Two Amnion-Derived Mesenchymal Stem-Cells Injections to Osteoarthritic Elbows in Dogs—Pilot Study
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Population Study
2.2. Isolation of MSC from Amniotic Tissue
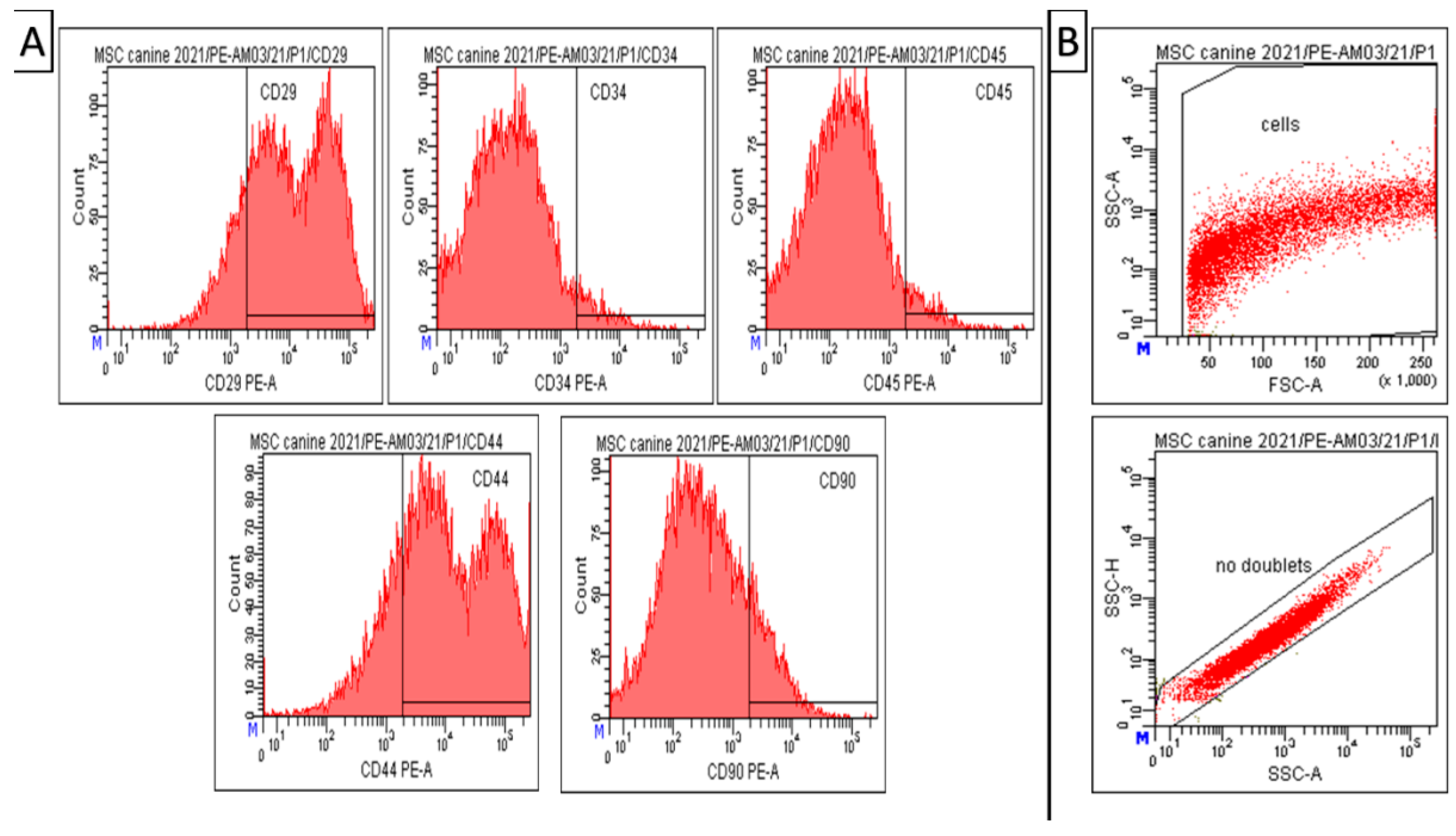
2.3. CD Characterization of Amniotic Mesenchymal Stem Cells
2.4. Multilineage Potential
2.5. Application of MSCs
2.6. Kinematic Gait Analysis
2.7. Synovial Fluid Analysis
- We added 200 µL of WashBuffer into each well of the plate. The plate was sealed and mixed on a plate shaker for 10 min at room temperature (20–25 °C);
- The WashBuffer was decanted and removed to the residual amount from all wells by inverting the plate and tapping it smartly onto absorbent towels several times;
- 25 µL of each standard or control was added into the appropriate wells. Assay Buffer was used for 0 pg/mL standard (background);
- 25 µL of Assay Buffer was added to the sample wells;
- 25 µL of appropriate matrix solution was added to the background, standards, and control wells;
- 25 μL of the sample was added into the appropriate wells;
- The mixing bottle was vortexed and 25 μL of the premixed beads were added to each well;
- The plate was sealed with a plate sealer. The plate was wrapped with foil and incubated with agitation on a plate shaker overnight at 4 °C or 2 h at room temperature (20–25 °C);
- The well contents were gently removed and the plate was washed twice following instructions listed in the plate-washing section;
- 25 µL of detection antibodies were added into each well;
- The cover was sealed with foil and incubated with agitation on a plate shaker for 1 h at room temperature (20–25 °C);
- 25 µL Streptavidin–Phycoerythrin was added to each well containing the 25 µL of detection antibodies;
- The cover was sealed with foil and incubated with agitation on a plate shaker for 30 min at room temperature (20–25 °C);
- The well contents were gently removed and the plate was washed twice following instructions listed in the plate-washing section;
- 150 µL of Drive Fluid PLUS was added to all wells. Beads were resuspended on a plate shaker for 5 min;
- The plate was run on MAGPIX®;
- Median fluorescent intensity (MFI) data were saved and analyzed using a spline curve-fitting method for calculating cytokine/chemokines concentrations in samples.
2.8. Statistical Analysis
3. Results
3.1. Isolation of Canine MSCs from Amnion
3.2. Phenotypization of MSCs
3.3. Multilineage Potential
3.4. Clinical Examination Post MSCs Application
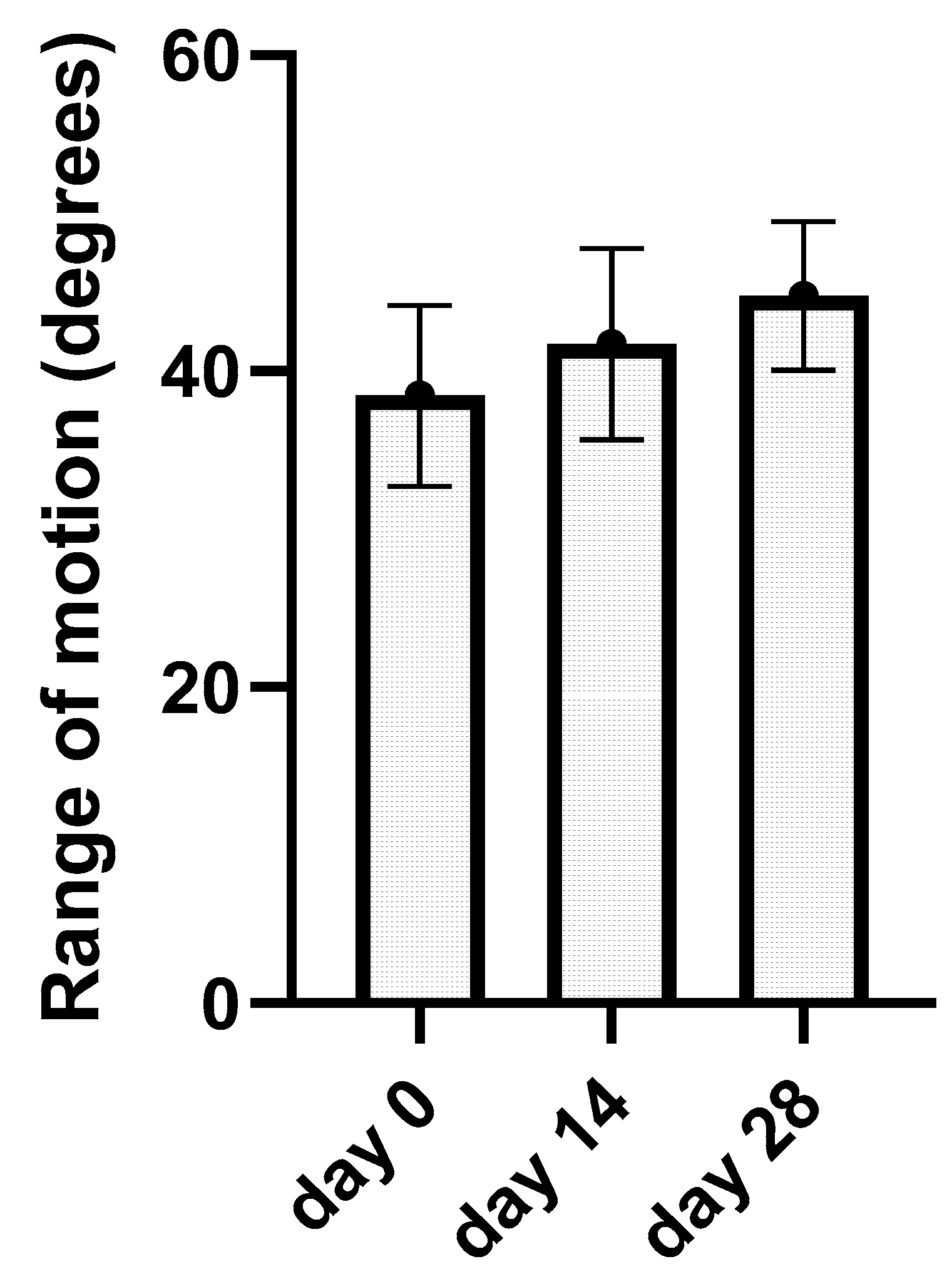
3.5. Kinematic Gait Analysis
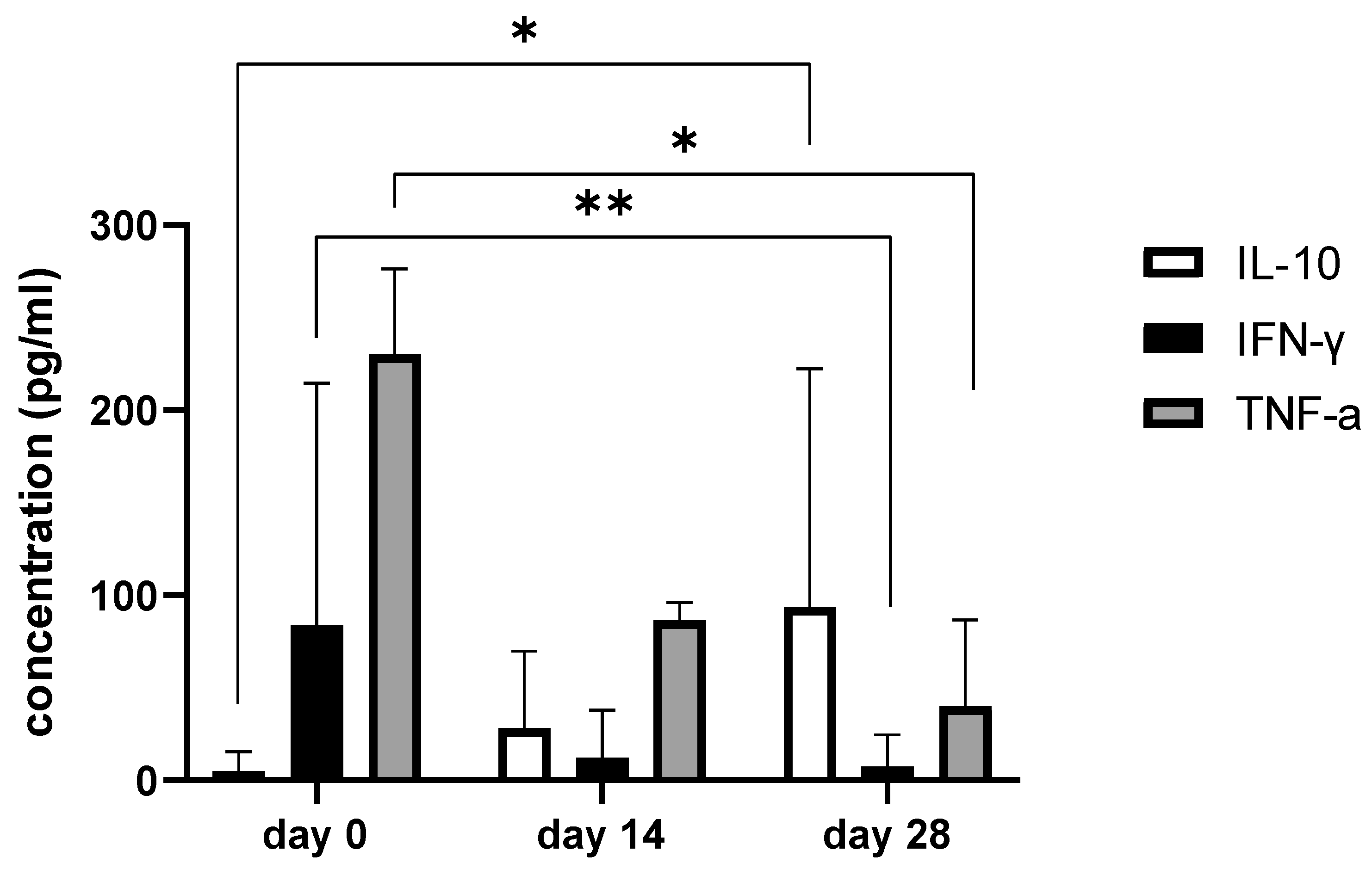
3.6. Synovial Fluid Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Anderson, K.L.; Zulch, H.; O’Neill, D.G.; Meeson, R.L.; Collins, L.M. Risk Factors for Canine Osteoarthritis and Its Predisposing Arthropathies: A Systematic Review. Front. Vet. Sci. 2020, 7, 220. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, D.G.; Church, D.B.; McGreevy, P.D.; Thomson, P.C.; Brodbelt, D.C. Prevalence of Disorders Recorded in Dogs Attending Primary-Care Veterinary Practices in England. PLoS ONE 2014, 9, e90501. [Google Scholar] [CrossRef] [PubMed]
- Clements, D.N.; Carter, S.D.; Innes, J.F.; Ollier, W.E.R. Genetic Basis of Secondary Osteoarthritis in Dogs with Joint Dysplasia. Am. J. Vet. Res. 2006, 67, 909–918. [Google Scholar] [CrossRef] [PubMed]
- Kuppa, S.S.; Kim, H.K.; Kang, J.Y.; Lee, S.C.; Seon, J.K. Role of Mesenchymal Stem Cells and Their Paracrine Mediators in Macrophage Polarization: An Approach to Reduce Inflammation in Osteoarthritis. Int. J. Mol. Sci. 2022, 23, 13016. [Google Scholar] [CrossRef]
- Bijlsma, J.W.J.; Berenbaum, F.; Lafeber, F.P.J.G. Osteoarthritis: An Update with Relevance for Clinical Practice. Lancet 2011, 377, 2115–2126. [Google Scholar] [CrossRef]
- Meeson, R.L.; Todhunter, R.J.; Blunn, G.; Nuki, G.; Pitsillides, A.A. Spontaneous Dog Osteoarthritis—A One Medicine Vision. Nat. Rev. Rheumatol. 2019, 15, 273–287. [Google Scholar] [CrossRef]
- Lane, N.E.; Brandt, K.; Hawker, G.; Peeva, E.; Schreyer, E.; Tsuji, W.; Hochberg, M.C. OARSI-FDA Initiative: Defining the Disease State of Osteoarthritis. Osteoarthr. Cartil. 2011, 19, 478–482. [Google Scholar] [CrossRef]
- Spector, T.D.; MacGregor, A.J. Risk Factors for Osteoarthritis: Genetics. Osteoarthr. Cartil. 2004, 12 (Suppl. A), S39–S44. [Google Scholar] [CrossRef]
- Tsezou, A. Osteoarthritis Year in Review 2014: Genetics and Genomics. Osteoarthr. Cartil. 2014, 22, 2017–2024. [Google Scholar] [CrossRef]
- Sallander, M.H.; Hedhammar, Å.; Trogen, M.E. Diet, Exercise, and Weight as Risk Factors in Hip Dysplasia and Elbow Arthrosis in Labrador Retrievers. J. Nutr. 2006, 136, 2050S–2052S. [Google Scholar] [CrossRef]
- Francisco, V.; Pérez, T.; Pino, J.; López, V.; Franco, E.; Alonso, A.; Gonzalez-Gay, M.A.; Mera, A.; Lago, F.; Gómez, R.; et al. Biomechanics, Obesity, and Osteoarthritis. The Role of Adipokines: When the Levee Breaks. J. Orthop. Res. 2017, 36, 594–604. [Google Scholar] [CrossRef]
- Coopman, F.; Verhoeven, G.; Saunders, J.; Duchateau, L.; Van Bree, H. Prevalence of Hip Dysplasia, Elbow Dysplasia and Humeral Head Osteochondrosis in Dog Breeds in Belgium. Vet. Rec. 2008, 163, 654–658. [Google Scholar] [CrossRef]
- Hou, Y.; Wang, Y.; Lu, X.; Zhang, X.; Zhao, Q.; Todhunter, R.J.; Zhang, Z. Correction: Monitoring Hip and Elbow Dysplasia Achieved Modest Genetic Improvement of 74 Dog Breeds over 40 Years in USA. PLoS ONE 2013, 8, e76390. [Google Scholar] [CrossRef]
- Lavrijsen, I.C.M.; Heuven, H.C.M.; Meij, B.P.; Theyse, L.F.H.; Nap, R.C.; Leegwater, P.A.J.; Hazewinkel, H.A.W. Prevalence and Co-Occurrence of Hip Dysplasia and Elbow Dysplasia in Dutch Pure-Bred Dogs. Prev. Vet. Med. 2014, 114, 114–122. [Google Scholar] [CrossRef]
- Wesseling, J.; Bierma-Zeinstra, S.M.A.; Kloppenburg, M.; Meijer, R.; Bijlsma, J.W.J. Worsening of Pain and Function over 5 Years in Individuals with “early” OA Is Related to Structural Damage: Data from the Osteoarthritis Initiative and CHECK (Cohort Hip & Cohort Knee) Study. Ann. Rheum. Dis. 2015, 74, 347–353. [Google Scholar] [CrossRef]
- Dieppe, P.A.; Lohmander, L.S. Pathogenesis and Management of Pain in Osteoarthritis. Lancet 2005, 365, 965–973. [Google Scholar] [CrossRef]
- Gierman, L.M.; van der Ham, F.; Koudijs, A.; Wielinga, P.Y.; Kleemann, R.; Kooistra, T.; Stoop, R.; Kloppenburg, M.; van Osch, G.J.V.M.; Stojanovic-Susulic, V.; et al. Metabolic Stress-Induced Inflammation Plays a Major Role in the Development of Osteoarthritis in Mice. Arthritis Rheum. 2012, 64, 1172–1181. [Google Scholar] [CrossRef]
- Clements, D.N.; Carter, S.D.; Innes, J.F.; Ollier, W.E.R.; Day, P.J.R. Gene Expression Profiling of Normal and Ruptured Canine Anterior Cruciate Ligaments. Osteoarthr. Cartil. 2008, 16, 195–203. [Google Scholar] [CrossRef]
- Duval, J.M.; Budsberg, S.C.; Flo, G.L.; Sammarco, J.L. Breed, Sex, and Body Weight as Risk Factors for Rupture of the Cranial Cruciate Ligament in Young Dogs. J. Am. Vet. Med. Assoc. 1999, 215, 811–814. [Google Scholar]
- O’Neill, D.G.; Brodbelt, D.C.; Hodge, R.; Church, D.B.; Meeson, R.L. Epidemiology and Clinical Management of Elbow Joint Disease in Dogs under Primary Veterinary Care in the UK. Canine Med. Genet. 2020, 7, 1. [Google Scholar] [CrossRef]
- Fitzpatrick, N.; Smith, T.J.; Evans, R.B.; Yeadon, R. Radiographic and Arthroscopic Findings in the Elbow Joints of 263 Dogs with Medial Coronoid Disease. Vet. Surg. 2009, 38, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Molnar, V.; Matišić, V.; Kodvanj, I.; Bjelica, R.; Jeleč, Ž.; Hudetz, D.; Rod, E.; Čukelj, F.; Vrdoljak, T.; Vidović, D.; et al. Cytokines and Chemokines Involved in Osteoarthritis Pathogenesis. Int. J. Mol. Sci. 2021, 22, 9208. [Google Scholar] [CrossRef] [PubMed]
- Johnston, S.A.; Tobias, K.M. (Eds.) Veterinary Surgery: Small Animal, 2nd ed.; Elsevier: St. Louis, MO, USA, 2018; ISBN 978-0-323-32065-8. [Google Scholar]
- Vonkeman, H.E.; van de Laar, M.A.F.J. Nonsteroidal Anti-Inflammatory Drugs: Adverse Effects and Their Prevention. Semin. Arthritis Rheum. 2010, 39, 294–312. [Google Scholar] [CrossRef] [PubMed]
- Patrignani, P.; Tacconelli, S.; Bruno, A.; Sostres, C.; Lanas, A. Managing the Adverse Effects of Nonsteroidal Anti-Inflammatory Drugs. Expert. Rev. Clin. Pharmacol. 2011, 4, 605–621. [Google Scholar] [CrossRef] [PubMed]
- Bhala, N.; Emberson, J.; Merhi, A.; Abramson, S.; Arber, N.; Baron, J.A.; Bombardier, C.; Cannon, C.; Farkouh, M.E.; FitzGerald, G.A.; et al. Vascular and Upper Gastrointestinal Effects of Non-Steroidal Anti-Inflammatory Drugs: Meta-Analyses of Individual Participant Data from Randomised Trials. Lancet 2013, 382, 769–779. [Google Scholar] [CrossRef]
- Dajani, E.Z.; Islam, K. Cardiovascular and Gastrointestinal Toxicity of Selective Cyclo-Oxygenase-2 Inhibitors in Man. J. Physiol. Pharmacol. 2008, 59 (Suppl. 2), 117–133. [Google Scholar]
- KuKanich, B. Pharmacokinetics of Acetaminophen, Codeine, and the Codeine Metabolites Morphine and Codeine-6-Glucuronide in Healthy Greyhound Dogs. J. Vet. Pharmacol. Ther. 2010, 33, 15–21. [Google Scholar] [CrossRef]
- Onakpoya, I.J. Paracetamol as First Line for Treatment of Knee and Hip Osteoarthritis. BMJ Evid. Based Med. 2020, 25, 40. [Google Scholar] [CrossRef]
- Bannuru, R.R.; Osani, M.C.; Vaysbrot, E.E.; Arden, N.K.; Bennell, K.; Bierma-Zeinstra, S.M.A.; Kraus, V.B.; Lohmander, L.S.; Abbott, J.H.; Bhandari, M.; et al. OARSI Guidelines for the Non-Surgical Management of Knee, Hip, and Polyarticular Osteoarthritis. Osteoarthr. Cartil. 2019, 27, 1578–1589. [Google Scholar] [CrossRef]
- Hunt, J.R.; Dean, R.S.; Davis, G.N.D.; Murrell, J.C. An Analysis of the Relative Frequencies of Reported Adverse Events Associated with NSAID Administration in Dogs and Cats in the United Kingdom. Vet. J. 2015, 206, 183–190. [Google Scholar] [CrossRef]
- Monteiro-Steagall, B.P.; Steagall, P.V.M.; Lascelles, B.D.X. Systematic Review of Nonsteroidal Anti-Inflammatory Drug-Induced Adverse Effects in Dogs. J. Vet. Intern. Med. 2013, 27, 1011–1019. [Google Scholar] [CrossRef]
- Monteiro, B.; Steagall, P.V. Antiinflammatory Drugs. Vet. Clin. N. Am. Small Anim. Pract. 2019, 49, 993–1011. [Google Scholar] [CrossRef]
- Toutain, C.E.; Brossard, P.; King, S.B.; Helbig, R. Six-Month Safety Evaluation of Robenacoxib Tablets (OnsiorTM) in Dogs after Daily Oral Administrations. BMC Vet. Res. 2018, 14, 242. [Google Scholar] [CrossRef]
- Enomoto, M.; Mantyh, P.W.; Murrell, J.; Innes, J.F.; Lascelles, B.D.X. Anti-Nerve Growth Factor Monoclonal Antibodies for the Control of Pain in Dogs and Cats. Vet. Rec. 2019, 184, 23. [Google Scholar] [CrossRef]
- Mancuso, P.; Raman, S.; Glynn, A.; Barry, F.; Murphy, J.M. Mesenchymal Stem Cell Therapy for Osteoarthritis: The Critical Role of the Cell Secretome. Front. Bioeng. Biotechnol. 2019, 7, 9. [Google Scholar] [CrossRef]
- Black, L.L.; Gaynor, J.; Adams, C.; Dhupa, S.; Sams, A.E.; Taylor, R.; Harman, S.; Gingerich, D.A.; Harman, R. Effect of Intraarticular Injection of Autologous Adipose-Derived Mesenchymal Stem and Regenerative Cells on Clinical Signs of Chronic Osteoarthritis of the Elbow Joint in Dogs. Vet. Ther. 2008, 9, 192–200. [Google Scholar]
- Guercio, A.; Di Marco, P.; Casella, S.; Cannella, V.; Russotto, L.; Purpari, G.; Di Bella, S.; Piccione, G. Production of Canine Mesenchymal Stem Cells from Adipose Tissue and Their Application in Dogs with Chronic Osteoarthritis of the Humeroradial Joints. Cell Biol. Int. 2012, 36, 189–194. [Google Scholar] [CrossRef]
- Kriston-Pál, É.; Czibula, Á.; Gyuris, Z.; Balka, G.; Seregi, A.; Sükösd, F.; Süth, M.; Kiss-Tóth, E.; Haracska, L.; Uher, F.; et al. Characterization and Therapeutic Application of Canine Adipose Mesenchymal Stem Cells to Treat Elbow Osteoarthritis. Can. J. Vet. Res. 2017, 81, 73–78. [Google Scholar]
- Spasovski, D.; Spasovski, V.; Baščarević, Z.; Stojiljković, M.; Vreća, M.; Anđelković, M.; Pavlović, S. Intra-Articular Injection of Autologous Adipose-Derived Mesenchymal Stem Cells in the Treatment of Knee Osteoarthritis. J. Gene Med. 2018, 20, e3002. [Google Scholar] [CrossRef]
- Jo, C.H.; Lee, Y.G.; Shin, W.H.; Kim, H.; Chai, J.W.; Jeong, E.C.; Kim, J.E.; Shim, H.; Shin, J.S.; Shin, I.S.; et al. Intra-Articular Injection of Mesenchymal Stem Cells for the Treatment of Osteoarthritis of the Knee: A Proof-of-Concept Clinical Trial. Stem Cells 2014, 32, 1254–1266. [Google Scholar] [CrossRef]
- Pers, Y.-M.; Ruiz, M.; Noël, D.; Jorgensen, C. Mesenchymal Stem Cells for the Management of Inflammation in Osteoarthritis: State of the Art and Perspectives. Osteoarthr. Cartil. 2015, 23, 2027–2035. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Du, H.; Dai, C.; Zhang, L.; Li, S.; Hunter, D.J.; Lu, L.; Bao, C. Human Adipose-Derived Mesenchymal Stem Cells for Osteoarthritis: A Pilot Study with Long-Term Follow-up and Repeated Injections. Regen. Med. 2018, 13, 295–307. [Google Scholar] [CrossRef] [PubMed]
- Davatchi, F.; Abdollahi, B.S.; Mohyeddin, M.; Shahram, F.; Nikbin, B. Mesenchymal Stem Cell Therapy for Knee Osteoarthritis. Preliminary Report of Four Patients. Int. J. Rheum. Dis. 2011, 14, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Orozco, L.; Munar, A.; Soler, R.; Alberca, M.; Soler, F.; Huguet, M.; Sentís, J.; Sánchez, A.; García-Sancho, J. Treatment of Knee Osteoarthritis with Autologous Mesenchymal Stem Cells: A Pilot Study. Transplantation 2013, 95, 1535–1541. [Google Scholar] [CrossRef]
- Tofiño-Vian, M.; Guillén, M.I.; Alcaraz, M.J. Extracellular Vesicles: A New Therapeutic Strategy for Joint Conditions. Biochem. Pharmacol. 2018, 153, 134–146. [Google Scholar] [CrossRef]
- Villatoro, A.J.; Alcoholado, C.; Martín-Astorga, M.C.; Fernández, V.; Cifuentes, M.; Becerra, J. Comparative Analysis and Characterization of Soluble Factors and Exosomes from Cultured Adipose Tissue and Bone Marrow Mesenchymal Stem Cells in Canine Species. Vet. Immunol. Immunopathol. 2019, 208, 6–15. [Google Scholar] [CrossRef]
- Uccelli, A.; Moretta, L.; Pistoia, V. Mesenchymal Stem Cells in Health and Disease. Nat. Rev. Immunol. 2008, 8, 726–736. [Google Scholar] [CrossRef]
- Lamo-Espinosa, J.M.; Mora, G.; Blanco, J.F.; Granero-Moltó, F.; Nuñez-Córdoba, J.M.; Sánchez-Echenique, C.; Bondía, J.M.; Aquerreta, J.D.; Andreu, E.J.; Ornilla, E.; et al. Intra-Articular Injection of Two Different Doses of Autologous Bone Marrow Mesenchymal Stem Cells versus Hyaluronic Acid in the Treatment of Knee Osteoarthritis: Multicenter Randomized Controlled Clinical Trial (Phase I/II). J. Transl. Med. 2016, 14, 246. [Google Scholar] [CrossRef]
- Park, K.-H.; Mun, C.H.; Kang, M.-I.; Lee, S.-W.; Lee, S.-K.; Park, Y.-B. Treatment of Collagen-Induced Arthritis Using Immune Modulatory Properties of Human Mesenchymal Stem Cells. Cell Transplant. 2016, 25, 1057–1072. [Google Scholar] [CrossRef]
- Oliveira, R.L.; Chagastelles, P.C.; Sesterheim, P.; Pranke, P. In Vivo Immunogenic Response to Allogeneic Mesenchymal Stem Cells and the Role of Preactivated Mesenchymal Stem Cells Cotransplanted with Allogeneic Islets. Stem Cells Int. 2017, 2017, 9824698. [Google Scholar] [CrossRef]
- Cabon, Q.; Febre, M.; Gomez, N.; Cachon, T.; Pillard, P.; Carozzo, C.; Saulnier, N.; Robert, C.; Livet, V.; Rakic, R.; et al. Long-Term Safety and Efficacy of Single or Repeated Intra-Articular Injection of Allogeneic Neonatal Mesenchymal Stromal Cells for Managing Pain and Lameness in Moderate to Severe Canine Osteoarthritis Without Anti-Inflammatory Pharmacological Support: Pilot Clinical Study. Front. Vet. Sci. 2019, 6, 10. [Google Scholar] [CrossRef]
- Redondo, J.I.; Rubio, M.; Soler, G.; Serra, I.; Soler, C.; Gómez-Villamandos, R.J. Normal Values and Incidence of Cardiorespiratory Complications in Dogs during General Anaesthesia. A Review of 1281 Cases. J. Vet. Med. A Physiol. Pathol. Clin. Med. 2007, 54, 470–477. [Google Scholar] [CrossRef]
- Shah, K.; Drury, T.; Roic, I.; Hansen, P.; Malin, M.; Boyd, R.; Sumer, H.; Ferguson, R. Outcome of Allogeneic Adult Stem Cell Therapy in Dogs Suffering from Osteoarthritis and Other Joint Defects. Stem Cells Int. 2018, 2018, 7309201. [Google Scholar] [CrossRef]
- English, K.; Barry, F.P.; Field-Corbett, C.P.; Mahon, B.P. IFN-γ and TNF-α Differentially Regulate Immunomodulation by Murine Mesenchymal Stem Cells. Immunol. Lett. 2007, 110, 91–100. [Google Scholar] [CrossRef]
- Chen, K.; Wang, D.; Du, W.T.; Han, Z.-B.; Ren, H.; Chi, Y.; Yang, S.G.; Zhu, D.; Bayard, F.; Han, Z.C. Human Umbilical Cord Mesenchymal Stem Cells HUC-MSCs Exert Immunosuppressive Activities through a PGE2-Dependent Mechanism. Clin. Immunol. 2010, 135, 448–458. [Google Scholar] [CrossRef]
- Martinet, L.; Fleury-Cappellesso, S.; Gadelorge, M.; Dietrich, G.; Bourin, P.; Fournié, J.-J.; Poupot, R. A Regulatory Cross-Talk between Vγ9Vδ2 T Lymphocytes and Mesenchymal Stem Cells: Immunomodulation. Eur. J. Immunol. 2009, 39, 752–762. [Google Scholar] [CrossRef]
- Sotiropoulou, P.A.; Perez, S.A.; Gritzapis, A.D.; Baxevanis, C.N.; Papamichail, M. Interactions between Human Mesenchymal Stem Cells and Natural Killer Cells. Stem Cells 2006, 24, 74–85. [Google Scholar] [CrossRef]
- Aggarwal, S.; Pittenger, M.F. Human Mesenchymal Stem Cells Modulate Allogeneic Immune Cell Responses. Blood 2005, 105, 1815–1822. [Google Scholar] [CrossRef]
- Wisniewski, H.-G.; Vilček, J. Cytokine-Induced Gene Expression at the Crossroads of Innate Immunity, Inflammation and Fertility: TSG-6 and PTX3/TSG-14. Cytokine Growth Factor. Rev. 2004, 15, 129–146. [Google Scholar] [CrossRef]
- Lee, K.B.L.; Wang, V.T.Z.; Chan, Y.H.; Hui, J.H.P. A Novel, Minimally-Invasive Technique of Cartilage Repair in the Human Knee Using Arthroscopic Microfracture and Injections of Mesenchymal Stem Cells and Hyaluronic Acid—A Prospective Comparative Study on Safety and Short-Term Efficacy. Ann. Acad. Med. Singap. 2012, 41, 511–517. [Google Scholar] [CrossRef]
- Mellor, A.L.; Munn, D.H. Ido Expression by Dendritic Cells: Tolerance and Tryptophan Catabolism. Nat. Rev. Immunol. 2004, 4, 762–774. [Google Scholar] [CrossRef] [PubMed]
- Saulnier, N.; Viguier, E.; Perrier-Groult, E.; Chenu, C.; Pillet, E.; Roger, T.; Maddens, S.; Boulocher, C. Intra-Articular Administration of Xenogeneic Neonatal Mesenchymal Stromal Cells Early after Meniscal Injury down-Regulates Metalloproteinase Gene Expression in Synovium and Prevents Cartilage Degradation in a Rabbit Model of Osteoarthritis. Osteoarthr. Cartil. 2015, 23, 122–133. [Google Scholar] [CrossRef] [PubMed]
- Shimazawa, Y.; Kusamori, K.; Tsujimura, M.; Shimomura, A.; Takasaki, R.; Takayama, Y.; Shimizu, K.; Konishi, S.; Nishikawa, M. Intravenous Injection of Mesenchymal Stem Cell Spheroids Improves the Pulmonary Delivery and Prolongs in Vivo Survival. Biotechnol. J. 2022, 17, 2100137. [Google Scholar] [CrossRef] [PubMed]
- Wan, P.-X. Importance of the Stem Cell Microenvironment for Ophthalmological Cell-Based Therapy. WJSC 2015, 7, 448. [Google Scholar] [CrossRef]
- Kook, M.G.; Lee, S.; Shin, N.; Kong, D.; Kim, D.-H.; Kim, M.-S.; Kang, H.K.; Choi, S.W.; Kang, K.-S. Repeated Intramuscular Transplantations of HUCB-MSCs Improves Motor Function and Survival in the SOD1 G93A Mice through Activation of AMPK. Sci. Rep. 2020, 10, 1572. [Google Scholar] [CrossRef]
- Huňáková, K.; Hluchý, M.; Špaková, T.; Matejová, J.; Mudroňová, D.; Kuricová, M.; Rosocha, J.; Ledecký, V. Study of Bilateral Elbow Joint Osteoarthritis Treatment Using Conditioned Medium from Allogeneic Adipose Tissue-Derived MSCs in Labrador Retrievers. Res. Vet. Sci. 2020, 132, 513–520. [Google Scholar] [CrossRef]
- Ha, C.-W.; Park, Y.-B.; Kim, S.H.; Lee, H.-J. Intra-Articular Mesenchymal Stem Cells in Osteoarthritis of the Knee: A Systematic Review of Clinical Outcomes and Evidence of Cartilage Repair. Arthrosc. J. Arthrosc. Relat. Surg. 2019, 35, 277–288.e2. [Google Scholar] [CrossRef]
- McLaughlin, R.M. Kinetic and Kinematic Gait Analysis in Dogs. Vet. Clin. N. Am. Small Anim. Pract. 2001, 31, 193–201. [Google Scholar] [CrossRef]
- Sellam, J.; Berenbaum, F. The Role of Synovitis in Pathophysiology and Clinical Symptoms of Osteoarthritis. Nat. Rev. Rheumatol. 2010, 6, 625–635. [Google Scholar] [CrossRef]
- Kapoor, M.; Martel-Pelletier, J.; Lajeunesse, D.; Pelletier, J.-P.; Fahmi, H. Role of Proinflammatory Cytokines in the Pathophysiology of Osteoarthritis. Nat. Rev. Rheumatol. 2011, 7, 33–42. [Google Scholar] [CrossRef]
- Miller, R.E.; Miller, R.J.; Malfait, A.-M. Osteoarthritis Joint Pain: The Cytokine Connection. Cytokine 2014, 70, 185–193. [Google Scholar] [CrossRef]
- Orita, S.; Koshi, T.; Mitsuka, T.; Miyagi, M.; Inoue, G.; Arai, G.; Ishikawa, T.; Hanaoka, E.; Yamashita, K.; Yamashita, M.; et al. Associations between Proinflammatory Cytokines in the Synovial Fluid and Radiographic Grading and Pain-Related Scores in 47 Consecutive Patients with Osteoarthritis of the Knee. BMC Musculoskelet. Disord. 2011, 12, 144. [Google Scholar] [CrossRef]
- McCoy, A.M. Animal Models of Osteoarthritis: Comparisons and Key Considerations. Vet. Pathol. 2015, 52, 803–818. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, S.; Wang, T. How the Mechanical Microenvironment of Stem Cell Growth Affects Their Differentiation: A Review. Stem Cell Res. Ther. 2022, 13, 415. [Google Scholar] [CrossRef]
- Li, L.; Chen, X.; Wang, W.E.; Zeng, C. How to Improve the Survival of Transplanted Mesenchymal Stem Cell in Ischemic Heart? Stem Cells Int. 2016, 2016, 9682757. [Google Scholar] [CrossRef]
- Zhang, B.; Wang, B.; Li, S.; Luo, D.; Zhan, X.; Chen, S.; Chen, Z.; Liu, C.; Ji, H.; Bai, Y.; et al. Evaluation of the Curative Effect of Umbilical Cord Mesenchymal Stem Cell Therapy for Knee Arthritis in Dogs Using Imaging Technology. Stem Cells Int. 2018, 2018, 1983025. [Google Scholar] [CrossRef]
- Harrell, C.R.; Sadikot, R.; Pascual, J.; Fellabaum, C.; Jankovic, M.G.; Jovicic, N.; Djonov, V.; Arsenijevic, N.; Volarevic, V. Mesenchymal Stem Cell-Based Therapy of Inflammatory Lung Diseases: Current Understanding and Future Perspectives. Stem Cells Int. 2019, 2019, 4236973. [Google Scholar] [CrossRef]
- Cachon, T.; Frykman, O.; Innes, J.F.; Lascelles, B.D.X.; Okumura, M.; Sousa, P.; Staffieri, F.; Steagall, P.V.; Van Ryssen, B. Face Validity of a Proposed Tool for Staging Canine Osteoarthritis: Canine OsteoArthritis Staging Tool (COAST). Vet. J. 2018, 235, 1–8. [Google Scholar] [CrossRef]
- Pelletier, J.P.; Faure, M.P.; DiBattista, J.A.; Wilhelm, S.; Visco, D.; Martel-Pelletier, J. Coordinate Synthesis of Stromelysin, Interleukin-1, and Oncogene Proteins in Experimental Osteoarthritis. An Immunohistochemical Study. Am. J. Pathol. 1993, 142, 95–105. [Google Scholar]
- Goldring, M.B. The Role of Cytokines as Inflammatory Mediators in Osteoarthritis: Lessons from Animal Models. Connect. Tissue Res. 1999, 40, 1–11. [Google Scholar] [CrossRef]
- Schlaak, J.F.; Schwarting, A.; Knolle, P.; Meyer zum Büschenfelde, K.H.; Mayet, W. Effects of Th1 and Th2 Cytokines on Cytokine Production and ICAM-1 Expression on Synovial Fibroblasts. Ann. Rheum. Dis. 1995, 54, 560–565. [Google Scholar] [CrossRef] [PubMed]
- Bondeson, J.; Wainwright, S.D.; Lauder, S.; Amos, N.; Hughes, C.E. The Role of Synovial Macrophages and Macrophage-Produced Cytokines in Driving Aggrecanases, Matrix Metalloproteinases, and Other Destructive and Inflammatory Responses in Osteoarthritis. Arthritis Res. Ther. 2006, 8, R187. [Google Scholar] [CrossRef] [PubMed]
- Baud, V.; Karin, M. Signal Transduction by Tumor Necrosis Factor and Its Relatives. Trends Cell Biol. 2001, 11, 372–377. [Google Scholar] [CrossRef] [PubMed]
- Bodmer, J.-L.; Schneider, P.; Tschopp, J. The Molecular Architecture of the TNF Superfamily. Trends Biochem. Sci. 2002, 27, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Nees, T.A.; Rosshirt, N.; Zhang, J.A.; Reiner, T.; Sorbi, R.; Tripel, E.; Walker, T.; Schiltenwolf, M.; Hagmann, S.; Moradi, B. Synovial Cytokines Significantly Correlate with Osteoarthritis-Related Knee Pain and Disability: Inflammatory Mediators of Potential Clinical Relevance. J. Clin. Med. 2019, 8, 1343. [Google Scholar] [CrossRef]
- Rosshirt, N.; Hagmann, S.; Tripel, E.; Gotterbarm, T.; Kirsch, J.; Zeifang, F.; Lorenz, H.-M.; Tretter, T.; Moradi, B. A Predominant Th1 Polarization Is Present in Synovial Fluid of End-Stage Osteoarthritic Knee Joints: Analysis of Peripheral Blood, Synovial Fluid and Synovial Membrane. Clin. Exp. Immunol. 2019, 195, 395–406. [Google Scholar] [CrossRef]
- Tsuchida, A.I.; Beekhuizen, M.; Rutgers, M.; van Osch, G.J.; Bekkers, J.E.; Bot, A.G.; Geurts, B.; Dhert, W.J.; Saris, D.B.; Creemers, L.B. Interleukin-6 Is Elevated in Synovial Fluid of Patients with Focal Cartilage Defects and Stimulates Cartilage Matrix Production in an in Vitro Regeneration Model. Arthritis Res. Ther. 2012, 14, R262. [Google Scholar] [CrossRef]
- Teunis, T.; Beekhuizen, M.; Kon, M.; Creemers, L.B.; Schuurman, A.H.; van Minnen, L.P. Inflammatory Mediators in Posttraumatic Radiocarpal Osteoarthritis. J. Hand Surg. Am. 2013, 38, 1735–1740. [Google Scholar] [CrossRef]
- Cuellar, V.G.; Cuellar, J.M.; Golish, S.R.; Yeomans, D.C.; Scuderi, G.J. Cytokine Profiling in Acute Anterior Cruciate Ligament Injury. Arthrosc. J. Arthrosc. Relat. Surg. 2010, 26, 1296–1301. [Google Scholar] [CrossRef]
- Larsson, S.; Struglics, A.; Lohmander, L.S.; Frobell, R. Surgical Reconstruction of Ruptured Anterior Cruciate Ligament Prolongs Trauma-Induced Increase of Inflammatory Cytokines in Synovial Fluid: An Exploratory Analysis in the KANON Trial. Osteoarthr. Cartil. 2017, 25, 1443–1451. [Google Scholar] [CrossRef]
- Tang, M.; Tian, L.; Luo, G.; Yu, X. Interferon-Gamma-Mediated Osteoimmunology. Front. Immunol. 2018, 9, 1508. [Google Scholar] [CrossRef]
- Henrotin, Y.E.; Zheng, S.X.; Labasse, A.H.; Deby, G.P.; Crielaard, J.M.; Reginster, J.Y. Modulation of Human Chondrocyte Metabolism by Recombinant Human Interferon. Osteoarthr. Cartil. 2000, 8, 474–482. [Google Scholar] [CrossRef][Green Version]
- Reginato, A.M.; Sanz-Rodriguez, C.; Diaz, A.; Dharmavaram, R.M.; Jimenez, S.A. Transcriptional Modulation of Cartilage-Specific Collagen Gene Expression by Interferon Gamma and Tumour Necrosis Factor Alpha in Cultured Human Chondrocytes. Biochem. J. 1993, 294(Pt. 3), 761–769. [Google Scholar] [CrossRef]
- Shan, Y.; Qi, C.; Liu, Y.; Gao, H.; Zhao, D.; Jiang, Y. Increased Frequency of Peripheral Blood Follicular Helper T Cells and Elevated Serum IL-21 Levels in Patients with Knee Osteoarthritis. Mol. Med. Rep. 2017, 15, 1095–1102. [Google Scholar] [CrossRef]
- De Waal Malefyt, R.; Abrams, J.; Bennett, B.; Figdor, C.G.; de Vries, J.E. Interleukin 10(IL-10) Inhibits Cytokine Synthesis by Human Monocytes: An Autoregulatory Role of IL-10 Produced by Monocytes. J. Exp. Med. 1991, 174, 1209–1220. [Google Scholar] [CrossRef]
- D’Andrea, A.; Aste-Amezaga, M.; Valiante, N.M.; Ma, X.; Kubin, M.; Trinchieri, G. Interleukin 10 (IL-10) Inhibits Human Lymphocyte Interferon Gamma-Production by Suppressing Natural Killer Cell Stimulatory Factor/IL-12 Synthesis in Accessory Cells. J. Exp. Med. 1993, 178, 1041–1048. [Google Scholar] [CrossRef]
- Fiorentino, D.F.; Zlotnik, A.; Mosmann, T.R.; Howard, M.; O’Garra, A. Pillars Article: IL-10 Inhibits Cytokine Production by Activated Macrophages. J. Immunol. 1991. 147: 3815-3822. J. Immunol. 2016, 197, 1539–1546. [Google Scholar]
- Scanzello, C.R.; Umoh, E.; Pessler, F.; Diaz-Torne, C.; Miles, T.; Dicarlo, E.; Potter, H.G.; Mandl, L.; Marx, R.; Rodeo, S.; et al. Local Cytokine Profiles in Knee Osteoarthritis: Elevated Synovial Fluid Interleukin-15 Differentiates Early from End-Stage Disease. Osteoarthr. Cartil. 2009, 17, 1040–1048. [Google Scholar] [CrossRef]
- Iannone, F.; De Bari, C.; Dell’Accio, F.; Covelli, M.; Cantatore, F.P.; Patella, V.; Lo Bianco, G.; Lapadula, G. Interleukin-10 and Interleukin-10 Receptor in Human Osteoarthritic and Healthy Chondrocytes. Clin. Exp. Rheumatol. 2001, 19, 139–145. [Google Scholar]
- Wang, Y.; Lou, S. Direct Protective Effect of Interleukin-10 on Articular Chondrocytes in Vitro. Chin. Med. J. 2001, 114, 723–725. [Google Scholar]
- Pearson, M.J.; Herndler-Brandstetter, D.; Tariq, M.A.; Nicholson, T.A.; Philp, A.M.; Smith, H.L.; Davis, E.T.; Jones, S.W.; Lord, J.M. IL-6 Secretion in Osteoarthritis Patients Is Mediated by Chondrocyte-Synovial Fibroblast Cross-Talk and Is Enhanced by Obesity. Sci. Rep. 2017, 7, 3451. [Google Scholar] [CrossRef] [PubMed]
- Deligne, C.; Casulli, S.; Pigenet, A.; Bougault, C.; Campillo-Gimenez, L.; Nourissat, G.; Berenbaum, F.; Elbim, C.; Houard, X. Differential Expression of Interleukin-17 and Interleukin-22 in Inflamed and Non-Inflamed Synovium from Osteoarthritis Patients. Osteoarthr. Cartil. 2015, 23, 1843–1852. [Google Scholar] [CrossRef] [PubMed]
- Rai, M.F.; Sandell, L.J. Inflammatory Mediators: Tracing Links between Obesity and Osteoarthritis. Crit. Rev. Eukaryot. Gene Expr. 2011, 21, 131–142. [Google Scholar] [CrossRef] [PubMed]
- Issa, R.I.; Griffin, T.M. Pathobiology of Obesity and Osteoarthritis: Integrating Biomechanics and Inflammation. Pathobiol. Aging Age Relat. Dis. 2012, 2, 17470. [Google Scholar] [CrossRef] [PubMed]
- Beekhuizen, M.; Gierman, L.M.; van Spil, W.E.; Van Osch, G.J.V.M.; Huizinga, T.W.J.; Saris, D.B.F.; Creemers, L.B.; Zuurmond, A.-M. An Explorative Study Comparing Levels of Soluble Mediators in Control and Osteoarthritic Synovial Fluid. Osteoarthr. Cartil. 2013, 21, 918–922. [Google Scholar] [CrossRef]
- Stannus, O.; Jones, G.; Cicuttini, F.; Parameswaran, V.; Quinn, S.; Burgess, J.; Ding, C. Circulating Levels of IL-6 and TNF-α Are Associated with Knee Radiographic Osteoarthritis and Knee Cartilage Loss in Older Adults. Osteoarthr. Cartil. 2010, 18, 1441–1447. [Google Scholar] [CrossRef]
- Goekoop, R.J.; Kloppenburg, M.; Kroon, H.M.; Frölich, M.; Huizinga, T.W.J.; Westendorp, R.G.J.; Gussekloo, J. Low Innate Production of Interleukin-1beta and Interleukin-6 Is Associated with the Absence of Osteoarthritis in Old Age. Osteoarthr. Cartil. 2010, 18, 942–947. [Google Scholar] [CrossRef]
- Eymard, F.; Pigenet, A.; Citadelle, D.; Flouzat-Lachaniette, C.-H.; Poignard, A.; Benelli, C.; Berenbaum, F.; Chevalier, X.; Houard, X. Induction of an Inflammatory and Prodegradative Phenotype in Autologous Fibroblast-like Synoviocytes by the Infrapatellar Fat Pad from Patients with Knee Osteoarthritis. Arthritis Rheumatol. 2014, 66, 2165–2174. [Google Scholar] [CrossRef]
- Sun, J.-M.; Sun, L.-Z.; Liu, J.; Su, B.; Shi, L. Serum Interleukin-15 Levels Are Associated with Severity of Pain in Patients with Knee Osteoarthritis. Dis. Markers 2013, 35, 203–206. [Google Scholar] [CrossRef]
- Tao, Y.; Qiu, X.; Xu, C.; Sun, B.; Shi, C. Expression and Correlation of Matrix Metalloproteinase-7 and Interleukin-15 in Human Osteoarthritis. Int. J. Clin. Exp. Pathol. 2015, 8, 9112–9118. [Google Scholar]
- Constantinescu, C.S.; Grygar, C.; Kappos, L.; Leppert, D. Interleukin 15 Stimulates Production of Matrix Metalloproteinase-9 and Tissue Inhibitor of Metalloproteinase-1 by Human Peripheral Blood Mononuclear Cells. Cytokine 2001, 13, 244–247. [Google Scholar] [CrossRef]
- Warner, S.C.; Nair, A.; Marpadga, R.; Chubinskaya, S.; Doherty, M.; Valdes, A.M.; Scanzello, C.R. IL-15 and IL15RA in Osteoarthritis: Association With Symptoms and Protease Production, but Not Structural Severity. Front. Immunol. 2020, 11, 1385. [Google Scholar] [CrossRef]
- Kleine, S.A.; Gogal, R.M.; George, C.; Thaliath, M.; Budsberg, S.C. Elevated Synovial Fluid Concentration of Monocyte Chemoattractant Protein-1 and Interleukin-8 in Dogs with Osteoarthritis of the Stifle. Vet. Comp. Orthop. Traumatol. 2020, 33, 147–150. [Google Scholar] [CrossRef]
- Bilgic, H.; Ytterberg, S.R.; Amin, S.; McNallan, K.T.; Wilson, J.C.; Koeuth, T.; Ellingson, S.; Newman, B.; Bauer, J.W.; Peterson, E.J.; et al. Interleukin-6 and Type I Interferon-Regulated Genes and Chemokines Mark Disease Activity in Dermatomyositis. Arthritis Rheum. 2009, 60, 3436–3446. [Google Scholar] [CrossRef]
- Raghu, H.; Lepus, C.M.; Wang, Q.; Wong, H.H.; Lingampalli, N.; Oliviero, F.; Punzi, L.; Giori, N.J.; Goodman, S.B.; Chu, C.R.; et al. CCL2/CCR2, but Not CCL5/CCR5, Mediates Monocyte Recruitment, Inflammation and Cartilage Destruction in Osteoarthritis. Ann. Rheum. Dis. 2017, 76, 914–922. [Google Scholar] [CrossRef]
- Dutta, P.; Sarkissyan, M.; Paico, K.; Wu, Y.; Vadgama, J.V. MCP-1 Is Overexpressed in Triple-Negative Breast Cancers and Drives Cancer Invasiveness and Metastasis. Breast Cancer Res. Treat. 2018, 170, 477–486. [Google Scholar] [CrossRef]
- Salah, S.M.; Meisenheimer, J.D.; Rao, R.; Peda, J.D.; Wallace, D.P.; Foster, D.; Li, X.; Li, X.; Zhou, X.; Vallejo, J.A.; et al. MCP-1 Promotes Detrimental Cardiac Physiology, Pulmonary Edema, and Death in the Cpk Model of Polycystic Kidney Disease. Am. J. Physiol. Renal Physiol. 2019, 317, F343–F360. [Google Scholar] [CrossRef]
- Liu, S.; Li, N.; Zhu, Q.; Zhu, B.; Wu, T.; Wang, G.; Liu, S.; Luo, Q. Increased Serum MCP-1 Levels in Systemic Vasculitis Patients with Renal Involvement. J. Interferon Cytokine Res. 2018, 38, 406–412. [Google Scholar] [CrossRef]
- Lee, W.-J.; Liao, Y.-C.; Wang, Y.-F.; Lin, I.-F.; Wang, S.-J.; Fuh, J.-L. Plasma MCP-1 and Cognitive Decline in Patients with Alzheimer’s Disease and Mild Cognitive Impairment: A Two-Year Follow-up Study. Sci. Rep. 2018, 8, 1280. [Google Scholar] [CrossRef]
- Hoff, P.; Buttgereit, F.; Burmester, G.-R.; Jakstadt, M.; Gaber, T.; Andreas, K.; Matziolis, G.; Perka, C.; Röhner, E. Osteoarthritis Synovial Fluid Activates Pro-Inflammatory Cytokines in Primary Human Chondrocytes. Int. Orthop. 2013, 37, 145–151. [Google Scholar] [CrossRef]
- Hampel, U.; Sesselmann, S.; Iserovich, P.; Sel, S.; Paulsen, F.; Sack, R. Chemokine and Cytokine Levels in Osteoarthritis and Rheumatoid Arthritis Synovial Fluid. J. Immunol. Methods 2013, 396, 134–139. [Google Scholar] [CrossRef] [PubMed]
- Morrison, N.A.; Day, C.J.; Nicholson, G.C. Dominant Negative MCP-1 Blocks Human Osteoclast Differentiation. J. Cell. Biochem. 2014, 115, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Villiger, P.M.; Terkeltaub, R.; Lotz, M. Monocyte Chemoattractant Protein-1 (MCP-1) Expression in Human Articular Cartilage. Induction by Peptide Regulatory Factors and Differential Effects of Dexamethasone and Retinoic Acid. J. Clin. Investig. 1992, 90, 488–496. [Google Scholar] [CrossRef] [PubMed]
- Chun, J.L.; Bang, H.T.; Ji, S.Y.; Jeong, J.Y.; Kim, M.; Kim, B.; Lee, S.D.; Lee, Y.K.; Reddy, K.E.; Kim, K.H. A Simple Method to Evaluate Body Condition Score to Maintain the Optimal Body Weight in Dogs. J. Anim. Sci. Technol. 2019, 61, 366–370. [Google Scholar] [CrossRef] [PubMed]
- Bruecker, K.A.; Benjamino, K.; Vezzoni, A.; Walls, C.; Wendelburg, K.L.; Follette, C.M.; Déjardin, L.M.; Guillou, R. Canine Elbow Dysplasia. Vet. Clin. N. Am. Small Anim. Pract. 2021, 51, 475–515. [Google Scholar] [CrossRef]
- Hebel, M.; Panek, W.K.; Ruszkowski, J.J.; Nabzdyk, M.; Niedzielski, D.; Pituch, K.C.; Jackson, A.M.; Kiełbowicz, M.; Pomorska-Mól, M. Computed Tomography Findings in a Cohort of 169 Dogs with Elbow Dysplasia—A Retrospective Study. BMC Vet. Res. 2021, 17, 296. [Google Scholar] [CrossRef]
- Deweese, M.; Brown, D.; Hayashi, K.; Blake, C.; Anglin, E.; Morris, K.; Agnello, K. Observer Variability of Arthroscopic Cartilage Grading Using the Modified Outerbridge Classification System in the Dog. Vet. Comp. Orthop. Traumatol. 2019, 32, 126–132. [Google Scholar] [CrossRef]
- Vezzoni, A.; Benjamino, K. Canine Elbow Dysplasia. Vet. Clin. N. Am. Small Anim. Pract. 2021, 51, 439–474. [Google Scholar] [CrossRef]
- Michelsen, J. Canine Elbow Dysplasia: Aetiopathogenesis and Current Treatment Recommendations. Vet. J. 2013, 196, 12–19. [Google Scholar] [CrossRef]
- Baers, G.; Keller, G.G.; Famula, T.R.; Oberbauer, A.M. Heritability of Unilateral Elbow Dysplasia in the Dog: A Retrospective Study of Sire and Dam Influence. Front. Vet. Sci. 2019, 6, 422. [Google Scholar] [CrossRef]
- Mammoto, T.; Ingber, D.E. Mechanical Control of Tissue and Organ Development. Development 2010, 137, 1407–1420. [Google Scholar] [CrossRef]
- Hao, J.; Zhang, Y.; Wang, Y.; Ye, R.; Qiu, J.; Zhao, Z.; Li, J. Role of Extracellular Matrix and YAP/TAZ in Cell Fate Determination. Cell. Signal. 2014, 26, 186–191. [Google Scholar] [CrossRef]
- Man, G.S.; Mologhianu, G. Osteoarthritis Pathogenesis—A Complex Process That Involves the Entire Joint. J. Med. Life 2014, 7, 37–41. [Google Scholar]
- Glyn-Jones, S.; Palmer, A.J.R.; Agricola, R.; Price, A.J.; Vincent, T.L.; Weinans, H.; Carr, A.J. Osteoarthritis. Lancet 2015, 386, 376–387. [Google Scholar] [CrossRef]
- Sulzbacher, I. Osteoarthritis: Histology and Pathogenesis. Wien. Med. Wochenschr. 2013, 163, 212–219. [Google Scholar] [CrossRef]
- Djouad, F.; Charbonnier, L.-M.; Bouffi, C.; Louis-Plence, P.; Bony, C.; Apparailly, F.; Cantos, C.; Jorgensen, C.; Noël, D. Mesenchymal Stem Cells Inhibit the Differentiation of Dendritic Cells Through an Interleukin-6-Dependent Mechanism. Stem Cells 2007, 25, 2025–2032. [Google Scholar] [CrossRef]
- Maggini, J.; Mirkin, G.; Bognanni, I.; Holmberg, J.; Piazzón, I.M.; Nepomnaschy, I.; Costa, H.; Cañones, C.; Raiden, S.; Vermeulen, M.; et al. Mouse Bone Marrow-Derived Mesenchymal Stromal Cells Turn Activated Macrophages into a Regulatory-Like Profile. PLoS ONE 2010, 5, e9252. [Google Scholar] [CrossRef]
- Van Buul, G.M.; Villafuertes, E.; Bos, P.K.; Waarsing, J.H.; Kops, N.; Narcisi, R.; Weinans, H.; Verhaar, J.A.N.; Bernsen, M.R.; van Osch, G.J.V.M. Mesenchymal Stem Cells Secrete Factors That Inhibit Inflammatory Processes in Short-Term Osteoarthritic Synovium and Cartilage Explant Culture. Osteoarthr. Cartil. 2012, 20, 1186–1196. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, X.; Cao, W.; Shi, Y. Plasticity of Mesenchymal Stem Cells in Immunomodulation: Pathological and Therapeutic Implications. Nat. Immunol. 2014, 15, 1009–1016. [Google Scholar] [CrossRef]
- YlÖstalo, J.H.; Bartosh, T.J.; Coble, K.; Prockop, D.J. Human Mesenchymal Stem/Stromal Cells Cultured as Spheroids Are Self-activated to Produce Prostaglandin E2 That Directs Stimulated Macrophages into an Anti-inflammatory Phenotype. Stem Cells 2012, 30, 2283–2296. [Google Scholar] [CrossRef]
- Pountos, I.; Giannoudis, P.V.; Jones, E.; English, A.; Churchman, S.; Field, S.; Ponchel, F.; Bird, H.; Emery, P.; McGonagle, D. NSAIDS Inhibit in Vitro MSC Chondrogenesis but Not Osteogenesis: Implications for Mechanism of Bone Formation Inhibition in Man. J. Cell. Mol. Med. 2011, 15, 525–534. [Google Scholar] [CrossRef] [PubMed]
- Sok, D.; Raval, S.; McKinney, J.; Drissi, H.; Mason, A.; Mautner, K.; Kaiser, J.M.; Willett, N.J. NSAIDs Reduce Therapeutic Efficacy of Mesenchymal Stromal Cell Therapy in a Rodent Model of Posttraumatic Osteoarthritis. Am. J. Sport. Med. 2022, 50, 1389–1398. [Google Scholar] [CrossRef] [PubMed]
- Almaawi, A.; Wang, H.T.; Ciobanu, O.; Rowas, S.A.L.; Rampersad, S.; Antoniou, J.; Mwale, F. Effect of Acetaminophen and Nonsteroidal Anti-Inflammatory Drugs on Gene Expression of Mesenchymal Stem Cells. Tissue Eng. Part A 2013, 19, 1039–1046. [Google Scholar] [CrossRef] [PubMed]
- Salem, O.; Wang, H.T.; Alaseem, A.M.; Ciobanu, O.; Hadjab, I.; Gawri, R.; Antoniou, J.; Mwale, F. Naproxen Affects Osteogenesis of Human Mesenchymal Stem Cells via Regulation of Indian Hedgehog Signaling Molecules. Arthritis Res. Ther. 2014, 16, R152. [Google Scholar] [CrossRef]
- Belinsky, G.S.; Antic, S.D. Mild Hypothermia Inhibits Differentiation of Human Embryonic and Induced Pluripotent Stem Cells. Biotechniques 2013, 55, 79–82. [Google Scholar] [CrossRef]
- Heng, B.C.; Cowan, C.M.; Basu, S. Temperature and Calcium Ions Affect Aggregation of Mesenchymal Stem Cells in Phosphate Buffered Saline. Cytotechnology 2008, 58, 69–75. [Google Scholar] [CrossRef]
- Rasheed, Z.; Akhtar, N.; Haqqi, T.M. Advanced Glycation End Products Induce the Expression of Interleukin-6 and Interleukin-8 by Receptor for Advanced Glycation End Product-Mediated Activation of Mitogen-Activated Protein Kinases and Nuclear Factor- B in Human Osteoarthritis Chondrocytes. Rheumatology 2011, 50, 838–851. [Google Scholar] [CrossRef]
- DeGroot, J.; Verzijl, N.; Wenting-Van Wijk, M.J.G.; Jacobs, K.M.G.; Van El, B.; Van Roermund, P.M.; Bank, R.A.; Bijlsma, J.W.J.; TeKoppele, J.M.; Lafeber, F.P.J.G. Accumulation of Advanced Glycation End Products as a Molecular Mechanism for Aging as a Risk Factor in Osteoarthritis. Arthritis Rheum. 2004, 50, 1207–1215. [Google Scholar] [CrossRef]
- Johnston, S.A.; McLaughlin, R.M.; Budsberg, S.C. Nonsurgical Management of Osteoarthritis in Dogs. Vet. Clin. N. Am. Small Anim. Pract. 2008, 38, 1449–1470. [Google Scholar] [CrossRef]
- Brondeel, C.; Pauwelyn, G.; de Bakker, E.; Saunders, J.; Samoy, Y.; Spaas, J.H. Review: Mesenchymal Stem Cell Therapy in Canine Osteoarthritis Research: “Experientia Docet” (Experience Will Teach Us). Front. Vet. Sci. 2021, 8, 668881. [Google Scholar] [CrossRef]
- Sarsenova, M.; Issabekova, A.; Abisheva, S.; Rutskaya-Moroshan, K.; Ogay, V.; Saparov, A. Mesenchymal Stem Cell-Based Therapy for Rheumatoid Arthritis. Int. J. Mol. Sci. 2021, 22, 11592. [Google Scholar] [CrossRef]
- Kearney, C.M.; Khatab, S.; van Buul, G.M.; Plomp, S.G.M.; Korthagen, N.M.; Labberté, M.C.; Goodrich, L.R.; Kisiday, J.D.; Van Weeren, P.R.; van Osch, G.J.V.M.; et al. Treatment Effects of Intra-Articular Allogenic Mesenchymal Stem Cell Secretome in an Equine Model of Joint Inflammation. Front. Vet. Sci. 2022, 9, 907616. [Google Scholar] [CrossRef]
- Ichiseki, T.; Shimasaki, M.; Ueda, Y.; Ueda, S.; Tsuchiya, M.; Souma, D.; Kaneuji, A.; Kawahara, N. Intraarticularly-Injected Mesenchymal Stem Cells Stimulate Anti-Inflammatory Molecules and Inhibit Pain Related Protein and Chondrolytic Enzymes in a Monoiodoacetate-Induced Rat Arthritis Model. Int. J. Mol. Sci. 2018, 19, 203. [Google Scholar] [CrossRef]
- Hached, F.; Vinatier, C.; Le Visage, C.; Gondé, H.; Guicheux, J.; Grimandi, G.; Billon-Chabaud, A. Biomaterial-Assisted Cell Therapy in Osteoarthritis: From Mesenchymal Stem Cells to Cell Encapsulation. Best Pract. Res. Clin. Rheumatol. 2017, 31, 730–745. [Google Scholar] [CrossRef]
- Slattery, C.; Kweon, C.Y. Classifications in Brief: Outerbridge Classification of Chondral Lesions. Clin. Orthop. Relat. Res. 2018, 476, 2101–2104. [Google Scholar] [CrossRef]
- Savkovic, V.; Li, H.; Seon, J.-K.; Hacker, M.; Franz, S.; Simon, J.-C. Mesenchymal Stem Cells in Cartilage Regeneration. CSCR 2014, 9, 469–488. [Google Scholar] [CrossRef]
- Gupta, P.K.; Das, A.K.; Chullikana, A.; Majumdar, A.S. Mesenchymal Stem Cells for Cartilage Repair in Osteoarthritis. Stem Cell Res. Ther. 2012, 3, 25. [Google Scholar] [CrossRef]
- Kon, E.; Filardo, G.; Roffi, A.; Andriolo, L.; Marcacci, M. New Trends for Knee Cartilage Regeneration: From Cell-Free Scaffolds to Mesenchymal Stem Cells. Curr. Rev. Musculoskelet. Med. 2012, 5, 236–243. [Google Scholar] [CrossRef]
- Yun, S.; Ku, S.-K.; Kwon, Y.-S. Adipose-Derived Mesenchymal Stem Cells and Platelet-Rich Plasma Synergistically Ameliorate the Surgical-Induced Osteoarthritis in Beagle Dogs. J. Orthop. Surg. Res. 2016, 11, 9. [Google Scholar] [CrossRef]
- Qiang, Y.; Yanhong, Z.; Jiang, P.; Shibi, L.; Quanyi, G.; Xinlong, M.; Qun, X.; Baoshan, X.; Bin, Z.; Aiyuan, W.; et al. Xenoimplantation of an Extracellular-Matrix-Derived, Biphasic, Cell-Scaffold Construct for Repairing a Large Femoral-Head High-Load-Bearing Osteochondral Defect in a Canine Model. Sci. World J. 2014, 2014, 127084. [Google Scholar] [CrossRef]
- Li, L.; Duan, X.; Fan, Z.; Chen, L.; Xing, F.; Xu, Z.; Chen, Q.; Xiang, Z. Mesenchymal Stem Cells in Combination with Hyaluronic Acid for Articular Cartilage Defects. Sci. Rep. 2018, 8, 9900. [Google Scholar] [CrossRef]
- Black, L.L.; Gaynor, J.; Gahring, D.; Adams, C.; Aron, D.; Harman, S.; Gingerich, D.A.; Harman, R. Effect of Adipose-Derived Mesenchymal Stem and Regenerative Cells on Lameness in Dogs with Chronic Osteoarthritis of the Coxofemoral Joints: A Randomized, Double-Blinded, Multicenter, Controlled Trial. Vet. Ther. 2007, 8, 272–284. [Google Scholar] [PubMed]
- Emadedin, M.; Aghdami, N.; Taghiyar, L.; Fazeli, R.; Moghadasali, R.; Jahangir, S.; Farjad, R.; Baghaban Eslaminejad, M. Intra-Articular Injection of Autologous Mesenchymal Stem Cells in Six Patients with Knee Osteoarthritis. Arch. Iran. Med. 2012, 15, 422–428. [Google Scholar]
- Vangsness, C.T.; Farr, J.; Boyd, J.; Dellaero, D.T.; Mills, C.R.; LeRoux-Williams, M. Adult Human Mesenchymal Stem Cells Delivered via Intra-Articular Injection to the Knee Following Partial Medial Meniscectomy: A Randomized, Double-Blind, Controlled Study. J. Bone Joint Surg. Am. 2014, 96, 90–98. [Google Scholar] [CrossRef] [PubMed]
- Centeno, C.J.; Schultz, J.R.; Cheever, M.; Freeman, M.; Faulkner, S.; Robinson, B.; Hanson, R. Safety and Complications Reporting Update on the Re-Implantation of Culture-Expanded Mesenchymal Stem Cells Using Autologous Platelet Lysate Technique. Curr. Stem Cell. Res. Ther. 2011, 6, 368–378. [Google Scholar] [CrossRef] [PubMed]
- Srzentić Dražilov, S.; Mrkovački, J.; Spasovski, V.; Fazlagić, A.; Pavlović, S.; Nikčević, G. The Use of Canine Mesenchymal Stem Cells for the Autologous Treatment of Osteoarthritis. Acta Vet. Hung. 2018, 66, 376–389. [Google Scholar] [CrossRef]
- Vilar, J.M.; Batista, M.; Morales, M.; Santana, A.; Cuervo, B.; Rubio, M.; Cugat, R.; Sopena, J.; Carrillo, J.M. Assessment of the Effect of Intraarticular Injection of Autologous Adipose-Derived Mesenchymal Stem Cells in Osteoarthritic Dogs Using a Double Blinded Force Platform Analysis. BMC Vet. Res. 2014, 10, 143. [Google Scholar] [CrossRef]
- Vilar, J.M.; Cuervo, B.; Rubio, M.; Sopena, J.; Domínguez, J.M.; Santana, A.; Carrillo, J.M. Effect of Intraarticular Inoculation of Mesenchymal Stem Cells in Dogs with Hip Osteoarthritis by Means of Objective Force Platform Gait Analysis: Concordance with Numeric Subjective Scoring Scales. BMC Vet. Res. 2016, 12, 223. [Google Scholar] [CrossRef]
- Vilar, J.M.; Morales, M.; Santana, A.; Spinella, G.; Rubio, M.; Cuervo, B.; Cugat, R.; Carrillo, J.M. Controlled, Blinded Force Platform Analysis of the Effect of Intraarticular Injection of Autologous Adipose-Derived Mesenchymal Stem Cells Associated to PRGF-Endoret in Osteoarthritic Dogs. BMC Vet. Res. 2013, 9, 131. [Google Scholar] [CrossRef]
- Daems, R.; Van Hecke, L.; Schwarzkopf, I.; Depuydt, E.; Broeckx, S.Y.; David, M.; Beerts, C.; Vandekerckhove, P.; Spaas, J.H. A Feasibility Study on the Use of Equine Chondrogenic Induced Mesenchymal Stem Cells as a Treatment for Natural Occurring Osteoarthritis in Dogs. Stem Cells Int. 2019, 2019, 4587594. [Google Scholar] [CrossRef]
- Cuervo, B.; Rubio, M.; Sopena, J.; Dominguez, J.; Vilar, J.; Morales, M.; Cugat, R.; Carrillo, J. Hip Osteoarthritis in Dogs: A Randomized Study Using Mesenchymal Stem Cells from Adipose Tissue and Plasma Rich in Growth Factors. Int. J. Mol. Sci. 2014, 15, 13437–13460. [Google Scholar] [CrossRef]
- Harman, R.; Carlson, K.; Gaynor, J.; Gustafson, S.; Dhupa, S.; Clement, K.; Hoelzler, M.; McCarthy, T.; Schwartz, P.; Adams, C. A Prospective, Randomized, Masked, and Placebo-Controlled Efficacy Study of Intraarticular Allogeneic Adipose Stem Cells for the Treatment of Osteoarthritis in Dogs. Front. Vet. Sci. 2016, 3, 81. [Google Scholar] [CrossRef]
- Wits, M.I.; Tobin, G.C.; Silveira, M.D.; Baja, K.G.; Braga, L.M.M.; Sesterheim, P.; Camassola, M.; Nardi, N.B. Combining Canine Mesenchymal Stromal Cells and Hyaluronic Acid for Cartilage Repair. Genet. Mol. Biol. 2020, 43, e20190275. [Google Scholar] [CrossRef]
- Kim, S.E.; Pozzi, A.; Yeh, J.; Lopez-Velazquez, M.; Au Yong, J.A.; Townsend, S.; Dunlap, A.E.; Christopher, S.A.; Lewis, D.D.; Johnson, M.D.; et al. Intra-Articular Umbilical Cord Derived Mesenchymal Stem Cell Therapy for Chronic Elbow Osteoarthritis in Dogs: A Double-Blinded, Placebo-Controlled Clinical Trial. Front. Vet. Sci. 2019, 6, 474. [Google Scholar] [CrossRef]
- Wieczorek, M.; Abualrous, E.T.; Sticht, J.; Álvaro-Benito, M.; Stolzenberg, S.; Noé, F.; Freund, C. Major Histocompatibility Complex (MHC) Class I and MHC Class II Proteins: Conformational Plasticity in Antigen Presentation. Front. Immunol. 2017, 8, 292. [Google Scholar] [CrossRef]
- Guercio, A.; Di Bella, S.; Casella, S.; Di Marco, P.; Russo, C.; Piccione, G. Canine Mesenchymal Stem Cells (MSCs): Characterization in Relation to Donor Age and Adipose Tissue-Harvesting Site: Adipose-Derived Mesenchymal Stem Cells in Dog. Cell Biol. Int. 2013, 37, 789–798. [Google Scholar] [CrossRef]
- Evangelista, M.; Soncini, M.; Parolini, O. Placenta-Derived Stem Cells: New Hope for Cell Therapy? Cytotechnology 2008, 58, 33–42. [Google Scholar] [CrossRef]
- Ilancheran, S.; Michalska, A.; Peh, G.; Wallace, E.M.; Pera, M.; Manuelpillai, U. Stem Cells Derived from Human Fetal Membranes Display Multilineage Differentiation Potential. Biol. Reprod. 2007, 77, 577–588. [Google Scholar] [CrossRef]
- Sarugaser, R.; Lickorish, D.; Baksh, D.; Hosseini, M.M.; Davies, J.E. Human Umbilical Cord Perivascular (HUCPV) Cells: A Source of Mesenchymal Progenitors. Stem Cells 2005, 23, 220–229. [Google Scholar] [CrossRef]
- Chahal, J.; Gómez-Aristizábal, A.; Shestopaloff, K.; Bhatt, S.; Chaboureau, A.; Fazio, A.; Chisholm, J.; Weston, A.; Chiovitti, J.; Keating, A.; et al. Bone Marrow Mesenchymal Stromal Cell Treatment in Patients with Osteoarthritis Results in Overall Improvement in Pain and Symptoms and Reduces Synovial Inflammation. Stem Cells Transl. Med. 2019, 8, 746–757. [Google Scholar] [CrossRef]
- Shegos, C.J.; Chaudhry, A.F. A Narrative Review of Mesenchymal Stem Cells Effect on Osteoarthritis. Ann. Joint 2022, 7, 26. [Google Scholar] [CrossRef]
- Agarwal, N.; Mak, C.; Bojanic, C.; To, K.; Khan, W. Meta-Analysis of Adipose Tissue Derived Cell-Based Therapy for the Treatment of Knee Osteoarthritis. Cells 2021, 10, 1365. [Google Scholar] [CrossRef] [PubMed]
- Yan, B.; Lv, S.; Tong, P.; Yan, L.; Chen, Z.; Zhou, L.; Yuan, Q.; Guo, L.; Shan, L. Intra-Articular Injection of Adipose-Derived Stem Cells Ameliorates Pain and Cartilage Anabolism/Catabolism in Osteoarthritis: Preclinical and Clinical Evidences. Front. Pharmacol. 2022, 13, 854025. [Google Scholar] [CrossRef] [PubMed]
- Borghesi, J.; Ferreira Lima, M.; Mario, L.C.; de Almeida da Anunciação, A.R.; Silveira Rabelo, A.C.; Giancoli Kato Cano da Silva, M.; Assunpção Fernandes, F.; Miglino, M.A.; Oliveira Carreira, A.C.; Oliveira Favaron, P. Canine Amniotic Membrane Mesenchymal Stromal/Stem Cells: Isolation, Characterization and Differentiation. Tissue Cell 2019, 58, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Humenik, F.; Maloveska, M.; Hudakova, N.; Petrouskova, P.; Hornakova, L.; Domaniza, M.; Mudronova, D.; Bodnarova, S.; Cizkova, D. A Comparative Study of Canine Mesenchymal Stem Cells Isolated from Different Sources. Animals 2022, 12, 1502. [Google Scholar] [CrossRef]
- Cifù, A.; Domenis, R.; Pozzi-Mucelli, M.; Di Benedetto, P.; Causero, A.; Moretti, M.; Stevanato, M.; Pistis, C.; Parodi, P.C.; Fabris, M.; et al. The Exposure to Osteoarthritic Synovial Fluid Enhances the Immunomodulatory Profile of Adipose Mesenchymal Stem Cell Secretome. Stem Cells Int. 2020, 2020, 4058760. [Google Scholar] [CrossRef]
- Huang, F.; Thokerunga, E.; He, F.; Zhu, X.; Wang, Z.; Tu, J. Research Progress of the Application of Mesenchymal Stem Cells in Chronic Inflammatory Systemic Diseases. Stem Cell. Res. Ther. 2022, 13, 1. [Google Scholar] [CrossRef]
- Tsai, S.-Y. Intra-Articular Transplantation of Porcine Adipose-Derived Stem Cells for the Treatment of Canine Osteoarthritis: A Pilot Study. WJT 2014, 4, 196. [Google Scholar] [CrossRef]
- Taroni, M.; Cabon, Q.; Fèbre, M.; Cachon, T.; Saulnier, N.; Carozzo, C.; Maddens, S.; Labadie, F.; Robert, C.; Viguier, E. Evaluation of the Effect of a Single Intra-Articular Injection of Allogeneic Neonatal Mesenchymal Stromal Cells Compared to Oral Non-Steroidal Anti-Inflammatory Treatment on the Postoperative Musculoskeletal Status and Gait of Dogs over a 6-Month Period after Tibial Plateau Leveling Osteotomy: A Pilot Study. Front. Vet. Sci. 2017, 4, 83. [Google Scholar] [CrossRef]
- Takemitsu, H.; Zhao, D.; Yamamoto, I.; Harada, Y.; Michishita, M.; Arai, T. Comparison of Bone Marrow and Adipose Tissue-Derived Canine Mesenchymal Stem Cells. BMC Vet. Res. 2012, 8, 150. [Google Scholar] [CrossRef]
- Prišlin, M.; Vlahović, D.; Kostešić, P.; Ljolje, I.; Brnić, D.; Turk, N.; Lojkić, I.; Kunić, V.; Karadjole, T.; Krešić, N. An Outstanding Role of Adipose Tissue in Canine Stem Cell Therapy. Animals 2022, 12, 1088. [Google Scholar] [CrossRef]
- Preda, M.B.; Neculachi, C.A.; Fenyo, I.M.; Vacaru, A.-M.; Publik, M.A.; Simionescu, M.; Burlacu, A. Short Lifespan of Syngeneic Transplanted MSC Is a Consequence of in Vivo Apoptosis and Immune Cell Recruitment in Mice. Cell Death Dis. 2021, 12, 566. [Google Scholar] [CrossRef]
- Guest, D.J.; Smith, M.R.W.; Allen, W.R. Monitoring the Fate of Autologous and Allogeneic Mesenchymal Progenitor Cells Injected into the Superficial Digital Flexor Tendon of Horses: Preliminary Study. Equine Vet. J. 2008, 40, 178–181. [Google Scholar] [CrossRef]
- Mammoto, A.; Mammoto, T.; Ingber, D.E. Mechanosensitive Mechanisms in Transcriptional Regulation. J. Cell Sci. 2012, 125, 3061–3073. [Google Scholar] [CrossRef]
- Agostinho, F.S.; Rahal, S.C.; Miqueleto, N.S.M.L.; Verdugo, M.R.; Inamassu, L.R.; El-Warrak, A.O. Kinematic Analysis of Labrador Retrievers and Rottweilers Trotting on a Treadmill. Vet. Comp. Orthop. Traumatol. 2011, 24, 185–191. [Google Scholar] [CrossRef]
- Sandberg, G.S.; Torres, B.T.; Budsberg, S.C. Review of Kinematic Analysis in Dogs. Vet. Surg. 2020, 49, 1088–1098. [Google Scholar] [CrossRef]
- Miqueleto, N.S.M.L.; Rahal, S.C.; Agostinho, F.S.; Siqueira, E.G.M.; Araújo, F.A.P.; El-Warrak, A.O. Kinematic Analysis in Healthy and Hip-Dysplastic German Shepherd Dogs. Vet. J. 2013, 195, 210–215. [Google Scholar] [CrossRef]
- Lorke, M.; Willen, M.; Lucas, K.; Beyerbach, M.; Wefstaedt, P.; Murua Escobar, H.; Nolte, I. Comparative Kinematic Gait Analysis in Young and Old Beagle Dogs. J. Vet. Sci. 2017, 18, 521. [Google Scholar] [CrossRef]
- De Souza, M.C.; Calesso, J.R.; Cenci, B.; Cardoso, M.J.L.; Moura, F.A.; Fagnani, R. Kinematics of Healthy American Pit Bull Terrier Dogs. Vet. Med. 2021, 66, 8–16. [Google Scholar] [CrossRef]
- Hottinger, H.A.; DeCamp, C.E.; Olivier, N.B.; Hauptman, J.G.; Soutas-Little, R.W. Noninvasive Kinematic Analysis of the Walk in Healthy Large-Breed Dogs. Am. J. Vet. Res. 1996, 57, 381–388. [Google Scholar]
- Holler, P.J.; Brazda, V.; Dal-Bianco, B.; Lewy, E.; Mueller, M.C.; Peham, C.; Bockstahler, B.A. Kinematic Motion Analysis of the Joints of the Forelimbs and Hind Limbs of Dogs during Walking Exercise Regimens. AJVR 2010, 71, 734–740. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.; Sharma, A.; Chakraborty, C.; Saibaba, B.; Ahn, M.-E.; Lee, S.-S. Review of Prospects of Biological Fluid Biomarkers in Osteoarthritis. Int. J. Mol. Sci. 2017, 18, 601. [Google Scholar] [CrossRef] [PubMed]
- Lampropoulou-Adamidou, K.; Lelovas, P.; Karadimas, E.V.; Liakou, C.; Triantafillopoulos, I.K.; Dontas, I.; Papaioannou, N.A. Useful Animal Models for the Research of Osteoarthritis. Eur. J. Orthop. Surg. Traumatol. 2014, 24, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Little, C.B.; Zaki, S. What Constitutes an “Animal Model of Osteoarthritis”—The Need for Consensus? Osteoarthr. Cartil. 2012, 20, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Teeple, E.; Jay, G.D.; Elsaid, K.A.; Fleming, B.C. Animal Models of Osteoarthritis: Challenges of Model Selection and Analysis. AAPS J. 2013, 15, 438–446. [Google Scholar] [CrossRef]
- Poole, R.; Blake, S.; Buschmann, M.; Goldring, S.; Laverty, S.; Lockwood, S.; Matyas, J.; McDougall, J.; Pritzker, K.; Rudolphi, K.; et al. Recommendations for the Use of Preclinical Models in the Study and Treatment of Osteoarthritis. Osteoarthr. Cartil. 2010, 18 (Suppl. 3), S10–S16. [Google Scholar] [CrossRef]
- Meehan, R.T.; Regan, E.A.; Hoffman, E.D.; Wolf, M.L.; Gill, M.T.; Crooks, J.L.; Parmar, P.J.; Scheuring, R.A.; Hill, J.C.; Pacheco, K.A.; et al. Synovial Fluid Cytokines, Chemokines and MMP Levels in Osteoarthritis Patients with Knee Pain Display a Profile Similar to Many Rheumatoid Arthritis Patients. J. Clin. Med. 2021, 10, 5027. [Google Scholar] [CrossRef]
- Wicks, I.P.; Roberts, A.W. Targeting GM-CSF in Inflammatory Diseases. Nat. Rev. Rheumatol. 2016, 12, 37–48. [Google Scholar] [CrossRef]
- Louis, C.; Souza-Fonseca-Guimaraes, F.; Yang, Y.; D’Silva, D.; Kratina, T.; Dagley, L.; Hediyeh-Zadeh, S.; Rautela, J.; Masters, S.L.; Davis, M.J.; et al. NK Cell–Derived GM-CSF Potentiates Inflammatory Arthritis and Is Negatively Regulated by CIS. J. Exp. Med. 2020, 217, e20191421. [Google Scholar] [CrossRef]
- Stankovic, A.; Slavic, V.; Stamenkovic, B.; Kamenov, B.; Bojanovic, M.; Mitrovic, D.R. Serum and Synovial Fluid Concentrations of CCL2 (MCP-1) Chemokine in Patients Suffering Rheumatoid Arthritis and Osteoarthritis Reflect Disease Activity. Bratisl. Lek. Listy 2009, 110, 641–646. [Google Scholar]
- Rabaan, A.A.; Al-Ahmed, S.H.; Muhammad, J.; Khan, A.; Sule, A.A.; Tirupathi, R.; Mutair, A.A.; Alhumaid, S.; Al-Omari, A.; Dhawan, M.; et al. Role of Inflammatory Cytokines in COVID-19 Patients: A Review on Molecular Mechanisms, Immune Functions, Immunopathology and Immunomodulatory Drugs to Counter Cytokine Storm. Vaccines 2021, 9, 436. [Google Scholar] [CrossRef]
- Clockaerts, S.; Bastiaansen-Jenniskens, Y.M.; Feijt, C.; De Clerck, L.; Verhaar, J.A.N.; Zuurmond, A.-M.; Stojanovic-Susulic, V.; Somville, J.; Kloppenburg, M.; van Osch, G.J.V.M. Cytokine Production by Infrapatellar Fat Pad Can Be Stimulated by Interleukin 1β and Inhibited by Peroxisome Proliferator Activated Receptor α Agonist. Ann. Rheum. Dis. 2012, 71, 1012–1018. [Google Scholar] [CrossRef]
- Min, S.; Wang, C.; Lu, W.; Xu, Z.; Shi, D.; Chen, D.; Teng, H.; Jiang, Q. Serum Levels of the Bone Turnover Markers Dickkopf-1, Osteoprotegerin, and TNF-α in Knee Osteoarthritis Patients. Clin. Rheumatol. 2017, 36, 2351–2358. [Google Scholar] [CrossRef]
- Catterall, J.B.; Stabler, T.V.; Flannery, C.R.; Kraus, V.B. Changes in Serum and Synovial Fluid Biomarkers after Acute Injury (NCT00332254). Arthritis Res. Ther. 2010, 12, R229. [Google Scholar] [CrossRef]
- Cook, A.D.; Pobjoy, J.; Steidl, S.; Dürr, M.; Braine, E.L.; Turner, A.L.; Lacey, D.C.; Hamilton, J.A. Granulocyte-Macrophage Colony-Stimulating Factor Is a Key Mediator in Experimental Osteoarthritis Pain and Disease Development. Arthritis Res. Ther. 2012, 14, R199. [Google Scholar] [CrossRef]
- Ni, F.; Zhang, Y.; Peng, X.; Li, J. Correlation between Osteoarthritis and Monocyte Chemotactic Protein-1 Expression: A Meta-Analysis. J. Orthop. Surg. Res. 2020, 15, 516. [Google Scholar] [CrossRef]
- Boffa, A.; Merli, G.; Andriolo, L.; Lattermann, C.; Salzmann, G.M.; Filardo, G. Synovial Fluid Biomarkers in Knee Osteoarthritis: A Systematic Review and Quantitative Evaluation Using BIPEDs Criteria. Cartilage 2021, 13, 82S–103S. [Google Scholar] [CrossRef]
- Chisari, E.; Yaghmour, K.M.; Khan, W.S. The Effects of TNF-Alpha Inhibition on Cartilage: A Systematic Review of Preclinical Studies. Osteoarthr. Cartil. 2020, 28, 708–718. [Google Scholar] [CrossRef]
- Manicourt, D.-H.; Poilvache, P.; Egeren, A.V.; Devogelaer, J.-P.; Lenz, M.-E.; Thonar, E.J.-M.A. Synovial Fluid Levels of Tumor Necrosis Factor α and Oncostatin M Correlate with Levels of Markers of the Degradation of Crosslinked Collagen and Cartilage Aggrecan in Rheumatoid Arthritis but Not in Osteoarthritis. Arthritis Rheum. 2000, 43, 281. [Google Scholar] [CrossRef]
- Arafat, S.; Kamel, S. The Effect of Single Versus Multiple Intra-articular Injection of Synovial Fluid Mesenchymal Stem Cells on rat Temporomandibular Joint with Induced Arthritis. Biochemical and Histologic Analysis. Egypt. Dent. J. 2020, 66, 2165–2172. [Google Scholar] [CrossRef]
- Niada, S.; Giannasi, C.; Gomarasca, M.; Stanco, D.; Casati, S.; Brini, A.T. Adipose-Derived Stromal Cell Secretome Reduces TNFα-Induced Hypertrophy and Catabolic Markers in Primary Human Articular Chondrocytes. Stem Cell Res. 2019, 38, 101463. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Zheng, D.; Xu, R.-H. Critical Role of Tumor Necrosis Factor Signaling in Mesenchymal Stem Cell-Based Therapy for Autoimmune and Inflammatory Diseases. Front. Immunol. 2018, 9, 1658. [Google Scholar] [CrossRef] [PubMed]
- Sutton, M.T.; Bonfield, T.L. Stem Cells: Innovations in Clinical Applications. Stem Cells Int. 2014, 2014, 516278. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, S.J.; Blain, E.J.; Mason, D.J. Interferon-Gamma Modulates Articular Chondrocyte and Osteoblast Metabolism through Protein Kinase R-Independent and Dependent Mechanisms. Biochem. Biophys. Rep. 2022, 32, 101323. [Google Scholar] [CrossRef] [PubMed]
- Page, C.E.; Smale, S.; Carty, S.M.; Amos, N.; Lauder, S.N.; Goodfellow, R.M.; Richards, P.J.; Jones, S.A.; Topley, N.; Williams, A.S. Interferon-Gamma Inhibits Interleukin-1beta-Induced Matrix Metalloproteinase Production by Synovial Fibroblasts and Protects Articular Cartilage in Early Arthritis. Arthritis Res. Ther. 2010, 12, R49. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Kwon, J.Y.; Kim, S.-Y.; Jung, K.; Cho, M.-L. Interferon-Gamma Regulates Inflammatory Cell Death by Targeting Necroptosis in Experimental Autoimmune Arthritis. Sci. Rep. 2017, 7, 10133. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Lu, Y.; Mao, Y.; Yu, Y.; Wu, T.; Zhao, W.; Zhu, Y.; Zhao, P.; Zhang, F. IFN-γ Enhances the Efficacy of Mesenchymal Stromal Cell-Derived Exosomes via MiR-21 in Myocardial Infarction Rats. Stem Cell Res. Ther. 2022, 13, 333. [Google Scholar] [CrossRef]
- Vuolteenaho, K.; Koskinen-Kolasa, A.; Laavola, M.; Nieminen, R.; Moilanen, T.; Moilanen, E. High Synovial Fluid Interleukin-6 Levels Are Associated with Increased Matrix Metalloproteinase Levels and Severe Radiographic Changes in Osteoarthritis Patients. Osteoarthr. Cartil. 2017, 25, S92–S93. [Google Scholar] [CrossRef]
- Mihailova, A. Interleukin 6 Concentration in Synovial Fluid of Patients with Inflammatoryand Degenerative Arthritis. CRR 2022, 18, 230–233. [Google Scholar] [CrossRef]
- Leijs, M.J.C.; van Buul, G.M.; Lubberts, E.; Bos, P.K.; Verhaar, J.A.N.; Hoogduijn, M.J.; van Osch, G.J.V.M. Effect of Arthritic Synovial Fluids on the Expression of Immunomodulatory Factors by Mesenchymal Stem Cells: An Explorative in Vitro Study. Front. Immun. 2012, 3, 231. [Google Scholar] [CrossRef]
- Kay, A.G.; Long, G.; Tyler, G.; Stefan, A.; Broadfoot, S.J.; Piccinini, A.M.; Middleton, J.; Kehoe, O. Mesenchymal Stem Cell-Conditioned Medium Reduces Disease Severity and Immune Responses in Inflammatory Arthritis. Sci. Rep. 2017, 7, 18019. [Google Scholar] [CrossRef]
- Schulze-Tanzil, G.; Zreiqat, H.; Sabat, R.; Kohl, B.; Halder, A.; Muller, R.; John, T. Interleukin-10 and Articular Cartilage: Experimental Therapeutical Approaches in Cartilage Disorders. CGT 2009, 9, 306–315. [Google Scholar] [CrossRef]
- Choi, J.-J.; Yoo, S.-A.; Park, S.-J.; Kang, Y.-J.; Kim, W.-U.; Oh, I.-H.; Cho, C.-S. Mesenchymal Stem Cells Overexpressing Interleukin-10 Attenuate Collagen-Induced Arthritis in Mice. Clin. Exp. Immunol. 2008, 153, 269–276. [Google Scholar] [CrossRef]
- Asadi, S.; Farzanegi, P.; Azarbayjani, M.A. Effect of Exercise, Ozone and Mesenchymal Stem Cells Therapies on Expression of IL-10 and TNF-α in the Cartilage Tissue of Overweight Rats with Knee Osteoarthritis. Soc. Determ. Health 2018, 4, 162–170. [Google Scholar] [CrossRef]
- Muir, P.; Hans, E.C.; Racette, M.; Volstad, N.; Sample, S.J.; Heaton, C.; Holzman, G.; Schaefer, S.L.; Bloom, D.D.; Bleedorn, J.A.; et al. Autologous Bone Marrow-Derived Mesenchymal Stem Cells Modulate Molecular Markers of Inflammation in Dogs with Cruciate Ligament Rupture. PLoS ONE 2016, 11, e0159095. [Google Scholar] [CrossRef]
- Harigai, M.; Hara, M.; Yoshimura, T.; Leonard, E.J.; Inoue, K.; Kashiwazaki, S. Monocyte Chemoattractant Protein-1 (MCP-1) in Inflammatory Joint Diseases and Its Involvement in the Cytokine Network of Rheumatoid Synovium. Clin. Immunol. Immunopathol. 1993, 69, 83–91. [Google Scholar] [CrossRef]
- Bjerre, M.; Hansen, T.K.; Flyvbjerg, A.; Tønnesen, E. Simultaneous Detection of Porcine Cytokines by Multiplex Analysis: Development of Magnetic Bioplex Assay. Vet. Immunol. Immunopathol. 2009, 130, 53–58. [Google Scholar] [CrossRef]
- Johannisson, A.; Jonasson, R.; Dernfalk, J.; Jensen-Waern, M. Simultaneous Detection of Porcine Proinflammatory Cytokines Using Multiplex Flow Cytometry by the XMAPTM Technology. Cytometry 2006, 69A, 391–395. [Google Scholar] [CrossRef]
- Hall, S.A.; Ison, S.H.; Owles, C.; Coe, J.; Sandercock, D.A.; Zanella, A.J. Development and Validation of a Multiplex Fluorescent Microsphere Immunoassay Assay for Detection of Porcine Cytokines. MethodsX 2019, 6, 1218–1227. [Google Scholar] [CrossRef]
- Fan, X.-L.; Zhang, Y.; Li, X.; Fu, Q.-L. Mechanisms Underlying the Protective Effects of Mesenchymal Stem Cell-Based Therapy. Cell. Mol. Life Sci. 2020, 77, 2771–2794. [Google Scholar] [CrossRef]
- Wang, Q.; Lepus, C.M.; Raghu, H.; Reber, L.L.; Tsai, M.M.; Wong, H.H.; von Kaeppler, E.; Lingampalli, N.; Bloom, M.S.; Hu, N.; et al. IgE-Mediated Mast Cell Activation Promotes Inflammation and Cartilage Destruction in Osteoarthritis. eLife 2019, 8, e39905. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, T.L.; Gomoll, A.H.; Lattermann, C.; Hernandez, A.J.; Bueno, D.F.; Amano, M.T. Macrophage: A Potential Target on Cartilage Regeneration. Front. Immunol. 2020, 11, 111. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Xu, J. Immune Modulation by Mesenchymal Stem Cells. Cell Prolif. 2020, 53, e12712. [Google Scholar] [CrossRef] [PubMed]
- Saldaña, L.; Bensiamar, F.; Vallés, G.; Mancebo, F.J.; García-Rey, E.; Vilaboa, N. Immunoregulatory Potential of Mesenchymal Stem Cells Following Activation by Macrophage-Derived Soluble Factors. Stem Cell Res. Ther. 2019, 10, 58. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Shen, S.; Fu, H.; Wang, Z.; Li, X.; Sui, X.; Yuan, M.; Liu, S.; Wang, G.; Guo, Q. Immunomodulatory Functions of Mesenchymal Stem Cells in Tissue Engineering. Stem Cells Int. 2019, 2019, 9671206. [Google Scholar] [CrossRef]
- Hamdalla, H.M.; Ahmed, R.R.; Galaly, S.R.; Ahmed, O.M.; Naguib, I.A.; Alghamdi, B.S.; Abdul-Hamid, M. Assessment of the Efficacy of Bone Marrow-Derived Mesenchymal Stem Cells against a Monoiodoacetate-Induced Osteoarthritis Model in Wistar Rats. Stem Cells Int. 2022, 2022, 1900403. [Google Scholar] [CrossRef]
- Babu, G.S.; Badrish, Y.; Oswal, V.M.; Jeyaraman, N.; Prajwal, G.S.; Jeyaraman, M.; Muthu, S.; Khanna, M. Immunomodulatory Actions of Mesenchymal Stromal Cells (MSCs) in Osteoarthritis of the Knee. Osteology 2021, 1, 209–224. [Google Scholar] [CrossRef]
- Kong, L.; Zheng, L.-Z.; Qin, L.; Ho, K.K.W. Role of Mesenchymal Stem Cells in Osteoarthritis Treatment. J. Orthop. Transl. 2017, 9, 89–103. [Google Scholar] [CrossRef]
- Ossendorff, R.; Walter, S.; Schildberg, F.; Khoury, M.; Salzmann, G. Controversies in Regenerative Medicine: Should Knee Joint Osteoarthritis Be Treated with Mesenchymal Stromal Cells? eCM 2022, 43, 89–111. [Google Scholar] [CrossRef]
- Ankrum, J.A.; Ong, J.F.; Karp, J.M. Mesenchymal Stem Cells: Immune Evasive, Not Immune Privileged. Nat. Biotechnol. 2014, 32, 252–260. [Google Scholar] [CrossRef]








| Breed | Gender | Age (Years) | COAST Rad. Assessment, IEWG | Number of Joints | Previous Surgical Procedures | Pain upon Manipulation | BCS/Weight (kg) | |
|---|---|---|---|---|---|---|---|---|
| 1 | GS | Male | 5 | Moderate | SE | SCO | Mild | 5/40 kg |
| 2 | CB | Male | 6 | Moderate | BE | SCO | Mild | 3/21 kg |
| 3 | LR | Male | 2 | Mild | BE | SCO | None | 3/36 kg |
| 4 | APT | Female | 7 | Moderate | SE | None | Mild | 3/25 kg |
| 5 | LR | Male | 3 | Mild | BE | SCO | None | 5/40.3 kg |
| 6 | CS | Male | 8 | Moderate | BE | SCO | Mild | 3/16.4 kg |
| 7 | APT | Female | 8 | Mild | BE | None | Mild | 3/22.5 kg |
| 8 | GS | Male | 4 | Mild | SE | APR | None | 3/36 kg |
| Breed | Gender | Age (Years) | Type of Joint | COAST Rad. Assessment | Pain Upon Manipulation | |
|---|---|---|---|---|---|---|
| 1 | GS | male | 2 | elbow | NRSOO | None |
| 2 | CAS | male | 2 | elbow | NRSOO | None |
| 3 | GS | female | 5 | elbow | NRSOO | None |
| 4 | CS | male | 2 | elbow | NRSOO | None |
| 5 | GS | male | 5 | elbow | NRSOO | None |
| 6 | LR | female | 5 | elbow | NRSOO | None |
| 7 | APT | male | 2 | elbow | NRSOO | None |
| 8 | SC | male | 2 | elbow | NRSOO | None |
| 9 | D | female | 3 | elbow | NRSOO | None |
| Patient | Statistical Significance in ROM |
|---|---|
| German shepherd | p value = 0.1300 ns |
| Crossbreed, RE | p value = 0.0013 ss |
| Crossbreed, LE | p value = 0.0011 ss |
| Labrador retriever, RE | p value = 0.0130 ss |
| Labrador retriever, LE | p value = 0.0110 ss |
| American pit bull terrier | p value = 0.0900 ns |
| Labrador retriever, RE | p value = 0.0006 ss |
| Labrador retriever, LE | p value = 0.0112 ss |
| Cocker spaniel, RE | p value = 0.3600 ns |
| Cocker spaniel, LE | p value = 0.0010 ss |
| American pit bull terrier, RE | p value = 0.0002 ss |
| American pit bull terrier, LE | p value = 0.0020 ss |
| German shepherd | p value = 0.4100 ns |
| Biomarker in SF | Day 0. Mean (Range) | Day14. Mean (Range) | Day28. Mean (Range) | p-Value | Statistical Significance |
|---|---|---|---|---|---|
| IFN-y (pg/mL) | 83.58 (4.7–345.7) | 12.1 (1.6–79.5) | 7.40 (1.1–68.9) | 0.001 | ** |
| IL-10 (pg/mL) | 4.9 (2.6–31.28) | 28.1 (12.3–109.6) | 93.64 (8.63–342.8) | 0.009 | * |
| IL-15 (pg/mL) | 1848.2 (201.1–7031.1) | 1018.2 (8.6–5674.5) | 206.6 (72.61–665.4) | 0.124 | ns |
| IL-6 (pg/mL) | 1468.7 (41.0–6409.2) | 565.2 (35.9–1930.2) | 50.7 (11.4–96.2) | 0.011 | * |
| MCP-1 (pg/mL) | 1066.2 (453.1–3271) | 977.5(452.1–2176.5) | 619.9 (369.2–1350.3) | 0.343 | ns |
| TNF-α (pg/mL) | 230.3 (14.8–668.4) | 86.3 (13.2–461.7) | 39.1 (11.3–100.6) | 0.012 | * |
| GM-CSF (pg/mL) | 549.1 (36.7–1567) | 344.4 (22.7–1828.3) | 172.1 (25.8–381.1) | 0.163 | ns |
| Mediator in SF | Mean (Range) |
|---|---|
| IFN-y (pg/mL) | 3.1 (0.7–10.5) |
| IL-10 (pg/mL) | OOR< |
| IL-15 (pg/mL) | OOR< |
| IL-6 (pg/mL) | 17.50 (1.04–113.38) |
| MCP-1 (pg/mL) | 60.50 (33.50–93.0) |
| TNF-α (pg/mL) | 0.83 (0.09–4.11) |
| GM-CSF (pg/mL) | 33.12 (1.14–92.10) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Domaniza, M.; Hluchy, M.; Cizkova, D.; Humenik, F.; Slovinska, L.; Hudakova, N.; Hornakova, L.; Vozar, J.; Trbolova, A. Two Amnion-Derived Mesenchymal Stem-Cells Injections to Osteoarthritic Elbows in Dogs—Pilot Study. Animals 2023, 13, 2195. https://doi.org/10.3390/ani13132195
Domaniza M, Hluchy M, Cizkova D, Humenik F, Slovinska L, Hudakova N, Hornakova L, Vozar J, Trbolova A. Two Amnion-Derived Mesenchymal Stem-Cells Injections to Osteoarthritic Elbows in Dogs—Pilot Study. Animals. 2023; 13(13):2195. https://doi.org/10.3390/ani13132195
Chicago/Turabian StyleDomaniza, Michal, Marian Hluchy, Dasa Cizkova, Filip Humenik, Lucia Slovinska, Nikola Hudakova, Lubica Hornakova, Juraj Vozar, and Alexandra Trbolova. 2023. "Two Amnion-Derived Mesenchymal Stem-Cells Injections to Osteoarthritic Elbows in Dogs—Pilot Study" Animals 13, no. 13: 2195. https://doi.org/10.3390/ani13132195
APA StyleDomaniza, M., Hluchy, M., Cizkova, D., Humenik, F., Slovinska, L., Hudakova, N., Hornakova, L., Vozar, J., & Trbolova, A. (2023). Two Amnion-Derived Mesenchymal Stem-Cells Injections to Osteoarthritic Elbows in Dogs—Pilot Study. Animals, 13(13), 2195. https://doi.org/10.3390/ani13132195





