Carcass Quality Profiles and Associated Genomic Regions of South African Goat Populations Investigated Using Goat SNP50K Genotypes
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animal Description and Management
2.2. Blood Collection, DNA Isolation and SNP Genotyping
2.3. Slaughter Procedure and Carcass Quality Measurements
2.4. Statistical Analysis of Carcass Quality Data
2.5. Association Analysis
2.6. Gene Annotation and SNP Association
3. Results
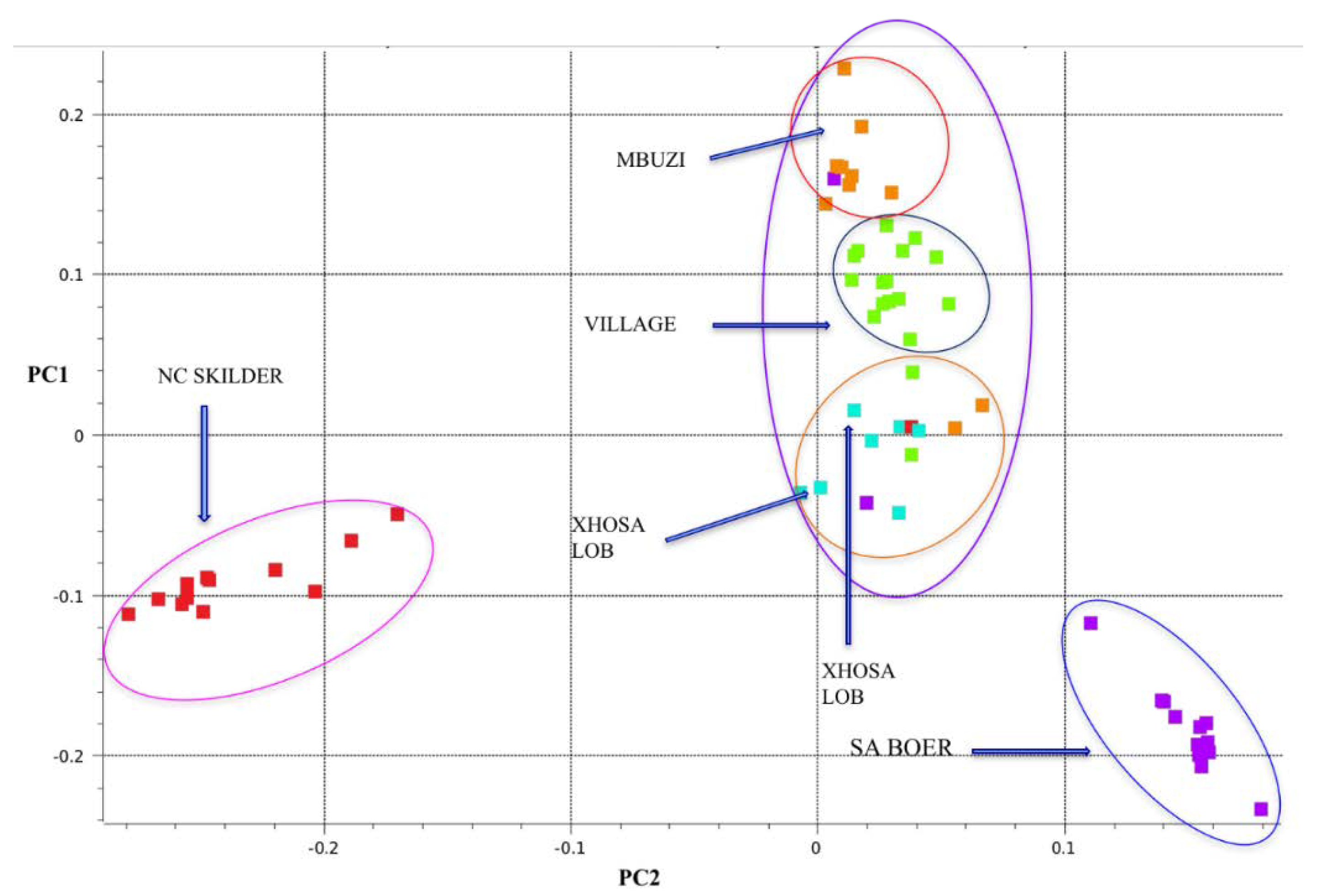
3.1. SNP Genotype Quality Control and Population Structure
3.2. Carcass Quality Traits
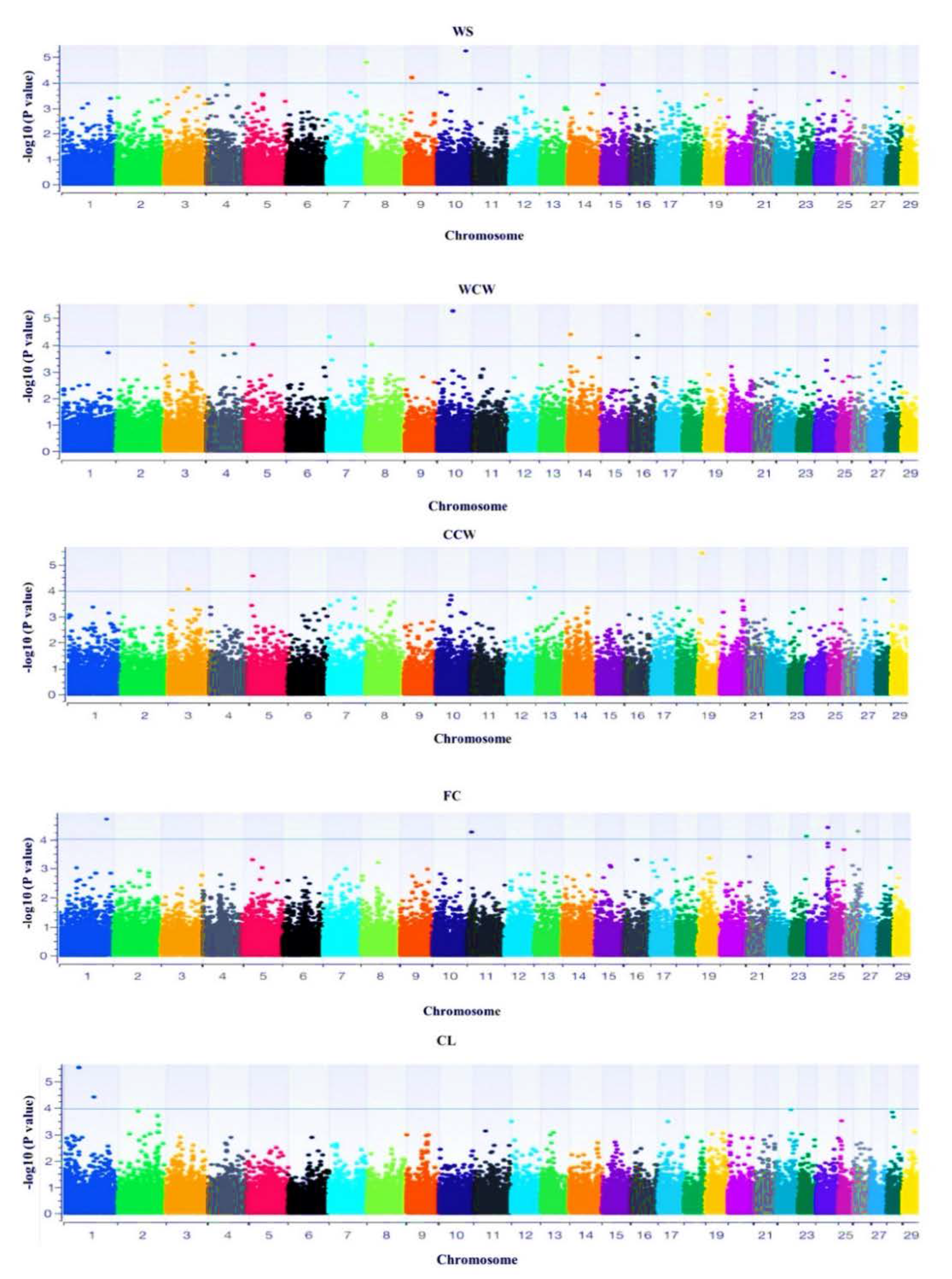
3.3. Genome-Wide Association
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mazhangara, I.R.; Chivandi, E.; Mupangwa, J.F.; Muchenje, V. The potential of goat meat in the red meat industry. Sustainability 2019, 11, 3671. [Google Scholar] [CrossRef] [Green Version]
- Lehohla, P. Community Survey Agricultural Households; Statistics South Africa: Pretoria, South Africa, 2016.
- Dzomba, E.F.; Mdladla, K.; Ncube, K.T.; Muchadeyi, F.C. Genomics for the Improvement of Productivity and Robustness of South African Goat Breeds; Springer: Berlin/Heidelberg, Germany, 2018. [Google Scholar]
- Morokolo, B. A Profile of the South African Goat Market Value Chain; Directorate Marketing Private Bag X 15 ARCADIA: South Africa, 2011; pp. 1–37. [Google Scholar]
- Webb, E.C. Goat meat production, composition, and quality. Anim. Front. 2014, 4, 33–37. [Google Scholar] [CrossRef] [Green Version]
- Casey, N.H.; Webb, E.C. Managing goat production for meat quality. Small Rumin. Res. 2010, 89, 218–224. [Google Scholar] [CrossRef] [Green Version]
- Mdladla, K.; Dzomba, E.F.; Huson, H.J.; Muchadeyi, F.C. Population genomic structure and linkage disequilibrium analysis of South African goat breeds using genome-wide SNP data. Anim. Genet. 2016, 47, 471–482. [Google Scholar] [CrossRef]
- Sapkota, S.; Kolakshyapati, M.; Gairhe, S.; Upadhyay, N.; Acharya, Y. Boer Goat Production Performance, Constraints and Opportunities in Nepal Monitoring and Evaluation Division 123, Planning Division 45 NARC. Imp. J. Interdiscip. Res. 2016, 2, 491–495. [Google Scholar]
- Jw, M. A Guide to the Identification of the Natural Indigenous Goats of Southern Africa; Compil Indig Veld Goat Breeders Soc: Pretoria, South Africa, 2007. [Google Scholar]
- Ncube, K.T.; Mdladla, K.; Dzomba, E.F.; Muchadeyi, F.C. Targeted high-throughput growth hormone 1 gene sequencing reveals high within-breed genetic diversity in South African goats. Anim. Genet. 2016, 47, 382–385. [Google Scholar] [CrossRef]
- DAFF. Goat Training Manual; District Livestock Service Office: Gorkha, Nepal, 2016; p. 1.
- Pophiwa, P.; Webb, E.C.; Frylinck, L. Meat quality characteristics of two South African goat breeds after applying electrical stimulation or delayed chilling of carcasses. Small Rumin. Res. 2016, 145, 107–114. [Google Scholar] [CrossRef]
- Ncube, K.T.; Hadebe, K.; Dzomba, E.F.; Soma, P.; Frylinck, L.; Muchadeyi, F.C. Relationship between population genomic structure and growth profiles of South African goats under different production systems. Trop. Anim. Health Prod. 2020, 52, 1277–1286. [Google Scholar] [CrossRef] [PubMed]
- Tosser-Klopp, G.; Bardou, P.; Bouchez, O.; Cabau, C.; Crooijmans, R.; Dong, Y.; Donnadieu-Tonon, C.; Eggen, A.; Heuven, H.C.M.; Jamli, S.; et al. Design and characterization of a 52K SNP chip for goats. PLoS ONE 2014, 9, e86227. [Google Scholar]
- Lashmar, S.F.; Visser, C.; Van Marle-Köster, E. Validation of the 50k Illumina goat SNP chip in the South African Angora goat. S. Afr. J. Anim. Sci. 2015, 45, 56–59. [Google Scholar] [CrossRef] [Green Version]
- Rahmatalla, S.A.; Arends, D.; Reissmann, M.; Wimmers, K.; Reyer, H.; Brockmann, G.A. Genome-wide association study of body morphological traits in Sudanese goats. Anim. Genet. 2018, 49, 478–482. [Google Scholar] [CrossRef] [PubMed]
- Mdladla, K.; Dzomba, E.F.; Muchadeyi, F.C. Landscape genomics and pathway analysis to understand genetic adaptation of South African indigenous goat populations. Heredity 2018, 120, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Purcell, S.; Neale, B.; Todd-Brown, K.; Thomas, L.; Ferreira, M.A.R.; Bender, D.; Maller, J.; Sklar, P.; de Bakker, P.I.W.; Daly, M.J.; et al. PLINK: A Tool Set for Whole-Genome Association and Population-Based Linkage Analyses. Am. J. Hum. Genet. 2007, 81, 559–575. [Google Scholar] [CrossRef] [Green Version]
- Cloete, J.J.E.; Hoffman, L.C.; Cloete, S.W.P.; Fourie, J.E. A comparison between the body composition, carcass characteristics and retail cuts of South African Mutton Merino and Dormer sheep. S. Afr. J. Anim. Sci. 2004, 34, 44–51. [Google Scholar] [CrossRef]
- Pophiwa, P.; Webb, E.C.; Frylinck, L. Carcass and meat quality of Boer and indigenous goats of South Africa under delayed chilling conditions. S. Afr. J. Anim. Sci. 2017, 47, 749–803. [Google Scholar] [CrossRef] [Green Version]
- Assan, N. Influence of Non-genetic Factors Weight and Carcass Traits in Indigenous Matebele Goat. J. Anim. Prod. Adv. 2012, 2, 57–64. [Google Scholar]
- Tshabalala, P.A.; Strydom, P.E.; Webb, E.C.; De Kock, H.L. Meat quality of designated South African indigenous goat and sheep breeds. Meat Sci. 2003, 65, 563–570. [Google Scholar] [CrossRef]
- Higgins, M.G.; Kenny, D.A.; Fitzsimons, C.; Blackshields, G.; Coyle, S.; McKenna, C.; McGee, M.; Morris, D.K.; Waters, S.M. The effect of breed and diet type on the global transcriptome of hepatic tissue in beef cattle divergent for feed efficiency. BMC Genom. 2019, 20, 525. [Google Scholar] [CrossRef] [PubMed]
- McCabe, M.; Waters, S.; Morris, D.; Kenny, D.; Lynn, D.; Creevey, C. RNA-seq analysis of differential gene expression in liver from lactating dairy cows divergent in negative energy balance. BMC Genom. 2012, 13, 193. [Google Scholar] [CrossRef] [Green Version]
- Berkowicz, E.W.; Magee, D.A.; Berry, D.P.; Sikora, K.M.; Howard, D.J.; Mullen, M.P.; Evans, R.D.; Spillane, C.; MacHugh, D.E. Single nucleotide polymorphisms in the imprinted bovine insulin-like growth factor 2 receptor gene (IGF2R) are associated with body size traits in Irish Holstein-Friesian cattle. Anim. Genet. 2012, 43, 81–87. [Google Scholar] [CrossRef]
- De Brito, G.F.; Ponnampalam, E.N.; Hopkins, D.L. The Effect of Extensive Feeding Systems on Growth Rate, Carcass Traits, and Meat Quality of Finishing Lambs. Compr. Rev. Food Sci. Food Saf. 2017, 16, 23–38. [Google Scholar] [CrossRef]
- Lee, K.T.; Lee, Y.M.; Alam, M.; Choi, B.H.; Park, M.R.; Kim, K.S.; Kim, T.-H.; Kim, J.-J. A whole genome association study on meat quality traits using high density SNP chips in a cross between Korean native pig and landrace. Asian-Australas. J. Anim. Sci. 2012, 25, 1529–1539. [Google Scholar] [CrossRef] [PubMed]
- Lian, T.; Wang, L.; Liu, Y. A new insight into the role of calpains in post-mortem meat tenderization in domestic animals: A review. Asian-Australas. J. Anim. Sci. 2013, 26, 443–454. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Idkowiak-Baldys, J.; Santhanam, U.; Buchanan, S.M.; Pfaff, K.L.; Rubin, L.L.; Lyga, J. Growth differentiation factor 11 (GDF11) has pronounced effects on skin biology. PLoS ONE 2019, 14, e0218035. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jeong, H.; Song, K.D.; Seo, M.; Caetano-Anolles, K.; Kim, J.; Kwak, W.; Oh, J.; Kim, E.; Jeong, D.K.; Cho, S.; et al. Exploring evidence of positive selection reveals genetic basis of meat quality traits in Berkshire pigs through whole genome sequencing. BMC Genet. 2015, 16, 104. [Google Scholar] [CrossRef] [Green Version]
- Frylinck, L.; van Wyk, G.L.; Smith, T.P.L.; Strydom, P.E.; van Marle-Köster, E.; Webb, E.C.; Koohmaraie, M.; Smith, M.F. Evaluation of biochemical parameters and genetic markers for association with meat tenderness in South African feedlot cattle. Meat Sci. 2009, 83, 657–665. [Google Scholar] [CrossRef] [PubMed]
- Chanoine, J.P.; De Waele, K.; Walia, P. Ghrelin and the growth hormone secretagogue receptor in growth and development. Int. J. Obes. 2009, 33, S48–S52. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manjula, P.; Choi, N.; Seo, D.; Lee, J.H. POU class 1 homeobox 1 gene polymorphisms associated with growth traits in Korean native chicken. Asian-Australas. J. Anim. Sci. 2018, 31, 643. [Google Scholar] [CrossRef]
| γBreed | |||||||
|---|---|---|---|---|---|---|---|
| SAB | NCS | XL | MBZ | VTI | VTE | Significance | |
| * Trait | (p < F) | ||||||
| WS (kg) | 40.9 a ± 3.6 | 33.1 b ± 4.1 | 29.8 c ± 5.9 | 23.0 d ± 3.4 | 24.2 d ± 2.8 | 22.0 d ± 4.6 | <0.001 |
| WCW (kg) | 17.2 a ± 2.3 | 14.1 b ± 1.9 | 12.2 c ± 2.6 | 9.4 d ± 1.5 | 9.9 d ± 1.1 | 9.1 d ± 2.0 | <0.001 |
| CCW (kg) | 16.3 a ± 2.3 | 12.9 b ± 1.7 | 11.4 b ± 2.4 | 8.7 c ± 1.4 | 9.2 c ± 1.2 | 8.4 c ± 1.9 | <0.001 |
| FC | 1.3 ± 0.5 | 1.1 ± 0.5 | 1.2 ± 0.4 | 0.4 bc ± 0.2 | 0.4 bc ± 0.3 | 0.3 ± 0.00 | <0.001 |
| DP (%) | 41.9 ± 2.4 | 42.5 ± 2.5 | 41.4 ± 1.7 | 40.9 ± 1.5 | 40.8 ± 1.7 | 41.2 ± 2.6 | 0.353 |
| CL (%) | 5.5 ± 1.7 | 8.4 ± 3.5 | 6.7 ± 1.7 | 7.4 ± 1.9 | 6.6 ± 2.1 | 7.8 y ± 2.7 | 0.059 |
| Breed × Sex | Significance * | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SAB | NCS | XL | MBZ | VTI | VTE | ||||||||
| Does | Bucks | Does | Bucks | Does | Bucks | Does | Bucks | Does | Bucks | Does | Bucks | (p < F) | |
| WS (kg) | 42.7 a ± 3.7 | 39.2 ab ± 3.0 | 31.9 c ± 4.1 | 34.9 bc ± 3.7 | 25.8 d ± 4.8 | 34.4 c ± 2.0 | 23.9 de ± 3.4 | 21.3 de ± 3.1 | 24.1 de ± 3.5 | 24.4 de ± 1.9 | 21.0 e ± 4.5 | 23.7 de ± 5.1 | 0.004 |
| WCW (kg) | 18.0 a ± 2.5 | 16.1 ab ± 1.6 | 13.4 c ± 1.9 | 15.1 bc ± 1.5 | 10.5 d ± 1.9 | 14.3 bc ± 1.4 | 9.7 d ± 1.5 | 8.9d ± 1.6 | 9.9 d ± 1.4 | 9.9d ± 0.9 | 8.5 d ± 1.7 | 10.0 d ± 2.4 | 0.004 |
| CCW (kg) | 17.1 a ± 2.4 | 15.0 ab ± 1.4 | 12.4 c ± 1.9 | 13.6 bc ± 1.1 | 9.9 d ± 1.8 | 13.3 bc ± 1.5 | 9.0 d ± 1.5 | 8.2 d ± 1.4 | 9.2 d ± 1.5 | 9.2d ± 0.9 | 7.9 d ± 1.8 | 9.2 d ± 2.1 | 0.006 |
| DP (%) | 42.6 ± 2.8 | 41.0 ± 1.2 | 42.0 ± 2.4 | 43.3 ± 2.8 | 41.3 ± 2.2 | 41.5 ± 1.1 | 40.4 ± 0.9 | 41.7 ± 2.1 | 40.9 ± 2.3 | 40.6 ± 1.0 | 40.6 ± 2.4 | 42.1 ± 3.1 | 0.492 |
| CL (%) | 4.8 ± 0.8 | 6.4 ± 2.1 | 7.5 ± 2.8 | 9.8 ± 4.2 | 6.2 ± 1.2 | 7.3 ± 2.1 | 7.4 ± 2.3 | 7.5 ± 1.0 | 6.5 ± 1.8 | 6.8 ± 2.8 | 7.7 ± 3.2 | 8.0 ± 2.3 | 0.862 |
| Trait | SNP | Chr | Position | p-Value | MAF | Gene | KEGG Pathway |
|---|---|---|---|---|---|---|---|
| WS | snp10578-scaffold1376-2185653 | 8 | 72,415,408 | 0.005 | 0.384 | CDCA2 | - |
| snp31819-scaffold356-851991 | 8 | 99,190,229 | 0.019 | 0.289 | MUSK | - | |
| snp51334-scaffold750-1385368 | 8 | 79,156,981 | 0.031 | 0.402 | GAS1 | Hedgehog signaling pathway | |
| snp42388-scaffold56-1009293 | 8 | 87,724,933 | 0.039 | 0.100 | GADD45G | FoxO signaling pathway, p53 signaling pathway, MAPK signaling pathway, NF-kappa B signaling pathway, apoptosis | |
| snp53226-scaffold802-11917 | 9 | 14,759,064 | 0.002 | 0.471 | FABP7 | PPAR signaling pathway | |
| snp36803-scaffold447-444443 | 9 | 82,960,991 | 0.003 | 0.478 | IGF2R | Endocytosis, lysosome | |
| snp39508-scaffold501-3016606 | 10 | 17,332,120 | 0.015 | 0.435 | VRTN | - | |
| snp16896-scaffold1766-13800 | 25 | 1,378,654 | 0.027 | 0.492 | GFER | - |
| Trait | SNP | Chr | Position | p-Value | MAF | Gene | KEGG Pathway |
|---|---|---|---|---|---|---|---|
| WCW | snp56476-scaffold89-151841 | 3 | 11,055,188 | 0.001 | 0.42 | THRAP3 | - |
| snp44023-scaffold595-5580840 | 3 | 89,714,538 | 0.002 | 0.196 | CAPZA1 | Endocytosis | |
| snp5156-scaffold118-634690 | 3 | 84,590,504 | 0.014 | 0.413 | VAV3 | B cell receptor signaling pathway, Leukocyte transendothelial migration, Rap1 signaling pathway, cAMP signaling pathway, chemokine signaling pathway, focal adhesion, natural kille mediated cyctoxicity, T cell receptor sigbaling pathway, Fc epsilon RI signaling pathway, Yersinia infection, lipid and atheroscierosis | |
| snp15511-scaffold164-2927845 | 3 | 41,839,503 | 0.039 | 0.486 | INSL5 | Relaxin signaling pathway, neuroactive ligand receptor interaction | |
| snp11534-scaffold1421-222892 | 10 | 16,932,843 | 0.001 | 0.268 | LTBP2 | - | |
| snp16574-scaffold1748-66549 | 10 | 15,612,288 | 0.005 | 0.199 | TGFB3 | Cytokine–cytokine receptor interaction, MAPK signaling pathway, FoxO signaling pathway, TGF-beta signaling pathway, Hippo signaling pathway, Chagas disease, leishmaniasis, toxoplamosis, tuberculosis | |
| snp39508-scaffold501-3016606 | 10 | 17,332,120 | 0.009 | 0.434 | VRTN | - | |
| snp11543-scaffold1421-651926 | 10 | 16,503,809 | 0.002 | 0.427 | PGF | MAPK signaling pathway, Ras signaling pathway, Rap1 signaling pathway, focal adhesion, PI3K-Akt signaling pathway, phospholipase D signaling pathway, neuroactive ligand receptor interaction | |
| snp23543-scaffold237-2500414 | 14 | 46,657,313 | 0.008 | 0.364 | MSC | Staphylococcus aureus infection | |
| snp17242-scaffold18-837754 | 16 | 24,869,668 | 0.012 | 0.435 | CAPN8 | - | |
| CAPN2 | Necroptosis, protein processing in endoplasmic reticulum, apoptosis, cellular senescence, focal adhesion | ||||||
| snp30146-scaffold331-1127999 | 16 | 49,917,608 | 0.049 | 0.289 | PERM1 | - | |
| snp9015-scaffold1328-778798 | 19 | 28,743,439 | 0.012 | 0.383 | GAS7 | - | |
| snp43335-scaffold5775-55878 | 19 | 55,399,362 | 0.032 | 0.478 | GRB2 | Growth hormone synthesis, secretion and action, Insulin signaling pathway, ErbB signaling pathway, T cell receptor sognaling pathway, EGFR tyrosine kinase inhibitor resistance, endocrine resistance, Ras signaling pathway, MAPK signaling pathway, FoxO signaling pathway, mTOR signaling pathway, PI3K-Akt signaling pathway, signaling pathways regulating pluripotency of stem cells, natural killer cell mediated cyctoxity, B cell receptor signaling pathway, Fc epsilon RI signaling pathway, thermogenesis, neurotrophin signaling pathway, JAK-STAT signaling pathway | |
| snp55723-scaffold864-1874947 | 27 | 25,697,142 | 0.001 | 0.407 | PDGFRL | - | |
| snp30755-scaffold34-2405052 | 27 | 4,053,757 | 0.001 | 0.478 | THRB | Thyroid hormone signaling pathway, neuroactive ligand-receptor interaction | |
| snp44450-scaffold604-977126 | 27 | 11,448,725 | 0.024 | 0.301 | FGFR1 | Parathyroid hormone synthesis, secretion and action, adherens junction, thermogenesis, MAPK signaling pathway, Ras signaling pathway, Rap1 signaling pathway, calcium signaling pathway, regulation, signaling pathways regulating pluripotency of stem cells | |
| snp55724-scaffold864-1906315 | 27 | 25,665,774 | 0.039 | 0.493 | PDGFRL | - |
| Trait | SNP | Chr | Position | p-Value | MAF | Gene | KEGG Pathway |
|---|---|---|---|---|---|---|---|
| CCW | snp11922-scaffold144-3610093 | 3 | 68,043,979 | 0.004 | 0.435 | TGFBR3 | - |
| snp15489-scaffold164-1962025 | 3 | 42,805,323 | 0.005 | 0.472 | GADD45A | FoxO signaling pathway, p53 sigaling pathway, MAPK signaling pathway, NF-kapp B signaling pathway, cellular senescence | |
| snp11896-scaffold144-2409377 | 3 | 69,244,695 | 0.009 | 0.311 | GFI1 | - | |
| snp43970-scaffold595-3028771 | 3 | 92,266,607 | 0.020 | 0.486 | TSHB | Thyroid hormone synthesis, cAMP signaling pathway Neuroactive-ligand receptor interaction, regulation of lipolysis in adipocytes | |
| NGF | neurotrophin signaling pathway, apoptosis, inflammatory mediator regulation of TRP channesl, aldosterone synthesis and secretion, MAPK signaling pathway, Ras signaling pathway, Rap1 signaling pathway, PI3-Akt signaling pathway, calcium signaling pathway, NF-kappa B signaling pathway, cortisol synthesis and secretion | ||||||
| snp12164-scaffold1450-542494 | 5 | 99,721,587 | 0.0020 | 0.478 | DPPA3 | - | |
| GDF3 | Cytokine–cytokine receptor interaction | ||||||
| snp48908-scaffold698-1623875 | 5 | 102,631,469 | 0.023 | 0.400 | ING4 | Circadian entrainment, retrograde endocannabinoid signaling, relaxin signaling pathway, GABaergic synapse, MAPK signaling pathway, Ras signaling pathway, PI3K-Akt signaling pathway, cardiac muscle contraction, serotonergic synapse, oxytocin signaling pathway | |
| snp12642-scaffold1482-13523 | 5 | 56,734,400 | 0.029 | 0.130 | GDF11 | Cytokine–cytokine receptor interaction | |
| snp43888-scaffold593-832260 | 5 | 92,679,764 | 0.038 | 0.136 | EPS8 | - | |
| RERG | - | ||||||
| snp10950-scaffold1393-138987 | 5 | 30,292,487 | 0.042 | 0.261 | LMBR1L | - |
| Trait | SNP | Chr | Position | p-Value | MAF | Gene | KEGG Pathway |
|---|---|---|---|---|---|---|---|
| FC | snp26446-scaffold276-6029619 | 1 | 94,587,913 | 0.011 | 0.420 | GHSR | Growth hormone synthesis, secretion, and action; neuroactive ligand receptor interaction, cAMP signaling pathway |
| snp37534-scaffold46-1530591 | 1 | 151,789,185 | 0.028 | 0.272 | COL6A5 | ECM–receptor interaction, PI3K-Akt signaling pathway, focal adhesion, protein digestion and absorption | |
| COLQ | - | ||||||
| CAPN7 | - | ||||||
| snp36302-scaffold435-1967666 | 1 | 74,307,126 | 0.029 | 0.351 | FGF12 | - | |
| snp23054-scaffold230-2979377 | 1 | 130,211,629 | 0.032 | 0.100 | MRAS | Ras signaling pathway, C-type lectin receptor signaling pathway, MAPK signaling pathway, Rap1 signaling pathway, phospholipase D signaling pathway, cellular senescence, apelin signaling pathway, regulation of cytoskeleton | |
| snp20787-scaffold204-4829802 | 11 | 77,889,065 | 0.042 | 0.447 | LDAH | - | |
| GDF7 | Axon guidance, cytokine–cytokine receptor interaction, TGF-beta signaling pathway, Hippo signaling pathway |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ncube, K.T.; Dzomba, E.F.; Hadebe, K.; Soma, P.; Frylinck, L.; Muchadeyi, F.C. Carcass Quality Profiles and Associated Genomic Regions of South African Goat Populations Investigated Using Goat SNP50K Genotypes. Animals 2022, 12, 364. https://doi.org/10.3390/ani12030364
Ncube KT, Dzomba EF, Hadebe K, Soma P, Frylinck L, Muchadeyi FC. Carcass Quality Profiles and Associated Genomic Regions of South African Goat Populations Investigated Using Goat SNP50K Genotypes. Animals. 2022; 12(3):364. https://doi.org/10.3390/ani12030364
Chicago/Turabian StyleNcube, Keabetswe Tebogo, Edgar Farai Dzomba, Khanyisile Hadebe, Pranisha Soma, Lorinda Frylinck, and Farai Catherine Muchadeyi. 2022. "Carcass Quality Profiles and Associated Genomic Regions of South African Goat Populations Investigated Using Goat SNP50K Genotypes" Animals 12, no. 3: 364. https://doi.org/10.3390/ani12030364
APA StyleNcube, K. T., Dzomba, E. F., Hadebe, K., Soma, P., Frylinck, L., & Muchadeyi, F. C. (2022). Carcass Quality Profiles and Associated Genomic Regions of South African Goat Populations Investigated Using Goat SNP50K Genotypes. Animals, 12(3), 364. https://doi.org/10.3390/ani12030364







