Feeding Fiber-Bound Polyphenol Ingredients at Different Levels Modulates Colonic Postbiotics to Improve Gut Health in Dogs
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Foods
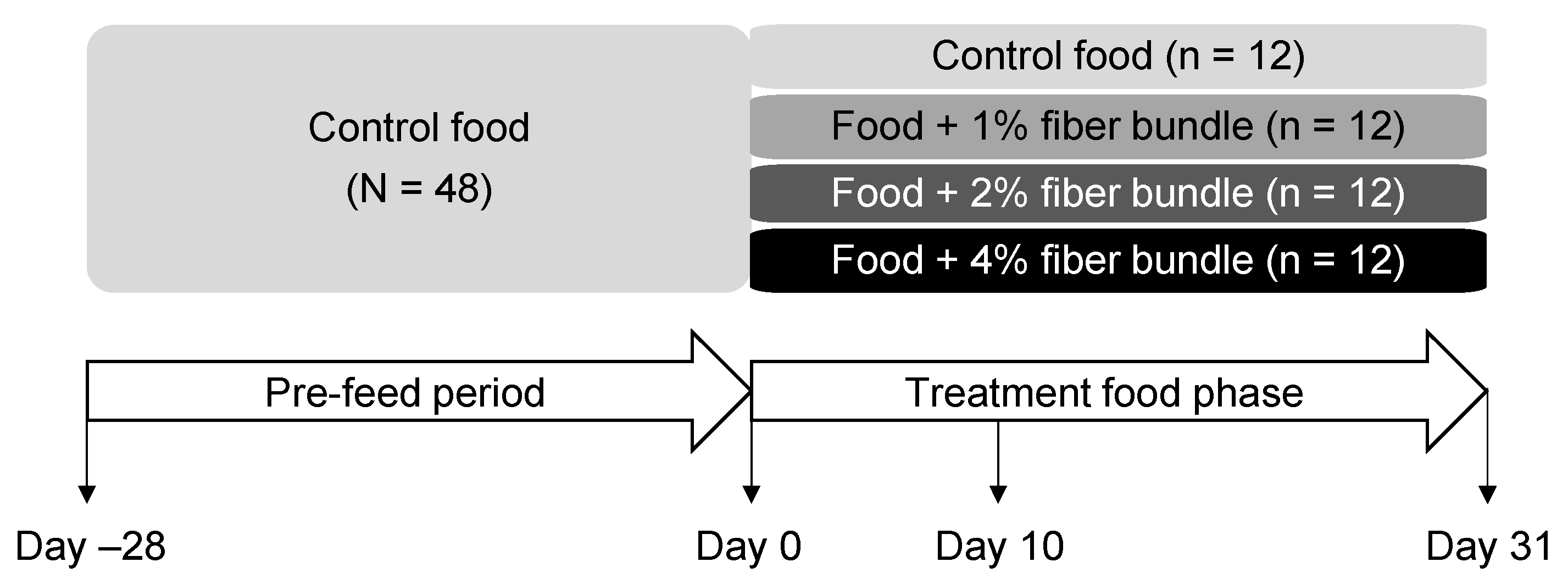
2.2. Animals and Experimental Design
2.3. Stool Scoring and Fecal Sample Processing
2.4. Serum and Metabolite Analyses
2.5. Fecal Microbiome Analysis and Bioinformatics Processing
2.6. Statistical Analysis
3. Results
3.1. Animal Demographics, Intakes, and Clinical Assessment
3.2. Fecal Parameters and Analytes
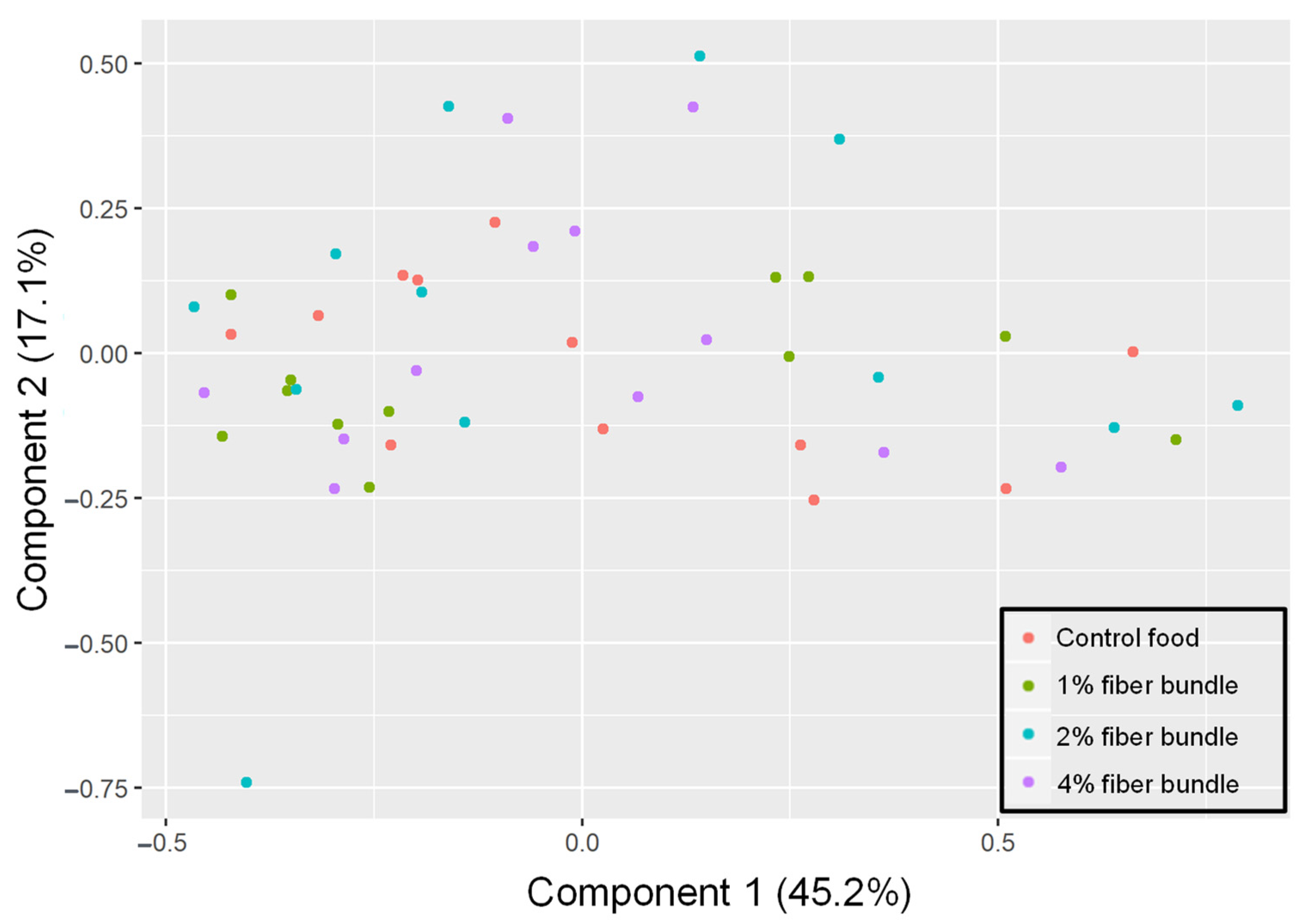
3.3. Fecal Microbiota
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shock, T.; Badang, L.; Ferguson, B.; Martinez-Guryn, K. The interplay between diet, gut microbes, and host epigenetics in health and disease. J. Nutr. Biochem. 2021, 95, 108631. [Google Scholar] [CrossRef] [PubMed]
- Wernimont, S.M.; Radosevich, J.; Jackson, M.; Ephraim, E.; Badri, D.V.; MacLeay, J.M.; Jewell, D.E.; Suchodolski, J.S. The effects of nutrition on the gastrointestinal microbiome of cats and dogs: Impact on health and disease. Front. Microbiol. 2020, 11, 1266. [Google Scholar] [CrossRef] [PubMed]
- Abbas, M.; Saeed, F.; Anjum, F.M.; Afzaal, M.; Tufail, T.; Bashir, M.S.; Ishtiaq, A.; Hussain, S.; Suleria, H.A.R. Natural polyphenols: An overview. Int. J. Food Prop. 2017, 20, 1689–1699. [Google Scholar] [CrossRef] [Green Version]
- Cardona, F.; Andrés-Lacueva, C.; Tulipani, S.; Tinahones, F.J.; Queipo-Ortuño, M.I. Benefits of polyphenols on gut microbiota and implications in human health. J. Nutr. Biochem. 2013, 24, 1415–1422. [Google Scholar] [CrossRef] [Green Version]
- Selma, M.V.; Espín, J.C.; Tomás-Barberán, F.A. Interaction between phenolics and gut microbiota: Role in human health. J. Agric. Food Chem. 2009, 57, 6485–6501. [Google Scholar] [CrossRef]
- Van Der Beek, C.M.; DeJong, C.H.C.; Troost, F.J.; Masclee, A.A.M.; Lenaerts, K. Role of short-chain fatty acids in colonic inflammation, carcinogenesis, and mucosal protection and healing. Nutr. Rev. 2017, 75, 286–305. [Google Scholar] [CrossRef]
- Ríos-Covián, D.; Ruas-Madiedo, P.; Margolles, A.; Gueimonde, M.; De Los Reyes-Gavilán, C.G.; Salazar, N. Intestinal short chain fatty acids and their link with diet and human health. Front. Microbiol. 2016, 7, 185. [Google Scholar] [CrossRef] [Green Version]
- Martinez, K.B.; Leone, V.; Chang, E.B. Microbial metabolites in health and disease: Navigating the unknown in search of function. J. Biol. Chem. 2017, 292, 8553–8559. [Google Scholar] [CrossRef] [Green Version]
- Middelbos, I.S.; Boler, B.M.V.; Qu, A.; White, B.A.; Swanson, K.S.; Fahey, G.C., Jr. Phylogenetic characterization of fecal microbial communities of dogs fed diets with or without supplemental dietary fiber using 454 pyrosequencing. PLoS ONE 2010, 5, e9768. [Google Scholar] [CrossRef] [Green Version]
- Panasevich, M.R.; Kerr, K.R.; Dilger, R.N.; Fahey, G.C.; Guérin-Deremaux, L.; Lynch, G.L.; Wils, D.; Suchodolski, J.S.; Steer, J.M.; Dowd, S.E.; et al. Modulation of the faecal microbiome of healthy adult dogs by inclusion of potato fibre in the diet. Br. J. Nutr. 2014, 113, 125–133. [Google Scholar] [CrossRef] [Green Version]
- Panasevich, M.R.; Serao, M.C.R.; De Godoy, M.R.C.; Swanson, K.; Guérin-Deremaux, L.; Lynch, G.L.; Wils, D.; Fahey, G.C., Jr.; Dilger, R.N. Potato fiber as a dietary fiber source in dog foods. J. Anim. Sci. 2013, 91, 5344–5352. [Google Scholar] [CrossRef]
- Vanhoutte, T.; Huys, G.; De Brandt, E.; Fahey, J.G.C.; Swings, J. Molecular monitoring and characterization of the faecal microbiota of healthy dogs during fructan supplementation. FEMS Microbiol. Lett. 2005, 249, 65–71. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maria, A.P.J.; Ayane, L.; Putarov, T.C.; Loureiro, B.A.; Neto, B.P.; Casagrande, M.F.; Gomes, M.O.S.; Glória, M.B.A.; Carciofi, A.C. The effect of age and carbohydrate and protein sources on digestibility, fecal microbiota, fermentation products, fecal IgA, and immunological blood parameters in dogs. J. Anim. Sci. 2017, 95, 2452–2466. [Google Scholar] [CrossRef] [PubMed]
- Kröger, S.; Vahjen, W.; Zentek, J. Influence of lignocellulose and low or high levels of sugar beet pulp on nutrient digestibility and the fecal microbiota in dogs. J. Anim. Sci. 2017, 95, 1598–1605. [Google Scholar] [CrossRef] [PubMed]
- Simpson, J.; Martineau, B.; Jones, W.; Ballam, J.; Mackie, R. Characterization of fecal bacterial populations in canines: Effects of age, breed and dietary fiber. Microb. Ecol. 2002, 44, 186–197. [Google Scholar] [CrossRef] [PubMed]
- Jackson, M.I.; Jewell, D.E. Balance of saccharolysis and proteolysis underpins improvements in stool quality induced by adding a fiber bundle containing bound polyphenols to either hydrolyzed meat or grain-rich foods. Gut Microbes 2018, 10, 298–320. [Google Scholar] [CrossRef] [Green Version]
- Hall, J.A.; Melendez, L.D.; Jewell, D.E. Using gross energy improves metabolizable energy predictive equations for pet foods whereas undigested protein and fiber content predict stool quality. PLoS ONE 2013, 8, e54405. [Google Scholar] [CrossRef] [Green Version]
- National Research Council. Guide for the Care and Use of Laboratory Animals, 8th ed.; The National Academies Press: Washington, DC, USA, 2011; ISBN 978-0-309-38629-6. [CrossRef]
- Ephraim, E.; Cochrane, C.-Y.; Jewell, D.E. Varying protein levels influence metabolomics and the gut microbiome in healthy adult dogs. Toxins 2020, 12, 517. [Google Scholar] [CrossRef]
- Schloss, P.D.; Westcott, S.L.; Ryabin, T.; Hall, J.R.; Hartmann, M.; Hollister, E.B.; Lesniewski, R.A.; Oakley, B.B.; Parks, D.H.; Robinson, C.J.; et al. Introducing mothur: Open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl. Environ. Microbiol. 2009, 75, 7537–7541. [Google Scholar] [CrossRef] [Green Version]
- DeSantis, T.Z.; Hugenholtz, P.; Larsen, N.; Rojas, M.; Brodie, E.L.; Keller, K.; Huber, T.; Dalevi, D.; Hu, P.; Andersen, G.L. Greengenes, a chimera-checked 16S rRNA gene database and workbench compatible with ARB. Appl. Environ. Microbiol. 2006, 72, 5069–5072. [Google Scholar] [CrossRef] [Green Version]
- Langille, M.G.I.; Zaneveld, J.; Caporaso, J.G.; McDonald, D.; Knights, D.; Reyes, J.A.; Clemente, J.C.; Burkepile, D.E.; Vega Thurber, R.L.; Knight, R.; et al. Predictive functional profiling of microbial communities using 16S rRNA marker gene sequences. Nat. Biotechnol. 2013, 31, 814–821. [Google Scholar] [CrossRef] [PubMed]
- Scoditti, E. Neuroinflammation and neurodegeneration: The promising protective role of the citrus flavanone hesperetin. Nutrients 2020, 12, 2336. [Google Scholar] [CrossRef] [PubMed]
- Stevens, Y.; Van Rymenant, E.; Grootaert, C.; Van Camp, J.; Possemiers, S.; Masclee, A.; Jonkers, D. The intestinal fate of citrus flavanones and their effects on gastrointestinal health. Nutrients 2019, 11, 1464. [Google Scholar] [CrossRef] [Green Version]
- Mas-Capdevila, A.; Teichenne, J.; Domenech-Coca, C.; Caimari, A.; Del Bas, J.M.; Escoté, X.; Crescenti, A. Effect of hesperidin on cardiovascular disease risk factors: The role of intestinal microbiota on hesperidin bioavailability. Nutrients 2020, 12, 1488. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, G.R.; Vasconcelos, A.B.S.; Wu, D.-T.; Li, H.-B.; Antony, P.J.; Li, H.; Geng, F.; Gurgel, R.Q.; Narain, N.; Gan, R.-Y. Citrus flavonoids as promising phytochemicals targeting diabetes and related complications: A systematic review of in vitro and in vivo studies. Nutrients 2020, 12, 2907. [Google Scholar] [CrossRef]
- Kim, D.-H.; Jung, E.-A.; Sohng, I.-S.; Han, J.-A.; Kim, T.-H.; Han, M.J. Intestinal bacterial metabolism of flavonoids and its relation to some biological activities. Arch. Pharmacal. Res. 1998, 21, 17–23. [Google Scholar] [CrossRef]
- Khan, A.; Ikram, M.; Hahm, J.R.; Kim, M.O. Antioxidant and anti-inflammatory effects of citrus flavonoid hesperetin: Special focus on neurological disorders. Antioxidants 2020, 9, 609. [Google Scholar] [CrossRef]
- Liu, P.; Li, J.; Liu, M.; Zhang, M.; Xue, Y.; Zhang, Y.; Han, X.; Jing, X.; Chu, L. Hesperetin modulates the Sirt1/Nrf2 signaling pathway in counteracting myocardial ischemia through suppression of oxidative stress, inflammation, and apoptosis. Biomed. Pharmacother. 2021, 139, 111552. [Google Scholar] [CrossRef]
- Noda, S.; Tanabe, S.; Suzuki, T. Differential effects of flavonoids on barrier integrity in human intestinal Caco-2 cells. J. Agric. Food Chem. 2012, 60, 4628–4633. [Google Scholar] [CrossRef]
- Duda-Chodak, A. The inhibitory effect of polyphenols on human gut microbiota. J. Physiol. Pharmacol. Off. J. Pol. Physiol. Soc. 2012, 63, 497–503. [Google Scholar]
- Duque, A.L.R.F.; Monteiro, M.; Adorno, M.A.T.; Sakamoto, I.K.; Sivieri, K. An exploratory study on the influence of orange juice on gut microbiota using a dynamic colonic model. Food Res. Int. 2016, 84, 160–169. [Google Scholar] [CrossRef]
- Kang, G.-D.; Kim, D.-H. Poncirin and its metabolite ponciretin attenuate colitis in mice by inhibiting LPS binding on TLR4 of macrophages and correcting Th17/Treg imbalance. J. Ethnopharmacol. 2016, 189, 175–185. [Google Scholar] [CrossRef] [PubMed]
- Kang, G.-D.; Kim, D.-H. Ponciretin attenuates ethanol-induced gastric damage in mice by inhibiting inflammatory responses. Int. Immunopharmacol. 2017, 43, 179–186. [Google Scholar] [CrossRef]
- Kim, D.-H.; Bae, E.-A.; Han, M.J. Anti-Helicobacter pylori activity of the metabolites of poncirin from Poncirus trifoliata by human intestinal bacteria. Biol. Pharm. Bull. 1999, 22, 422–424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Padda, J.; Khalid, K.; Cooper, A.C.; Jean-Charles, G. Association between Helicobacter pylori and gastric carcinoma. Cureus 2021, 13, e15165. [Google Scholar] [CrossRef] [PubMed]
- Kezimana, P.; Dmitriev, A.A.; Kudryavtseva, A.V.; Romanova, E.V.; Melnikova, N.V. Secoisolariciresinol diglucoside of flaxseed and its metabolites: Biosynthesis and potential for nutraceuticals. Front. Genet. 2018, 9, 641. [Google Scholar] [CrossRef] [Green Version]
- Imran, M.; Ahmad, N.; Anjum, F.M.; Khan, M.K.; Mushtaq, Z.; Nadeem, M.; Hussain, S. Potential protective properties of flax lignan secoisolariciresinol diglucoside. Nutr. J. 2015, 14, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Senizza, A.; Rocchetti, G.; Mosele, J.I.; Patrone, V.; Callegari, M.L.; Morelli, L.; Lucini, L. Lignans and gut microbiota: An interplay revealing potential health implications. Molecules 2020, 25, 5709. [Google Scholar] [CrossRef]
- Ephraim, E.; Jewell, D.E. Effect of added dietary betaine and soluble fiber on metabolites and fecal microbiome in dogs with early renal disease. Metabolites 2020, 10, 370. [Google Scholar] [CrossRef]
- Aqeel, T.; Gurumallu, S.C.; Bhaskar, A.; Hashimi, S.M.; Javaraiah, R. Secoisolariciresinol diglucoside protects against cadmium-induced oxidative stress-mediated renal toxicity in rats. J. Trace Elements Med. Biol. 2020, 61, 126552. [Google Scholar] [CrossRef]
- Wang, Z.; Chen, T.; Yang, C.; Bao, T.; Yang, X.; He, F.; Zhang, Y.; Zhu, L.; Chen, H.; Rong, S.; et al. Secoisolariciresinol diglucoside suppresses Dextran sulfate sodium salt-induced colitis through inhibiting NLRP1 inflammasome. Int. Immunopharmacol. 2019, 78, 105931. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Cheng, M.; Wang, Z.; Liu, Y.; Ren, Y.; Rong, S.; Wang, X. Secoisolariciresinol diglucoside exerts anti-inflammatory and antiapoptotic effects through inhibiting the akt/iκb/nf-κb pathway on human umbilical vein endothelial cells. Mediat. Inflamm. 2020, 2020, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Özgöçmen, M.; Bayram, D.; Türel, G.Y.; Toğay, V.A.; Calapoğlu, N. Secoisolariciresinol diglucoside induces caspase-3-mediated apoptosis in monolayer and spheroid cultures of human colon carcinoma cells. J. Food Biochem. 2021, 45, e13719. [Google Scholar] [CrossRef]
- Shin, M.-K.; Jeon, Y.-D.; Jin, J.-S. Apoptotic effect of enterodiol, the final metabolite of edible lignans, in colorectal cancer cells. J. Sci. Food Agric. 2018, 99, 2411–2419. [Google Scholar] [CrossRef]
- Basen, M.; Kurrer, S.E. A close look at pentose metabolism of gut bacteria. FEBS J. 2020, 288, 1804–1808. [Google Scholar] [CrossRef] [PubMed]
- Thornton, E.; Robinson, E.; Templeman, J.R.; Bruggink, L.; Bower, M.; Cant, J.P.; Holloway, G.P.; Swanson, K.S.; Squires, E.J.; Shoveller, A.K. Supplemental fiber affects body temperature and fecal metabolites but not respiratory rate or body composition in mid-distance training sled dogs. Front. Vet. Sci. 2021, 8, 639335. [Google Scholar] [CrossRef]
- Fernandez-Navarro, T.; Salazar, N.; Gutiérrez-Díaz, I.; Sánchez, B.; Rúas-Madiedo, P.; Reyes-Gavilan, C.D.L.; Margolles, A.; Gueimonde, M.; González, S. Bioactive compounds from regular diet and faecal microbial metabolites. Eur. J. Nutr. 2018, 57, 487–497. [Google Scholar] [CrossRef] [PubMed]
- Pilla, R.; Suchodolski, J.S. The gut microbiome of dogs and cats, and the influence of diet. Vet. Clin. N. Am. Small Anim. Pract. 2021, 51, 605–621. [Google Scholar] [CrossRef]
| Fiber Bundle Percentage in Food | ||||
|---|---|---|---|---|
| Nutrient Parameter | Control Food | 1% | 2% | 4% |
| Moisture | 8.11 | 8.38 | 8.50 | 8.41 |
| Protein, crude | 23.81 | 23.44 | 23.56 | 23.63 |
| Fat | 13.16 | 14.27 | 12.95 | 13.01 |
| Atwater energy 1 (kcal/kg) | 3569 | 3631 | 3567 | 3588 |
| Ash | 5.81 | 5.96 | 5.98 | 5.85 |
| Crude fiber | 2.4 | 2.3 | 2.1 | 1.8 |
| Total dietary fiber | 6.1 | 6.7 | 6.2 | 6.8 |
| Total insoluble fiber | 4.6 | 5.1 | 5.7 | 5.3 |
| Total soluble fiber | 1.5 | 1.6 | 0.5 | 1.5 |
| Calcium | 1.02 | 1.00 | 1.06 | 1.04 |
| Phosphorus | 0.75 | 0.77 | 0.79 | 0.78 |
| Sodium | 0.40 | 0.40 | 0.41 | 0.40 |
| Capric acid (10:0) | <0.02 | <0.02 | <0.02 | <0.02 |
| Lauric acid (12:0) | <0.02 | <0.02 | <0.02 | <0.02 |
| Myristic acid (14:0) | 0.07 | 0.07 | 0.07 | 0.07 |
| Palmitic acid (16:0) | 2.92 | 2.79 | 2.70 | 2.72 |
| Palmitoleic acid (16:1) | 0.70 | 0.66 | 0.63 | 0.64 |
| Steric acid (18:0) | 0.86 | 0.81 | 0.80 | 0.80 |
| Oleic acid (18:1) | 4.68 | 4.45 | 4.33 | 4.38 |
| Arachidic acid (20:0) | 0.02 | <0.02 | 0.02 | <0.02 |
| LA (18:2 (n-6)) | 2.69 | 2.73 | 2.71 | 2.74 |
| aLA (18:3 (n-3)) | 0.18 | 0.22 | 0.27 | 0.35 |
| ARA (20:4 (n-6)) | 0.05 | 0.05 | 0.05 | 0.05 |
| EPA (20:5 (n-3)) | <0.02 | <0.02 | <0.02 | <0.02 |
| DHA (22:6 (n-3)) | <0.02 | <0.02 | <0.02 | <0.02 |
| Lysine | 1.10 | 1.08 | 1.10 | 1.09 |
| Threonine | 0.89 | 0.85 | 0.84 | 0.85 |
| Methionine | 0.53 | 0.54 | 0.53 | 0.54 |
| Cystine | 0.31 | 0.31 | 0.31 | 0.31 |
| Tryptophan | 0.27 | 0.27 | 0.27 | 0.29 |
| Fiber Bundle Percentage in Food | ||||
|---|---|---|---|---|
| Parameter | Control Food | 1% | 2% | 4% |
| Body weight, kg | ||||
| Day 0 | 10.7 ± 0.55 | 10.6 ± 0.55 | 10.9 ± 0.55 | 10.5 ± 0.55 |
| Day 31 | 10.4 ± 0.56 | 10.3 ± 0.56 | 10.8 ± 0.56 | 10.3 ± 0.56 |
| Change | −0.3 ± 0.10 1 | −0.3 ± 0.10 1 | −0.1 ± 0.10 | −0.1 ± 0.10 |
| Food intake, kcal/(body weight in kg)0.75 | 112 ± 5.5 | 107 ± 5.5 | 119 ± 5.5 | 109 ± 5.5 |
| Albumin, mg/dL | ||||
| Day 0 | 3.57 ± 0.08 | 3.62 ± 0.08 | 3.45 ± 0.08 | 3.51 ± 0.08 |
| Day 31 | 3.68 ± 0.08 | 3.64 ± 0.08 | 3.52 ± 0.08 | 3.60 ± 0.08 |
| Change | 0.12 ± 0.04 | 0.02 ± 0.04 | 0.08 ± 0.04 | 0.09 ± 0.04 |
| Total protein, mg/dL | ||||
| Day 0 | 5.58 ± 0.09 | 5.78 ± 0.09 | 5.54 ± 0.09 | 5.65 ± 0.09 |
| Day 31 | 5.54 ± 0.11 | 5.60 ± 0.11 | 5.47 ± 0.11 | 5.64 ± 0.11 |
| Change | −0.04 ± 0.06 | −0.17 ± 0.06 1 | −0.08 ± 0.06 | −0.01 ± 0.06 |
| Urea nitrogen, mg/dL | ||||
| Day 0 | 12.2 ± 0.8 | 13.1 ± 0.8 | 14.1 ± 0.8 | 12.5 ± 0.8 |
| Day 31 | 11.5 ± 0.8 | 12.9 ± 0.8 | 13.9 ± 0.8 | 12.4 ± 0.8 |
| Change | −0.6 ± 0.6 | −0.2 ± 0.6 | −0.2 ± 0.6 | −0.1 ± 0.6 |
| Creatinine, mg/dL | ||||
| Day 0 | 0.71 ± 0.03 | 0.70 ± 0.03 | 0.68 ± 0.03 | 0.72 ± 0.03 |
| Day 31 | 0.75 ± 0.04 | 0.71 ± 0.04 | 0.73 ± 0.04 | 0.76 ± 0.04 |
| Change | 0.03 ± 0.02 | 0.01 ± 0.02 | 0.06 ± 0.02 1 | 0.04 ± 0.02 1 |
| Triglycerides, mg/dL | ||||
| Day 0 | 63.2 ± 5.6 | 59.1 ± 5.6 | 64.2 ± 5.6 | 65.6 ± 5.6 |
| Day 31 | 66.9 ± 5.1 | 62.7 ± 5.1 | 68.7 ± 5.1 | 61.6 ± 5.1 |
| Change | 3.7 ± 4.1 | 3.7 ± 4.1 | 4.5 ± 4.1 | −4.0 ± 4.1 |
| Cholesterol, mg/dL | ||||
| Day 0 | 195.1 ± 14.2 | 207.6 ± 14.2 | 191.2 ± 14.2 | 219.2 ± 14.2 |
| Day 31 | 203.5 ± 14.7 | 211.9 ± 14.7 | 184.0 ± 14.7 | 222.4 ± 14.7 |
| Change | 8.2 ± 5.8 | 4.3 ± 5.8 | −7.2 ± 5.8 | 3.2 ± 5.8 |
| Fiber Bundle Percentage in Food | ||||
|---|---|---|---|---|
| Parameter | Control Food | 1% | 2% | 4% |
| Moisture | ||||
| Day 0 | 68.3 ± 0.9 | 69.8 ± 0.9 | 68.1 ± 0.9 | 67.8 ± 0.9 |
| Day 10, % of Day 0 | 103 ± 1 a1 | 98 ± 1 b | 101 ± 1 a,b | 103 ± 1 a1 |
| Day 31, % of Day 0 | 105 ± 1 a1 | 100 ± 1 b | 104 ± 1 a1 | 104 ± 1 a1 |
| Ammonium, mmol/g | ||||
| Day 0 | 0.040 ± 0.002 | 0.044 ± 0.002 | 0.046 ± 0.002 | 0.045 ± 0.002 |
| Day 10, % of Day 0 | 97 ± 9 | 107 ± 9 | 94 ± 9 | 95 ± 9 |
| Day 31, % of Day 0 | 107 ± 9 | 110 ± 9 | 102 ± 9 | 104 ± 9 |
| pH | ||||
| Day 0 | 5.88 ± 0.06 | 5.92 ± 0.06 | 5.90 ± 0.06 | 5.97 ± 0.06 |
| Day 10, % of Day 0 | 100 ± 1 | 100 ± 1 | 100 ± 1 | 99 ± 1 |
| Day 31, % of Day 0 | 99 ± 1 | 102 ± 1 | 100 ± 1 | 98 ± 1 |
| Stool score | ||||
| Day 0 | 4.2 ± 0.20 | 4.2 ± 0.19 | 4.5 ± 0.19 | 4.0 ± 1.19 |
| Day 10 | 4.3 ± 0.19 | 4.4 ± 0.19 | 4.7 ± 0.19 | 4.5 ± 0.19 |
| Day 31 | 4.2 ± 0.19 | 4.4 ± 0.19 | 4.3 ± 0.19 | 4.5 ± 0.19 |
| Fiber Bundle Percentage in Food | ||||
|---|---|---|---|---|
| SCFA | Control Food | 1% | 2% | 4% |
| Acetic acid | ||||
| Day 0, μg/g | 4100 ± 180 | 4243 ± 173 | 4744 ± 173 | 4538 ± 173 |
| Day 10, % of Day 0 | 114 ± 7 | 103 ± 7 | 103 ± 7 | 104 ± 7 |
| Day 31, % of Day 0 | 112 ± 7 | 104 ± 7 | 93 ± 7 | 102 ± 7 |
| Propionic acid | ||||
| Day 0, μg/g | 3229 ± 191 | 3000 ± 183 | 3231 ± 183 | 3239 ± 183 |
| Day 10, % of Day 0 | 91 ± 9 | 104 ± 9 | 102 ± 9 | 96 ± 9 |
| Day 31, % of Day 0 | 86 ± 9 | 92 ± 9 | 90 ± 9 | 97 ± 9 |
| Butyric acid | ||||
| Day 0, μg/g | 3547 ± 449 | 3960 ± 430 | 3355 ± 430 | 3962 ± 430 |
| Day 10, % of Day 0 | 92 ± 25 | 102 ± 24 | 123 ± 24 | 104 ± 24 |
| Day 31, % of Day 0 | 120 ± 25 | 110 ± 24 | 126 ± 24 | 158 ± 24 1 |
| Valeric acid | ||||
| Day 0, μg/g | 530 ± 162 | 468 ± 155 | 640 ± 155 | 559 ± 155 |
| Day 10, % of initial | 100 ± 80 | 166 ± 76 | 160 ± 76 | 121 ± 76 |
| Day 31, % of initial | 141 ± 80 | 156 ± 76 | 189 ± 76 | 299 ± 76 1 |
| Hexanoic acid | ||||
| Day 0, μg/g | 27 ± 9.8 | 8 ± 9.5 | 30 ± 9.5 | 26 ± 9.5 |
| Day 10, % of Day 0 | 151 ± 251 | 316 ± 240 | 99 ± 240 | 107 ± 240 |
| Day 31, % of Day 0 | 120 ± 251 | 140 ± 240 | 167 ± 240 | 756 ± 240 1 |
| 2-methylpropionic acid | ||||
| Day 0, μg/g | 188 ± 21 | 215 ± 20 | 257 ± 20 | 253 ± 20 |
| Day 10, % of Day 0 | 108 ± 12 | 117 ± 12 | 95 ± 12 | 91 ± 12 |
| Day 31, % of Day 0 | 120 ± 12 | 115 ± 12 | 97 ± 12 | 98 ± 12 |
| 2-methylbutyric acid | ||||
| Day 0, μg/g | 137 ± 14 | 151 ± 14 | 181 ± 14 | 175 ± 14 |
| Day 10, % of Day 0 | 97 ± 11 | 113 ± 11 | 94 ± 11 | 91 ± 11 |
| Day 31, % of Day 0 | 107 ± 11 | 115 ± 11 | 92 ± 11 | 102 ± 11 |
| 3-methylbutyric acid | ||||
| Day 0, μg/g | 223 ± 23 | 229 ± 22 | 274 ± 22 | 275 ± 22 |
| Day 10, % of Day 0 | 99 ± 12 | 119 ± 12 | 102 ± 12 | 85 ± 12 |
| Day 31, % of Day 0 | 103 ± 12 | 109 ± 12 | 97 ± 12 | 102 ± 12 |
| Fiber Bundle Percentage in Food | ||||
|---|---|---|---|---|
| Metabolite | Control Food | 1% | 2% | 4% |
| Hesperidin | ||||
| Day 10 ratio | 0.42 ± 0.31 a | 2.15 ± 0.31 b1 | 3.63 ± 0.31 c1 | 4.55 ± 0.31 d1 |
| Day 31 ratio | 0.95 ± 0.31 a | 1.85 ± 0.31 b1 | 3.74 ± 0.31 c1 | 4.37 ± 0.31 c1 |
| Hesperetin | ||||
| Day 10 ratio | 0.62 ± 0.38 a | 3.59 ± 0.37 b1 | 4.69 ± 0.37 c1 | 5.39 ± 0.37 c1 |
| Day 31 ratio | 0.67 ± 0.38 a | 3.22 ± 0.37 b1 | 4.33 ± 0.37 c1 | 5.24 ± 0.37 c1 |
| Ponciretin | ||||
| Day 10 ratio | 0.53 ± 0.38 a | 3.19 ± 0.36 b1 | 3.93 ± 0.36 b1 | 4.76 ± 0.36 c1 |
| Day 31 ratio | 0.51 ± 0.38 a | 2.82 ± 0.36 b1 | 3.56 ± 0.36 b1 | 4.75 ± 0.36 c1 |
| Secoisolariciresinol diglucoside | ||||
| Day 10 ratio | 0.01 ± 0.24 a | 1.41 ± 0.23 b1 | 1.85 ± 0.23 b,c1 | 2.31 ± 0.23 c1 |
| Day 31 ratio | 0.43 ± 0.24 a | 0.66 ± 0.23 a1 | 1.42 ± 0.23 b1 | 1.83 ± 0.23 b1 |
| Secoisolariciresinol | ||||
| Day 10 ratio | 0.28 ± 0.26 a | 0.87 ± 0.25 b1 | 1.88 ± 0.25 c1 | 2.06 ± 0.25 c1 |
| Day 31 ratio | 0.50 ± 0.26 a | 0.62 ± 0.25 a,b1 | 1.17 ± 0.25 b,c1 | 1.80 ± 0.25 c1 |
| Enterodiol | ||||
| Day 10 ratio | −0.47 ± 0.30 a | 0.91 ± 0.29 b1 | 0.99 ± 0.29 b1 | 1.33 ± 0.29 b1 |
| Day 31 ratio | 0.32 ± 0.30 a | 0.97 ± 0.29 b1 | 1.00 ± 0.29 b1 | 2.07 ± 0.29 c1 |
| Arabinose | ||||
| Day 10 ratio | 0.50 ± 0.15 a1 | 0.14 ± 0.14 b | 0.22 ± 0.14 a,b | 0.56 ± 0.14 a1 |
| Day 31 ratio | 0.25 ± 0.15 a | −0.26 ± 0.14 b | −0.23 ± 0.14 b | 0.30 ± 0.14 a1 |
| Ribulose/xylulose | ||||
| Day 10 ratio | 0.12 ± 0.11 | 0.01 ± 0.15 | 0.22 ± 0.14 | 0.35 ± 0.14 1 |
| Day 31 ratio | 0.17 ± 0.11 | −0.01 ± 0.15 | −0.10 ± 0.14 | 0.17 ± 0.14 |
| Microbiome | Food Comparison | Mean Squares | p-Value Corrected * | p Value |
|---|---|---|---|---|
| Taxa | ||||
| Phylum | All | 0.025 | 0.929 | 0.888 |
| Family | All | 0.099 | 0.990 | 0.923 |
| Genus | All | 0.131 | 0.981 | 0.969 |
| KEGG pathways | ||||
| Arginine | All | 0.0000009 | 0.989 | 0.916 |
| Benzoate | All | 0.0000000 | 0.989 | 0.983 |
| Butyrate | All | 0.0000025 | 0.989 | 0.976 |
| Carbohydrate-active enzymes | All | 0.0000018 | 0.989 | 0.971 |
| Phenylalanine | All | 0.0000002 | 0.989 | 0.912 |
| Propionate | All | 0.0000016 | 0.989 | 0.928 |
| Tryptophan | All | 0.0000000 | 0.989 | 0.946 |
| Tyrosine | All | 0.0000002 | 0.989 | 0.903 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jewell, D.E.; Jackson, M.I.; Cochrane, C.-Y.; Badri, D.V. Feeding Fiber-Bound Polyphenol Ingredients at Different Levels Modulates Colonic Postbiotics to Improve Gut Health in Dogs. Animals 2022, 12, 627. https://doi.org/10.3390/ani12050627
Jewell DE, Jackson MI, Cochrane C-Y, Badri DV. Feeding Fiber-Bound Polyphenol Ingredients at Different Levels Modulates Colonic Postbiotics to Improve Gut Health in Dogs. Animals. 2022; 12(5):627. https://doi.org/10.3390/ani12050627
Chicago/Turabian StyleJewell, Dennis E., Matthew I. Jackson, Chun-Yen Cochrane, and Dayakar V. Badri. 2022. "Feeding Fiber-Bound Polyphenol Ingredients at Different Levels Modulates Colonic Postbiotics to Improve Gut Health in Dogs" Animals 12, no. 5: 627. https://doi.org/10.3390/ani12050627
APA StyleJewell, D. E., Jackson, M. I., Cochrane, C.-Y., & Badri, D. V. (2022). Feeding Fiber-Bound Polyphenol Ingredients at Different Levels Modulates Colonic Postbiotics to Improve Gut Health in Dogs. Animals, 12(5), 627. https://doi.org/10.3390/ani12050627






