1. Introduction
Rabies is transmissible to all mammals and is caused by the neurotropic rabies virus (RABV) of the genus
Lyssavirus. This zoonotic disease has a fatality rate of almost 100.00% in humans and animals and nowadays remains a global threat, resulting in around 59,000 human deaths per year [
1]. Annually, the occurrence of rabies results in a global estimated cost of USD 8.6 billion [
2]. Canines are considered by the World Health Organization as the most important source of infection for the zoonotic transmission of rabies worldwide, but the transmission through the bites of vampire bats or contact with saliva from infected herbivores on open cuts or wounds also occurs in countries such as those placed in South America [
2].
In the last decade in Brazil, rabies has been transmitted to humans via bites by hematophagous bats (
Desmodus (D.) rotundus) (52.60%), dogs (23.70%), non-human primates (10.50%), and felines (10.50%) [
3], and there has been a growing number of reports regarding
D. rotundus bites on humans, posing a potential threat to public health within this country [
4].
D. rotundus are also the main transmitters of rabies to domestic herbivores in Brazil (100.00%,
n = 50 [
5] and 99.20%,
n = 650 [
6]), causing a high caseload of vampire-bat variant rabies in areas of Brazil where geographic conditions support a high number of bat vampire shelters [
7,
8]. Rabies occurrence in livestock animals is frequently reported, with cattle being the most affected among the livestock species [
9]. This disease is considered an important cause of economic loss for cattle farmers [
10]. Additionally, the importance of this disease in equines has recently been on the rise in Brazil [
11].
For the control of rabies in cattle and equines in Brazil, the following control strategies are recommended by the federal government: the monitoring and control of hematophagous bats through the application of anticoagulant poison, mandatory vaccination of bovines and equines in focus and peripheral high-risk areas, voluntary vaccination of these animals in low-risk areas, mandatory notification of suspected cases of rabies or vampire-bat shelters detection, investigation of suspected cases, conducting laboratory tests for disease diagnosis, and implementing animal and human health education programs [
12]. Anticoagulant poisons (Warfarin paste) are fatal when ingested by bats. Population control using anticoagulant poison occurs via two methods: they are either applied topically to bats to be transmitted through allogrooming, or they are administered to livestock for subsequent ingestion by bats during blood feeding. After applying the poison to the animals individually, it is expected that the population control within the shelters will be achieved. However, the efficacy of culling bats for rabies management continues to be a contentious topic of debate in the scientific community due to risks to wildlife [
13].
Oliveira and colleagues [
11] emphasized the need for attention to be directed specifically to the state of Rondônia, where this study was performed, due to the potential risk of rabies spread to transboundary areas. The monitoring of bat lyssaviruses in this area has also been recommended recently [
14] and there is a need of adopting more effective prophylactic measures to control rabies in this area of the Brazilian’s Legal Amazon [
9]. The animal health agency responsible for rabies control in domestic herbivores in the state of Rondônia, Brazil, is known as the “
Agência de Defesa Sanitária Agrosilvopastoril do Estado de Rondônia” (IDARON), which translates to the Agency of Agrosilvopastoral Sanitary Defense of Rondônia State. IDARON operates in compliance with Ordinance No. 168, issued on 27 September 2005, by the Brazilian Ministry of Agriculture, Livestock, and Supply Chain (MAPA). This ordinance approves the Technical Manual for Herbivore Rabies Control, which guides IDARON’s actions [
12].
Considering these factors, this study aimed to describe the epidemiology of rabies in bovines and equines in the state of Rondônia, Brazil, from 2002 to 2021, and correlate these findings with the prophylactic strategies implemented by the health agency for rabies control (IDARON) in livestock animals.
4. Discussion
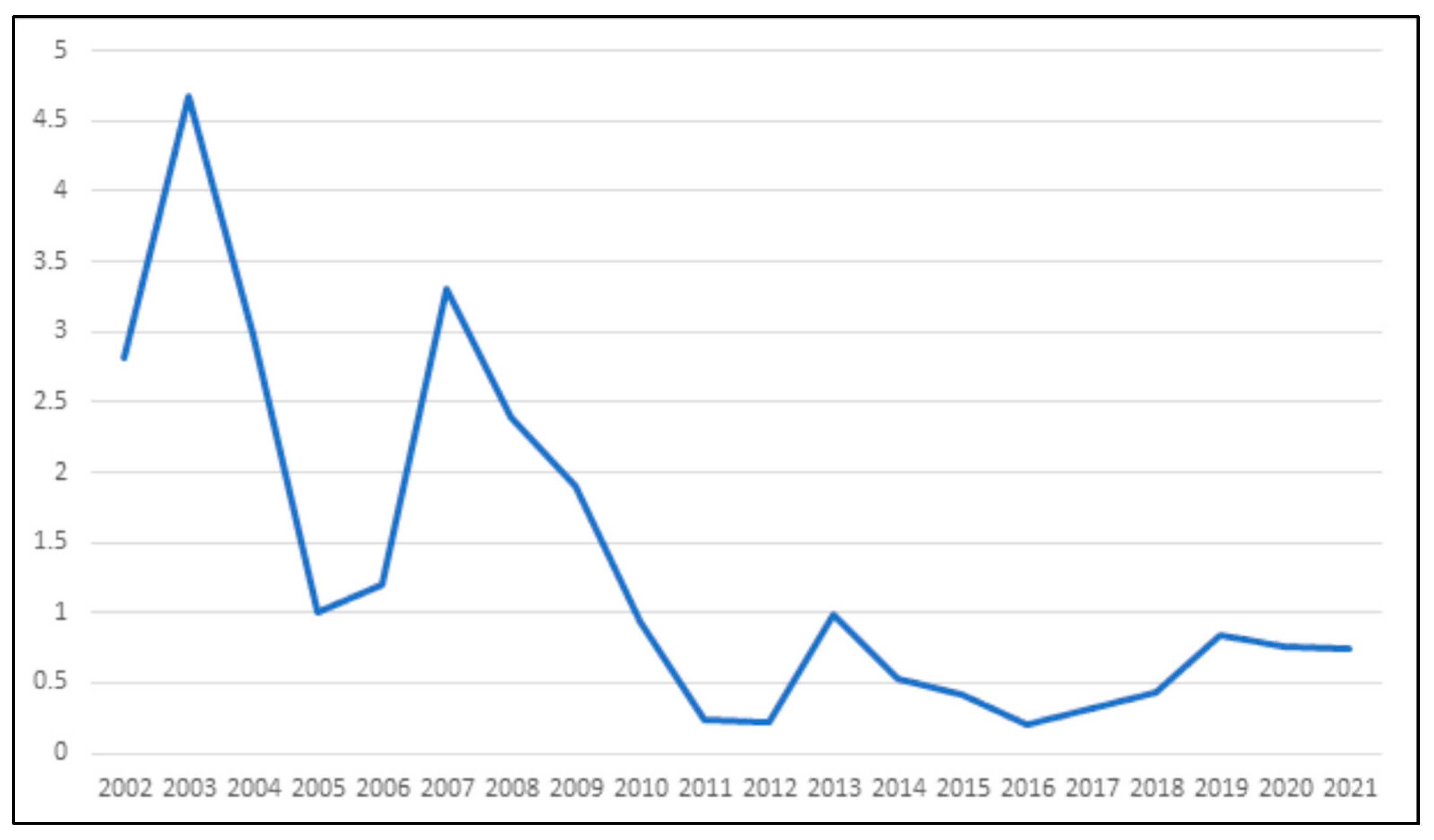
Controlling livestock in rabies was associated with a substantial decrease in rabies cases between 2002 and 2021. This effort resulted in a sustained four-fold reduction in rabies incidence among herbivores, measured per million animals, by the year 2011.
The available evidence strongly indicates a global trend of under-reporting in rabies cases [
2]. It is therefore plausible that under-reporting might also be present in the dataset used for this study. However, it is important to note that efforts have been made to mitigate this issue. The compulsory reporting of animals suspected to be rabies-infected to the local health agency (IDARON) has been in place [
12], contributing to minimizing the potential for under-reporting. It is noteworthy that all instances of rabies cases in cattle and equine from the comprehensive IDARON database were meticulously incorporated into our study. Considering these measures, we maintain a high level of confidence in the representation of the actual number of cases within our dataset. However, we cannot ensure that they are not underestimated due to the potential occurrence of suspected cases not reported by farmers or veterinaries to IDARON.
Rabies affecting domestic herbivores is frequently reported in various regions of Brazil, including, at least, the states of Paraná [
21], Mato Grosso [
22,
23], Rio Grande do Sul [
24], and states located in the North Region of Brazil [
8,
25,
26], where the state of Rondônia is located. The considerable majority (>99%) of rabies cases in herbivores attributed to the
D. rotundus-variant rabies virus in other parts of Brazil [
5,
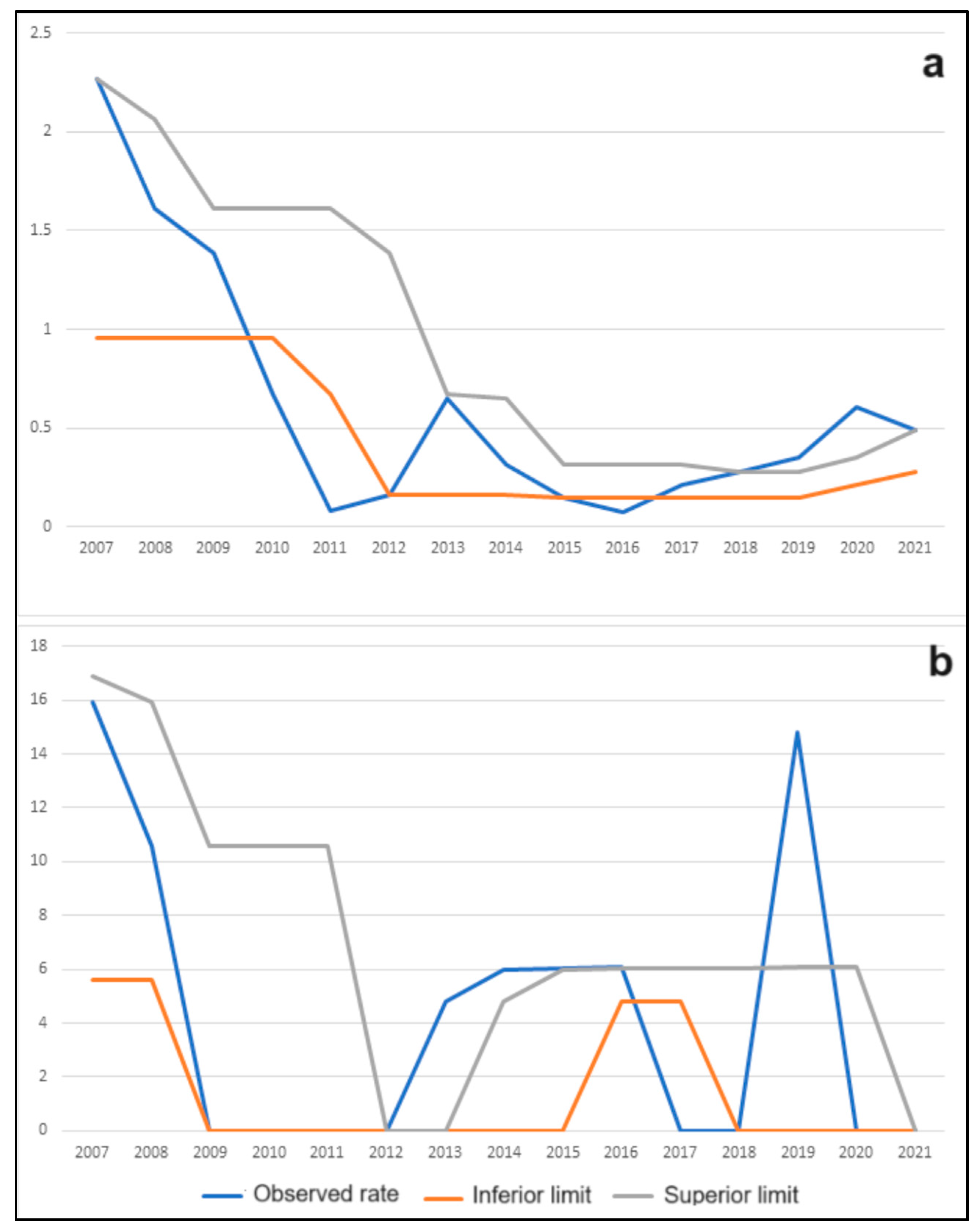
6] provides a solid rationale for investigating the potential connection between vampire-specific control strategies and the prevalence of rabies in cattle and equines examined in this study. The implemented control measures, which encompass efforts to manage
D. rotundus populations, have the potential to substantially diminish the risk of rabies incidence in the local cattle and equine populations within this region (
Table 1 and
Table 7,
Figure 2). In Brazil, human rabies also is predominantly transmitted via
D. rotundus bites, as the transmission through dogs has been significantly reduced in this country [
2]. Therefore, perhaps the control of rabies in hematophagous bats, focusing on the transmission of rabies to domestic herbivores, may also contribute to the prevention of rabies in humans.
In Rondônia, the incidence of rabies in equids was considered high (
Table 1). Oliveira and colleagues [
11] found a high incidence of equine rabies in Brazil (ranging from 2.8 to 137 per 100,000 horses), including an incidence in Rondonia (640 cases per 1 million equines) prior to our study period (2010–2019), which is considerably higher than that found in our study (127 per million). This difference may have occurred probably because we included a larger historical dataset. Furthermore, in our study, the incidence of rabies was significantly higher in equines. The impact of a relatively low number of cases in horses is more relevant within the horse population compared to the effect of bovine rabies on the cattle population. This is due to the considerably smaller size of the horse population in comparison to the cattle population, coupled with the fact that the number of cases in horses is also lower than that seen in cattle. It is worth highlighting that despite the difference in rabies incidences between cattle and horses in this study, prophylactic measures for this disease should be directed towards both species. This is because both species hold equal significance in the disease cycle and are managed through the same control measures.
Consistent with multiple other studies in Brazil [
26,
27,
28,
29,
30,
31,
32], our analysis found no statistically significant within-year pattern of domestic herbivore rabies. The climatic characteristics of the North region, which lacks a rigorous winter and experiences high temperatures throughout the year, may contribute to the absence of distinct within-year patterns in bat populations and consequently minimize the patterns of rabies occurrence [
5]. Our results demonstrate that control measures for rabies, including the annual vaccination of cattle and horses, should provide an effective reduction in the risk of rabies infection throughout the year.
The GEE model identified three control activities (negative associations: treating bats using anticoagulant poison and using vaccines in cattle and equines; positive association: printing educative materials) that were significantly associated (
p < 0.05) with the rabies incidence during the study period. Our results are consistent with those of one study conducted in Brazil, specifically in the state of São Paulo, using official data from the local health agency for rabies control in domestic herbivores [
31]. This study considered that controlling
D. rotundus and vaccinating herbivores are the strategies responsible for the reduction in the cases of rabies in production animals. Another study conducted in the state of Minas Gerais, Brazil, highlighted the decrease in official bovine rabies tests between 1998 and 2006, as well as a decline in clinical cases of rabies. These authors attributed these improvements to enhanced control measures, including rabies vaccinations and the treatment of vampire bats using anticoagulant poison [
32]. Our results corroborate what was reported by these authors, as we observed that the vaccination and application of anticoagulant poison were effective actions in reducing the occurrence of rabies in domestic herbivores.
A significantly more pronounced effect when employing anticoagulant poison for treating bats, in comparison to the impact of vaccinating cattle and equines, was observed. Employing anticoagulant poison to treat D. rotundus proves to be a potent control method. This approach targets multiple bats, thereby diminishing the potential for rabies transmission through the reduced interaction between all mammal populations, including herbivores and humans, and these bats. Consequently, the advantages of bat treatment likely extend beyond the realm of reducing rabies solely in herbivores and is likely to contribute to a decline in non-urban human rabies cases. In contrast, the vaccination’s impact is confined to disease prevention in the vaccinated individual, offering no direct benefit to human health.
Out of the six variables related to managing vampire bat populations, only one—treating bats with anticoagulant poison—showed a significant association with reducing the rabies incidence risk. Concerning shelter monitoring, capture events, and the number of captured bats, none of these factors displayed additional significance in the model. This was primarily because the datasets were intertwined and exhibited strong correlations. Treated bats constitute a subset of captured bats (rho = 0.99). Moreover, the numbers of captured bats (rho = 0.73) and capture events (rho = 0.79) correlate with the quantity of monitored shelters. Additionally, the rates of captured bats represent a subset of the capture events (rho = 0.91). While the figures related to shelter monitoring, capture events, and captured bats demonstrated high correlations (p < 0.1) in the Spearman correlation analysis, indicating a reduction in rabies cases, these associations were largely attributed to the impact of bat treatment alone. The capacity of the Generalized Estimating Equation (GEE) analysis to consider autocorrelation in the data allowed it to discern which specific bat shelter-based activity impacted rabies incidence. Despite not individually yielding significant reductions in rabies, the other shelter-based activities should remain integral to the control program, as they are essential for the effective treatment of bats.
Interestingly, while the application of anticoagulant poison to bats yielded significant results, the direct sale of these pastes to farmers did not exhibit a connection to reducing the incidence of rabies in cattle and equines. However, the values obtained for the number of farms that acquired anticoagulant poison were mixed with the acquisition of vaccines, which may have influenced our results (a bias). Despite both methods sharing the common approach of targeting bat populations in shelters through allogrooming behavior, the key distinction lies in the fact that one is directly applied to bats, whereas the other is administered to animals for ingestion by bats. Notably, the act of directly applying the poison to bats is executed by IDARON itself. Conversely, when it comes to the vampire bat pastes intended for application to animals, these are retailed in stores and then utilized by farmers to apply in domestic herbivores. Due to the unique nature of these actions, no correlation exists between them, elucidating why one strategy proved efficacious while the other did not. Possible reasons for this difference may include the sold poison not being used by farms, poison not being applied correctly, bats not coming into contact with poison, or the poison only affecting the contact bats (or fewer bats than when applied to captured bats).
The occurrence of rabies outbreaks in cattle is influenced by the remarkable adaptability of the bat species
D. rotundus to artificial shelters and their proximity to the primary food source, which happens to be cattle in farms [
33]. When cattle are granted free-range grazing access within expansive management systems encompassing forested areas, riparian zones, caves, secondary vegetation, as well as fragments and edges of vegetation, the ensuing conflict between vampire bats and cattle escalates in both magnitude and severity [
34]. This scenario is widely presented in the state of Rondônia. Therefore, the control of hematophagous bats carried out by IDARON through anticoagulant poison contributed to the prevention of cases in livestock animals and possibly in humans, given that most human cases in Brazil are caused by attacks from hematophagous bats [
3].
Furthermore, despite our data demonstrating the importance of controlling the populations of hematophagous bats for the transmission of rabies to domestic herbivores, the real effectiveness of these actions is currently questioned among the scientific community in a more general sense, as they may result in ecological impacts and risks for wildlife animals [
13,
34,
35,
36,
37]. Bats are important in the Amazon ecosystem and the excessive population control may result in local or regional extinctions and unintended ecological consequences, such as killing other carnivores that ingest dead bats or the contamination of non-hematophagous bats with anticoagulant poison in multispecies colonies [
37]. A promising alternative control involves vaccinating vampire bat populations using an effective transmissible rabies vaccine carried by a betaherpesvirus vector [
38]. This strategy offers benefits compared to lethal treatments for bats; however, it is not currently accessible. The vaccination of bats would effectively address the ethical debates surrounding bat extermination and the potential extinction risk of a protected species within complex ecosystems, such as the Amazon Rainforest.
Our results also demonstrated the importance of vaccination for the control of rabies in production animals, which was associated with its incidence reduction. Mello and colleagues [
9] reported 52 outbreaks of bovine rabies in 23 out of the 79 municipalities comprising the state of Mato Grosso do Sul, Brazil, spanning from 2010 to 2016. These authors concluded that the vaccination of cattle, regardless of herd size, was economically feasible and was recommended to minimize losses due to bovine rabies. Upon comprehensive analysis, the economic losses resulting from the documented outbreaks remained below the threshold of USD 5000.00 per farm. In a few outbreaks, losses ranged from USD 15,000.00 to USD 25,000.00. Among the 52 surveyed farms, the collective expense of vaccination would amount to less than USD 200.00 for 45 of them. When comparing vaccination costs to the losses incurred due to rabies-related fatalities, regardless of herd size, the average cost of vaccinations constituted less than 10% of the estimated economic loss. According to this research, this underlines the efficacy and economic viability of vaccinations as a viable approach for rabies control [
9].
From 2017 to 2021, the number of vaccine doses sold in Rondonia (
Table 4) relative to the population of cattle and horses (
Table 1) was sufficient to immunize approximately 23–34% of the herd. Both the Spearman correlation and GEE analysis indicate that this risk-based vaccination program has been successful in significantly reducing herbivore rabies in Rondonia, although it has not completely eradicated it. Implementing mandatory vaccination for the entire cattle population in Rondonia (which stood at 16 million in 2021) to address the remaining 5–10 annual rabies cases would pose a substantial financial burden. However, as suggested by the analysis in [
9], expanding either mandatory or recommended vaccination to all properties in Rondonia with a history of herbivore rabies is likely to be a cost-effective approach to further diminish rabies occurrences without introducing ecological risks.
Additionally, our data demonstrate that the incidence of rabies in cattle and horses is positively associated with the rate of printed educational materials. Printed material had no significant spearman correlation, yet had an extremely significant GEE estimation. We believe that this discrepancy may be related to the fact that the Spearman correlation test is a non-parametric method, where the specific value of the data is not relevant. Only the position of that value in relation to the positions of the others is considered. This is different from the Pearson correlation and GEE tests (which are significant), where the actual value of the data is considered.
We can hypothesize two distinct scenarios to explain this result: The first would be linked to IDARON’s actions of printing educational materials reactively in response to rabies occurrence in domestic herbivores, with the aim of educating farmers and preventing new cases. The second scenario would involve the effectiveness of distributing the printed materials to farmers leading to the increased reporting of suspected cases and, consequently, case detection and incidence increasing. On the other hand, the implementation of meetings, speeches, courses, and media dissemination (such as television and radio) did not appear to have a relationship with the incidence of rabies. However, we believe that these actions should not be discontinued, as they contribute to raising awareness among the general population (including farmers) about this infectious disease and can potentially contribute to rabies control through the reporting of suspected cases [
39]. Promoting awareness and the reporting of suspected cases and detection by testing supports the vaccination and bat treatment activities which were effective. Within cattle owners in Buthan, there was a noticeable absence of in-depth understanding concerning rabies, including vulnerable hosts, pathways of transmission, the impact of rabies infection on human health, and suitable health-related responses. This research underscores the urgency of bolstering rabies education initiatives in rural areas to bridge these informational voids [
40]. Similarly, our findings emphasize the ongoing importance of promoting health education in the state of Rondônia, Brazil.
Lastly, we emphasize that because we used data obtained from the IDARON’s database, which was supplied by different managers and professionals over approximately 20 years, there may be some biases present in this database, such as errors in data annotation and an inability to distinguish data = 0 (absence) from data that are unknown, missing, or not recorded (zero values in
Table 3,
Table 4 and
Table 5). When it comes to actions taken by official agencies, such as IDARON, the absence of information implies a value of zero because if any action had taken place, it would have been recorded. In the case of disease cases, we always consider the recorded number because there is no way to determine the real occurrence counts due to the possibility of under-reporting. However, in some other situations, it is possible that certain data may have only started being recorded after a certain period, and they might not have been zero. Assessing this impact on our results is challenging. Nevertheless, given the large span of years of the data included in this study, we believe this impact is likely to be minimal, though it is worth mentioning.