Antimicrobial Activity of Peptides Produced by Lactococcus lactis subsp. lactis on Swine Pathogens
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strains
2.2. Bacterial Growth Conditions
2.3. Antimicrobial Activity Assay
2.3.1. Agar Diffusion
2.3.2. Growth Inhibition Test
2.4. Analysis of the BLIS Produced by L. lactis L2
2.5. In Silico Prediction of Bacteriocinogenic Production by L. lactis L2
2.6. Statistical Analysis
3. Results
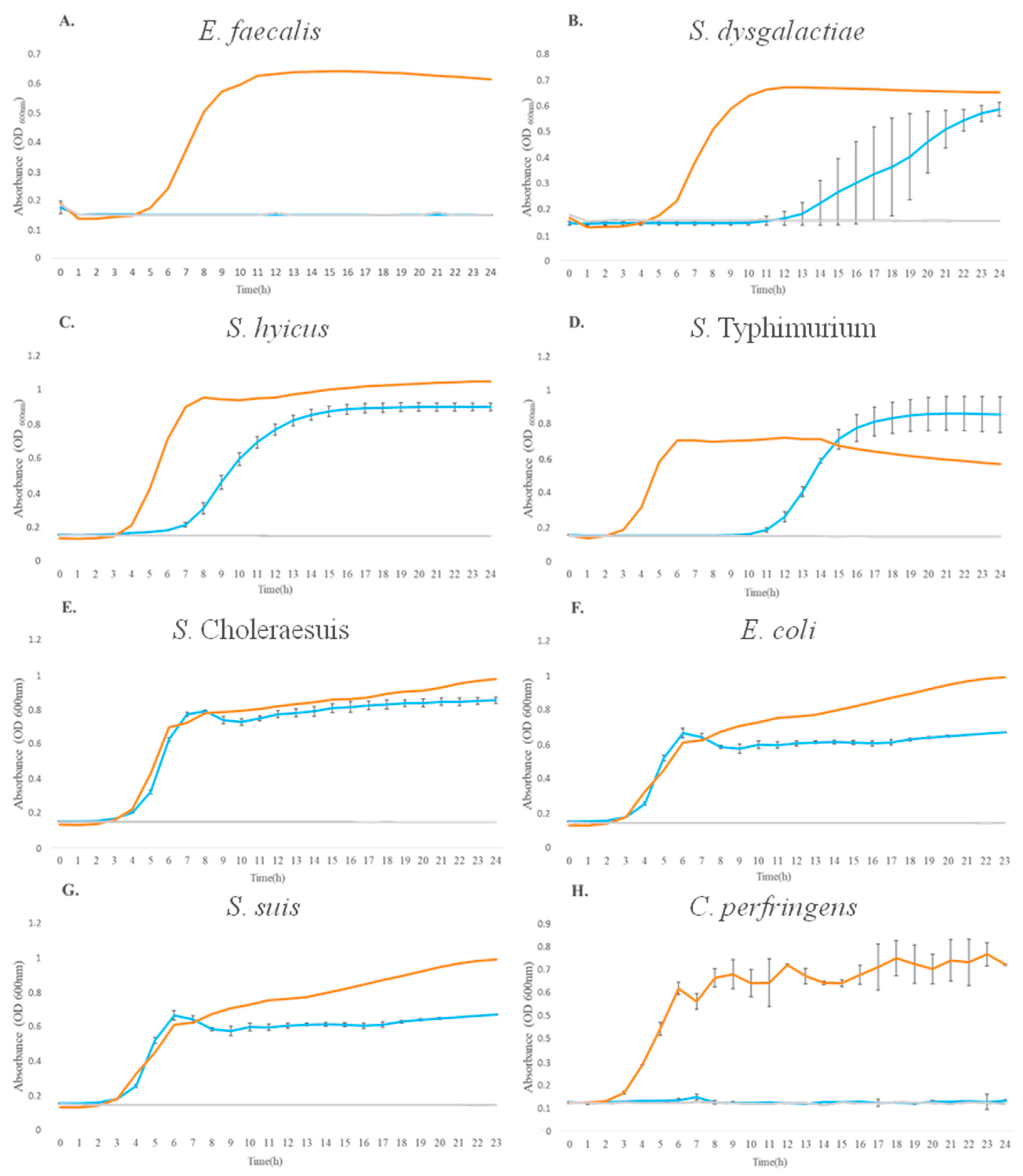
3.1. Assessment of Antibacterial Activity against Pathogenic Strains
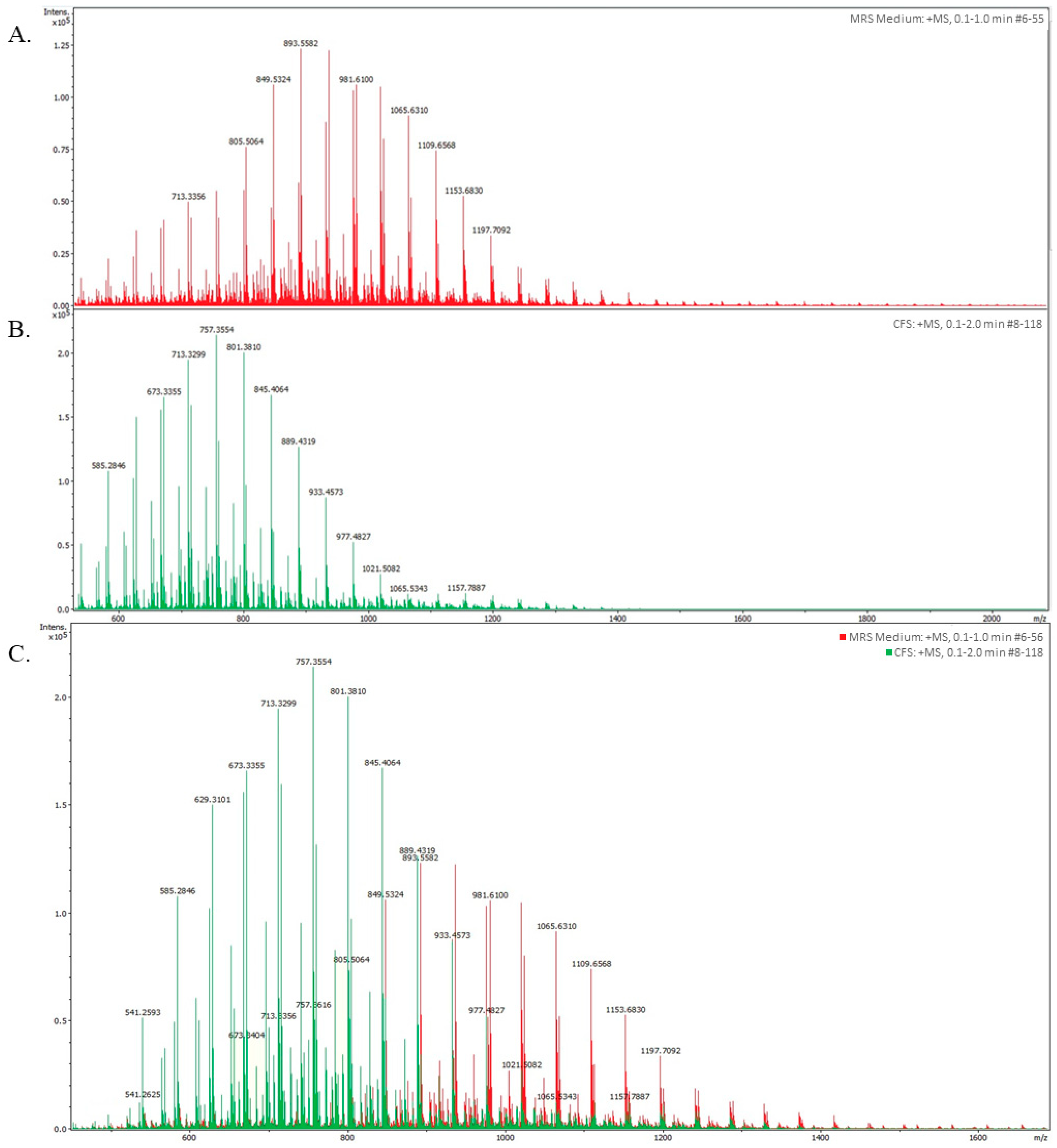
3.2. Analysis of L. lactis L2 BLIS by High-Performance Liquid Chromatography (HPLC)
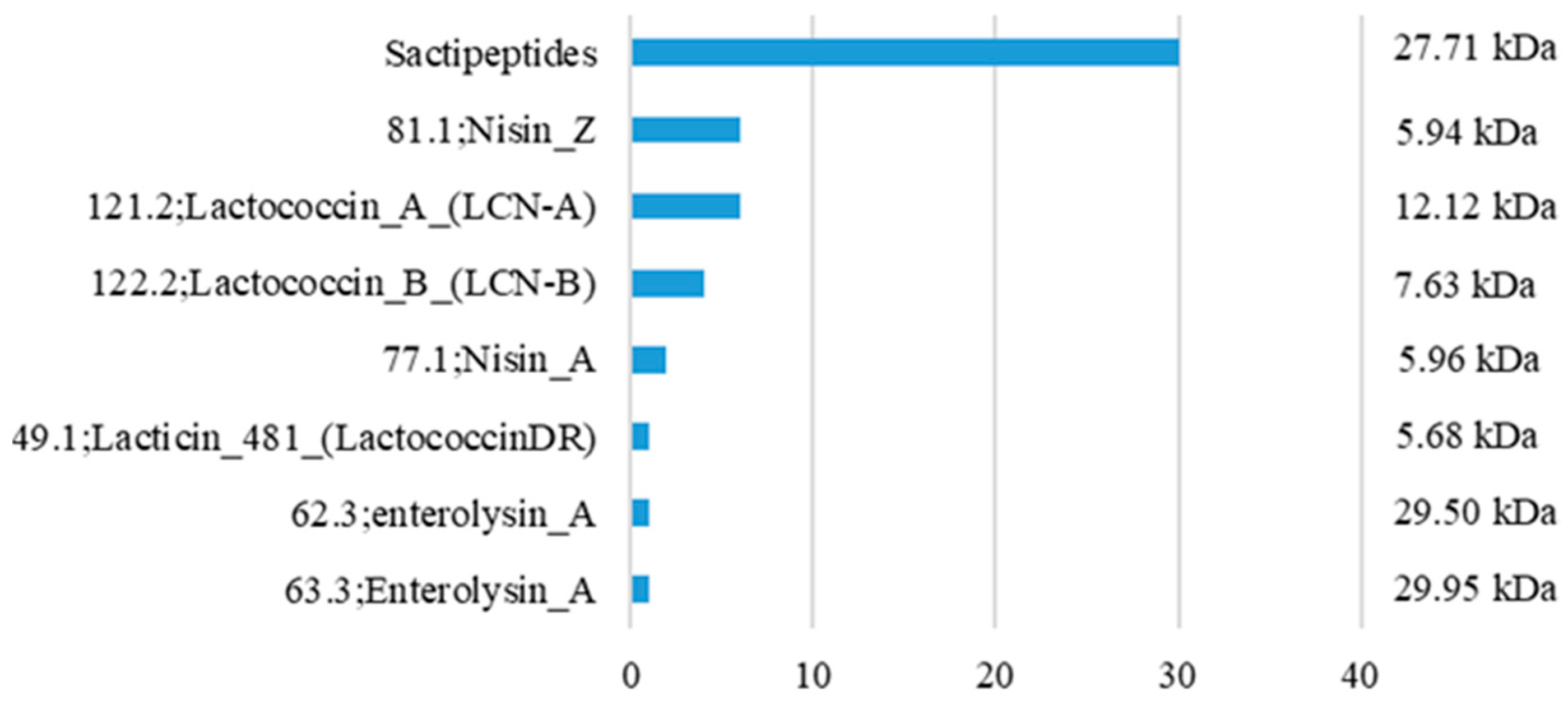
3.3. In Silico Prediction of Bacteriocin Genes
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Calderón Díaz, J.A.; Fitzgerald, R.M.; Shalloo, L.; Rodrigues da Costa, M.; Niemi, J.; Leonard, F.C.; Kyriazakis, I.; García Manzanilla, E. Financial Analysis of Herd Status and Vaccination Practices for Porcine Reproductive and Respiratory Syndrome Virus, Swine Influenza Virus, and Mycoplasma hyopneumoniae in Farrow-to-Finish Pig Farms Using a Bio-Economic Simulation Model. Front. Vet. Sci. 2020, 7, 556674. [Google Scholar] [CrossRef]
- Pietrosemoli, S.; Tang, C. Animal Welfare and Production Challenges Associated with Pasture Pig Systems: A Review. Agriculture 2020, 10, 223. [Google Scholar] [CrossRef]
- Thornton, P.K. Livestock production: Recent trends, future prospects. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2010, 365, 2853–2867. [Google Scholar] [CrossRef]
- Kim, H.B.; Isaacson, R.E. Salmonella in swine: Microbiota interactions. Annu. Rev. Anim. Biosci. 2017, 5, 43–63. [Google Scholar] [CrossRef]
- Álvarez-Pérez, S.; Blanco, J.L.; Astorga, R.J.; Gómez-Laguna, J.; Barrero-Domínguez, B.; Galán-Relaño, A.; Harmanus, C.; Kuijper, E.; García, M.E. Distribution and tracking of Clostridium difficile and Clostridium perfringens in a free-range pig abattoir and processing plant. Food Res. Int. 2018, 113, 456–464. [Google Scholar] [CrossRef]
- Segura, M. Streptococcus suis Research: Progress and Challenges. Pathogens 2020, 9, 707. [Google Scholar] [CrossRef]
- Hurst, C.J. Opportunistic bacteria associated with mammalian livestock disease. In The Connections between Ecology and Infectious Disease; Hurst, C.J., Ed.; Advances in Environmental Microbiology; Springer International Publishing: Cham, Switzerland, 2018; Volume 5, pp. 185–238. ISBN 978-3-319-92371-0. [Google Scholar] [CrossRef]
- Fairbrother, J.M.; Nadeau, E.; Gyles, C.L. Escherichia coli in postweaning diarrhea in pigs: An update on bacterial types, pathogenesis, and prevention strategies. Anim. Health Res. Rev. 2005, 6, 17–39. [Google Scholar] [CrossRef]
- Institute of Medicine (US). Improving Food Safety through a One Health Approach: Workshop Summary; The National Academies Collection: Reports funded by National Institutes of Health; National Academies Press (US): Washington, DC, USA, 2012; ISBN 0309259339. [Google Scholar]
- Larsson, D.G.J.; Flach, C.F. Antibiotic resistance in the environment. Nat. Rev. Microbiol. 2022, 20, 257–269. [Google Scholar] [CrossRef] [PubMed]
- Jo, H.E.; Kwon, M.-S.; Whon, T.W.; Kim, D.W.; Yun, M.; Lee, J.; Shin, M.-Y.; Kim, S.-H.; Choi, H.-J. Alteration of gut microbiota after antibiotic exposure in finishing swine. Front. Microbiol. 2021, 12, 596002. [Google Scholar] [CrossRef]
- Lambo, M.T.; Chang, X.; Liu, D. The recent trend in the use of multistrain probiotics in livestock production: An overview. Animals 2021, 11, 2805. [Google Scholar] [CrossRef]
- Soltani, S.; Hammami, R.; Cotter, P.D.; Rebuffat, S.; Said, L.B.; Gaudreau, H.; Bédard, F.; Biron, E.; Drider, D.; Fliss, I. Bacteriocins as a new generation of antimicrobials: Toxicity aspects and regulations. FEMS Microbiol. Rev. 2021, 45, fuaa039. [Google Scholar] [CrossRef]
- Vieco-Saiz, N.; Belguesmia, Y.; Raspoet, R.; Auclair, E.; Gancel, F.; Kempf, I.; Drider, D. Benefits and Inputs From Lactic Acid Bacteria and Their Bacteriocins as Alternatives to Antibiotic Growth Promoters During Food-Animal Production. Front. Microbiol. 2019, 10, 57. [Google Scholar] [CrossRef] [PubMed]
- Hernández-González, J.C.; Martínez-Tapia, A.; Lazcano-Hernández, G.; García-Pérez, B.E.; Castrejón-Jiménez, N.S. Bacteriocins from Lactic Acid Bacteria. A Powerful Alternative as Antimicrobials, Probiotics, and Immunomodulators in Veterinary Medicine. Animals 2021, 11, 979. [Google Scholar] [CrossRef]
- Silva, D.R.; Sardi, J.d.C.O.; Pitangui, N.d.S.; Roque, S.M.; Silva, A.C.B.d.; Rosalen, P.L. Probiotics as an alternative antimicrobial therapy: Current reality and future directions. J. Funct. Foods 2020, 73, 104080. [Google Scholar] [CrossRef]
- Sure, K.P.; Kotnis, P.V.; Bhagwat, P.K.; Ranveer, R.C.; Dandge, P.B.; Sahoo, A.K. Production and characterization of bacteriocin produced by Lactobacillus viridescence (NICM 2167). Braz. Arch. Biol. Technol. 2016, 59, e16150518. [Google Scholar] [CrossRef]
- Tagg, J.R.; Dajani, A.S.; Wannamaker, L.W. Bacteriocin of Gram-positive bacteria. Bacteriol. Rev. 1976, 40, 722–756. [Google Scholar] [CrossRef]
- Jack, R.W.; Tagg, J.R.; Ray, B. Bacteriocins of Gram-positive bacteria. Microbiol. Mol. Biol. Rev. 1995, 59, 171–200. [Google Scholar] [CrossRef]
- Azevedo, P.O.S.; Converti, A.; Gierus, M.; Oliveira, R.P.S. Antimicrobial activity of bacteriocin-like inhibitory substance produced by Pediococcus pentosaceus: From shake flasks to bioreactor. Mol. Biol. Rep. 2018, 45, 461–469. [Google Scholar] [CrossRef]
- Pereira, W.A.; Piazentin, A.C.M.; de Oliveira, R.C.; Mendonça, C.M.N.; Tabata, Y.A.; Mendes, M.A.; Fock, R.A.; Makiyama, E.N.; Corrêa, B.; Vallejo, M.; et al. Bacteriocinogenic probiotic bacteria isolated from an aquatic environment inhibit the growth of food and fish pathogens. Sci. Rep. 2022, 12, 5530. [Google Scholar] [CrossRef]
- Zulkhairi Amin, F.A.; Sabri, S.; Ismail, M.; Chan, K.W.; Ismail, N.; Mohd Esa, N.; Mohd Lila, M.A.; Zawawi, N. Probiotic Properties of Bacillus Strains Isolated from Stingless Bee (Heterotrigona itama) Honey Collected across Malaysia. Int. J. Environ. Res. Public Health 2019, 17, 278. [Google Scholar] [CrossRef]
- International Scientific Association for Probiotics and Prebiotics 2010 Meeting Report—International Scientific Association for Probiotics and Prebiotics (ISAPP). Available online: https://isappscience.org/publications/international-scientific-association-for-probiotics-and-prebiotics-2010-meeting-report/ (accessed on 8 November 2022).
- Riboulet-Bisson, E.; Sturme, M.H.J.; Jeffery, I.B.; O’Donnell, M.M.; Neville, B.A.; Forde, B.M.; Claesson, M.J.; Harris, H.; Gardiner, G.E.; Casey, P.G.; et al. Effect of Lactobacillus salivarius bacteriocin Abp118 on the mouse and pig intestinal microbiota. PLoS ONE 2012, 7, e31113. [Google Scholar] [CrossRef] [PubMed]
- de Souza de Azevedo, P.O.; Converti, A.; Gierus, M.; de Souza Oliveira, R.P. Application of nisin as biopreservative of pork meat by dipping and spraying methods. Braz. J. Microbiol. 2019, 50, 523–526. [Google Scholar] [CrossRef]
- Garcés, M.E.; Olivera, N.L.; Fernández, M.; Riva Rossi, C.; Sequeiros, C. Antimicrobial activity of bacteriocin-producing Carnobacterium spp. isolated from healthy Patagonian trout and their potential for use in aquaculture. Aquac. Res. 2020, 51, 4602–4612. [Google Scholar] [CrossRef]
- Kuniyoshi, T.M.; Mendonça, C.M.N.; Vieira, V.B.; Robl, D.; de Melo Franco, B.D.G.; Todorov, S.D.; Tomé, E.; O’Connor, P.M.; Converti, A.; Araújo, W.L.; et al. Pediocin PA-1 production by Pediococcus pentosaceus ET34 using non-detoxified hemicellulose hydrolysate obtained from hydrothermal pretreatment of sugarcane bagasse. Bioresour. Technol. 2021, 338, 125565. [Google Scholar] [CrossRef]
- Balouiri, M.; Sadiki, M.; Ibnsouda, S.K. Methods for in vitro evaluating antimicrobial activity: A review. J. Pharm. Anal. 2016, 6, 71–79. [Google Scholar] [CrossRef]
- Dias, M.; Lacerda, J.T.J.G.; Andrade, L.M.; Nascimento, C.A.O.; Rozas, E.E.; Mendes, M.A. Proteomic analysis of a copper mine isolated fungus Rhizopus microspores IOC 4686 when exposed to copper sulfate. Arch. Biochem. 2022, 112, 1447–1452. [Google Scholar] [CrossRef]
- van Heel, A.J.; de Jong, A.; Song, C.; Viel, J.H.; Kok, J.; Kuipers, O.P. BAGEL4: A user-friendly web server to thoroughly mine RiPPs and bacteriocins. Nucleic Acids Res. 2018, 46, W278–W281. [Google Scholar] [CrossRef]
- Wosinska, L.; Walsh, C.J.; O’Connor, P.M.; Lawton, E.M.; Cotter, P.D.; Guinane, C.M.; O’Sullivan, O. In Vitro and In Silico Based Approaches to Identify Potential Novel Bacteriocins from the Athlete Gut Microbiome of an Elite Athlete Cohort. Microorganisms 2022, 10, 701. [Google Scholar] [CrossRef]
- Stothard, P. The sequence manipulation suite: JavaScript programs for analyzing and formatting protein and DNA sequences. BioTechniques 2000, 28, 1102–1104. [Google Scholar] [CrossRef]
- Cardelle-Cobas, A.; Coy-Girón, L.; Cepeda, A.; Nebot, C. Swine production: Probiotics as an alternative to the use of antibiotics. In Antibiotics and Probiotics in Animal Food—Impact and Regulation [Working Title]; Veterinary medicine and science; IntechOpen: London, UK, 2022. [Google Scholar]
- Lanza, V.F.; Tedim, A.P.; Martínez, J.L.; Baquero, F.; Coque, T.M. The Plasmidome of Firmicutes: Impact on the Emergence and the Spread of Resistance to Antimicrobials. Microbiol. Spectr. 2015, 3, PLAS-0039. [Google Scholar] [CrossRef]
- Knecht, D.; Cholewińska, P.; Jankowska-Mąkosa, A.; Czyż, K. Development of Swine’s Digestive Tract Microbiota and Its Relation to Production Indices-A Review. Animals 2020, 10, 527. [Google Scholar] [CrossRef]
- Aarestrup, F.M.; Oliver Duran, C.; Burch, D.G.S. Antimicrobial resistance in swine production. Anim. Health Res. Rev. 2008, 9, 135–148. [Google Scholar] [CrossRef]
- Ben Lagha, A.; Haas, B.; Gottschalk, M.; Grenier, D. Antimicrobial potential of bacteriocins in poultry and swine production. Vet. Res. 2017, 48, 22. [Google Scholar] [CrossRef]
- Monteiro, M.S.; Matias, D.N.; Poor, A.P.; Dutra, M.C.; Moreno, L.Z.; Parra, B.M.; Silva, A.P.S.; Matajira, C.E.C.; de Moura Gomes, V.T.; Barbosa, M.R.F.; et al. Causes of Sow Mortality and Risks to Post-Mortem Findings in a Brazilian Intensive Swine Production System. Animals 2022, 12, 1804. [Google Scholar] [CrossRef]
- Loree, J.; Lappin, S.L. Bacteriostatic Antibiotics; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Hwanhlem, N.; Ivanova, T.; Haertlé, T.; Jaffrès, E.; Dousset, X. Inhibition of food-spoilage and foodborne pathogenic bacteria by a nisin Z-producing Lactococcus lactis subsp. lactis KT2W2L. LWT—Food Science and Technology 2017, 82, 170–175. [Google Scholar] [CrossRef]
- Noonpakdee, W.; Santivarangkna, C.; Jumriangrit, P.; Sonomoto, K.; Panyim, S. Isolation of nisin-producing Lactococcus lactis WNC 20 strain from nham, a traditional Thai fermented sausage. Int. J. Food Microbiol. 2003, 81, 137–145. [Google Scholar] [CrossRef]
- Pellegrino, M.S.; Frola, I.D.; Natanael, B.; Gobelli, D.; Nader-Macias, M.E.F.; Bogni, C.I. In Vitro Characterization of Lactic Acid Bacteria Isolated from Bovine Milk as Potential Probiotic Strains to Prevent Bovine Mastitis. Probiotics Antimicrob. Proteins 2019, 11, 74–84. [Google Scholar] [CrossRef]
- Nieto-Lozano, J.C.; Reguera-Useros, J.I.; Peláez-Martínez, M.D.C.; Hardisson de la Torre, A. Effect of a bacteriocin produced by Pediococcus acidilactici against Listeria monocytogenes and Clostridium perfringens on Spanish raw meat. Meat Sci. 2006, 72, 57–61. [Google Scholar] [CrossRef][Green Version]
- Hyun, W.B.; Kang, H.S.; Lee, J.W.; Abraha, H.B.; Kim, K.-P. A newly-isolated Bacillus subtilis BSC35 produces bacteriocin-like inhibitory substance with high potential to control Clostridium perfringens in food. LWT 2021, 138, 110625. [Google Scholar] [CrossRef]
- Heo, S.; Kim, M.G.; Kwon, M.; Lee, H.S.; Kim, G.-B. Inhibition of Clostridium perfringens using Bacteriophages and Bacteriocin Producing Strains. Korean J. Food Sci. Anim. Resour. 2018, 38, 88–98. [Google Scholar] [CrossRef]
- Talukdar, P.K.; Udompijitkul, P.; Hossain, A.; Sarker, M.R. Inactivation Strategies for Clostridium perfringens Spores and Vegetative Cells. Appl. Environ. Microbiol. 2017, 83, e02731-16. [Google Scholar] [CrossRef]
- García, S.; Heredia, N. Clostridium perfringens: A Dynamic Foodborne Pathogen. Food Bioprocess Technol. 2011, 4, 624–630. [Google Scholar] [CrossRef]
- Andersson, A.; Ronner, U.; Granum, P.E. What problems does the food industry have with the spore-forming pathogens Bacillus cereus and Clostridium perfringens? Int. J. Food Microbiol. 1995, 28, 145–155. [Google Scholar] [CrossRef]
- Griffith, R.W.; Carlson, S.A.; Krull, A.C. Salmonellosis. In Diseases of Swine; Zimmerman, J.J., Karriker, L.A., Ramirez, A., Schwartz, K.J., Stevenson, G.W., Zhang, J., Eds.; Wiley: Hoboken, NJ, USA, 2019; pp. 912–925. ISBN 9781119350859. [Google Scholar] [CrossRef]
- Shin, J.M.; Gwak, J.W.; Kamarajan, P.; Fenno, J.C.; Rickard, A.H.; Kapila, Y.L. Biomedical applications of nisin. J. Appl. Microbiol. 2016, 120, 1449–1465. [Google Scholar] [CrossRef]
- Pieterse, R.; Todorov, S.D. Bacteriocins—Exploring alternatives to antibiotics in mastitis treatment. Braz. J. Microbiol 2010, 41, 542–562. [Google Scholar] [CrossRef]
- Yount, N.Y.; Yeaman, M.R. Peptide antimicrobials: Cell wall as a bacterial target. Ann. N. Y. Acad. Sci. 2013, 1277, 127–138. [Google Scholar] [CrossRef]
- Gao, Y.; van Belkum, M.J.; Stiles, M.E. The outer membrane of gram-negative bacteria inhibits antibacterial activity of brochocin-C. Appl. Environ. Microbiol. 1999, 65, 4329–4333. [Google Scholar] [CrossRef]
- Zhu, H.; Han, L.; Ni, Y.; Yu, Z.; Wang, D.; Zhou, J.; Li, B.; Zhang, W.; He, K. In vitro and In vivo Antibacterial Effects of Nisin Against Streptococcus suis. Probiotics Antimicrob. Proteins 2021, 13, 598–610. [Google Scholar] [CrossRef]
- Lebel, G.; Piché, F.; Frenette, M.; Gottschalk, M.; Grenier, D. Antimicrobial activity of nisin against the swine pathogen Streptococcus suis and its synergistic interaction with antibiotics. Peptides 2013, 50, 19–23. [Google Scholar] [CrossRef] [PubMed]
- Takada, K.; Hirasawa, M.; Ikeda, T. Isolation and purification of bacteriocin from Prevotella intermedia (Bacteroides intermedius). J. Periodontol. 1991, 62, 439–444. [Google Scholar] [CrossRef]
- Singh, P.K.; Sharma, S.; Kumari, A.; Korpole, S. A non-pediocin low molecular weight antimicrobial peptide produced by Pediococcus pentosaceus strain IE-3 shows increased activity under reducing environment. BMC Microbiol. 2014, 14, 226. [Google Scholar] [CrossRef] [PubMed]
- Propionibacteria peptide microcin—Dimensions. Available online: https://app.dimensions.ai/details/patent/US-5635484-A (accessed on 20 November 2022).
- Gradisteanu Pircalabioru, G.; Popa, L.I.; Marutescu, L.; Gheorghe, I.; Popa, M.; Czobor Barbu, I.; Cristescu, R.; Chifiriuc, M.-C. Bacteriocins in the era of antibiotic resistance: Rising to the challenge. Pharmaceutics 2021, 13, 196. [Google Scholar] [CrossRef]
- Dowarah, R.; Verma, A.K.; Agarwal, N. The use of Lactobacillus as an alternative of antibiotic growth promoters in pigs: A review. Anim. Nutr. 2017, 3, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo-Cantabrana, C.; Delgado, S.; Ruiz, L.; Ruas-Madiedo, P.; Sánchez, B.; Margolles, A. Bifidobacteria and their health-promoting effects. In Bugs as Drugs: Therapeutic Microbes for the Prevention and Treatment of Disease; Britton, R.A., Cani, P.D., Eds.; ASM Press: Washington, DC, USA, 2018; pp. 73–98. ISBN 9781555819699. [Google Scholar] [CrossRef]
- Holmer, I.; Salomonsen, C.M.; Jorsal, S.E.; Astrup, L.B.; Jensen, V.F.; Høg, B.B.; Pedersen, K. Antibiotic resistance in porcine pathogenic bacteria and relation to antibiotic usage. BMC Vet. Res. 2019, 15, 449. [Google Scholar] [CrossRef] [PubMed]
- Thacker, P.A. Alternatives to antibiotics as growth promoters for use in swine production: A review. J. Anim. Sci. Biotechnol. 2013, 4, 35. [Google Scholar] [CrossRef]
- Gonzalez-Ronquillo, M.; Villegas-Estrada, D.; Robles-Jimenez, L.E.; Garcia Herrera, R.A.; Villegas-Vázquez, V.L.; Vargas-Bello-Pérez, E. Effect of the Inclusion of Bacillus spp. in Growing-Finishing Pigs’ Diets: A Meta-Analysis. Animals 2022, 12, 2269. [Google Scholar] [CrossRef]

| Pathogenic Bacteria | Isolation Site | Clinical Case | Age | State | Year |
|---|---|---|---|---|---|
| Streptococcus suis | Lung | Pneumonia | Nursery pig | São Paulo | 2016 |
| Streptococcus dysgalactiae | Brain | Meningitis | Nursery pig | Mato Grosso | 2019 |
| Staphylococcus hyicus | Skin | Pyoderma | Finishing pig | Santa Catarina | 2013 |
| Enterococcus faecalis | Spleen | Septicemia | Sow | São Paulo | 2017 |
| Salmonella enterica serovar Choleraesuis | Lung | Septicemia | Finishing pig | Paraná | 2015 |
| Salmonella enterica serovar Typhimurium | Feces | Enteritis | Sow | Santa Catarina | 2012 |
| Escherichia coli | Feces | Enteritis | Unweaned piglet | Rio Grande do Sul | 2012 |
| Clostridium perfringens | Feces | Enteritis | Unweaned piglet | São Paulo | 2022 |
| Pathogenic Strains | Area of Inhibition (cm2) | Diameter of Inhibition Halo (mm) | Antimicrobial Activity |
|---|---|---|---|
| E. coli | - | - | - |
| S. enterica serovar Typhimurium | - | - | - |
| S. enterica serovar Choleraesuis | - | - | - |
| S. hyicus | 0.46 | 5.20 | 5.80 |
| S. dysgalactiae | 0.28 | 4.60 | 3.49 |
| S. suis | 0.67 | 5.80 | 8.39 |
| E. faecalis | 1.58 | 7.90 | 19.69 |
| C. perfringens | 1.11 | 6.90 | 13.88 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sanca, F.M.M.; Blanco, I.R.; Dias, M.; Moreno, A.M.; Martins, S.M.M.K.; Stephano, M.A.; Mendes, M.A.; Mendonça, C.M.N.; Pereira, W.A.; Azevedo, P.O.S.; et al. Antimicrobial Activity of Peptides Produced by Lactococcus lactis subsp. lactis on Swine Pathogens. Animals 2023, 13, 2442. https://doi.org/10.3390/ani13152442
Sanca FMM, Blanco IR, Dias M, Moreno AM, Martins SMMK, Stephano MA, Mendes MA, Mendonça CMN, Pereira WA, Azevedo POS, et al. Antimicrobial Activity of Peptides Produced by Lactococcus lactis subsp. lactis on Swine Pathogens. Animals. 2023; 13(15):2442. https://doi.org/10.3390/ani13152442
Chicago/Turabian StyleSanca, Fernando M. M., Iago R. Blanco, Meriellen Dias, Andrea M. Moreno, Simone M. M. K. Martins, Marco A. Stephano, Maria A. Mendes, Carlos M. N. Mendonça, Wellison A. Pereira, Pamela O. S. Azevedo, and et al. 2023. "Antimicrobial Activity of Peptides Produced by Lactococcus lactis subsp. lactis on Swine Pathogens" Animals 13, no. 15: 2442. https://doi.org/10.3390/ani13152442
APA StyleSanca, F. M. M., Blanco, I. R., Dias, M., Moreno, A. M., Martins, S. M. M. K., Stephano, M. A., Mendes, M. A., Mendonça, C. M. N., Pereira, W. A., Azevedo, P. O. S., Gierus, M., & Oliveira, R. P. S. (2023). Antimicrobial Activity of Peptides Produced by Lactococcus lactis subsp. lactis on Swine Pathogens. Animals, 13(15), 2442. https://doi.org/10.3390/ani13152442








