The Untrained Response of Pet Dogs to Human Epileptic Seizures
Abstract
:Simple Summary
Abstract
1. Introduction
2. Methods
2.1. Ethical Note
- To conduct nonlicensed animal research using 19 pet dogs which were exposed to seizure and control sample odours and their reactions monitored and recorded to those odours, Ref No. QUB-BS-AREC-19-001.
- Ethical approval was also granted to obtain sweat samples from three epilepsy patient volunteers and three control volunteer participants and for 19 dog owners to accompany their dogs and sit passively while the reaction of their dogs to seizure and control odours were being recorded. REF: 04/19/PowellNR1.
2.2. Volunteers for Sweat Samples
- –
- Volunteer A—a mature female living with daily absence and tonic-clonic seizures. Cause of epilepsy unknown.
- –
- Volunteer B—a mature female, also experiencing daily absence and tonic-clonic seizures. Cause of epilepsy unknown.
- –
- Volunteer C—an adult female with the genetic Lennox–Gastaut syndrome and severe developmental and learning issues and experienced several daily recurring tonic-clonic seizures. She was cared for by her parents.
- –
- pre-seizure taken when their untrained pet dog characteristically indicated that a seizure was imminent,
- –
- seizure sample harvested immediately while a seizure was occurring
- –
- post-seizure taken 6 h after a seizure episode to allow time for potential seizure-associated odours to dissipate.
2.3. Sample Collection and Storage
2.4. Study Participants
2.5. Data Collection
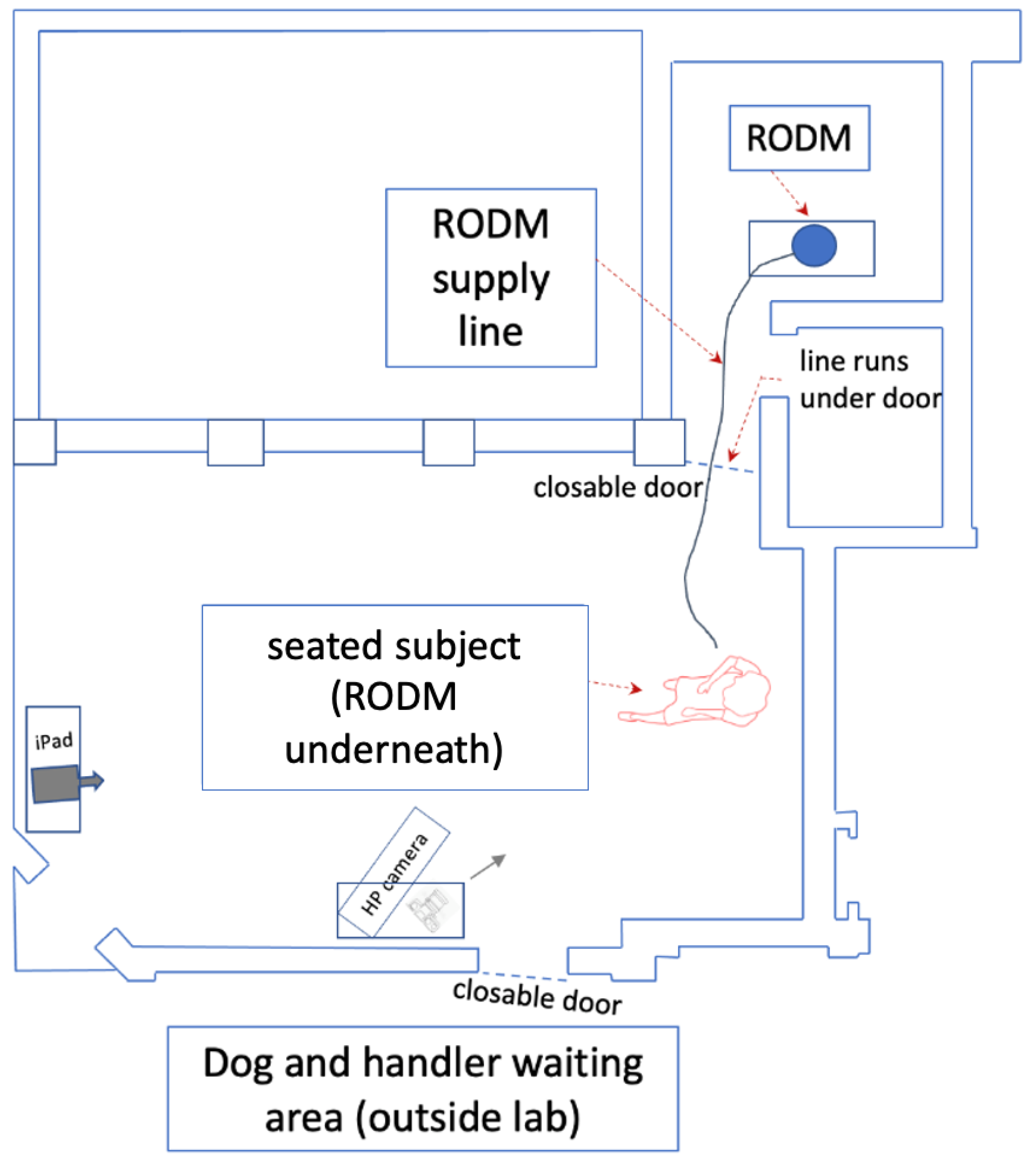
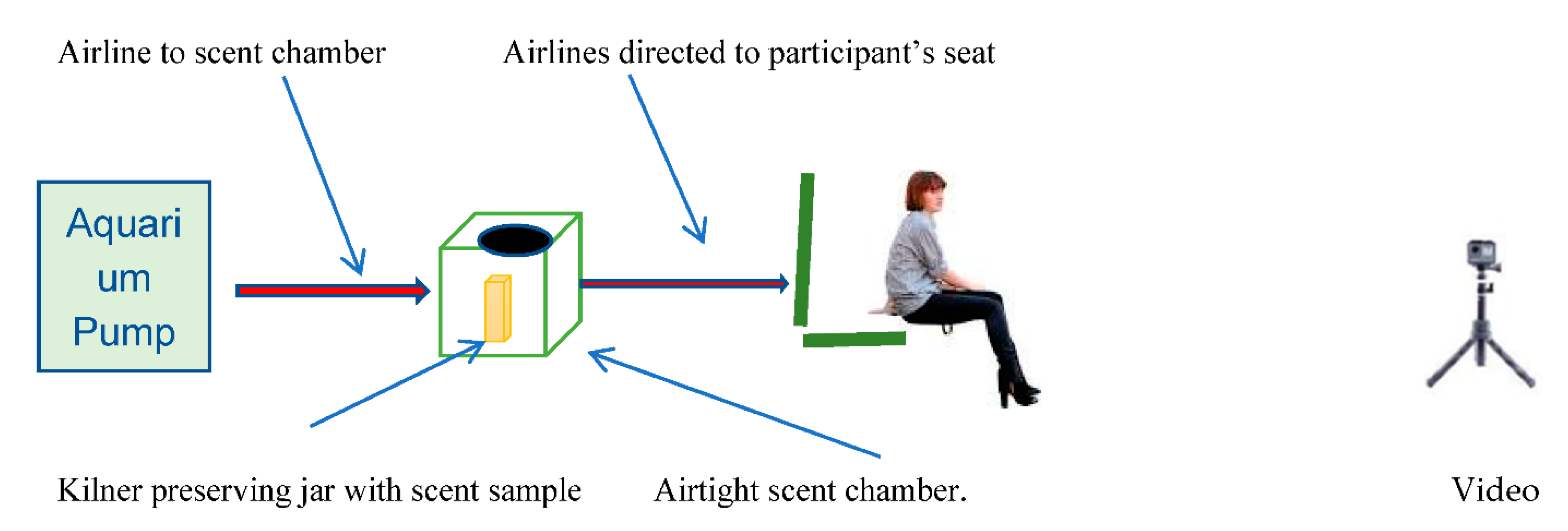
2.6. Experimental Procedure
- –
- Volunteer A: pre-seizure, 1, seizure, 2, post-seizure, 5
- –
- Volunteer B: pre-seizure, 3, seizure, 4, post-seizure, 6
- –
- Volunteer C: pre-seizure, 10, seizure, 11, post-seizure, 12
- –
- Control 1: 7
- –
- Control 2: 8
- –
- Pump delivers 200 Lt in 60 min = 18 s/1 L
- –
- Airtight keg volume = 3.6 Lt
- –
- Time to clear 3.6 Lt = 3.6 × 18 s = 64.8 s = 1 min approx.
- –
- Time to run sample scent before introducing dog = 1 min.
- –
- Time to run sample scent, dog in room = 3 min
- –
- Flush time after trial (directed outside window dog out of room) = 1 min.
- –
- Total time for each scent sample = 5 min
- –
- Total number of samples per dog = 5 samples
- –
- Total time needed for each dog = 5 × 5 = 25 min
- –
- +3 min initial habituation time = 28 min/dog
2.7. Statistical Analysis
3. Results
3.1. Demographic Information
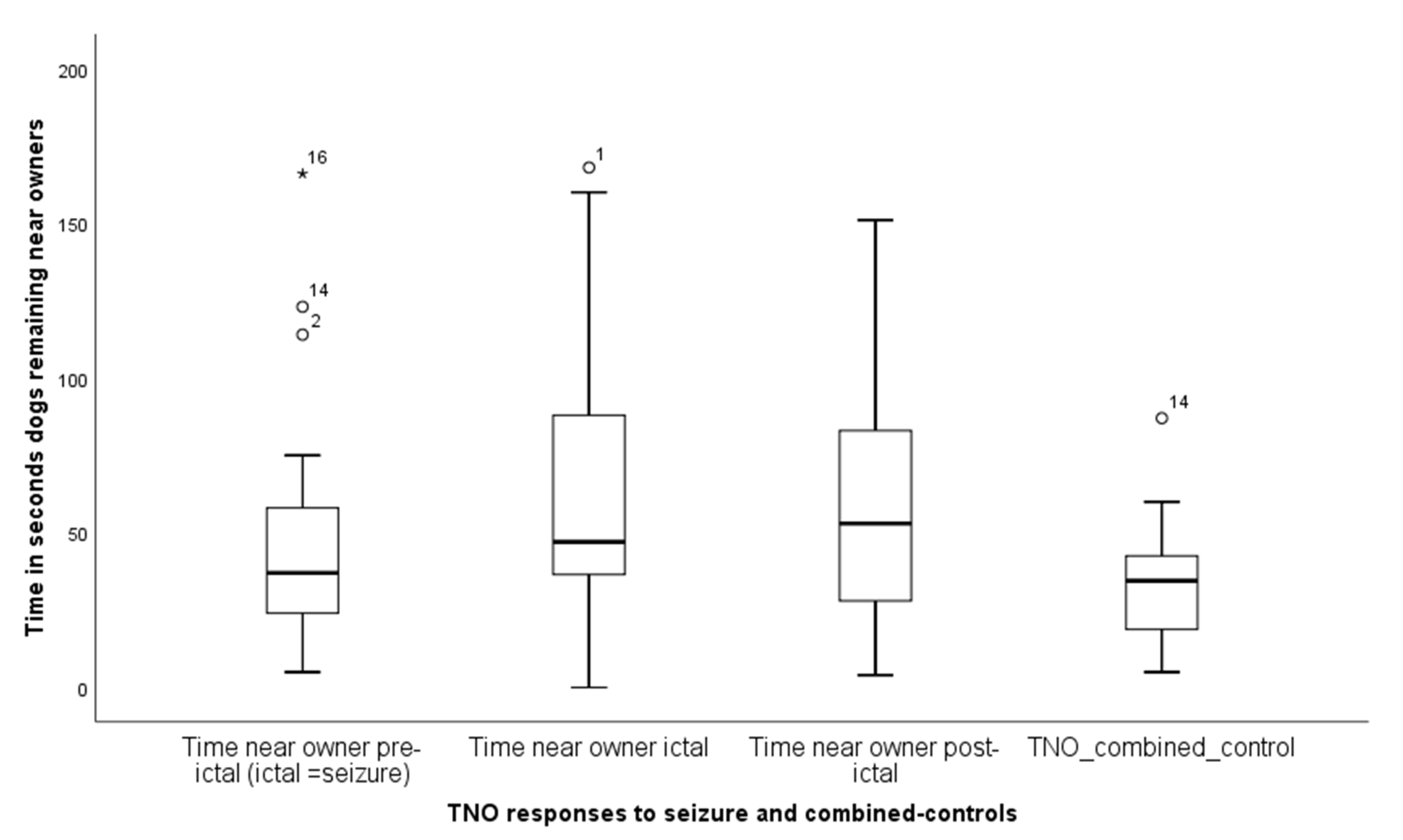
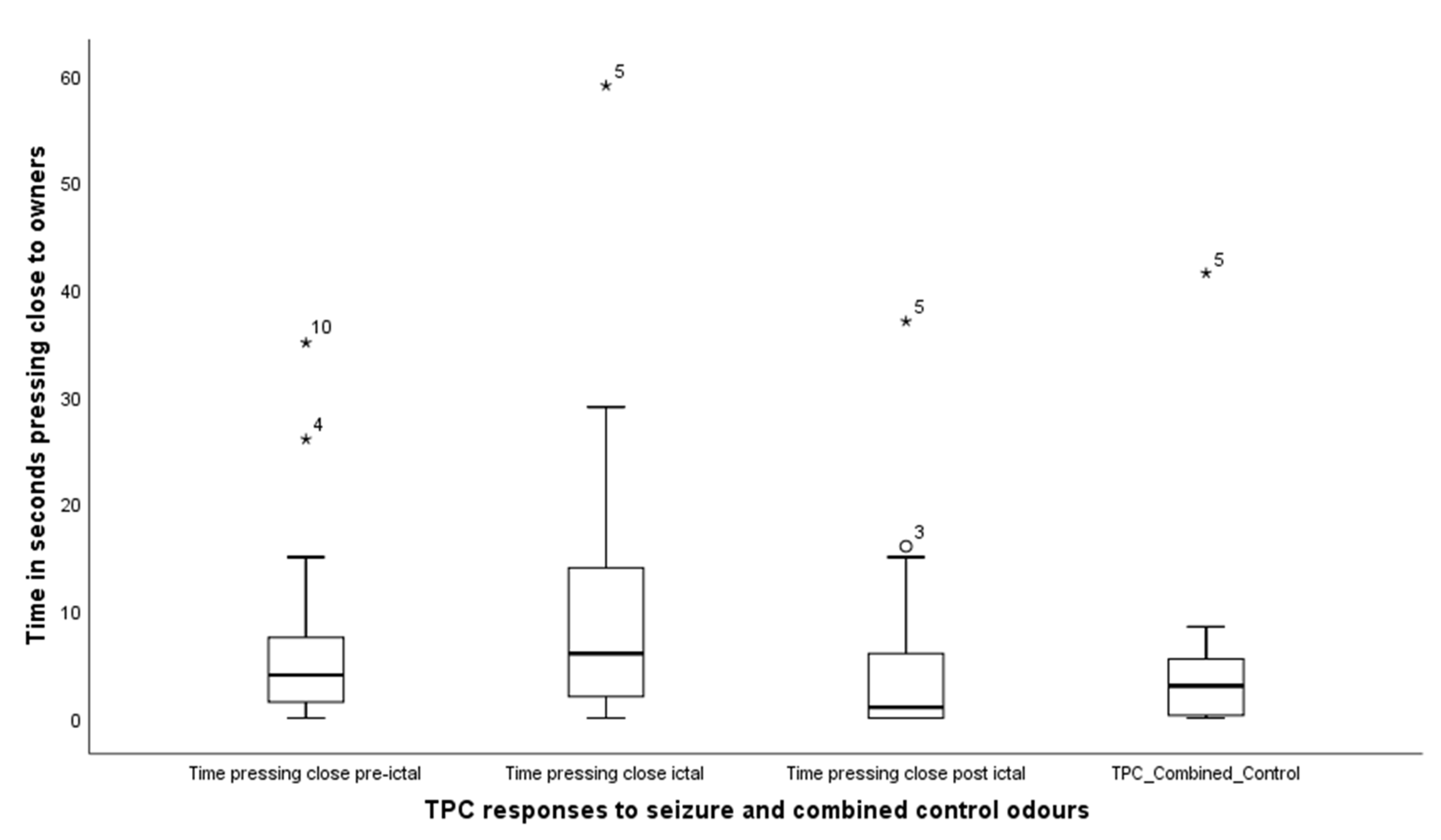
3.2. Behavioural Responses to Seizure-Related and Control Odours
4. Discussion
5. Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A. Instructions to Volunteers for Taking Sweat Samples
Appendix A.1. Pre-Seizure Samples
- When your dog warns of a seizure, put on fresh gloves, place 5 sterile pads in each armpit and clamp your arms tightly for 3 min to hold the pads in place.
- After 3 min, remove the samples and store them in the sealable bag labelled, Pre-Seizure. Add date and time.
- Place sample in the fridge for storage.
Appendix A.2. Seizure-Samples
- 4.
- Now in the time before a seizure, put on fresh gloves and place 5 new sterile pads in each armpit and leave them there until after the seizure passes.
- 5.
- After the seizure has passed, remove the pads and place them in the sealable bag labelled Seizure.
- 6.
- Write the date and time on the label and place in the fridge.
Appendix A.3. After-Seizure Samples
- 7.
- Six hours after the seizure, put on fresh sterile gloves and place 5 new pads under each arm
- 8.
- Clamp your arm tightly for 3 min, then place the pads in the sealable bag labelled Post-seizure. Write date and time on the jar and place in the fridge.
- 9.
- To return the samples, place them in the envelopes provided and post them back to me at:
Appendix B. Instructions for Taking Sweat Samples (No Epilepsy)
- Put on fresh gloves then, place 5 sterile pads in each armpit and clamp your arms tightly for 3 min to hold the pads in place.
- Clamp your arms tightly for 3 min to hold the pads in place.
- After 3 min, remove the samples and store them in the sealable bag labelled, Pre-Seizure. Add date and time.
- Place the sealed bag in the fridge for storage.
- To return the samples, place them in the envelopes provided and post them back to me at.
References
- Dalziel, D.J.; Uthman, B.M.; Mcgorray, S.P.; Reep, R.L. Seizure-alert dogs: A review and preliminary study. Seizure 2003, 12, 115–120. [Google Scholar] [CrossRef] [Green Version]
- Kirton, A.; Wirrell, E.; Zhang, J.; Hamiwka, L. Seizure-alerting and -response behaviors in dogs living with epileptic children. Am. Acad. Neurol. 2004, 62, 2303–2305. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kirton, A.; Winter, A.; Wirrell, E.; Snead, O. Seizure response dogs: Evaluation of a formal training program. Epilepsy Behav. 2008, 13, 499–504. [Google Scholar] [CrossRef]
- Mills, D.S.; Beral, A.; Lawson, S. Attention seeking behavior in dogs—what owners love and loathe! J. Vet. Behav. Clin. Appl. Res. 2010, 5, 60. [Google Scholar] [CrossRef]
- Martinez-Caja, A.M.; De Herdt, V.; Boon, P.; Brandl, U.; Cock, H.; Parra, J.; Perucca, E.; Thadani, V.; Moons, C.P.H. Seizure-alerting behavior in dogs owned by people experiencing seizures. Epilepsy Behav. 2019, 94, 104–111. [Google Scholar] [CrossRef]
- Catala, A.; Cousillas, H.; Hausberger, M.; Grandgeorge, M. Dog alerting and/or responding to epileptic seizures: A scoping review. PLoS ONE 2018, 13, e0208280. [Google Scholar] [CrossRef]
- Catala, A.; Grandgeorge, M.; Schaff, J.-L.; Cousillas, H.; Hausberger, M.; Cattet, J. Dogs demonstrate the existence of an epileptic seizure odour in humans. Sci. Rep. 2019, 9, 4103. [Google Scholar] [CrossRef] [PubMed]
- Powell, N. The Untrained Response of Pet Dogs to Human Epileptic Seizures. Ph.D. Thesis, Queens University Library, Belfast, UK, 2020. [Google Scholar]
- Csoltova, E.; Martineau, M.; Boissy, A.; Gilbert, C. Behavioral and physiological reactions in dogs to a veterinary examination: Owner-dog interactions improve canine well-being. Physiol. Behav. 2017, 177, 270–281. [Google Scholar] [CrossRef] [PubMed]
- Lensen, R.; Moons, C.P.; Diederich, C. Saliva sampling in dogs: How to select the most appropriate procedure for your study. J. Vet. Behav. 2015, 10, 504–512. [Google Scholar] [CrossRef]
- Lensen, R.C.; Betremieux, C.; Sys, S.U.; Moons, C.P.; Diederich, C.; Bavegems, V. Validity and reliability of cardiac measures during behavioural tests in pet dogs at home. Appl. Anim. Behav. Sci. 2017, 186, 56–63. [Google Scholar] [CrossRef]
- Lensen, R.C.M.M.; Moons, C.P.H.; Diederich, C. Physiological stress reactivity and recovery related to behavioral traits in dogs (Canis familiaris). PLoS ONE 2019, 14, e0222581. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Serpell, J.A.; Hsu, Y. Effects of breed, sex, and neuter status on train-ability in dogs. Anthrozoös 2005, 18, 196–207. [Google Scholar] [CrossRef] [Green Version]
- Whealy, M.; Wirrell, E.; Wong-Kisiel, L.; Nickels, K.; Payne, E.T. Prevalence and Risk Factors of Peri-ictal Autonomic Changes in Children With Temporal Lobe Seizures. Pediatr. Neurol. 2017, 67, 36–39. [Google Scholar] [CrossRef]
- Bruno, E.; Simblett, S.; LaNG, A.; Biondi, A.; Odoi, C.; Schulze-Bonhage, A.; Wykes, T.; Richardson, M.P. Wearable technology in epilepsy: The views of patients, caregivers and health care professionals. Epilepsy Behav. 2018, 85, 141–149. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hogg, M.C.; Raoof, R.; El Naggar, H.; Monsefi, N.; Delanty, N.; O’Brien, D.F.; Bauer, S.; Rosenow, F.; Henshall, D.C.; Prehn, J.H. Elevation of plasma tRNA fragments precedes seizures in human epilepsy. J. Clin. Investig. 2019, 129, 2946–2951. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nater, U.M.; Rohleder, N. Salivary alpha-amylase as a non-invasive biomarker for the sympathetic nervous system: Current state of research. Psychoneuroendocrinology 2009, 34, 486–496. [Google Scholar] [CrossRef] [PubMed]
- Linnemann, A.; Strahler, J.; Nater, U.M. The stress-reducing effect of music listening varies depending on the social context. Psychoneuroendocrinology 2016, 72, 97–105. [Google Scholar] [CrossRef]
- Skoluda, N.; La Marca, R.; Gollwitzer, M.; Müller, A.; Limm, H.; Marten-Mittag, B.; Gündel, H.; Angerer, P.; Nater, U.M. Long-term stability of diurnal salivary cortisol and alpha-amylase secretion patterns. Physiol. Behav. 2017, 175, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Pereira, V.H.; Campos, I.; Sousa, N. The role of autonomic nervous system in susceptibility and resilience to stress. Curr. Opin. Behav. Sci. 2017, 14, 102–107. [Google Scholar] [CrossRef]
- Burda, H.; Begall, S.; Cerveny, J.; Neef, J.; Nemec, P. Extremely low-frequency electromagnetic fields disrupt magnetic alignment of ruminants. Proc. Natl. Acad. Sci. USA 2009, 106, 5708–5713. [Google Scholar] [CrossRef] [Green Version]
- Buljubasic, F.; Buchbauer, G. The scent of human diseases: A review on specific volatile organic compounds as diagnostic biomarkers. Flavour Fragr. J. 2015, 30, 5–25. [Google Scholar] [CrossRef]
- Jadoon, S.; Sabiha, K.; Muhammad, R.A.; Abida, K.K.; Muhammad, A.Z.; Abdul, R.S.; Ghulam, M. Recent developments in sweat analysis and its applications. Int. J. Anal. Chem. 2015, 2015, 164974. [Google Scholar] [CrossRef] [PubMed]
- Edwards, T.L.; Browne, C.; Schoon, A.; Cox, C.; Poling, A. Animal olfactory detection of human diseases: Guidelines and systematic review. J. Vet. Behav. 2017, 20, 59–73. [Google Scholar] [CrossRef]
- Hackner, K.; Pleil, J. Canine olfaction as an alternative to analytical instruments for disease diagnosis: Understanding ‘dog personality’ to achieve reproducible results. J. Breath Res. 2017, 11, 012001. [Google Scholar] [CrossRef] [PubMed]
- Reeve, C.; Cummings, E.; McLaughlin, E.; Smith, S.; Gadbois, S. An Idiographic Investigation of Diabetic Alert Dogs’ Ability to Learn From a Small Sample of Breath Samples From People With Type 1 Diabetes. Can. J. Diabetes 2020, 44, 37–43.e1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tsukuda, M.; Nishiyama, Y.; Kawai, S.; Okumura, Y. Identifying stress biomarkers in skin gases by analysing gas collected from subjects undergoing the Trier social stress test performing statistical analysis. J. Breath Res. 2019, 13, 036003. [Google Scholar] [CrossRef]
- Catala, A.; Latour, P.; Martinez-Caja, A.M.; Cousillas, H.; Hausberger, M.; Grandgeorge, M. Is there a Profile of Spontaneous Seizure-Alert Pet Dogs? A Survey of French People with Epilepsy. Animals 2020, 10, 254. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maa, E.; Arnold, J.; Ninedorf, K.; Olsen, H. Canine Olfaction of volatile organic compounds unique to epileptic seizure. Epilepsy Behav. 2021, 115, 107690. [Google Scholar] [CrossRef] [PubMed]
- McCulloch, M.; Jezierski, T.; Broffman, M.; Hubbard, A.; Turner, K.; Janecki, T. Diagnostic Accuracy of Canine Scent Detection in Early- and Late-Stage Lung and Breast Cancers. Integr. Cancer Ther. 2006, 5, 30–39. [Google Scholar] [CrossRef]
- Sonoda, H.; Kohnoe, S.; Yamazato, T.; Satoh, Y.; Morizono, G.; Shikata, K.; Morita, M.; Watanabe, A.; Kakeji, Y.; Inoue, F.; et al. Colorectal cancer screening with odour material by canine scent detection. Gut 2011, 60, 814–819. [Google Scholar] [CrossRef]
- Walczak, M.; Jezierski, T.; Górecka-Bruzda, A.; Sobczyńska, M.; Ensminger, J. Impact of individual training parameters and manner of taking breath odor samples on the reliability of canines as cancer screeners. J. Vet. Behav. 2012, 7, 283–294. [Google Scholar] [CrossRef]
- Finamore, P.; Pedone, C.; Lelli, D.; Costanzo, L.; Bartoli, I.R.; De Vincentis, A.; Grasso, S.; Parente, F.R.; Pennazza, G.; Santonico, M.; et al. Analysis of volatile organic compounds: An innovative approach to heart failure characterization in older patients. J. Breath Res. 2018, 12, 026007. [Google Scholar] [CrossRef] [PubMed]
- Coren, S. How Dogs Think, Understanding the Canine Mind, 1st ed.; Free Press, Simon & Schuster UK Ltd., Africa House: London, UK, 2005; ISBN 0743222326. [Google Scholar]
- Walker, D.B.; Walker, J.C.; Cavnar, P.J.; Taylor, J.L.; Pickel, D.H.; Hall, S.B.; Suarez, J.C. Naturalistic quantification of canine olfactory sensitivity. Appl. Anim. Behav. Sci. 2006, 97, 241–254. [Google Scholar] [CrossRef]
- Bradshaw, J.W. In Defence of Dogs: Why Dogs Need Our Understanding; Penguin Group, Penguin Books Ltd.: London, UK, 2011; ISBN 978-1-846-14295-6. [Google Scholar]
- Quignon, P.; Rimbault, M.; Robin, S.; Galibert, F. Genetics of canine olfaction and receptor diversity. Mamm. Genome 2012, 23, 132–143. [Google Scholar] [CrossRef]
- Porritt, F.; Mansson, R.; Berry, A.; Cook, N.; Sibbald, N.; Nicklin, S. Validation of a short odour discrimination test for working dogs. Appl. Anim. Behav. Sci. 2015, 165, 133–142. [Google Scholar] [CrossRef] [Green Version]
- Lazarowski, L.; Dorman, D.C. Explosives detection by military working dogs: Olfactory generalization from components to mixtures. Appl. Anim. Behav. Sci. 2014, 151, 84–93. [Google Scholar] [CrossRef]
- Siniscalchi, M.; D’Ingeo, S.; Minunno, M.; Quaranta, A. Communication in Dogs. Animals 2018, 8, 131. [Google Scholar] [CrossRef] [Green Version]
- Miklósi, A.; Polgárdi, R.; Topál, J.; Csányi, V. Intentional behaviour in dog-human communication: An experimental analysis of "showing" behaviour in the dog. Anim. Cogn. 2000, 3, 159–166. [Google Scholar] [CrossRef]
- Miklósi, Á.; Kubinyi, E.; Topál, J.; Gácsi, M.; Virányi, Z.; Csányi, V. A Simple Reason for a Big Difference: Wolves Do Not Look Back at Humans, but Dogs Do. Curr. Biol. 2003, 13, 763–766. [Google Scholar] [CrossRef] [Green Version]
- Miklosi, A.; Topal, J.; Csanyi, V. Comparative social cognition: What can dogs teach us? Anim. Behav. 2004, 67, 995–1004. [Google Scholar] [CrossRef]
- Hare, B.; Brown, M.; Williamson, C.; Tomasello, M. The Domestication of Social Cognition in Dogs. Science 2002, 298, 1634–1636. [Google Scholar] [CrossRef] [Green Version]
- Hare, B.; Tomasello, M. Human-like social skills in dogs? Trends Cogn. Sci. 2005, 9, 439–444. [Google Scholar] [CrossRef]
- Miklosi, A.; Soproni, K. A comparative analysis of animals’ understanding of the human pointing gesture. Anim. Cogn. 2005, 9, 81–93. [Google Scholar] [CrossRef]
- Reeve, C.; Wentzell, P.; Wielens, B.; Bjorn, W.; Jones, C.; Stehouwer, K.; Gadbois, S. Assessing individual performance and maintaining breath sample integrity in biomedical detection dogs. Behav. Process. 2017, 155, 8–18. [Google Scholar] [CrossRef] [PubMed]
- D’Aniello, B.; Semin, G.R.; Alterisio, A.; Aria, M.; Scandurra, A. Interspecies transmission of emotional information via chemosignals: From humans to dogs (Canis lupus familiaris). Anim. Cogn. 2018, 21, 67–78. [Google Scholar] [CrossRef] [PubMed]
- Willis, C.M.; Church, S.M.; Guest, C.M.; Cook, W.A.; McCarthy, N.; Bransbury, A.J.; Church, M.R.T.; Church, J.C.T. Olfactory detection of human bladder cancer by dogs: Proof of principle study. BMJ 2004, 329, 712–714. [Google Scholar] [CrossRef] [Green Version]
- Forbes, S.; Rosenthal, N. Preparing the ground for tissue regeneration: From mechanism to therapy. Nat. Med. 2014, 20, 857–869. [Google Scholar] [CrossRef]
- Jezierski, T.; Adamkiewicz, E.; Walczak, M.; Sobczyńska, M.; Górecka-Bruzda, A.; Ensminger, J.; Papet, E. Efficacy of drug detection by fully-trained police dogs varies by breed, training level, type of drug and search environment. Forensic Sci. Int. 2014, 237, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Topál, J.; Miklósi, Á.; Csányi, V.; Dóka, A. Attachment behavior in dogs (Canis familiaris): A new application of Ainsworth’s (1969) Strange Situation Test. J. Comp. Psychol. 1998, 112, 219–229. Available online: http://ovidsp.ovid.com/ovidweb.cgi?T=JS&PAGE=reference&D=ovftc&NEWS=N&AN=00003159-199809000-00001 (accessed on 14 September 2020). [CrossRef] [Green Version]
- Prato-Previde, E.; Spiezio, C.; Sabatini, F.; Custance, D.M. Is the dog-human relationship an attachment bond? An observational study using Ainsworth’s strange situation. Behaviour 2003, 140, 225–254. [Google Scholar] [CrossRef]
- Palestrini, C.; Prato-Previde, E.; Spiezio, C.; Verga, M. Heart rate and behavioral responses of dogs in the Ainsworth’s strange situation: A pilot study. Appl. Anim. Behav. Sci. 2005, 94, 75–88. [Google Scholar] [CrossRef]
- Palmer, R.; Custance, D. A counterbalanced version of Ainsworth’s strange situation procedure reveals secure-base effects in dog–human relationships. Appl. Anim. Behav. Sci. 2008, 109, 306–319. [Google Scholar] [CrossRef]
- Rehn, T.; McGowan, R.T.; Keeling, L.J. Evaluating the situation procedure (SSP) to assess the bond between dogs and humans. PLoS ONE 2013, 8, 0056938. [Google Scholar] [CrossRef] [PubMed]
- Scandurra, A.; Alterisio, A.; D’Aniello, B. Behavioral effects of training on water rescue dogs in the Strange Situation Test. Appl. Anim. Behav. Sci. 2016, 174, 121–127. [Google Scholar] [CrossRef]
- Topal, J.; Kis, A.; Olah, K. Dogs’ sensitivity to human ostensive cues: A unique adaption? In The Social Dog: Behaviour and Cognition; Academic Press: Cambridge, MA, USA, 2014; p. 329. ISBN 9780124078185. [Google Scholar]
- Haidet, K.K.; Tate, J.; Divirgilio-Thomas, D.; Kolanowski, A.; Happ, M.B. Methods to improve reliability of video-recorded behavioural data. Res. Nurs. Health 2009, 32, 465–474. [Google Scholar] [CrossRef] [PubMed]
- Maa, E.H.; Arnold, J.; Bush, C.K. Epilespy and the smell of fear. Epilepsy Behav. 2021, 121, 108078. [Google Scholar] [CrossRef]
- Hoppe, C.; Feldmann, M.; Blachut, B.; Surges, R.; Elger, C.E.; Helmstaedter, C. Novel techniques for automated seizure registration: Patients’ wants and needs. Epilepsy Behav. 2015, 52, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Ramgopal, S.; Thome-Souza, S.; Jackson, M.; Kadish, N.; Fernández, I.S.; Klehm, J.; Bosl, W.; Reinsberger, C.; Schachter, S.; Loddenkemper, T. Seizure detection, seizure prediction, and closed-loop warning systems in epilepsy. Epilepsy Behav. 2014, 37, 291–307. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lazzaroni, M.; Marshall-Pescini, S.; Manzenreiter, H.; Gosch, S.; Přibilová, L.; Darc, L.; McGetrick, J.; Range, F. Why do dogs look back at the human in an impossible task? Looking back behaviour may be over-interpreted. Anim. Cogn. 2020, 23, 427–441. [Google Scholar] [CrossRef] [Green Version]
- Begall, S.; Červený, J.; Neef, J.; Vojtěch, O.; Burda, H. Magnetic alignment in grazing and resting cattle and deer. Proc. Natl. Acad. Sci. USA 2008, 105, 13451–13455. [Google Scholar] [CrossRef] [Green Version]
- Hart, V.; Nováková, P.; Malkemper, E.P.; Begall, S.; Hanzal, V.; Ježek, M.; Kušta, T.; Němcová, V.; Adámková, J.; Benediktova, K.; et al. Dogs are sensitive to small variations of the Earth’s magnetic field. Front. Zoöl. 2013, 10, 80. [Google Scholar] [CrossRef] [Green Version]
- Martini, S.; Begall, S.; Findeklee, T.; Schmitt, M.; Malkemper, E.P.; Burda, H. Dogs can be trained to find a bar magnet. PeerJ 2018, 6, e6117. [Google Scholar] [CrossRef] [PubMed]
- Bromfield, E.B.; Cavazos, J.E.; Reeves, J.L. (Eds.) Basic Mechanisms Underlying Seizures and Epilepsy. In An Introduction to Epilepsy [Internet]; American Epilepsy Society: West Hartford, CT, USA, 2006. Available online: https://www.ncbi.nlm.nih.gov/books/NBK2510/ (accessed on 14 October 2020).
- Sano, A.; Picard, R.W.; Stickgold, R. Quantitative analysis of wrist electrodermal activity during sleep. Int. J. Psychophysiol. 2014, 94, 382–389. [Google Scholar] [CrossRef]
- Bandarabadi, M.; Teixeira, C.; Rasekhi, J.; Dourado, A. Epileptic seizure prediction using relative spectral power features. Clin. Neurophysiol. 2015, 126, 237–248. [Google Scholar] [CrossRef]
- Moshe, L.S.; Perucca, E.; Ryvlin, P.; Torbjorn, T. Epilepsy New Advances. Lancet 2015, 385, 884–898. [Google Scholar] [CrossRef]
- Alvarez, V.; Reinsberger, C.; Scirica, B.; Obrien, M.H.; Avery, K.R.; Henderson, G.; Woo Lee, J. Continuous electrodermal activity as a potential novel neurophysical biomarker of prognosis after cardiac arrest—A pilot study. Resuscitation 2015, 93, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Epilepsy Action. 2020. Available online: https://www.epilepsy.org.uk/info/what-is-epilepsy (accessed on 2 August 2019).

| Demographic Factor Percentage | Number |
|---|---|
| Owner sex | |
| Male | 3 |
| 15.80% | |
| Female | 16 |
| 84.20% | |
| Owner age years | |
| 18–25 | 1 |
| 5.30% | |
| 26–35 | 7 |
| 36.80% | |
| 36–45 | 3 |
| 15.80% | |
| 46–55 | 1 |
| 5.30% | |
| 56–65 | 6 |
| 31.60% | |
| 66+ | 1 |
| 5.30% | |
| Dog sex | |
| Male | 8 |
| 42.10% | |
| Female | 11 |
| 57.90% | |
| Dog age months | |
| 6–35 | 7 |
| 36.80% | |
| 36–65 | 5 |
| 26.30% | |
| 66–95 | 3 |
| 15.80% | |
| 96–125 | 1 |
| 5.30% | |
| 126–155 | 2 |
| 10.50% | |
| 156–185 | 1 |
| 5.30% | |
| Dog breed | |
| Pedigree | 13 |
| 68.40% | |
| Mixed | 6 |
| 31.60% | |
| Length of ownership | |
| <12 months | 13 |
| 76.50% | |
| 1–5 years | 6 |
| 23.50% | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Powell, N.A.; Ruffell, A.; Arnott, G. The Untrained Response of Pet Dogs to Human Epileptic Seizures. Animals 2021, 11, 2267. https://doi.org/10.3390/ani11082267
Powell NA, Ruffell A, Arnott G. The Untrained Response of Pet Dogs to Human Epileptic Seizures. Animals. 2021; 11(8):2267. https://doi.org/10.3390/ani11082267
Chicago/Turabian StylePowell, Neil A., Alastair Ruffell, and Gareth Arnott. 2021. "The Untrained Response of Pet Dogs to Human Epileptic Seizures" Animals 11, no. 8: 2267. https://doi.org/10.3390/ani11082267
APA StylePowell, N. A., Ruffell, A., & Arnott, G. (2021). The Untrained Response of Pet Dogs to Human Epileptic Seizures. Animals, 11(8), 2267. https://doi.org/10.3390/ani11082267





