Moringa oleifera Leaves’ Extract Enhances Nonspecific Immune Responses, Resistance against Vibrio alginolyticus, and Growth in Whiteleg Shrimp (Penaeus vannamei)
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Moringa Preparation
2.2. Whiteleg Shrimp
2.3. In Vitro Study of Viability and Immune Response
2.4. Diet Preparation
2.5. In Vivo Study of the Immune Response
2.6. Immune-Related Gene Expression
2.7. Growth Performance
2.8. Challenge with V. alginolyticus
2.9. Statistical Analysis
3. Results
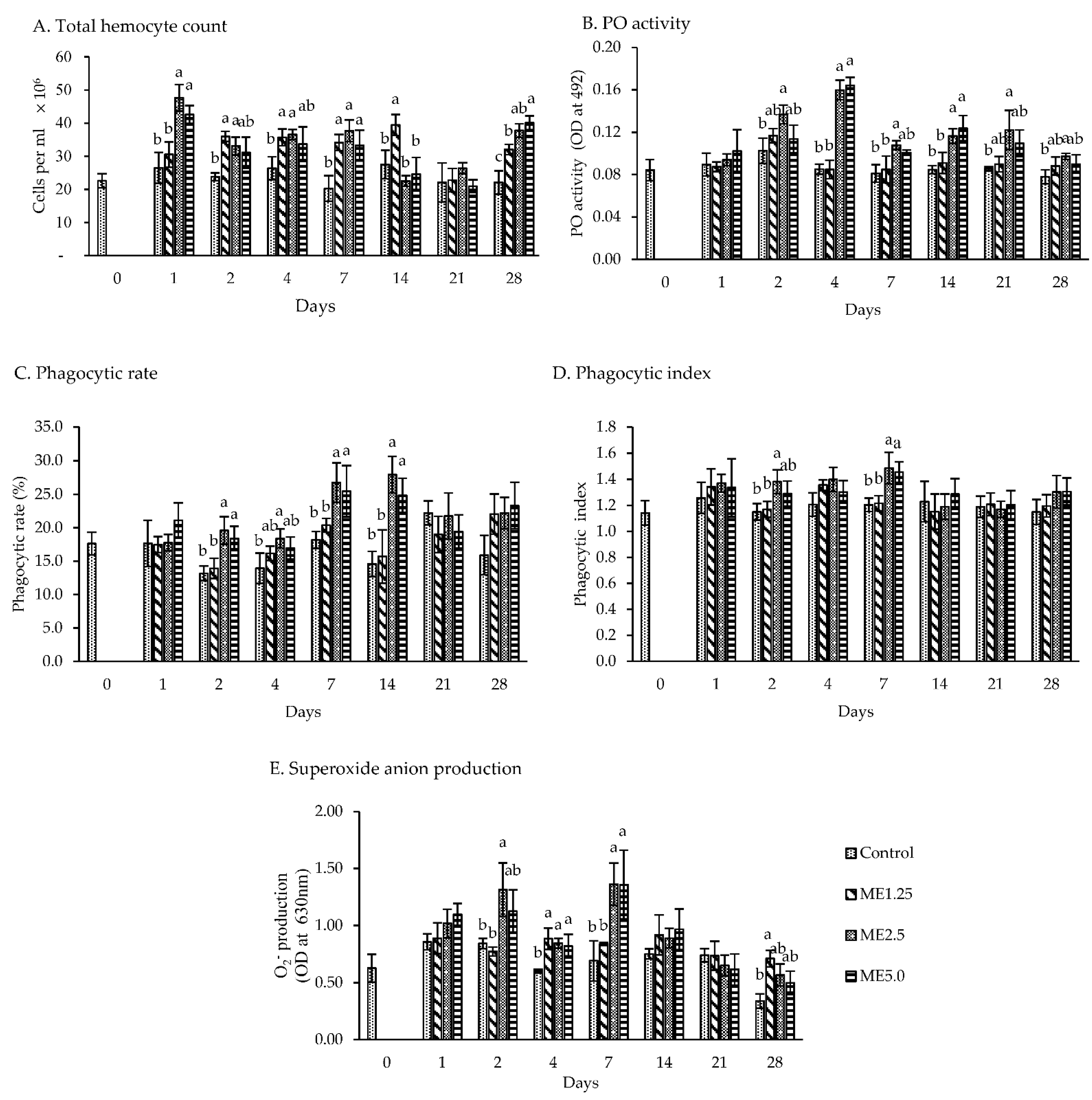
3.1. In Vitro Study of Viability and Immune Response
3.2. In Vivo Study of Immune Responses
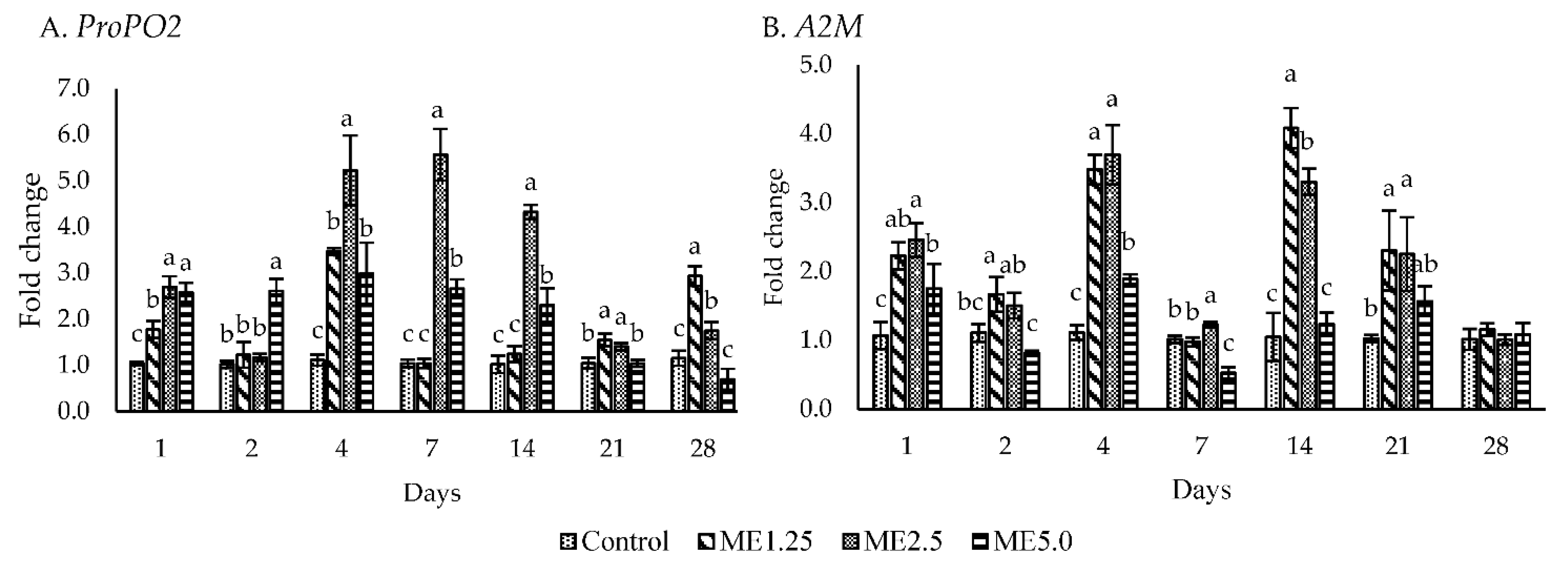
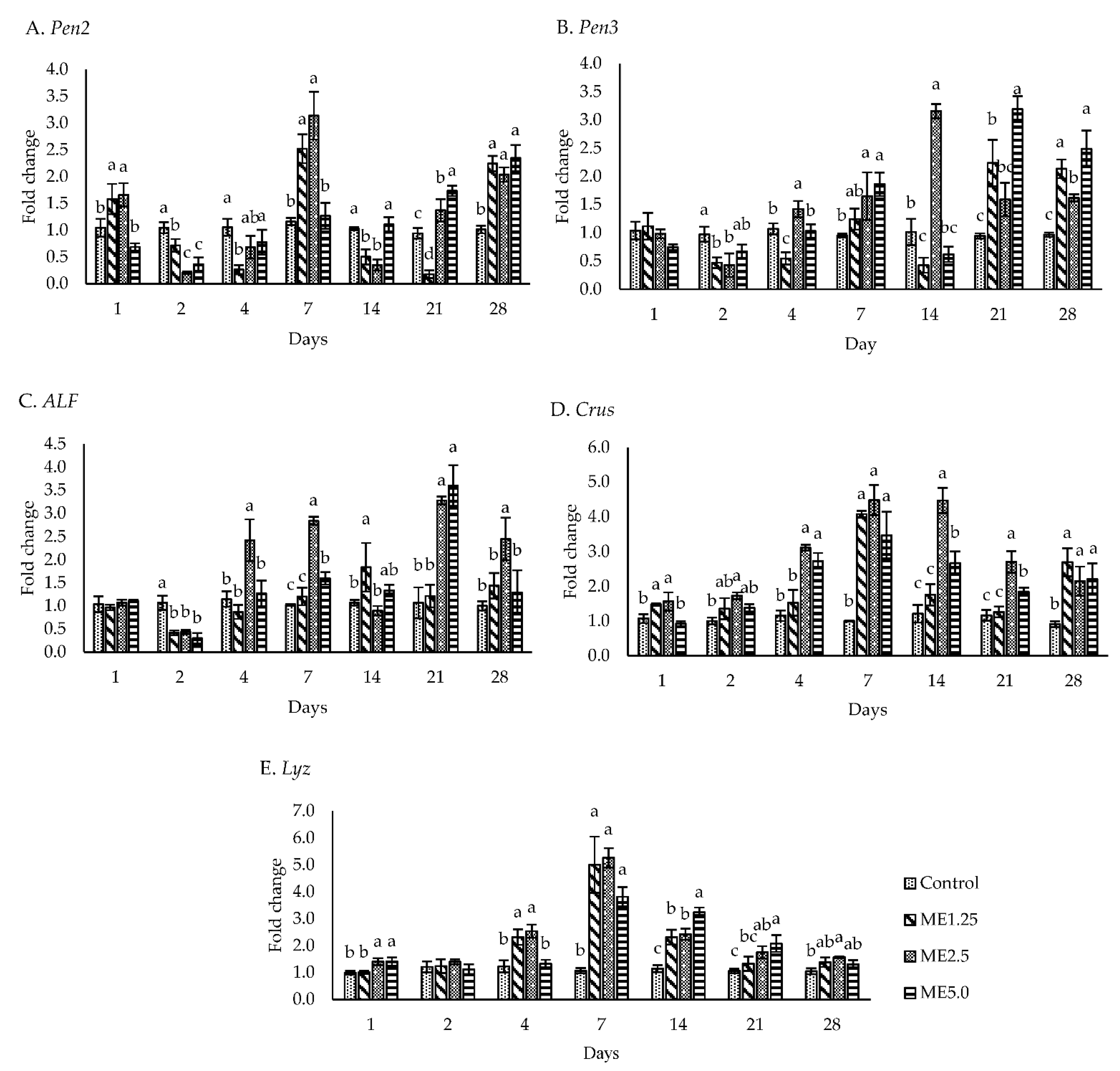
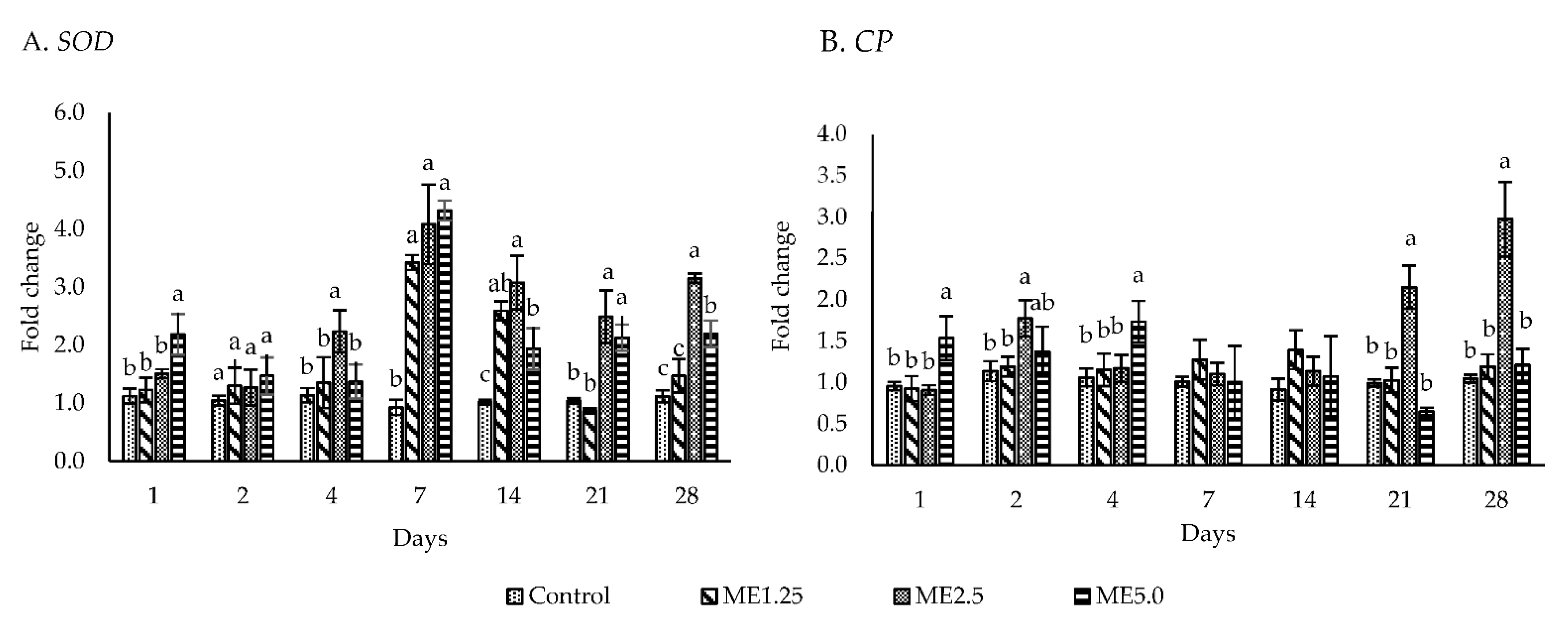
3.3. Immune-Related Gene Expression
3.4. Growth Performance
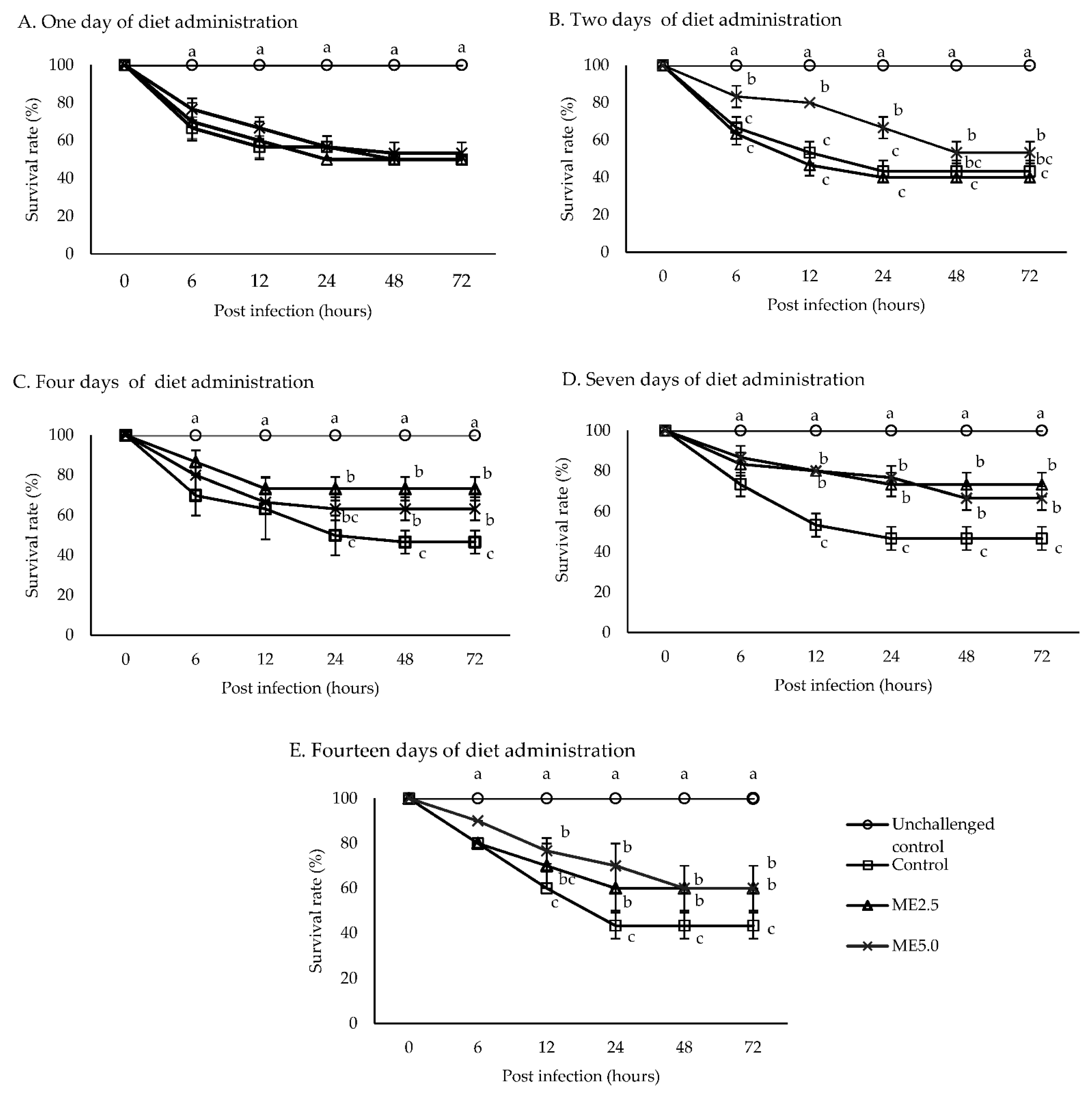
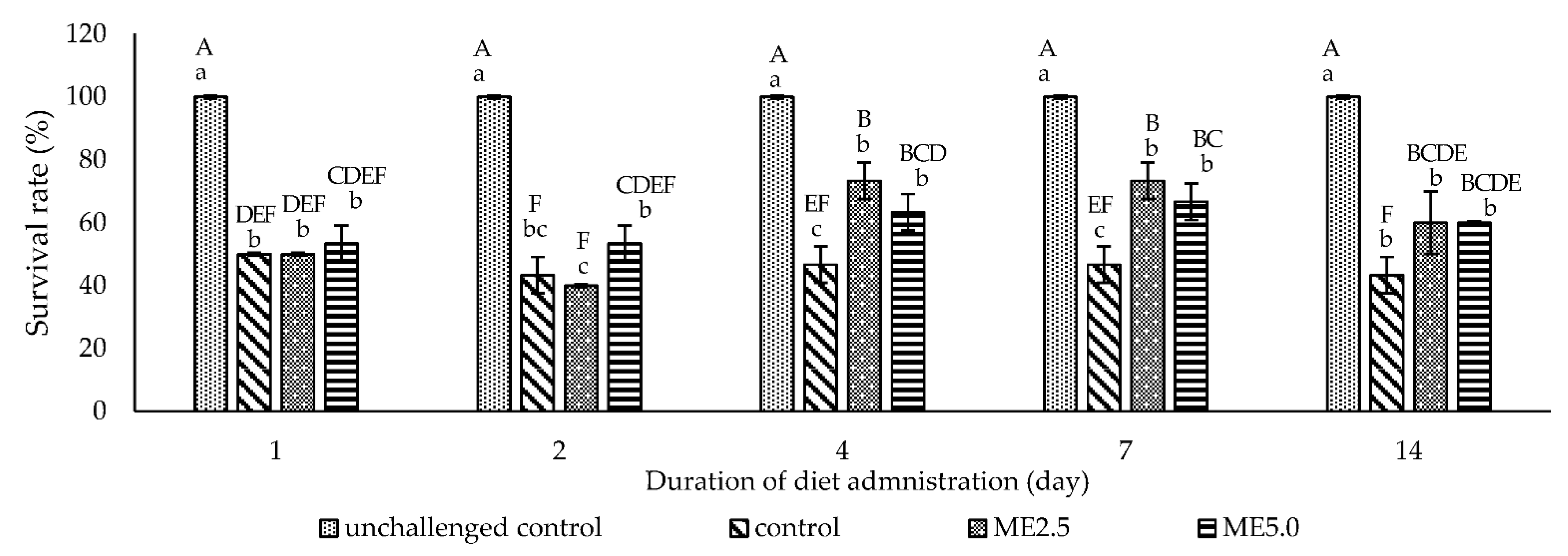
3.5. Challenge Test
4. Discussion
4.1. In Vitro Study of Viability and Immune Response
4.2. In Vivo Study of Immune Responses
4.3. Growth Performance
4.4. Challenge Test
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nath, S.; Haldar, C. Effects of Stress among Shrimp Post-Larvae stocked at High Stocking Density in Nursery Culture System: A Review. Int. J. Curr. Microbiol. App. Sci. 2020, 9, 2987–2996. [Google Scholar] [CrossRef]
- Kathyayani, S.A.; Poornima, M.; Sukumaran, S.; Nagavel, A.; Muralidhar, M. Effect of ammonia stress on immune variables of Pacific white shrimp Penaeus vannamei under varying levels of pH and susceptibility to white spot syndrome virus. Ecotoxicol. Environ. Saf. 2019, 184, 109626. [Google Scholar] [CrossRef]
- Gao, Y.; He, Z.; Zhao, B.; Li, Z.; He, J.; Lee, J.Y.; Chu, Z. Effect of stocking density on growth, oxidative stress and HSP 70 of pacific white shrimp Litopenaeus vannamei. Turk. J. Fish. Aquat. Sci. 2017, 17, 877–884. [Google Scholar] [CrossRef]
- Liu, C.H.; Chen, J.C. Effect of ammonia on the immune response of white shrimp Litopenaeus vannamei and its susceptibility to Vibrio alginolyticus. Fish Shellfish Immunol. 2004, 16, 321–334. [Google Scholar] [CrossRef]
- Wang, L.U.; Chen, J.C. The immune response of white shrimp Litopenaeus vannamei and its susceptibility to Vibrio alginolyticus at different salinity levels. Fish Shellfish Immunol. 2005, 18, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Cheng, W.; Wang, L.U.; Chen, J.C. Effect of water temperature on the immune response of white shrimp Litopenaeus vannamei to Vibrio alginolyticus. Aquaculture 2005, 250, 592–601. [Google Scholar] [CrossRef]
- Elumalai, P.; Kurian, A.; Lakshmi, S.; Faggio, C.; Esteban, M.A.; Ringø, E. Herbal Immunomodulators in Aquaculture. Rev. Fish. Sci. Aquac. 2021, 29, 33–57. [Google Scholar] [CrossRef]
- Angela, C.; Wang, W.; Lyu, H.; Zhou, Y.; Huang, X. The effect of dietary supplementation of Astragalus membranaceus and Bupleurum chinense on the growth performance, immune-related enzyme activities and genes expression in white shrimp, Litopenaeus vannamei. Fish Shellfish Immunol. 2020, 107, 379–384. [Google Scholar] [CrossRef]
- Mahfuz, S.; Piao, X.S. Application of Moringa (Moringa oleifera) as Natural Feed Supplement in Poultry Diets. Animals 2019, 9, 431. [Google Scholar] [CrossRef] [Green Version]
- Bolaños, X.C.; Vega, R.C.; Camacho, R.R.; Gómez, M.R.; Piña, G.F.L.; Maldonado, S.G. Moringa infusion (Moringa oleifera) rich in phenolic compounds and high antioxidant capacity attenuate nitric oxide pro-inflammatory mediator in vitro. Ind. Crop. Prod. 2018, 118, 95–101. [Google Scholar] [CrossRef]
- Ma, Z.F.; Ahmad, J.; Zhang, H.; Khan, I.; Muhammad, S. Evaluation of phytochemical and medicinal properties of Moringa (Moringa oleifera) as a potential functional food. S. Afr. J. Bot. 2020, 129, 40–46. [Google Scholar] [CrossRef]
- Mwamatope, B.; Tembo, D.; Chikowe, I.; Kampira, E.; Nyirenda, C. Total phenolic contents and antioxidant activity of Senna singueana, Melia azedarach, Moringa oleifera and Lannea discolor herbal plants. Sci. Afr. 2020, 9, e00481. [Google Scholar] [CrossRef]
- Kaleo, I.V.; Gao, Q.; Liu, B.; Sun, C.; Zhou, Q.; Zhang, H.; Shan, F.; Xiong, Z.; Bo, L.; Song, C. Effects of Moringa oleifera leaf extract on growth performance, physiological and immune response, and related immune gene expression of Macrobrachium rosenbergii with Vibrio anguillarum and ammonia stress. Fish Shellfish Immunol. 2019, 89, 603–613. [Google Scholar] [CrossRef]
- Akbary, P.; Gholamhosseini, A.; Ali, M.; Jahanbakhshi, A.; Tavabe, K.R.; Kuchaksaraei, B.S.; Mirghaed, A.T. Dietary administration of Moringa oleifera extract enhances growth, fatty acid composition, antioxidant activity and resistance of shrimp Litopenaeus vannamei against Photobacterium damselae. J. Agric. Nat. Resour. 2021, 55, 161–170. [Google Scholar]
- Gawad, E.E.A.A.E.; El Asely, A.M.; Soror, E.I.; Abbass, A.A.; Austin, B. Effect of dietary Moringa oleifera leaf on the immune response and control of Aeromonas hydrophila infection in Nile tilapia (Oreochromis niloticus) fry. Aquac. Int. 2020, 28, 389–402. [Google Scholar] [CrossRef]
- Labh, S.N. Expression of Immune Genes and Stress Enzyme Profiles of Rainbow Trout (Oncorhynchus mykiss) Fed Moringa Oleifera Leaf Meal (MLM). Int. J. Biol. Innov. 2020, 2, 155–164. [Google Scholar] [CrossRef]
- Faheem, M.; Khaliq, S.; Mustafa, N.; Rani, S.; Lone, K.P. Dietary Moringa oleferia leaf meal induce growth, innate immunity and cytokine expression in grass carp, Ctenopharyngodon idella. Aquac. Nutr. 2020, 26, 1164–1172. [Google Scholar] [CrossRef]
- Hamed, H.S.; Sayed, Y.S.E. Antioxidant activities of Moringa oleifera leaf extract against pendimethalin-induced oxidative stress and genotoxicity in Nile tilapia, Oreochromis niloticus (L.). Fish Physiol. Biochem. 2019, 45, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Arisa, I.I.; Agustina, S.; Nurfadillah, N.; Karina, S. In vivo test of Litopenaeus vannamei infected by Vibrio using Moringa oleifera leaf extract. Depik 2021, 10, 86–90. [Google Scholar] [CrossRef]
- Khalil, F.; KornI, F.M. Evaluation of Moringa oleifera leaves and their aqueous extract in improving growth, immunity and mitigating effect of stress on common carp (Cyprinus carpio) fingerlings. Aquat. Sci. Eng. 2017, 32, 170–177. [Google Scholar] [CrossRef]
- Ambi, A.A.; Abdurahman, E.; Katsayal, U.; Sule, M.; Pateh, U.; Ibrahim, N. Toxicity evaluation of Moringa oleifera leaves. Int. J. Pharmaceut. Res. Innovat. 2011, 4, 22–24. [Google Scholar]
- Asare, G.A.; Gyan, B.; Bugyei, K.; Adjei, S.; Mahama, R.; Addo, P.; Otu-Nyarko, L.; Wiredu, E.K.; Nyarko, A. Toxicity potentials of the nutraceutical Moringa oleifera at supra-supplementation levels. J. Ethnopharmacol. 2012, 139, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Asomugha, A.; Ezejindu, D.; Asomugha, R.; Anyabolu, A.; Ojukwu, P. Evaluation of toxicity effect of graded doses of Moringa oleifera leaf extract on blood indices using 20 adult Wistar rats. Int. J. Biom. Adv. Res. 2015, 6, 98–102. [Google Scholar]
- Guzman, G.A.; Martinez, J.G.S.; Cordova, A.I.C.; Gonzalez, A.L.; Ascencio, F. Penaeid shrimp immune system. Thai J. Vet. Med. 2009, 39, 205–215. [Google Scholar]
- Aftabuddin, S.; Siddique, M.A.M.; Romkey, S.S.; Shelton, W.L. Antibacterial function of herbal extracts on growth, survival and immunoprotection in the black tiger shrimp Penaeus monodon. Fish Shellfish Immunol. 2017, 65, 52–58. [Google Scholar] [CrossRef]
- Awad, E.; Awaad, A. Role of medicinal plants on growth performance and immune status in fish. Fish Shellfish Immunol. 2017, 67, 40–54. [Google Scholar] [CrossRef]
- Vico, G.D.; Guida, V.; Carella, F. Urtica dioica (Stinging Nettle): A Neglected Plant with Emerging Growth Promoter/Immunostimulant Properties for Farmed Fish. Front. Physiol. 2018, 9, 285. [Google Scholar] [CrossRef] [PubMed]
- Ngo, H.V.T.; Huang, H.T.; Lee, P.T.; Liao, Z.H.; Chen, H.Y.; Nan, F.H. Effects of Phyllanthus amarus extract on nonspecific immune responses, growth, and resistance to Vibrio alginolyticus in white shrimp Litopenaeus vannamei. Fish Shellfish Immunol. 2020, 107, 1–8. [Google Scholar] [CrossRef]
- Lee, P.T.; Tran, H.T.Q.; Huang, H.T.; Nan, F.H.; Lee, M.C. Sargassum horneri extracts stimulate innate immunity, enhance growth performance, and upregulate immune genes in the white shrimp Litopenaeus vannamei. Fish Shellfish Immunol. 2020, 102, 276–285. [Google Scholar] [CrossRef]
- Borbor, C.D.; Alvarado, B.C.; Rodríguez, J.A. A simple in vitro method to evaluate the toxicity of functional additives used in shrimp aquaculture. MethodsX 2018, 5, 90–95. [Google Scholar] [CrossRef]
- Declarador, R.S.; Serrano, A.E., Jr.; Corre, V.L., Jr. Ulvan extract acts as immunostimulant against white spot syndrome virus (WSSV) in juvenile black tiger shrimp Penaeus monodon. Aquac. Aquar. Conserv. Legis. 2014, 7, 153–161. Available online: http://www.bioflux.com.ro/docs/2014.153-161.pdf. (accessed on 10 August 2021).
- Sung, H.H.; Kuo, P.A.; Kao, W.Y. Effect of lipopolysaccharide on in vitro phagocytosis by hemocytes from giant freshwater prawn (Macrobrachium rosenbergii). Fish Pathol. 2000, 35, 109–116. [Google Scholar] [CrossRef]
- Córdova, A.C.; Saavedra, N.H.; De Philippis, R.; Ascencio, F. Generation of superoxide anion and SOD activity in haemocytes and muscle of American white shrimp (Litopenaeus vannamei) as a response to β-glucan and sulphated polysaccharide. Fish Shellfish Immunol. 2002, 12, 353–366. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Helrich, K. Official Methods of Analysis of the Association of Official Analytical Chemists; 0935584420; Association of Official Analytical Chemists: Rockville, MD, USA, 1990; p. 1298. [Google Scholar]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. MethodsX 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Chen, Y.Y.; Chen, J.C.; Lin, Y.C.; Putra, D.F.; Kitikiew, S.; Li, C.C.; Hsieh, J.F.; Liou, C.H.; Yeh, S.T. Shrimp that have received carrageenan via immersion and diet exhibit immunocompetence in phagocytosis despite a post-plateau in immune parameters. Fish Shellfish Immunol. 2014, 36, 352–366. [Google Scholar] [CrossRef]
- Wang, K.H.C.; Tseng, C.W.; Lin, H.Y.; Chen, I.T.; Chen, Y.H.; Chen, Y.M.; Chen, T.Y.; Yang, H.L. RNAi knock-down of the Litopenaeus vannamei Toll gene (LvToll) significantly increases mortality and reduces bacterial clearance after challenge with Vibrio harveyi. Dev. Comp. Immunol. 2010, 34, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Alcalá, J.S.; Ruiz, P.Á.; Naranjo, J.A.; Leal, H.E.; Villaseñor, I.L.; Godínez, J.E.; González, A.L.; Jiménez, C.G.; Plata, G.D. Transcriptional response of immune-related genes in Litopenaeus vannamei post-larvae cultured in recirculating aquaculture systems with and without biofloc. Aquac. Int. 2019, 27, 209–225. [Google Scholar] [CrossRef]
- Dewi, N.R.; Huang, H.-T.; Wu, Y.-S.; Liao, Z.-H.; Lin, Y.-J.; Lee, P.-T.; Nan, F.-H. Guava (Psidium guajava) leaf extract enhances immunity, growth, and resistance against Vibrio parahaemolyticus in white shrimp Penaeus vannamei. Fish Shellfish Immunol. 2021, 118, 1–10. [Google Scholar] [CrossRef]
- Huang, H.-T.; Liao, Z.-H.; Wu, Y.-S.; Lin, Y.-J.; Kang, Y.-S.; Nan, F.-H. Effects of Bidens alba and Plectranthus amboinicus dietary supplements on nonspecific immune responses, growth, and resistance to Vibrio alginolyticus in white leg shrimp (Penaeus vannamei). Aquaculture 2022, 546, 737306. [Google Scholar] [CrossRef]
- Helmiati, S.; Rustadi; Isnansetyo, A.; Zuprizal. The replacement of fish meal with fermented Moringa leaves meal and its effect on the immune response of red tilapia (Oreochromis sp.). IOP Conf. Ser. Earth Environ. Sci. 2021, 919, 012057. [Google Scholar] [CrossRef]
- García-Beltrán, J.M.; Mansour, A.T.; Alsaqufi, A.S.; Ali, H.M.; Esteban, M.Á. Effects of aqueous and ethanolic leaf extracts from drumstick tree (Moringa oleifera) on gilthead seabream (Sparus aurata L.) leucocytes, and their cytotoxic, antitumor, bactericidal and antioxidant activities. Fish Shellfish Immunol. 2020, 106, 44–55. [Google Scholar] [CrossRef] [PubMed]
- Elgendy, M.Y.; Awad, E.S.; Darwish, D.A.; Ibrahim, T.B.; Soliman, W.S.; Kenawy, A.M.; Abumourad, I.M.; Abbas, H.H.; Abbas, W. Investigations on the influence of Moringa oleifera on the growth, haematology, immunity and disease resistance in Oreochromis niloticus with special reference to the analysis of antioxidant activities by PAGE electrophoresis. Aquac. Res. 2021, 52, 4983–4995. [Google Scholar] [CrossRef]
- Jiménez-Monreal, A.M.; Guardiola, F.A.; Esteban, M.Á.; Tomás, M.A.M.; Martínez-Tomé, M. Antioxidant Activity in Gilthead Seabream (Sparus aurata L.) Fed with Diet Supplemented with Moringa. Antioxidants 2021, 10, 1423. [Google Scholar] [CrossRef] [PubMed]
- Sherif, A.H.; Prince, A.; Adel Seida, A.; Saad Sharaf, M.; Eldessouki, E.A.; Harfoush, M.A. Moringa oleifera mitigates oxytetracycline stress in Oreochromis niloticus. Aquac. Res. 2021, 52, 1–10. [Google Scholar] [CrossRef]
- Abdel-Latif, H.M.R.; Abdel-Daim, M.M.; Shukry, M.; Nowosad, J.; Kucharczyk, D. Benefits and applications of Moringa oleifera as a plant protein source in Aquafeed: A review. Aquaculture 2022, 547, 737369. [Google Scholar] [CrossRef]
- Aniballi, C.; Elmi, A.; Bertocchi, M.; Parmeggiani, A.; Govoni, N.; Scozzoli, M.; Ventrella, D.; Bacci, M.L. Effects of Gill Fish® on growth and welfare indices of farmed rainbow trout (Oncorhynchus mykiss) during early life stages. Aquaculture 2021, 533, 736158. [Google Scholar] [CrossRef]
- Billah, M.B.; Haque, M.E.; Sarkar, S.; Hossain, M.M.; Dey, S.K. Growth performance, hematological disorder and bacterial challenge on nile tilapia (Oreochromis niloticus) using moringa oleifera plant leaf as feed supplement. Bangladesh J. Zool. 2020, 48, 151–166. [Google Scholar] [CrossRef]
- LV, J. Effect of american ginseng saponins on enhancing immunity based on zebrafish model organisms. Chin. Tradit. Herb. Drugs 2020, 51, 3728–3733. [Google Scholar] [CrossRef]
- Peng, K.; Lv, X.; Zhao, H.; Chen, B.; Chen, X.; Huang, W. Antioxidant and intestinal recovery function of condensed tannins in Lateolabrax maculatus responded to in vivo and in vitro oxidative stress. Aquaculture 2022, 547, 737399. [Google Scholar] [CrossRef]
- Peng, K.; Zhou, Y.; Wang, Y.; Wang, G.; Huang, Y.; Cao, J. Inclusion of condensed tannins in Lateolabrax japonicus diets: Effects on growth, nutrient digestibility, antioxidant and immune capacity and copper sulphate stress resistance. Aquac. Rep. 2020, 18, 100525. [Google Scholar] [CrossRef]
- Johansson, M.W.; Keyser, P.; Sritunyalucksana, K.; Söderhäll, K. Crustacean haemocytes and haematopoiesis. Aquaculture 2000, 191, 45–52. [Google Scholar] [CrossRef]
- Liu, C.H.; Yeh, S.T.; Cheng, S.Y.; Chen, J.C. The immune response of the white shrimp Litopenaeus vannamei and its susceptibility to Vibrio infection in relation with the moult cycle. Fish Shellfish Immunol. 2004, 16, 151–161. [Google Scholar] [CrossRef]
- Ponprateep, S.; Vatanavicharn, T.; Lo, C.F.; Tassanakajon, A.; Rimphanitchayakit, V. Alpha-2-macroglobulin is a modulator of prophenoloxidase system in pacific white shrimp Litopenaeus vannamai. Fish Shellfish Immunol. 2017, 62, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Söderhäll, K.; Häll, L.; Unestam, T.; Nyhlén, L. Attachment of phenoloxidase to fungal cell walls in arthropod immunity. J. Invertebr. Pathol. 1979, 34, 285–294. [Google Scholar] [CrossRef]
- Leonard, C.; Ratcliffe, N.A.; Rowley, A.F. The role of prophenoloxidase activation in non-self recognition and phagocytosis by insect blood cells. J. Insect Physiol. 1985, 31, 789–799. [Google Scholar] [CrossRef]
- Holmblad, T.; Söderhäll, K. Cell adhesion molecules and antioxidative enzymes in a crustacean, possible role in immunity. Aquaculture 1999, 172, 111–123. [Google Scholar] [CrossRef]
- Bachere, E.; Mialhe, E.; Noel, D.; Boulo, V.; Morvan, A.; Rodriguez, J. Knowledge and research prospects in marine mollusc and crustacean immunology. Aquaculture 1995, 132, 17–32. [Google Scholar] [CrossRef]
- Chiu, C.H.; Guu, Y.K.; Liu, C.H.; Pan, T.M.; Cheng, W. Immune responses and gene expression in white shrimp, Litopenaeus vannamei, induced by Lactobacillus plantarum. Fish Shellfish Immunol. 2007, 23, 364–377. [Google Scholar] [CrossRef]
- Wu, Y.S.; Lee, M.C.; Huang, C.T.; Kung, T.C.; Huang, C.Y.; Nan, F.H. Effects of traditional medical herbs “Minor bupleurum decoction” on the non-specific immune responses of white shrimp (Litopenaeus vannamei). Fish Shellfish Immunol. 2017, 64, 218–225. [Google Scholar] [CrossRef]
- Shourbela, R.M.; El-Hawarry, W.N.; Abd El-Latif, A.M.; Abo-Kora, S.Y. Potentiality of Moringa oleifera aqueous extract as a growth modulator and antistress in acute hypoxic Nile tilapia Oreochromis niloticus. Iran. J. Fish. Sci. 2020, 19, 67–84. Available online: http://dorl.net/dor/20.1001.1.15622916.2020.19.1.24.6. (accessed on 9 December 2021).
- Tassanakajon, A.; Amparyup, P.; Somboonwiwat, K.; Supungul, P. Cationic antimicrobial peptides in penaeid shrimp. Mar. Biotechnol. 2011, 13, 639–657. [Google Scholar] [CrossRef]
- O’Leary, N.A.; Gross, P.S. Genomic structure and transcriptional regulation of the penaeidin gene family from Litopenaeus vannamei. Gene 2006, 371, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Destoumieux, D.; Bulet, P.; Strub, J.M.; van Dorsselaer, A.; Bachère, E. Recombinant expression and range of activity of penaeidins, antimicrobial peptides from penaeid shrimp. Eur. J. Biochem. 1999, 266, 335–346. [Google Scholar] [CrossRef] [Green Version]
- Cuthbertson, B.J.; Yang, Y.; Bachère, E.; Büllesbach, E.E.; Gross, P.S.; Aumelas, A. Solution structure of synthetic penaeidin-4 with structural and functional comparisons with penaeidin-3. J. Biol. Chem. 2005, 280, 16009–16018. [Google Scholar] [CrossRef] [Green Version]
- Somboonwiwat, K.; Marcos, M.; Tassanakajon, A.; Klinbunga, S.; Aumelas, A.; Romestand, B.; Gueguen, Y.; Boze, H.; Moulin, G.; Bachère, E. Recombinant expression and anti-microbial activity of anti-lipopolysaccharide factor (ALF) from the black tiger shrimp Penaeus monodon. Dev. Comp. Immunol. 2005, 29, 841–851. [Google Scholar] [CrossRef] [PubMed]
- Alambra, J.R.; Alenton, R.R.R.; Gulpeo, P.C.R.; Mecenas, C.L.; Miranda, A.P.; Thomas, R.C.; Velando, M.K.S.; Vitug, L.D.; Maningas, M.B.B. Immunomodulatory effects of turmeric, Curcuma longa (Magnoliophyta, Zingiberaceae) on Macrobrachium rosenbergii (Crustacea, Palaemonidae) against Vibrio alginolyticus (Proteobacteria, Vibrionaceae). Aquac. Aquar. Conserv. Legis. 2012, 5, 13–17. [Google Scholar]
- Sun, B.; Wang, Z.; Zhu, F. The crustin-like peptide plays opposite role in shrimp immune response to Vibrio alginolyticus and white spot syndrome virus (WSSV) infection. Fish Shellfish Immunol. 2017, 66, 487–496. [Google Scholar] [CrossRef] [PubMed]
- Shockey, J.E.; O’Leary, N.A.; de la Vega, E.; Browdy, C.L.; Baatz, J.E.; Gross, P.S. The role of crustins in Litopenaeus vannamei in response to infection with shrimp pathogens: An in vivo approach. Dev. Comp. Immunol. 2009, 33, 668–673. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Jiang, Q.; Wu, D.; Hu, Y.; Chen, S.; Ding, T.; Ye, X.; Liu, D.; Chen, J. What is new in lysozyme research and its application in food industry? A review. Food Chem. 2019, 274, 698–709. [Google Scholar] [CrossRef]
- Simser, J.A.; Macaluso, K.R.; Mulenga, A.; Azad, A.F. Immune-responsive lysozymes from hemocytes of the American dog tick, Dermacentor variabilis and an embryonic cell line of the Rocky Mountain wood tick, D. andersoni. Insect Biochem. Mol. Biol. 2004, 34, 1235–1246. [Google Scholar] [CrossRef]
- Yeh, M.S.; Chen, Y.L.; Tsai, I.H. The hemolymph clottable proteins of tiger shrimp, Penaeus monodon, and related species. Comp. Biochem. Physiol. 1998, 121, 169–176. [Google Scholar] [CrossRef]
- Maningas, M.B.B.; Kondo, H.; Hirono, I.; Saito-Taki, T.; Aoki, T. Essential function of transglutaminase and clotting protein in shrimp immunity. Mol. Immunol. 2008, 45, 1269–1275. [Google Scholar] [CrossRef] [PubMed]
- Su, B.; Chen, X. Current Status and Potential of Moringa oleifera Leaf as an Alternative Protein Source for Animal Feeds. Front. Vet. Sci. 2020, 7, 53. [Google Scholar] [CrossRef] [PubMed]
- Dongmeza, E.; Siddhuraju, P.; Francis, G.; Becker, K. Effects of dehydrated methanol extracts of moringa (Moringa oleifera Lam.) leaves and three of its fractions on growth performance and feed nutrient assimilation in Nile tilapia (Oreochromis niloticus (L.)). Aquaculture 2006, 261, 407–422. [Google Scholar] [CrossRef]
- Richter, N.; Siddhuraju, P.; Becker, K. Evaluation of nutritional quality of moringa (Moringa oleifera Lam.) leaves as an alternative protein source for Nile tilapia (Oreochromis niloticus L.). Aquaculture 2003, 217, 599–611. [Google Scholar] [CrossRef]
- Radhakrishnan, S.; Bhavan, P.S.; Seenivasan, C.; Shanthi, R.; Poongodi, R. Influence of medicinal herbs (Alteranthera sessilis, Eclipta alba and Cissus quadrangularis) on growth and biochemical parameters of the freshwater prawn Macrobrachium rosenbergii. Aquacult. Int. 2014, 22, 551–572. [Google Scholar] [CrossRef]
- Halim, A.M.; Lee, P.P.; Chang, Z.W.; Chang, C.C. The hot-water extract of leaves of noni, Morinda citrifolia, promotes the immunocompetence of giant freshwater prawn, Macrobrachium rosenbergii. Fish Shellfish Immunol. 2017, 64, 457–468. [Google Scholar] [CrossRef]

| Ingredients (g per kg Diet) | Control | ME1.25 | ME2.5 | ME5.0 |
|---|---|---|---|---|
| Fish meal | 500 | 500 | 500 | 500 |
| Shrimp meal | 60 | 60 | 60 | 60 |
| Yeast | 50 | 50 | 50 | 50 |
| Moringa extract | 0 | 1.25 | 2.5 | 5 |
| a-starch | 150 | 150 | 150 | 150 |
| fish oil | 15 | 15 | 15 | 15 |
| Lecithin | 5 | 5 | 5 | 5 |
| Cholesterol | 5 | 5 | 5 | 5 |
| Choline chloride | 5 | 5 | 5 | 5 |
| Vitamin D3 | 1 | 1 | 1 | 1 |
| Vitamin E | 1 | 1 | 1 | 1 |
| Vitamin A | 1 | 1 | 1 | 1 |
| Vitamin premix 1 | 40 | 40 | 40 | 40 |
| Mineral premix 2 | 40 | 40 | 40 | 40 |
| a-cellulose | 127 | 125.75 | 124.5 | 122 |
| Proximate analysis (% in dry weight) | ||||
| Crude protein | 50.11 | 50.63 | 50.65 | 50.84 |
| Crude lipid | 8.79 | 8.90 | 8.78 | 8.74 |
| Ash | 11.45 | 11.28 | 11.57 | 11.74 |
| Moisture | 7.8 | 7.9 | 7.5 | 7.2 |
| Gene | Primer Name | Sequence (5′-3′) | References |
|---|---|---|---|
| Prophenoloxidase2 | proPO2-F proPO2-R | ACCACTGGCACTGGCACCTCGTCTA TCGCCAGTTCTCGAGCTTCTGCAC | [36] |
| α2macroglobulin | A2M-F A2M-R | GCACGTAATCAAGATCCG CCCATCTCATTAGCACAAAC | [36] |
| Penaiedin2 | Pen2-F Pen2-R | TCGTGGTCTGCCTGGTCTT CAGGTCTGAACGGTGGTCTTC | [37] |
| Penaiedin3 | Pen3-F Pen3-R | CACCCTTCGTGAGACCTTTG AATATCCCTTTCCCACGTGAC | [37] |
| Anti-LPS factor | ALF-F ALF-R | CTGTGGAGGAACGAGGAGAC CCACCGCTTAGCATCTTGTT | [37] |
| Crustin | Crus-F Crus-R | GAGGGTCAAGCCTACTGCTG ACTTATCGAGGCCAGCACAC | [37] |
| Lysozyme | Lyz-F Lyz-R | GAAGCGACTACGGCAAGAAC AACCGTGAGACCAGCACTCT | [37] |
| Superoxide dismutase | SOD-F SOD-R | ATCCACCACACAAAGCATCA AGCTCTCGTCAATGGCTTGT | [38] |
| Clotting protein | CP-F CP-R | TCTTTGCGCAGTTGGTGATC TGAGGTGACCGAGTGCAAAA | [37] |
| EF1α | EF1α F EF1αR | ATGGTTGTCAACTTTGCCCC TTGACCTCCTTGATCACACC | [36] |
| Diet | Initial Weight (g) | Final Weight (g) | Weight Gain (%) | SGR (% Day−1) | FCR | Survival Rate |
|---|---|---|---|---|---|---|
| Control | 0.59 ± 0.05 | 5.57 ± 0.35 b | 855 ± 136 b | 3.74 ± 0.24 b | 1.39 ± 0.01 a | 91.67 ± 7.22 |
| ME1.25 | 0.59 ± 0.05 | 6.50 ± 0.57 ab | 1018 ± 183 ab | 4.01 ± 0.28 ab | 1.35 ± 0.03 ab | 91.67 ± 7.22 |
| ME2.5 | 0.54 ± 0.01 | 6.96 ± 0.47 a | 1200 ± 81 a | 4.26 ± 0.11 a | 1.29 ± 0.01 c | 91.67 ± 7.22 |
| ME5.0 | 0.56 ± 0.02 | 6.39 ± 0.10 ab | 1039 ± 40 ab | 4.04 ± 0.08 ab | 1.34 ± 0.02 bc | 95.83 ± 7.22 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abidin, Z.; Huang, H.-T.; Liao, Z.-H.; Chen, B.-Y.; Wu, Y.-S.; Lin, Y.-J.; Nan, F.-H. Moringa oleifera Leaves’ Extract Enhances Nonspecific Immune Responses, Resistance against Vibrio alginolyticus, and Growth in Whiteleg Shrimp (Penaeus vannamei). Animals 2022, 12, 42. https://doi.org/10.3390/ani12010042
Abidin Z, Huang H-T, Liao Z-H, Chen B-Y, Wu Y-S, Lin Y-J, Nan F-H. Moringa oleifera Leaves’ Extract Enhances Nonspecific Immune Responses, Resistance against Vibrio alginolyticus, and Growth in Whiteleg Shrimp (Penaeus vannamei). Animals. 2022; 12(1):42. https://doi.org/10.3390/ani12010042
Chicago/Turabian StyleAbidin, Zaenal, Huai-Ting Huang, Zhen-Hao Liao, Bo-Ying Chen, Yu-Sheng Wu, Yu-Ju Lin, and Fan-Hua Nan. 2022. "Moringa oleifera Leaves’ Extract Enhances Nonspecific Immune Responses, Resistance against Vibrio alginolyticus, and Growth in Whiteleg Shrimp (Penaeus vannamei)" Animals 12, no. 1: 42. https://doi.org/10.3390/ani12010042
APA StyleAbidin, Z., Huang, H.-T., Liao, Z.-H., Chen, B.-Y., Wu, Y.-S., Lin, Y.-J., & Nan, F.-H. (2022). Moringa oleifera Leaves’ Extract Enhances Nonspecific Immune Responses, Resistance against Vibrio alginolyticus, and Growth in Whiteleg Shrimp (Penaeus vannamei). Animals, 12(1), 42. https://doi.org/10.3390/ani12010042







