Improving Ovine Behavioral Pain Diagnosis by Implementing Statistical Weightings Based on Logistic Regression and Random Forest Algorithms
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Dataset
2.2. Statistical Description
2.2.1. Creation of the Multilevel Binomial Logistic Regression Algorithm
2.2.2. Creation of the Random Forest Algorithm
2.2.3. Quality of Pain Diagnosis
3. Results
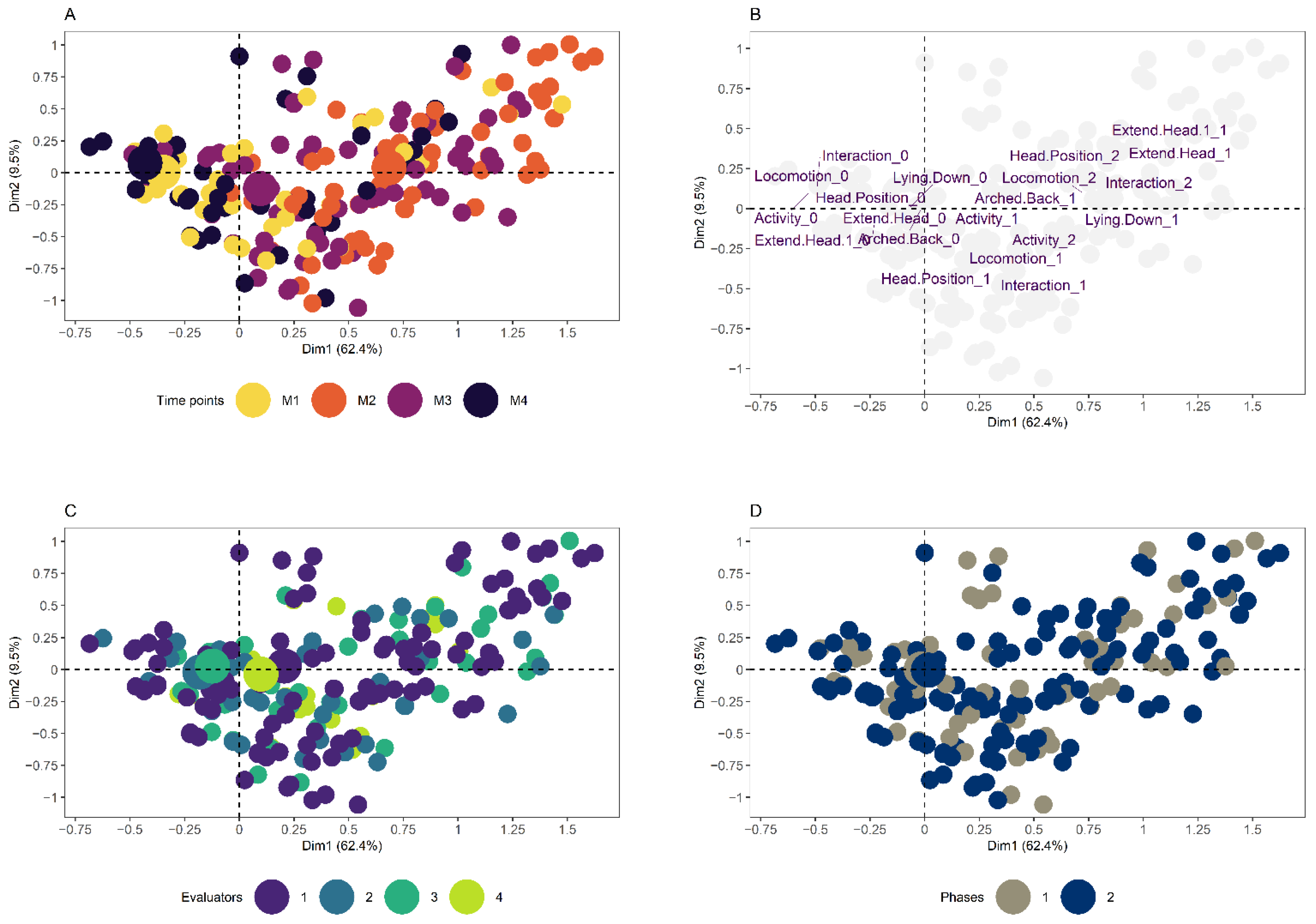
3.1. Creation of the Multilevel Binomial Logistic Regression Algorithm
3.2. Creation of the Random Forest Algorithm
3.3. Quality of Pain Diagnosis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Webster, J. Animal welfare: Freedoms, dominions and “A life worth living”. Animals 2016, 6, 35. [Google Scholar] [CrossRef]
- Steagall, P.V.; Bustamante, H.; Johnson, C.B.; Turner, P.V. Pain management in farm animals: Focus on cattle, sheep and pigs. Animals 2021, 11, 1483. [Google Scholar] [CrossRef]
- Mclennan, K.M. Why pain is still a welfare issue for farm animals, and how facial expression could be the answer. Agriculture 2018, 8, 127. [Google Scholar] [CrossRef]
- Leung, V.; Rousseau-Blass, F.; Beauchamp, G.; Pang, D.S.J. ARRIVE Has Not ARRIVEd: Support for the ARRIVE (Animal Research: Reporting of in Vivo Experiments) guidelines does not improve the reporting quality of papers in animal welfare, analgesia or anesthesia. PLoS ONE 2018, 13, e0197882. [Google Scholar] [CrossRef]
- Fisher, A.D. Addressing pain caused by mulesing in sheep. Appl. Anim. Behav. Sci. 2011, 135, 232–240. [Google Scholar] [CrossRef]
- Orihuela, A.; Ungerfeld, R. Tail Docking in Sheep (Ovis Aries): A review on the arguments for and against the procedure, advantages/disadvantages, methods, and new evidence to revisit the topic. Livest. Sci. 2019, 230, 103837. [Google Scholar] [CrossRef]
- Small, A.; Fisher, A.D.; Lee, C.; Colditz, I. Analgesia for sheep in commercial production: Where to next? Animals 2021, 11, 1127. [Google Scholar] [CrossRef]
- Taylor, K.; Rego, L.; Weber, T. Recommendations to improve the EU non-technical summaries of animal experiments. ALTEX Altern. To Anim. Exp. 2018, 35, 193–210. [Google Scholar] [CrossRef]
- Gigliuto, C.; De Gregori, M.; Malafoglia, V.; Raffaeli, W.; Compagnone, C.; Visai, L.; Petrini, P.; Avanzini, M.A.; Muscoli, C.; Viganò, J.; et al. Pain assessment in animal models: Do we need further studies? J. Pain Res. 2014, 7, 227–236. [Google Scholar]
- Gregory, M.H.; Capito, N.; Kuroki, K.; Stoker, A.M.; Cook, J.L.; Sherman, S.L. A review of translational animal models for knee osteoarthritis. Arthritis 2012, 2012, 14. [Google Scholar] [CrossRef]
- Piel, M.J.; Kroin, J.S.; Van Wijnen, A.J.; Kc, R.; Im, H.J. Pain assessment in animal models of osteoarthritis. Gene 2014, 537, 184–188. [Google Scholar] [CrossRef] [PubMed]
- Guatteo, R.; Guémené, D. Sources of known and/or potential pain in farm animals. Adv. Anim. Biosci. 2014, 5, 319–332. [Google Scholar] [CrossRef]
- Zufferey, R.; Minnig, A.; Thomann, B.; Zwygart, S.; Keil, N.; Schüpbach, G.; Miserez, R.; Zanolari, P.; Stucki, D. Animal-based indicators for on-farm welfare assessment in sheep. Animals 2021, 11, 2973. [Google Scholar] [CrossRef] [PubMed]
- Price, D.D. Psychological and neural mechanisms of the affective dimension of pain. Science 2000, 288, 1769–1772. [Google Scholar] [CrossRef]
- Fell, L.R.; Shutt, D.A. Behavioural and hormonal responses to acute surgical stress in sheep. Appl. Anim. Behav. Sci. 1989, 22, 283–294. [Google Scholar] [CrossRef]
- Mellor, D.J.; Murray, L. Effects of tail docking and castration on behaviour and plasma cortisol concentrations in young lambs. Res. Vet. Sci. 1989, 46, 387–391. [Google Scholar] [CrossRef]
- Lester, S.J.; Mellor, D.J.; Holmes, R.J.; Ward, R.N.; Stafford, K.J. Behavioural and cortisol responses of lambs to castration and tailing using different methods. N. Z. Vet. J. 1996, 44, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Paull, D.R.; Lee, C.; Atkinson, S.J.; Fisher, A.D. Effects of meloxicam or tolfenamic acid administration on the pain and stress responses of Merino lambs to mulesing. Aust. Vet. J. 2008, 86, 303–311. [Google Scholar] [CrossRef]
- Molony, V.; Kent, J.E.; Robertson, I.S. Behavioural responses of lambs of three ages in the first three hours after three methods of castration and tail docking. Res. Vet. Sci. 1993, 55, 236–245. [Google Scholar] [CrossRef]
- Molony, V.; Kent, J.E. Assessment of acute pain in farm animals using behavioral and physiological measurements. J. Anim. Sci. 1997, 75, 266–272. [Google Scholar] [CrossRef]
- Small, A.H.; Marini, D.; Dyall, T.; Paull, D.; Lee, C. A randomised field study evaluating the effectiveness of buccal meloxicam and topical local anaesthetic formulations administered singly or in combination at improving welfare of female Merino lambs undergoing surgical mulesing and hot knife tail docking. Res. Vet. Sci. 2018, 118, 305–311. [Google Scholar] [CrossRef] [PubMed]
- Futro, A.; Masłowska, K.; Dwyer, C.M. Ewes direct most maternal attention towards lambs that show the greatest pain-related behavioural responses. PLoS ONE 2015, 10, e0134024. [Google Scholar] [CrossRef]
- Molony, V.; Kent, J.E.; Viñuela-Fernández, I.; Anderson, C.; Dwyer, C.M. Pain in lambs castrated at 2 days using novel smaller and tighter rubber rings without and with local anaesthetic. Vet. J. 2012, 193, 81–86. [Google Scholar] [CrossRef]
- Thornton, P.; Waterman-Pearson, A. Behavioural responses to castration in lambs. Anim. Welf. 2002, 11, 203–212. [Google Scholar]
- Kent, J.E.; Molony, V.; Graham, M.J. Comparison of methods for the reduction of acute painproduced by rubber ring castration or tail docking of week-old lambs. Vet. J. 1998, 155, 39–51. [Google Scholar] [CrossRef]
- Dinniss, A.S.; Stafford, K.J.; Mellort, D.J.; Bruce, R.A.; Ward, R.N. The behaviour pattern of lambs after castration using a rubber ring and/or castrating clamp with or without local anaesthetic. N. Z. Vet. J. 1999, 47, 198–203. [Google Scholar] [CrossRef]
- Molony, V.; Kent, J.E.; McKendrick, I.J. Validation of a method for assessment of an acute pain in lambs. Appl. Anim. Behav. Sci. 2002, 76, 215–238. [Google Scholar] [CrossRef]
- Kent, J.E.; Molony, V.; Robertson, I.S. Comparison of the Burdizzo and rubber ring methods for castrating and tail docking lambs. Vet. Rec. 1995, 136, 192–196. [Google Scholar] [CrossRef]
- Grant, C. Behavioural responses of lambs to common painful husbandry procedures. Appl. Anim. Behav. Sci. 2004, 87, 255–273. [Google Scholar] [CrossRef]
- Lomax, S.; Dickson, H.; Sheil, M.; Windsor, P.A. Topical anaesthesia alleviates short-term pain of castration and tail docking in lambs. Aust. Vet. J. 2010, 88, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Melches, S.; Mellema, S.C.; Doherr, M.G.; Wechsler, B.; Steiner, A. Castration of lambs: A welfare comparison of different castration techniques in lambs over 10 weeks of age. Vet. J. 2007, 173, 554–563. [Google Scholar] [CrossRef] [PubMed]
- Small, A.H.; Jongman, E.C.; Niemeyer, D.; Lee, C.; Colditz, I.G. Efficacy of precisely injected single local bolus of lignocaine for alleviation of behavioural responses to pain during tail docking and castration of lambs with rubber rings. Res. Vet. Sci. 2020, 133, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Izer, J.M.; LaFleur, R.A.; Weiss, W.J.; Wilson, R.P. Development of a pain scoring system for use in sheep surgically implanted with ventricular assist devices. 2018, 32, 706–715. J. Investig. Surg. 2018, 32, 706–715. [Google Scholar] [CrossRef]
- Fitzpatrick, J.; Scott, M.; Nolan, A. Assessment of pain and welfare in sheep. Small Rumin. Res. 2006, 62, 55–61. [Google Scholar] [CrossRef]
- Silva, N.E.O.F.; Trindade, P.H.E.; Oliveira, A.R.; Taffarel, M.O.; Moreira, M.A.P.; Denadai, R.; Rocha, P.B.; Luna, S.P.L. Correction: Validation of the Unesp-Botucatu composite scale to assess acute postoperative abdominal pain in sheep (USAPS). PLoS ONE 2022, 17, e0268305. [Google Scholar] [CrossRef]
- Streiner, D.L.; Norman, G.R.; Cairney, J. Health Measurement Scales—A Practical Guide to Their Development and Use; Oxford University Press: Oxford, UK, 2015; Volume 60. [Google Scholar]
- Mokkink, L.B.; Prinsen, C.A.C.; Patrick, D.L.; Alonso, J.; Bouter, L.M.; de Vet, H.C.W.; Terwee, C.B. COSMIN Manual for Systematic Reviews of PROMs COSMIN Methodology for Systematic Reviews of Patient-Reported Outcome Measures (PROMs) User Manual. Available online: https://cosmin.nl/wp-content/uploads/COSMIN-syst-review-for-PROMs-manual_version-1_feb-2018.pdf (accessed on 14 August 2022).
- Lötsch, J.; Ultsch, A. Machine learning in pain research. Pain 2018, 159, 623. [Google Scholar] [CrossRef]
- Kapos, F.P.; Look, J.O.; Zhang, L.; Hodges, J.S.; Schiffman, E.L. Predictors of long-term TMD pain intensity: An 8-year cohort study. J. Oral Facial Pain Headache 2018, 32, 113. [Google Scholar] [CrossRef]
- Gugliotta, M.; Da Costa, B.R.; Dabis, E.; Theiler, R.; Jüni, P.; Reichenbach, S.; Landolt, H.; Hasler, P. Surgical versus conservative treatment for lumbar disc herniation: A prospective cohort study. BMJ Open 2016, 6, e012938. [Google Scholar] [CrossRef] [PubMed]
- Turner, D.; Griffiths, A.M.; Steinhart, A.H.; Otley, A.R.; Beaton, D.E. Mathematical weighting of a clinimetric index (Pediatric Ulcerative Colitis Activity Index) was superior to the judgmental approach. J. Clin. Epidemiol. 2009, 62, 738–744. [Google Scholar] [CrossRef] [PubMed]
- Turner, D.; Griffiths, A.M.; Walters, T.D.; Seah, T.; Markowitz, J.; Pfefferkorn, M.; Keljo, D.; Waxman, J.; Otley, A.; Leleiko, N.S.; et al. Mathematical weighting of the pediatric Crohn’s disease activity index (PCDAI) and comparison with its other short versions. Inflamm. Bowel Dis. 2012, 18, 55–62. [Google Scholar] [CrossRef]
- Nikaido, T.; Sumitani, M.; Sekiguchi, M.; Konno, S. The Spine PainDETECT questionnaire: Development and validation of a screening tool for neuropathic pain caused by spinal disorders. PLoS ONE 2018, 13, e0193987. [Google Scholar] [CrossRef]
- Lötsch, J.; Geisslinger, G.; Heinemann, S.; Lerch, F.; Oertel, B.G.; Ultsch, A. Quantitative sensory testing response patterns to capsaicin- and ultraviolet-B–induced local skin hypersensitization in healthy subjects: A machine-learned analysis. Pain 2018, 159, 11. [Google Scholar] [CrossRef]
- Sutton, G.A.; Dahan, R.; Turner, D.; Paltiel, O. A behaviour-based pain scale for horses with acute colic: Scale construction. Vet. J. 2013, 196, 394–401. [Google Scholar] [CrossRef]
- Kilkenny, C.; Browne, W.; Cuthill, I.C.; Emerson, M.; Altman, D.G. Animal research: Reporting in vivo experiments: The ARRIVE guidelines. Br. J. Pharmacol. 2010, 160, 1577. [Google Scholar] [CrossRef]
- Russell, W.M.S.; Burch, R.L. The Principles of Humane Experimental Technique; Methuen Publishing Ltd.: London, UK, 1959. [Google Scholar]
- Banks, R.E. The 4th R of research. Contemp. Top. Lab. Anim. Sci. 1995, 34, 50–51. [Google Scholar]
- Michael, K.; Nachtsheim, C.; Neter, J.; Li, W. Applied Linear Statistical Models, 5th ed.; McGraw-Hill/Irwin: New York, NY, USA, 2004. [Google Scholar]
- James, G.; Witten, D.; Hastie, T.; Tibshirani, R. An Introduction to Statistical Learning with Applications in R; Springer: New York, NY, USA, 2014. [Google Scholar]
- Heiberg, T.; Nordby, T.; Kvien, T.K.; Buanes, T. Development and Preliminary validation of the pancreatic cancer disease impact score. Support. Care Cancer 2013, 21, 1677–1684. [Google Scholar] [CrossRef][Green Version]
- Bashir, N.S.; Walters, T.D.; Griffiths, A.M.; Ungar, W.J. An assessment of the validity and reliability of the pediatric child health utility 9d in children with inflammatory bowel disease. Children 2021, 8, 343. [Google Scholar] [CrossRef]
- Sun, H.; Papadopoulos, E.J.; Hyams, J.S.; Griebel, D.; Lee, J.J.; Tomaino, J.; Mulberg, A.E. Well-defined and reliable clinical outcome assessments for pediatric Crohn disease: A critical need for drug development. J. Pediatr. Gastroenterol. Nutr. 2015, 60, 729–736. [Google Scholar] [CrossRef]
- Shaoul, R.; Day, A.S. An overview of tools to score severity in pediatric inflammatory bowel disease. Front. Pediatr. 2021, 9, 271. [Google Scholar] [CrossRef] [PubMed]
- Carlsen, K.; Frederiksen, N.W.; Wewer, V. Integration of EHealth into pediatric inflammatory bowel disease care is safe: 3 years of follow-up of daily care. J. Pediatr. Gastroenterol. Nutr. 2021, 72, 723–727. [Google Scholar] [CrossRef] [PubMed]
- Brondani, J.T.; Luna, S.P.L.; Padovani, C.R. Refinement and initial validation of a multidimensional composite scale for use in assessing acute postoperative pain in cats. Am. J. Vet. Res. 2011, 72, 174–183. [Google Scholar] [CrossRef] [PubMed]
- Brondani, J.T.; Mama, K.R.; Luna, S.P.L.; Wright, B.D.; Niyom, S.; Ambrosio, J.; Vogel, P.R.; Padovani, C.R. Validation of the english version of the UNESP-Botucatu multidimensional composite pain scale for assessing postoperative pain in cats. BMC Vet. Res. 2013, 9, 1–15. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, M.G.C.; Luna, S.P.L.; Nunes, T.L.; Firmino, P.R.; de Lima, A.G.A.; Ferreira, J.; Trindade, P.H.E.; Júnior, R.A.B.; de Paula, V.V. Post-operative pain behaviour associated with surgical castration in donkeys (Equus Asinus). Equine Vet. J. 2021, 53, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Pinho, R.H.; Leach, M.C.; Minto, B.W.; Rocha, F.D.L.; Luna, S.P.L. Postoperative pain behaviours in rabbits following orthopaedic surgery and effect of observer presence. PLoS ONE 2020, 15, e0240605. [Google Scholar] [CrossRef]
- Pinho, R.I.H.; Luna, S.I.P.L.; Trindade, P.H.E.; Justo, A.A.; Cima, D.S.; Fonseca, M.W.; Minto, B.W.; Rocha, F.D.L.; Miller, A.; Flecknell, P.; et al. Validation of the Rabbit Pain Behaviour Scale (RPBS) to assess acute postoperative pain in rabbits (Oryctolagus Cuniculus). PLoS ONE 2022, 17, e0268973. [Google Scholar] [CrossRef]
- Belli, M.; de Oliveira, A.R.; de Lima, M.T.; Trindade, P.H.E.; Steagall, P.V.; Luna, S.P.L. Clinical validation of the short and long UNESP-Botucatu scales for feline pain assessment. PeerJ 2021, 9, e11225. [Google Scholar] [CrossRef] [PubMed]
- Luna, S.P.L.; Trindade, P.H.E.; Monteiro, B.P.; Crosignani, N.; della Rocca, G.; Ruel, H.L.M.; Yamashita, K.; Kronen, P.; Te Tseng, C.; Teixeira, L.; et al. Multilingual validation of the short form of the Unesp-Botucatu feline pain scale (UFEPS-SF). PeerJ 2022, 10, e13134. [Google Scholar] [CrossRef]
- de Oliveira, F.A.; Luna, S.P.L.; do Amaral, J.B.; Rodrigues, K.A.; Sant’Anna, A.C.; Daolio, M.; Brondani, J.T. Validation of the UNESP-Botucatu unidimensional composite pain scale for assessing postoperative pain in cattle. BMC Vet. Res. 2014, 10, 1–14. [Google Scholar] [CrossRef]
- Gleerup, K.B.; Andersen, P.H.; Munksgaard, L.; Forkman, B. Pain evaluation in dairy cattle. Appl. Anim. Behav. Sci. 2015, 171, 25–32. [Google Scholar] [CrossRef]
- Luna, S.P.L.; de Araújo, A.L.; da Nóbrega Neto, P.I.; Brondani, J.T.; de Oliveira, F.A.; Azerêdo, L.M.D.S.; Telles, F.G.; Trindade, P.H.E. Validation of the UNESP-Botucatu pig composite acute pain scale (UPAPS). PLoS ONE 2020, 15, e0233552. [Google Scholar] [CrossRef]
- da Rocha, P.B.; Driessen, B.; McDonnell, S.M.; Hopster, K.; Zarucco, L.; Gozalo-Marcilla, M.; Hopster-Iversen, C.; Trindade, P.H.E.; da Rocha, T.K.G.; Taffarel, M.O.; et al. A critical evaluation for validation of composite and unidimensional postoperative pain scales in horses. PLoS ONE 2021, 16, e0255618. [Google Scholar] [CrossRef]
- Taffarel, M.O.; Luna, S.P.L.; de Oliveira, F.A.; Cardoso, G.S.; de Moura Alonso, J.; Pantoja, J.C.; Brondani, J.T.; Love, E.; Taylor, P.; White, K.; et al. Refinement and partial validation of the UNESP-Botucatu Multidimensional composite pain scale for assessing postoperative pain in horses. BMC Vet. Res. 2015, 11, 1–12. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, M.G.C.; de Paula, V.V.; Mouta, A.N.; Lima, I.D.O.; Macêdo, L.B.D.; Nunes, T.L.; Trindade, P.H.E.; Luna, S.P.L. Validation of the Donkey Pain Scale (DOPS) for assessing postoperative pain in donkeys. Front. Vet. Sci. 2021, 8, 532. [Google Scholar] [CrossRef] [PubMed]
- Trindade, P.H.E.; Taffarel, M.O.; Luna, S.P.L. Spontaneous behaviors of post-orchiectomy pain in horses regardless of the effects of time of day, anesthesia, and analgesia. Animals 2021, 11, 1629. [Google Scholar] [CrossRef] [PubMed]
- Box, G.E.; Draper, N.R. Empirical Model-Building and Response Surfaces; John Wiley & Sons: New York, NY, USA, 1987. [Google Scholar]
- Mead, B.E.; Karp, J.M. All models are wrong, but some organoids may be useful. Genome Biol. 2019, 20, 1–3. [Google Scholar] [CrossRef] [PubMed]
- McLennan, K.; Mahmoud, M. Development of an automated pain facial expression detection system for sheep (Ovis Aries). Animals 2019, 9, 196. [Google Scholar] [CrossRef]
- Noor, A.; Zhao, Y.; Koubaa, A.; Wu, L.; Khan, R.; Abdalla, F.Y.O. Automated sheep facial expression classification using deep transfer learning. Comput. Electron. Agric. 2020, 175, 105528. [Google Scholar] [CrossRef]
- Evangelista, M.C.; Watanabe, R.; Leung, V.S.Y.; Monteiro, B.P.; O’Toole, E.; Pang, D.S.J.; Steagall, P.V. Facial Expressions of pain in cats: The development and validation of a feline grimace scale. Sci. Rep. 2019, 9, 19128. [Google Scholar] [CrossRef]
- Viscardi, A.V.; Hunniford, M.; Lawlis, P.; Leach, M.; Turner, P.V. Development of a piglet grimace scale to evaluate piglet pain using facial expressions following castration and tail docking: A pilot study. Front. Vet. Sci. 2017, 4, 51. [Google Scholar] [CrossRef]


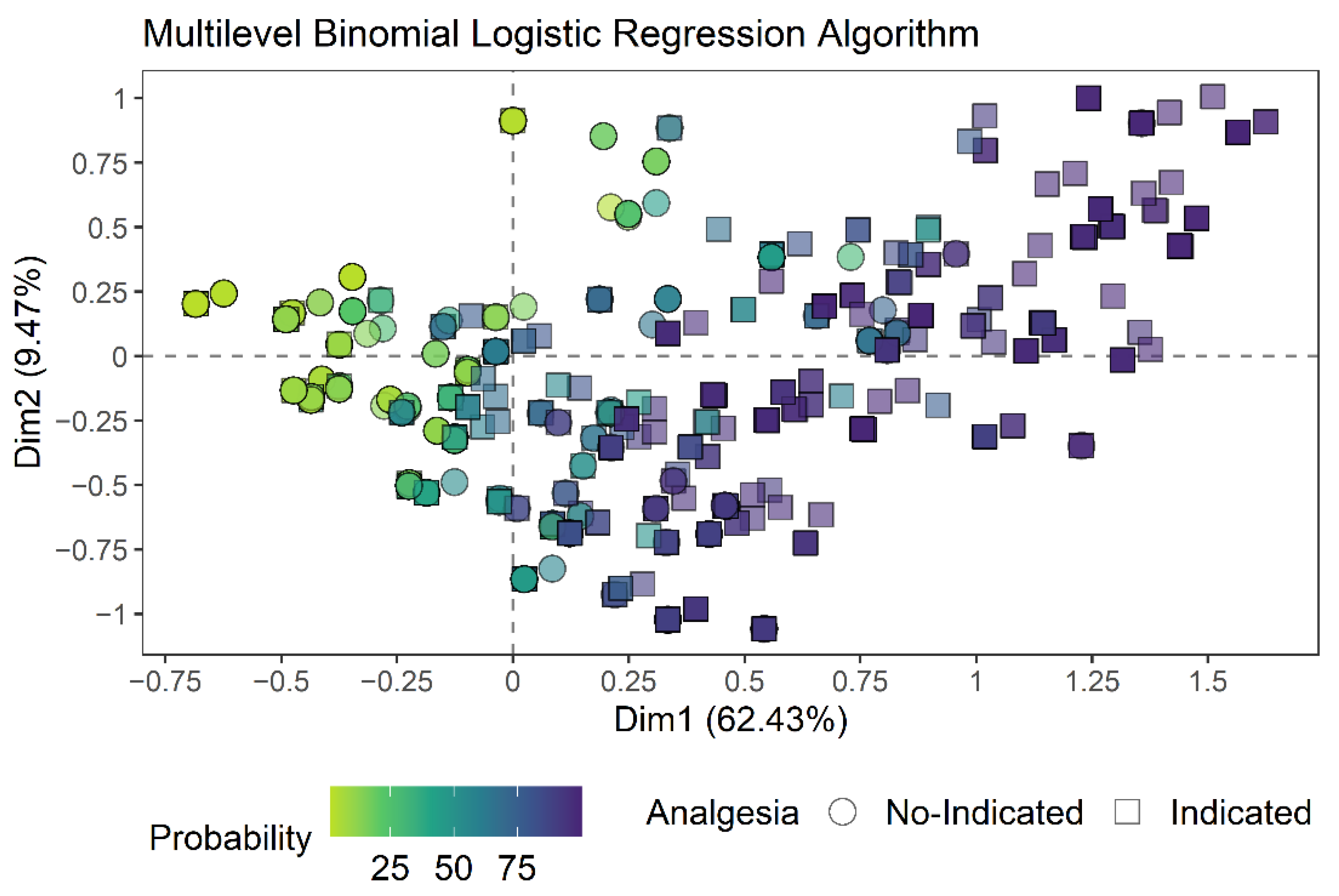
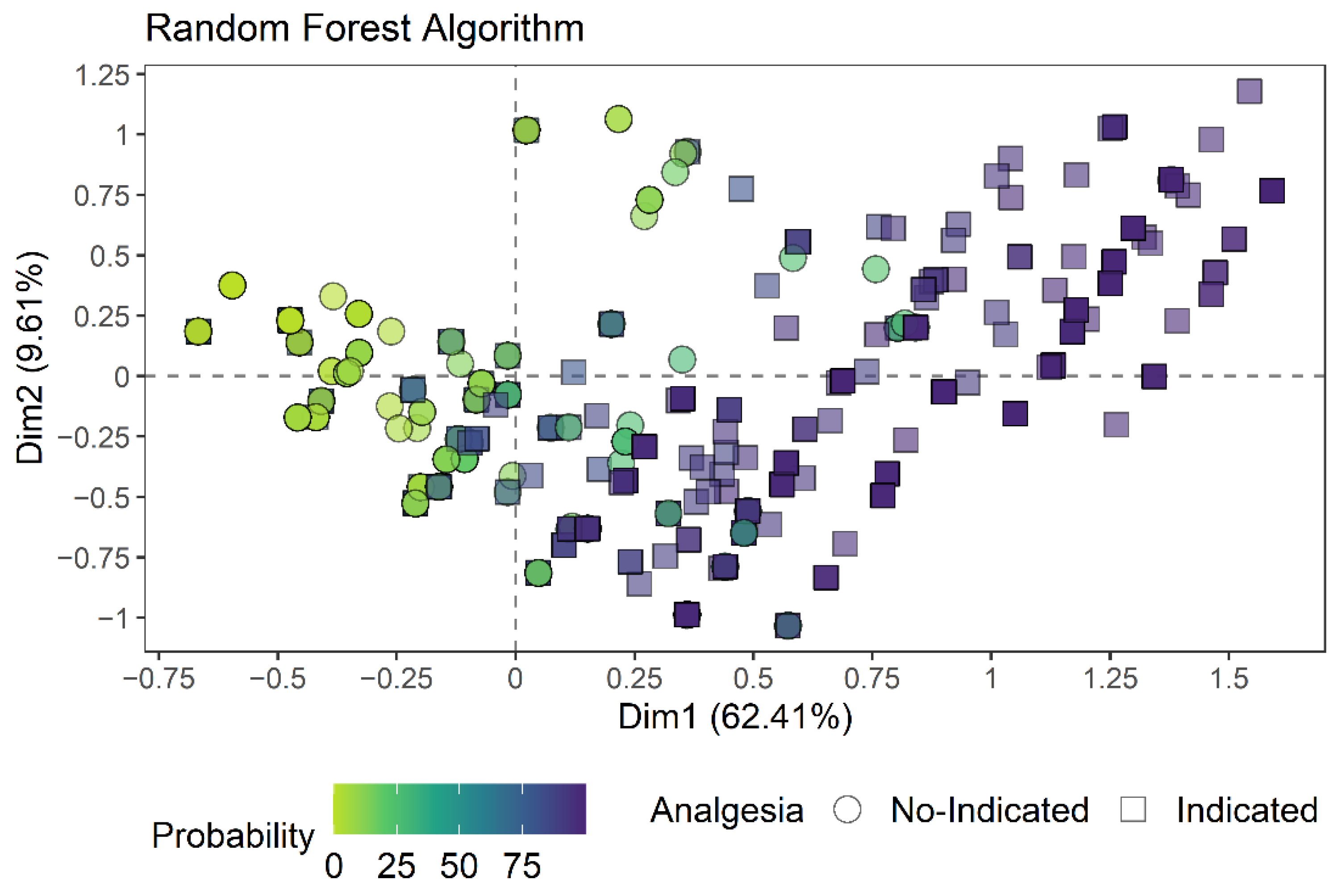
| N = 1536 | Slope Coefficient (β) | |||
|---|---|---|---|---|
| Fixed Effects | Estimate | SE | Z-Value | p-Value |
| Linear coefficient (α) | −4.0592 | 0.3949 | −10.2799 | 8.69−25 **** |
| Interaction score | ||||
| (0) Active, attentive to the environment, interacts and/or follows other animals | ||||
| (1) Apathetic: may remain close to other animals, but interacts little | 1.4327 | 0.2411 | 5.9415 | 2.83−9 **** |
| (2) Very apathetic: isolated or not interacting with other animals, not interested in the environment | 2.6128 | 0.4838 | 5.4004 | 6.65−8 **** |
| Locomotion score | ||||
| (0) Moves about freely, without altered locomotion; when stopped, the pelvic limbs are parallel to the thoracic limbs | ||||
| (1) Moves about with restriction and/or short steps and/or pauses and/or lameness; when stopped, the thoracic or pelvic limbs may be more open and further back than normal | 2.2143 | 0.2555 | 8.6650 | 4.51−18 **** |
| (2) Difficulty and/or reluctant to stand up and/or not moving and/or walking abnormally and/or limping; may lean against a surface | 2.8235 | 0.3096 | 9.1207 | 7.46−20 **** |
| Head position score | ||||
| (0) Head above the withers or eating | ||||
| (1) Head at the height of withers | 0.1648 | 0.2469 | 0.6677 | 0.5043 |
| (2) Head below the withers (except when eating) | 0.1400 | 0.3513 | 0.3984 | 0.6903 |
| Appetite score | ||||
| (0) Normorexia and/or rumination present | ||||
| (1) Hyporexia | 1.5283 | 0.4657 | 3.2821 | 0.0010 *** |
| (2) Anorexia | 0.4517 | 0.3172 | 1.4239 | 0.1545 |
| Activity score | ||||
| (0) Moves normally | ||||
| (1) Restless, moves more than normal, or lies down and stands up frequently | 1.9310 | 0.3507 | 5.5059 | 3.67−8 **** |
| (2) Moves less frequently or only when stimulated using a stick or does not move | 1.6895 | 0.2530 | 6.6781 | 2.42−11 **** |
| Posture score | ||||
| Arched back | 0.5527 | 0.4081 | 1.3543 | 0.1756 |
| Extends the head and neck | 0.6284 | 0.3031 | 2.0729 | 0.0382 ** |
| Lying down with head resting on the ground or close to the ground | 0.7387 | 0.4271 | 1.7295 | 0.0837 * |
| Moves the tail quickly (except when breastfeeding) and repeatedly and/or keeps the tail straight (except to defecate/urinate) | 2.0622 | 0.7126 | 2.8940 | 0.0038 *** |
| Random effects | Variance | SD | Groups | |
| Sheep (intercept) | 8.6422−9 | 9.2963−5 | 48 | |
| Observers (intercept) | 5.3755−2 | 2.3185−1 | 4 | |
| Moments (intercept) | 3.0738−1 | 5.5442−1 | 4 | |
| Phases (intercept) | 9.1439−8 | 3.0239−4 | 2 | |
| Model parameters | Full model | Null model | Short model | |
| Log-Likelihood (df) | −325.48 (19) | −683.97 (5) | −329.22 (14) | |
| P-value Log-Likelihood vs. Full Model | - | <2.2−16 **** | 0.1874 | |
| AIC | 688.96 | 1377.96 | 686.44 | |
| BIC | 790.36 | 1404.64 | 761.15 | |
| Pseudo-R² (fixed effects) | 0.76 | - | 0.75 | |
| Pseudo-R² (total) | 0.78 | 0.60 | 0.78 | |
| Dataset | Parameters | Original USAPS (Total Sum) | Weighted USAPS (Probability to Need Analgesia Based on Logistic Regression) | p-Value |
|---|---|---|---|---|
| 100% | Optimal cut-off | 03.50 (03.50–04.50) | 43.88 (29.61–61.38) | - |
| Specificity | 87.67 (85.50–93.49) | 91.21 (87.21–94.43) | - | |
| Sensitivity | 91.97 (85.61–93.94) | 92.88 (87.88–96.06) | - | |
| AUC | 95.32 (94.30–96.35) | 96.83 (95.98–97.68) | 1.381−9 | |
| Test data (30%) | Optimal cut-off | 03.50 (03.50–04.50) | 59.63 (43.14–66.67) | - |
| Specificity | 86.07 (80.74–93.03) | 92.21 (86.89–95.90) | - | |
| Sensitivity | 92.16 (83.82–96.08) | 92.16 (87.25–96.57) | - | |
| AUC | 94.87 (92.94–96.80) | 96.59 (95.02–98.15) | 4.891−4 |
| Dataset | Parameters | Original USAPS (Total Sum) | Weighted USAPS (Probability to Need Analgesia Based on Random Forest) | p-Value |
|---|---|---|---|---|
| Training data (70%) | Optimal cut-off | 03.50 (03.50–04.50) | 42.21 (20.68–64.09) | - |
| Specificity | 88.92 (86.55–94.94) | 94.62 (90.82–96.99) | - | |
| Sensitivity | 91.45 (85.09–94.08) | 93.42 (90.13–96.49) | - | |
| AUC | 95.47 (94.25–96.69) | 97.50 (96.56–98.45) | 1.822−9 | |
| Test data (30%) | Optimal cut-off | 03.50 (03.50–04.50) | 35.41 (35.26–65.13) | - |
| Specificity | 86.07 (80.74–93.03) | 89.34 (85.25–93.85) | - | |
| Sensitivity | 92.16 (83.82–96.08) | 95.10 (90.69–98.04) | - | |
| AUC | 94.87 (92.94–96.80) | 96.28 (94.17–97.85) | 0.0067 |
| Behavioral Items | Ranking | ||
|---|---|---|---|
| Multiple Binomial Logistic Regression | Random Forest | Delta | |
| ‘Activity 2′ | 1st | 3rd | 2 |
| ‘Locomotion 2′ | 2nd | 1st | −1 |
| ‘Interaction 2′ | 3rd | 6th | 3 |
| ‘Interaction 1′ | 4th | 4th | 0 |
| ‘Locomotion 1′ | 5th | 2nd | −3 |
| ‘Extend Head’ | 6th | 9th | 3 |
| ‘Head Position 2′ | 7th | 14th | 7 |
| ‘Appetite 1′ | 8th | 7th | −1 |
| ‘Head Position 1′ | 9th | 13th | 4 |
| ‘Activity 1′ | 10th | 5th | −5 |
| ‘Lying Down’ | 11th | 10th | −1 |
| ‘Moves Tail’ | 12th | 8th | −4 |
| ‘Appetite 2′ | 13th | 11th | −2 |
| ‘Arched Back’ | 14th | 12th | −2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Trindade, P.H.E.; Mello, J.F.S.R.d.; Silva, N.E.O.F.; Luna, S.P.L. Improving Ovine Behavioral Pain Diagnosis by Implementing Statistical Weightings Based on Logistic Regression and Random Forest Algorithms. Animals 2022, 12, 2940. https://doi.org/10.3390/ani12212940
Trindade PHE, Mello JFSRd, Silva NEOF, Luna SPL. Improving Ovine Behavioral Pain Diagnosis by Implementing Statistical Weightings Based on Logistic Regression and Random Forest Algorithms. Animals. 2022; 12(21):2940. https://doi.org/10.3390/ani12212940
Chicago/Turabian StyleTrindade, Pedro Henrique Esteves, João Fernando Serrajordia Rocha de Mello, Nuno Emanuel Oliveira Figueiredo Silva, and Stelio Pacca Loureiro Luna. 2022. "Improving Ovine Behavioral Pain Diagnosis by Implementing Statistical Weightings Based on Logistic Regression and Random Forest Algorithms" Animals 12, no. 21: 2940. https://doi.org/10.3390/ani12212940
APA StyleTrindade, P. H. E., Mello, J. F. S. R. d., Silva, N. E. O. F., & Luna, S. P. L. (2022). Improving Ovine Behavioral Pain Diagnosis by Implementing Statistical Weightings Based on Logistic Regression and Random Forest Algorithms. Animals, 12(21), 2940. https://doi.org/10.3390/ani12212940







