A High Prevalence of Cardiopulmonary Worms Detected in the Iberian Wolf (Canis lupus): A Threat for Wild and Domestic Canids
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
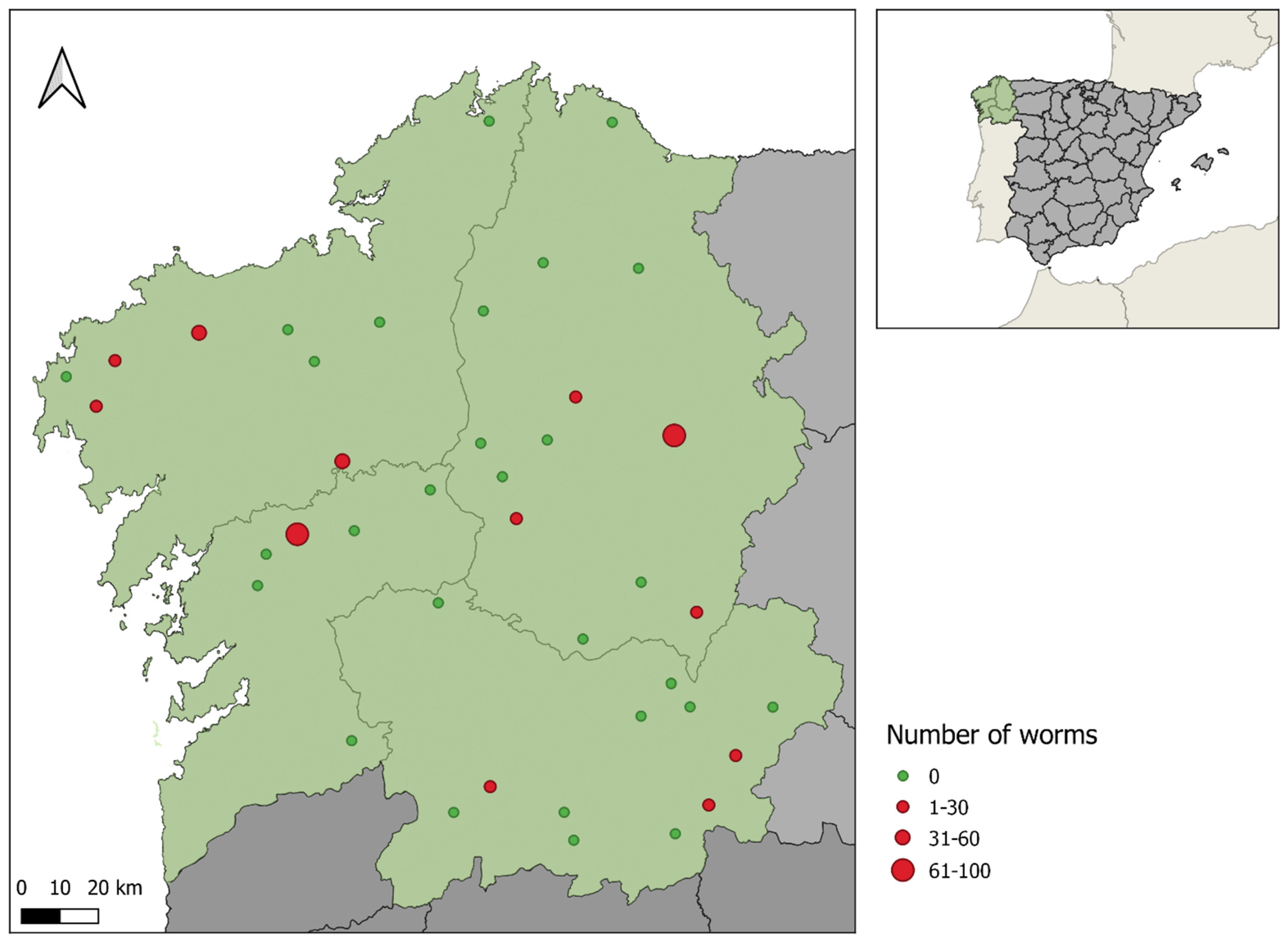
2.1. Study Area
2.2. Sample and Data Collection
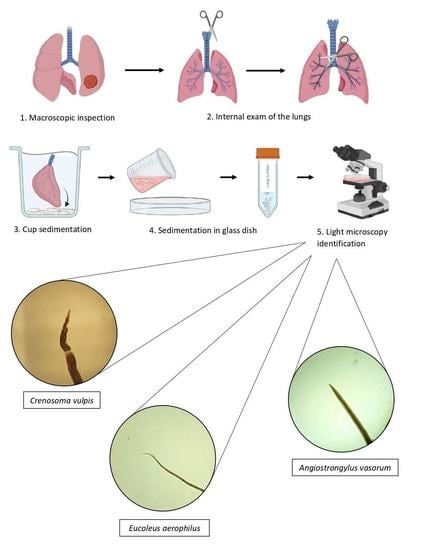
2.3. Cardiorespiratory Parasites
2.4. Statistical Analysis
3. Results
3.1. Nematode Parasite Species and Prevalence
3.2. Epidemiological Data Recorded in Wolves with Nematode Worms
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Arija, C.M. Biología y Conservación del Lobo Ibérico: Crónica de un conflicto. REDVET. Rev. Electrónica de Vet. 2010, 11, 1–18. [Google Scholar]
- Blanco, J.C.; Saénz de Buruaga, M.; Llaneza, L. Canis Lupus Linnaeus, 1758. In Atlas y Libro Rojo de mamíferos terrestres de España; Palomo, L.J., Gisbert, J., Blanco, J.C., Eds.; Dirección General para la Biodiversidad -SECEM-SECEMU: Madrid, Spain, 2007; pp. 272–276. [Google Scholar]
- Blanco, J.C. La Gestión Del Lobo En España. Controversias Científicas En Torno a Su Caza. arbor 2017, 193, a418. [Google Scholar] [CrossRef]
- Tanner, E.; White, A.; Acevedo, P.; Balseiro, A.; Marcos, J.; Gortázar, C. Wolves Contribute to Disease Control in a Multi-Host System. Sci. Rep. 2019, 9, 7940. [Google Scholar] [CrossRef] [PubMed]
- Blanco, J.C.; Cortés, Y. Ecología, Censos, Percepción y Evolución Del Lobo En España: Análisis de Un Conflicto; SECEM: Málaga, Spain, 2002; ISBN 84-607-3896-5. [Google Scholar]
- Figueiredo, A.; Oliveira, L.; de Carvalho, L.M.; Fonseca, C.; Torres, R.T. Parasite Species of the Endangered Iberian Wolf (Canis lupus signatus) and a Sympatric Widespread Carnivore. Int. J. Parasitol. Parasites Wildl. 2016, 5, 164–167. [Google Scholar] [CrossRef]
- Viejo, J.L.; Montesinos, V. La Fauna de La Sierra de Guadarrama. Somera Aproximación a La Riqueza Faunística de Un Nuevo Parque Nacional. Ambienta 2019, 103, 2–25. [Google Scholar]
- Martínez-Rondán, F.J.; de Ybáñez, M.R.R.; López-Beceiro, A.M.; Fidalgo, L.E.; Berriatua, E.; Lahat, L.; Sacristán, I.; Oleaga, Á.; Martínez-Carrasco, C. Cardiopulmonary Nematode Infections in Wild Canids: Does the Key Lie on Host-Prey-Parasite Evolution? Res. Vet. Sci. 2019, 126, 51–58. [Google Scholar] [CrossRef]
- Traversa, D.; Di Cesare, A.; Conboy, G. Canine and Feline Cardiopulmonary Parasitic Nematodes in Europe: Emerging and Underestimated. Parasites Vectors 2010, 3, 62. [Google Scholar] [CrossRef]
- 10 Segovia, J.-M.; Torres, J.; Miquel, J.; Sospedra, E.; Guerrero, R.; Feliu, C. Analysis of Helminth Communities of the Pine Marten, Martes martes, in Spain: Mainland and Insular Data. Acta Parasitol. 2007, 52, 156–164. [Google Scholar] [CrossRef]
- Balmori, A.; Rico, M.; Naves, J.; Llamazares, E. Contribución al Estudio de Los Endoparásitos Del Lobo En La Península Ibérica: Una Investigación Coprológica. Galemys 2000, 12, 13–26. [Google Scholar]
- Segovia, J.; Torres, J.; Miquel, J.; Llaneza, L.; Feliu, C. Helminths in the Wolf, Canis lupus, from North-Western Spain. J. Helminthol. 2001, 75, 183–192. [Google Scholar]
- Ferdushy, T.; Hasan, M.T. Angiostrongylus vasorum: The ‘French Heartworm. Parasitol. Res. 2010, 107, 765–771. [Google Scholar] [CrossRef] [PubMed]
- Tolnai, Z.; Széll, Z.; Sréter, T. Environmental Determinants of the Spatial Distribution of Angiostrongylus vasorum, Crenosoma vulpis and Eucoleus aerophilus in Hungary. Vet. Parasitol. 2015, 207, 355–358. [Google Scholar] [CrossRef] [PubMed]
- Traversa, D.; Di Cesare, A.; Lia, R.; Castagna, G.; Meloni, S.; Heine, J.; Strube, K.; Milillo, P.; Otranto, D.; Meckes, O.; et al. New Insights into Morphological and Biological Features of Capillaria aerophila (Trichocephalida, Trichuridae). Parasitol. Res. 2011, 109 (Suppl. 1), S97–S104. [Google Scholar] [CrossRef]
- Bowman, D. Georgis’ Parasitology for Veterinarians, 9th ed.; Elsevier España: St. Louis, MI, USA, 2011; ISBN 84-8086-705-1. [Google Scholar]
- Miró, G.; Montoya, A.; Roura, X.; Gálvez, R.; Sainz, A. Seropositivity Rates for Agents of Canine Vector-Borne Diseases in Spain: A Multicentre Study. Parasites Vectors 2013, 6, 117. [Google Scholar] [CrossRef]
- Caro-Vadillo, A.; Martínez-Merlo, E.; García-Real, I.; Fermín-Rodríguez, M.L.; Mateo, P. Verminous Pneumonia Due to Filaroides hirthi in a Scottish Terrier in Spain. Vet. Rec. 2005, 157, 586–589. [Google Scholar] [CrossRef] [PubMed]
- Luengo, M.; Arata, N. Filaroides osleri as a Finding in Canine Necropsy. Bol. Chil. Parasitol. 1970, 25, 87–88. [Google Scholar]
- Carrasco, L.; Herva’s, J. Massive Filaroides hirthi Infestation Associated with Caninedistemper. Vet. Rec. 1997, 140, 72–73. [Google Scholar] [CrossRef]
- Georgi, J.R.; Georgi, M.E.; Fahnestock, G.R.; Theodorides, V.J. Transmission and Control of Filaroides hirthi Lungworm Infection in Dogs. Am. J. Vet. Res. 1979, 40, 829–831. [Google Scholar]
- Carretón, E.; Morchón, R.; Falcón-Cordón, Y.; Matos, J.; Costa-Rodríguez, N.; Montoya-Alonso, J.A. First Epidemiological Survey of Angiostrongylus vasorum in Domestic Dogs from Spain. Parasites Vectors 2020, 13, 306. [Google Scholar] [CrossRef]
- Morchón, R.; Montoya-Alonso, J.A.; Sánchez-Agudo, J.Á.; de Vicente-Bengochea, J.; Murcia-Martínez, X.; Carretón, E. Angiostrongylus vasorum in Domestic Dogs in Castilla y León, Iberian Peninsula, Spain. Animals 2021, 11, 1513. [Google Scholar] [CrossRef]
- Mathews, F. Zoonoses in Wildlife: Integrating Ecology into Management. Adv. Parasitol. 2009, 68, 185–209. [Google Scholar] [PubMed]
- Agencia Estatal de Meteorología (AEMET) Atlas Climático. Available online: http://agroclimap.aemet.es/# (accessed on 21 July 2020).
- Schug, K.; Krämer, F.; Schaper, R.; Hirzmann, J.; Failing, K.; Hermosilla, C.; Taubert, A. Prevalence Survey on Lungworm (Angiostrongylus vasorum, Crenosoma vulpis, Eucoleus aerophilus) Infections of Wild Red Foxes (Vulpes vulpes) in Central Germany. Parasites Vectors 2018, 11, 85. [Google Scholar] [CrossRef]
- Schnyder, M.; Fahrion, A.; Ossent, P.; Kohler, L.; Webster, P.; Heine, J.; Deplazes, P. Larvicidal Effect of Imidacloprid/Moxidectin Spot-on Solution in Dogs Experimentally Inoculated with Angiostrongylus vasorum. Vet. Parasitol. 2009, 166, 326–332. [Google Scholar] [CrossRef] [PubMed]
- Conboy, G. Helminth Parasites of the Canine and Feline Respiratory Tract. Vet. Clin. N. Am. Small Anim. Pract. 2009, 39, 1109–1126. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.C.; Chabaud, A.G.; Willmott, S. Keys to the Nematode Parasites of Vertebrates: Archival Volume; CABI: Wallingford, UK, 2009; ISBN 1-84593-572-1. [Google Scholar]
- Taylor, M.A.; Coop, R.; Wall, R.L. Veterinary Parasitology; John Wiley & Sons: Hoboken, NJ, USA, 2015; ISBN 1-119-07369-3. [Google Scholar]
- Torres, J.; Segovia, J.; Miquel, J.; Feliu, C.; Llaneza, L.; Petrucci-Fonseca, F. Helmintofauna Del Lobo Ibérico (Canis lupus signatus Cabrera, 1907). Aspectos Potencialmente Útiles En Mastozoologia. Galemys 2000, 12, 1–11. [Google Scholar]
- Čabanová, V.; Guimaraes, N.; Hurníková, Z.; Chovancová, G.; Urban, P.; Miterpáková, M. Endoparasites of the Grey Wolf (Canis lupus) in Protected Areas of Slovakia. Anna. Parasitol. 2017, 63, 283–289. [Google Scholar]
- Hermosilla, C.; Kleinertz, S.; Silva, L.M.; Hirzmann, J.; Huber, D.; Kusak, J.; Taubert, A. Protozoan and Helminth Parasite Fauna of Free-Living Croatian Wild Wolves (Canis lupus) Analyzed by Scat Collection. Vet. Parasitol. 2017, 233, 14–19. [Google Scholar] [CrossRef]
- De Liberato, C.; Grifoni, G.; Lorenzetti, R.; Meoli, R.; Cocumelli, C.; Mastromattei, A.; Scholl, F.; Rombolà, P.; Calderini, P.; Bruni, G.; et al. Angiostrongylus vasorum in Wolves in Italy: Prevalence and Pathological Findings. Parasites Vectors 2017, 10, 386. [Google Scholar] [CrossRef]
- Bagrade, G.; Kirjusina, M.; Vismanis, K.; Ozolins, J. Helminth Parasites of the Wolf Canis Lupus from Latvia. J. Helminthol. 2009, 83, 63. [Google Scholar] [CrossRef]
- Shimalov, V.; Shimalov, V. Helminth Fauna of the Wolf (Canis lupus Linnaeus, 1758) in Belorussian Polesie. Parasitol. Res. 2000, 86, 163–164. [Google Scholar] [CrossRef]
- Humm, K.; Adamantos, S. Is Evaluation of a Faecal Smear a Useful Technique in the Diagnosis of Canine Pulmonary Angiostrongylosis? J. Small Anim. Pract. 2010, 51, 200–203. [Google Scholar] [CrossRef]
- Schnyder, M.; Tanner, I.; Webster, P.; Barutzki, D.; Deplazes, P. An ELISA for Sensitive and Specific Detection of Circulating Antigen of Angiostrongylus vasorum in Serum Samples of Naturally and Experimentally Infected Dogs. Vet. Parasitol. 2011, 179, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Jefferies, R.; Morgan, E.R.; Helm, J.; Robinson, M.; Shaw, S.E. Improved Detection of Canine Angiostrongylus vasorum Infection Using Real-Time PCR and Indirect ELISA. Parasitol. Res. 2011, 109, 1577–1583. [Google Scholar] [CrossRef] [PubMed]
- Houpin, E.; McCarthy, G.; Ferrand, M.; Waal, T.D.; O’Neill, E.J.; Zintl, A. Comparison of Three Methods for the Detection of Angiostrongylus vasorum in the Final Host. Vet. Parasitol. 2016, 220, 54–58. [Google Scholar] [CrossRef][Green Version]
- Elsheikha, H.M.; Holmes, S.A.; Wright, I.; Morgan, E.R.; Lacher, D.W. Recent Advances in the Epidemiology, Clinical and Diagnostic Features, and Control of Canine Cardio-Pulmonary Angiostrongylosis. Vet. Res. 2014, 45, 92. [Google Scholar] [CrossRef]
- Gortázar, C.; Villafuerte, R.; Lucientes, J.; Fernández-de-Luco, D. Habitat Related Differences in Helminth Parasites of Red Foxes in the Ebro Valley. Vet. Parasitol. 1998, 80, 75–81. [Google Scholar] [CrossRef]
- Segovia, J.; Torres, J.; Miquel, J. Helminth Parasites of the Red Fox [Vulpes vulpes L., 1758] in the Iberian Peninsula: An Ecological Study. Acta Parasitol. 2004, 49, 67–79. [Google Scholar]
- Mañas, S. Cardiopulmonary Helminth Parasites of Red Foxes (Vulpes vulpes) in Catalonia, Northeastern Spain. Vet. J. 2005, 169, 118–120. [Google Scholar] [CrossRef]
- Gerrikagoitia, X.; Barral, M.; Juste, R. Angiostrongylus Species in Wild Carnivores in the Iberian Peninsula. Vet. Parasitol. 2010, 174, 175–180. [Google Scholar] [CrossRef]
- Torres, J.; Miquel, J.; Motjé, M. Helminth Parasites of the Eurasian Badger (Meles meles L.) in Spain: A Biogeographic Approach. Parasitol. Res. 2001, 87, 259–263. [Google Scholar] [CrossRef]
- Gillis-Germitshc, N.; Kapel, C.M.O.; Thamsborg, S.M.; Deplazes, P.; Schnyder, M. Host-Specific Serological Response to Angiostrongylus vasorum Infection in Red Foxes (Vulpes vulpes): Implications for Parasite Epidemiology. Parasitology 2017, 144, 1144–1153. [Google Scholar] [CrossRef] [PubMed]
- Morgan, E.R.; Shaw, S.E.; Brennan, S.F.; Waal, T.D.D.; Jones, B.R.; Mulcahy, G. Angiostrongylus Vasorum: A Real Heartbreaker. Trends Parasitol. 2005, 21, 49–51. [Google Scholar] [CrossRef] [PubMed]
- Morgan, E.R.; Jefferies, R.; Krajewski, M.; Ward, P.; Shaw, S.E. Canine Pulmonary Angiostrongylosis: The Influence of Climate on Parasite Distribution. Parasitol. Int. 2009, 58, 406–410. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Carrasco, C.; Berriatua, E.; Garijo, M.; Martínez, J.; Alonso, F.; Ruiz de Ybáñez, R. Epidemiological Study of Non-systemic Parasitism in Dogs in Southeast Mediterranean Spain Assessed by Coprological and Post-mortem Examination. Zoonoses Public health 2007, 54, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Colella, V.; Mutafchiev, Y.; Cavalera, M.A.; Giannelli, A.; Lia, R.P.; Dantas-Torres, F.; Otranto, D. Development of Crenosoma vulpis in the Common Garden Snail Cornu Aspersum: Implications for Epidemiological Studies. Parasites Vectors 2016, 9, 208. [Google Scholar] [CrossRef]
- Jeffery, R.A.; Lankester, M.W.; McGrath, M.J.; Whitney, H.G. Angiostrongylus vasorum and Crenosoma vulpis in Red Foxes (Vulpes vulpes) in Newfoundland, Canada. Can. J. Zool. 2004, 82, 66–74. [Google Scholar] [CrossRef]
- Miró, G.; Montoya, A.; Jiménez, S.; Frisuelos, C.; Mateo, M.; Fuentes, I. Prevalence of Antibodies to Toxoplasma gondii and Intestinal Parasites in Stray, Farm and Household Cats in Spain. Vet. Parasitol. 2004, 126, 249–255. [Google Scholar] [CrossRef]
- Muñoz, S.; Ramos, P.L.; Carretón, E.; Diosdado, A.; González-Miguel, J.; Simón, F.; Morchón, R. Intestinal helminths in Iberian wolves (Canis lupus signatus) from Northwest Spain. The Open Parasitology J. 2018, 6, 106–111. [Google Scholar] [CrossRef]
- Martínez-Carrasco, C.; Ruiz de Ybanez, M.; Sagarminaga, J.; Garijo, M.; Moreno, F.; Acosta, I.; Hernandez, S.; Alonso, F. Parasites of the Red Fox (Vulpes vulpes Linnaeus, 1758) in Murcia, Southeast Spain. Rev. Med. Vet. 2007, 158, 331–335. [Google Scholar]
- Criado-Fornelio, A.; Gutierrez-Garcia, L.; Rodriguez-Caabeiro, F.; Reus-Garcia, E.; Roldan-Soriano, M.; Diaz-Sanchez, M. A Parasitological Survey of Wild Red Foxes (Vulpes vulpes) from the Province of Guadalajara, Spain. Vet. Parasitol. 2000, 92, 245–251. [Google Scholar] [CrossRef]
- Rutgers, M.; Orgiazzi, A.; Gardi, C.; Römbke, J.; Jänsch, S.; Keith, A.M.; Neilson, R.; Boag, B.; Schmidt, O.; Murchie, A.K. Mapping Earthworm Communities in Europe. Appl. Soil Ecol. 2016, 97, 98–111. [Google Scholar] [CrossRef]
- Neubert, E.; Seddon, E.; Allen, M.; Backeljau, T. European Red List of Terrestrial Molluscs: Snails, Slugs, and Semi-Slugs; IUCN: Brussels, Belgium, 2019. [Google Scholar]
- Jefferies, R.; Shaw, S.; Willesen, J.; Viney, M.; Morgan, E. Elucidating the Spread of the Emerging Canid Nematode Angiostrongylus vasorum between Palaearctic and Nearctic Ecozones. Infect. Genet. Evol. 2010, 10, 561–568. [Google Scholar] [CrossRef] [PubMed]
- Millán, J.; García, E.J.; Oleaga, Á.; López-Bao, J.V.; Llaneza, L.; Palacios, V.; Candela, M.G.; Cevidanes, A.; Rodríguez, A.; León-Vizcaíno, L. Using a Top Predator as a Sentinel for Environmental Contamination with Pathogenic Bacteria: The Iberian Wolf and Leptospires. Memórias do Inst. Oswaldo Cruz 2014, 109, 1041–1044. [Google Scholar] [CrossRef] [PubMed]
| Species Identified | No. Positive Wolves (%) | No. Total Worms | Female Worms | Male Worms | Mean (SD) | No. Worms per Wolf (min.–max.) |
|---|---|---|---|---|---|---|
| A. vasorum | 11 (19.2) | 374 * | 265 | 71 | 34 (35.1) | 1–99 |
| C. vulpis | 4 (7) | 8 | 7 | 1 | 2 (0.8) | 1–3 |
| E. aerophilus | 2 (3.5) | 3 | 3 | 0 | 1.5 (0.7) | 1–2 |
| A. vasorum single infection | 8 (14) | 348 * | 245 | 65 | 43.5 (36.7) | 7–99 |
| C. vulpis single infection | 3 (5.3) | 6 | 5 | 1 | 2 (1.0) | 1–3 |
| A. vasorum + C. vulpis | 1 (1.7) | 18 | 13 | 5 | 18 (0.0) | 18 |
| A. vasorum + E. aerophilus | 2 (3.5) | 13 | 12 | 1 | 6.5 (6.4) | 2–11 |
| Category | Total No. Wolves | Angiostrongylus vasorum | Crenosoma vulpis | Eucoleus aerophilus | Total No. of Worms | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| No. Positive Wolves | % | p-Value | No. Positive Wolves | % | p-Value | No. Positive Wolves | % | p-Value | No. Positive Wolves | % | p-Value | |||
| Province | A Coruna | 15 | 3 | 20 | 0.959 | 2 | 13.3 | 0.265 | 0 | 0.0 | 1 | 4 | 26.7 | 1 |
| Lugo | 21 | 5 | 23.8 | 0 | 0.0 | 1 | 4.8 | 5 | 23.8 | |||||
| Ourense | 15 | 2 | 13.3 | 2 | 13.3 | 1 | 6.7 | 4 | 26.7 | |||||
| Pontevedra | 6 | 1 | 16.7 | 0 | 0.0 | 0 | 0 | 1 | 16.7 | |||||
| Sex | Male | 29 | 5 | 17.2 | 0.747 | 2 | 6.9 | 1 | 0 | 0.0 | 0.236 | 7 | 24.1 | 1 |
| Female | 28 | 6 | 21.4 | 2 | 7.1 | 2 | 7.1 | 7 | 25.0 | |||||
| Body condition (1–5) | 1 | 2 | 0 | 0.0 | 0.221 | 0 | 0.0 | 0.444 | 0 | 0.0 | 1 | 0 | 0.0 | 0.174 |
| 2 | 10 | 4 | 40.0 | 2 | 20.0 | 0 | 0.0 | 5 | 50.0 | |||||
| 3 | 20 | 5 | 25.0 | 1 | 5.0 | 1 | 5.0 | 6 | 30.0 | |||||
| 4 | 23 | 2 | 8.7 | 1 | 4.3 | 1 | 4.3 | 3 | 13.0 | |||||
| 5 | 1 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | 0 | 0.0 | |||||
| Age | Pup | 14 | 3 | 21.4 | 1 | 4 | 28.6 | 0.002 | 1 | 7.1 | 0.714 | 6 | 42.9 | 0.243 |
| Young | 24 | 5 | 20.8 | 0 | 0.0 | 1 | 4.1 | 5 | 28.8 | |||||
| Adult | 19 | 3 | 15.8 | 0 | 0.0 | 0 | 0.0 | 3 | 15.8 | |||||
| Wolf Pup (n = 14) | Young (n = 24) | Adults (n = 19) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| No. of Parasites | No. | % | p-Value | No. | % | p-Value | No. | % | p-Value |
| 0 | 8 | 57.1 | 0.083 | 19 | 79.2 | 0.757 | 16 | 84.2 | 0.343 |
| 1–29 | 6 | 42.9 | 0.004 | 2 | 8.3 | 0.277 | 1 | 5.26 | 0.246 |
| 30-60 | 0 | 0 | 0.567 | 2 | 8.3 | 0.566 | 1 | 5.26 | 1.000 |
| >60 | 0 | 0 | 1.000 | 1 | 4.2 | 1.000 | 1 | 5.26 | 1.000 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Estévez-Sánchez, E.; Checa, R.; Montoya, A.; Barrera, J.P.; López-Beceiro, A.M.; Fidalgo, L.E.; Miró, G. A High Prevalence of Cardiopulmonary Worms Detected in the Iberian Wolf (Canis lupus): A Threat for Wild and Domestic Canids. Animals 2022, 12, 2289. https://doi.org/10.3390/ani12172289
Estévez-Sánchez E, Checa R, Montoya A, Barrera JP, López-Beceiro AM, Fidalgo LE, Miró G. A High Prevalence of Cardiopulmonary Worms Detected in the Iberian Wolf (Canis lupus): A Threat for Wild and Domestic Canids. Animals. 2022; 12(17):2289. https://doi.org/10.3390/ani12172289
Chicago/Turabian StyleEstévez-Sánchez, Efrén, Rocío Checa, Ana Montoya, Juan Pedro Barrera, Ana María López-Beceiro, Luis Eusebio Fidalgo, and Guadalupe Miró. 2022. "A High Prevalence of Cardiopulmonary Worms Detected in the Iberian Wolf (Canis lupus): A Threat for Wild and Domestic Canids" Animals 12, no. 17: 2289. https://doi.org/10.3390/ani12172289
APA StyleEstévez-Sánchez, E., Checa, R., Montoya, A., Barrera, J. P., López-Beceiro, A. M., Fidalgo, L. E., & Miró, G. (2022). A High Prevalence of Cardiopulmonary Worms Detected in the Iberian Wolf (Canis lupus): A Threat for Wild and Domestic Canids. Animals, 12(17), 2289. https://doi.org/10.3390/ani12172289