Environmental and Social Influences on the Behaviour of Free-Living Mandarin Ducks in Richmond Park
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Population and Study Site
2.2. Data Collection
2.3. Data Analysis
3. Results
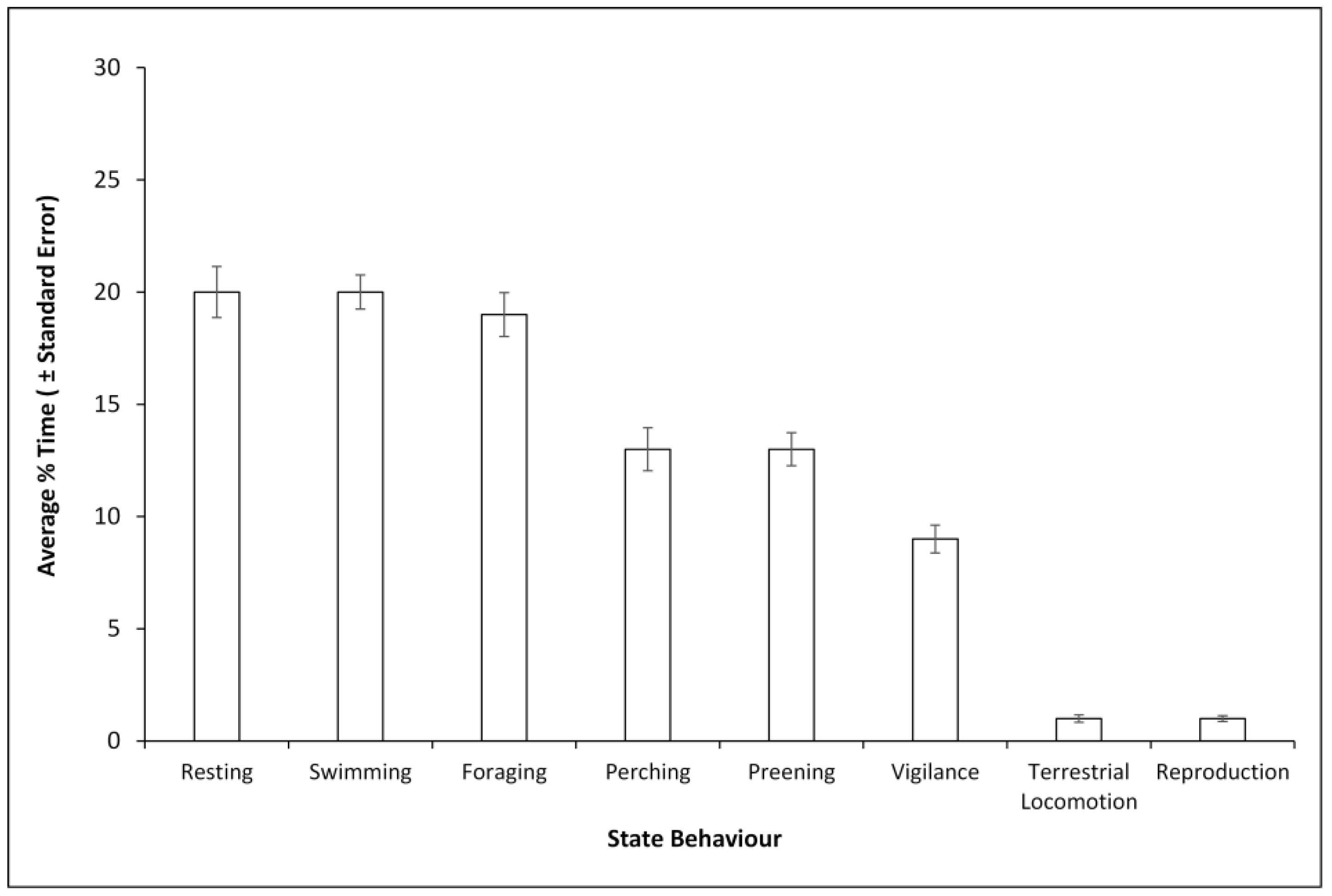
3.1. Time Budget
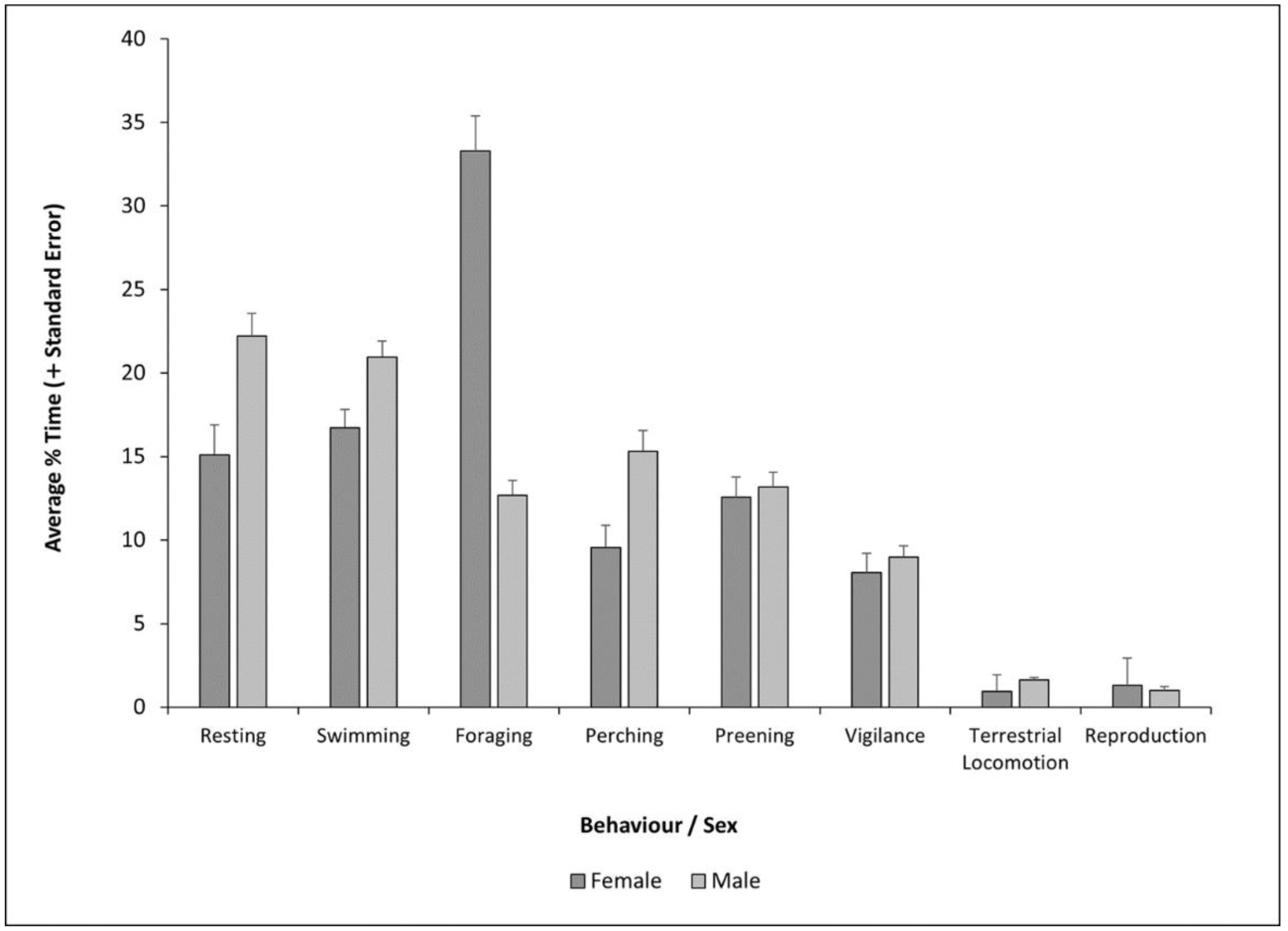
3.2. Sex Effects on Behaviour
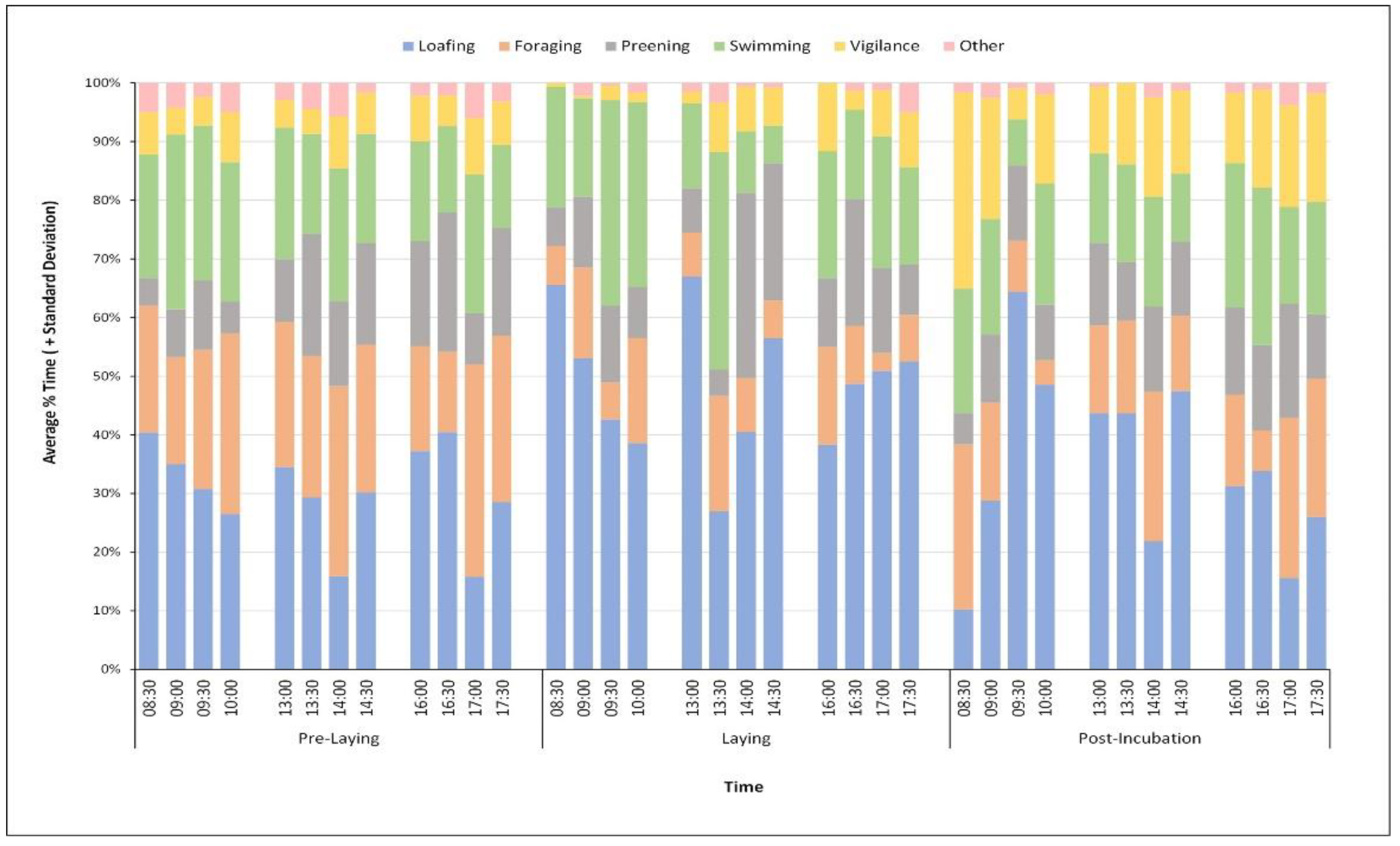
3.3. Habitat, Temporal, and Social Variables
3.4. Weather Variables
3.5. Effects of Humans and Number of Ducks on Behaviour
3.6. Wild Ducks Compared to Captive Ducks
4. Discussion
4.1. Time-Activity Budget
4.2. Sex Effects on Behaviour
4.3. Temporal and Environmental Factors
4.3.1. Effects of Weather
4.3.2. Effects of Human Presence and Number of Ducks
4.4. Comparing Wild and Captive Mandarin Behaviour
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mallapur, A.; Chellam, R. Environmental influences on stereotypy and the activity budget of Indian leopards (Panthera pardus) in four zoos in Southern India. Zoo Biol. 2002, 21, 585–595. [Google Scholar] [CrossRef]
- Rose, P.; Robert, R. Evaluating the activity patterns and enclosure usage of a little-studied zoo species, the sitatunga (Tragelaphus spekii). J. Zoo Aquar. Res. 2013, 1, 14–19. [Google Scholar] [CrossRef]
- Huettner, T.; Dollhaeupl, S.; Simon, R.; Baumgartner, K.; von Fersen, L. Activity Budget Comparisons Using Long-Term Observations of a Group of Bottlenose Dolphins (Tursiops truncatus) under Human Care: Implications for Animal Welfare. Animals 2021, 11, 2107. [Google Scholar] [CrossRef]
- Wolfensohn, S.; Shotton, J.; Bowley, H.; Davies, S.; Thompson, S.; Justice, W.S.M. Assessment of Welfare in Zoo Animals: Towards Optimum Quality of Life. Animals 2018, 8, 110. [Google Scholar] [CrossRef]
- Rose, P.E. Ensuring a good quality of life in the zoo. Underpinning welfare-positive animal management with ecological evidence. In Zoo Animals: Behavior, Welfare and Public Interactions; Berger, M., Corbett, S., Eds.; Nova Science Publishers Inc.: New York, NY, USA, 2018; pp. 141–198. [Google Scholar]
- Dawkins, M.S. Observing Animal Behaviour: Design and Analysis of Quantitative Data; Oxford University Press: Oxford, UK, 2007. [Google Scholar]
- Manning, A.; Dawkins, M.S. An Introduction to Animal Behaviour; Cambridge University Press: Cambridge, UK, 2012; Volume 6. [Google Scholar]
- Mench, J. Why It is Important to Understand Animal Behavior. ILAR J. 1998, 39, 20–26. [Google Scholar] [CrossRef]
- Dawkins, M.S. The Science of Animal Welfare: Understanding What Animals Want; Oxford University Press: Oxford, UK, 2021. [Google Scholar]
- Kleiman, D.G. Introduction (Conservation & Research). In Wild Mammals in Captivity: Principles and Techniques for Zoo Management; Kleiman, D.G., Thompson, K.V., Baer, C.K., Eds.; University of Chicago Press: Chicago, IL, USA, 2010; pp. 217–218. [Google Scholar]
- Sutherland, W.J. The importance of behavioural studies in conservation biology. Anim. Behav. 1998, 56, 801–809. [Google Scholar] [CrossRef]
- Kleiman, D.G.; Thompson, K.V.; Baer, C.K. Wild Mammals in Captivity: Principles and Techniques for Zoo Management; University of Chicago Press: Chicago, IL, USA, 2010. [Google Scholar]
- De Azevedo, C.S.; Cipreste, C.F.; Young, R.J. Environmental enrichment: A GAP analysis. Appl. Anim. Behav. Sci. 2007, 102, 329–343. [Google Scholar] [CrossRef]
- Melfi, V.A. There are big gaps in our knowledge, and thus approach, to zoo animal welfare: A case for evidence-based zoo animal management. Zoo Biol. 2009, 28, 574–588. [Google Scholar] [CrossRef]
- Rose, P.E.; Brereton, J.E.; Rowden, L.J.; De Figueiredo, R.L.; Riley, L. What’s new from the zoo? An analysis of ten years of zoo-themed research output. Palgrave Commun. 2019, 5, 128. [Google Scholar] [CrossRef]
- Stoinski, T.S.; Lukas, K.E.; Maple, T.L. A survey of research in North American zoos and aquariums. Zoo Biol. 1998, 17, 167–180. [Google Scholar] [CrossRef]
- Binding, S.; Farmer, H.; Krusin, L.; Cronin, K. Status of animal welfare research in zoos and aquariums: Where are we, where to next? J. Zoo Aquar. Res. 2020, 8, 166–174. [Google Scholar]
- Rose, P.; O’Brien, M. Welfare Assessment for Captive Anseriformes: A Guide for Practitioners and Animal Keepers. Animals 2020, 10, 1132. [Google Scholar] [CrossRef] [PubMed]
- McKinney, F. The Comfort Movements of Anatidae. Behaviour 1965, 25, 120–220. [Google Scholar] [CrossRef]
- Kear, J.; Hulme, M. Ducks, Geese and Swans (Bird Families of the World); Oxford University Press: Oxford, UK, 2005. [Google Scholar]
- Bruggers, R.L.; Jackson, W.B. Time budgets of mandarin ducks under semi-natural conditions. Wildfowl 1977, 28, 7. [Google Scholar]
- Yijin, Z.; Mingqin, S.; Peng, C.; Bin, C. Time Budget and Activity Rhythm of the Mandarin Duck Aix galericulata in the Poyang Lake Watershed. Pak. J. Zool. 2019, 51, 725–730. [Google Scholar] [CrossRef]
- Scott, P. A Coloured Key to the Wildfowl of the World; The Wildfowl & Wetlands Trust: Slimbridge, UK, 2006. [Google Scholar]
- Shurtleff, L.L.; Savage, C. The Wood Duck and the Mandarin: The Northern Wood Ducks; University of California Press: Berkeley, CA, USA, 1996. [Google Scholar]
- Krapu, G.L.; Reinecke, K.J. Foraging ecology and nutrition. In Ecology and Management of Breeding Waterfowl; Batt, B.D.J., Afton, A.D., Anderson, M.G., Ankney, C.D., Johnson, D.H., Kadlec, J.A., Krapu, G.L., Eds.; University of Minnesota Press: Minnesota, MN, USA, 1992; pp. 1–29. [Google Scholar]
- Krapu, G.L.; Swanson, G.A. Some Nutritional Aspects of Reproduction in Prairie Nesting Pintails. J. Wildl. Manag. 1975, 39, 156. [Google Scholar] [CrossRef]
- Wong, B.B.M.; Candolin, U. Behavioral responses to changing environments. Behav. Ecol. 2014, 26, 665–673. [Google Scholar] [CrossRef]
- Heise, J.R.; Williams, C.K.; Castelli, P.M. Factors Influencing a 24-Hour Time-Budget for Wintering Atlantic Brant. J. Fish Wildl. Manag. 2019, 10, 79–90. [Google Scholar] [CrossRef]
- Ali, E.; Ismahan, H.; Moussa, H. Time budget patterns and complementary use of a Mediterranean wetland (Tonga, Northeast Algeria) by migrant and resident waterbirds. Riv. Ital. Di Ornitol. 2016, 86, 55. [Google Scholar] [CrossRef]
- Rose, P.E. Investigating the Behaviour and Welfare of Captive Flamingos (Phoenicopterformes) in College of Life & Environmental Sciences; University of Exeter: Exeter, UK, 2018. [Google Scholar]
- Sebastián-González, E.; Green, A.J. Habitat Use by Waterbirds in Relation to Pond Size, Water Depth, and Isolation: Lessons from a Restoration in Southern Spain. Restor. Ecol. 2014, 22, 311–318. [Google Scholar] [CrossRef]
- Bennett, J.W.; Bolen, E.G. Stress Response in Wintering Green-Winged Teal. J. Wildl. Manag. 1978, 42, 81. [Google Scholar] [CrossRef]
- Elkins, N. Weather and Bird Behaviour; T & A. D. Poyser: London, UK, 1983. [Google Scholar]
- Liechti, F. Birds: Blowin by the wind? J. Ornithol. 2006, 147, 202–211. [Google Scholar] [CrossRef]
- Shao, M.; Chen, B. Effect of sex, temperature, time and flock size on the diving behavior of the wintering Scaly-sided Merganser (Mergus squamatus). Avian Res. 2017, 8, 9. [Google Scholar] [CrossRef]
- Tassicker, A.L.; Kutt, A.S.; Vanderduys, E.; Mangru, S. The effects of vegetation structure on the birds in a tropical savanna woodland in north-eastern Australia. Rangel. J. 2006, 28, 139–152. [Google Scholar] [CrossRef]
- Burger, J.; Gochfeld, M. Effects of ecotourists on bird behaviour at Loxahatchee National Wildlife Refuge, Florida. Environ. Conserv. 1998, 25, 13–21. [Google Scholar] [CrossRef]
- Price, M. The impact of human disturbance on birds: A selective review. Aust. Zool. 2008, 34, 163–196. [Google Scholar] [CrossRef]
- Udvardy, M.D.F. The Significance of Interspecific Competition in Bird Life. Oikos 1951, 3, 98. [Google Scholar] [CrossRef]
- Hurtado, M.J.; Ebensperger, L.A. Seasonal changes in the time budget of degus, Octodon degus. Behaviour 2005, 142, 91–112. [Google Scholar] [CrossRef]
- Veasey, J.S.; Waran, N.K.; Young, R.J. On comparing the behaviour of zoo housed animals with wild conspecifics as a welfare indicator. Anim. Welf. 1996, 5, 13–24. [Google Scholar]
- Waran, N.; Randle, H. What we can measure, we can manage: The importance of using robust welfare indicators in Equitation Science. Appl. Anim. Behav. Sci. 2017, 190, 74–81. [Google Scholar] [CrossRef]
- Rose, P.; Roper, A.; Banks, S.; Giorgio, C.; Timms, M.; Vaughan, P.; Hatch, S.; Halpin, S.; Thomas, J.; O’Brien, M. Evaluation of the time-activity budgets of captive ducks (Anatidae) compared to wild counterparts. Appl. Anim. Behav. Sci. 2022, 251, 105626. [Google Scholar] [CrossRef]
- The Royal Parks. History and Architecture. Available online: https://www.royalparks.org.uk/parks/richmond-park/about-richmond-park/history-and-architecture (accessed on 25 August 2022).
- The Royal Parks. Wildlife. Available online: https://www.royalparks.org.uk/parks/richmond-park/things-to-see-and-do/wildlife (accessed on 25 August 2022).
- The Royal Parks. Isabella Plantation. Available online: https://www.royalparks.org.uk/parks/richmond-park/things-to-see-and-do/isabella-plantation (accessed on 25 August 2022).
- The Royal Parks. ‘Help Nature Thrive’ in the Royal Parks by Not Feeding Wildlife. Available online: https://www.royalparks.org.uk/media-centre/press-releases/help-nature-thrive-in-the-royal-parks-by-not-feeding-wildlife (accessed on 25 August 2022).
- Lever, C. The Mandarin Duck; Bloomsbury Publishing: London, UK, 2013. [Google Scholar]
- BOU. The British List. Available online: https://bou.org.uk/british-list/ (accessed on 25 August 2022).
- RSPB. Mandarin Duck. Available online: https://www.rspb.org.uk/birds-and-wildlife/wildlife-guides/bird-a-z/mandarin/ (accessed on 25 August 2022).
- Woodward, I.D.; Massimino, D.; Hammond, M.J.; Barber, L.; Barimore, C.; Harris, S.J.; Leech, D.I.; Noble, D.G.; Walker, R.H.; Baillie, S.R.; et al. BirdTrends 2020: Trends in Numbers, Breeding Success and Survival for UK Breeding Birds in BTO Research Report 732; British Trust for Ornithology: Thetford, UK, 2020. [Google Scholar]
- Cosgrove, P. Mandarin ducks in northern Scotland and the potential consequences for breeding goldeneye. Scott. Birds 2003, 24, 1–10. [Google Scholar]
- Downs, J.; Loraamm, R.; Anderson, J.; Perry, J.; Bullock, J. Habitat Use and Behaviours of Introduced Muscovy Ducks (Cairina moschata) in Urban and Suburban Environments. Suburb. Sustain. 2017, 5, 1. [Google Scholar] [CrossRef]
- Yang, S.; Zhou, L.; Lin, W.; Li, X.; Lu, M.; Liu, C. Behavioral Differentiation between Anas poecilorhyncha and Domestic Duck. J. Agric. Sci. Technol. 2016, 6, 270–282. [Google Scholar] [CrossRef][Green Version]
- Bateson, M.; Martin, P. Measuring Behaviour: An Introductory Guide, 4th ed.; Cambridge University Press: Cambridge, UK, 2021. [Google Scholar]
- IBM Corp. IBM SPSS Statistics for Windows, Version 26.0; IBM: Armonk, NY, USA, 2019. [Google Scholar]
- Plowman, A.B. Analysing the results. In Handbook of Zoo and Aquarium Research; BIAZA: London, UK, 2013; pp. 22–45. [Google Scholar]
- Giroux, J.-F.; Bédard, J.; Bédard, Y. Time budget of greater snow geese during the brood-rearing period. Wildfowl 1986, 37, 46–50. [Google Scholar]
- Wlodarczyk, R. The daily activity pattern in males and females of the Mute Swan (Cygnus olor, Anseriformes) during different parts of the breeding season. North-West. J. Zool. 2017, 13, 85–93. [Google Scholar]
- Afton, A.D. Time Budget of Breeding Northern Shovelers. Wilson Bull. 1979, 91, 42–49. [Google Scholar]
- Asplund, C. Time budgets of breeding mallard in northern Sweden. Wildfowl 1981, 32, 55–64. [Google Scholar]
- Chettibi, F.; Khelifa, R.; Aberkane, M.; Bouslama, Z.; Houhamdi, M. Diurnal activity budget and breeding ecology of the white-headed duck Oxyura leucocephala at Lake Tonga (Northeast Algeria). Zool. Ecol. 2013, 23, 183–190. [Google Scholar] [CrossRef]
- Dwyer, T.J. Time Budget of Breeding Gadwalls. Wilson Bull. 1975, 87, 335–343. [Google Scholar]
- Arzel, C.; Elmberg, J. Time use, foraging behavior and microhabitat use in a temporary guild of spring-staging dabbling ducks (Anas spp.). Ornis Fenn. 2004, 81, 157–168. [Google Scholar]
- Dwyer, T.J. Social behavior of breeding gadwalls in North Dakota. Auk 1974, 91, 375–386. [Google Scholar]
- Seymour, N.R.; Titman, R.D. Changes in activity patterns, agonistic behavior, and territoriality of black ducks (Anas rubripes) during the breeding season in a Nova Scotia tidal marsh. Can. J. Zool. 1978, 56, 1773–1785. [Google Scholar] [CrossRef]
- Ricklefs, R.E. The energetics of reproduction in birds. In Avian Energetics; Paynter, R.A., Ed.; Nuttall Ornithological Club: Cambridge, MA, USA, 1974; pp. 152–297. [Google Scholar]
- Christensen, T. Female pre-nesting foraging and male vigilance in Common Eider, Somateria mollissima. Bird Study 2000, 47, 311–319. [Google Scholar] [CrossRef][Green Version]
- Jedlikowski, J.; Chibowski, P.; Karasek, T.; Brambilla, M. Multi-scale habitat selection in highly territorial bird species: Exploring the contribution of nest, territory and landscape levels to site choice in breeding rallids (Aves: Rallidae). Acta Oecol. 2016, 73, 10–20. [Google Scholar] [CrossRef]
- Kennamer, R.A.; Harvey, W.F., IV.; Hepp, G.R. Embryonic Development and Nest Attentiveness of Wood Ducks during Egg Laying. Condor 1990, 92, 587–592. [Google Scholar] [CrossRef]
- Finney, S.K.; Wanless, S.; Harris, M.P. The Effect of Weather Conditions on the Feeding Behaviour of a Diving Bird, the Common Guillemot Uria aalge. J. Avian Biol. 1999, 30, 23–30. [Google Scholar] [CrossRef]
- Cooper, S.J.; Swanson, D.L. Seasonal Acclimatization of Thermoregulation in the Black-Capped Chickadee. Condor 1994, 96, 638–646. [Google Scholar] [CrossRef]
- Dawson, W.R.; Marsh, R.L.; Buttemer, W.A.; Carey, C. Seasonal and Geographic Variation of Cold Resistance in House Finches Carpodacus mexicanus. Physiol. Zool. 1983, 56, 353–369. [Google Scholar] [CrossRef]
- Whyte, R.J.; Bolen, E.G. Impact of Winter Stress on Mallard Body Composition. Condor 1984, 86, 477–482. [Google Scholar] [CrossRef]
- Rijke, A.M. Wettability and Phylogenetic Development of Feather Structure in Water Birds. J. Exp. Biol. 1970, 52, 469–479. [Google Scholar] [CrossRef]
- Şekercioğlu, Ç.H.; Primack, R.B.; Wormworth, J. The effects of climate change on tropical birds. Biol. Conserv. 2012, 148, 1–18. [Google Scholar] [CrossRef]
- Rodenburg, T.B.; Bracke, M.B.M.; Berk, J.; Cooper, J.E.; Faure, J.M.; Guémené, D.G.; Guy, G.; Harlander, A.; Jones, T.; Knierim, U. Welfare of ducks in European duck husbandry systems. World’s Poult. Sci. J. 2005, 61, 633–646. [Google Scholar] [CrossRef]
- García-Arroyo, M.; MacGregor-Fors, I. Tolerant to humans? Assessment of alert and flight initiation distances of two bird species in relation to sex, flock size, and environmental characteristics. Ethol. Ecol. Evol. 2020, 32, 445–456. [Google Scholar] [CrossRef]
- De Azevedo, C.S.; Lima, M.F.F.; da Silva, V.C.A.; Young, R.J.; Rodrigues, M. Visitor influence on the behavior of captive greater rheas (Rhea americana, Rheidae Aves). J. Appl. Anim. Welf. Sci. 2012, 15, 113–125. [Google Scholar] [CrossRef] [PubMed]
- Rose, P.E.; Scales, J.S.; Brereton, J.E. Why the “visitor effect” is complicated. Unraveling individual animal, visitor number, and climatic influences on behavior, space use and interactions with keepers—A case study on captive hornbills. Front. Vet. Sci. 2020, 7, 236. [Google Scholar] [CrossRef]
- Samia, D.S.M.; Nakagawa, S.; Nomura, F.; Rangel, T.F.; Blumstein, D.T. Increased tolerance to humans among disturbed wildlife. Nat. Commun. 2015, 6, 8877. [Google Scholar] [CrossRef]
- Murray, M.H.; Becker, D.J.; Hall, R.J.; Hernandez, S.M. Wildlife health and supplemental feeding: A review and management recommendations. Biol. Conserv. 2016, 204, 163–174. [Google Scholar] [CrossRef]
- Clayton, D.A. Socially facilitated behavior. Q. Rev. Biol. 1978, 53, 373–392. [Google Scholar] [CrossRef]
- Clayton, D.A. The effects of pre-test conditions on social facilitation of drinking in ducks. Anim. Behav. 1976, 24, 125–134. [Google Scholar] [CrossRef]
- Liste, G.; Asher, L.; Broom, D.M. When a Duck Initiates Movement, Do Others Follow? Testing Preference in Groups. Ethology 2014, 120, 1199–1206. [Google Scholar] [CrossRef]
- Dickens, M.J.; Bentley, G.E. Stress, captivity, and reproduction in a wild bird species. Horm. Behav. 2014, 66, 685–693. [Google Scholar] [CrossRef] [PubMed]
- El-Lethey, H.; Huber-Eicher, B.; Jungi, T.W. Exploration of stress-induced immunosuppression in chickens reveals both stress-resistant and stress-susceptible antigen responses. Veter. Immunol. Immunopathol. 2003, 95, 91–101. [Google Scholar] [CrossRef]
- Shini, S.; Huff, G.R.; Shini, A.; Kaiser, P. Understanding stress-induced immunosuppression: Exploration of cytokine and chemokine gene profiles in chicken peripheral leukocytes. Poult. Sci. 2010, 89, 841–851. [Google Scholar] [CrossRef] [PubMed]
- Treep, J.; Bohrer, G.; Shamoun-Baranes, J.; Duriez, O.; de Moraes Frasson, R.P.; Bouten, W. Using High-Resolution GPS Tracking Data of Bird Flight for Meteorological Observations. Bull. Am. Meteorol. Soc. 2016, 97, 951–961. [Google Scholar] [CrossRef]
- Williams, H.J.; Shepard, E.L.C.; Duriez, O.; Lambertucci, S.A. Can accelerometry be used to distinguish between flight types in soaring birds? Anim. Biotelemetry 2015, 3, 45. [Google Scholar] [CrossRef]


| Category | Behaviour | Description |
|---|---|---|
| States | ||
| Active | Natural Foraging | Surface feeding, diving feeding, catching, or swallowing food or duck seeks out food (naturally) in and out of water |
| Feeding by Visitor | Surface feeding, diving feeding, catching, or swallowing food or duck seeks out food (from visitors) in and out of water | |
| Preening | Any element of the preening sequence including nibbling feathers, head rolls and shaking, that occurs when duck is either in the water or on land | |
| Terrestrial Locomotion | Slow or rapid movement on the ground, out of the water | |
| Swimming | Slow or rapid movement on the water with no foraging behaviour | |
| Reproduction | Courtship: Drake performs a drinking-preening-behind-the-wing sequence in the water/Hen nibbles the throat region of mate and utters coquette call. Hen flattens herself on the water in copulation posture, turning around and around Copulation: Drake is on top of a hen mating in the water | |
| Vigilance | Duck is stood upright, motionless, alert, and watchful, focusing on a particular alarming stimulus for a relatively long duration | |
| Flying | Flying for a relatively long duration, usually away from the study site | |
| Inactive | Resting | Loaf or sleeping such as eyes are closed (or one eye is closed), neck is short, no head movements and/or or bill is tucked under wing on either water or land |
| Perching | Loaf or sleeping such as eyes are closed (or one eye is closed), neck is short, no head movements and/or bill is tucked under wing in either a tree, on a branch or above ground | |
| Events | ||
| Vocalisation | Display call is like a thin, whistling and rapidly rising “hueessst, accompanied by deeper clappering sounds. Other sounds include a short and sharp, coot-like “ket”, and a short “ack”. | |
| Alert | Duck raises head and is attentive for a very brief moment during the performance of a state behaviour and then resumes the original behaviour after a few seconds. | |
| Fleeing | Duck is moving quickly away from another animal, usually in response to a threat or other aggressive behaviour | |
| Conspecific Social Interaction | Any brief interaction with another Mandarin including pecking, aggression or chasing | |
| Interspecific Social Interaction | Any brief interaction with another species of bird, including pecking, aggression or chasing | |
| Flight | Any brief flight which usually occurs within the study site | |
| Maintenance | Mostly body fluffing, body shaking and wing flapping but also sometimes stretching, scratching, flapping, bathing, head dip in water |
| 95% Walk Confidence Interval for Difference | |||||||
|---|---|---|---|---|---|---|---|
| Factor | Behaviour | Median ± IQR | p Value | RR | Lower | Upper | |
| Sex | Female | Natural Foraging | 20% ± 50 | <0.001 | 3.53 | 2.88 | 4.34 |
| Male | 0% ± 15 | 1 | |||||
| Female | Vigilance | 0% ± 5 | <0.001 | 0.08 | 0.46 | 0.13 | |
| Male | 0% ± 15 | 1 | |||||
| Female | Vocalisation | 0% ± 0 | <0.001 | 0.64 | 0.51 | 0.81 | |
| Male | 0% ± 0.00084 | 1 | |||||
| Pond ID | Peg’s Pond | Swimming | 15% ± 35 | 0.006 | 1.39 | 1.01 | 1.75 |
| Thomson’s Pond | 15% ± 20 | 1 | |||||
| Social Period | Pre-Laying | Natural Foraging | 7.5 ± 30 | 0.006 | 0.54 | 0.35 | 0.84 |
| Post-Incubation | 0% ± 20 | 1 | |||||
| Pre-Laying | Natural Foraging | 7.5 ± 30 | 0.003 | 2.09 | 1.29 | 3.38 | |
| Laying | 0% ± 10 | 1 | |||||
| Pre-Laying | Activity | 27.5% ± 80 | <0.001 | 1.40 | 1.17 | 1.67 | |
| Laying | 20% ± 65.5 | 1 | |||||
| Pre-Laying | Activity | 27.5% ± 80 | 0.020 | 1.30 | 1.05 | 1.60 | |
| Post-Incubation | 25% ± 95 | 1 | |||||
| Laying | Inactivity | 25% ± 65 | <0.001 | 1.51 | 1.21 | 1.89 | |
| Pre-Laying | 5% ± 35 | 1 | |||||
| Laying | Inactivity | 25% ± 65 | <0.001 | 1.65 | 1.26 | 2.16 | |
| Post-Incubation | 0% ± 36 | 1 | |||||
| Vegetation Coverage | Open Water | Vigilance | 0% ± 15 | <0.001 | 1.82 | 1.41 | 2.34 |
| Close to Veg | 0% ± 5 | 1 | |||||
| Open Water | Resting | 0% ± 20 | <0.001 | 0.48 | 0.37 | 0.62 | |
| Close to Veg | 15% ± 60 | 1 | |||||
| Open Water | Activity | 35% ± 95 | <0.001 | 1.96 | 1.66 | 2.30 | |
| Close to Veg | 10% ± 47.5 | 1 | |||||
| Weather Description | Light Cloud | Natural Foraging | 5% ± 25 | 0.016 | 0.10 | 0.02 | 0.65 |
| Light Rain | 0% ± 15 | 0.023 | 10.10 | .0.02 | 0.72 | ||
| Sunny | 5% ± 29 | 0.025 | 0.11 | 0.02 | 0.76 | ||
| Sunny Intervals | 5% ± 30 | 0.008 | 10.08 | .0.02 | 0.51 | ||
| Thick Cloud | 5% ± 36 | 0.032 | 0.09 | 0.01 | 0.81 | ||
| Heavy Rain | 0% ± 0 | 1 | |||||
| Light Cloud | Swimming | 15% ± 30 | <0.001 | 0.21 | 0.09 | 0.50 | |
| Light Rain | 15% ± 40 | <0.001 | 0.16 | 0.07 | 0.39 | ||
| Sunny | 15% ± 20 | <0.001 | 0.18 | 0.08 | 0.44 | ||
| Sunny Intervals | 15% ± 20 | <0.001 | 10.21 | 0.09 | 0.50 | ||
| Thick Cloud | 15% ± 26 | <0.001 | 0.25 | 0.09 | 0.72 | ||
| Heavy Rain | 0% ± 7.5 | 1 | |||||
| Light Rain | Alertness | 0 ± 0 | 0.036 | 1.69 | 1.04 | 2.76 | |
| Sunny Intervals | 0 ± 0.000833 | 1 | |||||
| Temperature | Swimming | 0.048 | 0.97 | 0.93 | 0.05 | ||
| Precipitation | Maintenance | 0.001 | 1.01 | 1.00 | 1.01 | ||
| Wind Speed | Fleeing | 0.015 | 1.01 | 1.01 | 1.10 | ||
| Flight | <0.001 | 0.90 | 0.84 | 0.97 | |||
| Mandarin Duck N° | Natural Foraging | 0.016 | 1.11 | 1.02 | 1.21 | ||
| Vigilance | 0.028 | 1.098 | 1.010 | 1.193 | |||
| Interspecific Interaction | <0.001 | 1.292 | 1.170 | 1.427 | |||
| Other Waterfowl N° | Natural Foraging | <0.001 | 0.93 | 0.90 | 0.97 | ||
| 95% Confidence Interval for Difference | |||||||
|---|---|---|---|---|---|---|---|
| Behaviour | Sex | Social Period | Median ± IQR | p Value | RR | Lower | Upper |
| Natural Foraging | Female | Pre-Laying | 30% ± 55 | <0.001 | 2.54 | 1.65 | 3.91 |
| Post-Incubation | 0% ± 15 | 1 | |||||
| Female | Laying | 50% ± 60 | 0.034 | 1.76 | 1.21 | 2.56 | |
| Pre-Laying | 30% ± 55 | 1 | |||||
| Female | Laying | 50% ± 60 | <0.001 | 4.49 | 2.62 | 7.68 | |
| Post-Incubation | 0% ± 15 | 1 | |||||
| Male | Pre-Laying | 5% ± 15 | 0.019 | 0.66 | .0.45 | 0.95 | |
| Post-Incubation | 0% ± 23 | 1 | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Munday, C.; Rose, P. Environmental and Social Influences on the Behaviour of Free-Living Mandarin Ducks in Richmond Park. Animals 2022, 12, 2554. https://doi.org/10.3390/ani12192554
Munday C, Rose P. Environmental and Social Influences on the Behaviour of Free-Living Mandarin Ducks in Richmond Park. Animals. 2022; 12(19):2554. https://doi.org/10.3390/ani12192554
Chicago/Turabian StyleMunday, Camille, and Paul Rose. 2022. "Environmental and Social Influences on the Behaviour of Free-Living Mandarin Ducks in Richmond Park" Animals 12, no. 19: 2554. https://doi.org/10.3390/ani12192554
APA StyleMunday, C., & Rose, P. (2022). Environmental and Social Influences on the Behaviour of Free-Living Mandarin Ducks in Richmond Park. Animals, 12(19), 2554. https://doi.org/10.3390/ani12192554






