3. Results
Due to the low participation rate in the species-specific part (section B), only the data of the general section (section A) are presented here. Of the 44 participants who took part in the survey, 14 surveys were excluded due to incomplete data sets with less than 40% answered questions. In total, 30 surveys were included and evaluated. On average, the included participants answered 81% of the questions. Open fields were classified as “no information”.
In the included surveys, pigs were the most frequently housed farm animal species (n = 28) followed by small ruminants (n = 23) and large ruminants (n = 9). All the following results are based on these datasets.
Question 1:
Please provide detailed information about the nature of your facility, the housed species, the housing capacities and the age groups of the housed animals.
Pigs and small ruminants
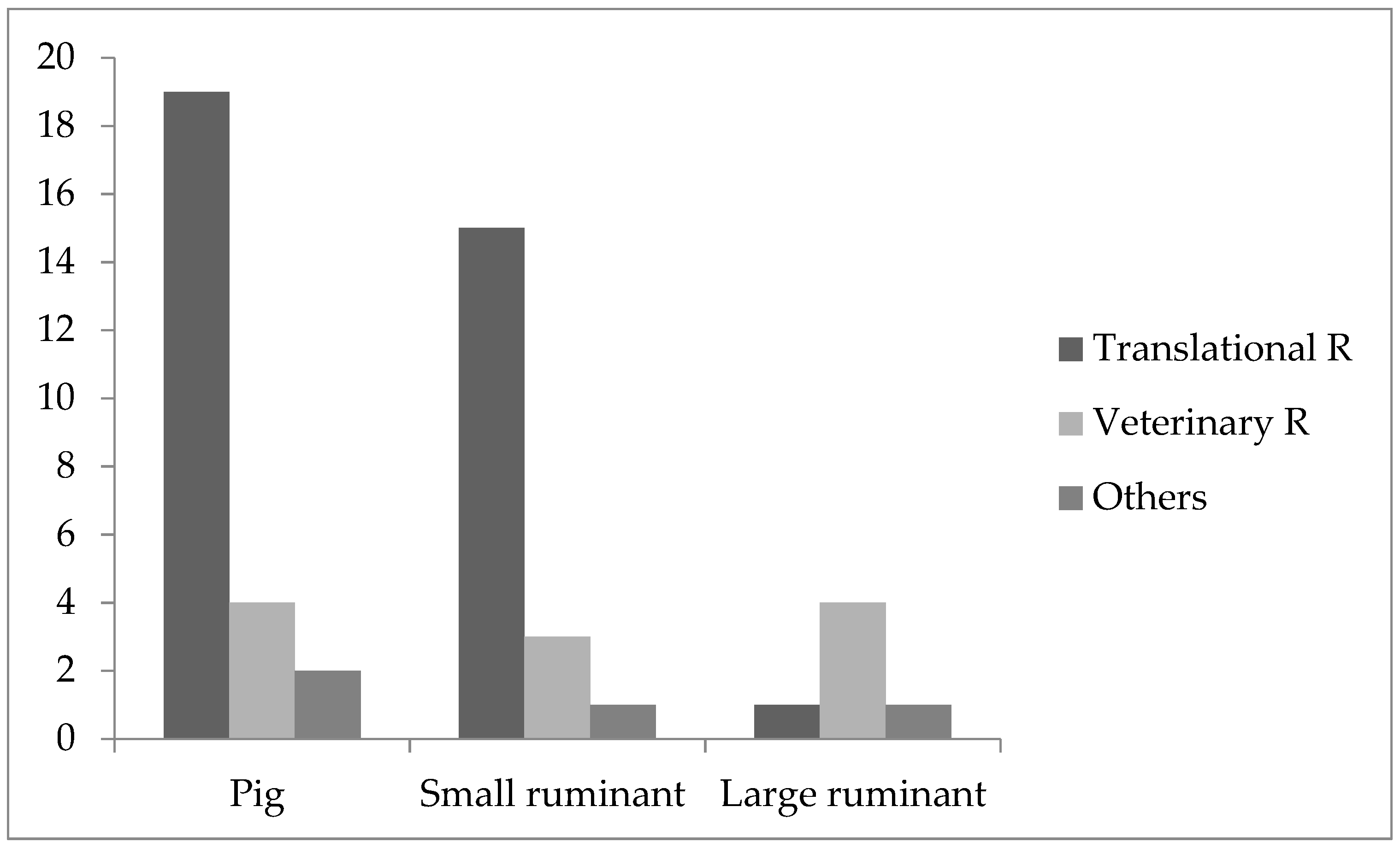
The majority of the participants used pigs and small ruminants in the field of medical research (pigs = 25; small ruminants = 19) with a focus on translational research (pigs = 19; small ruminants = 15), followed by veterinary research (pigs = 4; small ruminants = 3) and other fields (pigs= 2; small ruminants = 1;
Figure 1).
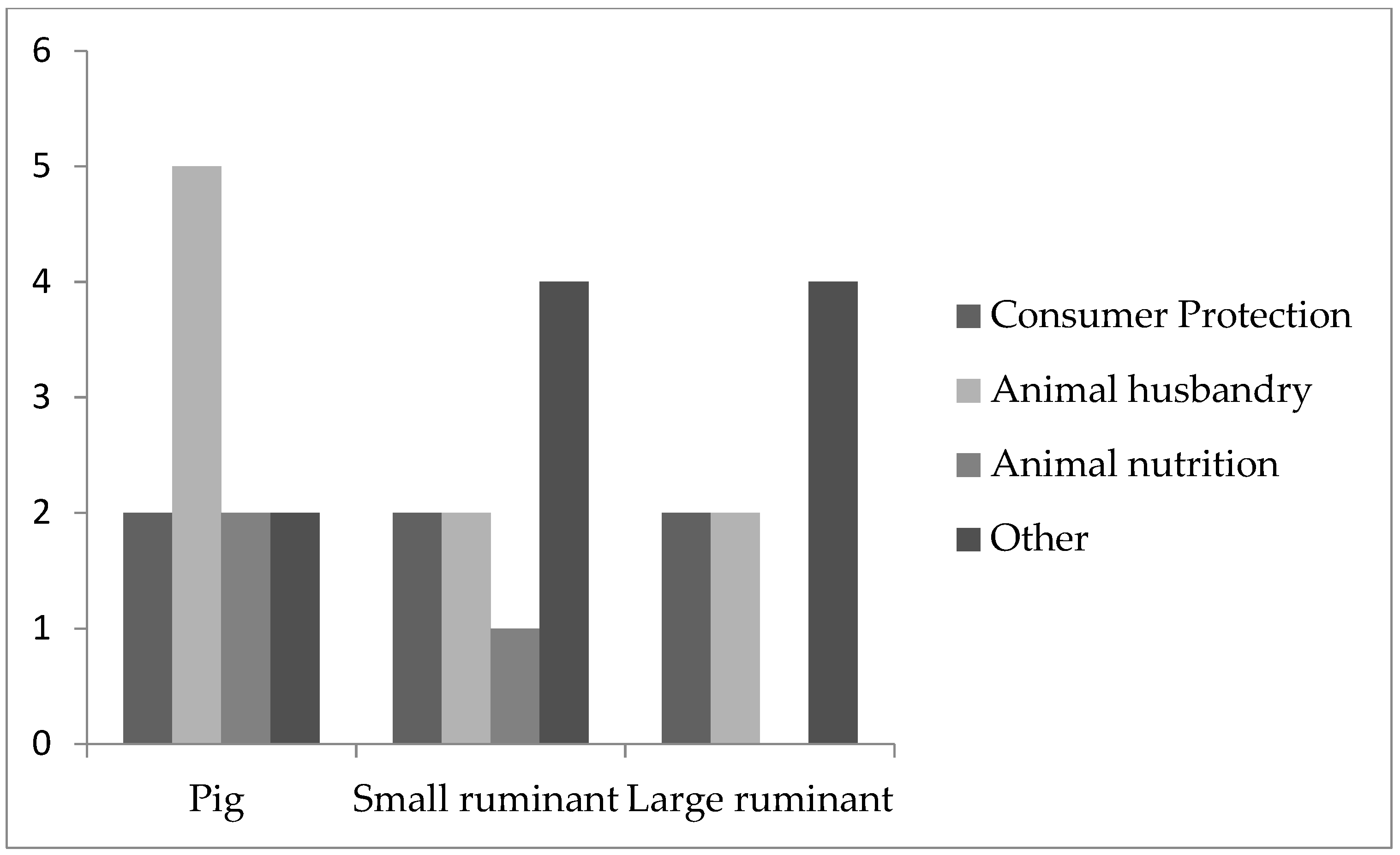
Pigs used in agricultural research (
n = 11;
Figure 2) were mostly used for research addressing animal husbandry (
n = 5). Two participants mentioned using pigs in consumer protection research, animal nutrition or other fields.
Small ruminants used for agricultural research (
n = 9) were mostly used in other fields (
n = 4) followed by consumer protection (
n = 2), animal husbandry (
n = 2) and animal nutrition (
n = 1) (
Figure 2).
Large ruminants
Large ruminants were more often used in agricultural research (
n = 8) compared to medical (
n = 6) research. In medical research (
Figure 1), most participants used large ruminants for veterinary research (
n = 4) followed by translational research and others (each = 1). In agricultural research, large ruminants were mostly used in other fields (
n = 4), followed by consumer protection (
n = 2) and animal husbandry (
n = 2) (
Figure 2).
Comments
In the general comments, fourteen participants mentioned using farm animals for teaching purposes, i.e., either in the translational research field for surgical courses or in animal experimentation courses for students, animal caretakers and researchers. In translational research, cardiology, orthopedics and regenerative medicine, vaccination development, plasma proteins and coagulation, imaging, anesthesia, biotechnology and testing of medical devices were reported. In agricultural or veterinary research, animal nutrition and health, housing, behavior, infection medicine and diagnostics of animal diseases were specified.
A total of 16 participant institutions were also breeding facilities, predominantly breeding for their own use, but also including two commercial breeders (pigs and small ruminants).
Furthermore, we asked for the age groups and total housing capacities of animals (
Table 1 and
Table 2). If participants responded, they provided information on both topics; however, the total number of answers was low regarding these topics (
Table 1 and
Table 2). Most of the facilities housed young and adult animals. For small ruminants, some facilities kept only adult animals. This could not be observed in facilities that kept pigs and large ruminants.
Question 2:
What are the access restrictions in your livestock facilities?
Pigs
Most participants had restricted access (yes = 25; no = 3) and changed their clothing and shoes (yes = 24; no = 4). Nine of the participants described their facility as a strike barrier (yes = 9; no = 17; no information = 2), three indicated working with showers (yes = 3; no = 23; no information = 2). Only seven participants reported having no access barriers (yes = 7; no = 19; no information = 2).
Small ruminants
Most participants had restricted access (yes = 19; no = 3; no information = 1) and changed their clothing and shoes (yes = 17; no = 5; no information = 1). Two of the participants described their facility as a strike barrier (yes = 2; no = 19; no information = 2). No participant indicated working with showers (yes = 0; no = 22; no information = 1). Only five participants reported having no access barriers (yes = 5; no = 16; no information = 2).
Large ruminants
Only one participant declared their facility as having restricted access (yes = 1; no = 8; no information = 0), but eight participants stated that they changed their clothing and shoes (yes = 8; no = 1; no information = 0). None of the participants described their facility as a strike barrier (yes = 0; no = 9; no information = 0) or indicated working with showers (yes = 0; no = 9; no information = 0). Two participants reported having no access barriers (yes = 2; no = 6; no information = 1).
Question 3:
Please describe your methods for germ reduction, in particular your ventilation and the type of water and feed used, as well as if roughage and green feed is provided.
Pigs
Six participants did not use any ventilation system in their facilities or did not provide information about the presence of a ventilation system. Eight participants reported having ventilation with overpressure and a HEPA-filtered air supply (n = 5) or without a HEPA-filtered air supply (n = 2). Eight participants reported using vacuum pressure ventilation with a HEPA-filtered air supply (n = 4) or without a HEPA-filtered air supply (n = 4).
In most of the facilities, untreated tap water was provided to the animals for drinking (n = 22), four supplied germ-reduced tap water and two did not provide information.
Most participants reported using standardized finished feed (n = 24) and four reported that they do not use finished food.
Twelve participants stated that they fed their animals roughage, one stated that they fed their animals germ-reduced roughage, two did not feed their animals roughage and 13 did not specify what they fed their animals. Three of the participants stated that they fed their animals green feed, 11 did not feed their animals green feed and 14 did not provide specific information.
Small ruminants
Most participants reported that there was no ventilation system in their facilities (n = 8) or did not provide information (n = 9). In four facilities, a ventilation system was present, either with overpressure and a HEPA-filtered air supply (n = 3) or without a HEPA-filtered air supply (n = 1). Two participants reported using vacuum pressure ventilation with a HEPA-filtered air supply.
Most of the participants supplied untreated tap water to the animals (n = 16), two used germ-reduced tap water and five did not provide information.
Most participants reported using standardized finished feed (n = 18), three reported that they did not use finished food and two did not provide any information. Ten of the participants stated that their animals received roughage, two did not feed their animals roughage and 11 did not provide specific information. Four of the participants stated that they fed their animals green feed, six did not feed their animals green feed and 13 did not provide specific information.
Large ruminants
Most participants did not provide information (n = 6). One participant declared the absence of a ventilation system in the facility (n = 1). Two participants reported having a ventilation system: One with overpressure and one with negative pressure, but both with a HEPA-filtered air supply.
In three facilities, untreated tap water was supplied to the animals (n = 3). One participant used germ-reduced tap water and five did not provide any information. Three participants reported using standardized finished feed (n = 3), but six did not provide any information. One participant stated that they fed their animals roughage, but eight did not provide specific information. One of the participants stated that they did not feed their animals green feed and the other eight did not provide specific information.
Question 4:
Please provide information regarding whether you purchase the animals from a special breeder for experimental use or from livestock production.
Pigs
Most of the participants purchased pigs from livestock production breeders (
Table 3,
n = 23; no information = 5), mostly consistently from one livestock farm (
n = 14).
Three participants indicated that they purchased pigs from an animal trader. Two participants received pigs from various livestock farms. One participant consistently purchased pigs from both a livestock farm and a breeder of experimental animals. One participant stated that they purchased pigs from a livestock farm and also bred pigs themselves. One participant indicated that they obtained pigs from various livestock farms and also bred pigs themselves. Another participant purchased pigs from various livestock farms and a trader.
Fifteen participants indicated that they obtained pigs from a special breeder (n = 15; no information = 13), mostly from one particular specialized breeder of experimental animals (n = 7), and also bred pigs themselves (n = 4).
Two participants purchased their pigs from various breeders of laboratory animals. Two participants indicated that they bred their own pigs but also purchased animals from various experimental animal breeders or animal traders.
Due to the possibility of leaving a comment in answer to this question, we received one answer stating that the origin of pigs depends on the number of animals that need to be ordered and their availability.
Small ruminants
From the participants who obtained small ruminants from a special breeder (
Table 4,
n = 11; no information = 12), most participants also bred animals themselves (
n = 5). Two participants stated that they bred their own animals and purchased animals from one breeder of experimental animals as well. Two participants purchased small ruminants from one breeder of experimental animals. Two participants purchased small ruminants from various specialized breeders of animals used for experimental purposes.
From the participants that purchased small ruminants from livestock production breeders (n = 18; no information = 5), the majority (n = 11) indicated that they obtained animals consistently from one farm. Two participants responded that they purchased small ruminants from various farms and two participants obtained small ruminants from a trader who obtained animals from various livestock farms. One participant indicated that they bred their own animals and purchased them from various livestock farms. Another participant bred their own animals and purchased animals from a trader who obtained small ruminants from a livestock farm.
Large ruminants
From the participants who stated that they obtained large ruminants from special breeders (n = 6; no information = 3), most participants served as their own breeding facility (n = 5). One participant bred animals independently and purchased animals used for experimentation from a specialized breeder.
From the participants who purchased large ruminants from livestock production (n = 7; no information = 2), three participants indicated that they bred their own large ruminants and also purchased large ruminants from one farm. Two participants consistently purchased large ruminants from one farm. One participant stated that they purchased large ruminants from various farms. One participant received animals from a trader who obtained animals from various unknown livestock farms.
Question 5:
Please describe the health status and hygiene monitoring performed when purchasing animals.
- 1.
Are the animals transported separately when obtained from different origins?
Pigs and small ruminants
When the animals were purchased from different origins, transportation of pigs and small ruminants was mostly performed separately (pigs: Transport separate = 22; transport not separate = 1; no information = 5; small ruminants: Transport separate = 16; transport not separate = 2; no information = 5).
Large ruminants
Large ruminants were more frequently transported together when purchased from different origins (transport separate = 2; transport not separate = 3; no information = 4).
- 2.
In general, do you purchase animals with or without a health certificate? Furthermore, please describe if the certificate includes information about disease history and serological, parasitological or bacteriological results.
Pigs
Ten participants purchased pigs without health certificates, eleven participants purchased pigs with health certificates and five participants answered that they purchased pigs both with and without health certificates (
Figure 3). Two participants provided no information.
Regarding the participants who purchased pigs with health certificates, most participants (n = 4) received a health certificate that included only the disease history and no further information on the serological, parasitological or bacteriological results. Three participants received the serological results, and one of them also included either the bacteriological or parasitological results. One participant received parasitological results and another parasitological and bacteriological results. Three participants gave no further information on the included analysis.
In the comments, it was noted that miniature pigs were specifically obtained in a pathogen-free state, according to the FELASA recommendations.
Small ruminants
Eight participants generally purchased small ruminants without a health certificate, eleven participants purchased small ruminants with a health certificate and two participants answered that they purchased small ruminants both with and without a health certificate (
Figure 4). Two participants provided no information.
Regarding the participants who received a health certificate, most participants (n = 4) received a health certificate without the disease history, serological, parasitological or bacteriological results. Three participants received a health certificate that included only the disease history. Two participants received a health certificate with serological results and one of them received a certificate with additional bacteriological results. Two participants received a health certificate with parasitological results and one of them received a certificate with additional bacteriological results. Only one participant received a health certificate with serological, parasitological and bacteriological results. One participant gave no further information on the analyses included in the certificate.
It was commented that rams are purchased with a health certificate stating their current status.
Large ruminants
Four participants generally purchased large ruminants without a health certificate and two participants purchased large ruminants with a health certificate. Three participants provided no information.
Regarding the participants who received a health certificate, one participant received a health certificate with the disease history and the other participants received a health certificate without any further information.
It was commented that when stock bulls are purchased, a health certificate with their current status is mandatory.
Comments
A few participants commented that the farm from which they consistently purchase animals monitored the livestock to determine their health status. In small ruminants and pigs an endoparasite treatment, and in small ruminants an ectoparasite treatment, are performed routinely before transportation in some cases.
- 3.
Does your health certificate include any information about the exclusion of a pregnancy and about their/other measures to avoid/exclude an unwanted pregnancy?
Pigs
Regarding the 16 participants who received a health certificate, most participants (n = 9) received a health certificate without the exclusion of pregnancy. Four participants received a health certificate with the exclusion of pregnancy and three provided no information.
Regarding all participants who housed pigs, most participants (n = 14) stated that they housed different sexes separate from each other to prevent unwanted pregnancies without performing additional ultrasound examinations or medicinal treatments. Eight participants answered that they performed no ultrasounds, medicinal treatments or separate housing of different sexes after delivery to detect or avoid a pregnancy. Six participants provided no information.
Small ruminants
Regarding the 13 participants who received a health certificate, most participants (n = 9) answered that the health certificate did not exclude pregnancy. Three participants received health certificates with the exclusion of pregnancy and one provided no information.
Regarding all participants housing small ruminants, most participants (n = 7) responded that they housed different sexes separate from each other to prevent unwanted pregnancies and without performing additional ultrasound examinations or medicinal treatments. Five participants answered that they performed no ultrasounds, medicinal treatments or separate housing of different sexes after delivery to detect or avoid pregnancies. Three participants responded that they performed ultrasounds and medicinal treatments, and did house different sexes separately. Two participants stated that they performed ultrasounds and had separate housing for different sexes to avoid pregnancies. Five participants provided no information on any aspect of the housing of small ruminants.
Large ruminants
Regarding the two participants who received a health certificate, one of them responded that they received a certificate that included the exclusion of pregnancy, while the other participant received a certificate without this information.
Regarding all participants housing large ruminants (n = 9), most participants (n = 3) responded that they performed no ultrasound or medicinal treatments and did not house different sexes separately. One participant responded that they did house different sexes separately. Five participants provided no information on any aspect of the housing of large ruminants.
Comments
As a comment, we received the answer that (in contrast to the exclusion of pregnancy), when pregnant farm animals are ordered, a certificate of pregnancy is mandatory.
Question 6:
The quarantine procedure, prophylactic treatment management and the restocking of the housing rooms were queried.
- 1.
Quarantine procedures
Pigs
While 13 facilities did not report the use of any quarantine program, 15 facilities confirmed that they quarantined the animals. Pigs were mostly quarantined routinely in their own holdings after delivery (n = 8), while quarantining was also performed in the holdings of origin either in suspected cases (n = 4) or routinely (n = 3) according to other participants.
Small ruminants
In 12 facilities, animals were quarantined in their own holdings either routinely (n = 8) or in suspected cases (n = 4). Nine participants answered that no quarantine procedures were carried out and two users provided no information.
Large ruminants
In five facilities, large ruminants were quarantined in their own holdings either routinely (n = 2) or only in suspected cases (n = 3). Two participants answered that they had no quarantine procedures or provided no information.
- 2.
Prophylactic treatments and restocking procedures of housing rooms (in–out principle or no in–out principle)
Pigs
Most participants (n = 18) performed prophylactic treatments; however, regarding choice, frequency and location, no clear trend was visible. Antiparasitic treatments alone were performed in five facilities (in participants’ own holdings—single treatment (n = 1) or repeated (n = 2); or in the holdings of origin—single treatment (n = 2)). Vaccinations alone were performed in four facilities (in participants’ own holdings—repeated (n = 1); or in the housing of origin—single (n = 1) or repeated (n = 2).
Combined treatments were confirmed by nine participants (in participants’ own holdings—single treatment (n = 1) or repeated treatments (n = 3); or in the holdings of origin—single (n = 3) or repeated (n = 2)).
No prophylactic treatments were confirmed by eight facilities. Two participants provided no information.
In most of the facilities (n = 22), new occupancy of the housing rooms was approached following the in–out principle, meaning that after occupation by one animal group, the room was disinfected before new animals were introduced. Three facilities did not restock following the in–out principle and three did not provide any information.
Small ruminants
Nineteen participants confirmed that prophylactic treatments were applied, while their frequency and application varied. Prophylactic antiparasitic treatments alone were applied in 12 facilities (in participants’ own holdings—single treatment (n = 2) or repeated treatments (n = 4); or in the holdings of origin—single treatment (n = 5) or repeated treatments = 1)) compared to vaccinations alone, which were applied in two facilities (in participants’ own holdings—repeated treatments). Five participants combined antiparasitic treatments and vaccination (in the holdings of origin—single (n = 1) or repeated treatments (n = 1); or in participants’ own holdings—repeated treatment = 3).
Two respondents answered that they did not perform any treatments and two participants did not provide any information.
The restocking of housing rooms for small ruminants followed the in–out principle in 13 facilities. In eight facilities, the in–out principle was not applied and two users did not provide any information on restocking procedures.
Large ruminants
Prophylactic treatments were carried out in five facilities in participants’ own holdings after the entry of the animals to the research premises. Single (n = 2) prophylaxis or repeated antiparasitic treatments (n = 1) were mainly performed. In only one facility were vaccinations or combinations of vaccinations and antiparasitic treatments performed. Four participants provided no information on prophylactic treatments. No trend regarding restocking procedures was visible (yes: 3; no: 4; no information: 2).
Question 7:
Are health-monitoring programs for farm animals established in your facility?
Do you perform a necropsy after the death of an animal to monitor health issues and, if so, is this carried out internally or externally?
Pigs
Most of the participants (n = 26) confirmed that they had an established health-monitoring program that is performed routinely in 17 facilities, while nine facilities only followed a health-monitoring program in suspected cases. Only one facility had no health-monitoring program or provided no information.
In most of the facilities, autopsies in pigs were performed in house in suspected cases (n = 14). In six facilities, in-house necropsies were performed routinely. Five participants mentioned that they did not perform in-house necropsies and three provided no information.
An examination of the sacrificed pigs in external institutes was performed only in suspected cases in 13 facilities, whereas nine facilities never performed external necropsies. Six participants provided no information.
Small ruminants
Most of the participants (n = 20) confirmed the presence of an established health-monitoring program in their facilities, which was either performed routinely (n = 12) or in suspected cases only (n = 8). Three facilities had no health-monitoring program.
In-house necropsies were performed in most of the facilities in suspected cases (n = 12), while some routinely performed necropsies (n = 4). In six facilities, no in-house pathology was performed. One participant provided no information.
Autopsies of small ruminants in external institutes was reported, but only in suspected cases (n = 15). Five participants answered that their facilities never demanded external necropsies. Three participants did not provide any information.
Large ruminants
Eight participants confirmed that they carried out routine monitoring of animal health within their facility. In one facility, health examinations were only performed in suspected cases.
Necropsies were performed externally more often (n = 7), versus in-house (n = 3) autopsies that were performed in suspected cases only. Examinations of animals’ external pathology were reported on two occasions, in suspected cases (n = 3) and routinely (n = 4). Five participants mentioned that they never performed necropsies of large ruminants in house. Two participants mentioned that they never required external pathological examinations of large ruminants. One participant provided no information.
Question 8:
What type of livestock animal husbandry is used?
Pigs
Most participants housed their pigs in groups in a stable all year round with (n = 14) or without (n = 8) the possibility of single housing. One participant stated that pigs were housed in single boxes in the stable all year round (n = 1). Two participants reported the possibility of group housing in the stable with year-round pasture keeping and the possibility of separate housing of individual animals (n = 2). Three participants did not provide information.
Small ruminants
The majority of the participants reported that they housed small ruminants in groups in a stable with temporary pasture keeping (n = 7) or in groups in a stable all year round with the possibility of single housing (n = 4). Two participants reported that they housed small ruminants in groups in a stable (n = 2) or kept them in a pasture all year round with (n = 2) or without the possibility of single housing (n = 2). Six participants did not provide information.
Large ruminants
From the nine participants that answered the question, one reported housing their large ruminants in groups in a stable with temporary pasture keeping (n = 1). Eight participants did not provide information.
Question 9:
Have you established any kind of internal or external program to share organs and tissues?
Are animals released, rehomed or returned to the food chain after the end of procedures?
- 1.
Sharing Organs and tissue
Pigs
The majority (n = 22) of the participants reported that they use programs for sharing the organs and tissues of sacrificed animals. This result was crosslinked to the only use of institutional (internal) sharing programs. Two participants provided no information and four participants used neither an internal nor an external program.
Small ruminants
A total of 15 participants confirmed the use of programs to share organs and tissues. As with pigs, only internal programs were used. Only one user provided no information and seven participants used neither an internal nor an external program.
Large ruminants
In total, five participants endorsed the use of sharing programs. In three cases, this was related to internal programs and two participants confirmed the use of external programs. Two participants used neither an internal nor an external program and one participant did not provide information.
- 2.
Animal release, rehoming or return to food chain
Pigs
The majority of participants did not give animals away after the end of use (n = 17). Private release (rehoming) was confirmed in four cases and commercial release to return pigs to the food chain (external slaughtering) was confirmed in five cases. However, only three participants confirmed that they carried out commercial animal release and external slaughtering with the presence of an official veterinary certification. In addition, a total number of four participants confirmed that they carried out internal slaughtering. Two participants did not provide any information.
Small ruminants
Most of the participating facilities did not give animals away after the end of their use, neither privately nor commercially (n = 15). Private release (rehoming) was confirmed in four cases and commercial release to return small ruminants to the food chain (external slaughtering) was confirmed in four cases. However, only two participants confirmed that they carried out commercial animal release and external slaughtering with the presence of an official veterinary certification. In addition, a total number of four participants confirmed that they carried out internal slaughtering. One participant did not provide information.
Large ruminants
In total, two participants stated that they do not give animals away after the end of their use, neither privately nor commercially. Private release (rehoming) was confirmed in one case and commercial release to return large ruminants to the food chain (external slaughtering) was confirmed in five cases. Three participants confirmed that they carried out commercial animal release and external slaughtering with the presence of an official veterinary certification. In addition, a total number of two participants confirmed that they carried out internal slaughtering. Two participants did not provide information.