1. Introduction
Consumers are increasingly becoming concerned about healthy and safe products, and the demand for these products is escalating [
1]. Consumers expect the meat products on the market to have the required nutritional values, to be wholesome; fresh; lean and have adequate juiciness, flavor and tenderness and be harvested with a high animal welfare standard [
2,
3,
4]. Game and venison meat are, nowadays, perceived by some categories of consumers as more animal welfare-friendly, because hunted game meat is considered more ethically justifiable than farmed meat, since the animals roam free until the moment of harvest [
5]. A recent study proved that German consumers strongly prefer hunting to intensive farming [
6].
Boar hunting is mainly divided into two categories: (i) collective hunting, consisting of the presence of a large number of participants with drive hunts, and (ii) individual hunting, carried out by a single hunter or by a number not exceeding three by stalking or posting [
7]. Both rifled-bore and smooth-bore weapons can be used for wild boar hunting. In addition to the metal sights, the weapon can be set with red dot or telescope attachments. For smooth-bore weapons, only 12- or 20-gauge dry bullet ammunitions are allowed. These guns can be both swinging (like shotguns) and fixed (like semi-automatic). Instead, for rifled-core weapons, there are various models such as: bolt-action, express, falling block, lever and semi-automatic. The recommended caliber of the latter is from 7 mm upwards. The weapon of choice for wild boar hunting depends on the type of hunt and the local regulations [
8].
Once bled, eviscerated and cooled, the carcass must be sent to a point where it can be sectioned or kept refrigerated for maturation. The precautions to be observed during this phase are few and simple but essential: protect the meat and start the cooling process [
9]. After preparing the animal (bleeding, evisceration and cooling), it may be necessary to face a period of transport that is sometimes long and not very easy; while small animals can be easily transported in a backpack, larger ones must necessarily be carried by several people or sometimes dragged to the vehicle capable of transporting them. It is obvious that a wild boar opened from the jaws to the pelvis and dragged for hundreds of meters will tend to be contaminated with blades of grass and, sometimes, earth; however, these contaminations are to be considered secondary in the preservation of the meat, compared to the contamination by the intestinal contents, and can be easily eliminated with a simple washing on arrival at home [
9]. The practice of not opening the thorax of a prey before arriving home (the abdomen is still opened due to weight problems during transport) is instead absolutely wrong, as it hinders the cooling of the meat and can favor the exit of liquids from the esophagus, liquids capable of contaminating the meat due to their bacterial components. Of the two types of meat contamination, ground contamination is much more tolerable, but it is still preferable to avoid it; it will be possible to have a plasticized sheet to tie around the torso of the animal, which, in the case of not excessively uneven terrain, as well as protecting the carcass, will facilitate its sliding [
10]. For our areas, among the animals that cause the greatest transport problems, due to their size wild boars certainly apply. As soon as you arrive at a motorized vehicle capable of transporting the carcass, it is necessary to remember that this is used to transport the prey as soon as possible to a suitable place to start the cooling process (cold room or cold cellar) and not to transport the hunters in a suitable place to celebrate the capture, forgetting the prey perhaps under the sun; it is, in fact, essential to be able to quickly put the meat away from possible alterations [
9,
10]. Once at home, the animal is hung, washed on the inside of the carcass with plenty of water to clean off the dirt and blood that will pour out during the transport without wetting the hair and leave it hanging to drain off; once drained and dried (be careful not to put water where it is unable to drain—for example, inside the wounds), the carcass will be ready to pass into maturation and subsequent sectioning. During the draining and drying phases, it would be good to keep the chest of the carcass open with a special retractor (wooden stick) in order to favor its internal drying and, also, keep it in place in the cell, until cooling has taken place. It is necessary to remember the role of the external temperature in the processing and conservation of meat; the storage temperatures of the meat are 3–7 °C, but the cells are generally set at −1 °C, as, at this temperature, the meat does not freeze, so, if we have external temperatures of 18–20 °C (in the summer or autumn) and a cool cellar at 8 °C, this will be suitable to favor cooling but not suitable for the preservation of the carcass, which, if stored in that place, will tend to rot rapidly.
Refrigeration is done in cold rooms, at temperatures ranging from −1 to 3 °C. The cold acts on the meat by slowing down both the degradation processes of its nutrients, proteins and fats, in particular, and the proliferation of germs and moulds, which, in a more-or-less abundant way, contaminate the carcass. It is clear that, to obtain prolonged conservation, the cooling must be carried out as quickly as possible after recovery of the carcass and after the latter has undergone the practices of bleeding and evisceration, scrupulously observing the hygiene rules. This is of the utmost importance, because the bacteria that contaminate the meat in these phases drastically reduces its shelf life, due to the fact that many bacteria have adapted to survive in the soil and surface waters of cold areas, and so, they replicate well, albeit more slowly, at refrigeration conditions [
11]. Furthermore, low temperatures also slow down the activities of the enzymes responsible for maturation, which will require longer times for their development. In addition to the temperature, humidity and air speed must also be considered for the purposes of maturation inside the refrigerator. These factors, if well-regulated, contribute to keep the bacterial population low on the surface and, at the same time, favor the maturation of the meat in depth. Refrigeration therefore performs two functions at the same time: it prolongs the life of the meat by promoting its maturation. It is clear that this method preserves the meat for a limited period of time, which, for furred game, hardly exceeds 10–15 days (in larger animals). The bacteria that contaminate the meat after the death of the animal can come from multiple sources, such as the animal itself, the external environment and the operator who handles it [
11]. Perishable foods such as meats can be easily polluted, so it is important to strictly observe the hygiene rules to avoid bringing in bacterial colonies capable of drastically reducing its storage time (spoilage bacteria, e.g.,
Pseudomonas spp.) or even make it dangerous for the health of the consumer (pathogenic bacteria, e.g.,
Salmonella [
12,
13,
14]. The speed with which game meat, from the initial contamination, undergoes the alterative process of putrefaction depends to a decisive extent of the type of bacteria present and their quantity, as well as on the atmospheric temperature, which can influence their multiplication in different ways [
11].
In an attempt to investigate the effects of delayed refrigeration on wild boar carcass contamination, the aim of this work was to determine a correlation between several hunting and logistic parameters (age, sex, animal weight, shooting distance, guns caliber, number of shots, weather and atmospheric temperature, time from shot to refrigeration and to analysis) to the bacterial population of the carcass.
3. Results and Discussion
The average bacterial count of the carcass surfaces was 3.27-log cfu cm
−2 (SD 1.43, minimum 0.6 and maximum 7.04).
Table 4 brings to light a lot of data. The most important findings should be discussed.
The relationship between the bacterial population and some risk factors, determined by a regression analysis, is shown in
Figure 3.
The correlation coefficient, r, was found to be 0.038 for the eviscerated body weight (
p < 0.05) (
Figure 3a), 0.091 for the external temperature on the day of hunting (
p < 0.05) (
Figure 3b), 0.038 for the time from refrigeration to analysis (
p < 0.05) (
Figure 3c), 0.027 for the time from shot to refrigeration (
p = 0.0811) (
Figure 3d) and 0.043 for the time from shot to analysis (
p < 0.05) (
Figure 3e).
Table 5 shows the average values for the factors correlated to the bacterial population split by contamination levels above and below 5-log cfu cm
−2These results stand for a negative correlation between the bacterial population and eviscerated carcass weight and between the bacterial population and external temperature and for a positive correlation between the time from shot to analysis and from refrigeration to analysis. No association was demonstrated between the bacterial population and the time from shot to refrigeration.
The multivariate analysis (
Table 6 identified “temperature” as a factor inversely related to carcass contamination above 5-log cfu cm
−2: OR 0.905 (CI = 0.842 − 0.972,
p = 0.0062) indicates a lower probability of carcass contamination above 5 log in animals hunted with higher external temperatures.
However, the differences between the odd ratios from multiple logistic regression and simple logistic regression indicate that there are some confounding effects of other factors on the relationship between the external temperature and microbial population of a carcass. In fact, the simple logistic regression (
Table 7) also showed statistically significant differences (
p < 0.05) for “refr/sampling” (OR = 1.009,
p = 0.0227) and “shot/sampling” (OR = 1.008,
p = 0.0271), where, apparently, the longer the time, the higher the probability of a microbial population above 5-log cfu cm
−2.
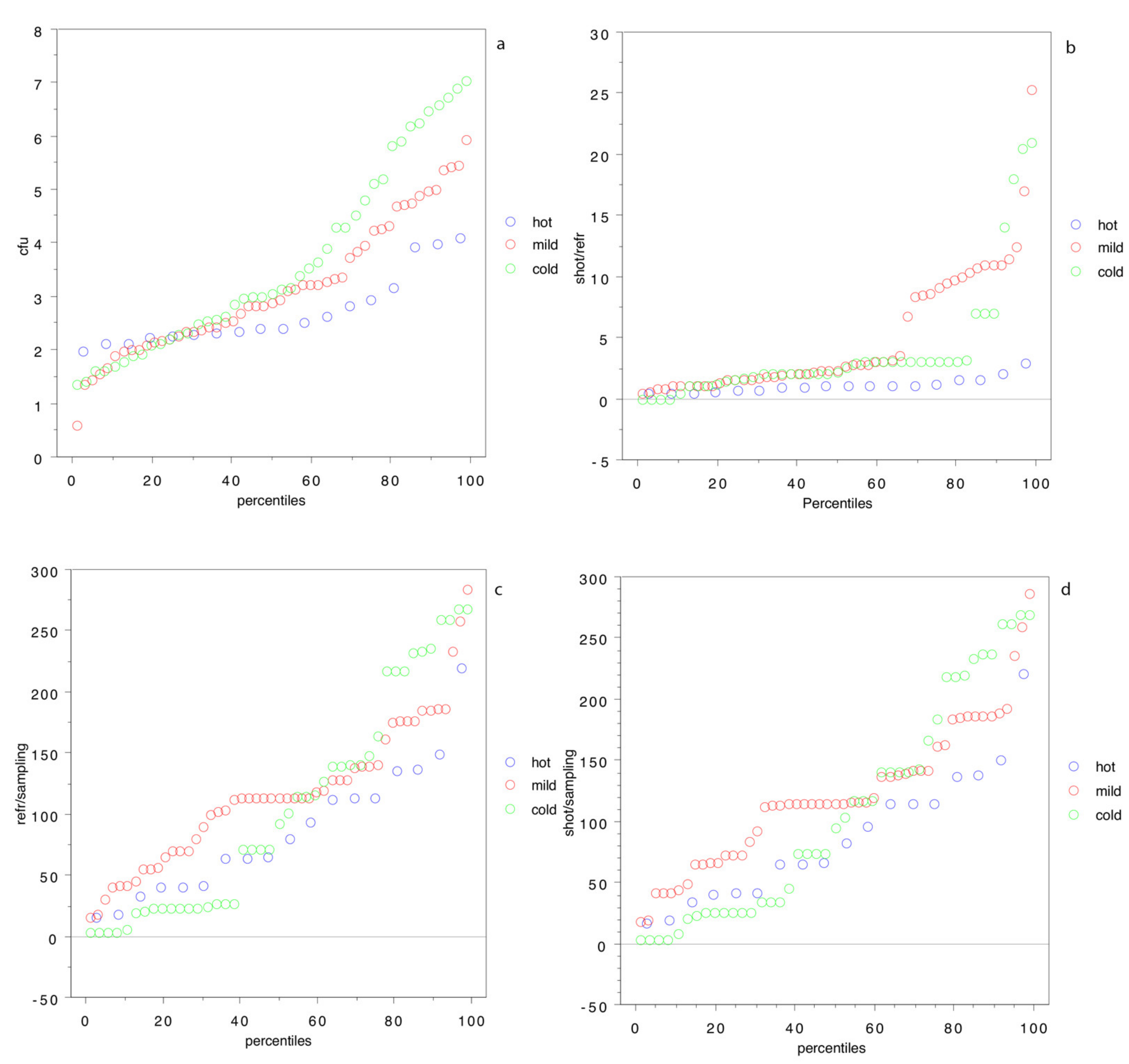
Figure 4 sheds some light: the percentile distribution of log cfu cm
−2 (
Figure 4a) clearly shows that, from the 50th percentile, the bacterial population of the carcasses is higher in wild boars hunted in the cold season.
Figure 4b–d, on the other hand, shows that the time from shot to refrigeration, from refrigeration to analysis and, in general, from shot to analysis are higher in the cold or mild weather at the time of hunting.
This last result is of the utmost importance, because it is well-known that hunters are concerned by the external temperatures, and for hunting trip done during the hot season or in the warmest part of the day, they tend to shorten the delay between the shot and the proper refrigeration of the carcass. On the other hand, in the winter and the coldest hours of the day, hunters tend to delay carcass refrigeration, because they rely on low temperatures [
10]. The results of our study, however, demonstrated that proper and prompt refrigeration is of the utmost importance also in the winter and in cold weather, because the carcass needs an immediate refrigeration for the proper storage and aging of meat. The industry standard cold chain, in fact, relies on low temperatures and ventilation to lower the internal carcass temperature to 0–4 °C within the first 48 h, i.e., within four times the so-called semi-cooling time.
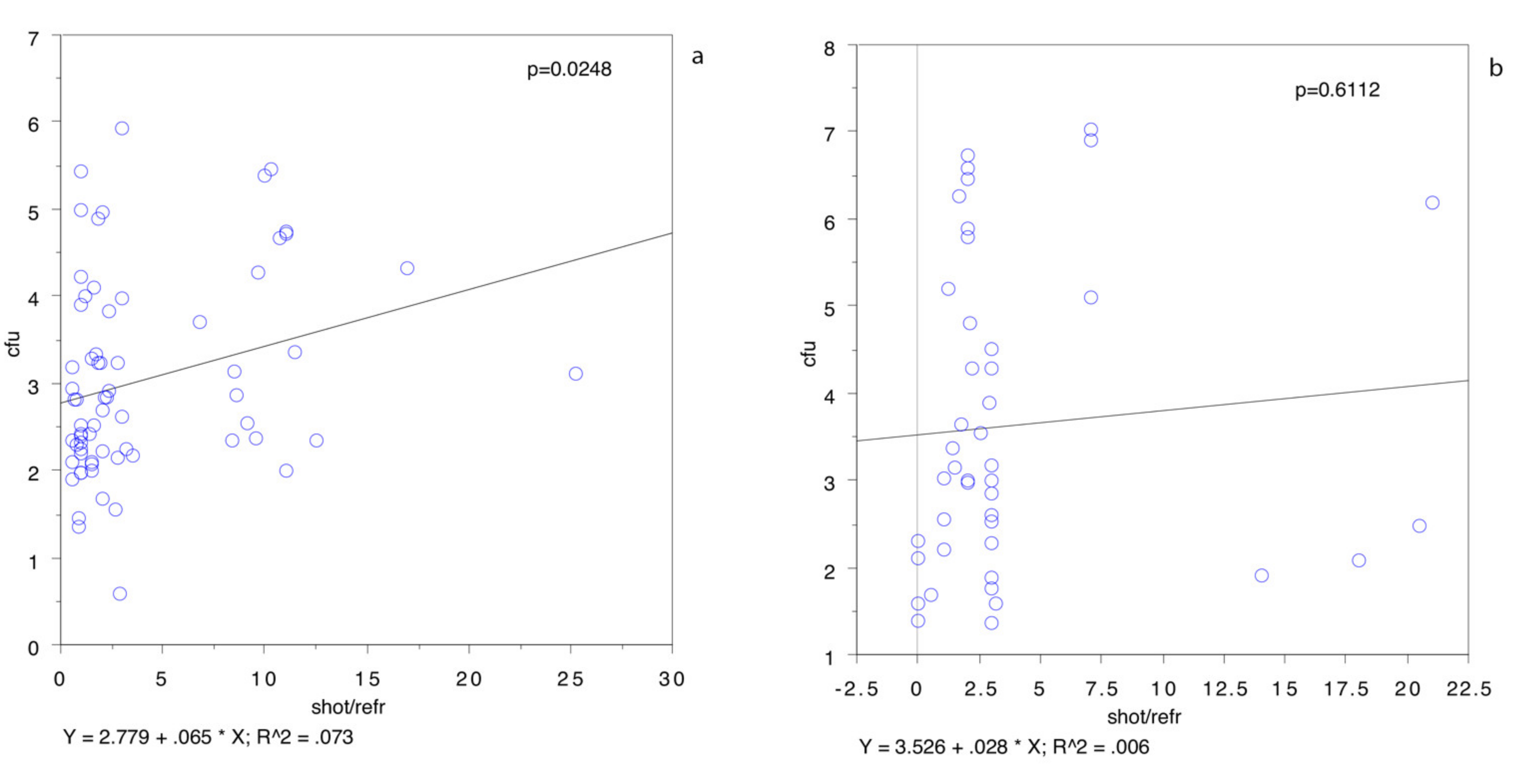
Our study also demonstrated that the bacterial population of wild boar carcasses is not correlated to the mere time from shot to refrigeration but is correlated to the refrigeration time from chilling to analysis. The study results revealed a correlation between the time from shot to analysis and from refrigeration to analysis but a lack of correlation between the time from shot to refrigeration. This is another important point to consider, because the prolonged refrigerated storage of hunted meat, especially when the carcass has been contaminated by environmental microorganisms, may increase the probability of a higher bacterial population at the time of analysis. Our results demonstrated, however, a lower bacterial population in the heavier carcasses (
Figure 3a), not confirmed by a univariate analysis. Furthermore, when the regression plot was rerun after omitting all the animals hunted in the winter or in the colder parts of the day, the time between the shot and refrigeration were positively correlated (
Figure 5a): correlation coefficient, r, was 0.073,
p < 0.05. On the other hand, the same test, run with only the animals hunted in the winter or in the coldest parts of the day (
Figure 5b), showed no statistically significant differences (r = 0.006,
p = 0.6112).
Data on the factors affecting the bacterial population in wild boar carcasses are available in the literature, although the results are contradictory. For instance, Avagnina, et al. [
15] found that bacterial contamination—in particular, the aerobic viable counts—were not influenced by the time elapsed between shooting and evisceration and sampling. For these authors, the statistical analyses revealed that the aerobic viable counts and Enterobacteriaceae counts in each species did not differ significantly across either the shooting–evisceration or shooting–sampling time ranges. These authors postulated that a lack of any statistical significance could be related to the grouping of the data into time categories that subsequently reduced the sample sizes of each group. On the other hand, Stella et al. [
11] found that the total viable counts were positively influenced by high environmental temperatures, and higher Enterobacteriaceae counts were detected in heavy male carcasses than females. More recently, Orsoni et al. [
16] found that the aerobic colony count mean value in the whole sample was 4.67-log cfu cm
−2 and varied with the boar’s total weight, time between evisceration and skinning and by cleaning. These authors found also that the aerobic colony counts increased with the boar’s total weight. Since a boar’s weight increases with age, they suggested that older animals may be more contaminated than younger ones and that their carcasses should therefore be managed with particular care. Our data regarding body weight proved that the contamination is higher for lighter carcasses during the regression analysis, but no association was statistically significant during the logistic regression (
Figure 3a and
Table 6). An experimental study by Soriano et al. [
17] in wild red deer proved that no differences in the aerobic mesophilic bacteria of M.
Longissinumus dorsi were detected for the carcasses eviscerated immediately or after 4 h.
The data on the average carcass contamination of hunted wild boar is very consistent across the literature and is distributed in the range of 3–6-log cfu cm
−2 [
9,
11,
15,
16,
17,
18]. To assess the microbiological status of the wild boar carcasses analyzed, our data were compared with the criteria specified by Regulation (EC) No. 2073/2005. However, the reference to these criteria is for guidance only, since they are provided for the carcasses of domestic pigs slaughtered in licensed premises (slaughterhouses). According to EU microbiological criteria for pig carcasses, the results are interpreted as satisfactory if the daily mean log for the total viable counts is <4-log cfu cm
−2, acceptable if between 4 and 5-log cfu/cm
−2 and unsatisfactory if the daily mean log is >5-log cfu cm
−2. In our study, the prevalence of total viable counts below 4-log cfu cm
−2 (satisfactory) occurred in 82 out of 112 carcasses (73.2%), between 4 and 5-log cfu cm
−2 (acceptable) occurred in 14 carcasses (12.5%) and above 5-log cfu cm
−2 (unsatisfactory) occurred in 16 out of 112 (14.3%) carcasses (
Table 1). The comparison of our results with the EU microbiological criteria that are used to demonstrate the microbiological quality of the production process indicates that the hygienic quality of the handling and dressing procedures with wild boar carcasses in our study was acceptable or satisfactory in 96 out of 112 carcasses (85.7%).