Multidrug-Resistant Campylobacer jejuni on Swine Processing at a Slaughterhouse in Eastern Spain
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Sample Collection
2.3. Campylobacter Isolation
2.4. Campylobacter Speciation
2.5. Antimicrobial Susceptibility
2.6. Statistical Analyses
3. Results
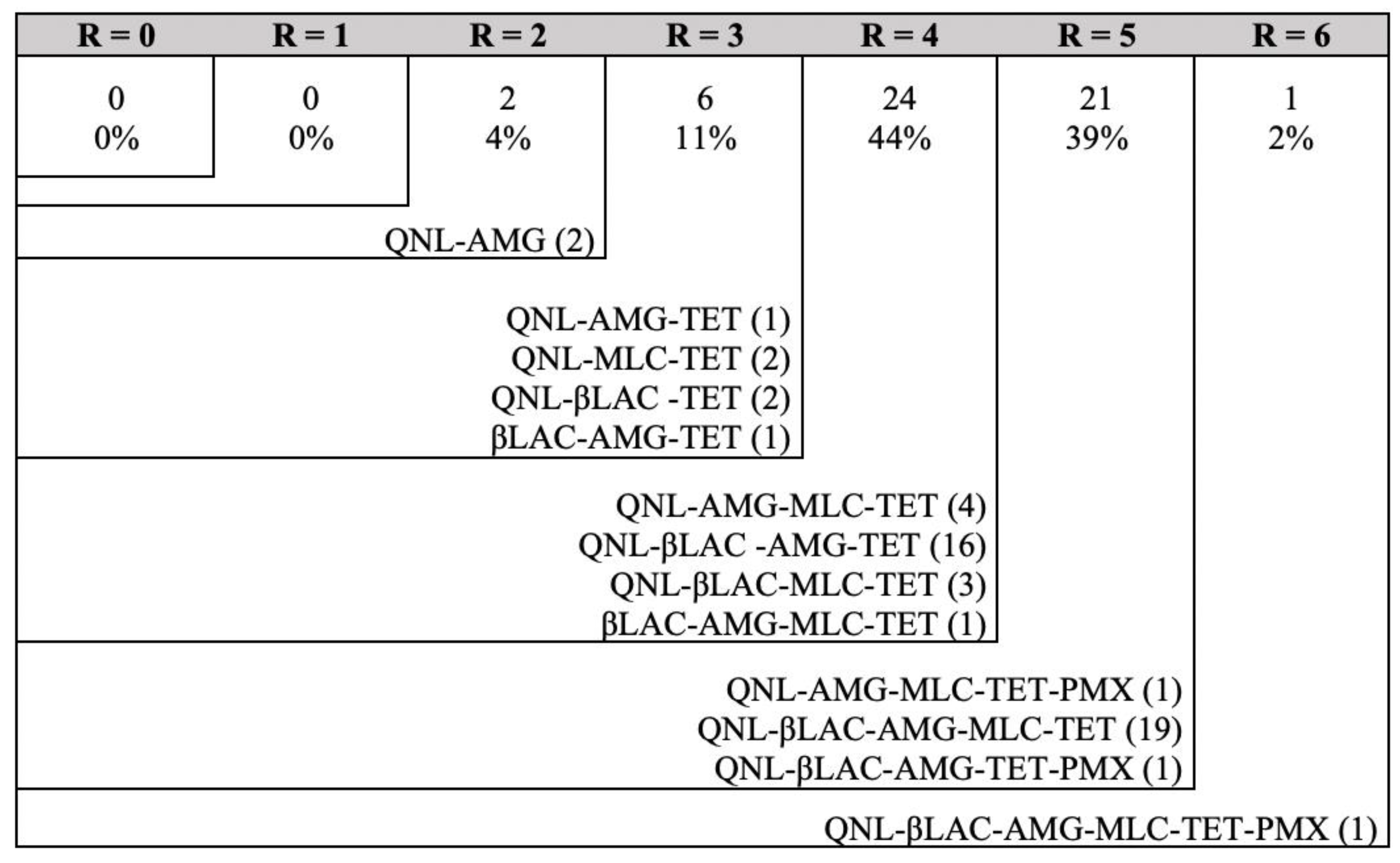
Antimicrobial Susceptibility
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO. Ten Threats to Global Health in 2019. Available online: https://www.who.int/news-room/feature-stories/ten-threats-to-global-health-in-2019 (accessed on 12 May 2020).
- Hutchings, M.; Truman, A.; Wilkinson, B. Antibiotics: Past, present and future. Curr. Opin. Microbiol. 2019, 51, 72–80. [Google Scholar] [CrossRef]
- O’Neill, J. Antimicrobial Resistance: Tackling a crisis for the health and wealth of nations. Rev. Antimicrob. Resist. Chaired 2014. Available online: http://www.jpiamr.eu/wp-content/uploads/2014/12/AMR-Review-Paper-Tackling-a-crisis-for-the-health-and-wealth-of-nations_1-2.pdf (accessed on 8 May 2021).
- ECDC. 33000 People Die Every Year Due to Infections with Antibiotic-Resistant Bacteria. Available online: https://www.ecdc.europa.eu/en/news-events/33000-people-die-every-year-due-infections-antibiotic-resistant-bacteria (accessed on 24 February 2021).
- FAO. One Health Legislation: Contributing to Pandemic Prevention through Law “An Ounce of Prevention is Worth a Pound of”; FAO: Roma, Italy, 2020. [Google Scholar]
- Tang, K.L.; Caffrey, N.P.; Nóbrega, D.B.; Cork, S.C.; Ronksley, P.E.; Barkema, H.W.; Polachek, A.J.; Ganshorn, H.; Sharma, N.; Kellner, J.D.; et al. Restricting the use of antibiotics in food-producing animals and its associations with antibiotic resistance in food-producing animals and human beings: A systematic review and meta-analysis. Lancet Planet Health 2017, 1, e316–e327. [Google Scholar] [CrossRef]
- Tacconelli, E.; Carrara, E.; Savoldi, A.; Harbarth, S.; Mendelson, M.; Monnet, D.L.; Pulcini, C.; Kahlmeter, G.; Kluytmans, J.; Carmeli, Y.; et al. Discovery, research, and development of new antibiotics: The WHO priority list of antibiotic-resistant bacteria and tuberculosis. Lancet Infect. Dis. 2018, 18, 318–327. [Google Scholar] [CrossRef]
- Altekruse, S.F.; Stern, N.J.; Fields, P.I.; Swerdlow, D.L. Campylobacter jejuni—An emerging foodborne pathogen. Emerg. Infect. Dis. 1999, 5, 28–35. [Google Scholar] [CrossRef] [PubMed]
- WHO. Campylobacter. Available online: https://www.who.int/news-room/fact-sheets/detail/campylobacter (accessed on 19 June 2020).
- EFSA; ECDC. The European Union One Health 2019 Zoonoses Report. EFSA J. 2021, 19. [Google Scholar] [CrossRef]
- O’Neill, J. Tackling drug-resistant infections globally. Rev. Antimicrob. Resist. 2016, 7, 110. [Google Scholar] [CrossRef]
- EFSA; ECDC. The European Union Summary Report on Antimicrobial Resistance in zoonotic and indicator bacteria from humans, animals and food in 2017/2018. EFSA J. 2020, 18. [Google Scholar] [CrossRef]
- CDC. Information for Health Professionals|Campylobacter|CDC. Available online: https://www.cdc.gov/campylobacter/technical.html (accessed on 22 June 2020).
- Figueredo, L.; Milán, W.; Font, J.; Zamora, V.; Rosales, A. Decomiso de vísceras de cerdos sacrificados en matadero y su impacto económico. Rev. Vet. Argent. 2018, 35, 1–8. [Google Scholar]
- MAPA. El Sector de la Carne de Cerdo en Cifras: Principales Indicadores Económicos; MAPA: Madrid, Spain, 2020. [Google Scholar]
- Sacristán, S. Plans to Voluntarily Reduce the Consumption of Antibiotics in Animal Health; Spanish Agency of Medicines and Medical Devices: Madrid, Spain, 2018.
- Rossler, E.; Signorini, M.L.; Romero-Scharpen, A.; Soto, L.P.; Berisvil, A.; Zimmermann, J.A.; Fusari, M.L.; Olivero, C.; Zbrun, M.V.; Frizzo, L.S. Meta-analysis of the prevalence of thermotolerant Campylobacter in food-producing animals worldwide. Zoonoses Public Health 2019, 66, 359–369. [Google Scholar] [CrossRef]
- Mencía-Ares, O.; Argüello, H.; Puente, H.; Gómez-García, M.; Álvarez-Ordóñez, A.; Manzanilla, E.G.; Carvajal, A.; Rubio, P. Effect of antimicrobial use and production system on Campylobacter spp., Staphylococcus spp. and Salmonella spp. resistance in Spanish swine: A cross-sectional study. Zoonoses Public Health 2021, 68, 54–66. [Google Scholar] [CrossRef]
- EC COMMISSION REGULATION (EU). 2017/1495 of 23 August 2017 amending Regulation (EC) No 2073/2005 as regards Campylobacter in broiler carcases. Off. J. Eur. Union 2017, 14, 1–6. [Google Scholar]
- Melero, B.; Juntunen, P.; Hänninen, M.L.; Jaime, I.; Rovira, J. Tracing Campylobacter jejuni strains along the poultry meat production chain from farm to retail by pulsed-field gel electrophoresis, and the antimicrobial resistance of isolates. Food Microbiol. 2012, 32, 124–128. [Google Scholar] [CrossRef]
- García-Sánchez, L.; Melero, B.; Jaime, I.; Hänninen, M.L.; Rossi, M.; Rovira, J. Campylobacter jejuni survival in a poultry processing plant environment. Food Microbiol. 2017, 65, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Pearce, R.A.; Wallace, F.M.; Call, J.E.; Dudley, R.L.; Oser, A.; Yoder, L.; Sheridan, J.J.; Luchansky, J.B. Prevalence of Campylobacter within a Swine Slaughter and Processing Facility. J. Food Prot. 2003, 66, 1550–1556. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Posch, J.; Feierl, G.; Wuest, G.; Sixl, W.; Schmidt, S.; Haas, D.; Reinthaler, F.F.; Marth, E. Transmission of Campylobacter spp. in a poultry slaughterhouse and genetic characterisation of the isolates by pulsed-field gel electrophoresis. Br. Poult. Sci. 2006, 47, 286–293. [Google Scholar] [CrossRef] [PubMed]
- Gruntar, I.; Biasizzo, M.; Kušar, D.; Pate, M.; Ocepek, M. Campylobacter jejuni contamination of broiler carcasses: Population dynamics and genetic profiles at slaughterhouse level. Food Microbiol. 2015, 50, 97–101. [Google Scholar] [CrossRef]
- Corry, J.E.L.; Atabay, H.I. Poultry as a source of Campylobacter and related organisms. Symp. Ser. Soc. Appl. Microbiol. 2001, 90, 96S–114S. [Google Scholar] [CrossRef] [PubMed]
- Oosterom, J.; Dekker, R.; de Wilde, G.J.; van Kempen-de Troye, F.; Engels, G.B. Prevalence of Campylobacter jejuni and Salmonella during pig slaughtering. Vet. Q. 1985, 7, 31–34. [Google Scholar] [CrossRef] [PubMed]
- Shang, K.; Wei, B.; Jang, H.K.; Kang, M. Phenotypic characteristics and genotypic correlation of antimicrobial resistant (AMR) Salmonella isolates from a poultry slaughterhouse and its downstream retail markets. Food Control 2019, 100, 35–45. [Google Scholar] [CrossRef]
- Marin, C.; Chinillac, M.C.; Cerdà-Cuéllar, M.; Montoro-Dasi, L.; Sevilla-Navarro, S.; Ayats, T.; Marco-Jimenez, F.; Vega, S. Contamination of pig carcass with Salmonella enterica serovar Typhimurium monophasic variant 1,4 [5],12: i: Originates mainly in live animals. Sci. Total Environ. 2020, 703, 134609. [Google Scholar] [CrossRef] [PubMed]
- Ceccarelli, M.; Leprini, E.; Sechi, P.; Iulietto, M.F.; Grispoldi, L.; Goretti, E.; Cenci-Goga, B.T. Analysis of the causes of the seizure and destruction of carcasses and organs in a slaughterhouse in central Italy in the 2010–2016 period. Ital. J. Food Saf. 2018, 7, 40–44. [Google Scholar] [CrossRef] [PubMed]
- Pielsticker, C.; Glünder, G.; Rautenschlein, S. Colonization properties of Campylobacter jejuni in chickens. Eur. J. Microbiol. Immunol. 2012, 2, 61–65. [Google Scholar] [CrossRef] [PubMed]
- Di Donato, G.; Marotta, F.; Nuvoloni, R.; Zilli, K.; Neri, D.; Di Sabatino, D.; Calistri, P.; Di Giannatale, E. Prevalence, population diversity and antimicrobial resistance of Campylobacter coli isolated in Italian swine at slaughterhouse. Microorganisms 2020, 8, 222. [Google Scholar] [CrossRef]
- ISO 10272-1:2018. Microbiology of the Food Chain—Horizontal Method for Detection and Enumeration of Campylobacter spp.—Part 1: Detection Method (ISO 10272-1:2017); ISO: Geneve, Switzerland, 2018. [Google Scholar]
- Ingresa-Capaccioni, S.; González-Bodí, S.; Jiménez-Trigos, E.; Marco-Jiménez, F.; Catalá, P.; Vega, S.; Marin, C. Comparison of different sampling types across the rearing period in broiler flocks for isolation of Campylobacter spp. Poult. Sci. 2015, 94, 766–771. [Google Scholar] [CrossRef]
- Ingresa-Capaccioni, S.; Jiménez-Trigos, E.; Marco-Jiménez, F.; Catalá, P.; Vega, S.; Marin, C. Campylobacter epidemiology from breeders to their progeny in Eastern Spain. Poult. Sci. 2016, 95, 676–683. [Google Scholar] [CrossRef]
- Marin, C.; Sevilla-Navarro, S.; Lonjedo, R.; Catalá-Gregori, P.; Ferrús, M.A.; Vega, S.; Jiménez-Belenguer, A. Genotyping and molecular characterisation of antimicrobial resistance in thermophilic Campylobacter isolated from poultry breeders and their progeny in Eastern Spain. Poult. Sci. 2020. [Google Scholar] [CrossRef]
- European Commission. Commission Implementing Decision of 12 November 2013 on the Monitoring and Reporting of Antimicrobial Resistance in Zoonotic and Commensal Bacteria (notified under document C(2013) 7145) (Text with EEA relevance). Off. J. Eur. Union 2013, 26–39. [Google Scholar]
- EFSA; ECDC. The European Union summary report on antimicrobial resistance in zoonotic and indicator bacteria from humans, animals and food in 2015. EFSA J. 2017, 15, 1–4694. [Google Scholar] [CrossRef]
- AEMPS. Plan Nacional Frente a la Resistencia a los Antibióticos 2019–2021. 2019. Available online: https://resistenciaantibioticos.es/es/system/files/field/files/pran_2019-2021_0.pdf?file=1&type=node&id=497&force=0 (accessed on 8 May 2021).
- Ghimire, L.; Singh, D.K.; Basnet, H.B.; Bhattarai, R.K.; Dhakal, S.; Sharma, B. Prevalence, antibiogram and risk factors of thermophilic campylobacter spp. in dressed porcine carcass of Chitwan, Nepal. BMC Microbiol. 2014, 14, 1–7. [Google Scholar] [CrossRef]
- Berger, S. Gideon Guide to Antimicrobial Agents, 2020 ed.; Gideon Informatics, Inc.: Los Angeles, CA, USA, 2020; ISBN 9781498825382. [Google Scholar]
- EUCAST New Definitions of S, I and R from 2019. Available online: https://www.eucast.org/newsiandr/ (accessed on 15 May 2020).
- Schill, F.; Abdulmawjood, A.; Klein, G.; Reich, F. Prevalence and characterization of extended-spectrum β-lactamase (ESBL) and AmpC β-lactamase producing Enterobacteriaceae in fresh pork meat at processing level in Germany. Int. J. Food Microbiol. 2017, 257, 58–66. [Google Scholar] [CrossRef]
- Stella, S.; Soncini, G.; Ziino, G.; Panebianco, A.; Pedonese, F.; Nuvoloni, R.; Di Giannatale, E.; Colavita, G.; Alberghini, L.; Giaccone, V. Prevalence and quantification of thermophilic Campylobacter spp. in Italian retail poultry meat: Analysis of influencing factors. Food Microbiol. 2017, 62, 232–238. [Google Scholar] [CrossRef]
- Slader, J.; Domingue, G.; Jørgensen, F.; McAlpine, K.; Owen, R.J.; Bolton, F.J.; Humphrey, T.J. Impact of transport crate reuse and of catching and processing on Campylobacter and Salmonella contamination of broiler chickens. Appl. Environ. Microbiol. 2002, 68, 713–719. [Google Scholar] [CrossRef] [PubMed]
- Abley, M.J.; Wittum, T.E.; Moeller, S.J.; Zerby, H.N.; Funk, J.A. Quantification of campylobacter in swine before, during, and after the slaughter process. J. Food Prot. 2012, 75, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Scanlon, K.A.; Cagney, C.; Walsh, D.; McNulty, D.; Carroll, A.; McNamara, E.B.; McDowell, D.A.; Duffy, G. Occurrence and characteristics of fastidious Campylobacteraceae species in porcine samples. Int. J. Food Microbiol. 2013, 163, 6–13. [Google Scholar] [CrossRef] [PubMed]
- Ghafir, Y.; China, B.; Dierick, K.; de Zutter, L.; Daube, G. A seven-year survey of Campylobacter contamination in meat at different production stages in Belgium. Int. J. Food Microbiol. 2007, 116, 111–120. [Google Scholar] [CrossRef]
- Hong, S.H.; Kim, H.S.; Yoon, K.S. Survival and risk comparison of Campylobacter jejuni on various processed meat products. Int. J. Environ. Res. Public Health 2016, 13, 580. [Google Scholar] [CrossRef]
- Brown, H.L.; Reuter, M.; Salt, L.J.; Cross, K.L.; Betts, R.P.; van Vliet, A.H.M. Chicken juice enhances surface attachment and biofilm formation of Campylobacter jejuni. Appl. Environ. Microbiol. 2014, 80, 7053–7060. [Google Scholar] [CrossRef]
- Trigui, H.; Thibodeau, A.; Fravalo, P.; Letellier, A.; Faucher, S.P. Survival in water of Campylobacter jejuni strains isolated from the slaughterhouse. Springerplus 2015, 4, 1–7. [Google Scholar] [CrossRef][Green Version]
- Di Giannatale, E.; Di Serafino, G.; Zilli, K.; Alessiani, A.; Sacchini, L.; Garofolo, G.; Aprea, G.; Marotta, F. Characterization of antimicrobial resistance patterns and detection of virulence genes in Campylobacter isolates in Italy. Sensors 2014, 14, 3308–3322. [Google Scholar] [CrossRef]
- Carter, D.L.; Docherty, K.M.; Gill, S.A.; Baker, K.; Teachout, J.; Vonhof, M.J. Antibiotic resistant bacteria are widespread in songbirds across rural and urban environments. Sci. Total Environ. 2018, 627, 1234–1241. [Google Scholar] [CrossRef]
- Lee, J.; Jeong, J.; Lee, H.; Ha, J.; Kim, S.; Choi, Y.; Oh, H.; Seo, K.; Yoon, Y.; Lee, S. Antibiotic susceptibility, genetic diversity, and the presence of toxin producing genes in campylobacter isolates from poultry. Int. J. Environ. Res. Public Health 2017, 14, 1400. [Google Scholar] [CrossRef] [PubMed]
- Notario, R.; Borda, N.; Gambande, T.; Bermejo, J.; Ponessa, A.; Toledo, V. Cepas de Campylobacter jejuni resistentes a quinolonas aisladas de humanos, gallinas y pollos. Medicina 2011, 71, 331–335. [Google Scholar] [PubMed]
- Rivera, N.; Bustos, R.; Montenegro, S.; Sandoval, M.; Castillo, J.; Fernández, H.; Maturana, M.; Delgado, L.; Contreras, A.; Chávez, D.; et al. Genotipificación y resistencia antibacteriana de cepas de Campylobacter spp. aisladas en niños y en aves de corral. Rev. Chil. Infect. 2011, 28, 555–562. [Google Scholar] [CrossRef]
- EFSA; ECDC. The European union summary report on antimicrobial resistance in zoonotic and indicator bacteria from humans, animals and food in 2017. EFSA J. 2019, 17, e05598. [Google Scholar] [CrossRef]
- Sartelli, M.; Weber, D.G.; Ruppé, E.; Bassetti, M.; Wright, B.J.; Ansaloni, L.; Catena, F.; Coccolini, F.; Abu-Zidan, F.M.; Coimbra, R.; et al. Antimicrobials: A global alliance for optimizing their rational use in intra-abdominal infections (AGORA). World J. Emerg. Surg. 2016, 11, 33. [Google Scholar] [CrossRef]
- Aarestrup, F.M. The livestock reservoir for antimicrobial resistance: A personal view on changing patterns of risks, effects of interventions and the way forward. Philos. Trans. R. Soc. B Biol. Sci. 2015, 370, 20140085. [Google Scholar] [CrossRef] [PubMed]
- ECDC/EFSA/EMA. Second joint report on the integrated analysis of the consumption of antimicrobial agents and occurrence of antimicrobial resistance in bacteria from humans and food-producing animals: Joint Interagency Antimicrobial Consumption and Resistan. EFSA J. 2017, 15, e04872. [Google Scholar] [CrossRef]
- Woolhouse, M.; Ward, M.; van Bunnik, B.; Farrar, J. Antimicrobial resistance in humans, livestock and the wider environment. Philos. Trans. R. Soc. B Biol. Sci. 2015, 370, 20140083. [Google Scholar] [CrossRef]
- Bengtsson, B.; Greko, C. Antibiotic resistance-consequences for animal health, welfare, and food production. Ups. J. Med. Sci. 2014, 119, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Agyare, C.; Etsiapa Boamah, V.; Ngofi Zumbi, C.; Boateng Osei, F. Antibiotic Use in Poultry Production and Its Effects on Bacterial Resistance. In Antimicrobial Resistance—A Global Threat; IntechOpen: London, UK, 2019. [Google Scholar]
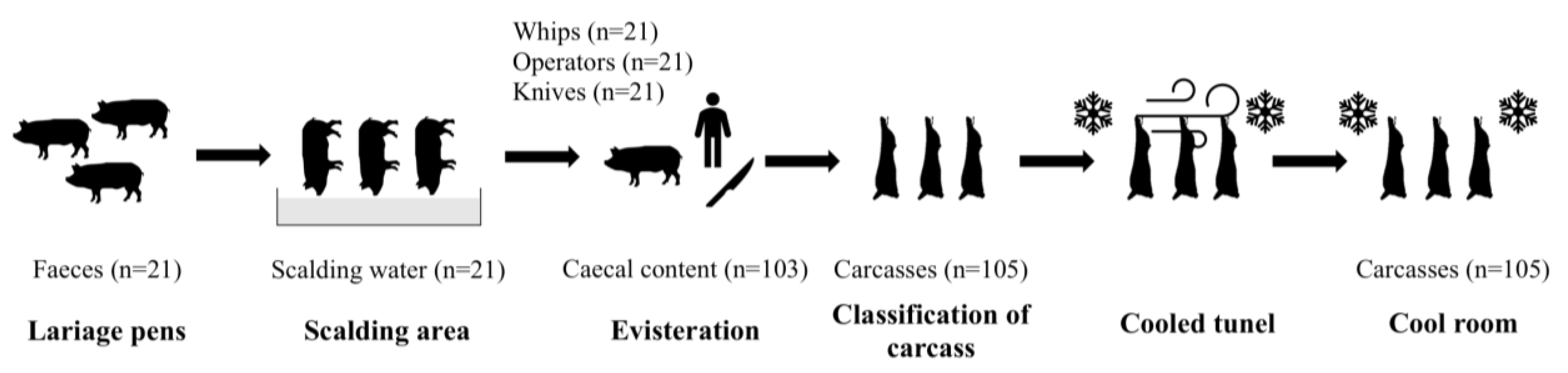
| Sample Type | nT | Campylobacter spp. (%) | nc | C. jejuni (%) | ncj | MDR C. jejuni (%) | |
|---|---|---|---|---|---|---|---|
| Animal samples | Faeces | 21 | 57.0 ± 10.8 ab | 12 | 75.0 ± 12.5 a | 8 | 100 ± 0.0 |
| Caeca | 103 | 70.0 ± 4.5 a | 65 | 31.0 ± 5.7 c | 18 | 89.0 ± 7.4 | |
| Carcass BC | 105 | 49.0 ± 4.9 b | 50 | 38.0 ± 6.9 bc | 14 | 100 ± 0.0 | |
| Carcass AC | 105 | 27.0 ± 4.3 c | 28 | 54.0 ± 9.4 ab | 11 | 100 ± 0.0 | |
| Environmental samples | Whips | 21 | 14.0 ± 7.6 c | 3 | 67.0 ± 27.2 abc | 2 | 100 ± 0.0 |
| Operator | 21 | 29.0 ± 9.9 bc | 5 | 60.0 ± 21.9 abc | 1 | 100 ± 0.0 | |
| Knives | 21 | 14.0 ± 7.6 c | 1 | 0 d | - | - | |
| p-value | <0.001 | 0.000 | >0.05 | ||||
| Type of Sample | n | CIP | NA | AMP | AMC | GEN | S | E | TE | CST |
|---|---|---|---|---|---|---|---|---|---|---|
| Faeces | 8 | 87.5 | 87.5 | 87.5 ab | 0 | 12.5 c | 100 a | 62.5 b | 100 | 12.5 |
| Caeca | 18 | 100 | 88.9 | 77.8 b | 0 | 22.2 bc | 88.9 ab | 55.6 b | 88.9 | 5.6 |
| Carcass before cooling | 14 | 92.9 | 92.9 | 85.7 ab | 0 | 14.3 c | 64.3 bc | 42.9 b | 100 | 0 |
| Carcass after cooling | 11 | 100 | 81.8 | 72.7 b | 0 | 54.5 b | 54.5 c | 63.6 b | 100 | 9.1 |
| Whips | 2 | 100 | 100 | 50 ab | 0 | 50 abc | 100 a | 100 a | 100 | 0 |
| Operator | 1 | 100 | 100 | 100 a | 0 | 100 a | 100 a | 100 a | 100 | 0 |
| p-value | >0.05 | >0.05 | 0.012 | - | 0.000 | 0.000 | 0.000 | >0.05 | >0.05 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marin, C.; Lorenzo-Rebenaque, L.; Moreno-Moliner, J.; Sevilla-Navarro, S.; Montero, E.; Chinillac, M.C.; Jordá, J.; Vega, S. Multidrug-Resistant Campylobacer jejuni on Swine Processing at a Slaughterhouse in Eastern Spain. Animals 2021, 11, 1339. https://doi.org/10.3390/ani11051339
Marin C, Lorenzo-Rebenaque L, Moreno-Moliner J, Sevilla-Navarro S, Montero E, Chinillac MC, Jordá J, Vega S. Multidrug-Resistant Campylobacer jejuni on Swine Processing at a Slaughterhouse in Eastern Spain. Animals. 2021; 11(5):1339. https://doi.org/10.3390/ani11051339
Chicago/Turabian StyleMarin, Clara, Laura Lorenzo-Rebenaque, Judith Moreno-Moliner, Sandra Sevilla-Navarro, Estefania Montero, Mᵃ Carmen Chinillac, Jaume Jordá, and Santiago Vega. 2021. "Multidrug-Resistant Campylobacer jejuni on Swine Processing at a Slaughterhouse in Eastern Spain" Animals 11, no. 5: 1339. https://doi.org/10.3390/ani11051339
APA StyleMarin, C., Lorenzo-Rebenaque, L., Moreno-Moliner, J., Sevilla-Navarro, S., Montero, E., Chinillac, M. C., Jordá, J., & Vega, S. (2021). Multidrug-Resistant Campylobacer jejuni on Swine Processing at a Slaughterhouse in Eastern Spain. Animals, 11(5), 1339. https://doi.org/10.3390/ani11051339








