Contamination Status of Pet Cats in Thailand with Organohalogen Compounds (OHCs) and Their Hydroxylated and Methoxylated Derivatives and Estimation of Sources of Exposure to These Contaminants
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling
2.2. Chemicals
2.3. Analysis of Organohalogen Compounds
2.4. Analysis of Lipid Contents in Cat Sera
2.5. Quality Assurance/Quality Control
2.6. Estimation of Daily Intake
2.7. Statistical Analysis
3. Results
3.1. OHC Contamination in Cat Sera
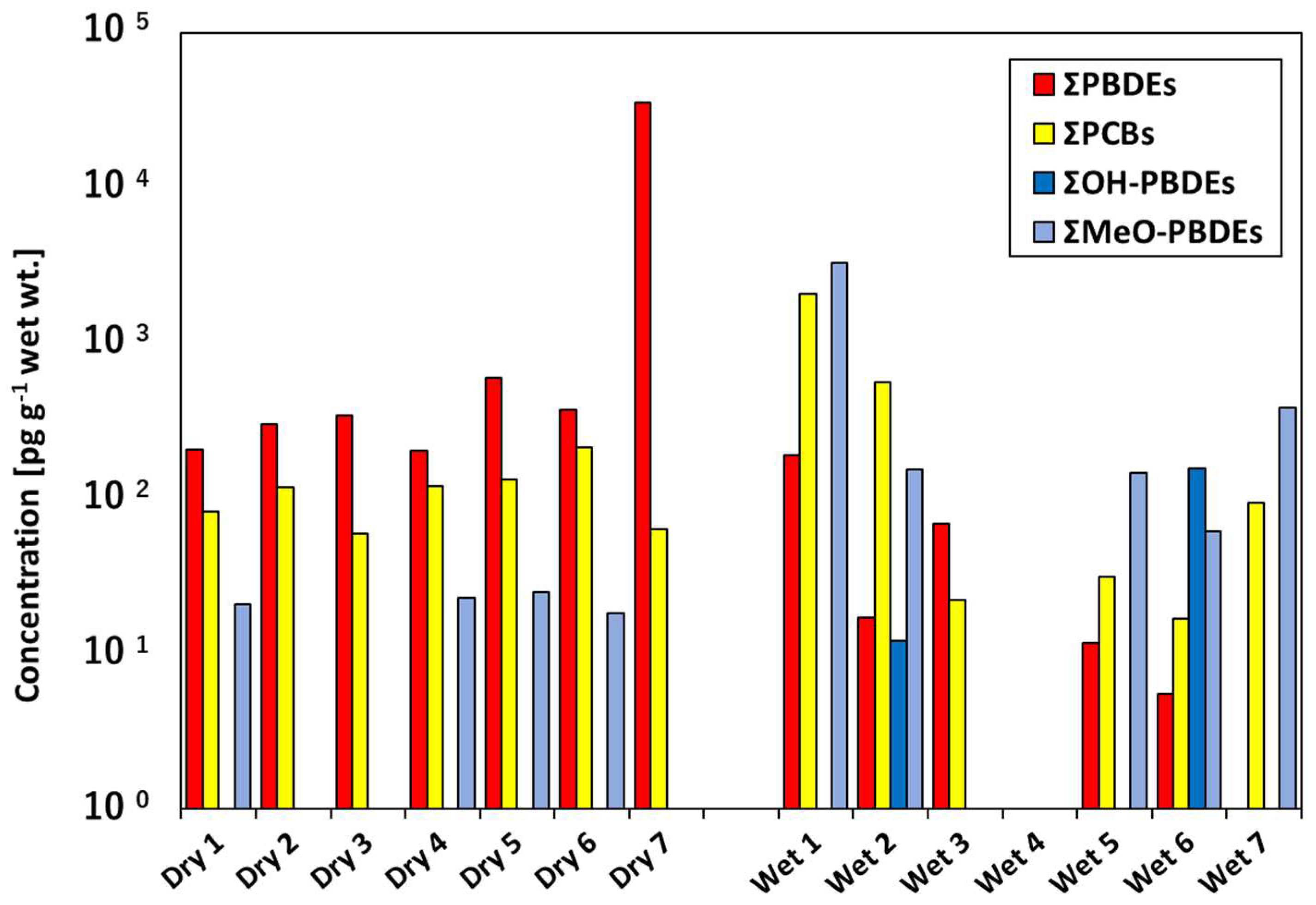
3.2. OHC Contamination in Cat Food Samples
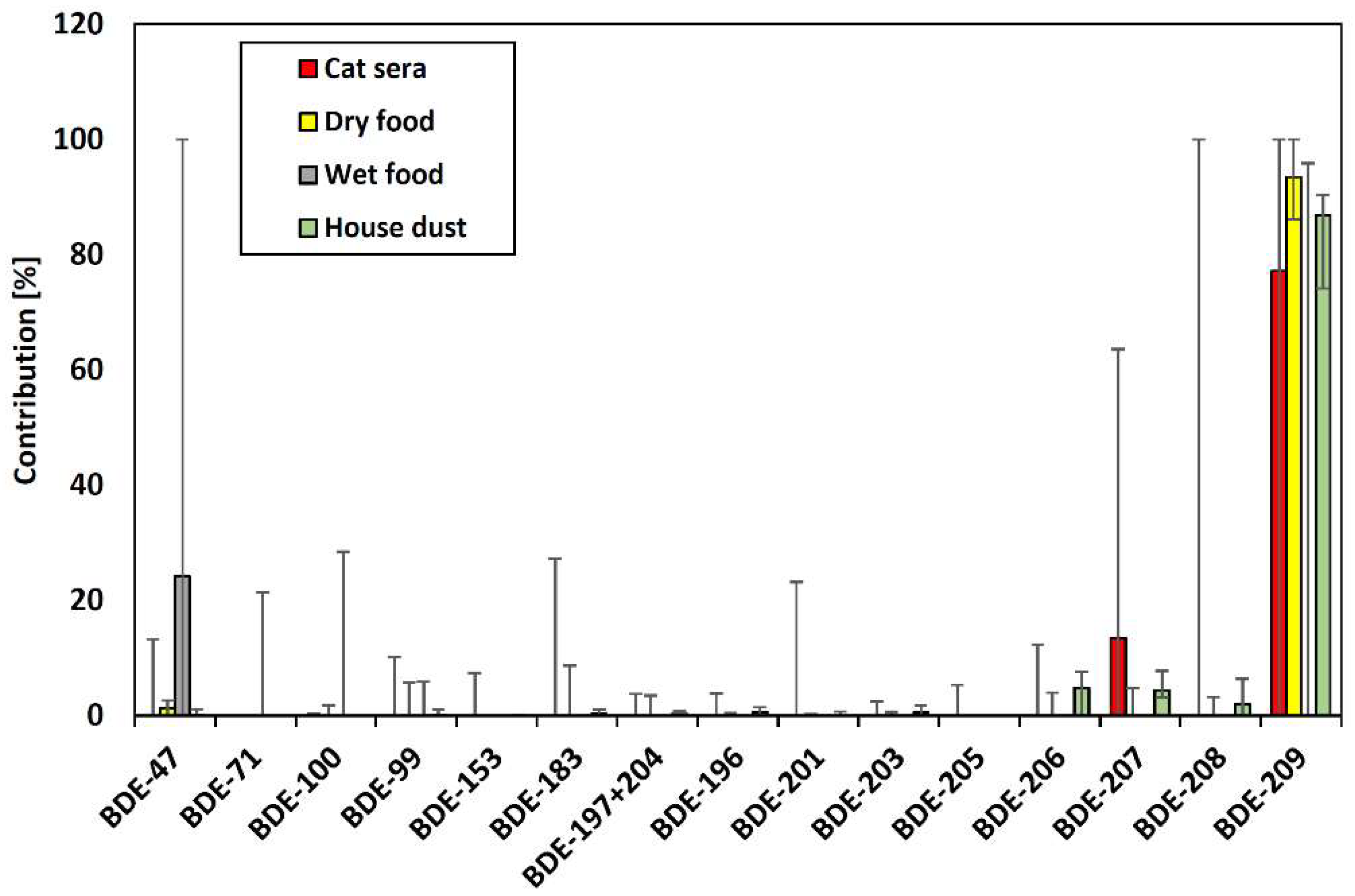
3.3. OHC Contamination in House Dust Samples
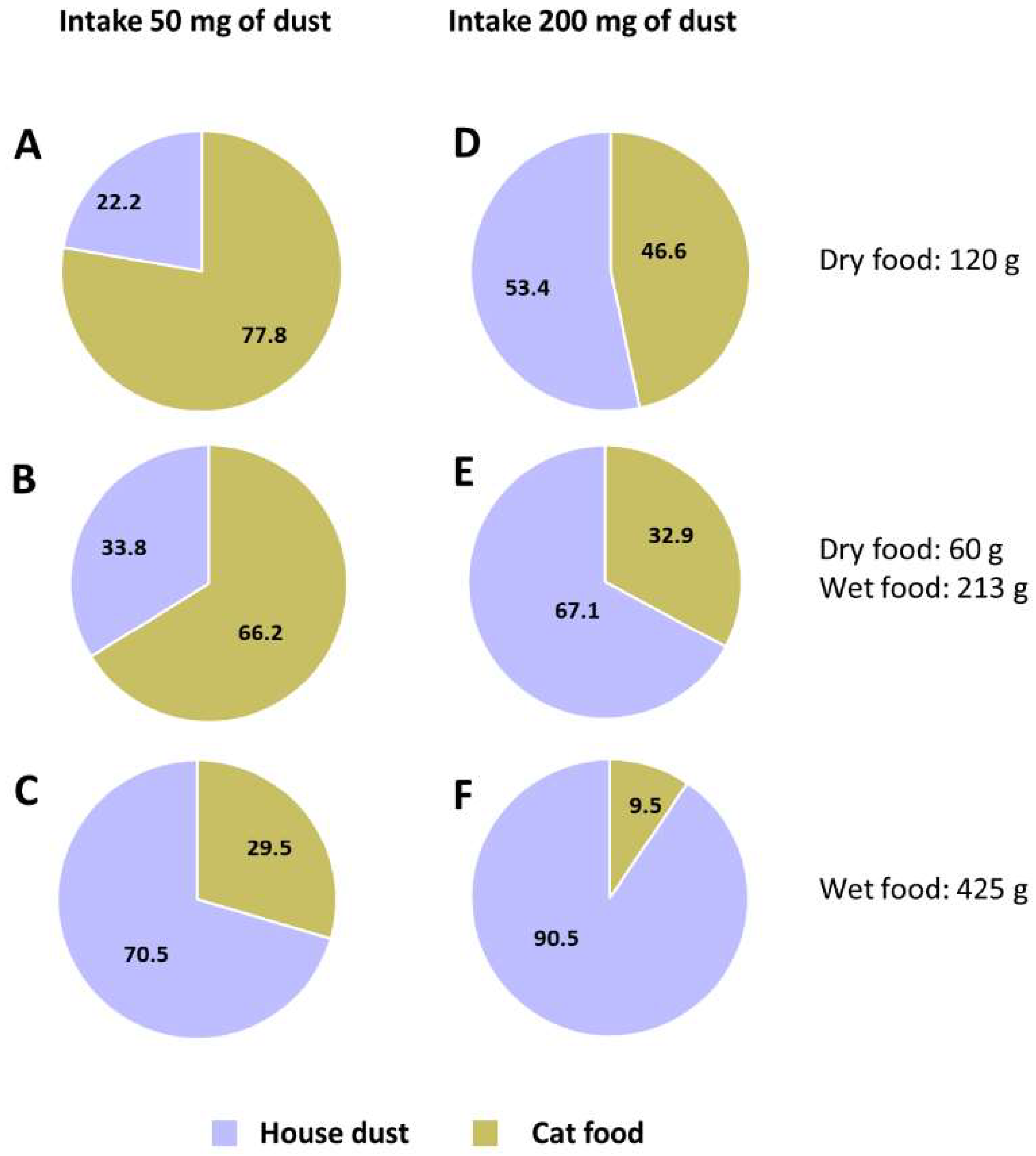
3.4. Estimated Exposure of Pet Cats to OHCs via Cat Food and House Dust and Risk Assessment
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mizukawa, H.; Nomiyama, K.; Nakatsu, S.; Yachimori, S.; Hayashi, T.; Tashiro, Y.; Nagano, Y.; Tanabe, S. Species-specific differences in the accumulation features of organohalogen contaminants and their metabolites in the blood of Japanese terrestrial mammals. Environ. Pollut. 2013, 174, 28–37. [Google Scholar] [CrossRef] [PubMed]
- Aznar-Alemany, Ò.; Yang, X.; Alonso, M.B.; Costa, E.S.; Torres, J.P.M.; Malm, O.; Barceló, D.; Eljarrat, E. Preliminary study of long-range transport of halogenated flame retardants using Antarctic marine mammals. Sci. Total Environ. 2019, 650, 1889–1897. [Google Scholar] [CrossRef] [PubMed]
- Grimm, F.A.; Hu, D.; Kania-Korwel, I.; Lehmler, H.J.; Ludewig, G.; Hornbuckle, K.C.; Duffel, M.W.; Bergman, A.; Robertson, L.W. Metabolism and metabolites of polychlorinated biphenyls (PCBs). Crit. Rev. Toxicol. 2015, 45, 245–272. [Google Scholar] [CrossRef] [PubMed]
- Hakk, H.; Letcher, R.J. Metabolism in the toxicokinetics and fate of brominated flame retardants—A review. Environ. Int. 2003, 29, 801–828. [Google Scholar] [CrossRef] [PubMed]
- Mizukawa, H.; Nomiyama, K.; Nakatsu, S.; Iwata, H.; Yoo, J.; Kubota, A.; Yamamoto, M.; Isizuka, M.; Ikenaka, Y.; Nakayama, S.M.M.; et al. Organohalogen compounds in pet dog and cat: Do pets biotransform natural brominated products in food to harmful hydroxlated substances? Environ. Sci. Technol. 2016, 50, 444–452. [Google Scholar] [CrossRef]
- Khidkhan, K.; Mizukawa, H.; Ikenaka, Y.; Nakayama, S.M.; Nomiyama, K.; Yokoyama, N.; Ichii, O.; Darwish, W.S.; Takiguchi, M.; Tanabe, S.; et al. Tissue distribution and characterization of feline cytochrome P450 genes related to polychlorinated biphenyl exposure. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2019, 226, 108613. [Google Scholar] [CrossRef]
- Kakehi, M.; Ikenaka, Y.; Nakayama, S.M.; Kawai, Y.K.; Watanabe, K.P.; Mizukawa, H.; Nomiyama, K.; Tanabe, S.; Ishizuka, M. Uridine diphosphate-glucuronosyltransferase (UGT) xenobiotic metabolizing activity and genetic evolution in pinniped species. Toxicol. Sci. 2015, 147, 360–369. [Google Scholar] [CrossRef]
- Kondo, T.; Ikenaka, Y.; Nakayama, S.M.; Kawai, Y.K.; Mizukawa, H.; Mitani, Y.; Nomiyama, K.; Tanabe, S.; Ishizuka, M. Uridine diphosphate-glucuronosyltransferase (UGT) 2B subfamily interspecies differences in carnivores. Toxicol. Sci. 2017, 158, 90–100. [Google Scholar] [CrossRef]
- Dye, J.A.; Venier, M.; Zhu, L.; Ward, C.R.; Hites, R.A.; Birnbaum, L.S. Elevated PBDE levels in pet cats: Sentinels for humans? Environ. Sci. Technol. 2007, 41, 6350–6356. [Google Scholar] [CrossRef]
- Ali, N.; Malik, R.N.; Mehdi, T.; Eqani, S.A.M.A.S.; Javeed, A.; Neels, H.; Covaci, A. Organohalogenated contaminants (OHCs) in the serum and hair of pet cats and dogs: Biosentinels of indoor pollution. Sci. Total Environ. 2013, 449, 29–36. [Google Scholar] [CrossRef]
- Boada, L.D.; Sangil, M.; Álvarez-León, E.E.; Hernández-Rodríguez, G.; Henríquez-Hernández, L.A.; Camacho, M.; Zumbado, M.; Serra-Majem, L.; Luzardo, O.P. Consumption of foods of animal origin as determinant of contamination by organochlorine pesticides and polychlorobiphenyls: Results from a population-based study in Spain. Chemosphere 2014, 114, 121–128. [Google Scholar] [CrossRef]
- Malisch, R.; Kotz, A. Dioxins and PCBs in feed and food—Review from European perspective. Sci. Total Environ. 2014, 491, 2–10. [Google Scholar] [CrossRef]
- Wu, N.; Herrmann, T.; Paepke, O.; Tickner, J.; Hale, R.; Harvey, E.; Guardia, M.L.; McClean, M.D.; Webster, T.F. Human exposure to PBDEs: Associations of PBDE body burdens with food consumption and house dust concentrations. Environ. Sci. Technol. 2007, 41, 1584–1589. [Google Scholar] [CrossRef]
- Redfern, F.M.; Lee, W.J.; Yan, P.; Mwangi, J.K.; Wang, L.C.; Shih, C.H. Overview and perspectives on emissions of polybrominated diphenyl ethers on a global basis: Evaporative and fugitive releases from commercial PBDE mixtures and emissions from combustion sources. Aerosol Air Qual. Res. 2017, 17, 1117–1131. [Google Scholar] [CrossRef]
- Norrgran Engdahl, J.; Bignert, A.; Jones, B.; Athanassiadis, I.; Bergman, Å.; Weiss, J.M. Cats’ internal exposure to selected brominated flame retardants and organochlorines correlated to house dust and cat food. Environ. Sci. Technol. 2017, 51, 3012–3020. [Google Scholar] [CrossRef]
- Hamers, T.; Kamstra, J.H.; Sonneveld, E.; Murk, A.J.; Visser, T.J.; Van Velzen, M.J.; Brouwer, A.; Bergman, Å. Biotransformation of brominated flame retardants into potentially endocrine-disrupting metabolites, with special attention to 2,2′,4,4′-tetrabromodiphenyl ether (BDE-47). Mol. Nutr. Food Res. 2008, 52, 284–298. [Google Scholar] [CrossRef]
- Li, F.; Xie, Q.; Li, X.; Li, N.; Chi, P.; Chen, J.; Wang, Z.; Hao, C. Hormone activity of hydroxylated polybrominated diphenyl ethers on human thyroid receptor-β: In vitro and in silico investigations. Environ. Health Perspect. 2010, 118, 602–606. [Google Scholar] [CrossRef]
- Legradi, J.; Dahlberg, A.K.; Cenijn, P.; Marsh, G.; Asplund, L.; Bergman, Å.; Legler, J. Disruption of oxidative phosphorylation (OXPHOS) by hydroxylated polybrominated diphenyl ethers (OH-PBDEs) present in the marine environment. Environ. Sci. Technol. 2014, 48, 14703–14711. [Google Scholar] [CrossRef]
- Hendriks, H.S.; Antunes Fernandes, E.C.; Bergman, Å.; van den Berg, M.; Westerink, R.H. PCB-47, PBDE-47, and 6-OH-PBDE-47 differentially modulate human GABAA and α4β2 nicotinic acetylcholine receptors. Toxicol. Sci. 2010, 118, 635–642. [Google Scholar] [CrossRef]
- McLean, J.L.; Lobetti, R.G.; Schoeman, J.P. Worldwide prevalence and risk factors for feline hyperthyroidism: A review. J. S. Afr. Vet. Assoc. 2014, 85, 1–6. [Google Scholar] [CrossRef]
- Guo, W.; Park, J.S.; Wang, Y.; Gardner, S.; Baek, C.; Petreas, M.; Hooper, K. High polybrominated diphenyl ether levels in California house cats: House dust a primary source? Environ. Toxicol. Chem. 2012, 31, 301–306. [Google Scholar] [CrossRef] [PubMed]
- Dirtu, A.C.; Niessen, S.J.; Jorens, P.G.; Covaci, A. Organohalogenated contaminants in domestic cats’ plasma in relation to spontaneous acromegaly and type 2 diabetes mellitus: A clue for endocrine disruption in humans? Environ. Int. 2013, 57, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Baldé, C.P.; Forti, V.; Gray, V.; Kuehr, R.; Stegmann, P. The Global E-waste Monitor–2017; Electronic Version; United Nations University (UNU): Bonn, Germany; International Telecommunication Union (ITU): Geneva, Switzerland; International Solid Waste Association (ISWA): Vienna, Austria, 2017; pp. 978–992. [Google Scholar]
- Kwan, C.S.; Takada, H.; Mizukawa, K.; Rinawati, M.S.; Santiago, E.C. Sedimentary PBDEs in urban areas of tropical Asian countries. Mar. Pollut. Bull. 2013, 76, 95–105. [Google Scholar] [CrossRef] [PubMed]
- Muenhor, D.; Harrad, S. Polybrominated diphenyl ethers (PBDEs) in car and house dust from Thailand: Implication for human exposure. J. Environ. Sci. Health Part A 2018, 53, 629–642. [Google Scholar] [CrossRef] [PubMed]
- Future Market Insight. Southeast Asia Pet Care Market to Grow at a CAGR of 6.8% from 2014 to 2020, Driven by Pet Humanization in the Region: FMI Estimates. 2015. Available online: https://www.futuremarketinsights.com/press-release/southeast-asia-pet-care-market (accessed on 6 November 2022).
- Eguchi, A.; Nomiyama, K.; Ochiai, M.; Mizukawa, H.; Nagano, Y.; Nakagawa, K.; Tanaka, K.; Miyagawa, H.; Tanabe, S. Simultaneous detection of multiple hydroxylated polychlorinated biphenyls from a complex tissue matrix using gas chromatography/isotope dilution mass spectrometry. Talanta 2014, 118, 253–261. [Google Scholar] [CrossRef]
- Eguchi, A.; Isobe, T.; Ramu, K.; Tanabe, S. Optimisation of the analytical method for octa-, nona- and deca-brominated diphenyl ethers using gas chromatography–quadrupole mass spectrometry and isotope dilution. Int. J. Environ. Anal. Chem. 2011, 91, 348–357. [Google Scholar] [CrossRef]
- Covaci, A.; Voorspoels, S.; Thomsen, C.; van Bavel, B.; Neels, H. Evaluation of total lipids using enzymatic methods for the normalization of persistent organic pollutant levels in serum. Sci. Total Environ. 2006, 366, 361–366. [Google Scholar] [CrossRef]
- Ministry of the Environment. Guidance on Chemical Substance Environmental Survey [Online]. 2016. Available online: http://www.env.go.jp/chemi/kurohon/tebiki/mat01.pdf (accessed on 6 November 2022).
- Dirtu, A.C.; Covaci, A. Estimation of daily intake of organohalogenated contaminants from food consumption and indoor dust ingestion in Romania. Environ. Sci. Technol. 2010, 44, 6297–6304. [Google Scholar] [CrossRef]
- Mensching, D.A.; Slater, M.; Scott, J.W.; Ferguson, D.C.; Beasley, V.R. The feline thyroid gland: A model for endocrine disruption by polybrominated diphenyl ethers (PBDEs)? J. Toxicol. Environ. Health A 2012, 75, 201–212. [Google Scholar] [CrossRef]
- Guo, W.; Gardner, S.; Yen, S.; Petreas, M.; Park, J.S. Temporal changes of PBDE levels in California house cats and a link to cat hyperthyroidism. Environ. Sci. Technol. 2016, 50, 1510–1518. [Google Scholar] [CrossRef]
- Suzuki, G.; Kida, A.; Sakai, S.-I.; Takigami, H. Existence State of Bromine as an Indicator of the Source of Brominated Flame Retardants in Indoor Dust. Environ. Sci. Technol. 2009, 43, 1437–1442. [Google Scholar] [CrossRef]
- Ruiz-Suárez, N.; Camacho, M.; Boada, L.D.; Henríquez-Hernández, L.A.; Rial, C.; Valerón, P.F.; Zumbado, M.; Gonzáiez, M.A.; Luzardo, O.P. The assessment of daily dietary intake reveals the existence of a different pattern of bioaccumulation of chlorinated pollutants between domestic dogs and cats. Sci. Total Environ. 2015, 530, 45–52. [Google Scholar] [CrossRef]
- Serpe, F.P.; Fiorito, F.; Esposito, M.; Ferrari, A.; Fracassi, F.; Miniero, R.; Pietra, M.; Roncada, P.; Brambilla, G. Polychlorobiphenyl levels in the serum of cats from residential flats in Italy: Role of the indoor environment. J. Environ. Sci. Health A Tox. Hazard Subst. Environ. Eng. 2018, 53, 777–785. [Google Scholar] [CrossRef]
- Nomiyama, K.; Uchiyama, Y.; Horiuchi, S.; Eguchi, A.; Mizukawa, H.; Horai Hirata, S.; Shinohara, R.; Tanabe, S. Organohalogen compounds and their metabolites in the blood of Japanese amberjack (Seriola quinqueradiata) and scalloped hammerhead shark. Chemosphere 2011, 85, 315–321. [Google Scholar] [CrossRef]
- Vénisseau, A.; Bichon, E.; Brosseaud, A.; Vaccher, V.; Lesquin, E.; Larvor, F.; Durand, S.; Dervilly-Pinel, G.; Marchand, P.; Le Bizec, B. Occurence of legacy and novel brominated flame retardants in food and feed in France for the period 2014 to 2016. Chemosphere 2018, 207, 497–506. [Google Scholar] [CrossRef]
- Sjodin, A.; Papke, O.; McGahee, E.; Focant, J.F.; Jones, R.S.; Pless-Mulloli, T.; Toms, L.M.; Herrmann, T.; Muller, J.; Needham, L.L.; et al. Concentration of polybrominated diphenyl ethers (PBDEs) in household dust from various countries. Chemosphere 2008, 73, S131–S136. [Google Scholar] [CrossRef]
- Anh, H.Q.; Tri, T.M.; Minh, T.B.; Takahashi, S. Polybrominated diphenyl ethers in indoor and outdoor dust from Southeast Asia: An updated review on contamination status, human exposure, and future perspectives. Environ. Pollut. 2021, 272, 116012. [Google Scholar]
- Muenhor, D.; Moon, H.B.; Lee, S.; Goosey, E. Polybrominated diphenyl ethers (PBDEs) in floor and road dust from a manual e-waste dismantling facility and adjacent communities in Thailand. J. Environ. Sci. Health Part A 2017, 52, 1284–1294. [Google Scholar] [CrossRef]
- Huwe, J.K.; Smith, D.J. Accumulation, Whole-Body Depletion, and Debromination of Decabromodiphenyl Ether in Male Sprague−Dawley Rats Following Dietary Exposure. Environ. Sci. Technol. 2007, 41, 2371–2377. [Google Scholar] [CrossRef]
- Chow, K.; Hearn, L.K.; Zuber, M.; Beatty, J.A.; Mueller, J.F.; Barrs, V.R. Evaluation of polybrominated diphenyl ethers (PBDEs) in matched cat sera and house dust samples: Investigation of a potential link between PBDEs and spontaneous feline hyperthyroidism. Environ. Res. 2015, 136, 173–179. [Google Scholar] [CrossRef] [PubMed]
| Sample ID | Hospital Location | Lipid Content [%] *1 | Age [Year] | Weight [kg] | Breed | Sex | Diet | Housing | Q1 *2 | Q2 *3 | Q3 *4 | Q4 *5 | Q5 *6 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| BK04 | Bangkok | 0.66 | 2.0 | 4.85 | Domestic short hair | Female | Dry | Outdoor | 14 | 0 | 0 | 0 | no |
| BK05 | Bangkok | 0.31 | 2.0 | 3.4 | Domestic short hair | Female | Dry | Indoor | 14 | 0 | 0 | 0 | no |
| BK06 | Bangkok | 0.24 | 3.5 | 7 | Domestic short hair | Male | Dry, Wet | Both | 21 | 7 | 0 | 0 | yes |
| BK08 | Bangkok | 0.48 | 0.3 | 4.4 | Domestic short hair | Male | Dry, Wet | Indoor | 14 | 0 | 14 | 0 | yes |
| BK09 | Bangkok | 0.24 | 4.25 | 5.15 | Domestic short hair | Male | Dry, Wet | Indoor | 21 | 7 | 0 | 0 | yes |
| BK10 | Bangkok | 0.23 | 1.0 | 4.14 | Domestic short hair | Male | Dry | Indoor | 21 | 0 | 0 | 0 | no |
| BK11 | Bangkok | 0.30 | 5.1 | 5.0 | Persian | Male | Dry | Indoor | 14 | 0 | 0 | 0 | yes |
| BK19 | Bangkok | 0.40 | 5.0 | 8.0 | Domestic short hair | Male | Dry, Wet | Both | 20 | 4 | 2 | 0 | yes |
| BK24 | Bangkok | 0.51 | 2.0 | 3.7 | Domestic short hair | Female | Dry, Home, Wet | Both | 14 | 0 | 1 | 7 | no |
| BK34 | Bangkok | 0.41 | 3.0 | 7.2 | Domestic short hair | Male | Dry, Home, Wet | Both | 14 | 0 | 1 | 7 | no |
| BKK001 | Bangkok | NA *7 | 1.0 | 5.0 | Domestic short hair | Female | Dry | Indoor | 7 | 0 | 0 | 0 | yes |
| BKK013 | Bangkok | NA | 1.0 | 4.0 | Domestic short hair | Male | Dry | Indoor | 21 | 0 | 0 | 0 | no |
| BKK017 | Bangkok | NA | 1.0 | 4.0 | Domestic short hair | Male | Dry | Indoor | 21 | 0 | 0 | 0 | no |
| HH02 | Hau Hin | 0.19 | 7.0 | 5.8 | British short hair | Male | Dry, Wet | Indoor | 7 | 1 | 0 | 0 | yes |
| HH03 | Hau Hin | 0.22 | 1.2 | 2.6 | Domestic short hair | Male | Dry | Indoor | 14 | 0 | 0 | 0 | yes |
| HH04 | Hau Hin | 0.23 | 0.3 | 1.6 | Domestic short hair | Male | Dry | Indoor | 14 | 0 | 0 | 0 | yes |
| HH05 | Hau Hin | NA | 5.0 | 3.3 | Domestic short hair | Female | Dry, Wet | Indoor | 7 | 0 | 7 | 0 | yes |
| HH10 | Hau Hin | 0.41 | 1.3 | 3.7 | Domestic short hair | Male | Dry | Indoor | 14 | 0 | 0 | 0 | yes |
| HH12 | Hau Hin | 0.22 | 3.0 | 4.5 | Domestic short hair | Female | Dry, Wet | Outdoor | 8 | 4 | 0 | 0 | yes |
| HH13 | Hau Hin | 0.14 | 3.2 | 1.0 | Domestic short hair | Female | Dry, Wet | Indoor | 30 | 10 | 15 | 0 | no |
| HH14 | Hau Hin | NA | 2.0 | 3.8 | Domestic short hair | Female | Dry, Wet | Indoor | 30 | 10 | 15 | 0 | no |
| HH15 | Hau Hin | NA | 5.0 | 3.2 | Domestic short hair | Female | Dry, Wet | Outdoor | 14 | 0 | 14 | 0 | no |
| HH18 | Hau Hin | 0.31 | 3.0 | 5.1 | Domestic short hair | Male | Wet, Home, Dry | Both | 7 | 0 | 14 | 14 | yes |
| HH24 | Hau Hin | 0.12 | 5.0 | 4.1 | Domestic short hair | Female | Dry | Outdoor | 14 | 0 | 0 | 0 | no |
| HH25 | Hau Hin | 0.23 | 3.0 | 4.0 | Domestic short hair | Male | Wet, Home, Dry | Both | 7 | 0 | 14 | 14 | yes |
| HH28 | Hau Hin | 0.18 | 10 | 5.2 | Mixed | Female | Dry, Wet | Indoor | 7 | 0 | 7 | 0 | Yes |
| HH32 | Hau Hin | 0.15 | 3.8 | 1.5 | Maine coon | Female | Dry | Indoor | 7 | 0 | 0 | 0 | Yes |
| HH34 | Hau Hin | 0.29 | 3.0 | 2.2 | Domestic short hair | Female | Dry, Home | Outdoor | 2 | 0 | 0 | 2 | No |
| HH35 | Hau Hin | 0.12 | 4.0 | 5.2 | Unknown | Male | Dry, Wet, Home | Both | 7 | 3 | 0 | 2 | No |
| HH37 | Hau Hin | NA | 0.5 | 3.5 | Unknown | Male | Dry | Indoor | 7 | 0 | 0 | 0 | No |
| HH41 | Hau Hin | 0.56 | 7.0 | 6.2 | Mixed | Male | Wet, Dry, Home | Both | 7 | 0 | 14 | 7 | Yes |
| NP01 | Nong Pho | NA | 3.0 | 2.2 | Persian | Female | Dry | Indoor | 14 | 0 | 0 | 0 | yes |
| NP02 | Nong Pho | NA | 7.0 | 4.4 | Domestic short hair | Male | Dry, Wet | Both | 14 | 2 | 2 | 0 | yes |
| NP05 | Nong Pho | NA | 0.6 | 3.8 | Domestic short hair | Male | Dry, Home, Wet | Indoor | 14 | 1 | 0 | 14 | yes |
| NP07 | Nong Pho | NA | 1.0 | 3.0 | Domestic short hair | Female | Dry | Indoor | 14 | 0 | 0 | 0 | no |
| NP10 | Nong Pho | NA | 0.5 | 2.7 | Domestic short hair | Female | Dry, Home | Both | 21 | 0 | 0 | 7 | no |
| NP11 | Nong Pho | NA | 0.5 | 2.5 | Domestic short hair | Female | Home | Both | 0 | 0 | 0 | 21 | no |
| NP15 | Nong Pho | NA | 0.7 | 3.8 | Domestic short hair | Male | Dry | Outdoor | 35 | 0 | 0 | 0 | yes |
| NP16 | Nong Pho | NA | 0.7 | 2.5 | Domestic short hair | Female | Dry, Wet | Indoor | 6 | 1 | 1 | 0 | yes |
| NP18 | Nong Pho | NA | 0.7 | 2.5 | Domestic short hair | Female | Dry, Home | Indoor | 14 | 0 | 0 | 3 | yes |
| NP20 | Nong Pho | NA | 0.9 | 4.0 | Domestic short hair | Male | Dry, Home, Wet | Indoor | 21 | 3 | 0 | 7 | yes |
| Type | Sample ID | Produced Country | Maker | Main Ingredient |
|---|---|---|---|---|
| Dry | Dry 1 | France | Maker A | Poultry meat, Rice, Animal fat, Corn, Fish oil |
| Dry | Dry 2 | France | Maker A | Poultry meat, Rice, Wheat gluten, Animal fat, Corn, Fish oil |
| Dry | Dry 3 | France | Maker A | Poultry meat, Wheat gluten, Rice, Corn, Animal fat, Flour |
| Dry | Dry 4 | France | Maker A | Poultry meat, Wheat gluten, Rice, Corn, Animal fat, Flour |
| Dry | Dry 5 | Thailand | Maker B | Cereal, Byproduct-meal, Vegetable protein, Fish meal, Tuna-byproduct |
| Dry | Dry 6 | Thailand | Maker B | Cereal, Byproduct-meal, Vegetable protein, Fish meal, Shrimp-byproduct |
| Dry | Dry 7 | USA | Maker C | Chicken meal, Corn, Rice, Fish oil |
| Wet (Canned) | Wet 1 | Thailand | Maker D | Tuna, Shrimp |
| Wet (Canned) | Wet 2 | Thailand | Maker B | Pilchards, Shrimp |
| Wet (Pouch) | Wet 3 | Thailand | Maker B | Mackerel, Vegetable oil |
| Wet (Canned) | Wet 4 | Thailand | Maker E | Chicken breast |
| Wet (Pouch) | Wet 5 | Thailand | Maker E | Tuna, Ship jack tuna, Mackerel, Beef |
| Wet (Canned) | Wet 6 | Thailand | Maker F | Tuna, Ocean fish, Vegetable oil, |
| Wet (Pouch) | Wet 7 | Thailand | Maker F | Mackerel, Soybean oil |
| Cat Serum (pg mL−1 Serum) | Dry Food (pg g−1 Wet wt.) | Wet Food (pg g−1 Wet wt.) | House Dust (pg g−1 Dry wt.) | |||||
|---|---|---|---|---|---|---|---|---|
| Compound | Median (Range) | DF (%) | Median | DF (%) | Median | DF (%) | Median | DF (%) |
| BDE-47 | <MDL (<MDL–70) | 26 | 4.9 (<MDL–15) | 71 | 5.6 (<MDL–46) | 71 | 260 (110–890) | 100 |
| BDE-99 | <MDL (<MDL–95) | 21 | <MDL (<MDL–12) | 29 | <MDL (<MDL–11) | 14 | 210 (130–1400) | 100 |
| BDE-209 | 350 (<MDL–37,000) | 79 | 330 (180–31,000) | 100 | <MDL (<MDL–69) | 29 | 200,000 (21,000–520,000) | 100 |
| ΣPBDEs | 490 (<MDL–48,000) | 100 | 350 (200–36,000) | 100 | 12 (<MDL–190) | 71 | 240,000 (26,000–590,000) | 100 |
| CB-153 | 8 (<MDL–340) | 51 | 24 (20–43) | 100 | 12 (<MDL–310) | 71 | <MDL (<MDL) | 0 |
| CB-138 | <MDL (<MDL–250) | 49 | 19 (15–32) | 100 | 9.1 (<MDL–240) | 71 | <MDL (<MDL) | 0 |
| ΣPCBs | 18 (<MDL–1500) | 74 | 120 (59–210) | 100 | 31 (<MDL–2100) | 86 | <MDL (<MDL–600) | 29 |
| 4′-OH-CB-25/26/4-OH-CB-31 | 7 (<MDL–270) | 82 | <MDL (<MDL) | 0 | <MDL (<MDL) | 0 | <MDL (<MDL) | 0 |
| 4′-OH-CB-72 | 14 (<MDL–53) | 82 | <MDL (<MDL) | 0 | <MDL (<MDL) | 0 | <MDL (<MDL) | 0 |
| ΣOH-PCBs | 59 (1.9–1000) | 100 | <MDL (<MDL) | 0 | <MDL (<MDL) | 0 | <MDL (<MDL–95) | 14 |
| 2′-OH-BDE-68 | 16 (<MDL–190) | 74 | <MDL (<MDL) | 0 | <MDL (<MDL–120) | 14 | <MDL (<MDL) | 0 |
| 6-OH-BDE-47 | 55 (9.9–370) | 100 | <MDL (<MDL) | 0 | <MDL (<MDL–35) | 29 | <MDL (<MDL) | 0 |
| ΣOH-PBDEs | 77 (9.9–440) | 100 | <MDL (<MDL) | 0 | <MDL (<MDL–160) | 29 | <MDL (<MDL) | 0 |
| 2′-MeO-BDE-68 | 14 (<MDL–130) | 59 | <MDL (<MDL) | 0 | 71 (<MDL–2800) | 71 | <MDL (<MDL) | 0 |
| 6-MeO-BDE-47 | <MDL (<MDL–380) | 28 | 18 (<MDL–25) | 57 | 17 (<MDL–420) | 57 | <MDL (<MDL) | 0 |
| ΣMeO-PBDEs | 19 (<MDL–460) | 62 | 18 (<MDL–25) | 57 | 150 (<MDL–3300) | 71 | <MDL (<MDL) | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shimasaki, M.; Mizukawa, H.; Takaguchi, K.; Saengtienchai, A.; Ngamchirttakul, A.; Pencharee, D.; Khidkhan, K.; Ikenaka, Y.; Nakayama, S.M.M.; Ishizuka, M.; et al. Contamination Status of Pet Cats in Thailand with Organohalogen Compounds (OHCs) and Their Hydroxylated and Methoxylated Derivatives and Estimation of Sources of Exposure to These Contaminants. Animals 2022, 12, 3520. https://doi.org/10.3390/ani12243520
Shimasaki M, Mizukawa H, Takaguchi K, Saengtienchai A, Ngamchirttakul A, Pencharee D, Khidkhan K, Ikenaka Y, Nakayama SMM, Ishizuka M, et al. Contamination Status of Pet Cats in Thailand with Organohalogen Compounds (OHCs) and Their Hydroxylated and Methoxylated Derivatives and Estimation of Sources of Exposure to These Contaminants. Animals. 2022; 12(24):3520. https://doi.org/10.3390/ani12243520
Chicago/Turabian StyleShimasaki, Makoto, Hazuki Mizukawa, Kohki Takaguchi, Aksorn Saengtienchai, Araya Ngamchirttakul, Disdanai Pencharee, Kraisiri Khidkhan, Yoshinori Ikenaka, Shouta M. M. Nakayama, Mayumi Ishizuka, and et al. 2022. "Contamination Status of Pet Cats in Thailand with Organohalogen Compounds (OHCs) and Their Hydroxylated and Methoxylated Derivatives and Estimation of Sources of Exposure to These Contaminants" Animals 12, no. 24: 3520. https://doi.org/10.3390/ani12243520
APA StyleShimasaki, M., Mizukawa, H., Takaguchi, K., Saengtienchai, A., Ngamchirttakul, A., Pencharee, D., Khidkhan, K., Ikenaka, Y., Nakayama, S. M. M., Ishizuka, M., & Nomiyama, K. (2022). Contamination Status of Pet Cats in Thailand with Organohalogen Compounds (OHCs) and Their Hydroxylated and Methoxylated Derivatives and Estimation of Sources of Exposure to These Contaminants. Animals, 12(24), 3520. https://doi.org/10.3390/ani12243520







