Chronic Intestinal Disorders in Humans and Pets: Current Management and the Potential of Nutraceutical Antioxidants as Alternatives
Abstract
:Simple Summary
Abstract
1. Introduction
2. Oxidative Stress-Induced Damage in Chronic Intestinal Disorders
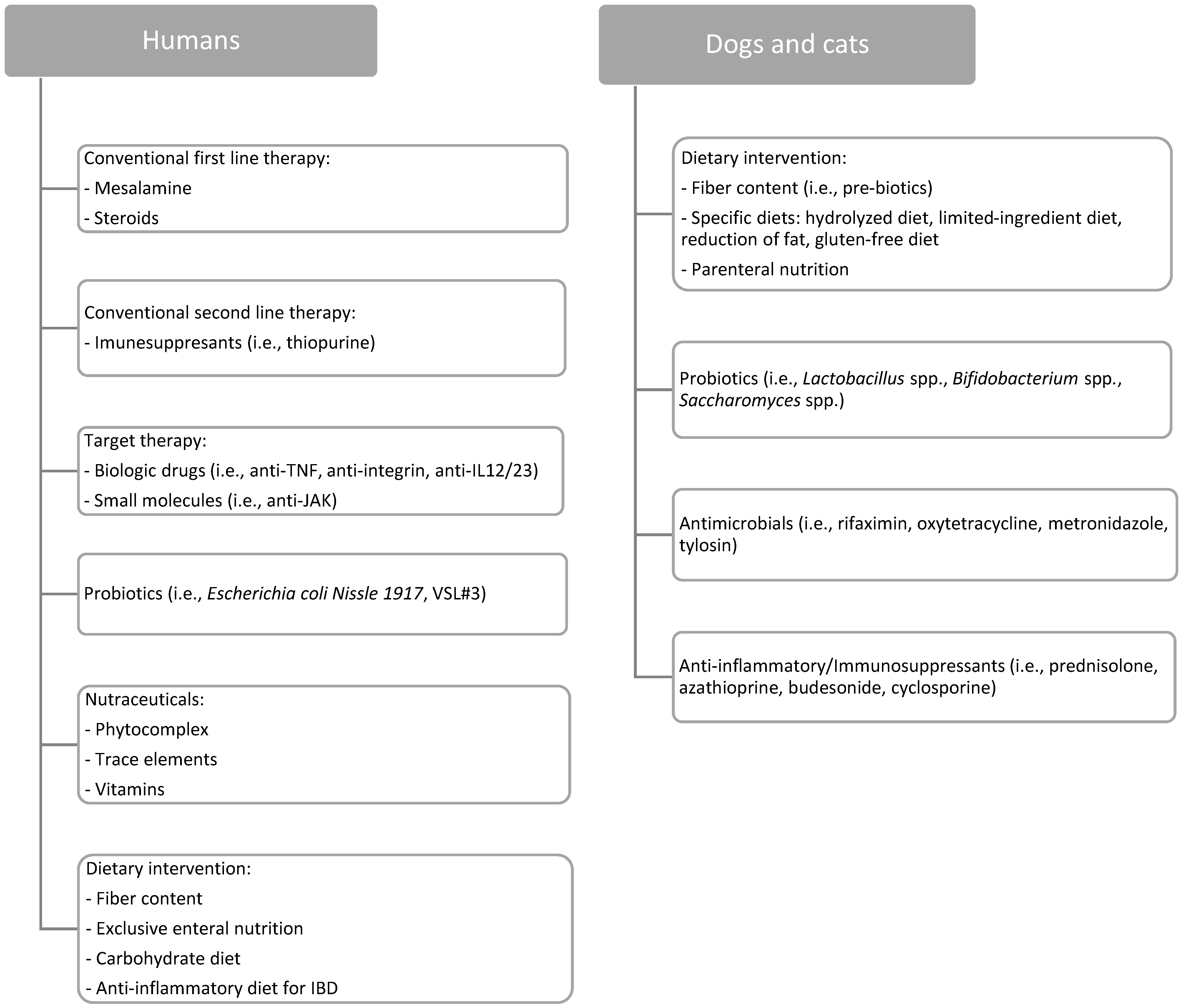
3. Therapeutic Management of Chronic Intestinal Disorders
3.1. Humans
3.1.1. Conventional Therapy
3.1.2. Advanced Therapy
3.2. Dogs and Cats
Conventional Therapy
4. Dietary Interventions in Chronic Intestinal Disorders
4.1. Humans
4.1.1. Fiber Content
4.1.2. Specific Diets
Exclusive Enteral Nutrition (EEN)
Carbohydrate Diet (SCD)
Anti-Inflammatory Diet for IBD (IBD-AID)
4.2. Dogs and Cats
4.2.1. Fiber Content
4.2.2. Specific Diets
Hydrolyzed Diets
Limited-Ingredient Diets
Fat Reduced Diets
Gluten-Free Diets
Parenteral Nutrition (PN)
5. Role of Nutraceuticals as Antioxidants in Chronic Intestinal Disorders: Phytocomplex, Trace Elements, and Vitamins
5.1. Phytocomplex
5.1.1. Curcuma longa
5.1.2. Aloe vera
5.1.3. Boswellia serrata
5.1.4. Triticum aestivum
5.1.5. Plantago spp.
5.1.6. Serpylli herba
5.1.7. Vaccinium myrtillus
5.1.8. Camellia sinensis
5.1.9. Citrus
5.1.10. Pomegranate
5.2. Trace Elements
5.2.1. Zinc
5.2.2. Selenium
5.3. Vitamins
5.3.1. Vitamin A
5.3.2. Vitamin E
5.3.3. Vitamin C
| Nutraceutical | Human Trial [Reference] | Proved Effectiveness in Human CID Patients |
|---|---|---|
| Phytocomplex | ||
| Curcuma longa | Yes [62] | More effective than placebo in keeping human patients with quiescent UC in remission |
| Aloe vera | Yes [70] | Produces clinical response more frequently than placebo and reduction in histological disease activity |
| Boswellia serrata | Yes [73,74] | Maintains remission over a prolonged period in UC patients |
| Triticum aestivum | Yes [78] | Efficient treatment for active distal UC as a single or adjuvant treatment |
| Plantago ovata | Yes [82] | Its seeds have been shown to be as effective as mesalazine in preventing UC relapse |
| Serpylli herba | no | NA |
| Vaccinium myrtillus | Yes [86] | Endoscopic and histologic disease activity, as well as fecal calprotectin levels, were considerably reduced in UC |
| Camellia sinensis | no | NA |
| Citrus | no | NA |
| Trace elements | ||
| Zinc | no | NA |
| Selenium | no | NA |
| Vitamins | ||
| Vitamin A | no | NA |
| Vitamin E | Yes [130] | Enemas containing vitamin E reduced the disease symptoms of mild and moderately active UC |
| Vitamin C | no | NA |
6. Concluding Remarks
Author Contributions
Funding
Conflicts of Interest
References
- Actis, G.C.; Pellicano, R.; Fagoonee, S.; Ribaldone, D.G. History of inflammatory bowel diseases. J. Clin. Med. 2019, 8, 1970. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Actis, G.C.; Rosina, F. Inflammatory bowel disease: An archetype disorder of outer environment sensor systems. World J. Gastrointest. Pharmacol. Ther. 2013, 4, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Ho, G.T.; Porter, R.J.; Kalla, R. Ulcerative colitis: Recent advances in the understanding of disease pathogenesis. F1000Research 2020, 9, Rev-294. [Google Scholar]
- De Souza, H.S.P.; Fiocchi, C.; Iliopoulos, D. The IBD interactome: An integrated view of aetiology, pathogenesis and therapy. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 739–749. [Google Scholar] [CrossRef] [PubMed]
- Dandrieux, J.R.S.; Mansfield, C.S. Chronic Enteropathy In Canines: Prevalence, Impact And Management Strategies. Vet. Med. 2019, 10, 203–214. [Google Scholar] [CrossRef] [Green Version]
- Hemida, M.; Vuori, K.A.; Moore, R.; Anturaniemi, J.; Hielm-Björkman, A. Early Life Modifiable Exposures and Their Association With Owner Reported Inflammatory Bowel Disease Symptoms in Adult Dogs. Front. Vet. Sci. 2021, 8, 552350. [Google Scholar] [CrossRef]
- Cerquetella, M.; Spaterna, A.; Laus, F.; Tesei, B.; Rossi, G.; Antonelli, E.; Villanacci, V.; Bassotti, G. Inflammatory bowel disease in the dog: Differences and similarities with humans. World J. Gastroenterol. 2010, 16, 1050–1056. [Google Scholar] [CrossRef]
- Jergens Albert, E. Inflammatory bowel disease in veterinary medicine. Front. Biosci. 2012, E4, 1404. [Google Scholar] [CrossRef]
- Makielski, K.; Cullen, J.; O’Connor, A.; Jergens, A.E. Narrative review of therapies for chronic enteropathies in dogs and cats. J. Vet. Intern. Med. 2019, 33, 11–22. [Google Scholar] [CrossRef] [Green Version]
- Allenspach, K. Clinical immunology and immunopathology of the canine and feline intestine. Vet. Clin. N. Am. Small Anim. Pract. 2011, 41, 345–360. [Google Scholar] [CrossRef]
- Kathrani, A.; Werling, D.; Allenspach, K. Canine breeds at high risk of developing inflammatory bowel disease in the south-eastern UK. Vet. Rec. 2011, 169, 635. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kathrani, A. Dietary and Nutritional Approaches to the Management of Chronic Enteropathy in Dogs and Cats. Vet. Clin. N. Am. Small Anim. Pract. 2021, 51, 123–136. [Google Scholar] [CrossRef] [PubMed]
- Pizzino, G.; Irrera, N.; Cucinotta, M.; Pallio, G.; Mannino, F.; Arcoraci, V.; Squadrito, F.; Altavilla, D.; Bitto, A. Oxidative Stress: Harms and Benefits for Human Health. Oxid. Med. Cell. Longev. 2017, 2017, 8416763. [Google Scholar] [CrossRef] [PubMed]
- Moret-Tatay, I.; Iborra, M.; Cerrillo, E.; Tortosa, L.; Nos, P.; Beltrán, B. Possible biomarkers in blood for Crohn’s disease: Oxidative stress and microRNAs—Current evidences and further aspects to unravel. Oxid. Med. Cell. Longev. 2016, 2016, 2325162. [Google Scholar] [CrossRef] [Green Version]
- Balmus, I.; Ciobica, A.; Trifan, A.; Stanciu, C. The implications of oxidative stress and antioxidant therapies in Inflammatory Bowel Disease: Clinical aspects and animal models. Saudi J. Gastroenterol. 2016, 22, 3–17. [Google Scholar] [CrossRef]
- Zhu, H.; Li, Y.R. Oxidative stress and redox signaling mechanisms of inflammatory bowel disease: Updated experimental and clinical evidence. Exp. Biol. Med. 2012, 237, 474–480. [Google Scholar] [CrossRef]
- Minamoto, Y.; Otoni, C.C.; Steelman, S.M.; Büyükleblebici, O.; Steiner, J.M.; Jergens, A.E.; Suchodolski, J.S. Alteration of the fecal microbiota and serum metabolite profiles in dogs with idiopathic inflammatory bowel disease. Gut Microbes 2015, 6, 33–47. [Google Scholar] [CrossRef] [Green Version]
- Gionchetti, P.; Dignass, A.; Danese, S.; Magro Dias, F.J.; Rogler, G.; Lakatos, P.L.; Adamina, M.; Ardizzone, S.; Buskens, C.J.; Sebastian, S.; et al. 3rd European Evidence-based Consensus on the Diagnosis and Management of Crohn’s Disease 2016: Part 2: Surgical Management and Special Situations. J. Crohn’s Colitis 2017, 11, 135–149. [Google Scholar] [CrossRef] [Green Version]
- Harbord, M.; Eliakim, R.; Bettenworth, D.; Karmiris, K.; Katsanos, K.; Kopylov, U.; Kucharzik, T.; Molnár, T.; Raine, T.; Sebastian, S.; et al. Third European evidence-based consensus on diagnosis and management of ulcerative colitis. Part 2: Current management. J. Crohn’s Colitis 2017, 11, 769–784. [Google Scholar] [CrossRef] [Green Version]
- Lichtenstein, G.R.; Loftus, E.V.; Isaacs, K.L.; Regueiro, M.D.; Gerson, L.B.; Sands, B.E. ACG Clinical Guideline: Management of Crohn’s Disease in Adults. Am. J. Gastroenterol. 2018, 113, 481–517. [Google Scholar] [CrossRef]
- Criscuoli, V.; Modesto, I.; Orlando, A.; Cottone, M. Mesalazine for the treatment of inflammatory bowel disease. Expert Opin. Pharmacother. 2013, 14, 1669–1678. [Google Scholar] [CrossRef] [PubMed]
- Salice, M.; Rizzello, F.; Calabrese, C.; Privitera Hrustemovic, H.; Gionchetti, P. Budesonide MMX: Efficacy and safety profile in the treatment of ulcerative colitis. Expert Rev. Gastroenterol. Hepatol. 2019, 13, 607–613. [Google Scholar] [CrossRef] [PubMed]
- Cushing, K.; Higgins, P.D.R. Management of Crohn Disease: A Review. JAMA J. Am. Med. Assoc. 2021, 325, 69–80. [Google Scholar] [CrossRef] [PubMed]
- Actis, G.C.; Pellicano, R.; Ribaldone, D.G. A concise history of thiopurines for inflammatory bowel disease: From anecdotal reporting to treat-to-target algorithms. Rev. Recent Clin. Trials 2019, 14, 4–9. [Google Scholar] [CrossRef]
- Saettone, V.; Biasato, I.; Radice, E.; Schiavone, A.; Bergero, D.; Meineri, G. State-of-the-Art of the Nutritional Alternatives to the Use of Antibiotics in Humans and Monogastric Animals. Animals 2020, 10, 2199. [Google Scholar] [CrossRef]
- Tursi, A.; Mocci, G.; Elisei, W.; Allegretta, L.; Colucci, R.; Della Valle, N.; De Medici, A.; Faggiani, R.; Ferronato, A.; Forti, G.; et al. Long-term, Real-life, Observational Study in Treating Outpatient Ulcerative Colitis with Golimumab. J. Gastrointestin. Liver Dis. 2021, 30, 456–461. [Google Scholar] [CrossRef]
- Roda, G.; Jharap, B.; Neeraj, N.; Colombel, J.F. Loss of Response to Anti-TNFs: Definition, Epidemiology, and Management. Clin. Transl. Gastroenterol. 2016, 7, e135. [Google Scholar] [CrossRef]
- Bertani, L.; Caviglia, G.P.; Antonioli, L.; Pellicano, R.; Fagoonee, S.; Astegiano, M.; Saracco, G.M.; Bugianesi, E.; Blandizzi, C.; Costa, F.; et al. Serum Interleukin-6 and -8 as Predictors of Response to Vedolizumab in Inflammatory Bowel Diseases. J. Clin. Med. 2020, 9, 1323. [Google Scholar] [CrossRef]
- Osuna, C.G.; Gómez-Vila, B.; Pariente, J.A.; Losada, B.V.; de Castro, C.G.; López, S.R.; Velázquez, Á.D.; García, L.P.; Fernández, L.O.; Diez, S.G.; et al. Ustekinumab drug survival in patients with psoriasis: A retrospective study of real clinical practice. Medicina 2020, 56, 584. [Google Scholar] [CrossRef]
- Sandborn, W.J.; Lawendy, N.; Danese, S.; Su, C.; Loftus, E.V.; Hart, A.; Dotan, I.; Damião, A.O.M.C.; Judd, D.T.; Guo, X.; et al. Safety and efficacy of tofacitinib for treatment of ulcerative colitis: Final analysis of OCTAVE Open, an open-label, long-term extension study with up to 7.0 years of treatment. Aliment. Pharmacol. Ther. 2022, 55, 464–478. [Google Scholar] [CrossRef]
- Sturgess, K. Diagnosis and management of idiopathic inflammatory bowel disease in dogs and cats. In Pract. 2005, 27, 293–301. [Google Scholar] [CrossRef]
- Kilpinen, S.; Spillmann, T.; Westermarck, E. Efficacy of two low-dose oral tylosin regimens in controlling the relapse of diarrhea in dogs with tylosin-responsive diarrhea: A prospective, single-blinded, two-arm parallel, clinical field trial. Acta Vet. Scand. 2014, 56, 43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Menozzi, A.; Dall’Aglio, M.; Quintavalla, F.; Dallavalle, L.; Meucci, V.; Bertini, S. Rifaximin is an effective alternative to metronidazole for the treatment of chronic enteropathy in dogs: A randomised trial. BMC Vet. Res. 2016, 12, 217. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malewska, K.; Rychlik, A.; Nieradka, R.; Kander, M. Treatment of inflammatory bowel disease (IBD) in dogs and cats. Pol. J. Vet. Sci. 2011, 14, 165–171. [Google Scholar] [CrossRef] [Green Version]
- Viviano, K.R. Update on immununosuppressive therapies for dogs and cats. Vet. Clin. N. Am. Small Anim. Pract. 2013, 43, 1149–1170. [Google Scholar] [CrossRef]
- Ruemmele, F.M.; Veres, G.; Kolho, K.L.; Griffiths, A.; Levine, A.; Escher, J.C.; Amil Dias, J.; Barabino, A.; Braegger, C.P.; Bronsky, J.; et al. Consensus guidelines of ECCO/ESPGHAN on the medical management of pediatric Crohn’s disease. J. Crohn’s Colitis 2014, 8, 1179–1207. [Google Scholar] [CrossRef] [Green Version]
- Bischoff, S.C.; Escher, J.; Hébuterne, X.; Kłęk, S.; Krznaric, Z.; Schneider, S.; Shamir, R.; Stardelova, K.; Wierdsma, N.; Wiskin, A.E.; et al. ESPEN practical guideline: Clinical Nutrition in inflammatory bowel disease. Clin. Nutr. 2020, 39, 632–653. [Google Scholar] [CrossRef] [Green Version]
- Dandrieux, J.R.S. Inflammatory bowel disease versus chronic enteropathy in dogs: Are they one and the same? J. Small Anim. Pract. 2016, 57, 589–599. [Google Scholar] [CrossRef]
- Howeler, J.F.; Hewson, A.D. Dietary fibre and toxaemia of pregnancy. Med. J. Aust. 1957, 44, 761–763. [Google Scholar] [CrossRef]
- Hamaker, B.R.; Tuncil, Y.E. A perspective on the complexity of dietary fiber structures and their potential effect on the gut microbiota. J. Mol. Biol. 2014, 426, 3838–3850. [Google Scholar] [CrossRef]
- Valcheva, R.; Dieleman, L.A. Prebiotics: Definition and protective mechanisms. Best Pract. Res. Clin. Gastroenterol. 2016, 30, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Mudgil, D.; Barak, S. Composition, properties and health benefits of indigestible carbohydrate polymers as dietary fiber: A review. Int. J. Biol. Macromol. 2013, 61, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, H.; Mander, I.; Zhang, Z.; Armstrong, D.; Wine, E. Not All Fibers Are Born Equal; Variable Response to Dietary Fiber Subtypes in IBD. Front. Pediatr. 2021, 8, 620189. [Google Scholar] [CrossRef] [PubMed]
- Cockburn, D.W.; Koropatkin, N.M. Polysaccharide Degradation by the Intestinal Microbiota and Its Influence on Human Health and Disease. J. Mol. Biol. 2016, 428, 3230–3252. [Google Scholar] [CrossRef]
- Amre, D.K.; D’Souza, S.; Morgan, K.; Seidman, G.; Lambrette, P.; Grimard, G.; Israel, D.; MacK, D.; Ghadirian, P.; Deslandres, C.; et al. Imbalances in dietary consumption of fatty acids, vegetables, and fruits are associated with risk for crohn’s disease in children. Am. J. Gastroenterol. 2007, 102, 2016–2025. [Google Scholar] [CrossRef]
- Issa, M.; Saeian, K. Diet in inflammatory bowel disease. Nutr. Clin. Pract. 2011, 26, 151–154. [Google Scholar] [CrossRef]
- Maslowski, K.M.; MacKay, C.R. Diet, gut microbiota and immune responses. Nat. Immunol. 2011, 12, 5–9. [Google Scholar] [CrossRef]
- Cohen, A.B.; Lee, D.; Long, M.D.; Kappelman, M.D.; Martin, C.F.; Sandler, R.S.; Lewis, J.D. Dietary patterns and self-reported associations of diet with symptoms of inflammatory bowel disease. Dig. Dis. Sci. 2013, 58, 1322–1328. [Google Scholar] [CrossRef]
- Sood, A.; Ahuja, V.; Kedia, S.; Midha, V.; Mahajan, R.; Mehta, V.; Sudhakar, R.; Singh, A.; Kumar, A.; Puri, A.S.; et al. Diet and inflammatory bowel disease: The Asian Working Group guidelines. Indian J. Gastroenterol. 2019, 38, 220–246. [Google Scholar] [CrossRef] [Green Version]
- Triantafillidis, J.K.; Vagianos, C.; Papalois, A.E. The role of enteral nutrition in patients with inflammatory bowel disease: Current aspects. BioMed Res. Int. 2015, 2015, 197167. [Google Scholar] [CrossRef] [Green Version]
- Yu, Y.; Chen, K.C.; Chen, J. Exclusive enteral nutrition versus corticosteroids for treatment of pediatric Crohn’s disease: A meta-analysis. World J. Pediatr. 2019, 15, 26–36. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Walton, C.; Montoya, M.P.B.; Fowler, D.P.; Turner, C.; Jia, W.; Whitehead, R.N.; Griffiths, L.; Waring, R.H.; Ramsden, D.B.; Cole, J.A.; et al. Enteral feeding reduces metabolic activity of the intestinal microbiome in Crohn’s disease: An observational study. Eur. J. Clin. Nutr. 2016, 70, 1052–1056. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burgis, J.C.; Nguyen, K.; Park, K.T.; Cox, K. Response to strict and liberalized specific carbohydrate diet in pediatric Crohn’s disease. World J. Gastroenterol. 2016, 22, 2111–2117. [Google Scholar] [CrossRef]
- Olendzki, B.C.; Silverstein, T.D.; Persuitte, G.M.; Ma, Y.; Baldwin, K.R.; Cave, D. An anti-inflammatory diet as treatment for inflammatory bowel disease: A case series report. Nutr. J. 2014, 13, 5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ambrosini, Y.M.; Neuber, S.; Borcherding, D.; Seo, Y.J.; Segarra, S.; Glanemann, B.; Garden, O.A.; Müller, U.; Adam, M.G.; Dang, V.; et al. Treatment With Hydrolyzed Diet Supplemented With Prebiotics and Glycosaminoglycans Alters Lipid Metabolism in Canine Inflammatory Bowel Disease. Front. Vet. Sci. 2020, 7, 451. [Google Scholar] [CrossRef]
- Hand, M.S.; Lewis, L.D. Small Animal Clinical Nutrition; Mark Morris Institute: Topeka, KS, USA, 2010; ISBN 9780615297019. [Google Scholar]
- Biesalski, H.K. Nutraceuticals: The link between nutrition and medicine. J. Toxicol. Cutan. Ocul. Toxicol. 2002, 21, 9–30. [Google Scholar] [CrossRef]
- Gupta, D.D.; Mishra, S.; Verma, S.S.; Shekher, A.; Rai, V.; Awasthee, N.; Das, T.J.; Paul, D.; Das, S.K.; Tag, H.; et al. Evaluation of antioxidant, anti-inflammatory and anticancer activities of diosgenin enriched Paris polyphylla rhizome extract of Indian Himalayan landraces. J. Ethnopharmacol. 2021, 270, 113842. [Google Scholar] [CrossRef]
- Mannucci, C.; Casciaro, M.; Sorbara, E.E.; Calapai, F.; Di Salvo, E.; Pioggia, G.; Navarra, M.; Calapai, G.; Gangemi, S. Nutraceuticals against Oxidative Stress in Autoimmune Disorders. Antioxidants 2021, 10, 261. [Google Scholar] [CrossRef]
- Day, A.S. Use of complementary and alternative therapies and probiotic agents by children attending gastroenterology outpatient clinics. J. Paediatr. Child Health 2002, 38, 343–346. [Google Scholar] [CrossRef]
- Lin, Z.; Liu, H.; Yang, C.; Zheng, H.; Zhang, Y.; Su, W.; Shang, J. Curcumin mediates autophagy and apoptosis in granulosa cells: A study of integrated network pharmacology and molecular docking to elucidate toxicological mechanisms. Drug Chem. Toxicol. 2021, 1–13. [Google Scholar] [CrossRef]
- Coelho, M.R.; Romi, M.D.; Ferreira, D.M.T.P.; Zaltman, C.; Soares-Mota, M. The Use of Curcumin as a Complementary Therapy in Ulcerative Colitis: A Systematic Review of Randomized Controlled Clinical Trials. Nutrients 2020, 12, 2296. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.C.; Patchva, S.; Aggarwal, B.B. Therapeutic roles of curcumin: Lessons learned from clinical trials. AAPS J. 2013, 15, 195–218. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hanai, H.; Iida, T.; Takeuchi, K.; Watanabe, F.; Maruyama, Y.; Andoh, A.; Tsujikawa, T.; Fujiyama, Y.; Mitsuyama, K.; Sata, M.; et al. Curcumin maintenance therapy for ulcerative colitis: Randomized, multicenter, double-blind, placebo-controlled trial. Clin. Gastroenterol. Hepatol. 2006, 4, 1502–1506. [Google Scholar] [CrossRef]
- Campigotto, G.; Alba, D.F.; Sulzbach, M.M.; Dos Santos, D.S.; Souza, C.F.; Baldissera, M.D.; Gundel, S.; Ourique, A.F.; Zimmer, F.; Petrolli, T.G.; et al. Dog food production using curcumin as antioxidant: Effects of intake on animal growth, health and feed conservation. Arch. Anim. Nutr. 2020, 74, 397–413. [Google Scholar] [CrossRef] [PubMed]
- Dulbecco, P.; Savarino, V. Therapeutic potential of curcumin in digestive diseases. World J. Gastroenterol. 2013, 19, 9256–9270. [Google Scholar] [CrossRef]
- Huang, W.T.; Niu, K.C.; Chang, C.K.; Lin, M.T.; Chang, C.P. Curcumin inhibits the increase of glutamate, hydroxyl radicals and PGE2 in the hypothalamus and reduces fever during LPS-induced systemic inflammation in rabbits. Eur. J. Pharmacol. 2008, 593, 105–111. [Google Scholar] [CrossRef]
- Hęś, M.; Dziedzic, K.; Górecka, D.; Jędrusek-Golińska, A.; Gujska, E. Aloe vera (L.) Webb.: Natural Sources of Antioxidants—A Review. Plant Foods Hum. Nutr. 2019, 74, 255–265. [Google Scholar] [CrossRef] [Green Version]
- Nejatzadeh-Barandozi, F. Antibacterial activities and antioxidant capacity of Aloe vera. Org. Med. Chem. Lett. 2013, 3, 5. [Google Scholar] [CrossRef] [Green Version]
- Langmead, L.; Feakins, R.M.; Goldthorpe, S.; Holt, H.; Tsironi, E.; De Silva, A.; Jewell, D.P.; Rampton, D.S. Randomized, double-blind, placebo-controlled trial of oral aloe vera gel for active ulcerative colitis. Aliment. Pharmacol. Ther. 2004, 19, 739–747. [Google Scholar] [CrossRef]
- Lans, C.; Turner, N.; Khan, T.; Brauer, G. Ethnoveterinary medicines used to treat endoparasites and stomach problems in pigs and pets in British Columbia, Canada. Vet. Parasitol. 2007, 148, 325–340. [Google Scholar] [CrossRef]
- Ammon, H.P.T. Boswellic acids and their role in chronic inflammatory diseases. In Advances in Experimental Medicine and Biology; Springer: New York, NY, USA, 2016; Volume 928, pp. 291–327. [Google Scholar]
- Pellegrini, L.; Milano, E.; Franceschi, F. Managing ulcerative colitis in remission phase: Usefulness of Casperome®, an innovative lecithin-based delivery system of Boswellia serrata extract—PubMed. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 2695–2700. [Google Scholar] [PubMed]
- Gupta, I.; Parihar, A.; Malhotra, P.; Gupta, S.; Lüdtke, R.; Safayhi, H.; Ammon, H.P.T. Effects of gum resin of Boswellia serrata in patients with chronic colitis. Planta Med. 2001, 67, 391–395. [Google Scholar] [CrossRef] [PubMed]
- Umar, S.; Umar, K.; Sarwar, A.H.M.G.; Khan, A.; Ahmad, N.; Ahmad, S.; Katiyar, C.K.; Husain, S.A.; Khan, H.A. Boswellia serrata extract attenuates inflammatory mediators and oxidative stress in collagen induced arthritis. Phytomedicine 2014, 21, 847–856. [Google Scholar] [CrossRef] [PubMed]
- Caterino, C.; Aragosa, F.; della Valle, G.; Costanza, D.; Lamagna, F.; Piscitelli, A.; Nieddu, A.; Fatone, G. Clinical efficacy of Curcuvet and Boswellic acid combined with conventional nutraceutical product: An aid to canine osteoarthritis. PLoS ONE 2021, 16, e0252279. [Google Scholar] [CrossRef]
- Martello, E.; Biasibetti, E.; Bigliati, M.; Meineri, G.; Bruni, N. Preliminary results on the efficacy of a dietary supplement combined with physiotherapy in dogs with osteoarthritis on biomarkers of oxidative stress and inflammation. Ital. J. Anim. Sci. 2021, 20, 2131–2133. [Google Scholar] [CrossRef]
- Ben-Arye, E.; Goldin, E.; Wengrower, D.; Stamper, A.; Kohn, R.; Berry, E. Wheat grass juice in the treatment of active distal ulcerative colitis: A randomized double-blind placebo-controlled trial. Scand. J. Gastroenterol. 2002, 37, 444–449. [Google Scholar] [CrossRef]
- Marsili, V.; Calzuola, I.; Gianfranceschi, G.L. Nutritional relevance of wheat sprouts containing high levels of organic phosphates and antioxidant compounds. J. Clin. Gastroenterol. 2004, 38, S123–S126. [Google Scholar] [CrossRef]
- Madgulkar, A.R.; Rao, M.R.P.; Warrier, D. Characterization of Psyllium (Plantago ovata) Polysaccharide and Its Uses. Polysaccharides 2015, 871–890. [Google Scholar] [CrossRef]
- Baghizadeh, A.; Davati, A.; Heidarloo, A.J.; Emadi, F.; Aliasl, J. Efficacy of Plantago major seed in management of ulcerative colitis symptoms: A randomized, placebo controlled, clinical trial. Complement. Ther. Clin. Pract. 2021, 44, 101444. [Google Scholar] [CrossRef]
- Fernández-Bañares, F.; Hinojosa, J.; Sánchez-Lombraña, J.L.; Navarro, E.; Martínez-Salmerón, J.F.; García-Pugés, A.; González-Huix, F.; Riera, J.; González-Lara, V.; Domínguez-Abascal, F.; et al. Randomized Clinical Trial of Plantago Ovata Seeds (Dietary Fiber) As Compared With Mesalamine in Maintaining Remission in Ulcerative Colitis. Am. J. Gastroenterol. 1999, 94, 427–433. [Google Scholar] [CrossRef]
- Najafian, Y.; Hamedi, S.S.; Kaboli Farshchi, M.; Feyzabadi, Z. Plantago major in Traditional Persian Medicine and modern phytotherapy: A narrative review. Electron. Physician 2018, 10, 6390–6399. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Algieri, F.; Rodriguez-Nogales, A.; Garrido-Mesa, N.; Zorrilla, P.; Burkard, N.; Pischel, I.; Sievers, H.; Benedek, B.; Feistel, B.; Walbroel, B.; et al. Intestinal anti-inflammatory activity of the Serpylli herba extract in experimental models of rodent colitis. J. Crohns. Colitis 2014, 8, 775–788. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nakajima, J.I.; Tanaka, I.; Seo, S.; Yamazaki, M.; Saito, K. LC/PDA/ESI-MS Profiling and Radical Scavenging Activity of Anthocyanins in Various Berries. J. Biomed. Biotechnol. 2004, 2004, 241–247. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Biedermann, L.; Mwinyi, J.; Scharl, M.; Frei, P.; Zeitz, J.; Kullak-Ublick, G.A.; Vavricka, S.R.; Fried, M.; Weber, A.; Humpf, H.U.; et al. Bilberry ingestion improves disease activity in mild to moderate ulcerative colitis—An open pilot study. J. Crohns. Colitis 2013, 7, 271–279. [Google Scholar] [CrossRef] [Green Version]
- Sgorlon, S.; Stefanon, B.; Sandri, M.; Colitti, M. Nutrigenomic activity of plant derived compounds in health and disease: Results of a dietary intervention study in dog. Res. Vet. Sci. 2016, 109, 142–148. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Wang, X.; Chen, Q.; Luo, L.; Ma, M.; Xiao, B.; Zeng, L. Camellia sinensis and Litsea coreana Ameliorate Intestinal Inflammation and Modulate Gut Microbiota in Dextran Sulfate Sodium-Induced Colitis Mice. Mol. Nutr. Food Res. 2020, 64, 1900943. [Google Scholar] [CrossRef]
- Oz, H.S. Chronic Inflammatory Diseases and Green Tea Polyphenols. Nutrients 2017, 9, 561. [Google Scholar] [CrossRef]
- Doustfatemeh, S.; Imanieh, M.H.; Mohagheghzade, A.; Zarshenas, M.M.; Torkamani, Z.; Yousefi, G.; Farahangiz, S.; Salehi, A. The Effect of Black Tea (Camellia sinensis (L) Kuntze) on Pediatrics With Acute Nonbacterial Diarrhea: A Randomized Controlled Trial. J. Evid. Based. Complement. Altern. Med. 2017, 22, 114. [Google Scholar] [CrossRef]
- Rahman, S.U.; Huang, Y.; Zhu, L.; Chu, X.; Junejo, S.A.; Zhang, Y.; Khan, I.M.; Li, Y.; Feng, S.; Wu, J.; et al. Tea polyphenols attenuate liver inflammation by modulating obesity-related genes and down-regulating COX-2 and iNOS expression in high fat-fed dogs. BMC Vet. Res. 2020, 16, 234. [Google Scholar] [CrossRef]
- Hininger-Favier, I.; Benaraba, R.; Coves, S.; Anderson, R.A.; Roussel, A.M. Green tea extract decreases oxidative stress and improves insulin sensitivity in an animal model of insulin resistance, the fructose-fed rat. J. Am. Coll. Nutr. 2009, 28, 355–361. [Google Scholar] [CrossRef]
- Li, H.; Wei, Y.; Li, X.; Zhang, S.; Zhang, R.; Li, J.; Ma, B.; Shao, S.; Lv, Z.; Ruan, H.; et al. Diosmetin has therapeutic efficacy in colitis regulating gut microbiota, inflammation, and oxidative stress via the circ-Sirt1/Sirt1 axis. Acta Pharmacol. Sin. 2021; Online ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Larrosa, M.; González-Sarrías, A.; Yáñez-Gascón, M.J.; Selma, M.V.; Azorín-Ortuño, M.; Toti, S.; Tomás-Barberán, F.; Dolara, P.; Espín, J.C. Anti-inflammatory properties of a pomegranate extract and its metabolite urolithin-A in a colitis rat model and the effect of colon inflammation on phenolic metabolism. J. Nutr. Biochem. 2010, 21, 717–725. [Google Scholar] [CrossRef] [PubMed]
- Vaghari-Tabari, M.; Jafari-Gharabaghlou, D.; Sadeghsoltani, F.; Hassanpour, P.; Qujeq, D.; Rashtchizadeh, N.; Ghorbanihaghjo, A. Zinc and Selenium in Inflammatory Bowel Disease: Trace Elements with Key Roles? Biol. Trace Elem. Res. 2020, 199, 3190–3204. [Google Scholar] [CrossRef]
- Zentrichová, V.; Pechová, A.; Kovaříková, S. Selenium and Dogs: A Systematic Review. Animals 2021, 11, 418. [Google Scholar] [CrossRef] [PubMed]
- Gammoh, N.Z.; Rink, L. Zinc in infection and inflammation. Nutrients 2017, 9, 624. [Google Scholar] [CrossRef] [Green Version]
- Gîlcǎ-Blanariu, G.E.; Diaconescu, S.; Ciocoiu, M.; Tefǎnescu, G. New Insights into the Role of Trace Elements in IBD. BioMed Res. Int. 2018, 2018, 1813047. [Google Scholar] [CrossRef]
- Ranaldi, G.; Ferruzza, S.; Canali, R.; Leoni, G.; Zalewski, P.D.; Sambuy, Y.; Perozzi, G.; Murgia, C. Intracellular zinc is required for intestinal cell survival signals triggered by the inflammatory cytokine TNFα. J. Nutr. Biochem. 2013, 24, 967–976. [Google Scholar] [CrossRef]
- Mohammadi, E.; Qujeq, D.; Taheri, H.; Hajian-Tilaki, K. Evaluation of Serum Trace Element Levels and Superoxide Dismutase Activity in Patients with Inflammatory Bowel Disease: Translating Basic Research into Clinical Application. Biol. Trace Elem. Res. 2017, 177, 235–240. [Google Scholar] [CrossRef]
- Oteiza, P.I. Zinc and the modulation of redox homeostasis. Free Radic. Biol. Med. 2012, 53, 1748–1759. [Google Scholar] [CrossRef] [Green Version]
- Sato, M.; Bremner, I. Oxygen free radicals and metallothionein. Free Radic. Biol. Med. 1993, 14, 325–337. [Google Scholar] [CrossRef]
- Colombo, G.; Clemente, N.; Zito, A.; Bracci, C.; Colombo, F.S.; Sangaletti, S.; Jachetti, E.; Ribaldone, D.G.; Caviglia, G.P.; Pastorelli, L.; et al. Neutralization of extracellular NAMPT (nicotinamide phosphoribosyltransferase) ameliorates experimental murine colitis. J. Mol. Med. 2020, 98, 595–612. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.; Liu, X.; Pan, Z. Zinc deficiency and cellular oxidative stress: Prognostic implications in cardiovascular diseases review-article. Acta Pharmacol. Sin. 2018, 39, 1120–1132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miyoshi, Y.; Tanabe, S.; Suzuki, T. Cellular zinc is required for intestinal epithelial barrier maintenance via the regulation of claudin-3 and occludin expression. Am. J. Physiol. Gastrointest. Liver Physiol. 2016, 311, G105–G116. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chinni, V.; El-Khoury, J.; Perera, M.; Bellomo, R.; Jones, D.; Bolton, D.; Ischia, J.; Patel, O. Zinc supplementation as an adjunct therapy for COVID-19: Challenges and opportunities. Br. J. Clin. Pharmacol. 2021, 87, 3737–3746. [Google Scholar] [CrossRef]
- Amorim Zafalon, R.V.; Perini, M.P.; Annibale Vendramini, T.H.; Pedrinelli, V.; Rentas, M.F.; Morilha, I.B.; Fiuza Henríquez, L.B.; Consentino Conti, R.M.; Brunetto, M.A. Vitamin-mineral supplements do not guarantee the minimum recommendations and may imply risks of mercury poisoning in dogs and cats. PLoS ONE 2021, 16, e0250738. [Google Scholar]
- Sakai, K.; Hatoya, S.; Furuya, M.; Shimamura, S.; Nabetani, T.; Tani, H.; Shimada, T. Decreased serum zinc concentration in dogs with lymphocytic-plasmacytic enteritis, and its associations with disease severity and prognosis. J. Vet. Med. Sci. 2020, 82, 759–763. [Google Scholar] [CrossRef] [Green Version]
- Cummings, J.E.; Kovacic, J.P. The ubiquitous role of zinc in health and disease. J. Vet. Emerg. Crit. Care 2009, 19, 215–240. [Google Scholar] [CrossRef]
- Naveh, Y.; Bentur, L.; Diamond, E. Site of zinc absorption in dog small intestine. J. Nutr. 1988, 118, 61–64. [Google Scholar] [CrossRef]
- Ren, Z.; Zhao, Z.; Wang, Y.; Huang, K. Preparation of selenium/zinc-enriched probiotics and their effect on blood selenium and zinc concentrations, antioxidant capacities, and intestinal microflora in canine. Biol. Trace Elem. Res. 2011, 141, 170–183. [Google Scholar] [CrossRef]
- Riggio, O.; Merli, M.; Capocaccia, L.; Caschera, M.; Zullo, A.; Pinto, G.; Gaudio, E.; Franchitto, A.; Spagnoli, R.; D’aquilino, E.; et al. Zinc supplementation reduces blood ammonia and increases liver ornithine transcarbamylase activity in experimental cirrhosis. Hepatology 1992, 16, 785–789. [Google Scholar] [CrossRef]
- Brewer, G.J.; Dick, R.D.; Schall, W.; Yuzbasiyan-Gurkan, V.; Mullaney, T.P.; Pace, C.; Lindgren, J.; Thomas, M.; Padgett, G. Use of zinc acetate to treat copper toxicosis in dogs. J. Am. Vet. Med. Assoc. 1992, 201, 564–568. [Google Scholar] [PubMed]
- Li, J.; Chen, H.; Wang, B.; Cai, C.; Yang, X.; Chai, Z.; Feng, W. ZnO nanoparticles act as supportive therapy in DSS-induced ulcerative colitis in mice by maintaining gut homeostasis and activating Nrf2 signaling. Sci. Rep. 2017, 7, 43126. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Filburn, C.; Kettenacker, R.; Griffin, D. Safety and Bioavailability in Beagles of Zinc and Vitamin E Combined with Silybin and Phosphatidylcholine. Int. J. Appl. Res. Vet. Med. 2006, 4, 326–334. [Google Scholar]
- Pereira, A.M.; Maia, M.R.G.; Fonseca, A.J.M.; Cabrita, A.R.J. Zinc in Dog Nutrition, Health and Disease: A Review. Animals 2021, 11, 978. [Google Scholar] [CrossRef] [PubMed]
- Nutritional Guidelines—FEDIAF. Available online: https://fediaf.org/self-regulation/nutrition.html (accessed on 15 December 2021).
- Stoffaneller, R.; Morse, N.L. A review of dietary selenium intake and selenium status in Europe and the Middle East. Nutrients 2015, 7, 1494–1537. [Google Scholar] [CrossRef]
- Vaghari-Tabari, M.; Moein, S.; Qujeq, D.; Kashifard, M.; Hajian-Tilaki, K. Positive Correlation of Fecal Calprotectin With Serum Antioxidant Enzymes in Patients With Inflammatory Bowel Disease: Accidental Numerical Correlation or a New Finding? Am. J. Med. Sci. 2018, 355, 449–455. [Google Scholar] [CrossRef]
- Nunes, C.; Teixeira, N.; Serra, D.; Freitas, V.; Almeida, L.; Laranjinha, J. Red wine polyphenol extract efficiently protects intestinal epithelial cells from inflammation via opposite modulation of JAK/STAT and Nrf2 pathways. Toxicol. Res. 2015, 5, 53–65. [Google Scholar] [CrossRef]
- El-Demerdash, F.M.; Nasr, H.M. Antioxidant effect of selenium on lipid peroxidation, hyperlipidemia and biochemical parameters in rats exposed to diazinon. J. Trace Elem. Med. Biol. 2014, 28, 89–93. [Google Scholar] [CrossRef]
- Bitiren, M.; Karakilcik, A.Z.; Zerin, M.; Ozardalı, I.; Selek, S.; Nazlıgül, Y.; Ozgonul, A.; Musa, D.; Uzunkoy, A. Protective effects of selenium and vitamin E combination on experimental colitis in blood plasma and colon of rats. Biol. Trace Elem. Res. 2010, 136, 87–95. [Google Scholar] [CrossRef]
- Hadrup, N.; Ravn-Haren, G. Acute human toxicity and mortality after selenium ingestion: A review. J. Trace Elem. Med. Biol. 2020, 58, 126435. [Google Scholar] [CrossRef]
- Griffiths, J.C.; Matulka, R.A.; Power, R. Acute and subchronic toxicity studies on Sel-Plex, a standardized, registered high-selenium yeast. Int. J. Toxicol. 2006, 25, 465–476. [Google Scholar] [CrossRef] [PubMed]
- Forrer, R.; Gautschi, K.; Lutz, H. Comparative determination of selenium in the serum of various animal species and humans by means of electrothermal atomic absorption spectrometry. J. Trace Elem. Electrolytes Health Dis. 1991, 5, 101–113. [Google Scholar] [PubMed]
- Trivedi, P.P.; Jena, G.B. Mechanistic insight into beta-carotene-mediated protection against ulcerative colitis-associated local and systemic damage in mice. Eur. J. Nutr. 2015, 54, 639–652. [Google Scholar] [CrossRef] [PubMed]
- Carazo, A.; Macáková, K.; Matoušová, K.; Krčmová, L.K.; Protti, M.; Mladěnka, P. Vitamin A Update: Forms, Sources, Kinetics, Detection, Function, Deficiency, Therapeutic Use and Toxicity. Nutrients 2021, 13, 1703. [Google Scholar] [CrossRef]
- Morris, P.J.; Salt, C.; Raila, J.; Brenten, T.; Kohn, B.; Schweigert, F.J.; Zentek, J. Safety evaluation of vitamin A in growing dogs. Br. J. Nutr. 2012, 108, 18. [Google Scholar] [CrossRef]
- Green, A.S.; Fascetti, A.J. Meeting the Vitamin A Requirement: The Efficacy and Importance of β-Carotene in Animal Species. Sci. World J. 2016, 2016, 7393620. [Google Scholar] [CrossRef] [Green Version]
- Mirbagheri, S.A.; Nezami, B.G.; Assa, S.; Hajimahmoodi, M. Rectal administration of d-alpha tocopherol for active ulcerative colitis: A preliminary report. World J. Gastroenterol. 2008, 14, 5990–5995. [Google Scholar] [CrossRef]
- Jewell, D.E.; Toll, P.W.; Wedekind, K.J.; Zicker, S.C. Effect of increasing dietary antioxidants on concentrations of vitamin E and total alkenals in serum of dogs and cats. Vet. Ther. 2000, 1, 264–272. [Google Scholar]
- Kondo, K.; Hiramoto, K.; Yamate, Y.; Goto, K.; Sekijima, H.; Ooi, K. Ameliorative Effect of High-Dose Vitamin C Administration on Dextran Sulfate Sodium-Induced Colitis Mouse Model. Biol. Pharm. Bull. 2019, 42, 954–959. [Google Scholar] [CrossRef] [Green Version]
- Ratajczak, A.E.; Szymczak-Tomczak, A.; Skrzypczak-Zielińska, M.; Rychter, A.M.; Zawada, A.; Dobrowolska, A.; Krela-Kaźmierczak, I. Vitamin C Deficiency and the Risk of Osteoporosis in Patients with an Inflammatory Bowel Disease. Nutrients 2020, 12, 2263. [Google Scholar] [CrossRef]
- Packialakshmi, B.; Stewart, I.J.; Burmeister, D.M.; Chung, K.K.; Zhou, X. Large animal models for translational research in acute kidney injury. Ren. Fail. 2020, 42, 1042–1058. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meineri, G.; Martello, E.; Radice, E.; Bruni, N.; Saettone, V.; Atuahene, D.; Armandi, A.; Testa, G.; Ribaldone, D.G. Chronic Intestinal Disorders in Humans and Pets: Current Management and the Potential of Nutraceutical Antioxidants as Alternatives. Animals 2022, 12, 812. https://doi.org/10.3390/ani12070812
Meineri G, Martello E, Radice E, Bruni N, Saettone V, Atuahene D, Armandi A, Testa G, Ribaldone DG. Chronic Intestinal Disorders in Humans and Pets: Current Management and the Potential of Nutraceutical Antioxidants as Alternatives. Animals. 2022; 12(7):812. https://doi.org/10.3390/ani12070812
Chicago/Turabian StyleMeineri, Giorgia, Elisa Martello, Elisabetta Radice, Natascia Bruni, Vittorio Saettone, David Atuahene, Angelo Armandi, Giulia Testa, and Davide Giuseppe Ribaldone. 2022. "Chronic Intestinal Disorders in Humans and Pets: Current Management and the Potential of Nutraceutical Antioxidants as Alternatives" Animals 12, no. 7: 812. https://doi.org/10.3390/ani12070812
APA StyleMeineri, G., Martello, E., Radice, E., Bruni, N., Saettone, V., Atuahene, D., Armandi, A., Testa, G., & Ribaldone, D. G. (2022). Chronic Intestinal Disorders in Humans and Pets: Current Management and the Potential of Nutraceutical Antioxidants as Alternatives. Animals, 12(7), 812. https://doi.org/10.3390/ani12070812









