Magnetic Compass Orientation in a Palaearctic–Indian Night Migrant, the Red-Headed Bunting
Abstract
Simple Summary
Abstract
1. Introduction
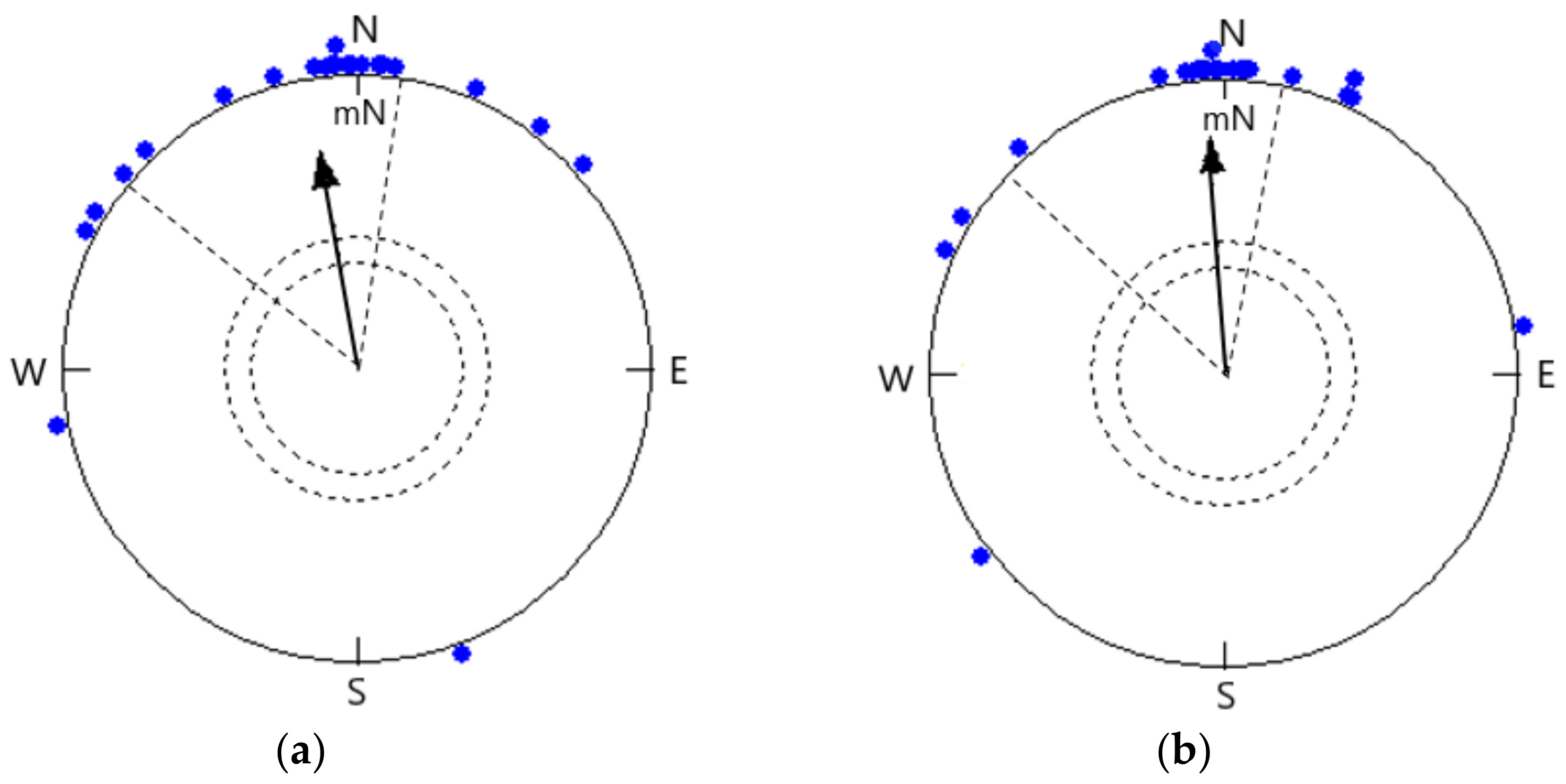
- (a)
- Open skies when celestial cues (sunset, horizon glow, and skylight polarization patterns) were visible to the songbirds in the natural magnetic field.
- (b)
- Simulated overcast skies in the natural magnetic field with no celestial cues visible.
- (c)
- The magnetic field shifted −120° under simulated overcast skies.
- (d)
- The effect of different temperatures (22 °C and 38 °C) on orientation, tested under simulated overcast skies.
2. Materials and Methods
2.1. Study Species and Animal Husbandry
2.2. Experimental Procedure
2.3. Helmholtz Coil System
2.4. Experiment 1
- (a)
- Clear sky condition: Twenty-two birds, each tested four times in the natural magnetic field under the open sky. Sunset and horizon glow was clearly visible to the birds.
- (b)
- Simulated overcast condition: The same birds were tested under simulated overcast skies in rooms (dimension = 4 m × 4 m × 3 m) in the natural magnetic field. No celestial cues were visible to the birds. The simulated overcast sky was made using translucent plexiglass sheets of 3 mm thickness that allow natural light to pass through them.
- (c)
- −120° rotated magnetic field under simulated overcast condition: The same 22 birds were tested under simulated overcast skies in rooms (dimension = 4 m × 4 m × 3 m) in a 120° counter-clockwise rotated magnetic field with the help of the Helmholtz coil system. Under this experiment, no celestial cues were visible to the birds. Helmholtz coil apparatus HCS01CL of 2 m × 2 m × 2 m was used to rotate the horizontal component of the magnetic field. The experimental birds were exposed to a deflected magnetic field (−120°) indoors under the simulated overcast sky in orientation funnels for a period of 60 minutes.
2.5. Experiment 2
2.6. Statistics
3. Results
3.1. Orientation in the Local Magnetic Field
3.2. Orientation in a −120° Rotated Magnetic Field under Simulated Overcast Skies
3.3. Orientation of Birds Tested at 22 °C and 38 °C in the Natural Magnetic Field under Simulated Overcast Skies
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wiltschko, W.; Wiltschko, R. Magnetic Compass of European Robins. Science 1972, 176, 62–64. [Google Scholar] [CrossRef] [PubMed]
- Wiltschko, R.; Wiltschko, W. Magnetic Orientation in Animals; Zoophysiology; Springer: Berlin/Heidelberg, Germany, 1995; ISBN 978-3-642-79751-4. [Google Scholar]
- Mouritsen, H. Redstarts, Phoenicurus Phoenicurus, Can Orient in a True-Zero Magnetic Field. Anim. Behav. 1998, 55, 1311–1324. [Google Scholar] [CrossRef] [PubMed]
- Cochran, W.W.; Mouritsen, H.; Wikelski, M. Migrating Songbirds Recalibrate Their Magnetic Compass Daily from Twilight Cues. Science 2004, 304, 405–408. [Google Scholar] [CrossRef]
- Wiltschko, R.; Wiltschko, W. Avian Navigation: A Combination of Innate and Learned Mechanisms. Adv. Study Behav. 2015, 47, 229–310. [Google Scholar] [CrossRef]
- Hore, P.J.; Mouritsen, H. The Radical-Pair Mechanism of Magnetoreception. Annu. Rev. Biophys. 2016, 45, 299–344. [Google Scholar] [CrossRef]
- Mouritsen, H. Long-Distance Navigation and Magnetoreception in Migratory Animals. Nature 2018, 558, 50–59. [Google Scholar] [CrossRef] [PubMed]
- Sauer, F. Die Sternenorientierung Nächtlich Ziehender Grasmücken (Sylvia Atricapilla, Borin Und Curruca). Z. Tierpsychol. 1957, 14, 20–70. [Google Scholar] [CrossRef]
- Emlen, S.T. Celestial Rotation: Its Importance in the Development of Migratory Orientation. Science 1970, 170, 1198–1201. [Google Scholar] [CrossRef]
- Emlen, S.T. The Stellar-Orientation System of a Migratory Bird. Sci. Am. 1975, 233, 102–111. [Google Scholar] [CrossRef]
- Bingman, V.P. Night Sky Orientation of Migratory Pied Flycatchers Raised in Different Magnetic Fields. Behav. Ecol. Sociobiol. 1984, 15, 77–80. [Google Scholar] [CrossRef]
- Wiltschko, W.; Daum, P.; Fergenbauer-Kimmel, A.; Wiltschko, R. The Development of the Star Compass in Garden Warblers, Sylvia Borin. Ethology 1987, 74, 285–292. [Google Scholar] [CrossRef]
- Mouritsen, H.; Larsen, O.N. Migrating Songbirds Tested in Computer-Controlled Emlen Funnels Use Stellar Cues for a Time-Independent Compass. J. Exp. Biol. 2001, 204, 3855–3865. [Google Scholar] [CrossRef]
- Michalik, A.; Alert, B.; Engels, S.; Lefeldt, N.; Mouritsen, H. Star Compass Learning: How Long Does It Take? J. Ornithol. 2013, 155, 225–234. [Google Scholar] [CrossRef]
- Able, K.P. Skylight Polarization Patterns at Dusk Influence Migratory Orientation in Birds. Nature 1982, 299, 550–551. [Google Scholar] [CrossRef]
- Moore, F.R. Sunset and the Orientation Behaviour of Migrating Birds. Biol. Rev. 1987, 62, 65–86. [Google Scholar] [CrossRef]
- Moore, F.R.; Phillips, J.B. Sunset, Skylight Polarization and the Migratory Orientation of Yellow-Rumped Warblers, Dendroica Coronata. Anim. Behav. 1988, 36, 1770–1778. [Google Scholar] [CrossRef]
- Schmidt-Koenig, K. The Sun Compass. Experientia 1990, 46, 336–342. [Google Scholar] [CrossRef]
- Helbig, A.J. Dusk Orientation of Migratory European Robins, Erithacus Rubecula: The Role of Sun-Related Directional Information. Anim. Behav. 1991, 41, 313–322. [Google Scholar] [CrossRef]
- Phillips, J.B.; Moore, F.R. Calibration of the Sun Compass by Sunset Polarized Light Patterns in a Migratory Bird. Behav. Ecol. Sociobiol. 1992, 31, 189–193. [Google Scholar] [CrossRef]
- Muheim, R.; Phillips, J.B.; Åkesson, S. Polarized Light Cues Underlie Compass Calibration in Migratory Songbirds. Science 2006, 313, 837–839. [Google Scholar] [CrossRef]
- Mettke-Hofmann, C.; Gwinner, E. Long-Term Memory for a Life on the Move. Proc. Natl. Acad. Sci. USA 2003, 100, 5863–5866. [Google Scholar] [CrossRef] [PubMed]
- Viguier, C. Le Sens de l’orientation et Ses Organes Chez Les Animaux et Chez l’homme. Rev. Philos. Fr. Etranger 1882, 14, 1–36. [Google Scholar]
- Skiles, D.D. The Geomagnetic Field Its Nature, History, and Biological Relevance. In Magnetite Biomineralization and Magnetoreception in Organisms; Kirschvink, J.L., Jones, D.S., MacFadden, B.J., Eds.; Topics in Geobiology; Springer US: Boston, MA, USA, 1985; Volume 5, pp. 43–102. ISBN 978-1-4613-7992-8. [Google Scholar]
- Wiltschko, W. Further Analysis of the Magnetic Compass of Migratory Birds. In Animal Migration, Navigation, and Homing; Schmidt-Koenig, K., Keeton, W.T., Eds.; Springer: Berlin/Heidelberg, Germany, 1978; pp. 302–310. [Google Scholar]
- Kishkinev, D.; Packmor, F.; Zechmeister, T.; Winkler, H.-C.; Chernetsov, N.; Mouritsen, H.; Holland, R.A. Navigation by Extrapolation of Geomagnetic Cues in a Migratory Songbird. Curr. Biol. 2021, S0960982221001160. [Google Scholar] [CrossRef]
- Schulten, K.; Swenberg, C.E.; Weller, A. A Biomagnetic Sensory Mechanism Based on Magnetic Field Modulated Coherent Electron Spin Motion. Z. Phys. Chem. 1978, 111, 1–5. [Google Scholar] [CrossRef]
- Ritz, T.; Adem, S.; Schulten, K. A Model for Photoreceptor-Based Magnetoreception in Birds. Biophys. J. 2000, 78, 707–718. [Google Scholar] [CrossRef]
- Rodgers, C.T.; Hore, P.J. Chemical Magnetoreception in Birds: The Radical Pair Mechanism. Proc. Natl. Acad. Sci. USA 2009, 106, 353–360. [Google Scholar] [CrossRef]
- Wiltschko, R.; Stapput, K.; Thalau, P.; Wiltschko, W. Directional Orientation of Birds by the Magnetic Field under Different Light Conditions. J. R. Soc. Interface 2010, 7, S163–S177. [Google Scholar] [CrossRef]
- Solov’yov, I.A.; Mouritsen, H.; Schulten, K. Acuity of a Cryptochrome and Vision-Based Magnetoreception System in Birds. Biophys. J. 2010, 99, 40–49. [Google Scholar] [CrossRef]
- Mouritsen, H.; Hore, P.J. The Magnetic Retina: Light-Dependent and Trigeminal Magnetoreception in Migratory Birds. Curr. Opin. Neurobiol. 2012, 22, 343–352. [Google Scholar] [CrossRef]
- Mouritsen, H.; Feenders, G.; Liedvogel, M.; Wada, K.; Jarvis, E.D. Night-Vision Brain Area in Migratory Songbirds. Proc. Natl. Acad. Sci. USA 2005, 102, 8339–8344. [Google Scholar] [CrossRef]
- Zapka, M.; Heyers, D.; Hein, C.M.; Engels, S.; Schneider, N.-L.; Hans, J.; Weiler, S.; Dreyer, D.; Kishkinev, D.; Wild, J.M.; et al. Visual but Not Trigeminal Mediation of Magnetic Compass Information in a Migratory Bird. Nature 2009, 461, 1274–1277. [Google Scholar] [CrossRef]
- Löhrl, H. Zur Frage Des Zeitpunkts Einer Prägung Auf Die Heimatregion Beim Halsbandschnäpper (Ficedula Albicollis). J. Orn. 1959, 100, 132–140. [Google Scholar] [CrossRef]
- Sokolov, L.V.; Bolshakov, K.V.; Vinogradova, N.V.; Dotnik, T.V.; Lyuleeva, D.S.; Payesky, V.A.; Shumakov, M.E.; Yablonkevich, M.L. The Testing of the Ability for Imprinting and Finding the Site of Future Nesting in Young Chaffinches (in Russian). Acad. Sci. USSR Zool. J. 1984, 63, 1671–1681. [Google Scholar]
- Alerstam, T. Ecological Causes and Consequences of Bird Orientation. In Orientation in Birds; Berthold, P., Ed.; Experientia Supplementum; Birkhäuser: Basel, Switzerland, 1991; pp. 202–225. ISBN 978-3-0348-7208-9. [Google Scholar]
- Dawson, A. Control of the Annual Cycle in Birds: Endocrine Constraints and Plasticity in Response to Ecological Variability. Philos. Trans. R. Soc. B Biol. Sci. 2008, 363, 1621–1633. [Google Scholar] [CrossRef]
- Cochran, W.W.; Wikelski, M. Individual Migratory Tactics of New World Catharus Thrushes: Current Knowledge and Future Tracking Options from Space; Johns Hopkins University Press: Baltimore, MD, USA, 2005; pp. 274–289. ISBN 978-0-8018-8107-7. [Google Scholar]
- Rubolini, D.; Massi, A.; Spina, F. Replacement of Body Feathers Is Associated with Low Pre-Migratory Energy Stores in a Long-Distance Migratory Bird, the Barn Swallow (Hirundo Rustica). J. Zool. 2002, 258, 441–447. [Google Scholar] [CrossRef]
- Stutchbury, B.J.M.; Tarof, S.A.; Done, T.; Gow, E.; Kramer, P.M.; Tautin, J.; Fox, J.W.; Afanasyev, V. Tracking Long-Distance Songbird Migration by Using Geolocators. Science 2009, 323, 896. [Google Scholar] [CrossRef]
- Able, K.P. Skylight Polarization Patterns and the Orientation of Migratory Birds. J. Exp. Biol. 1989, 141, 241–256. [Google Scholar] [CrossRef]
- Sjöberg, S.; Alerstam, T.; Akesson, S.; Muheim, R. Ecological Factors Influence Timing of Departures in Nocturnally Migrating Songbirds at Falsterbo, Sweden. Anim. Behav. 2017, 127, 253–269. [Google Scholar] [CrossRef]
- Nilsson, C.; Klaassen, R.; Alerstam, T. Differences in Speed and Duration of Bird Migration between Spring and Autumn. Am. Nat. 2013, 181, 837–845. [Google Scholar] [CrossRef]
- Mitrus, C.; Sparks, T.; Tryjanowski, P. First Evidence of Phenological Change in a Transcontinental Migrant Overwintering in the Indian Sub-Continent: The Red-breasted Flycatcher Ficedula Parva. Ornis Fenn. 2005, 82, 13–19. [Google Scholar]
- Ali, S.; Ripley, S.D. Handbook of the Birds of India and Pakistan, Together with Those of Bangladesh, Nepal, Bhutan, and Sri Lanka; Oxford University Press: New Delhi, India, 1980; ISBN 978-0-19-561201-1. [Google Scholar]
- Olsson, U.; Byers, C.; Curson, J. Buntings and Sparrows; Bloomsbury Publishing Plc.: London, UK, 2013; ISBN 978-1-4081-8906-1. [Google Scholar]
- Morbey, Y.E.; Ydenberg, R.C. Protandrous Arrival Timing to Breeding Areas: A Review. Ecol. Lett. 2001, 4, 663–673. [Google Scholar] [CrossRef]
- Malik, S.; Rani, S.; Kumar, V. Wavelength Dependency of Light-Induced Effects on Photoperiodic Clock in the Migratory Blackheaded Bunting (Emberiza Melanocephala). Chronobiol. Int. 2004, 21, 367–384. [Google Scholar] [CrossRef]
- Åkesson, S. Effect of Geomagnetic Field on Orientation of the Marsh Warbler, Acrocephalus Palustris, in Sweden and Kenya. Anim. Behav. 1993, 46, 1157–1167. [Google Scholar] [CrossRef]
- Åkesson, S.; Alerstam, T.; Hedenström, A. Flight Initiation of Nocturnal Passerine Migrants in Relation to Celestial Orientation Conditions at Twilight. J. Avian Biol. 1996, 27, 95–102. [Google Scholar] [CrossRef]
- Emlen, S.T.; Emlen, J.T. A Technique for Recording Migratory Orientation of Captive Birds. Auk 1966, 83, 361–367. [Google Scholar] [CrossRef]
- Helbig, A.J.; Berthold, P.; Wiltschko, W. Migratory Orientation of Blackcaps (Sylvia Atricapilla): Population-Specific Shifts of Direction during the Autumn. Ethology 1989, 82, 307–315. [Google Scholar] [CrossRef]
- Muheim, R.; Henshaw, I.; Sjöberg, S.; Deutschlander, M. BirdOriTrack: A New Video-Tracking Program for Orientation Research with Migratory Birds. J. Field Ornithol. 2014, 85, 91–105. [Google Scholar] [CrossRef]
- Sandberg, R.; Pettersson, J. Magnetic Orientation of Snow Buntings (Plectrophenax Nivalis), a Species Breeding in the High Arctic: Passage Migration through Temperate-Zone Areas. J. Exp. Biol. 1996, 199, 1899–1905. [Google Scholar] [CrossRef]
- Merkel, F.W.; Wiltschko, W. Magnetismus Und Richtungsfinden Zugunruhiger Rotkehlchen (Erithacus Rubecula). Vogelwarte 1965, 23, 71–77. [Google Scholar]
- Helbig, A.J. Depolarization of Natural Skylight Disrupts Orientation of an Avian Nocturnal Migrant. Experientia 1990, 46, 755–758. [Google Scholar] [CrossRef]
- Sandberg, R.; Ottosson, U.; Pettersson, J. Magnetic Orientation of Migratory Wheatears (Oenanthe Oenanthe) in Sweden and Greenland. J. Exp. Biol. 1991, 155, 51–64. [Google Scholar] [CrossRef]
- Åkesson, S.; Ottosson, U.; Sandberg, R. Bird Orientation: Displacement Experiments with Young Autumn Migrating Wheatears, Oenanthe Oenanthe, along the Arctic Coast of Russia. Proc. R. Soc. Lond. B Biol. Sci. 1995, 262, 189–195. [Google Scholar] [CrossRef]
- Sandberg, R.; Bäckman, J.; Ottosson, U. Orientation of Snow Buntings (Plectrophenax Nivalis) Close to the Magnetic North Pole. J. Exp. Biol. 1998, 201, 1859–1870. [Google Scholar] [CrossRef] [PubMed]
- Gudmundsson, G.A.; Sandberg, R. Sanderlings (Calidris Alba) Have a Magnetic Compass: Orientation Experiments during Spring Migration in Iceland. J. Exp. Biol. 2000, 203, 3137–3144. [Google Scholar] [CrossRef]
- Åkesson, S.; Morin, J.; Muheim, R.; Ottosson, U. Avian Orientation at Steep Angles of Inclination: Experiments with Migratory White–Crowned Sparrows at the Magnetic North Pole. Proc. R. Soc. Lond. B Biol. Sci. 2001, 268, 1907–1913. [Google Scholar] [CrossRef]
- Muheim, R.; Åkesson, S. Clock-Shift Experiments with Savannah Sparrows, Passerculus Sandwichensis, at High Northern Latitudes. Behav. Ecol. Sociobiol. 2002, 51, 394–401. [Google Scholar] [CrossRef]
- Gauthreaux, S.A. A Radar and Direct Visual Study of Passerine Spring Migration in Southern Louisiana. Auk 1971, 88, 343–365. [Google Scholar] [CrossRef]
- Wiltschko, W.; Gwinner, E. Evidence for an Innate Magnetic Compass in Garden Warblers. Naturwissenschaften 1974, 61, 406. [Google Scholar] [CrossRef]
- Beck, W.; Wiltschko, W. The Magnetic Field as a Reference System for Genetically Encoded Migratory Direction in Pied Flycatchers (Ficedula Hypoleuca Pallas). Z. Tierpsychol. 1982, 60, 41–46. [Google Scholar] [CrossRef]
- Bingman, V.P. Importance of Earth’s Magnetism for the Sunset Orientation of Migratory Naive Savannah Sparrows. Ital. J. Zool. 1983, 17, 395–400. [Google Scholar] [CrossRef]
- Shumakov, M.E. The development of orientation capabilities of young night-migrants under natural and experimental conditions. Baltic Birds 1990, 5, 146–149. [Google Scholar]
- Schneider, T.; Thalau, H.P.; Semm, P.; Wiltschko, W. Melatonin Is Crucial for the Migratory Orientation of Pied Flycatchers (Ficedula Hypoleuca Pallas). J. Exp. Biol. 1994, 194, 255–262. [Google Scholar] [CrossRef]
- Bletz, H.; Weindler, P.; Wiltschko, R.; Wiltschko, W.; Berthold, P. The Magnetic Field as Reference for the Innate Migratory Direction in Blackcaps, Sylvia Atricapilla. Naturwissenschaften 1996, 83, 430–432. [Google Scholar] [CrossRef]
- Wiltschko, W.; Gesson, M.; Wiltschko, R. Magnetic Compass Orientation of European Robins under 565 Nm Green Light. Naturwissenschaften 2001, 88, 387–390. [Google Scholar] [CrossRef]
- Engels, S.; Schneider, N.-L.; Lefeldt, N.; Hein, C.M.; Zapka, M.; Michalik, A.; Elbers, D.; Kittel, A.; Hore, P.J.; Mouritsen, H. Anthropogenic Electromagnetic Noise Disrupts Magnetic Compass Orientation in a Migratory Bird. Nature 2014, 509, 353–356. [Google Scholar] [CrossRef]
- Wiltschko, W. Über Den Einfluß Statischer Magnetfelder Auf Die Zugorientierung Der Rotkehlchen (Erithacus Rubecula). Z. Tierpsychol. 1968, 25, 537–558. [Google Scholar] [CrossRef]
- Wiltschko, W. The influence of magnetic total intensity and inclination on directions chosen by migrating European Robins. In Animal Orientation and Navigation; NASA SP-262; U.S. Goverment Publishing Office: Washington, DC, USA, 1972; pp. 569–578. [Google Scholar]
- Wiltschko, W.; Höck, H.; Merkel, F.W. Outdoor Experiments with Migrating European Robins in Artificial Magnetic Fields. Z. Tierpsychol. 1971, 29, 409–415. [Google Scholar] [CrossRef]
- Wiltschko, W.; Wiltschko, R. The Interaction of Stars and Magnetic Field in the Orientation System of Night Migrating Birds. II. Spring Experiments with European Robins (Erithacus Rubecula). Z. Tierpsychol. 1975, 39, 265–282. [Google Scholar] [CrossRef]
- Wiltschko, W.; Wiltschko, R. The Interaction of Stars and Magnetic Field in the Orientation System of Night Migrating Birds. I. Autumn Experiments with European Warblers (Gen. Sylvia). Z. Tierpsychol. 1975, 37, 337–355. [Google Scholar] [CrossRef]
- Bingman, V.P. Earth’s Magnetism and the Nocturnal Orientation of Migratory European Robins. Auk 1987, 104, 523–525. [Google Scholar] [CrossRef]
- Bingman, V.P.; Wiltschko, W. Orientation of Dunnocks (Prunella Modularis) at Sunset. Ethology 1988, 77, 1–9. [Google Scholar] [CrossRef]
- Quay, T.L. The Savannah Sparrow (Passerculus Sandwichensis Gmelin) in Winter in the Lower Piedmont of North Carolina. J. Elisha Mitchell Sci. Soc. 1957, 73, 378–388. [Google Scholar]
- Norris, R.A.; Hight, G.L. Subspecific Variation in Winter Populations of Savannah Sparrows: A Study in Field Taxonomy. Condor 1957, 59, 40–52. [Google Scholar] [CrossRef]
- Parslow, J.L.F. The Migration of Passerine Night Migrants across the English Channel Studied by Radar. Ibis 1969, 111, 48–79. [Google Scholar] [CrossRef]
- Metcalfe, J.; Schmidt, K.L.; Kerr, W.B.; Guglielmo, C.G.; MacDougall-Shackleton, S.A. White-Throated Sparrows Adjust Behaviour in Response to Manipulations of Barometric Pressure and Temperature. Anim. Behav. 2013, 6, 1285–1290. [Google Scholar] [CrossRef]
- Bauer, S.; Gienapp, P.; Madsen, J. The Relevance of Environmental Conditions for Departure Decision Changes En Route in Migrating Geese. Ecology 2008, 89, 1953–1960. [Google Scholar] [CrossRef] [PubMed]
- Wikelski, M.; Tarlow, E.M.; Raim, A.; Diehl, R.H.; Larkin, R.P.; Visser, G.H. Costs of Migration in Free-Flying Songbirds. Nature 2003, 423, 704. [Google Scholar] [CrossRef]
- Hüppop, O.; Winkel, W. Climate Change and Timing of Spring Migration in the Long-Distance Migrant Ficedula Hypoleuca in Central Europe. J. Ornithol. 2006, 147, 344–353. [Google Scholar] [CrossRef]
- Marra, P.P.; Francis, C.M.; Mulvihill, R.S.; Moore, F.R. The Influence of Climate on the Timing and Rate of Spring Bird Migration. Oecologia 2005, 142, 307–315. [Google Scholar] [CrossRef]
- Ahola, M.; Laaksonen, T.; Sippola, K.; Eeva, T.; Rainio, K.; Lehikoinen, E. Variation in Climate Warming Uncouples Arrival and Breeding Dates. Glob. Change Biol. 2004, 10, 1610–1617. [Google Scholar] [CrossRef]
- Dawson, A.; Visser, M.E. The Effects of Temperature on Photoperiodic Responses: Implications for Climate Change. In Proceedings of the BOU Climate Change and Birds Conference, Leicester, UK, 6–8 April 2010. [Google Scholar]
- Trivedi, A.; Sur, S.; Sharma, A.; Taufique, S.K.; Gupta, N.; Kumar, V. Temperature Alters the Hypothalamic Transcription of Photoperiod Responsive Genes in Induction of Seasonal Response in Migratory Redheaded Buntings. Mol. Cell. Endocrinol. 2019, 493, 110454. [Google Scholar] [CrossRef]
- Bridge, E.; Kelly, J.; Contina, A.; Gabrielson, R.; MacCurdy, R.; Winkler, D. Advances in Tracking Small Migratory Birds: A Technical Review of Light-Level Geolocation. J. Field Ornithol. 2013, 84, 121–137. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tyagi, T.; Bhardwaj, S.K. Magnetic Compass Orientation in a Palaearctic–Indian Night Migrant, the Red-Headed Bunting. Animals 2021, 11, 1541. https://doi.org/10.3390/ani11061541
Tyagi T, Bhardwaj SK. Magnetic Compass Orientation in a Palaearctic–Indian Night Migrant, the Red-Headed Bunting. Animals. 2021; 11(6):1541. https://doi.org/10.3390/ani11061541
Chicago/Turabian StyleTyagi, Tushar, and Sanjay Kumar Bhardwaj. 2021. "Magnetic Compass Orientation in a Palaearctic–Indian Night Migrant, the Red-Headed Bunting" Animals 11, no. 6: 1541. https://doi.org/10.3390/ani11061541
APA StyleTyagi, T., & Bhardwaj, S. K. (2021). Magnetic Compass Orientation in a Palaearctic–Indian Night Migrant, the Red-Headed Bunting. Animals, 11(6), 1541. https://doi.org/10.3390/ani11061541





