Simple Summary
One of the greatest challenges to achieving a sustainable aquaculture is finding alternatives to fishmeal as a primary protein source in aquafeeds. Insects represent one of the most promising alternatives being explored and produced as replacements for this ingredient. This review addresses the use of two insect species (black soldier fly, Hermetia illucens, and yellow mealworm, Tenebrio molitor) in freshwater and marine fish diet formulations and the effect of insect meal on fish gut microbiota. Furthermore, the effects of a probiotic, namely, Lactococcus lactis subsp. lactis, are considered. The study of fish gut microbiota is very important for aquaculture practice as gut microbiota plays a significant role in nutrition metabolism, also affecting a number of other physiological functions, including fish growth and development, immune response, and pathogen resistance. Along with recent and promising results in this field, new insights and future directions on fish gut microbiota research are highlighted.
Abstract
Aquaculture is the fastest-growing agricultural industry in the world. Fishmeal is an essential component of commercial fish diets, but its long-term sustainability is a concern. Therefore, it is important to find alternatives to fishmeal that have a similar nutritional value and, at the same time, are affordable and readily available. The search for high-quality alternatives to fishmeal and fish oil has interested researchers worldwide. Over the past 20 years, different insect meals have been studied as a potential alternate source of fishmeal in aquafeeds. On the other hand, probiotics—live microbial strains—are being used as dietary supplements and showing beneficial effects on fish growth and health status. Fish gut microbiota plays a significant role in nutrition metabolism, which affects a number of other physiological functions, including fish growth and development, immune regulation, and pathogen resistance. One of the key reasons for studying fish gut microbiota is the possibility to modify microbial communities that inhabit the intestine to benefit host growth and health. The development of DNA sequencing technologies and advanced bioinformatics tools has made metagenomic analysis a feasible method for researching gut microbes. In this review, we analyze and summarize the current knowledge provided by studies of our research group on using insect meal and probiotic supplements in aquafeed formulations and their effects on different fish gut microbiota. We also highlight future research directions to make insect meals a key source of proteins for sustainable aquaculture and explore the challenges associated with the use of probiotics. Insect meals and probiotics will undoubtedly have a positive effect on the long-term sustainability and profitability of aquaculture.
1. Introduction
Aquaculture is one of the fastest-growing food production sectors in the world, supplying more than half of the global fish supply. By 2050, it is expected that global aquaculture consumption will double. To guarantee long-term food security, efficient and sustainable animal production methods are urgently required. Aquafeeds are mainly based on fishmeal (FM) and fish oil (FO), the most abundant dietary protein source. However, the global increase in aquaculture production has required alternative feedstuff, which often has a detrimental effect on the growth, intestinal health, and immune response of farmed marine fish [1,2,3].
One problem the aquaculture feed industry needs to solve is that of replacing FM with other protein sources. If this cannot be done, serious concerns exist about the industry’s capacity to remain economically and environmentally stable. Sardine, anchovy, herring, capelin, mackerel, and other forage and small pelagic marine fish species are sources of FM and FO. However, owing to the gradual decline in wild marine fish stocks [4,5,6,7,8,9,10,11,12,13,14], it will soon no longer be viable to use these aquafeed raw materials. Plant-based proteins and oils comprise the primary substitutes for FM and FO due to their greater availability and lower cost [4]. Indeed, soybean and other protein- and lipid-rich plants have replaced FM and FO in farmed fish diets [5,6,7,8,9,10]. Soybean meal is a top-rated source of protein in plant-based diets. However, plant-based diets may decrease fish growth and disease resistance due to anti-nutritional substances in plant meals that affect fish feed intake, digestion, and nutrient utilization, causing inflammation in many fish species’ intestines [4,5,6,7,8,9,10,11,12,13,14,15,16,17,18]. Furthermore, plant-based feed ingredients are deficient in protein, lack a balanced amino acid profile, are unpalatable, and compete with other food industrial sectors [16,19,20].
As a result of all of these factors, the need to find better ways of getting protein from other valuable alternatives to FM, for example, animal feed ingredients such as by-products from slaughterhouses or insect meals (IM), has increased [18,21,22]. From this perspective, insects have the potential to open a new world of sustainable, protein-rich ingredients for aquafeeds. Furthermore, single-cell proteins (SCP) deriving from microalgae, bacteria, and yeast are also being used as fish feed ingredients. Insect, micro- and macroalgae, and microbial meals are becoming more popular as aquafeed components [23,24,25]. Among the scientific publications currently available in indexed databases, just 19% were focused on IM, whereas 16% discussed the use of microalgae: IM and microalgae are both components that offer much promise in the aquafeed sector. Animal health and metabolism are influenced by a complex relationship between the host, the gut microbiota, and their feed. A balanced microbiota is important for the overall health and well-being of the host. The fish gut microbiota significantly affects fish health and physiology [8]. It helps develop the immune system and promotes nutrient utilization [26,27]. The structure of the gut microbial communities, including microbial diversity, is highly influenced by the ingredients of the diet because the microbiota reacts quickly to dietary changes [28]. It is well established that replacing FM with plants, yeast, IM, or animal by-products influences the biodiversity and number of gut bacteria [22,29,30,31,32,33,34]. Furthermore, the bioactive compounds present in insects can alter the complex communities of intestinal microbiota. Consequently, the variety and richness of fish gut bacteria have changed as a result of replacing FM in their diets with IM, either from Hermetia illucens (HI) or Tenebrio molitor (TM) [35,36,37].
The gut microbiota is usually called an “extra organ” because of its significant role in many physiological processes of the host, including digestion, metabolism, reproduction, development, and immunological response. In recent years, new alternative components have been investigated and used in aquafeeds. Since gut microbes are important for digestion and health, a number of studies have been carried out to determine how diet affects the gut microbiota of aquatic species. Therefore, this review highlights current developments and future perspectives of alternatives to conventional protein sources, that is, how IMs used as aquafeed ingredients affect the gut microbiota of marine fish. Furthermore, we review the effects of probiotics on fish intestinal microbiota.
2. Insects for Sustainable Aquaculture
Insects are an environmentally sustainable protein-rich feed ingredient for farmed fish. IM is being considered a potential alternative to FM as a protein source in aquaculture feeds. Interest in using IM as an alternative to FM has increased since the European Union (EU) authorized the use of IM from seven distinct insect species in aquaculture feeds [38,39]. According to the circular economy concept, insects are worthy candidates for aquafeed ingredients. Many country’s aquaculture industries increasingly depend on IM instead of FM. Insects are nutritionally valuable due to their high protein (60–80%), fat (31–43%), essential amino acids, and mineral and vitamin content [24]. Due to their high protein content and balanced amino acid profile, IM has emerged as a popular alternative to FM and a new source of protein in terrestrial and aquatic animal diets [40,41]. Therefore, insects constitute an excellent alternative to conventionally produced animal-based protein sources for use as feed [42,43]. Many studies have investigated the effects of FM/IM substitution in various fish species diets. The European Commission has withdrawn the ban on using processed animal proteins generated from insects in aquafeed for farm fish under regulation EU-2017/893. As a result, IM can now be used in aquafeeds. The regulation lists the seven types of insects that are allowed: black soldier fly, Hermetia illucens; common housefly, Musca domestica; yellow mealworm Tenebrio molitor; lesser mealworm, Alphitobius diaperinus; house cricket, Acheta domesticus; banded cricket, Gryllodes sigillatus; and field cricket, Gryllus assimilis. Of these, flies in particular have been the focus of aquafeed industry research in recent years owing to their many advantages over other animal protein sources [44]. HI and TM are the main species presently receiving considerable attention for aquaculture feed formulations [45]. Most studies have shown that replacing FM with IM is a good approach to increase aquaculture sustainability; however, the results vary based on the fish and insect species used. We recently obtained promising findings in marine and freshwater carnivorous fish species with the dietary use of different inclusion rates of black soldier fly and yellow mealworm meals [36,37,46,47,48,49]. Table 1 represents the research that our group has done on the effects of IM on fish gut microbiota.

Table 1.
Studies evaluating the effects of insect meal on fish gut microbiota using Next Generation Sequencing platforms.
2.1. Black Soldier Fly (Hermetia illucens, HI)
When producing IM, the black soldier fly HI is an excellent potential species because its amino acid profile is similar to that of FM, making it a suitable alternative protein source [24]. HI is the most widely studied and used insect species, representing our research group’s primary alternative to FM raw material. Indeed, HI can be raised quickly, have a high fertility rate, and turn waste into high-quality protein [50]. An increasing number of feeding trials have been conducted, demonstrating that HI meals can be a suitable FM replacement in aquaculture diets [41,44,51]. During the last few decades, approximately 130 research publications with the terms “Black soldier fly,” “Larvae meal,” and “Aquaculture” have been indexed in PubMed, Scopus, Web of Science, and other databases. Prepupae of HI comprise an intriguing choice for producing IM since mass-rearing procedures for high-quality output currently exist [24]. Using HI in fish feed provides a way to solve problems in the aquaculture industry related to managing a sustainable aquatic environment. According to several studies, HI can replace conventional FM and totally replace SBM in aquaculture feeds without negatively influencing fish growth, feed efficiency, digestion, or fillet quality [37,52,53,54]. Our experiments have shown that rainbow trout (Oncorhynchus mykiss) can tolerate up to 50% HI meal in their diet with no negative effects on fish growth and survival [36,37,46,48,55] and with positive effects on the gut microbiota of fish.
Effects of FM/HI Meal Replacement on Fish Gut Microbiota
HI meals are becoming more popular in aquaculture feeds, but ideal inclusion levels still must be determined to ensure fish growth and health. An increasing number of studies have examined the effects of substituting HI meals for FM in the diets of different species of fish. Most research recommended partial replacements of FM with HI meals. However, some recent studies revealed 100% replacement without affecting fish growth, especially for carnivorous fish [52].
Regarding fish growth, health, and gut microbiome, our group’s work has shown that partial or up to 50% inclusion of HI meal in the diet is well tolerated and has no negative effects on fish growth or survival. Diet has a significant role in shaping the gut microbiota, but the surrounding environment and environmental factors can also significantly impact microbiota composition. Our research group previously evaluated the effects of different HI inclusion levels in high-FM diets on fish gut microbiota using high-throughput sequencing technologies [36,37,46]. In all the experiments, we applied high-throughput sequencing of the 16S rRNA gene to assess the dynamics of major gut bacterial taxa in response to diet. PICRUSt1 bioinformatics software was used to determine gut microorganisms’ key active biological pathways. We reported that the partial substitution of dietary FM with 10%, 20%, or 30% of a defatted HI meal had an important effect in modulating the intestinal transient (allochthonous) and resident (autochthonous) bacterial communities in trout [36,37,46].
HI diet increased butyrate-producing bacteria in the fish gut [36,37,49] and led to diversification and other alterations in the intestinal bacterial makeup of rainbow trout [37,53,56]. In addition, dietary IM increased the colonization of beneficial bacteria, such as lactic acid bacteria (LAB), which are often used as probiotics in animal nutrition [36,37]. This was a good result as it is known that beneficial bacteria species compete with gut detrimental bacteria for niche space and produce and secrete antimicrobial peptides, thus protecting the host from colonization and proliferation of environmental pathogens [57].
Based on the metabarcoding results, three phyla, Firmicutes, Proteobacteria, and Tenericutes, were found to be the most abundant in the digestive tract of rainbow trout [37], and in fish fed with 10–30% HI meal; diversity was higher in allochthonous, but not in autochthonous gut microbiota [36,37]. Instead, another study [53] observed that trout fed a diet containing 20% HI meal had a higher species richness in their gut microbiota. Furthermore, the autochthonous bacterial community significantly influenced host metabolism and health status more than the allochthonous intestinal bacteria.
Fish gut microbiota studies vary in many ways, including the techniques used to analyze the microbiome. The dietary HI meal’s effects on autochthonous microbiota of trout were first explored using the gradient gel electrophoresis (DGGE) method [53], which identified a lower number of bacterial species than the Illumina MiSeq method, which we used in all our studies [36]. We analyzed the inclusion of 10%, 20%, and 30% HI meals on the autochthonous intestinal microbiota of rainbow trout (O. mykiss) and found a reduced abundance of Proteobacteria and an increased abundance of Mycoplasma, which produce lactic and acetic acid as final products of its fermentation [36,37]. These differences in the composition of the autochthonous intestinal microbiota are due to the prebiotic characteristics of fermentable chitin. In one of our previous studies [37], Proteobacteria, Firmicutes, and Actinobacteria dominated trout’s allochthonous gut microbial community. Interestingly, also other studies reported that LAB (Firmicutes phylum) were only found in large numbers in the gut contents of trout that had been fed IM but they were absent in gut mucosa [53]. In contrast, trout intestinal mucosa in our study contained many Proteobacteria (Gammaproteobacteria) bacteria, which was in line with previous work on rainbow trout [53,58]. The most common phyla are not the only ones for which differences between these findings and our previously published data were observed. Fish mucosa samples contained considerably fewer operational taxonomic units (OTUs) (74 vs. 450, respectively) than fish gut digesta samples [37]. These results agree with another study [59], which found that microbial diversity was lower in the gut mucosa than in the luminal part. This indicates that certain species of bacteria colonize the gut mucosal layer poorly and that the number of bacteria and the diversification of the autochthonous bacterial community may be different from the allochthonous microbiota [60]. In our studies, 20% IM increased biodiversity (Shannon and Simpson evenness indices) but not bacterial richness [36,37]. In line with previous research, we found that HI meal inclusion in the trout diet had positive effects on gut bacterial biodiversity [37,53,56]. Furthermore, since dietary effects may in part be biased by taxa from the feed microbiome [61], we included the feed as control and did not use digesta as a proxy for the intestinal microbiome [36,37]. Indeed, to fully unveil the response of gut microbiota to dietary changes, we performed concurrent profiling of feed microbiota, and digesta- and mucosa-associated gut microbiota.
In addition to their protein and fat content, insects contain a large amount of chitin, which is the building material that gives strength to the exoskeletons of insects. Studies have shown that the gut microbiota of fish may be altered by chitin [62]. In Atlantic salmon, a chitin-rich diet altered gut microbiota, revealing over 100 autochthonous bacterial species [63]. Dietary chitin or chitosan modulates fish gut microbiotas due to its prebiotic, antibacterial, and immunomodulatory properties [37,46,49,64,65]. Many fish cannot digest chitin, so it is possible to consider it as an insoluble fiber with possible prebiotic qualities. These properties may help maintain a well-balanced and healthy gut microbiota. The gut microbiota helps digest otherwise indigestible feed ingredients, generating short-chain fatty acids (SCFAs), which are the main energy source for intestinal epithelial cells [66]. Furthermore, our latest research [47] on the effects of chitin-rich shrimp head meal (SHM) and HI pupal exuviae on the gut microbiota of rainbow trout demonstrated that HI exuviae exert a modulatory influence on the fish gut microbiota by increasing the number of Firmicutes and Actinobacteria. Pupal exuviae thus represent a promising prebiotic for fish gut microbiota, increasing gut bacterial richness and the amount of beneficial chitin-degrading bacteria, such as Bacillus species, which promotes SCFA synthesis, especially butyrate. Similarly, adding krill or chitin into salmonid diets increased bacterial alpha diversity [62]. Therefore, our findings should not be unexpected when considering the chitin level of the IM.
Chitin is a prebiotic that increases the diversity of the bacteria in the gut. A healthy gut is typically characterized by a diverse bacterial population. In contrast, decreased diversity is typically associated with dysbiosis and illness risk, due to low bacterial competition for space and resources and enteric pathogen colonization [67,68]. The addition of HI meal to the trout diet significantly decreased indigenous Proteobacteria in the intestinal digesta [36,37]. The same finding was obtained in a study on the digesta and mucosa-associated trout microbiota [56]. Chitin, an insoluble fiber, may reduce Proteobacteria in IM-fed groups. According to several investigations, chitin and deacetylated chitin derivatives are antibacterial and bacteriostatic against Gram-negative pathogens [47,69]. We reported that trout-fed HI meal showed decreased Gammaproteobacteria, including the genera Shewanella, Aeromonas, Citrobacter, and Kluyera, which are considered responsible for some diseases in fish [36]. Therefore, including IM meal in trout diets has a positive effect that inhibits potential pathogen growth. Fish fed 20% and 30% HI diets had more Mycoplasma-genus bacteria in their intestines, and these may be beneficial [36,37,47]. Many studies have identified Mycoplasma as the predominant genus in the distal intestines of rainbow trout and other farmed salmonids [33,70,71]. Bacilli and Clostridium, which are also included in the phylum Firmicutes, are closely related to Mycoplasma. They are generally obligate symbiotic microbes of the gastrointestinal ecosystem because their small genome size makes it unlikely that complex metabolic functions take place in the fish gut [36]. Lactic and acetic acids are the main metabolites of Mycoplasma bacteria [71,72]. Mycoplasma maintains intestinal homeostasis in trout by using fermentable substrates and releasing end products from bacterial fermentations [73]. Recent research on trout revealed that a lower level of Mycoplasma in the gastrointestinal tract makes the fish more susceptible to disease [74]. These findings suggest that Mycoplasma produces antimicrobial chemicals, such as lactic and acetic acids, which are the main metabolites that benefit host health.
2.2. Yellow Mealworm (Tenebrio molitor, TM)
Yellow mealworms are becoming more popular as an alternative source of protein in aquaculture diets due to their high efficiency in converting organic waste, being considered an ideal circular economy insect. Defatted TM provides up to 63.84% crude protein and an amino acid composition similar to that of FM [75]. Furthermore, TM contain anti-tumoral, antibacterial, antioxidant, and immunomodulatory, physiologically active compounds [76,77]. TM has been evaluated as a potential alternative to FM as a protein source in the diets of various fish species. The nutritional value of TM varies with its substrate composition and rearing settings. Although most studies have indicated that 25% to 30% of TM be included in the diet [78], rainbow trout fed different FM/TM meal replacement levels showed better performance [49]. Significant growth improvement was seen in red seabream (Pagrus major) fed diets containing 65% defatted TM larval meal, completely replacing FM [79]. TM showed the highest apparent digestibility coefficient of the four IMs tested in Nile tilapia [80]. This proves that TM larvae can replace FM as a protein source in fish diets. One of our studies examined the impact of replacing FM with TM meal in rainbow trout diets on fish weight gain and gut and skin microbiome [49]. Dietary FM substitution with TM has been explored extensively on fish development performance but less on host symbiotic microbial population [49]. Like HI, TM contains bioactive chemicals that are abundant in chitin and lauric acid and affect the gut microbiota [35,36,37]. Most current research on fish microbiota has focused on the bacterial diversity that may be discovered in the fish’s gut; however, fish also have distinct microbial diversity in other important body sites. Particularly, the skin microbiota of fish and most farm animals has not been thoroughly studied but would require careful consideration. Fish skin constitutes one of their vital mucosal barriers to the outer world. Thus, skin microbiota plays a very important role in preventing fish diseases. In one of our studies, therefore, we investigated how the gut and skin microbiota of trout changed when FM was replaced with TM larvae meal [49].
Effects of FM/TM Meal Replacement on Fish Gut Microbiota
A considerable amount of research has been conducted on mealworm meals in aquafeeds. TM is an excellent alternative to FM, positively influencing fish growth rates and gut microbiota. The appropriate TM meal inclusion rates in feeds for different fish species depend on the nutritional requirements of a given fish species and the nutritional quality of the TM, which in turn depends on the diet and culture conditions of the larvae. Insect meal manufacturers have increased defatted insect meal production in recent years. Defatting insect meal increases crude protein and degradation resistance [24]. In rainbow trout diets, 25% or 50% TM meal did not affect fish weight but significantly improved feed conversion and the protein efficiency ratio [81,82]. The amount of protein, amino acids, micronutrients, lipids, and fatty acids in TM meal makes it a suitable replacement for FM in aquafeeds based on its effects on fish growth performance. In contrast, 50% of full-fat TM diets in European seabass reduced fish growth compared to FM diets [83]. In marine carnivorous fish species, high TM levels in aquafeed have led to reduced growth [84]. Our study found no statistically significant differences in growth performance features in rainbow trout after 90 days of feeding with either a 100% substitution of FM with a partially defatted TM diet or a diet without TM [49].
Many studies have focused on the impact of substituting TM meal for FM on growth and development without attempting to understand the processes that underlie these effects. It is important to use molecular genetics and genome sequencing to determine how TM meal works, how it is metabolized, and how it is absorbed by the digestive systems of different cultured fish species [49]. The gut microbiota plays an important role in enhancing feed digestion, which benefits the general health of fish [85]. As TM is being used in fish diets as a raw material, it is important to understand how gut microbes respond to adding TM to the diet. Several fish species, including rainbow trout, have been investigated to determine how dietary TM affects the composition and diversity of gut microbiota [35,49,86]. Our research group demonstrated how 100% of TM influences rainbow trout gut microbial populations [49]. Substituting FM with TM meal did not influence the species richness and variety of gut mucosal bacteria [50], a finding similar to that obtained from our previous studies [36,37]. Consistent with our findings, feeding rainbow trout (O. mykiss) or sea trout (Salmo trutta m. trutta) a hydrolyzed TM meal diet did not affect digesta-associated bacteria [86,87]. According to the results of our metagenomic analysis, the phylum Tenericutes was most represented in trout intestine irrespective of diet, followed by Proteobacteria and Firmicutes in descending order [49]; all these bacteria taxa play a key role in the host’s nutrition and metabolism. Furthermore, the abundance of Lactobacillus and Enterococcus bacteria increased in the intestines of juvenile rainbow trout fed a diet containing with 20% TM meal [86]. The prebiotic characteristics of chitin in dietary IM may be responsible for the increase in lactic acid bacteria. However, in our study on 100% FM substitution with a partially defatted TM diet, intestinal LAB did not increase [49]. This was a surprising result, especially compared to what we had seen in the intestines of trout-fed diets with HI meal [36,37]. Indeed, substituting FM with IM from HI larvae positively modulated rainbow trout gut microbiota by raising the levels of LAB, which are helpful bacteria commonly used as probiotics in the diet of fish and other vertebrates [36,37,53]. There is no doubt that LAB is crucial for degrading dietary fiber. In addition, they actively participate in host defense against pathogenic organisms by generating bactericidal chemicals, such as lactic acid, hydrogen peroxide, bacteriocins, and biosurfactants, which inhibit pathogen colonization of the intestinal epithelium [88,89]. The relative abundance of Actinobacteria increased in the digestive tracts of trout when TM larvae meal was added to their diet, but this effect was not evident in European sea bass or gilthead sea bream [35]. Indeed, the gut microbiota is usually changed towards Firmicutes and/or Actinobacteria when dietary fiber such as chitin is included [46,49,53,90]. Taken together, our data revealed that there were no negative effects on rainbow trout intestinal microbiota populations when FM was completely replaced with TM. No noticeable dysbiosis symptoms were found, but only slight microbial changes were seen [49]. The research revealed that TM larvae meal is a valid substitute for FM as an animal protein in aquafeeds.
3. Probiotics for Sustainable Aquaculture
Probiotics are living microorganisms that, when administered correctly, positively regulate an organism’s health [91]. They are regarded as important modulators of many biological processes such as digestion, immunological activation, restoring microbial balance, and modulating the microbiota composition and have potent antioxidant qualities due to their effects on the gut microbiota [92]. Probiotics can inhibit pathogens in various ways, including by directly competing for nutrients and cell attachment space and generating inhibitory molecules, such as lactoferrin, lysozyme, bacteriocins, siderophores, and enzymes. Probiotics secrete proteases, amylases, and lipases that degrade those feed ingredients the fish gut cannot digest, leading to enhanced growth and nutrient conversion efficiency [93,94,95]. In aquaculture, many probiotic microbial strains are now used [96]. LAB, such as Lactobacillus sp., Bacillus sp., Enterococcus sp., and yeast, Saccharomyces cerevisiae, are the most common probiotics used in aquaculture [97,98]. These microorganisms are widely distributed in nature in the digestive tracts of farmed fish and regulate the fish microbiota as permanent or transitory inhabitants [99]. Probiotics in aquaculture are generally also used to reduce antibiotic use and promote aquaculture industry sustainability. The misuse of antibiotics has a negative effect on the aquatic environment, particularly in aquatic ecosystems where antimicrobials can persist for a long time and help bacteria become resistant to multiple antibiotics [100]. Antibiotics also help fish grow but their use as growth promoters has reduced the variety and abundance of indigenous gut microbiota, negatively affecting fish immune systems [101]. For these reasons, antibiotics have been restricted in farmed animals in the EU since 2006 [102,103]; therefore, several research projects have attempted to substitute antibiotics with probiotics to help the growth and development of farmed animals [104].
Probiotics boost feed digestibility and nutrient absorption in cultured fish, leading to better fish growth and conversion rates [105]. They also maintain gut microbiota balance, especially at larval stages, when vaccination is challenging [101]. Probiotics are also being used more in aquaculture, and studies have confirmed the advantages for commercially important farmed fish [106,107,108]. In our recent work, gilthead sea bream fed low and high dosages of probiotic Lc. lactis subspecies lactis showed higher weight gain than control fish fed a diet without probiotics [109]. High-throughput sequencing was used in our study to analyze the alterations in sea bream gut microbial populations after Lc. lactis subsp. lactis feeding. The findings here indicate that digestion and nutrient utilization had improved in gilthead sea bream fed probiotics. The same results were seen when Lactobacillus spp. and Shewanella putrefaciens Pdp11 were administered to gilthead sea bream [110]. Many other farmed fish species showed improved growth performance when L. lactis was used as a probiotic [111,112,113]. Concerning the microbiota analysis, we also analyzed the microbiota populations associated with feeds at the end of our feeding trial to determine how stable the probiotics were in the fish diets. Firmicutes and Proteobacteria represented the most numerous bacterial phyla, followed by the Bacteriodetes and Fusobacteria in descending order. Compared to the most representative genera of Firmicutes phylum, the relative abundance of the probiotic L. lactis was higher.
The proportion of L. lactis included in the control diet was 0%, while it was 64% and 71% in the treatment diets, respectively, which corresponds with administering a low and high dose of probiotics to fish [109]. Gilthead sea bream fed high dosages of Lc. lactis had an increase in Spirochete bacteria phylum in their gut, which were almost absent in fish fed low doses of Lc. lactis and in the control fish. Around 200 different genera in the Firmicutes phylum, including Lactobacillus, help maintain fish intestine health [37,109,114]. Commensal Firmicutes and Bacteroidetes produce butyrate, acetate, and propionate SCFAs by dietary fiber fermentation. The gut microbiota of sea bream on high-probiotic diets had a Proteobacteria/Firmicutes ratio five times higher than in the other groups. This result is not surprising since Lc. lactis subsp. lactis produces the antibiotic nisin, displaying strong activity against Gram-positive bacteria, and a vast majority of Firmicutes are Gram-positive [109,115].
In our study, the analysis of gut-adherent (autochthonous) microbiota showed a lack of colonization of the probiotic Lc. lactis in the host’s intestinal mucosa [109]. This result was expected because it is well-recognized that the underlying mechanisms of establishing probiotics in the host intestinal mucosa are challenging and influenced by complex molecular interactions. Our probiotic modified the fish gut microbiota without colonizing the host’s intestinal mucosa, proving that colonization is not always required to trigger host modification [109]. In terms of diversity indices, the analyses of intestinal microbiota also found significant and controversial differences between groups of fish. There was a significant difference in the variety and diversification characterized by alpha diversity parameters in fish fed a low-probiotic diet compared to the control or high-probiotic diet fish [109].
Consistent with our findings, the bacterial diversity in the intestinal mucosa of Atlantic salmon supplemented with LAB was higher [116]. On the other hand, in the probiotic-rich diet group, gut bacteria diversity was lowest despite reaching the highest growth rates. A functionally unbalanced ecosystem may reduce competition for opportunistic or invading bacteria if bacterial diversity decreases and is generally regarded as a negative outcome [110,117,118]. While it has been documented that administering prebiotics (specialized plant fibers that stimulate the growth of healthy bacteria) increases the microbial richness of the gut, evidence of the benefits of probiotics on fish remains less clear. According to findings in the literature, the dietary probiotic Bacillus subtilis, alone or in combination with prebiotics or microalgae, decreased gilthead sea bream species richness and diversity indexes [110,119]. Moreover, probiotics such as LAB produce antimicrobial substances that limit the growth of other microbes, which can change the gut microbiota’s composition and biodiversity [120].
The correlations found in the aforementioned studies between diet and fish gut microbiota suggest that well-designed probiotics could provide a potential way to improve fish growth performance and digestive ability. However, traditional probiotics, such as lactic acid bacteria and yeasts are not the dominant indigenous microbes in the digestive tract of fish, and their use in fish may risk causing microbial dysbiosis in some cases [121]. Therefore, identifying commensal beneficial bacteria in fish is of great value for the development of novel probiotics for aquaculture [121].
4. Metagenomic Analysis for the Identification of Gut Microbiota
Different culture-dependent methods followed by identification based on biochemical and phenotypic characteristics of bacteria were used to identify and characterize fish microbiota in previous times. Unfortunately, culture-dependent techniques give a limited picture of intestinal microbiota because only a low fraction, down to about 1% of the bacteria from fish intestine, can be cultivated [22]. Therefore, culture-independent molecular technologies, such as next-generation sequencing (NGS) technologies, targeted amplicon sequencing of the 16S rRNA gene [122,123,124], polymerase chain reaction-denaturing gradient gel electrophoresis (PCR-DGGE) [125,126,127], or 16S rDNA PCR-DGGE and RNA polymerase β-subunit gene quantitative PCR [128], have been used more recently to evaluate the intestinal microbiota. New research approaches have boosted our understanding of the interplay between microbes and their hosts. In particular, NGS enabled the identification and quantification of fish gut bacteria at unprecedented resolution, providing novel insights into the role of the microbiota in fish growth and health [66,129].
Metagenomics has thoroughly changed the study of the fish gut microbiota. With these methods, it is possible to directly look at the genome of microorganisms from samples taken from the environment [130,131]. It can provide a deeper understanding of the information that the retrieved DNA reveals about the host or environment-specific host species and help researchers understand microbial diversity in aquaculture. The use of 16S rRNA sequencing as the gold standard for identifying variability of the 16S rRNA to assess the composition of whole bacterial communities through culture-independent methods is being used by many researchers [59,132,133]. Additionally, metagenomics methods have been successfully used to find novel genes and microbial pathways as well as to discover functional dysbiosis [134].
To investigate the effect of aquafeed ingredients on gut microbiota composition, we used the Illumina MiSeq platform (Illumina, Italy) for high-throughput sequencing of the 16S rRNA gene (Figure 1) to analyze and characterize the complete gut microbiome of different fish species [22,36,37,46,47,48,49]. Using the bioinformatics application PICRUSt (Phylogenetic Investigation of Communities by Reconstruction of Unobserved States), the active biological pathways of gut bacteria could be identified. This method is being used more frequently in fish studies. It can quickly and cost-effectively capture detailed sequencing data that provides additional information on even minute amounts of bacteria [66]. The Illumina, Roche 454, and Ion Torrent PGM (Personal Genome Machine) are the three leading platforms used to study fish gut microbiota [135]. Many studies have been performed using these platforms on the microbiota that live in different parts of a fish’s body, such as the gastrointestinal tract, gills, and skin that evolve to permit colonization on the mucosal surfaces by complex commensal microorganisms [136]. The skin, gills, and digestive tract of fish are the main entry routes for pathogens [136]. Thus, most research on microbial communities has focused on these body regions. Owing to the development and continuous growth of aquaculture, microbiota study has recently increased. More focus is now being directed to the microorganisms that inhabit the gastrointestinal systems of different finfish and crustaceans, such as prawns and crabs [136]. Furthermore, there is an increasing interest in the impact of aquafeed on the fish gut microbiome, as some recent findings [61] are consistent with a model wherein gut microbial profiles are to a different degree influenced by bacterial DNA present in the feed itself through a “feed microbiome” carry-over effect.
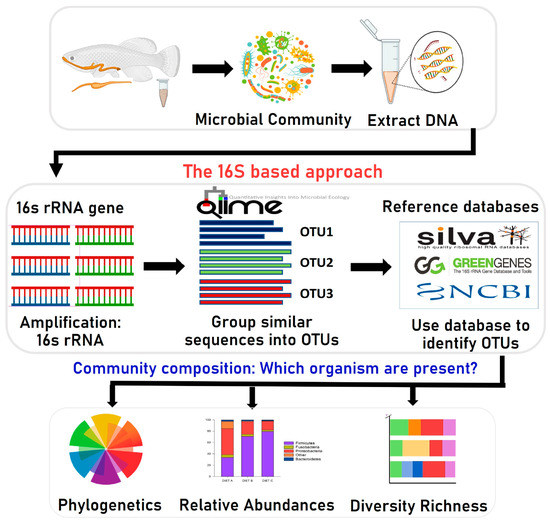
Figure 1.
Overview of bioinformatics methods for metagenomic analysis of gut microbiota based on the 16S rRNA gene sequencing. OTU; Operational Taxonomic Unit.
5. Conclusion and Future Prospects for Applied Research
This review summarizes the results of studies from our research group on the effects of diets formulated to contain IM from either T. molitor or H. illucens to replace dietary FM on freshwater fish gut microbiota. It also emphasizes the connection between diet and fish gut microbiota, suggesting that “tuning” the microbiota composition through the use of new raw materials could offer a promising strategy towards a sustainable aquaculture. The effects of a probiotic (Lactococcus lactis subs lactis) used as a feed supplement on marine fish gut microbiota are also reviewed. According to metagenomic data, IMs from H. illucens, or T. molitor constitute valid alternative protein sources that can affect gut microbiota composition and overall fish health. With regard to Lc lactis, although this probiotic did not colonize the host’s intestinal mucosa, it positively influenced the fish gut microbiota by changing the abundance of different beneficial bacterial taxa and by impacting many metabolic pathways associated with protein absorption and digestion. Therefore, insects and probiotics used in fish diets have a positive effect on the composition of gut microbiota and on fish nutritional physiology.
We believe this information will be helpful to researchers and aquaculture experts in fish nutrition, particularly when developing novel feed formulations or experimenting with various feed components and additives.
Author Contributions
Conceptualization: G.T. and I.H.; methodology: S.R.; software, G.S.; validation: I.H., S.R., G.S. and G.T.; formal analysis: I.H. and G.S.; investigation: I.H., S.R. and G.S.; resources: G.T.; data curation: S.R. and G.S.; writing—original draft preparation, I.H.; writing—review and editing: G.T. and G.S; supervision: G.T.; project administration: G.T.; funding acquisition, G.T. All authors have read and agreed to the published version of the manuscript.
Funding
This work was funded by the EU Horizon 2020 AquaIMPACT project (Genomic and nutritional innovations for genetically superior farmed fish to improve efficiency in European aquaculture; 818367).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The authors declare that no new data were created.
Acknowledgments
Special thanks to Micaela Antonini at the Aquaculture Unit of the University of Insubria for her technical assistance. I.H. and G.S. are students of the “Dottorato in Scienze della Vita e Biotecnologie” at the “Università degli Studi dell’Insubria,” Varese, Italy.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Aragão, C.; Cabano, M.; Colen, R.; Fuentes, J.; Dias, J. Alternative formulations for gilthead seabream diets: Towards a more sustainable production. Aquac. Nutr. 2020, 26, 444–455. [Google Scholar] [CrossRef]
- Estensoro, I.; Ballester-Lozano, G.; Benedito-Palos, L.; Grammes, F.; Martos-Sitcha, J.A.; Mydland, L.-T.; Calduch-Giner, J.A.; Fuentes, J.; Karalazos, V.; Ortiz, A. Dietary butyrate helps to restore the intestinal status of a marine teleost (Sparus aurata) fed extreme diets low in fish meal and fish oil. PLoS ONE 2016, 11, e0166564. [Google Scholar] [CrossRef]
- Krogdahl, Å.; Gajardo, K.; Kortner, T.M.; Penn, M.; Gu, M.; Berge, G.M.; Bakke, A.M. Soya saponins induce enteritis in Atlantic salmon (Salmo salar L.). J. Agric. Food Chem. 2015, 63, 3887–3902. [Google Scholar] [CrossRef] [PubMed]
- Kalhoro, H.; Zhou, J.; Hua, Y.; Ng, W.K.; Ye, L.; Zhang, J.; Shao, Q. Soy protein concentrate as a substitute for fish meal in diets for juvenile Acanthopagrus schlegelii: Effects on growth, phosphorus discharge and digestive enzyme activity. Aquac. Res. 2018, 49, 1896–1906. [Google Scholar] [CrossRef]
- Hedrera, M.I.; Galdames, J.A.; Jimenez-Reyes, M.F.; Reyes, A.E.; Avendaño-Herrera, R.; Romero, J.; Feijóo, C.G. Soybean meal induces intestinal inflammation in zebrafish larvae. PLoS ONE 2013, 8, e69983. [Google Scholar] [CrossRef]
- Francis, G.; Makkar, H.P.; Becker, K. Antinutritional factors present in plant-derived alternate fish feed ingredients and their effects in fish. Aquaculture 2001, 199, 197–227. [Google Scholar] [CrossRef]
- Merrifield, D.L.; Olsen, R.E.; Myklebust, R.; Ringø, E.; El-Shemy, H. Dietary effect of soybean (Glycine max) products on gut histology and microbiota of fish. In Soybean and Nutrition; InteckOpen: London, UK, 2011; pp. 231–250. [Google Scholar]
- Egerton, S.; Culloty, S.; Whooley, J.; Stanton, C.; Ross, R.P. The gut microbiota of marine fish. Front. Microbiol. 2018, 9, 873. [Google Scholar] [CrossRef] [PubMed]
- Penn, M.H.; Bendiksen, E.Å.; Campbell, P.; Krogdahl, Å. High level of dietary pea protein concentrate induces enteropathy in Atlantic salmon (Salmo salar L.). Aquaculture 2011, 310, 267–273. [Google Scholar] [CrossRef]
- Santigosa, E.; García-Meilán, I.; Valentín, J.M.; Navarro, I.; Pérez-Sánchez, J.; Gallardo, M.Á. Plant oils’ inclusion in high fish meal-substituted diets: Effect on digestion and nutrient absorption in gilthead sea bream (Sparus aurata L.). Aquac. Res. 2011, 42, 962–974. [Google Scholar] [CrossRef]
- Hardy, R.W. Collaborative opportunities between fish nutrition and other disciplines in aquaculture: An overview. Aquaculture 1999, 177, 217–230. [Google Scholar] [CrossRef]
- Naylor, R.L.; Goldburg, R.J.; Primavera, J.H.; Kautsky, N.; Beveridge, M.C.; Clay, J.; Folke, C.; Lubchenco, J.; Mooney, H.; Troell, M. Effect of aquaculture on world fish supplies. Nature 2000, 405, 1017–1024. [Google Scholar] [CrossRef]
- Pauly, D.; Zeller, D. Comments on FAOs state of world fisheries and aquaculture (SOFIA 2016). Mar. Policy 2017, 77, 176–181. [Google Scholar] [CrossRef]
- Tacon, A.G.; Metian, M. Global overview on the use of fish meal and fish oil in industrially compounded aquafeeds: Trends and future prospects. Aquaculture 2008, 285, 146–158. [Google Scholar] [CrossRef]
- Espe, M.; Lemme, A.; Petri, A.; El-Mowafi, A. Can Atlantic salmon (Salmo salar) grow on diets devoid of fish meal? Aquaculture 2006, 255, 255–262. [Google Scholar] [CrossRef]
- Gatlin III, D.M.; Barrows, F.T.; Brown, P.; Dabrowski, K.; Gaylord, T.G.; Hardy, R.W.; Herman, E.; Hu, G.; Krogdahl, Å.; Nelson, R. Expanding the utilization of sustainable plant products in aquafeeds: A review. Aquac. Res. 2007, 38, 551–579. [Google Scholar] [CrossRef]
- Aksnes, A.; Hope, B.; Albrektsen, S. Size-fractionated fish hydrolysate as feed ingredient for rainbow trout (Oncorhynchus mykiss) fed high plant protein diets. II: Flesh quality, absorption, retention and fillet levels of taurine and anserine. Aquaculture 2006, 261, 318–326. [Google Scholar] [CrossRef]
- Oliva-Teles, A.; Enes, P.; Peres, H. Replacing fishmeal and fish oil in industrial aquafeeds for carnivorous fish. In Feed and Feeding Practices in Aquaculture; Woodhead Publishing: Sawston, UK, 2015; pp. 203–233. [Google Scholar]
- Glencross, B.D.; Booth, M.; Allan, G.L. A feed is only as good as its ingredients–a review of ingredient evaluation strategies for aquaculture feeds. Aquac. Nutr. 2007, 13, 17–34. [Google Scholar] [CrossRef]
- Krogdahl, Å.; Penn, M.; Thorsen, J.; Refstie, S.; Bakke, A.M. Important antinutrients in plant feedstuffs for aquaculture: An update on recent findings regarding responses in salmonids. Aquac. Res. 2010, 41, 333–344. [Google Scholar] [CrossRef]
- Moutinho, S.; Martínez-Llorens, S.; Tomás-Vidal, A.; Jover-Cerdá, M.; Oliva-Teles, A.; Peres, H. Meat and bone meal as partial replacement for fish meal in diets for gilthead seabream (Sparus aurata) juveniles: Growth, feed efficiency, amino acid utilization, and economic efficiency. Aquaculture 2017, 468, 271–277. [Google Scholar] [CrossRef]
- Rimoldi, S.; Terova, G.; Ascione, C.; Giannico, R.; Brambilla, F. Next generation sequencing for gut microbiome characterization in rainbow trout (Oncorhynchus mykiss) fed animal by-product meals as an alternative to fishmeal protein sources. PLoS ONE 2018, 13, e0193652. [Google Scholar] [CrossRef]
- Biancarosa, I.; Sele, V.; Belghit, I.; Ørnsrud, R.; Lock, E.-J.; Amlund, H. Replacing fish meal with insect meal in the diet of Atlantic salmon (Salmo salar) does not impact the amount of contaminants in the feed and it lowers accumulation of arsenic in the fillet. Food Addit. Contam. 2019, 36, 1191–1205. [Google Scholar] [CrossRef]
- Henry, M.; Gasco, L.; Piccolo, G.; Fountoulaki, E. Review on the use of insects in the diet of farmed fish: Past and future. Anim. Feed Sci. Technol. 2015, 203, 1–22. [Google Scholar] [CrossRef]
- Sarker, P.K.; Kapuscinski, A.R.; Vandenberg, G.W.; Proulx, E.; Sitek, A.J. Towards sustainable and ocean-friendly aquafeeds: Evaluating a fish-free feed for rainbow trout (Oncorhynchus mykiss) using three marine microalgae species. Elem. Sci. Anthr. 2020, 8, 5. [Google Scholar] [CrossRef]
- Llewellyn, M.S.; Boutin, S.; Hoseinifar, S.H.; Derome, N. Teleost microbiomes: The state of the art in their characterization, manipulation and importance in aquaculture and fisheries. Front. Microbiol. 2014, 5, 207. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.R.; Ran, C.; Ringø, E.; Zhou, Z.G. Progress in fish gastrointestinal microbiota research. Rev. Aquac. 2018, 10, 626–640. [Google Scholar] [CrossRef]
- Daniel, H.; Gholami, A.M.; Berry, D.; Desmarchelier, C.; Hahne, H.; Loh, G.; Mondot, S.; Lepage, P.; Rothballer, M.; Walker, A. High-fat diet alters gut microbiota physiology in mice. ISME J. 2014, 8, 295–308. [Google Scholar] [CrossRef] [PubMed]
- Desai, A.R.; Links, M.G.; Collins, S.A.; Mansfield, G.S.; Drew, M.D.; Van Kessel, A.G.; Hill, J.E. Effects of plant-based diets on the distal gut microbiome of rainbow trout (Oncorhynchus mykiss). Aquaculture 2012, 350, 134–142. [Google Scholar] [CrossRef]
- Gajardo, K.; Jaramillo-Torres, A.; Kortner, T.M.; Merrifield, D.L.; Tinsley, J.; Bakke, A.M.; Krogdahl, Å. Alternative protein sources in the diet modulate microbiota and functionality in the distal intestine of Atlantic salmon (Salmo salar). Appl. Environ. Microbiol. 2017, 83, e02615–e02616. [Google Scholar] [CrossRef] [PubMed]
- Hartviksen, M.; Vecino, J.; Ringø, E.; Bakke, A.M.; Wadsworth, S.; Krogdahl, Å.; Ruohonen, K.; Kettunen, A. Alternative dietary protein sources for Atlantic salmon (Salmo salar L.) effect on intestinal microbiota, intestinal and liver histology and growth. Aquac. Nutr. 2014, 20, 381–398. [Google Scholar] [CrossRef]
- Huyben, D.; Nyman, A.; Vidaković, A.; Passoth, V.; Moccia, R.; Kiessling, A.; Dicksved, J.; Lundh, T. Effects of dietary inclusion of the yeasts Saccharomyces cerevisiae and Wickerhamomyces anomalus on gut microbiota of rainbow trout. Aquaculture 2017, 473, 528–537. [Google Scholar] [CrossRef]
- Huyben, D.; Sun, L.; Moccia, R.; Kiessling, A.; Dicksved, J.; Lundh, T. Dietary live yeast and increased water temperature influence the gut microbiota of rainbow trout. J. Appl. Microbiol. 2018, 124, 1377–1392. [Google Scholar] [CrossRef]
- Ingerslev, H.-C.; von Gersdorff Jørgensen, L.; Strube, M.L.; Larsen, N.; Dalsgaard, I.; Boye, M.; Madsen, L. The development of the gut microbiota in rainbow trout (Oncorhynchus mykiss) is affected by first feeding and diet type. Aquaculture 2014, 424, 24–34. [Google Scholar] [CrossRef]
- Antonopoulou, E.; Nikouli, E.; Piccolo, G.; Gasco, L.; Gai, F.; Chatzifotis, S.; Mente, E.; Kormas, K.A. Reshaping gut bacterial communities after dietary Tenebrio molitor larvae meal supplementation in three fish species. Aquaculture 2019, 503, 628–635. [Google Scholar] [CrossRef]
- Rimoldi, S.; Gini, E.; Iannini, F.; Gasco, L.; Terova, G. The effects of dietary insect meal from Hermetia illucens prepupae on autochthonous gut microbiota of rainbow trout (Oncorhynchus mykiss). Animals 2019, 9, 143. [Google Scholar] [CrossRef]
- Terova, G.; Rimoldi, S.; Ascione, C.; Gini, E.; Ceccotti, C.; Gasco, L. Rainbow trout (Oncorhynchus mykiss) gut microbiota is modulated by insect meal from Hermetia illucens prepupae in the diet. Rev. Fish Biol. Fish. 2019, 29, 465–486. [Google Scholar] [CrossRef]
- Alfiko, Y.; Xie, D.; Astuti, R.T.; Wong, J.; Wang, L. Insects as a feed ingredient for fish culture: Status and trends. Aquac. Fish. 2022, 7, 166–178. [Google Scholar] [CrossRef]
- Tran, H.Q.; Nguyen, T.T.; Prokešová, M.; Gebauer, T.; Doan, H.V.; Stejskal, V. Systematic review and meta-analysis of production performance of aquaculture species fed dietary insect meals. Rev. Aquac. 2022, 14, 1637–1655. [Google Scholar] [CrossRef]
- Makkar, H.P.; Tran, G.; Heuzé, V.; Ankers, P. State-of-the-art on use of insects as animal feed. Anim. Feed Sci. Technol. 2014, 197, 1–33. [Google Scholar] [CrossRef]
- Nogales-Mérida, S.; Gobbi, P.; Józefiak, D.; Mazurkiewicz, J.; Dudek, K.; Rawski, M.; Kierończyk, B.; Józefiak, A. Insect meals in fish nutrition. Rev. Aquac. 2019, 11, 1080–1103. [Google Scholar] [CrossRef]
- Barroso, F.G.; de Haro, C.; Sánchez-Muros, M.-J.; Venegas, E.; Martínez-Sánchez, A.; Pérez-Bañón, C. The potential of various insect species for use as food for fish. Aquaculture 2014, 422, 193–201. [Google Scholar] [CrossRef]
- Van Huis, A.; Oonincx, D.G. The environmental sustainability of insects as food and feed. A review. Agron. Sustain. Dev. 2017, 37, 43. [Google Scholar] [CrossRef]
- Lock, E.-J.; Biancarosa, I.; Gasco, L. Insects as raw materials in compound feed for aquaculture. In Edible Insects in Sustainable Food Systems; Springer: Berlin/Heidelberg, Germany, 2018; pp. 263–276. [Google Scholar]
- Gasco, L.; Gai, F.; Maricchiolo, G.; Genovese, L.; Ragonese, S.; Bottari, T.; Caruso, G.; Gasco, L.; Gai, F.; Maricchiolo, G. Fishmeal alternative protein sources for aquaculture feeds. In Feeds for the Aquaculture Sector: Current Situation and Alternative Sources; Springer: Berlin/Heidelberg, Germany, 2018; pp. 1–28. [Google Scholar]
- Rimoldi, S.; Antonini, M.; Gasco, L.; Moroni, F.; Terova, G. Intestinal microbial communities of rainbow trout (Oncorhynchus mykiss) may be improved by feeding a Hermetia illucens meal/low-fishmeal diet. Fish. Physiol. Biochem. 2021, 47, 365–380. [Google Scholar] [CrossRef] [PubMed]
- Rimoldi, S.; Ceccotti, C.; Brambilla, F.; Faccenda, F.; Antonini, M.; Terova, G. Potential of shrimp waste meal and insect exuviae as sustainable sources of chitin for fish feeds. Aquaculture 2023, 567, 739256. [Google Scholar] [CrossRef]
- Terova, G.; Ceccotti, C.; Ascione, C.; Gasco, L.; Rimoldi, S. Effects of partially defatted Hermetia illucens meal in rainbow trout diet on hepatic methionine metabolism. Animals 2020, 10, 1059. [Google Scholar] [CrossRef]
- Terova, G.; Gini, E.; Gasco, L.; Moroni, F.; Antonini, M.; Rimoldi, S. Effects of full replacement of dietary fishmeal with insect meal from Tenebrio molitor on rainbow trout gut and skin microbiota. J. Anim. Sci. Biotechnol. 2021, 12, 30. [Google Scholar] [CrossRef]
- Smetana, S.; Schmitt, E.; Mathys, A. Sustainable use of Hermetia illucens insect biomass for feed and food: Attributional and consequential life cycle assessment. Resour. Conserv. Recycl. 2019, 144, 285–296. [Google Scholar] [CrossRef]
- Gasco, L.; Acuti, G.; Bani, P.; Dalle Zotte, A.; Danieli, P.P.; De Angelis, A.; Fortina, R.; Marino, R.; Parisi, G.; Piccolo, G. Insect and fish by-products as sustainable alternatives to conventional animal proteins in animal nutrition. Ital. J. Anim. Sci. 2020, 19, 360–372. [Google Scholar] [CrossRef]
- Bruni, L.; Belghit, I.; Lock, E.J.; Secci, G.; Taiti, C.; Parisi, G. Total replacement of dietary fish meal with black soldier fly (Hermetia illucens) larvae does not impair physical, chemical or volatile composition of farmed Atlantic salmon (Salmo salar L.). J. Sci. Food Agric. 2020, 100, 1038–1047. [Google Scholar] [CrossRef]
- Bruni, L.; Pastorelli, R.; Viti, C.; Gasco, L.; Parisi, G. Characterisation of the intestinal microbial communities of rainbow trout (Oncorhynchus mykiss) fed with Hermetia illucens (black soldier fly) partially defatted larva meal as partial dietary protein source. Aquaculture 2018, 487, 56–63. [Google Scholar] [CrossRef]
- Magalhães, R.; Sánchez-López, A.; Leal, R.S.; Martínez-Llorens, S.; Oliva-Teles, A.; Peres, H. Black soldier fly (Hermetia illucens) pre-pupae meal as a fish meal replacement in diets for European seabass (Dicentrarchus labrax). Aquaculture 2017, 476, 79–85. [Google Scholar] [CrossRef]
- Bosi, A.; Banfi, D.; Moroni, F.; Ceccotti, C.; Giron, M.C.; Antonini, M.; Giaroni, C.; Terova, G. Effect of partial substitution of fishmeal with insect meal (Hermetia illucens) on gut neuromuscular function in Gilthead sea bream (Sparus aurata). Sci. Rep. 2021, 11, 21788. [Google Scholar] [CrossRef]
- Huyben, D.; Vidaković, A.; Hallgren, S.W.; Langeland, M. High-throughput sequencing of gut microbiota in rainbow trout (Oncorhynchus mykiss) fed larval and pre-pupae stages of black soldier fly (Hermetia illucens). Aquaculture 2019, 500, 485–491. [Google Scholar] [CrossRef]
- Butt, R.L.; Volkoff, H. Gut microbiota and energy homeostasis in fish. Front. Endocrinol. 2019, 10, 9. [Google Scholar] [CrossRef]
- Etyemez, M.; Balcázar, J.L. Bacterial community structure in the intestinal ecosystem of rainbow trout (Oncorhynchus mykiss) as revealed by pyrosequencing-based analysis of 16S rRNA genes. Res. Vet. Sci. 2015, 100, 8–11. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Brunt, J.; Austin, B. Microbial diversity of intestinal contents and mucus in rainbow trout (Oncorhynchus mykiss). J. Appl. Microbiol. 2007, 102, 1654–1664. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wang, J.; Lu, K.; Song, K.; Mai, K.; Zhang, C.; Rahimnejad, S. Total replacement of fish meal with soybean meal in diets for bullfrog (Lithobates catesbeianus): Effects on growth performance and gut microbial composition. Aquaculture 2020, 524, 735236. [Google Scholar] [CrossRef]
- Karlsen, C.; Tzimorotas, D.; Robertsen, E.M.; Kirste, K.H.; Bogevik, A.S.; Rud, I. Feed microbiome: Confounding factor affecting fish gut microbiome studies. ISME Commun. 2022, 2, 14. [Google Scholar] [CrossRef]
- Ringø, E.; Zhou, Z.; Olsen, R.; Song, S. Use of chitin and krill in aquaculture–the effect on gut microbiota and the immune system: A review. Aquac. Nutr. 2012, 18, 117–131. [Google Scholar] [CrossRef]
- Askarian, F.; Zhou, Z.; Olsen, R.E.; Sperstad, S.; Ringø, E. Culturable autochthonous gut bacteria in Atlantic salmon (Salmo salar L.) fed diets with or without chitin. Characterization by 16S rRNA gene sequencing, ability to produce enzymes and in vitro growth inhibition of four fish pathogens. Aquaculture 2012, 326, 1–8. [Google Scholar] [CrossRef]
- Ahmed, F.; Soliman, F.M.; Adly, M.A.; Soliman, H.A.; El-Matbouli, M.; Saleh, M. Dietary chitosan nanoparticles: Potential role in modulation of rainbow trout (Oncorhynchus mykiss) antibacterial defense and intestinal immunity against enteric redmouth disease. Mar. Drugs 2021, 19, 72. [Google Scholar] [CrossRef]
- Dawood, M.A.; Gewaily, M.S.; Soliman, A.A.; Shukry, M.; Amer, A.A.; Younis, E.M.; Abdel-Warith, A.-W.A.; Van Doan, H.; Saad, A.H.; Aboubakr, M. Marine-derived chitosan nanoparticles improved the intestinal histo-morphometrical features in association with the health and immune response of grey mullet (Liza ramada). Mar. Drugs 2020, 18, 611. [Google Scholar] [CrossRef]
- Ghanbari, M.; Kneifel, W.; Domig, K.J. A new view of the fish gut microbiome: Advances from next-generation sequencing. Aquaculture 2015, 448, 464–475. [Google Scholar] [CrossRef]
- Apper, E.; Weissman, D.; Respondek, F.; Guyonvarch, A.; Baron, F.; Boisot, P.; Rodiles, A.; Merrifield, D. Hydrolysed wheat gluten as part of a diet based on animal and plant proteins supports good growth performance of Asian seabass (Lates calcarifer), without impairing intestinal morphology or microbiota. Aquaculture 2016, 453, 40–48. [Google Scholar] [CrossRef]
- Dailey, F.E.; McGraw, J.E.; Jensen, B.J.; Bishop, S.S.; Lokken, J.P.; Dorff, K.J.; Ripley, M.P.; Munro, J.B. The microbiota of freshwater fish and freshwater niches contain omega-3 fatty acid-producing Shewanella species. Appl. Environ. Microbiol. 2016, 82, 218–231. [Google Scholar] [CrossRef]
- Nawaz, A.; Irshad, S.; Hoseinifar, S.H.; Xiong, H. The functionality of prebiotics as immunostimulant: Evidences from trials on terrestrial and aquatic animals. Fish Shellfish. Immunol. 2018, 76, 272–278. [Google Scholar] [CrossRef] [PubMed]
- Fogarty, C.; Burgess, C.M.; Cotter, P.D.; Cabrera-Rubio, R.; Whyte, P.; Smyth, C.; Bolton, D.J. Diversity and composition of the gut microbiota of Atlantic salmon (Salmo salar) farmed in Irish waters. J. Appl. Microbiol. 2019, 127, 648–657. [Google Scholar] [CrossRef] [PubMed]
- Lyons, P.; Turnbull, J.; Dawson, K.A.; Crumlish, M. Phylogenetic and functional characterization of the distal intestinal microbiome of rainbow trout Oncorhynchus mykiss from both farm and aquarium settings. J. Appl. Microbiol. 2017, 122, 347–363. [Google Scholar] [CrossRef]
- Rasmussen, J.A.; Villumsen, K.R.; Duchêne, D.A.; Puetz, L.C.; Delmont, T.O.; Sveier, H.; Jørgensen, L.v.G.; Præbel, K.; Martin, M.D.; Bojesen, A.M. Genome-resolved metagenomics suggests a mutualistic relationship between Mycoplasma and salmonid hosts. Commun. Biol. 2021, 4, 579. [Google Scholar] [CrossRef] [PubMed]
- Melo-Bolívar, J.F.; Ruiz Pardo, R.Y.; Hume, M.E.; Nisbet, D.J.; Rodriguez-Villamizar, F.; Alzate, J.F.; Junca, H.; Villamil Díaz, L.M. Establishment and characterization of a competitive exclusion bacterial culture derived from Nile tilapia (Oreochromis niloticus) gut microbiomes showing antibacterial activity against pathogenic Streptococcus agalactiae. PLoS ONE 2019, 14, e0215375. [Google Scholar] [CrossRef]
- Brown, R.M.; Wiens, G.D.; Salinas, I. Analysis of the gut and gill microbiome of resistant and susceptible lines of rainbow trout (Oncorhynchus mykiss). Fish Shellfish. Immunol. 2019, 86, 497–506. [Google Scholar] [CrossRef]
- Zhang, J.; Dong, Y.; Song, K.; Wang, L.; Li, X.; Tan, B.; Lu, K.; Zhang, C. Effects of the Replacement of Dietary Fish Meal with Defatted Yellow Mealworm (Tenebrio molitor) on Juvenile Large Yellow Croakers (Larimichthys crocea) Growth and Gut Health. Animals 2022, 12, 2659. [Google Scholar] [CrossRef]
- Benzertiha, A.; Kierończyk, B.; Kołodziejski, P.; Pruszyńska–Oszmałek, E.; Rawski, M.; Józefiak, D.; Józefiak, A. Tenebrio molitor and Zophobas morio full-fat meals as functional feed additives affect broiler chickens’ growth performance and immune system traits. Poult. Sci. 2020, 99, 196–206. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.; Yu, J.S.; Parker, B.L.; Skinner, M.; Je, Y.H.; Kim, J.S. Production of antibacterial Bombyx mori cecropin A in mealworm-pathogenic Beauveria bassiana ERL1170. J. Ind. Microbiol. Biotechnol. 2015, 42, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Sankian, Z.; Khosravi, S.; Kim, Y.-O.; Lee, S.-M. Effects of dietary inclusion of yellow mealworm (Tenebrio molitor) meal on growth performance, feed utilization, body composition, plasma biochemical indices, selected immune parameters and antioxidant enzyme activities of mandarin fish (Siniperca scherzeri) juveniles. Aquaculture 2018, 496, 79–87. [Google Scholar]
- Ido, A.; Hashizume, A.; Ohta, T.; Takahashi, T.; Miura, C.; Miura, T. Replacement of fish meal by defatted yellow mealworm (Tenebrio molitor) larvae in diet improves growth performance and disease resistance in red seabream (Pargus major). Animals 2019, 9, 100. [Google Scholar] [CrossRef]
- Fontes, T.V.; de Oliveira, K.R.B.; Gomes Almeida, I.L.; Orlando, T.M.; Rodrigues, P.B.; da Costa, D.V.; Rosa, P.V.E. Digestibility of insect meals for Nile tilapia fingerlings. Animals 2019, 9, 181. [Google Scholar] [CrossRef]
- Belforti, M.; Gai, F.; Lussiana, C.; Renna, M.; Malfatto, V.; Rotolo, L.; De Marco, M.; Dabbou, S.; Schiavone, A.; Zoccarato, I. Tenebrio molitor meal in rainbow trout (Oncorhynchus mykiss) diets: Effects on animal performance, nutrient digestibility and chemical composition of fillets. Ital. J. Anim. Sci. 2015, 14, 4170. [Google Scholar] [CrossRef]
- Piccolo, G.; Iaconisi, V.; Marono, S.; Gasco, L.; Loponte, R.; Nizza, S.; Bovera, F.; Parisi, G. Effect of Tenebrio molitor larvae meal on growth performance, in vivo nutrients digestibility, somatic and marketable indexes of gilthead sea bream (Sparus aurata). Anim. Feed Sci. Technol. 2017, 226, 12–20. [Google Scholar] [CrossRef]
- Gasco, L.; Henry, M.; Piccolo, G.; Marono, S.; Gai, F.; Renna, M.; Lussiana, C.; Antonopoulou, E.; Mola, P.; Chatzifotis, S. Tenebrio molitor meal in diets for European sea bass (Dicentrarchus labrax L.) juveniles: Growth performance, whole body composition and in vivo apparent digestibility. Anim. Feed Sci. Technol. 2016, 220, 34–45. [Google Scholar] [CrossRef]
- Sánchez-Muros, M.-J.; Barroso, F.G.; Manzano-Agugliaro, F. Insect meal as renewable source of food for animal feeding: A review. J. Clean. Prod. 2014, 65, 16–27. [Google Scholar] [CrossRef]
- Ganguly, S.; Prasad, A. Microflora in fish digestive tract plays significant role in digestion and metabolism. Rev. Fish Biol. Fish. 2012, 22, 11–16. [Google Scholar] [CrossRef]
- Józefiak, A.; Nogales-Mérida, S.; Mikołajczak, Z.; Rawski, M.; Kierończyk, B.; Mazurkiewicz, J. The Utilization of Full-Fat Insect Meal in Rainbow Trout (Oncorhynchus mykiss) Nutrition: The Effects on Growth Performance, Intestinal Microbiota and Gastrointestinal Tract Histomorphology. Ann. Anim. Sci. 2019, 19, 747–765. [Google Scholar] [CrossRef]
- Mikołajczak, Z.; Rawski, M.; Mazurkiewicz, J.; Kierończyk, B.; Józefiak, D. The effect of hydrolyzed insect meals in sea trout fingerling (Salmo trutta m. trutta) diets on growth performance, microbiota and biochemical blood parameters. Animals 2020, 10, 1031. [Google Scholar] [CrossRef]
- Gudiña, E.J.; Fernandes, E.C.; Rodrigues, A.I.; Teixeira, J.A.; Rodrigues, L.R. Biosurfactant production by Bacillus subtilis using corn steep liquor as culture medium. Front. Microbiol. 2015, 6, 59. [Google Scholar]
- Ringø, E.; Hoseinifar, S.H.; Ghosh, K.; Doan, H.V.; Beck, B.R.; Song, S.K. Lactic acid bacteria in finfish—An update. Front. Microbiol. 2018, 9, 1818. [Google Scholar] [CrossRef]
- Gaudioso, G.; Marzorati, G.; Faccenda, F.; Weil, T.; Lunelli, F.; Cardinaletti, G.; Marino, G.; Olivotto, I.; Parisi, G.; Tibaldi, E. Processed animal proteins from insect and poultry by-products in a fish meal-free diet for rainbow trout: Impact on intestinal microbiota and inflammatory markers. Int. J. Mol. Sci. 2021, 22, 5454. [Google Scholar] [CrossRef]
- Bajagai, Y.S.; Klieve, A.V.; Dart, P.J.; Bryden, W.L. Probiotics in Animal Nutrition: Production, Impact and Regulation; FAO: Rome, Italy, 2016. [Google Scholar]
- Kiron, V. Fish immune system and its nutritional modulation for preventive health care. Anim. Feed Sci. Technol. 2012, 173, 111–133. [Google Scholar] [CrossRef]
- Sugimura, Y.; Hagi, T.; Hoshino, T. Correlation between in vitro mucus adhesion and the in vivo colonization ability of lactic acid bacteria: Screening of new candidate carp probiotics. Biosci. Biotechnol. Biochem. 2011, 75, 511–515. [Google Scholar] [CrossRef]
- Pérez-Sánchez, T.; Balcázar, J.L.; Merrifield, D.L.; Carnevali, O.; Gioacchini, G.; de Blas, I.; Ruiz-Zarzuela, I. Expression of immune-related genes in rainbow trout (Oncorhynchus mykiss) induced by probiotic bacteria during Lactococcus garvieae infection. Fish Shellfish. Immunol. 2011, 31, 196–201. [Google Scholar] [CrossRef]
- Dahiya, T. Applications and possible modes of action of probiotics in aquaculture. Int. J. Agric. Sci. 2020, 12, 9753–9755. [Google Scholar]
- Newaj-Fyzul, A.; Al-Harbi, A.; Austin, B. Developments in the use of probiotics for disease control in aquaculture. Aquaculture 2014, 431, 1–11. [Google Scholar] [CrossRef]
- Adel, M.; El-Sayed, A.-F.M.; Yeganeh, S.; Dadar, M.; Giri, S.S. Effect of potential probiotic Lactococcus lactis subsp. lactis on growth performance, intestinal microbiota, digestive enzyme activities, and disease resistance of Litopenaeus vannamei. Probiotics Antimicrob. 2017, 9, 150–156. [Google Scholar] [CrossRef] [PubMed]
- Adel, M.; Yeganeh, S.; Dawood, M.; Safari, R.; Radhakrishnan, S. Effects of Pediococcus pentosaceus supplementation on growth performance, intestinal microflora and disease resistance of white shrimp, Litopenaeus vannamei. Aquac. Nutr. 2017, 23, 1401–1409. [Google Scholar] [CrossRef]
- Encarnação, P. Functional feed additives in aquaculture feeds. In Aquafeed Formulation; Elsevier: Amsterdam, The Netherlands, 2016; pp. 217–237. [Google Scholar]
- Cabello, F.C. Heavy use of prophylactic antibiotics in aquaculture: A growing problem for human and animal health and for the environment. Environ. Microbiol. 2006, 8, 1137–1144. [Google Scholar] [CrossRef] [PubMed]
- Borch, K.; Pederson, I.; Hogmo, R.O. The use of probiotics in fish feed for intensive aquaculture to promote healthy guts. Adv. Aquac. Fish. Manag. 2015, 3, 264–273. [Google Scholar]
- Casewell, M.; Friis, C.; Marco, E.; McMullin, P.; Phillips, I. The European ban on growth-promoting antibiotics and emerging consequences for human and animal health. J. Antimicrob. Chemother. 2003, 52, 159–161. [Google Scholar] [CrossRef]
- European Union. Regulation (EU) 2019/6 of the European Parliament and of the Council of 11 December 2018 on veterinary medicinal products and repealing Directive 2001/82/EC. Off. J. Eur. Union 2019, 4, 43–167. [Google Scholar]
- Ezema, C. Probiotics in animal production: A review. J. Vet. Med. Anim. Health 2013, 5, 308–316. [Google Scholar]
- Dimitroglou, A.; Merrifield, D.L.; Carnevali, O.; Picchietti, S.; Avella, M.; Daniels, C.; Güroy, D.; Davies, S.J. Microbial manipulations to improve fish health and production–a Mediterranean perspective. Fish Shellfish. Immunol. 2011, 30, 1–16. [Google Scholar] [CrossRef]
- Merrifield, D.; Bradley, G.; Baker, R.; Davies, S. Probiotic applications for rainbow trout (Oncorhynchus mykiss Walbaum) II. Effects on growth performance, feed utilization, intestinal microbiota and related health criteria postantibiotic treatment. Aquac. Nutr. 2010, 16, 496–503. [Google Scholar] [CrossRef]
- Chauhan, A.; Singh, R. Probiotics in aquaculture: A promising emerging alternative approach. Symbiosis 2019, 77, 99–113. [Google Scholar] [CrossRef]
- Ridha, M.T.; Azad, I.S. Preliminary evaluation of growth performance and immune response of Nile tilapia Oreochromis niloticus supplemented with two putative probiotic bacteria. Aquac. Res. 2012, 43, 843–852. [Google Scholar] [CrossRef]
- Moroni, F.; Naya-Català, F.; Piazzon, M.C.; Rimoldi, S.; Calduch-Giner, J.; Giardini, A.; Martínez, I.; Brambilla, F.; Pérez-Sánchez, J.; Terova, G. The effects of nisin-producing Lactococcus lactis strain used as probiotic on gilthead sea bream (Sparus aurata) growth, gut microbiota, and transcriptional response. Front. Mar. Sci. 2021, 8, 659519. [Google Scholar] [CrossRef]
- Cerezuela, R.; Fumanal, M.; Tapia-Paniagua, S.T.; Meseguer, J.; Moriñigo, M.Á.; Esteban, M.Á. Changes in intestinal morphology and microbiota caused by dietary administration of inulin and Bacillus subtilis in gilthead sea bream (Sparus aurata L.) specimens. Fish Shellfish. Immunol. 2013, 34, 1063–1070. [Google Scholar] [CrossRef]
- Feng, J.; Chang, X.; Zhang, Y.; Yan, X.; Zhang, J.; Nie, G. Effects of Lactococcus lactis from Cyprinus carpio L. as probiotics on growth performance, innate immune response and disease resistance against Aeromonas hydrophila. Fish Shellfish. Immunol. 2019, 93, 73–81. [Google Scholar] [CrossRef]
- Heo, W.-S.; Kim, Y.-R.; Kim, E.-Y.; Bai, S.C.; Kong, I.-S. Effects of dietary probiotic, Lactococcus lactis subsp. lactis I2, supplementation on the growth and immune response of olive flounder (Paralichthys olivaceus). Aquaculture 2013, 376, 20–24. [Google Scholar] [CrossRef]
- Xia, Y.; Lu, M.; Chen, G.; Cao, J.; Gao, F.; Wang, M.; Liu, Z.; Zhang, D.; Zhu, H.; Yi, M. Effects of dietary Lactobacillus rhamnosus JCM1136 and Lactococcus lactis subsp. lactis JCM5805 on the growth, intestinal microbiota, morphology, immune response and disease resistance of juvenile Nile tilapia, Oreochromis niloticus. Fish Shellfish. Immunol. 2018, 76, 368–379. [Google Scholar] [CrossRef]
- Kim, S.-K.; Bhatnagar, I.; Kang, K.-H. Development of marine probiotics: Prospects and approach. Adv. Food. Nutr. 2012, 65, 353–362. [Google Scholar]
- Li, Q.; Montalban-Lopez, M.; Kuipers, O.P. Increasing the antimicrobial activity of nisin-based lantibiotics against Gram-negative pathogens. Appl. Environ. Microbiol. 2018, 84, e00052-18. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Fečkaninová, A.; Lokesh, J.; Koščová, J.; Sørensen, M.; Fernandes, J.; Kiron, V. Lactobacillus dominate in the intestine of Atlantic salmon fed dietary probiotics. Front. Microbiol. 2019, 9, 3247. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Ni, J.; Li, J.; Wang, C.; Li, X.; Wu, S.; Zhang, T.; Yu, Y.; Yan, Q. Comparative study on gastrointestinal microbiota of eight fish species with different feeding habits. J. Appl. Microbiol. 2014, 117, 1750–1760. [Google Scholar] [CrossRef]
- Rimoldi, S.; Gini, E.; Koch, J.; Iannini, F.; Brambilla, F.; Terova, G. Effects of hydrolyzed fish protein and autolyzed yeast as substitutes of fishmeal in the gilthead sea bream (Sparus aurata) diet, on fish intestinal microbiome. BMC Vet. Res. 2020, 16, 118. [Google Scholar]
- Cerezuela, R.; Fumanal, M.; Tapia-Paniagua, S.T.; Meseguer, J.; Moriñigo, M.Á.; Esteban, M.Á. Histological alterations and microbial ecology of the intestine in gilthead seabream (Sparus aurata L.) fed dietary probiotics and microalgae. Cell Tissue Res. 2012, 350, 477–489. [Google Scholar] [CrossRef]
- Collado, M.; Meriluoto, J.; Salminen, S. Role of commercial probiotic strains against human pathogen adhesion to intestinal mucus. Lett. Appl. Microbiol. 2007, 45, 454–460. [Google Scholar] [CrossRef]
- Luan, Y.; Li, M.; Zhou, W.; Yao, Y.; Yang, Y.; Zhang, Z.; Ringø, E.; Olsen, R.E.; Clarke, J.L.; Xie, S. The Fish Microbiota: Research Progress and Potential Applications. Engineering 2023. [Google Scholar] [CrossRef]
- Kristiansen, M.; Ringø, E. Evaluation of prebiotic and probiotic effects on the intestinal gut microbiota and histology of Atlantic salmon (Salmo salar L.). J. Aquac. Res. Dev. 2011, S1, 9. [Google Scholar]
- Løvmo Martinsen, L.; Salma, W.; Myklebust, R.; Mayhew, T.M.; Ringø, E. Carnobacterium maltaromaticum vs. Vibrio (Listonella) anguillarum in the midgut of Atlantic cod (Gadus morhua L.): An ex vivo study. Aquac. Res. 2011, 42, 1830–1839. [Google Scholar] [CrossRef]
- Ringø, E.; Løvmo, L.; Kristiansen, M.; Bakken, Y.; Salinas, I.; Myklebust, R.; Olsen, R.E.; Mayhew, T.M. Lactic acid bacteria vs. pathogens in the gastrointestinal tract of fish: A review. Aquac. Res. 2010, 41, 451–467. [Google Scholar] [CrossRef]
- Chen, T.; Wang, M.; Jiang, S.; Xiong, S.; Wei, H. The application of polymerase chain reaction-denaturing gradient gel electrophoresis (PCR-DGGE) method in microbial screening. Afr. J. Biotechnol. 2011, 10, 9387–9395. [Google Scholar]
- Salma, W.; Zhou, Z.; Wang, W.; Askarian, F.; Kousha, A.; Ebrahimi, M.T.; Myklebust, R.; Ringø, E. Histological and bacteriological changes in intestine of beluga (Huso huso) following ex vivo exposure to bacterial strains. Aquaculture 2011, 314, 24–33. [Google Scholar] [CrossRef]
- de Araújo, J.C.; Schneider, R.P. DGGE with genomic DNA: Suitable for detection of numerically important organisms but not for identification of the most abundant organisms. Water Res. 2008, 42, 5002–5010. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Zhou, Z.; Liu, Y.; Cao, Y.; Meng, K.; Shi, P.; Yao, B.; Ringø, E. Effects of the antibiotic growth promoters flavomycin and florfenicol on the autochthonous intestinal microbiota of hybrid tilapia (Oreochromis niloticus♀ × O. aureus♂). Arch. Microbiol. 2010, 192, 985–994. [Google Scholar] [CrossRef] [PubMed]
- Ringø, E.; Zhou, Z.; Vecino, J.G.; Wadsworth, S.; Romero, J.; Krogdahl, Å.; Olsen, R.E.; Dimitroglou, A.; Foey, A.; Davies, S. Effect of dietary components on the gut microbiota of aquatic animals. A never-ending story? Aquac. Nutr. 2016, 22, 219–282. [Google Scholar] [CrossRef]
- Handelsman, J.; Rondon, M.R.; Brady, S.F.; Clardy, J.; Goodman, R.M. Molecular biological access to the chemistry of unknown soil microbes: A new frontier for natural products. Chem. Biol. 1998, 5, R245–R249. [Google Scholar] [CrossRef]
- Riesenfeld, C.S.; Schloss, P.D.; Handelsman, J. Metagenomics: Genomic analysis of microbial communities. Annu. Rev. Genet. 2004, 38, 525–552. [Google Scholar] [CrossRef]
- Clements, K.D.; Pasch, I.B.; Moran, D.; Turner, S.J. Clostridia dominate 16S rRNA gene libraries prepared from the hindgut of temperate marine herbivorous fishes. Mar. Biol. 2007, 150, 1431–1440. [Google Scholar] [CrossRef]
- Gao, X.; Wang, P.; Tang, Y. Engineered polyketide biosynthesis and biocatalysis in Escherichia coli. Appl. Microbiol. Biotechnol. 2010, 88, 1233–1242. [Google Scholar] [CrossRef]
- Vasquez, R.; Oh, J.K.; Song, J.H.; Kang, D.-K. Gut microbiome-produced metabolites in pigs: A review on their biological functions and the influence of probiotics. J. Anim. Sci. Technol. 2022, 64, 671–695. [Google Scholar] [CrossRef]
- Cui, X.; Zhang, Q.; Zhang, Q.; Zhang, Y.; Chen, H.; Liu, G.; Zhu, L. Research progress of the Gut microbiome in hybrid fish. Microorganisms 2022, 10, 891. [Google Scholar] [CrossRef]
- Diwan, A.D.; Harke, S.N.; Panche, A.N. Aquaculture industry prospective from gut microbiome of fish and shellfish: An overview. J. Anim. Physiol. Anim. Nutr. 2022, 106, 441–469. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).