Use of Blood Lactate in Assessment of Manual Capture Techniques of Zoo-Housed Crocodilians
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Sites and Animals
2.2. Manual Capture and Restraint Protocol
2.3. Data Collection
2.4. Data Analysis
2.4.1. Predictors of Lactate
2.4.2. Relationship between Lactate, Time from Capture to Blood Collection, and Weight
2.4.3. Visual Differences in Lactate with Capture Factors
3. Results
3.1. Predictors of Lactate
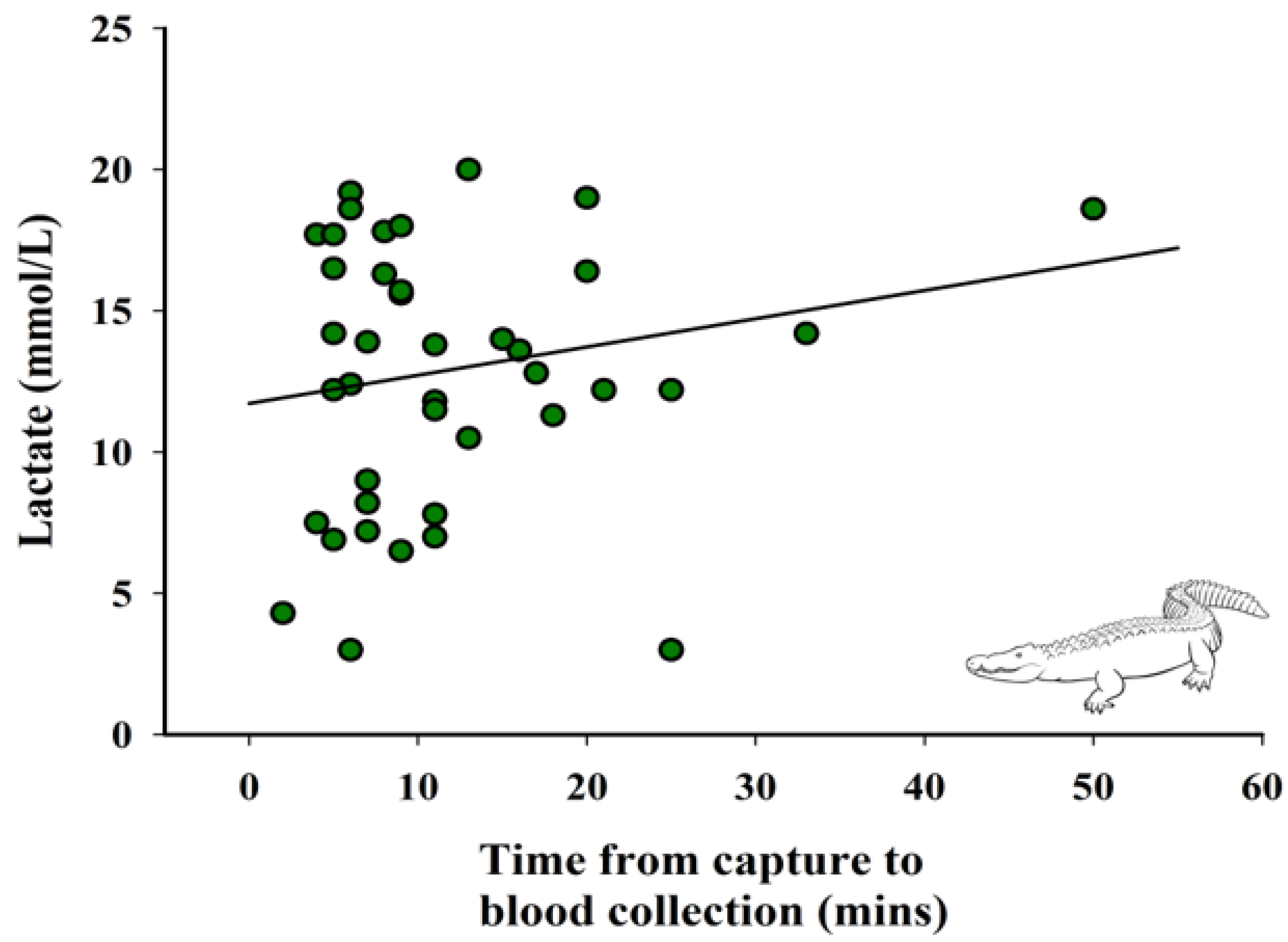
3.2. Relationship between Time from Capture to Blood Collection and Lactate
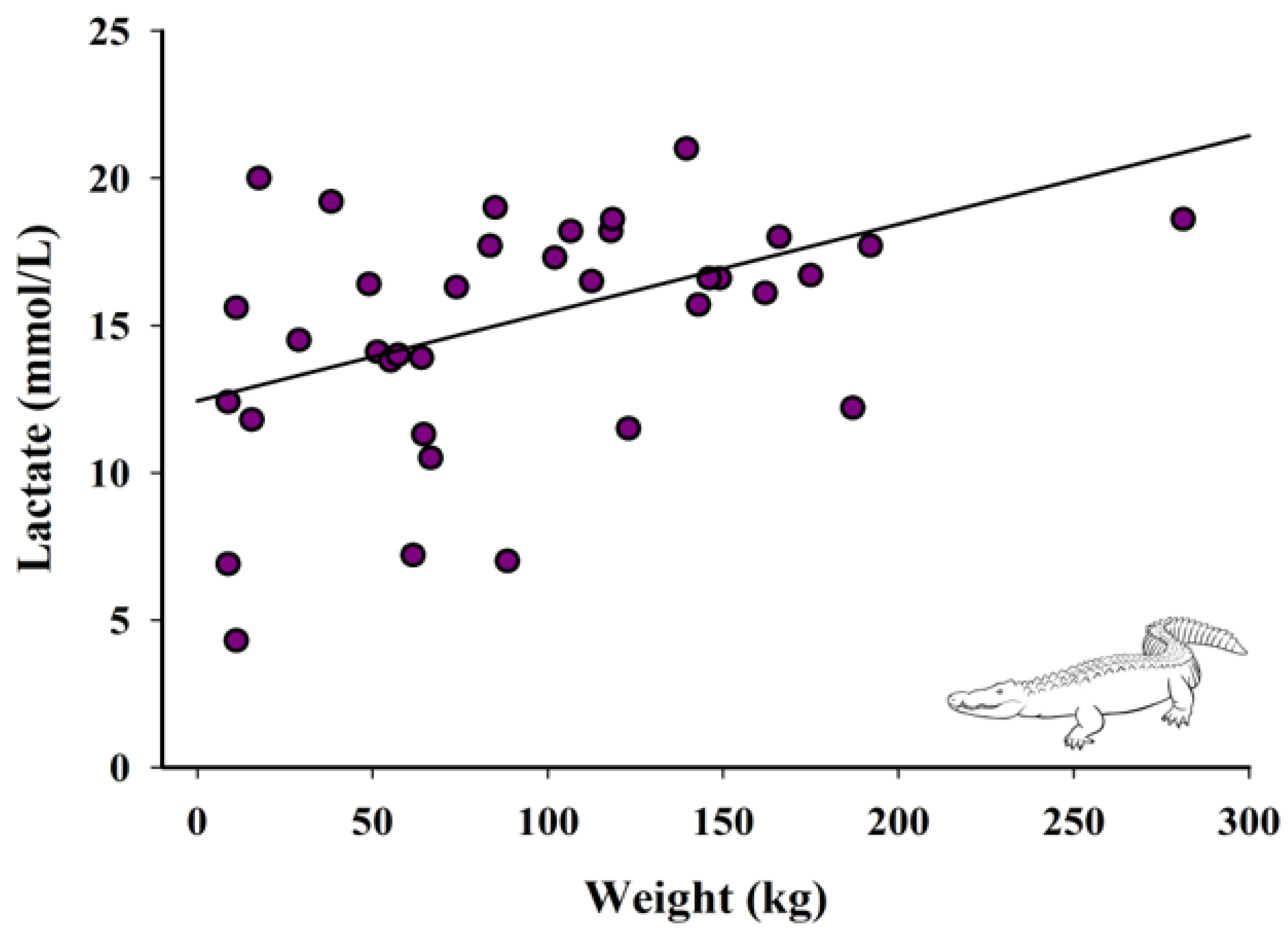
3.3. Relationship between Weight and Lactate
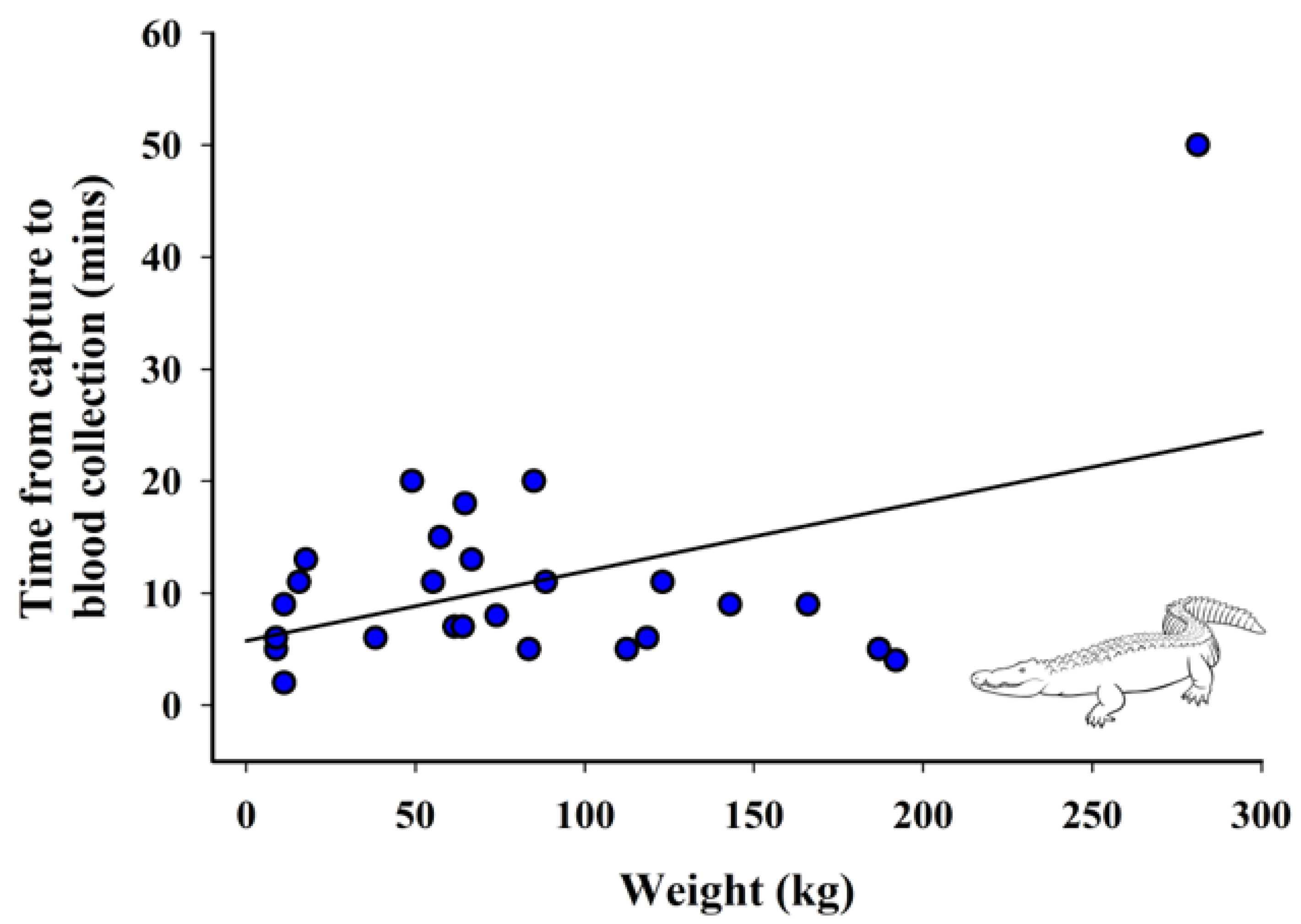
3.4. Relationship between Weight and Time from Capture to Blood Collection
3.5. Visual Differences in Lactate with Capture Factors
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Benn, A.L.; McLelland, D.J.; Whittaker, A.L. A review of welfare assessment methods in reptiles, and preliminary application of the welfare quality® protocol to the pygmy blue-tongue skink, Tiliqua adelaidensis, using animal-based measures. Animals 2019, 9, 27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Melfi, V.A.; McCormick, W.; Gibbs, A. A preliminary assessment of how zoo visitors evaluate animal welfare according to enclosure style and the expression of behavior. Anthrozoös 2004, 17, 98–108. [Google Scholar] [CrossRef]
- Lambert, H.; Carder, G.; D’Cruze, N. Given the Cold Shoulder: A Review of the Scientific Literature for Evidence of Reptile Sentience. Animals 2019, 9, 821. [Google Scholar] [CrossRef] [Green Version]
- Warwick, C.; Arena, P.; Lindley, S.; Jessop, M.; Steedman, C. Assessing reptile welfare using behavioural criteria. Practice 2013, 35, 123–131. [Google Scholar] [CrossRef] [Green Version]
- Huchzermeyer, F.W. Crocodiles—Biology, Husbandry and Diseases; CABI Publishing: Wallingford, UK, 2003. [Google Scholar]
- Isberg, S. Farming dinosaur cousins: The unique welfare challenges of farming crocodiles. In Breeding Focus 2016—Improving Welfare; Animal Genetics and Breeding Unit, University of New England: Armidale, NSW, Australia, 2016; pp. 67–76. [Google Scholar]
- Fowler, M. Restraint and Handling of Wild and Domestic Animals; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2008. [Google Scholar]
- Zeiler, G.E.; Meyer, L.C.R. Chemical capture of impala (Aepyceros melampus): A review of factors contributing to morbidity and mortality. Vet. Anaesth. Analg. 2017, 44, 991–1006. [Google Scholar] [CrossRef]
- McMahon, C.R.; Wiggins, N.L.; French, V.; McCallum, H.I.; Bowman, D.M.J.S. A report of capture myopathy in the Tas-manian pademelon (Thylogale billardierii). Anim. Welf. 2013, 22, 1–4. [Google Scholar] [CrossRef]
- Bennett, R.A. A Review of Anesthesia and Chemical Restraint in Reptiles. J. Zoo Wildl. Med. 2009, 22, 282–303. Available online: http://www.jstor.org/stable/20095160 (accessed on 13 April 2021).
- Silvestre, A.M. How to Assess Stress in Reptiles. J. Exot. Pet Med. 2014, 23, 240–243. [Google Scholar] [CrossRef]
- Wolfensohn, S.; Shotton, J.; Bowley, H.; Davies, S.; Thompson, S.; Justice, W.S.M. Assessment of Welfare in Zoo Animals: Towards Optimum Quality of Life. Animals 2018, 8, 110. [Google Scholar] [CrossRef] [Green Version]
- Wielebnowski, N. Stress and distress: Evaluating their impact for the well-being of zoo animals. J. Am. Vet. Med Assoc. 2003, 223, 973–977. [Google Scholar] [CrossRef]
- Greenberg, N.; Wingfield, J.C. Stress and Reproduction: Reciprocal Relationships. In Hormones and Reproduction in Fishes, Amphibians, and Reptiles; Norris, D., Jones, R.E., Eds.; Springer: New York, NY, USA, 1987; pp. 461–503. [Google Scholar]
- Elsey, R.M.; Joanen, T.; McNease, L.; Lance, V. Stress and plasma corticosterone levels in the american alliga-tor-relationships with stocking density and nesting success. Comp. Biochem. Physiol. Part A Physiol. 1990, 95, 55–63. [Google Scholar] [CrossRef]
- Jessop, T.S.; Tucker, A.D.; Limpus, C.J.; Whittier, J.M. Interactions between ecology, demography, capture stress, and profiles of corticosterone and glucose in a free-living population of Australian freshwater crocodiles. Gen. Comp. Endocrinol. 2003, 132, 161–170. [Google Scholar] [CrossRef]
- Hamilton, M.T.; Finger, J.W.; Elsey, R.M.; Mastromonaco, G.F.; Tuberville, T.D. Corticosterone in American alligator (Alligator mississippiensis) tail scutes: Evaluating the feasibility of using unconventional samples for investigating environmental stressors. Gen. Comp. Endocrinol. 2018, 268, 7–13. [Google Scholar] [CrossRef]
- Goessling, J.M.; Mendonça, M.T. Physiological responses of gopher tortoises (Gopherus polyphemus) to trapping. Conserv. Physiol. 2021, 9, coab003. [Google Scholar] [CrossRef] [PubMed]
- Miguel, C.; Becker, J.H.; de Freitas, B.S.; Touguinha, L.B.A.; Salvador, M.; Oliveira, G.T. Physiological effects of incidental capture and seasonality on juvenile green sea turtles (Chelonia mydas). J. Exp. Mar. Biol. Ecol. 2020, 533, 151460. [Google Scholar] [CrossRef]
- Seymour, R.S.; Bennett, A.F.; Bradford, D.F. Blood Gas Tensions and Acid-Base Regulation in the Salt-Water Crocodile, Crocodylus Porosus, at Rest and After Exhaustive Exercise. J. Exp. Biol. 1985, 118, 143–159. [Google Scholar] [CrossRef]
- Pereira, C.M.; Booth, D.T.; Bradley, A.J.; Limpus, C.J. Blood concentrations of lactate, glucose and corticosterone in dispersing hatchling sea turtles. Biol. Open 2013, 2, 63–67. [Google Scholar] [CrossRef] [Green Version]
- Pfitzer, S.; Ganswindt, A.; Fosgate, G.T.; Botha, P.J.; Myburgh, J.G. Capture of farmed Nile crocodiles (Crocodylus niloticus): Comparison of physiological parameters after manual capture and after capture with electrical stunning. Vet. Rec. 2014, 175, 304. [Google Scholar] [CrossRef] [Green Version]
- Gleeson, T.T. Patterns of Metabolic Recovery from Exercise in Amphibians and Reptiles. J. Exp. Biol. 1991, 160, 187–207. [Google Scholar] [CrossRef]
- Lang, J.W. Crocodilian Behaviour: Implications for Management. In Wildlife Management: Crocodilies and Alligators; Surrey Beatty: Sydney, Australia, 1987; pp. 273–294. [Google Scholar]
- Gleeson, B.Y.T.T.; Dalessio, P.M. Lactate and Glycogen Metabolism in the Lizard Dipsosaurus dorsalis following Exhaustive Exercise. J. Exp. Biol. 1989, 144, 377–393. [Google Scholar] [CrossRef]
- Seymour, R.S.; Webb, G.J.W.; Bennett, A.F.; Bradford, D.F. No Effect of Capture on the Physiology of Crocodylus porosus. In Wildlife Management: Crocodilies and Alligators; Surrey Beatty: Sydney, Australia, 1987. [Google Scholar]
- Gleeson, T.T. Post-exercise lactate metabolism: A comparative review of sites, pathways, and regulation. Annu. Rev. Physiol. 1996, 58, 565–581. [Google Scholar] [CrossRef] [PubMed]
- Bennett, A.F.; Seymour, R.S.; Bradford, D.F.; Webb, G.J.W. Mass-Dependence of Anaerobic Metabolism and Acid-Bas Disturbance During Activity in the Saltwater Crocodiles Crocodylus porosus. J. Exp. Biol. 1985, 118, 161–171. [Google Scholar] [CrossRef]
- Edwards, L.; Grandin, T.; Engle, T.; Porter, S.; Ritter, M.; Sosnicki, A.; Anderson, D. Use of exsanguination blood lactate to assess the quality of pre-slaughter pig handling. Meat Sci. 2010, 86, 384–390. [Google Scholar] [CrossRef] [PubMed]
- Correa, J.A.; Gonyou, H.; Torrey, S.; Widowski, T.; Bergeron, R.; Crowe, T.; Laforest, J.-P.; Faucitano, L. Welfare of Pigs Being Transported over Long Distances Using a Pot-Belly Trailer during Winter and Summer. Animals 2014, 4, 200–213. [Google Scholar] [CrossRef] [PubMed]
- Di Lorenzo, E.; Rossi, R.; Ferrari, F.; Martini, V.; Comazzi, S. Blood L-Lactate Concentration as an Indicator of Outcome in Roe Deer (Capreolus capreolus) Admitted to a Wildlife Rescue Center. Animals 2020, 10, 1066. [Google Scholar] [CrossRef]
- Klein, K.; Adamovicz, L.; Phillips, C.A.; Allender, M.C. Blood lactate concentrations in eastern box turtles (Terrapene carolina carolina) following capture by a canine search team. J. Zoo Wildl. Med. 2021, 52, 259–267. [Google Scholar] [CrossRef]
- Cerreta, A.J.; Cannizzo, S.A.; Smith, D.C.; Minter, L.J. Venous hematology, biochemistry, and blood gas analysis of free-ranging Eastern Copperheads (Agkistrodon contortrix) and Eastern Ratsnakes (Pantherophis alleghaniensis). PLoS ONE 2020, 15, e0229102. [Google Scholar] [CrossRef]
- Burgdorf-Moisuk, A.; Wack, R.; Ziccardi, M.; Larsen, R.S.; Hopper, K. Validation of lactate measurement in american fla-mingo (Phoenicopterus ruber) plasma and correlation with duration and difficulty of capture. J. Zoo Wildl. Med. 2012, 43, 450–458. [Google Scholar] [CrossRef]
- Vliet, K.A. Crocodilian Capture and Restraint. In Zoo Animal and Wildlife Immobilization and Anesthesia; West, G., Heard, D., Caulkett, N., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2014. [Google Scholar]
- Olsson, A.; Phalen, D. Comparison of biochemical stress indicators in juvenile captive estuarine crocodiles (Crocodylus Porosus) following physical restraint or chemical restraint by midazolam injection. J. Wildl. Dis. 2013, 49, 560–567. [Google Scholar] [CrossRef] [Green Version]
- Franklin, C.E.; Davis, B.M.; Peucker, S.; Stephenson, H.; Mayer, R.; Whittier, J.; Lever, J.; Grigg, G. Comparison of stress induced by manual restraint and immobilisation in the estuarine crocodile, Crocodylus porosus. J. Exp. Zool. Part A Comp. Exp. Biol. 2003, 298, 86–92. [Google Scholar] [CrossRef]
- Augustine, L.; Miller, K.; Peters, A.; Franklin, A.D.; Steinbeiser, C.M.; Brown, J.L.; Prado, N.A. Impacts of the season and reproductive status on fecal reproductive and adrenocortical steroid metabolites in zoo Cuban crocodiles (Crocodylus rhombifer). Zoo Biol. 2020, 39, 411–421. [Google Scholar] [CrossRef] [PubMed]
- Brien, M.L.; Cherkiss, M.S.; Parry, M.W.; Mazzotti, F.J. Housing Crocodilians in Captivity: Considerations for Central America and Caribbean. EDIS 2008, 2008, 1–26. [Google Scholar] [CrossRef]
- Myburgh, J.G.; Kirberger, R.M.; Steyl, J.; Soley, J.; Booyse, D.G.; Huchzermeyer, F.W.; Lowers, R.H.; Guillette, L.J., Jr. The post-occipital spinal venous sinus of the Nile crocodile (Crocodylus niloticus): Its anatomy and use for blood sample collection and intravenous infusions. J. S. Afr. Vet. Assoc. 2014, 85, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Viotto, E.V.; Navarro, J.L.; Piña, C.I. Growth Curves of Wild and Reintroduced Broad-Snouted Caimans (Caiman latirostris) and Their Management Implications. South Am. J. Herpetol. 2020, 16, 34. [Google Scholar] [CrossRef]
- Rosenberg, J.F.; Wellehan, J.F.X.; Crevasse, S.E.; Cray, C.; Stacy, N.I. Reference intervals for erythrocyte sedimentation rate, lactate, fibrinogen, hematology, and plasma protein electrophoresis in clinically healthy captive gopher tortoises (Gopherus polyphemus). J. Zoo Wildl. Med. 2018, 49, 520–527. [Google Scholar] [CrossRef] [PubMed]
- Hartzler, L.K.; Munns, S.L.; Bennett, A.F.; Hicks, J.W. Recovery from an activity-induced metabolic acidosis in the American alligator, Alligator mississippiensis. Comp. Biochem. Physiol.-A Mol. Integr. Physiol. 2006, 143, 368–374. [Google Scholar] [CrossRef]
- Patnaik, B.K. Ageing in Reptiles. Gerontology 1994, 40, 200–220. [Google Scholar] [CrossRef]
- Divers, S.J. Clinical Evaluation of Reptiles. Vet. Clin. N. Am. Exot. Anim. Pract. 1999, 2, 291–331. [Google Scholar] [CrossRef]
- McCulloch, J.S.G.; Talbot, L.M. Comparison of Weight Estimation Methods for Wild Animals and Domestic Livestock. J. Appl. Ecol. 1965, 2, 59. [Google Scholar] [CrossRef]
- Wright, J.C. Effects of Body Temperature, Mass, and Activity on Aerobic and Anaerobic Metabolism in Juvenile Crocodylus porosus. Physiol. Zool. 1986, 59, 505–513. [Google Scholar] [CrossRef]
- Kamel, S.; Gatten, R.E. Aerobic and Anaerobic Activity Metabolism of Limbless and Fossorial Reptiles. Physiol. Zool. 1983, 56, 419–429. [Google Scholar] [CrossRef]
- Hellmuth, H.; Augustine, L.; Watkins, B.; Hope, K. Using Operant Conditioning and Desensitization to Facilitate Veterinary Care with Captive Reptiles. Vet. Clin. N. Am.-Exot. Anim. Pract. 2012, 15, 425–443. [Google Scholar] [CrossRef] [PubMed]
- Hawthorne, W.H.; Goessling, J.M. A Test of Tradeoffs between Performance and Immunity in Water Snakes (Nerodia clarkii X Nerodia fasciata). Copeia 2020, 108, 453. [Google Scholar] [CrossRef]
- Nye, C.J.; Musulin, S.E.; Hanel, R.M.; Mariani, C.L. Evaluation of the Lactate Plus monitor for plasma lactate concentration measurement in dogs. J. Vet. Emerg. Crit. Care 2017, 27, 66–70. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hauss, A.A.; Stablein, C.K.; Fisher, A.L.; Greene, H.M.; Nout-Lomas, Y.S. Validation of the Lactate Plus Lactate Meter in the Horse and Its Use in a Conditioning Program. J. Equine Vet. Sci. 2014, 34, 1064–1068. [Google Scholar] [CrossRef]
- Seebacher, F.; Guderley, H.; Elsey, R.M.; Trosclair, P.L. Seasonal acclimatisation of muscle metabolic enzymes in a reptile (Alligator mississippiensis). J. Exp. Biol. 2003, 206, 1193–1200. [Google Scholar] [CrossRef] [Green Version]


| Species | Sex | Average Weight (kg) | Lactate (mmol/L) |
|---|---|---|---|
| A. mississippiensis | F = 9 M = 10 | F = 11.2 M = 281.22 | 10.02 ± 4.94 (n = 19) |
| A. sinensis | F = 1 | F = 15.6 | 11.8 (n = 1) |
| C. acutus | M = 1 Unknown = 15 | M = 166 U = 128.77 | 16.59 ± 2.4 (n = 16) |
| C. halli | M = 2 | M = 59 | 12.3 ± 2.54 (n = 2) |
| C. intermedius | F = 1 | F = 64.6 | 11.30 (n = 1) |
| C. rhombifer | F = 3 M = 1 | M = 55.2 F = 69.13 | 10.5 ± 3.93 (n = 4) |
| C. suchus | F = 2 M = 2 | M = 93.5 F = 39 | 16.8 ± 1.87 (n = 4) |
| G. gangeticus | M = 2 | M = 162 | 8.65 ± 10.53 (n = 2) |
| P. trigonatus | F = 1 M = 1 | M = 38.2 F = 17.6 | 19.6 ± 0.56 (n = 2) |
| T. schlegelii | M = 2 | M = 8.8 | 9.65 ± 3.88 (n = 2) |
| Age Class | Lactate (mmol/L) |
|---|---|
| Adult (n = 44) | 13.90 ± 4.75 A |
| Non-adult (n = 9) | 8.51 ± 4.10 B |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Molinaro, H.G.; Anderson, G.S.; Gruny, L.; Sperou, E.S.; Heard, D.J. Use of Blood Lactate in Assessment of Manual Capture Techniques of Zoo-Housed Crocodilians. Animals 2022, 12, 397. https://doi.org/10.3390/ani12030397
Molinaro HG, Anderson GS, Gruny L, Sperou ES, Heard DJ. Use of Blood Lactate in Assessment of Manual Capture Techniques of Zoo-Housed Crocodilians. Animals. 2022; 12(3):397. https://doi.org/10.3390/ani12030397
Chicago/Turabian StyleMolinaro, Holly Grace, Gen S. Anderson, Lauren Gruny, Emily S. Sperou, and Darryl J. Heard. 2022. "Use of Blood Lactate in Assessment of Manual Capture Techniques of Zoo-Housed Crocodilians" Animals 12, no. 3: 397. https://doi.org/10.3390/ani12030397
APA StyleMolinaro, H. G., Anderson, G. S., Gruny, L., Sperou, E. S., & Heard, D. J. (2022). Use of Blood Lactate in Assessment of Manual Capture Techniques of Zoo-Housed Crocodilians. Animals, 12(3), 397. https://doi.org/10.3390/ani12030397







