Association between Increased the De Ritis Quotient and Renal Azotaemia in Canine Babesiosis
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Sample
2.2. Renal Indices
2.3. Groups and Subgroups of Dogs
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Irwin, P.J. Canine babesiosis: From molecular taxonomy to control. Parasit. Vectors 2009, 2, S4. [Google Scholar] [CrossRef] [PubMed]
- Matijatko, V.; Torti, M.; Schetters, T.P. Canine babesiosis in Europe: How many diseases? Trends Parasitol. 2012, 28, 99–105. [Google Scholar] [CrossRef]
- Baneth, G.; Cardoso, L.; Brilhante-Simões, P.; Schnittger, L. Establishment of Babesia vulpes n. sp. (Apicomplexa: Babesiidae), a piroplasmid species pathogenic for domestic dogs. Parasit. Vectors 2019, 12, 129. [Google Scholar] [CrossRef] [PubMed]
- Zygner, W.; Wędrychowicz, H. Influence of anaemia on azotaemia in dogs infected with Babesia canis in Poland. Bull. Vet. Inst. Pulawy 2009, 53, 663–668. [Google Scholar]
- Máthé, A.; Vörös, K.; Papp, L.; Reiczigel, J. Clinical manifestations of canine babesiosis in Hungary (63 cases). Acta Vet. Hung. 2006, 54, 367–385. [Google Scholar] [CrossRef] [PubMed]
- Gójska-Zygner, O.; Bartosik, J.; Górski, P.; Zygner, W. Hyponatraemia and syndrome of inappropriate antidiuretic hormone secretion in non-azotaemic dogs with babesiosis associated with decreased arterial blood pressure. J. Vet. Res. 2019, 63, 339–344. [Google Scholar] [CrossRef]
- Schoeman, J.P.; Rees, P.; Herrtage, M.E. Endocrine predictors of mortality in canine babesiosis caused by Babesia canis rossi. Vet. Parasitol. 2007, 148, 75–82. [Google Scholar] [CrossRef]
- Matijatko, V.; Kiš, I.; Torti, M.; Brkljačić, M.; Kučer, N.; Rafaj, R.B.; Grden, D.; Živičnjak, T.; Mrljak, V. Septic shock in canine babesiosis. Vet. Parasitol. 2009, 162, 263–270. [Google Scholar] [CrossRef]
- Winiarczyk, D.; Michalak, K.; Adaszek, L.; Winiarczyk, M.; Winiarczyk, S. Urinary proteome of dogs with kidney injury during babesiosis. BMC Vet. Res. 2019, 15, 439. [Google Scholar] [CrossRef]
- Zygner, W.; Rodo, A.; Gójska-Zygner, O.; Górski, P.; Bartosik, J.; Kotomski, G. Disorders in blood circulation as a probable cause of death in dogs infected with Babesia canis. J. Vet. Res. 2021, 65, 277–285. [Google Scholar] [CrossRef]
- Lobetti, R.G.; Jacobson, L.S. Renal involvement in dogs with babesiosis. J. S. Afr. Vet. Assoc. 2001, 72, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Zygner, W.; Gójska-Zygner, O.; Wesołowska, A.; Wędrychowicz, H. Urinary creatinine to serum creatinine ratio and renal failure index in dogs infected with Babesia canis. Acta Parasitol. 2013, 58, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Zygner, W.; Gójska-Zygner, O.; Norbury, L.J.; Wędrychowicz, H. Increased AST/ALT ratio in azotaemic dogs infected with Babesia canis. Pol. J. Vet. Sci. 2012, 15, 483–486. [Google Scholar] [CrossRef][Green Version]
- Zygner, W.; Gójska-Zygner, O.; Wędrychowicz, H. Strong monovalent electrolyte imbalances in serum of dogs infected with Babesia canis. Ticks Tick Borne Dis. 2012, 3, 107–113. [Google Scholar] [CrossRef]
- Rymaszewska, A.; Adamska, M. Molecular evidence of vector-borne pathogens coinfecting dogs from Poland. Acta Vet. Hung. 2011, 59, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Myczka, A.W.; Steiner-Bogdaszewska, Ż.; Filip-Hutsch, K.; Oloś, G.; Czopowicz, M.; Laskowski, Z. Detection of Anaplasma phagocytophilum in Wild and Farmed Cervids in Poland. Pathogens 2021, 10, 1190. [Google Scholar] [CrossRef]
- Walls, J.J.; Caturegli, P.; Bakken, J.S.; Asanovich, K.M.; Dumler, J.S. Improved sensitivity of PCR for diagnosis of human granulocytic ehrlichiosis using epank1 genes of Ehrlichia phagocytophila-group ehrlichiae. J. Clin. Microbiol. 2000, 38, 354–356. [Google Scholar] [CrossRef]
- Lobetti, R.G. Changes in the serum urea: Creatinine ratio in dogs with babesiosis, haemolytic anaemia, and experimental haemoglobinaemia. Vet. J. 2012, 191, 253–256. [Google Scholar] [CrossRef]
- Waldrop, J.E. Urinary Electrolytes, Solutes, and Osmolality. Vet. Clin. N. Am. Small Anim. Pract. 2008, 38, 503–512. [Google Scholar] [CrossRef]
- Zygner, W.; Gójska-Zygner, O. Increased serum urea to creatinine ratio and its negative correlation with arterial pressure in canine babesiosis. Acta Parasitol. 2014, 59, 548–551. [Google Scholar] [CrossRef]
- De Scally, M.P.; Leisewitz, A.L.; Lobetti, R.G.; Thompson, P.N. The elevated serum urea: Creatinine ratio in canine babesiosis in South Africa is not of renal origin. J. S. Afr. Vet. Assoc. 2006, 77, 175–178. [Google Scholar] [CrossRef] [PubMed]
- Sutherland, R.J. Biochemical evaluation of the hepatobiliary system in dogs and cats. Vet. Clin. N. Am. Small Anim. Pract. 1989, 19, 899–927. [Google Scholar] [CrossRef]
- Furlanello, T.; Fiorio, F.; Caldin, M.; Lubas, G.; Solano-Gallego, L. Clinicopathological findings in naturally occurring cases of babesiosis caused by large form Babesia from dogs of northeastern Italy. Vet. Parasitol. 2005, 134, 77–85. [Google Scholar] [CrossRef]
- Adaszek, Ł.; Winiarczyk, S.; Skrzypczak, M. The clinical course of babesiosis in 76 dogs infected with protozoan parasites Babesia canis canis. Pol. J. Vet. Sci. 2009, 12, 81–87. [Google Scholar] [PubMed]
- Valentine, B.A.; Blue, J.T.; Shelley, S.M.; Cooper, B.J. Increased serum alanine aminotransferase activity associated with muscle necrosis in the dog. J. Vet. Intern. Med. 1990, 4, 140–143. [Google Scholar] [CrossRef]
- Jacobson, L.S.; Lobetti, R.G. Rhabdomyolysis as a complication of canine babesiosis. J. Small Anim. Pract. 1996, 37, 286–291. [Google Scholar] [CrossRef]
- Jacobson, L.S. The South African form of severe and complicated canine babesiosis: Clinical advances 1994–2004. Vet. Parasitol. 2006, 138, 126–139. [Google Scholar] [CrossRef]
- Visser, M.P.; Krill, M.T.; Muijtjens, A.M.; Willems, G.M.; Hermens, W.T. Distribution of enzymes in dog heart and liver; significance for assessment of tissue damage from data on plasma enzyme activities. Clin. Chem. 1981, 27, 1845–1850. [Google Scholar] [CrossRef]
- Dvir, E.; Lobetti, R.G.; Jacobson, L.S.; Pearson, J.; Becker, P.J. Electrocardiographic changes and cardiac pathology in canine babesiosis. J. Vet. Cardiol. 2004, 6, 15–23. [Google Scholar] [CrossRef]
- Beugnet, F.; Halos, L.; Larsen, D.; Labuschagné, M.; Erasmus, H.; Fourie, J. The ability of an oral formulation of afoxolaner to block the transmission of Babesia canis by Dermacentor reticulatus ticks to dogs. Parasit. Vectors 2014, 7, 283. [Google Scholar] [CrossRef]
- Taenzler, J.; Liebenberg, J.; Roepke, R.K.A.; Heckeroth, A.R. Prevention of transmission of Babesia canis by Dermacentor reticulatus ticks to dogs treated orally with fluralaner chewable tablets (Bravecto™). Parasit. Vectors 2015, 8, 305. [Google Scholar] [CrossRef] [PubMed]
- Teodorowski, O.; Winiarczyk, S.; Debiak, P.; Skrzypczak, M.; Mazurek, Ł.; Adaszek, Ł. Clinical course of granulocytic anaplasmosis in hunting dogs. Pol. J. Vet. Sci. 2021, 24, 175–181. [Google Scholar] [PubMed]
- Kuleš, J.; Bilić, P.; Beer Ljubić, B.; Gotić, J.; Crnogaj, M.; Brkljačić, M.; Mrljak, V. Glomerular and tubular kidney damage markers in canine babesiosis caused by Babesia canis. Ticks Tick Borne Dis. 2018, 9, 1508–1517. [Google Scholar] [CrossRef] [PubMed]
- Defauw, P.; Schoeman, J.P.; Leisewitz, A.L.; Goddard, A.; Duchateau, L.; Aresu, L.; Meyer, E.; Daminet, S. Evaluation of acute kidney injury in dogs with complicated or uncomplicated Babesia rossi infection. Ticks Tick Borne Dis. 2020, 11, 101406. [Google Scholar] [CrossRef] [PubMed]
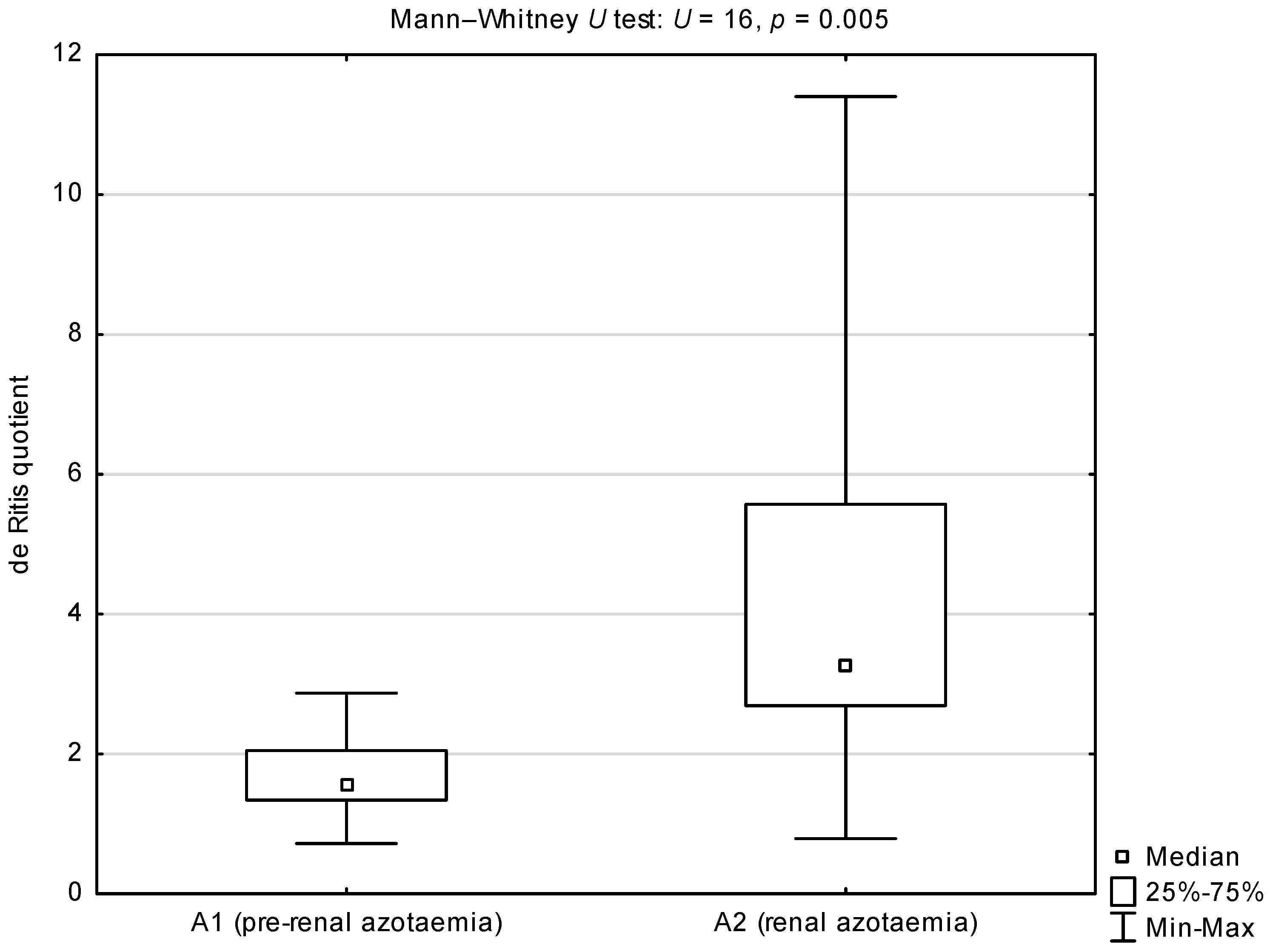
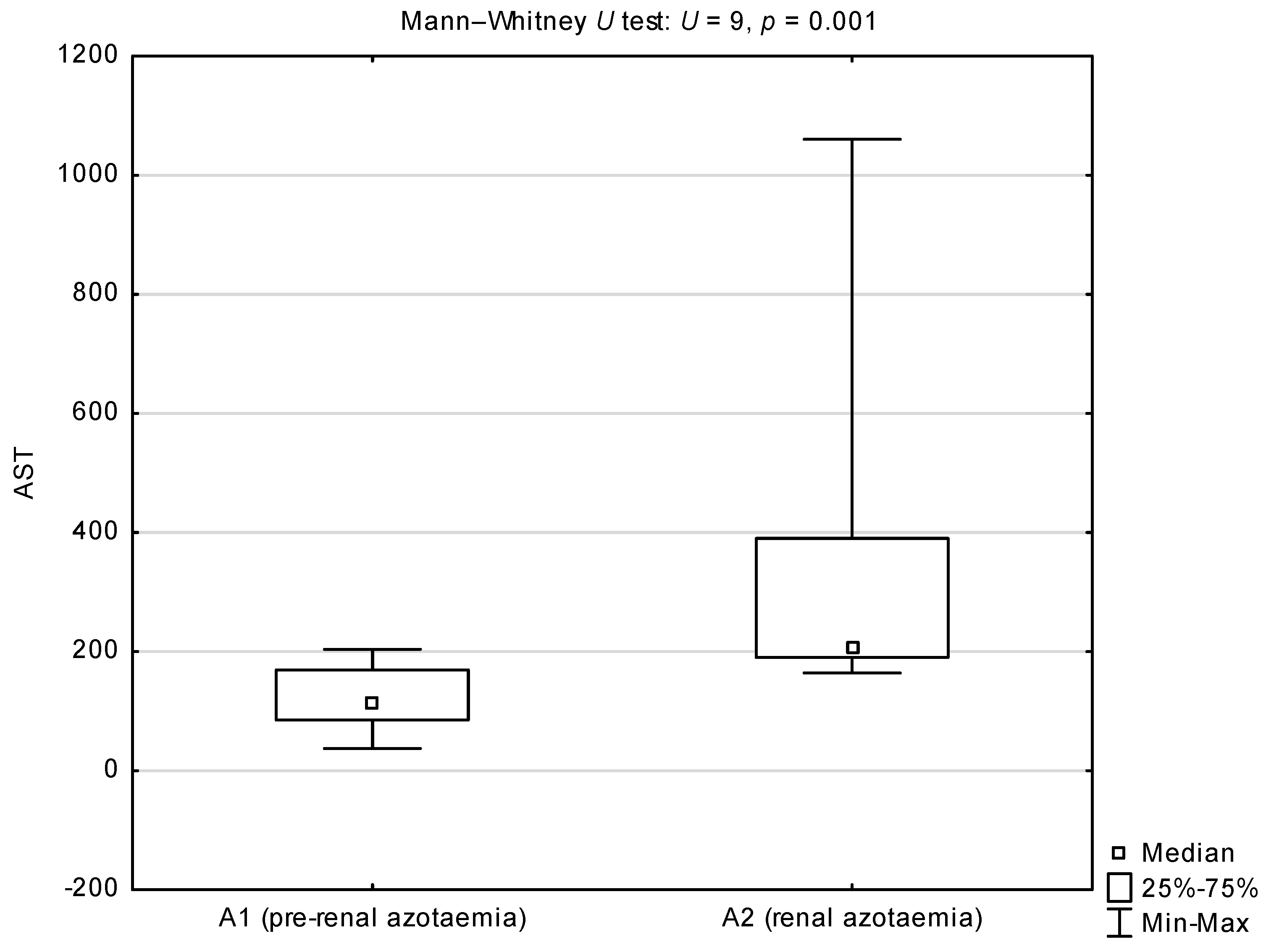
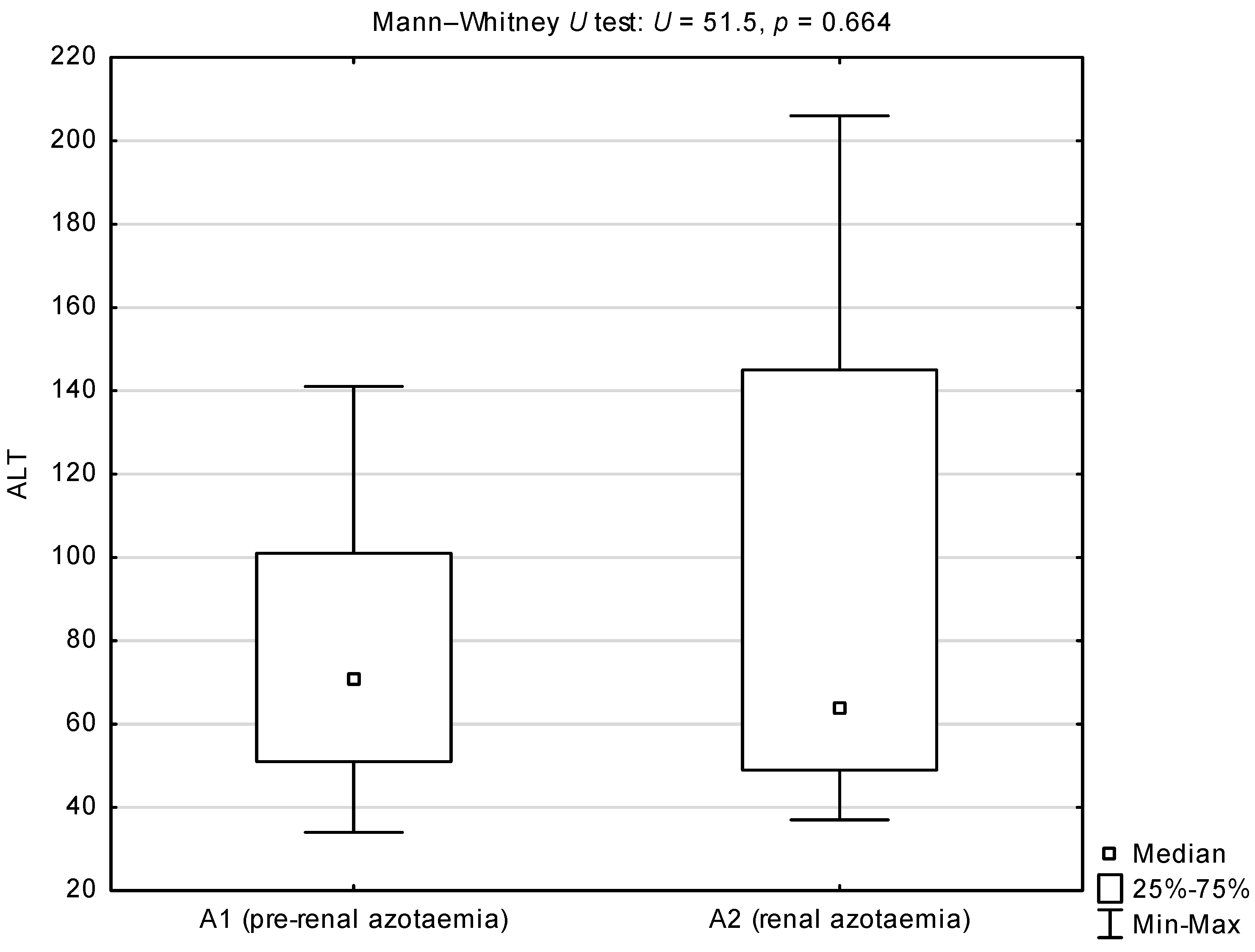
| Group or Sub-Group | Median (25th–75th Percentile) | |
|---|---|---|
| Urea (mg/dL) | Creatinine (mg/dL) | |
| A (azotaemic) | 194.5 (110–311) | 3.1 (1.5–4.1) |
| B (non-azotemic) | 41 (36.5–42) | 1.25 (1–1.35) |
| A1 (pre-renal azotaemia) | 132 (85–181) | 1.8 (1.5–2.9) |
| A2 (renal azotaemia) | 265 (227–341) | 5.1 (3.7–6.9) |
| Parameter | Median (25th%–75th%) | Min–Max | U | p | ||
|---|---|---|---|---|---|---|
| A (Azotaemic) | B (Non-Azotaemic) | A | B | |||
| AST/ALT ratio | 2.17 (1.35–3.22) | 1.08 (0.77–1.375) | 0.72–11.4 | 0.15–2.54 | 48 | 0.002 * |
| AST (IU/L) | 171.5 (95–204) | 89.5 (46–118.5) | 37–1060 | 27–159 | 49.5 | 0.003 * |
| ALT (IU/L) | 67.5 (49–125) | 66.5 (48–91) | 34–206 | 32–1050 | 125.5 | 0.828 |
| Correlations | R | p | |
|---|---|---|---|
| De Ritis quotient | RFI | 0.520 | 0.013 * |
| FE(Na+) | 0.522 | 0.012 * | |
| SU/SCr | 0.074 | 0.74 | |
| UCr/SCr | −0.460 | 0.032 * | |
| AST | RFI | 0.715 | 0.0002 * |
| FE(Na+) | 0.739 | 0.00008 * | |
| SU/SCr | −0.081 | 0.721 | |
| UCr/SCr | −0.732 | 0.0001 * | |
| ALT | RFI | 0.143 | 0.526 |
| FE(Na+) | 0.186 | 0.407 | |
| SU/SCr | −0.031 | 0.891 | |
| UCr/SCr | −0.282 | 0.203 | |
| Correlation | R | p |
|---|---|---|
| Groups A and B combined | 0.407 | 0.017 * |
| Group A (azotaemic) | 0.296 | 0.181 |
| Group B (non-azotaemic) | 0.714 | 0.009 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gójska-Zygner, O.; Karabowicz, J.; Bartosik, J.; Zygner, W. Association between Increased the De Ritis Quotient and Renal Azotaemia in Canine Babesiosis. Animals 2022, 12, 626. https://doi.org/10.3390/ani12050626
Gójska-Zygner O, Karabowicz J, Bartosik J, Zygner W. Association between Increased the De Ritis Quotient and Renal Azotaemia in Canine Babesiosis. Animals. 2022; 12(5):626. https://doi.org/10.3390/ani12050626
Chicago/Turabian StyleGójska-Zygner, Olga, Justyna Karabowicz, Justyna Bartosik, and Wojciech Zygner. 2022. "Association between Increased the De Ritis Quotient and Renal Azotaemia in Canine Babesiosis" Animals 12, no. 5: 626. https://doi.org/10.3390/ani12050626
APA StyleGójska-Zygner, O., Karabowicz, J., Bartosik, J., & Zygner, W. (2022). Association between Increased the De Ritis Quotient and Renal Azotaemia in Canine Babesiosis. Animals, 12(5), 626. https://doi.org/10.3390/ani12050626






