. 2018, 34, 180–185. [Google Scholar] [CrossRef]
- Garcia, M.; Shin, J.H.; Schlaefli, A.; Greco, L.F.; Maunsell, F.P.; Santos, J.E.P.; Staples, C.R.; Thatcher, W.W. Increasing Intake of Essential Fatty Acids from Milk Replacer Benefits Performance, Immune Responses, and Health of Preweaned Holstein Calves. J. Dairy Sci. 2015, 98, 458–477. [Google Scholar] [CrossRef] [PubMed]
- Salehi, R.; Colazo, M.G.; Tsoi, S.; Behrouzi, A.; Tsang, B.K.; Dyck, M.K.; Oba, M.; Ambrose, D.J. Morphologic and Transcriptomic Assessment of Bovine Embryos Exposed to Dietary Long-Chain Fatty Acids. Reproduction 2016, 152, 715–726. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Saini, R.K.; Keum, Y.-S. Omega-3 and Omega-6 Polyunsaturated Fatty Acids: Dietary Sources, Metabolism, and Significance—A Review. Life Sci. 2018, 203, 255–267. [Google Scholar] [CrossRef]
- Zeng, X.; Li, S.; Liu, L.; Cai, S.; Ye, Q.; Xue, B.; Wang, X.; Zhang, S.; Chen, F.; Cai, C.; et al. Role of Functional Fatty Acids in Modulation of Reproductive Potential in Livestock. J. Anim. Sci. Biotechnol. 2023, 14, 24. [Google Scholar] [CrossRef]
- Sambra, V.; Echeverria, F.; Valenzuela, A.; Chouinard-Watkins, R.; Valenzuela, R. Docosahexaenoic and Arachidonic Acids as Neuroprotective Nutrients throughout the Life Cycle. Nutrients 2021, 13, 986. [Google Scholar] [CrossRef]
- Terry, S.A.; Basarab, J.A.; Guan, L.L.; McAllister, T.A. Strategies to Improve the Efficiency of Beef Cattle Production. Can. J. Anim. Sci. 2021, 101, 1–19. [Google Scholar] [CrossRef]
- Trevisi, E.; Minuti, A. Assessment of the Innate Immune Response in the Periparturient Cow. Res. Vet. Sci. 2018, 116, 47–54. [Google Scholar] [CrossRef]
- Bertoni, G.; Minuti, A.; Trevisi, E. Immune System, Inflammation and Nutrition in Dairy Cattle. Anim. Prod. Sci. 2015, 55, 943. [Google Scholar] [CrossRef]
- Sordillo, L.M. Nutritional Strategies to Optimize Dairy Cattle Immunity. J. Dairy Sci. 2016, 99, 4967–4982. [Google Scholar] [CrossRef] [PubMed]
- Arnott, G.; Ferris, C.P.; O’Connell, N.E. Review: Welfare of Dairy Cows in Continuously Housed and Pasture-Based Production Systems. Animal 2017, 11, 261–273. [Google Scholar] [CrossRef]
- Hulbert, L.E.; Moisá, S.J. Stress, Immunity, and the Management of Calves. J. Dairy Sci. 2016, 99, 3199–3216. [Google Scholar] [CrossRef] [PubMed]
- Chase, C.; Kaushik, R.S. Mucosal Immune System of Cattle. Vet. Clin. N. Am. Food Anim. Pract. 2019, 35, 431–451. [Google Scholar] [CrossRef] [PubMed]
- Van Knegsel, A.T.M.; Remmelink, G.J.; Jorjong, S.; Fievez, V.; Kemp, B. Effect of Dry Period Length and Dietary Energy Source on Energy Balance, Milk Yield, and Milk Composition of Dairy Cows. J. Dairy Sci. 2014, 97, 1499–1512. [Google Scholar] [CrossRef]
- Jahan, N.; Minuti, A.; Trevisi, E. Assessment of Immune Response in Periparturient Dairy Cows Using Ex Vivo Whole Blood Stimulation Assay with Lipopolysaccharides and Carrageenan Skin Test. Vet. Immunol. Immunopathol. 2015, 165, 119–126. [Google Scholar] [CrossRef]
- Wankhade, P.R.; Manimaran, A.; Kumaresan, A.; Jeyakumar, S.; Ramesha, K.P.; Sejian, V.; Rajendran, D.; Varghese, M.R. Metabolic and Immunological Changes in Transition Dairy Cows: A Review. Vet. World 2017, 10, 1367–1377. [Google Scholar] [CrossRef]
- Vlasova, A.N.; Saif, L.J. Bovine Immunology: Implications for Dairy Cattle. Front. Immunol. 2021, 12, 643206. [Google Scholar] [CrossRef]
- Ulfman, L.H.; Leusen, J.H.W.; Savelkoul, H.F.J.; Warner, J.O.; Van Neerven, R.J.J. Effects of Bovine Immunoglobulins on Immune Function, Allergy, and Infection. Front. Nutr. 2018, 5, 52. [Google Scholar] [CrossRef]
- Sordillo, L.M.; Mavangira, V. The Nexus between Nutrient Metabolism, Oxidative Stress and Inflammation in Transition Cows. Anim. Prod. Sci. 2014, 54, 1204. [Google Scholar] [CrossRef]
- Abuelo, A.; Hernández, J.; Benedito, J.L.; Castillo, C. The Importance of the Oxidative Status of Dairy Cattle in the Periparturient Period: Revisiting Antioxidant Supplementation. J. Anim. Physiol. Anim. Nutr. 2015, 99, 1003–1016. [Google Scholar] [CrossRef] [PubMed]
- Hirano, T. IL-6 in Inflammation, Autoimmunity and Cancer. Int. Immunol. 2021, 33, 127–148. [Google Scholar] [CrossRef] [PubMed]
- Ginhoux, F.; Jung, S. Monocytes and Macrophages: Developmental Pathways and Tissue Homeostasis. Nat. Rev. Immunol. 2014, 14, 392–404. [Google Scholar] [CrossRef]
- Hansen, T.R.; Smirnova, N.P.; Webb, B.T.; Bielefeldt-Ohmann, H.; Sacco, R.E.; Van Campen, H. Innate and Adaptive Immune Responses to in Utero Infection with Bovine Viral Diarrhea Virus. Anim. Health Res. Rev. 2015, 16, 15–26. [Google Scholar] [CrossRef]
- Riera Romo, M.; Pérez-Martínez, D.; Castillo Ferrer, C. Innate Immunity in Vertebrates: An Overview. Immunology 2016, 148, 125–139. [Google Scholar] [CrossRef] [PubMed]
- Bordon, Y. Innate Memory Training. Nat. Rev. Immunol. 2014, 14, 713. [Google Scholar] [CrossRef]
- Bronzo, V.; Lopreiato, V.; Riva, F.; Amadori, M.; Curone, G.; Addis, M.F.; Cremonesi, P.; Moroni, P.; Trevisi, E.; Castiglioni, B. The Role of Innate Immune Response and Microbiome in Resilience of Dairy Cattle to Disease: The Mastitis Model. Animals 2020, 10, 1397. [Google Scholar] [CrossRef]
- Radzikowska, U.; Rinaldi, A.O.; Çelebi Sözener, Z.; Karaguzel, D.; Wojcik, M.; Cypryk, K.; Akdis, M.; Akdis, C.A.; Sokolowska, M. The Influence of Dietary Fatty Acids on Immune Responses. Nutrients 2019, 11, 2990. [Google Scholar] [CrossRef]
- Wysoczański, T.; Sokoła-Wysoczańska, E.; Pękala, J.; Lochyński, S.; Czyż, K.; Bodkowski, R.; Herbinger, G.; Patkowska-Sokoła, B.; Librowski, T. Omega-3 Fatty Acids and Their Role in Central Nervous System—A Review. Curr. Med. Chem. 2016, 23, 816–831. [Google Scholar] [CrossRef]
- Gutiérrez, S.; Svahn, S.L.; Johansson, M.E. Effects of Omega-3 Fatty Acids on Immune Cells. Int. J. Mol. Sci. 2019, 20, 5028. [Google Scholar] [CrossRef]
- Ryman, V.E.; Packiriswamy, N.; Norby, B.; Schmidt, S.E.; Lock, A.L.; Sordillo, L.M. Supplementation of Linoleic Acid (C18:2n-6) or α-Linolenic Acid (C18:3n-3) Changes Microbial Agonist-Induced Oxylipid Biosynthesis. J. Dairy Sci. 2017, 100, 1870–1887. [Google Scholar] [CrossRef] [PubMed]
- Olmo, I.; Teuber, S.; Larrazabal, C.; Alarcon, P.; Raipane, F.; Burgos, R.A.; Hidalgo, M.A. Docosahexaenoic Acid and TUG-891 Activate Free Fatty Acid-4 Receptor in Bovine Neutrophils. Vet. Immunol. Immunopathol. 2019, 209, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Ayrle, H.; Mevissen, M.; Kaske, M.; Nathues, H.; Gruetzner, N.; Melzig, M.; Walkenhorst, M. Medicinal Plants—Prophylactic and Therapeutic Options for Gastrointestinal and Respiratory Diseases in Calves and Piglets? A Systematic Review. BMC Vet. Res. 2016, 12, 89. [Google Scholar] [CrossRef] [PubMed]
- Osorio, J.S. Gut Health, Stress, and Immunity in Neonatal Dairy Calves: The Host Side of Host-Pathogen Interactions. J. Anim. Sci. Biotechnol. 2020, 11, 105. [Google Scholar] [CrossRef]
- Carter, H.S.M.; Renaud, D.L.; Steele, M.A.; Fischer-Tlustos, A.J.; Costa, J.H.C. A Narrative Review on the Unexplored Potential of Colostrum as a Preventative Treatment and Therapy for Diarrhea in Neonatal Dairy Calves. Animals 2021, 11, 2221. [Google Scholar] [CrossRef]
- Ziętek, J. (Ed.) Choroby Zakaźne Cieląt; Biblioteka Weterynaria w Terenie; Wydawnictwo Elamed: Katowice, Poland, 2013. [Google Scholar]
- Boccardo, A.; Belloli, A.; Biffani, S.; Locatelli, V.; Dall’Ara, P.; Filipe, J.; Restelli, I.; Proverbio, D.; Pravettoni, D. Intravenous Immunoglobulin Transfusion in Colostrum-Deprived Dairy Calves. Vet. J. 2016, 209, 93–97. [Google Scholar] [CrossRef]
- Wilm, J.; Costa, J.H.C.; Neave, H.W.; Weary, D.M.; Von Keyserlingk, M.A.G. Technical Note: Serum Total Protein and Immunoglobulin G Concentrations in Neonatal Dairy Calves over the First 10 Days of Age. J. Dairy Sci. 2018, 101, 6430–6436. [Google Scholar] [CrossRef]
- Grodkowska, K.; Gołębiewski, M.; Slósarz, J.; Sakowski, T.; Puppel, K. The Effect of Supplementation Using a Mixture of Fish Oil and Linseed on the Level of Immunomodulatory Components in Bovine Colostrum. Molecules 2023, 28, 2154. [Google Scholar] [CrossRef]
- Richard, C.; Lewis, E.D.; Goruk, S.; Field, C.J. The Content of Docosahexaenoic Acid in the Maternal Diet Differentially Affects the Immune Response in Lactating Dams and Suckled Offspring. Eur. J. Nutr. 2016, 55, 2255–2264. [Google Scholar] [CrossRef]
- Miciński, J.; Miciński, J.; Zwierzchowski, G.; Fijałkowska, M.; Pogorzelska, J.; Kobzhassarov, T.; Bermagambetova, N.; Siwicki, A.K.; Wójcik, R.; Shaikamal, G.I. Influence of A Diet Containing β-Carotene and Omega-3 Fatty Acids on the Biochemical and Nonspecific Humoral Immunity Indicators and on the Results of Experimental Calf Rearing. J. Elem. 2015, 21, 283–302. [Google Scholar] [CrossRef]
- Welboren, A.C.; Van Baal, J.; Ghaffari, M.H.; Sauerwein, H.; Renaud, J.B.; Martín-Tereso, J.; Steele, M.A.; Leal, L.N. Gastrointestinal Permeability and Inflammatory Status of Preweaning Dairy Calves in Response to Decreasing the Ratio of N-6 to n-3 Fatty Acid in Milk Replacer. J. Dairy Sci. 2023, 106, 3662–3679. [Google Scholar] [CrossRef] [PubMed]
- Hill, T.M.; VandeHaar, M.J.; Sordillo, L.M.; Catherman, D.R.; Bateman, H.G.; Schlotterbeck, R.L. Fatty Acid Intake Alters Growth and Immunity in Milk-Fed Calves. J. Dairy Sci. 2011, 94, 3936–3948. [Google Scholar] [CrossRef] [PubMed]
- Karcher, E.L.; Hill, T.M.; Bateman, H.G.; Schlotterbeck, R.L.; Vito, N.; Sordillo, L.M.; VandeHaar, M.J. Comparison of Supplementation of N-3 Fatty Acids from Fish and Flax Oil on Cytokine Gene Expression and Growth of Milk-Fed Holstein Calves. J. Dairy Sci. 2014, 97, 2329–2337. [Google Scholar] [CrossRef] [PubMed]
- Wilms, J.N.; Hare, K.S.; Fischer-Tlustos, A.J.; Vahmani, P.; Dugan, M.E.R.; Leal, L.N.; Steele, M.A. Fatty Acid Profile Characterization in Colostrum, Transition Milk, and Mature Milk of Primi- and Multiparous Cows during the First Week of Lactation. J. Dairy Sci. 2022, 105, 2612–2630. [Google Scholar] [CrossRef]
- Mellors, S.C.; Wilms, J.N.; Welboren, A.C.; Ghaffari, M.H.; Leal, L.N.; Martín-Tereso, J.; Sauerwein, H.; Steele, M.A. Gastrointestinal Structure and Function of Preweaning Dairy Calves Fed a Whole Milk Powder or a Milk Replacer High in Fat. J. Dairy Sci. 2023, 106, 2408–2427. [Google Scholar] [CrossRef] [PubMed]
- Kadkhoday, A.; Riasi, A.; Alikhani, M.; Dehghan-Banadaky, M.; Kowsar, R. Effects of Fat Sources and Dietary C 18:2 to C 18:3 Fatty Acids Ratio on Growth Performance, Ruminal Fermentation and Some Blood Components of Holstein Calves. Livest. Sci. 2017, 204, 71–77. [Google Scholar] [CrossRef]
- Melendez, P.; Roeschmann, C.F.; Baudo, A.; Tao, S.; Pinedo, P.; Kalantari, A.; Coarsey, M.; Bernard, J.K.; Naikare, H. Effect of Fish Oil and Canola Oil Supplementation on Immunological Parameters, Feed Intake, and Growth of Holstein Calves. J. Dairy Sci. 2022, 105, 2509–2520. [Google Scholar] [CrossRef]
- Flaga, J.; Korytkowski, Ł.; Górka, P.; Kowalski, Z.M. The Effect of Docosahexaenoic Acid-Rich Algae Supplementation in Milk Replacer on Performance and Selected Immune System Functions in Calves. J. Dairy Sci. 2019, 102, 8862–8873. [Google Scholar] [CrossRef]
- Abu El-Hamd, M.; El-Diahy, Y.; El-Maghraby, M.; Elshora, M. Effect of flaxseed oil on digestibility, blood parameters, immuno-response and productive performance of suckling friesian calves. J. Anim. Poult. Prod. 2015, 6, 755–765. [Google Scholar] [CrossRef]
- Parikh, M.; Netticadan, T.; Pierce, G.N. Flaxseed: Its Bioactive Components and Their Cardiovascular Benefits. Am. J. Physiol.-Heart Circ. Physiol. 2018, 314, H146–H159. [Google Scholar] [CrossRef]
- Kouamé, K.J.E.-P.; Bora, A.F.M.; Li, X.; Sun, Y.; Liu, L. Novel Trends and Opportunities for Microencapsulation of Flaxseed Oil in Foods: A Review. J. Funct. Foods 2021, 87, 104812. [Google Scholar] [CrossRef]
- Śpitalniak-Bajerska, K.; Szumny, A.; Pogoda-Sewerniak, K.; Kupczyński, R. Effects of N-3 Fatty Acids on Growth, Antioxidant Status, and Immunity of Preweaned Dairy Calves. J. Dairy Sci. 2020, 103, 2864–2876. [Google Scholar] [CrossRef] [PubMed]
- Śpitalniak-Bajerska, K.; Szumny, A.; Kucharska, A.Z.; Kupczyński, R. Effect of Natural Antioxidants on the Stability of Linseed Oil and Fish Stored under Anaerobic Conditions. J. Chem. 2018, 2018, 9375085. [Google Scholar] [CrossRef]
- Kuhn, M.J.; Mavangira, V.; Gandy, J.C.; Zhang, C.; Jones, A.D.; Sordillo, L.M. Differences in the Oxylipid Profiles of Bovine Milk and Plasma at Different Stages of Lactation. J. Agric. Food Chem. 2017, 65, 4980–4988. [Google Scholar] [CrossRef] [PubMed]
- Chandra, M.; Panchatcharam, M.; Miriyala, S. Biomarkers in ROS and Role of Isoprostanes in Oxidative Stress. In Free Radicals and Diseases; Ahmad, R., Ed.; InTech: London, UK, 2016. [Google Scholar] [CrossRef]
| Experimental Animals | Experiment Design | Share of Fatty Acids in the Ration/Fat Supplement | Effects of Supplementation | Source |
|---|---|---|---|---|
| 48 Holstein calves aged 2–5 days | Calves were randomly assigned to 1 of 3 diets (16 calves/diet): milk replacer with 17% pork fat, milk replacer with 15% pork fat and 2% DM flaxseed oil (flax), or milk replacer with 15% DM pork fat and 2% DM fish oil (fish) Experiment duration: 56 days | Control: n-3—1.7% n-6—22.4% n-6/n-3—12.8% Flax: n-3—12.5% n-6—21.3% n-6/n-3—1.7% Fish: n-3—6.1% n-6—21.4% n-6/n-3—3.5% | FLAX group: IL-1β ↓ IL-8 ↓ Osteopontin ↓ FISH group: TNF-α ↓ | [196] |
| 54 calves of the Holstein Friesian breed at 6 days old | Animals were divided into 3 groups (n =18 calves each): control, the group receiving milk replacer + beta-carotene (25 mg/calf/day), and the group receiving milk replacer + addition of liver oil suspension, as a source of n-3 (5 g/calf/day) Experiment duration: 54 days | Liver oil: SFA—15.2% MUFA—47.5% PUFA—31.4% n-3—27.2% | IgG ↑ ALT ↑ AST ↓ ALP ↑ LDH-L ↑ UREA ↓ CREA ↓ Glucose ↓ Cholesterol ↑ Occurrence of diarrhea ↓ Diseases of the upper tract ↓ | [193] |
| 15 Holstein Friesian calves at 7 days of age | Animals were divided into 3 groups (n=18 calves each): control, the group receiving milk replacer + beta-carotene (25 mg/calf/day), and the group receiving milk replacer + addition of liver oil suspension, as a source of n-3 (5 g/calf/day) Experiment duration: 54 days | Canola oil: * SFA—45.79% MUFA—24.06% PUFA—28.77% n-3—13.36% Fish oil: * SFA—33.37% MUFA—40.73% PUFA—24.96% n-3—11.23% | Fish oil (source of n-3) group: Hepatoglobins ↓ IL-1β ↓ TNF-α ↓ | [200] |
| 30 Holstein calves aged 1–4 days | Animals were randomly assigned to be fed a milk replacer with an n-6:n-3 FA ratio of 40:1 or 6.5:1 The PUFA ratio in the milk replacer was adjusted by including 1% flaxseed oil and 1% algae oil (as a share in fat content in the diet) Experiment duration: 25 days | Control: SFA—63.7% UFA—36.3% n-3—0.16% n-6—6.49% n-6:n-3—40.6% Experimental diet: SFA— 62.6% UFA—37.4% n-3—1.04% n-6 —6.76 n-6:n-3—6.5% | Acute phase proteins ↑ Haptoglobin ↑ Amyloid A ↑ | [194] |
| 40 Holstein Friesian calves at 8.6 days of age | Animals were assigned to 4 experimental groups: control, milk replacer + addition of 9 g of DHA-rich algae; milk replacer + addition of 9, 18, 27 g of DHA-rich algae Experiment duration: 49 days | Algae: * SFA—63.57% MUFA—1.24% PUFA—34.47% n-3—33.69% | IL-1β ↓ TNFα ↓ IgG ↓ | [201] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fabjanowska, J.; Kowalczuk-Vasilev, E.; Klebaniuk, R.; Milewski, S.; Gümüş, H. N-3 Polyunsaturated Fatty Acids as a Nutritional Support of the Reproductive and Immune System of Cattle—A Review. Animals 2023, 13, 3589. https://doi.org/10.3390/ani13223589
Fabjanowska J, Kowalczuk-Vasilev E, Klebaniuk R, Milewski S, Gümüş H. N-3 Polyunsaturated Fatty Acids as a Nutritional Support of the Reproductive and Immune System of Cattle—A Review. Animals. 2023; 13(22):3589. https://doi.org/10.3390/ani13223589
Chicago/Turabian StyleFabjanowska, Julia, Edyta Kowalczuk-Vasilev, Renata Klebaniuk, Szymon Milewski, and Hıdır Gümüş. 2023. "N-3 Polyunsaturated Fatty Acids as a Nutritional Support of the Reproductive and Immune System of Cattle—A Review" Animals 13, no. 22: 3589. https://doi.org/10.3390/ani13223589
APA StyleFabjanowska, J., Kowalczuk-Vasilev, E., Klebaniuk, R., Milewski, S., & Gümüş, H. (2023). N-3 Polyunsaturated Fatty Acids as a Nutritional Support of the Reproductive and Immune System of Cattle—A Review. Animals, 13(22), 3589. https://doi.org/10.3390/ani13223589





