Development of Calibration Models to Predict Mean Fibre Diameter in Llama (Lama glama) Fleeces with Near Infrared Spectroscopy
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Samples: Characterisation and Treatments
2.2. Reference Analysis
2.3. Spectra Collection
2.4. Spectral Preprocessing and Calibration Models Development
- -
- Vis–NIR: range from 400 to 2500 nm (4200 datapoints).
- -
- NIR: range from 1100 to 2500 nm (2800 datapoints). Visible and a section of the NIR region of the spectra was discarded due to a large variability originated by pigmented fibres.
- -
3. Results
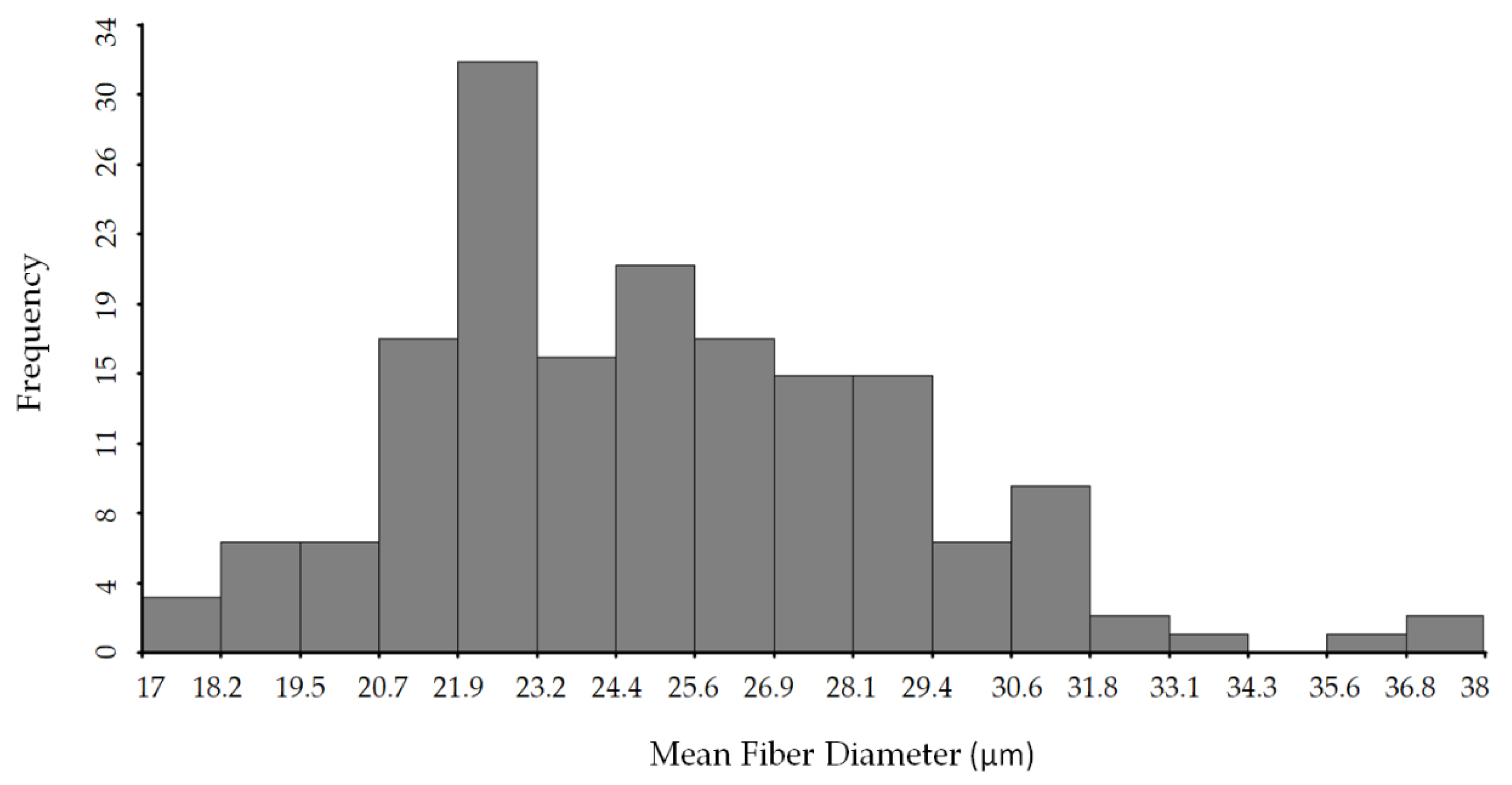
3.1. Reference Analysis
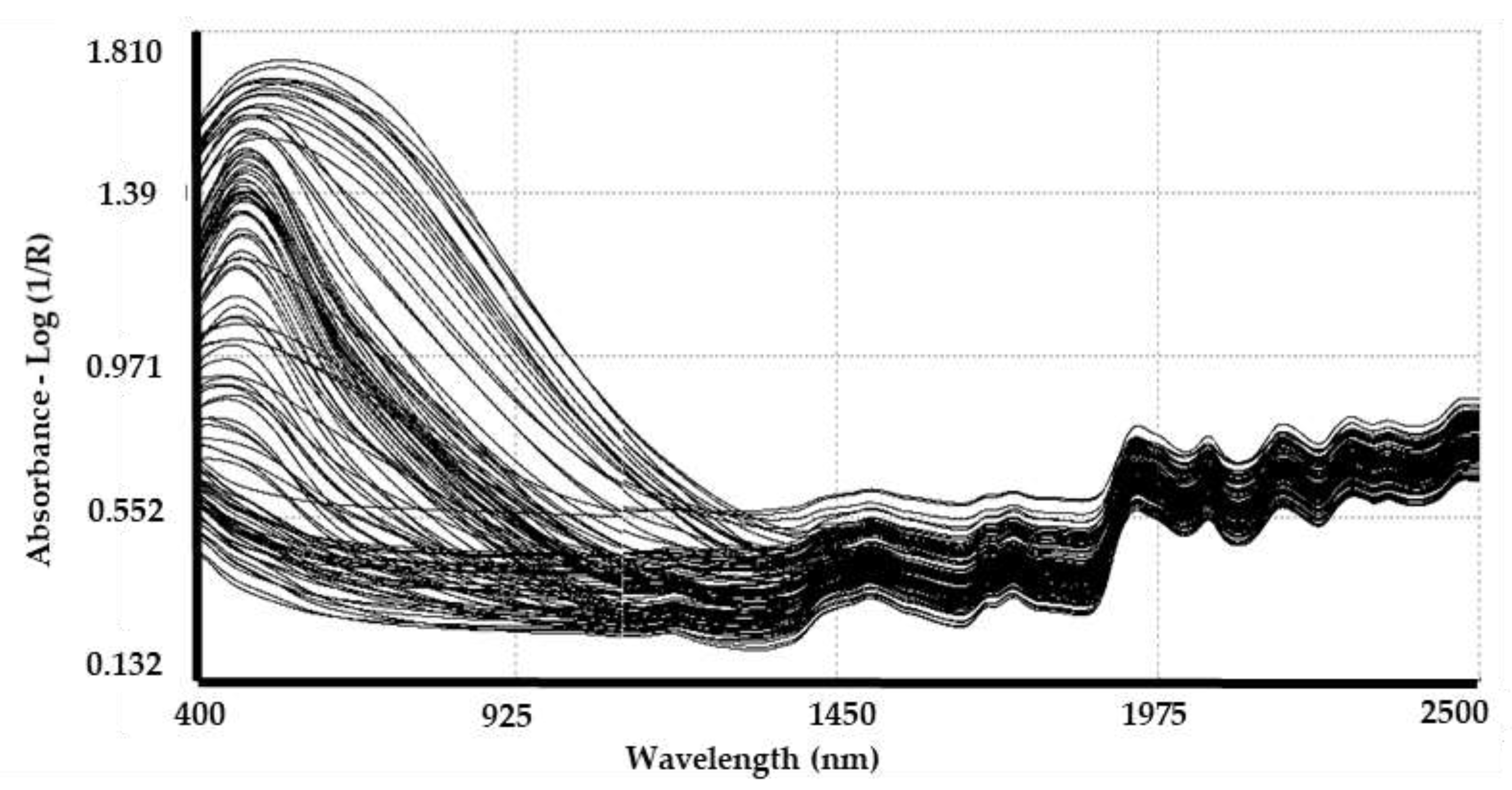
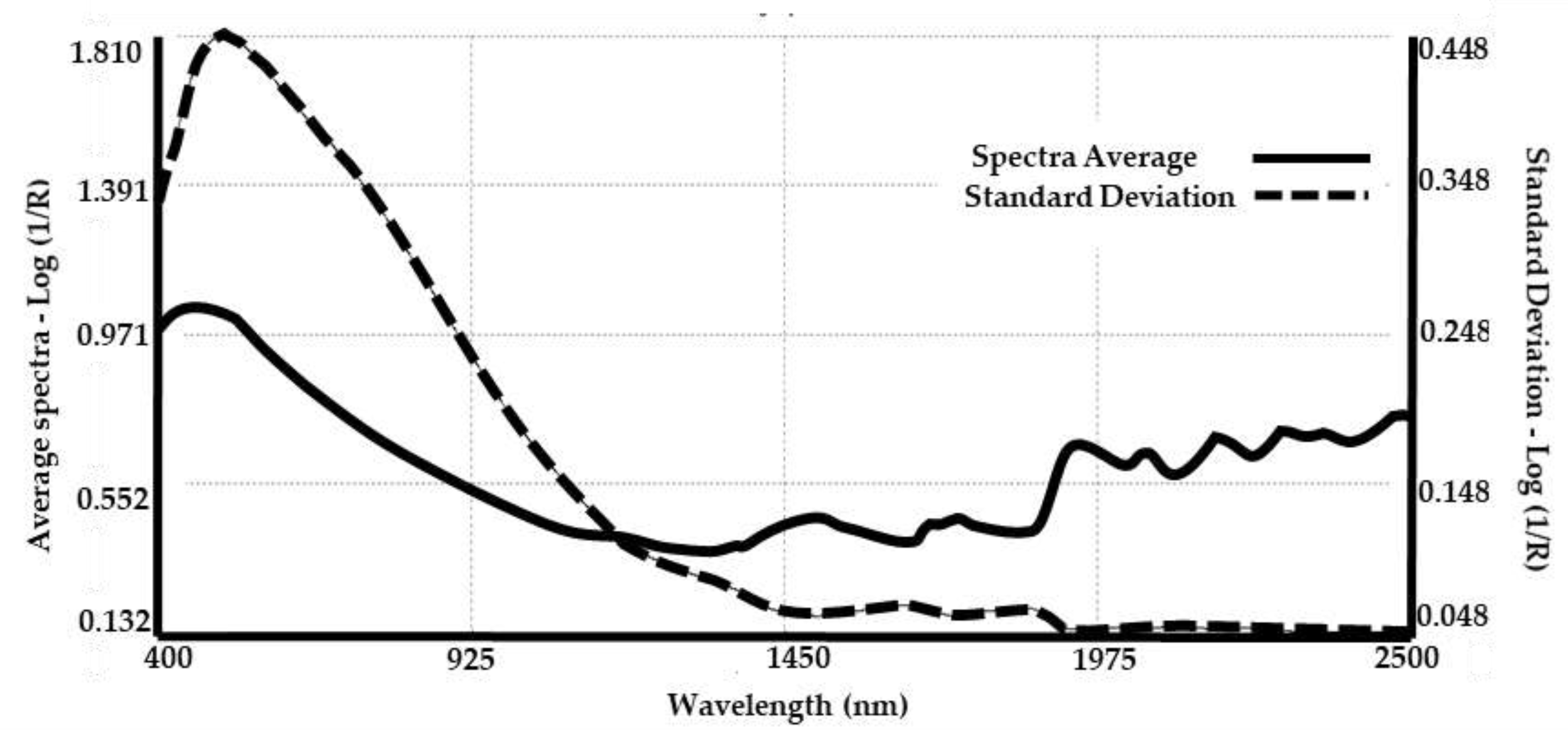
3.2. Spectra Collection
3.3. Spectral Processing and Calibration Models Analysis
4. Discussion
4.1. Reference Analysis
4.2. Spectral Pretreatments and Calibration Models
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Geyer, R.; Jambeck, J.R.; Law, K.L. Production, Use, and Fate of All Plastics Ever Made. Sci. Adv. 2017, 3, 25–29. [Google Scholar] [CrossRef]
- Mengüç, G.S.; Özdil, N.; Hes, L. Prickle and Handle Properties of Fabrics Produced from Specialty Animal Fibers. Text. Res. J. 2015, 85, 2155–2167. [Google Scholar] [CrossRef]
- Rojo, V.; Arzamendia, Y.; Vilá, B. Uso Del Hábitad Por Vicuñas (Vicugna Vicugna) En Un Sistema Agropastoril En Suripujio, Jujuy. Mastozool. Neotrop. 2012, 19, 127–138. [Google Scholar]
- Echenique, M.; Echenique, J.; Fava, R.; Alcoba, D.; Quiroga Mendiola, M. La Producción y Comercialización de Carne de La Agricultura Familiar En La Puna Jujeña, 1st ed.; INTA Ediciones: Ciudad Autónoma de Buenos Aires, Argentina, 2015; ISBN 9789875216402. [Google Scholar]
- Hick, M.; Frank, E.; Prieto, A.; Castillo, M. Ethno-Zootechnical Characterization of the Population of Fiber Bearing Lamas from the Province of Jujuy, Argentina. Arch. Latinoam. Prod. Anim. 2014, 22, 1–8. [Google Scholar]
- Mueller, J.P.; Rigalt, F.; Lamas, H.; Sacchero, D.M.; Cancino, A.K.; Wurzinger, M. Fibre Quality of South American Camelids in Argentina: A Review. Anim. Genet. Resour. 2015, 56, 97–109. [Google Scholar] [CrossRef]
- Rajabinejad, H.; Zoccola, M.; Patrucco, A.; Montarsolo, A.; Rovero, G.; Tonin, C. Physicochemical Properties of Keratin Extracted from Wool by Various Methods. Text. Res. J. 2018, 88, 2415–2424. [Google Scholar] [CrossRef]
- Quispe, E.C.; Chipa, L.; Pinares, R. Análisis Económico y de La Producción Del Descerdado Manual de La Fibra de Llamas (Lama Glama) Chaku. Arch. Zootec. 2015, 64, 191–198. [Google Scholar] [CrossRef][Green Version]
- McGregor, B.A. Properties, Processing and Performance of Rare and Natural Fibres, 1st ed.; Rural Industries Research and Development Corporation: Canberra, Australia, 2012; ISBN 9781742543338.
- Imasheva, A.; Otinshiev, M.; Alikhanov, D.; Daskalov, P. Development of Technology and Means of Cashmere down Processing. Bulg. J. Agric. Sci. 2014, 20, 980–987. [Google Scholar]
- Brodtmann, L.I.; Hick, M.H.; Castillo, M.F.; Frank, E.N. Conceptual Description of the Llama Fleece Structure and the Potential of Classing and Dehairing. Text. Res. J. 2017, 88, 1–18. [Google Scholar] [CrossRef]
- Adot, O.G.; Frank, E.N. Industrialization and Commercialisation of the Fibre of South American Camelids in Argentina. Int. J. Sci. Res. Innov. Technol. 2015, 2, 52–59. [Google Scholar]
- Canaza-Cayo, A.W.; Alomar, D.; Quispe, E. Prediction of Alpaca Fibre Quality by Near-Infrared Reflectance Spectroscopy. Animal 2013, 7, 1–7. [Google Scholar] [CrossRef]
- Sommerville, P. Fundamental Principles of Fibre Fineness Measurement; Australian Wool Testing Authority Ltd.: Kensington, Australia, 2007; ISBN 18328679. [Google Scholar]
- Cottle, D.J.; Baxter, B.P. Wool Metrology Research and Development to Date. Text. Prog. 2015, 47, 163–315. [Google Scholar] [CrossRef]
- Chen, H.; Lin, Z.; Tan, C. Classification of Different Animal Fibers by near Infrared Spectroscopy and Chemometric Models. Microchem. J. 2019, 144, 489–494. [Google Scholar] [CrossRef]
- Prieto, N.; Pawluczyk, O.; Dugan, M.E.R.; Aalhus, J.L. A Review of the Principles and Applications of Near-Infrared Spectroscopy to Characterize Meat, Fat, and Meat Products. Appl. Spectrosc. 2017, 71, 1403–1426. [Google Scholar] [CrossRef]
- Fearn, T. Assesing Calibrations: SEP, RPD, RER, R2. NIR News 2002, 13, 3. [Google Scholar] [CrossRef]
- Igne, B.; Reeves, J.B.; Mccarty, G.; Hively, W.D.; Lund, E.; Hurburgh, C.R. Evaluation of Spectral Pretreatments, Partial Least Squares, Least Squares—Support Vector Machines and Locally Weighted Regression for Quantitative Spectroscopic. J. Near Infrared Spectrosc. 2010, 18, 167–176. [Google Scholar] [CrossRef]
- Dahm, D.J. Explaining Some Light Scattering Properties of Milk Using Representative Layer Theory. J. Near Infrared Spectrosc. 2013, 339, 323–339. [Google Scholar] [CrossRef]
- Shenk, J.S.; Workman, J.; Westerhaus, M.O. Application of NIR spectroscopy to agricultural products. In Handbook of Near-Infrared Analysis; Burns, D.A., Ciurczak, E.W., Eds.; CRC Press Taylor & Francis Group: Boca Ratón, FL, USA, 2008; pp. 347–386. [Google Scholar]
- Fearn, T.; Riccioli, C.; Garrido-Varo, A.; Guerrero-Ginel, J.E. On the Geometry of SNV and MSC. Chemom. Intell. Lab. Syst. 2009, 96, 22–26. [Google Scholar] [CrossRef]
- Connell, J.P.; Brown, G.H. The Yield Testing Of Wool By Reflectance Spectroscopy. J. Text. Inst. 1978, 69, 357–363. [Google Scholar] [CrossRef]
- Hammersley, M.J.; Townsend, P.E. NIR Analysis of Wool. In Handbook of Near Infrared Analysis; Burns, D.A., Ciurczak, E.W., Eds.; CRC Press Taylor & Francis Group: Boca Raton, FL, USA, 2007; pp. 465–478. [Google Scholar]
- Davis, C.B.; Busch, K.W.; Rabbe, D.H.; Busch, M.A.; Lusk, J.R. Rapid, Non-Destructive, Textile Classification Using SIMCA on Diffuse Near-Infrared Reflectance Spectra. J. Mod. Phys. 2015, 06, 711–718. [Google Scholar] [CrossRef][Green Version]
- Boguslavsky, A.; Botha, A.; Hunter, L. Measuring Medullation in Mohair with Near Infrared Reflectance Analysis. Text. Res. J. 1992, 62, 433–437. [Google Scholar] [CrossRef]
- Lupton, C.J.; McColl, A. Measurement of Luster in Suri Alpaca Fiber. Small Rumin. Res. 2011, 99, 178–186. [Google Scholar] [CrossRef]
- Alomar, D.; Alarcón, M.; Kusanovic, A. Predicción de La Calidad de Lana Mediante Espectroscopia de Reflectancia En El Infrarrojo Cercano (NIRS). Agro-Sur 2015, 43, 19–24. [Google Scholar] [CrossRef]
- Gishen, M.; Cozzolino, D. Feasibility Study on the Potential of Visible and near Infrared Reflectance Spectroscopy to Measure Alpaca Fibre Characteristics. Animal 2007, 1, 899–904. [Google Scholar] [CrossRef]
- Hick, M.V.H.; Frank, E.N.; Castillo, M.F.; Prieto, A.; del Ahumada, M.R. Determination of the Textile Potential of Small Ruminants Primary (Creole) Populations in Less Favored Areas. Rev. Argentina Prod. Anim. 2016, 36, 91–102. [Google Scholar]
- Hick, M.V.H.; Lamas, H.E.; Echenique, J.; Prieto, A.; Castillo, M.F.; Frank, E.N. Estudio Demográfico de Los Atributos Morfológicos y Productivos En Poblaciones de Llamas (Lama Glama) de La Provincia de Jujuy, Argentina. Anim. Genet. Resour. Inf. 2009, 45, 71–78. [Google Scholar] [CrossRef][Green Version]
- Frank, E.; Hick, M.V.H.; Adot, O. Determination of Dehairing, Carding, Combing and Spinning Difference from Lama Type of Fleeces. Int. J. Appl. Sci. Technol. 2012, 2, 61–70. [Google Scholar]
- Quispe, M.D.; Benavidez, G.; Sauri, R.A.; Bengoechea, J.J.; Quispe, E.C. Development and Preliminary Validation of an Automatic Digital Analysis System for Animal Fibre Analysis. S. Afr. J. Anim. Sci. 2017, 47, 822–833. [Google Scholar] [CrossRef]
- Cervantes, I.; Pérez-Cabal, M.A.; Morante, R.; Burgos, A.; Salgado, C.; Nieto, B.; Goyache, F.; Gutiérrez, J.P. Genetic Parameters and Relationships between Fibre and Type Traits in Two Breeds of Peruvian Alpacas. Small Rumin. Res. 2010, 88, 6–11. [Google Scholar] [CrossRef]
- International Wool Textile Organisation (Ed.) International Wool Textile Organisation IWTO-47-2013: Measurement of the mean and distribution of fibre diameter of wool using an Optical Fibre Diameter Analyser (OFDA). In IWTO Red Book; 2013–2014; International Wool Textile Organisation: Brussels, Belgium, 2017; p. 37. [Google Scholar]
- Mark, H.; Workman, J. Effect of Repack on Calibrations Produced for Near-Infrared Reflectance Analysis. Anal. Chem. 1986, 58, 1454–1459. [Google Scholar] [CrossRef]
- Alarcón Buhofer, M.M.I. Predicción de La Calidad de Lana Mediante Espectroscopía de Reflectancia En El Infrarrojo Cercano (NIRS); Universidad Austral de Chile: Valdivia, Chile, 2012. [Google Scholar]
- Zoccola, M.; Lu, N.; Mossotti, R.; Innocenti, R.; Montarsolo, A. Identification of Wool, Cashmere, Yak, and Angora Rabbit Fibers and Quantitative Determination of Wool and Cashmere in Blend: A near Infrared Spectroscopy Study. Fibers Polym. 2013, 14, 1283–1289. [Google Scholar] [CrossRef]
- Fernández de Ahumada, E. Control de Productos y Procesos En La Industria de Fabricación de Piensos Compuestos Mediante Espectroscopía Del Infrarrojo Cercano; Universidad de Córdoba: Córdoba, España, 2008. [Google Scholar]
- Prananto, J.A.; Minasny, B.; Weaver, T. Near Infrared (NIR) Spectroscopy as a Rapid and Cost-Effective Method for Nutrient Analysis of Plant Leaf Tissues, 1st ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2020; Volume 164, ISBN 9780128207710. [Google Scholar]
- Wan, J.; Chen, Y.C.; Morris, A.J.; Thennadil, S.N. A Comparative Investigation of the Combined Effects of Pre-Processing, Wavelength Selection, and Regression Methods on Near-Infrared Calibration Model Performance. Appl. Spectrosc. 2017, 71, 1432–1446. [Google Scholar] [CrossRef] [PubMed]
- Goi, A.; Simoni, M.; Righi, F.; Visentin, G.; De Marchi, M. Application of a Handheld Near-Infrared Sprectrometer to Predict Gelatinized Starch, Fiber Fractions, and Mineral Content of Ground and Intact Extruded Dry Dog Food. Animals 2020, 10, 1660. [Google Scholar] [CrossRef] [PubMed]
- Jaconi, A.; Vos, C.; Don, A. Near Infrared Spectroscopy as an Easy and Precise Method to Estimate Soil Texture. Geoderma 2019, 337, 906–913. [Google Scholar] [CrossRef]
- Canaza-Cayo, A.W.; Cozzolino, D.; Alomar, D.; Quispe, E. A Feasibility Study of the Classification of Alpaca (Lama Pacos) Wool Samples from Different Ages, Sex and Color by Means of Visible and near Infrared Reflectance Spectroscopy. Comput. Electron. Agric. 2012, 88, 141–147. [Google Scholar] [CrossRef]
- Williams, P.; Dardenne, P.; Flinn, P. Tutorial: Items to Be Included in a Report on a near Infrared Spectroscopy Project. J. Near Infrared Spectrosc. 2017, 25, 85–90. [Google Scholar] [CrossRef]
- Laime Huarcaya, F.D.M.; Huamaní, R.P.; Ocsa, V.P.; Machaca, V.M.; Peña, E.C.Q. Características Tecnológicas de La Fibra de Llama (Lama Glama) Chaku Antes y Después de Descerdar. Rev. Investig. Vet. Peru 2016, 27, 209–217. [Google Scholar] [CrossRef]
- Quispe Peña, E.; Gutiérrez Poma, A.; Unanua Purroy, A. Características Productivas y Textiles de La Fibras de Alpacas de Raza Huacaya. Rev. Complut. Ciencias Vet. 2013, 7, 1–29. [Google Scholar] [CrossRef]
- Zoccola, M.; Mossotti, R.; Innocenti, R.; Loria, D.I.; Rosso, S.; Zanetti, R.; Group, H.W. A New Approach in the Determination of Eumelanin in Human Hair Using near Infrared Spectroscopy. NIR News 2005, 16, 10–12. [Google Scholar] [CrossRef]
- Workman, J.; Weyer, L. Practical Guide to Interpretive Near-Infrared Spectroscopy, 1st ed.; CRC Press: Boca Ratón, FL, USA, 2008; ISBN 9781574447842. [Google Scholar]
- Frank, E.N.; Hick, M.V.H. Comercialización de Fibras de Camélidos Sudamericanos; Universidad Católica de Córdoba: Córdoba, Argentina, 2017; Volume 5. [Google Scholar]
- Frank, E.N.; Hick, M.V.H.; Molina, M.G.; Caruso, L.M. Genetic Parameters for Fleece Weight and Fibre Attributes in Argentinean Llamas Reared Outside the Altiplano. Small Rumin. Res. 2011, 99, 54–60. [Google Scholar] [CrossRef]
- Wurzinger, M.; Delgado, J.; Nürnberg, M.; Valle Zárate, A.; Stemmer, A.; Ugarte, G.; Sölkner, J. Genetic and Non-Genetic Factors Influencing Fibre Quality of Bolivian Llamas. Small Rumin. Res. 2006, 61, 131–139. [Google Scholar] [CrossRef]
- Frank, E.N. El Confort de Los Tejidos Confeccionados Con Fibra de Camélidos Sudamericanos. In Proceedings of the VI Congreso Mundial de Camélidos; Raggi, L.A., Rojas, I., Parraguez, V.H., Sepúlveda, N., Eds.; Universidad de Chile: Arica, Chile, 2012; pp. 65–69. [Google Scholar]
- Amorena, J.I.; de Ahumada, E.F.; Álvarez, D.M.E.; Rigalt, F. Desarrollo Preliminar de Metodología NIRS Para La Evaluación Cuantitativa y Cualitativa de Fibra de Llama (Lama Glama). In Proceedings of the 8vo Congreso Mundial sobre Camélidos; Delgado Callisaya, P., Gutiérrez Ramirez, L., Bustos Fernández, N., Eds.; Ministerio de Desarrollo Rural y Tierras: Oruro, Bolivia, 2018; p. 2. [Google Scholar]
- Cozzolino, D.; Montossi, F.; San Julian, R. The Use of Visible (VIS) and near Infrared (NIR) Reflectance Spectroscopy to Predict Fibre Diameter in Both Clean and Greasy Wool Samples. Anim. Sci. 2005, 80, 333–337. [Google Scholar] [CrossRef]
- McGregor, B.A.; Quispe Peña, E.C. Cuticle and Cortical Cell Morphology of Alpaca and Other Rare Animal Fibres. J. Text. Inst. 2018, 109, 767–774. [Google Scholar] [CrossRef]
- Recena, R.; Fernández-Cabanás, V.M.; Delgado, A. Soil Fertility Assessment by Vis-NIR Spectroscopy: Predicting Soil Functioning Rather than Availability Indices. Geoderma 2019, 337, 368–374. [Google Scholar] [CrossRef]
- Cozzolino, D. Wet or Dry? The Challenges of NIR to Analyse Soil Samples. NIR News 2017, 28, 3–5. [Google Scholar] [CrossRef]
- Su, H.; Zhang, S.; Li, H.; Xie, P.; Sun, B. Using Near-Infrared Reflectance Spectroscopy to Predict Physical Parameters of Beef. Spectrosc. Lett. 2018, 51, 163–168. [Google Scholar] [CrossRef]
| Reference Variables | Mean± SD | Minimum | Maximum | CV |
|---|---|---|---|---|
| MFD (µm) | 25.16 ± 3.75 | 17.82 | 37.71 | 14.93 |
| SDMFD (µm) | 8.43 ± 1.83 | 5.12 | 13.62 | 21.76 |
| CVMFD (%) | 33.46 ± 4.03 | 22.7 | 45.7 | 12.05 |
| CF (%) | 79.03 ± 13.21 | 24.4 | 97.3 | 16.71 |
| Model ID | Sample Treatment 1 | Spectral Range | Pretreatments | Loadings | R2 2 | SECV 3 | SEV 4 | RPD5 | |
|---|---|---|---|---|---|---|---|---|---|
| Multiplicative | Derivative | ||||||||
| 01 | Control A | Vis–NIR | NONE | 0-0-1-1 | 5 | 0.53 | 2.372 | 2.672 | 1.58 |
| 10 | NIR | NONE | 0-0-1-1 | 5 | 0.59 | 2.236 | 2.547 | 1.68 | |
| 19 | Discrete | NONE | 0-0-1-1 | 5 | 0.57 | 2.286 | 2.504 | 1.64 | |
| 29 | Carded B | Vis–NIR | NONE | 1-5-3-1 | 5 | 0.67 | 1.965 | 2.235 | 1.91 |
| 37 | NIR | NONE | 0-0-1-1 | 8 | 0.68 | 2.088 | 2.067 | 1.80 | |
| 46 | Discrete | NONE | 0-0-1-1 | 7 | 0.64 | 2.210 | 2.007 | 1.70 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amorena, J.I.; Álvarez, D.M.E.; Fernández-Ahumada, E. Development of Calibration Models to Predict Mean Fibre Diameter in Llama (Lama glama) Fleeces with Near Infrared Spectroscopy. Animals 2021, 11, 1998. https://doi.org/10.3390/ani11071998
Amorena JI, Álvarez DME, Fernández-Ahumada E. Development of Calibration Models to Predict Mean Fibre Diameter in Llama (Lama glama) Fleeces with Near Infrared Spectroscopy. Animals. 2021; 11(7):1998. https://doi.org/10.3390/ani11071998
Chicago/Turabian StyleAmorena, José Ignacio, Dolores María Eugenia Álvarez, and Elvira Fernández-Ahumada. 2021. "Development of Calibration Models to Predict Mean Fibre Diameter in Llama (Lama glama) Fleeces with Near Infrared Spectroscopy" Animals 11, no. 7: 1998. https://doi.org/10.3390/ani11071998
APA StyleAmorena, J. I., Álvarez, D. M. E., & Fernández-Ahumada, E. (2021). Development of Calibration Models to Predict Mean Fibre Diameter in Llama (Lama glama) Fleeces with Near Infrared Spectroscopy. Animals, 11(7), 1998. https://doi.org/10.3390/ani11071998






