Integration of Metabolomics and Transcriptomicsto Comprehensively Evaluate the Metabolic Effects of Gelsemium elegans on Pigs
Abstract
Simply Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. Animals and Treatments
2.3. Plasma Biochemical and Immunity Parameters
2.4. Transcriptome Analysis
2.5. Metabolomic Analysis
2.5.1. Plasma Sample Collection and Preparation
2.5.2. UPLC–MS/MS Conditions
2.5.3. Metabolomics Data Processing
3. Results
3.1. Growth Performance and Routine Blood Analysis
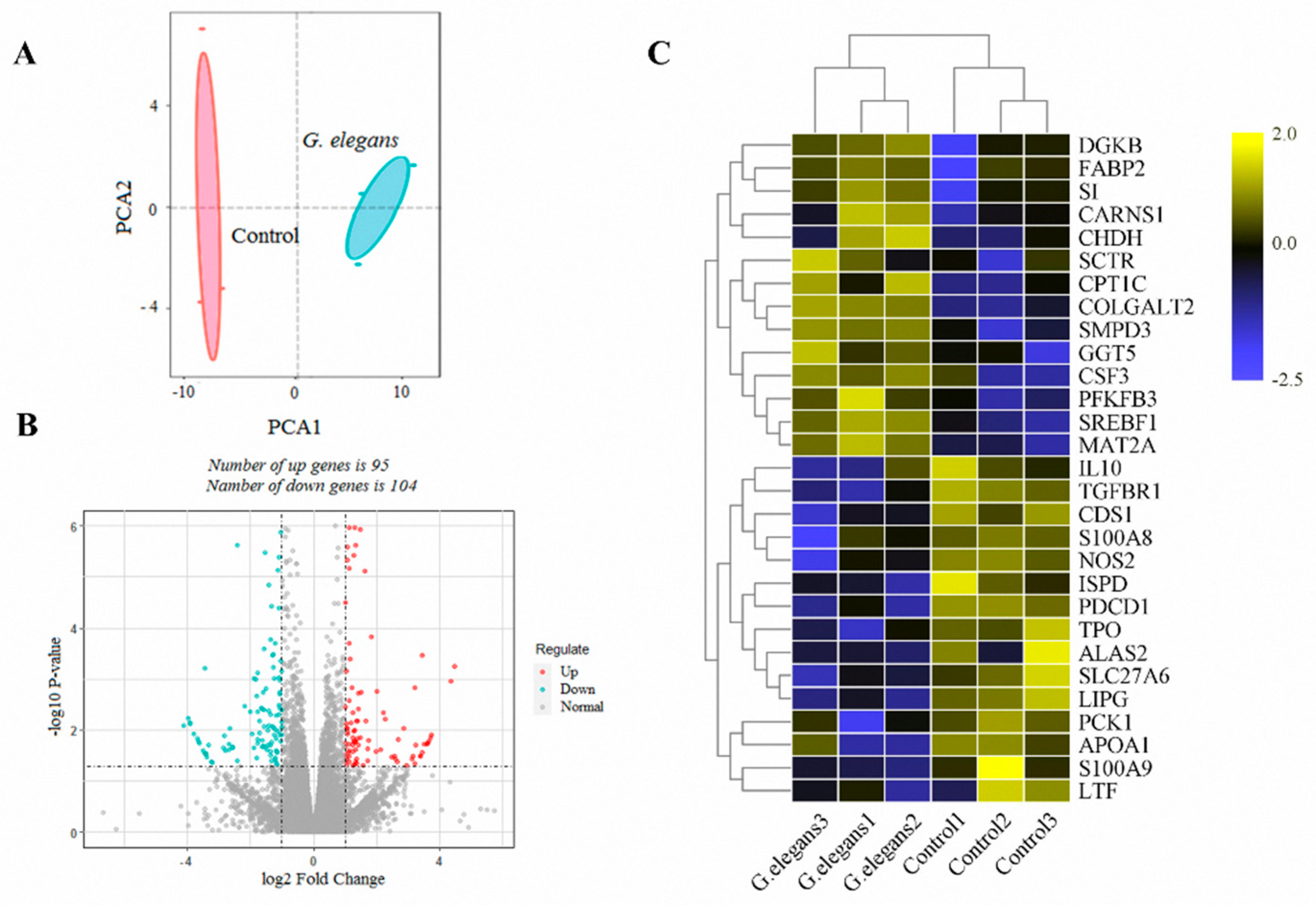
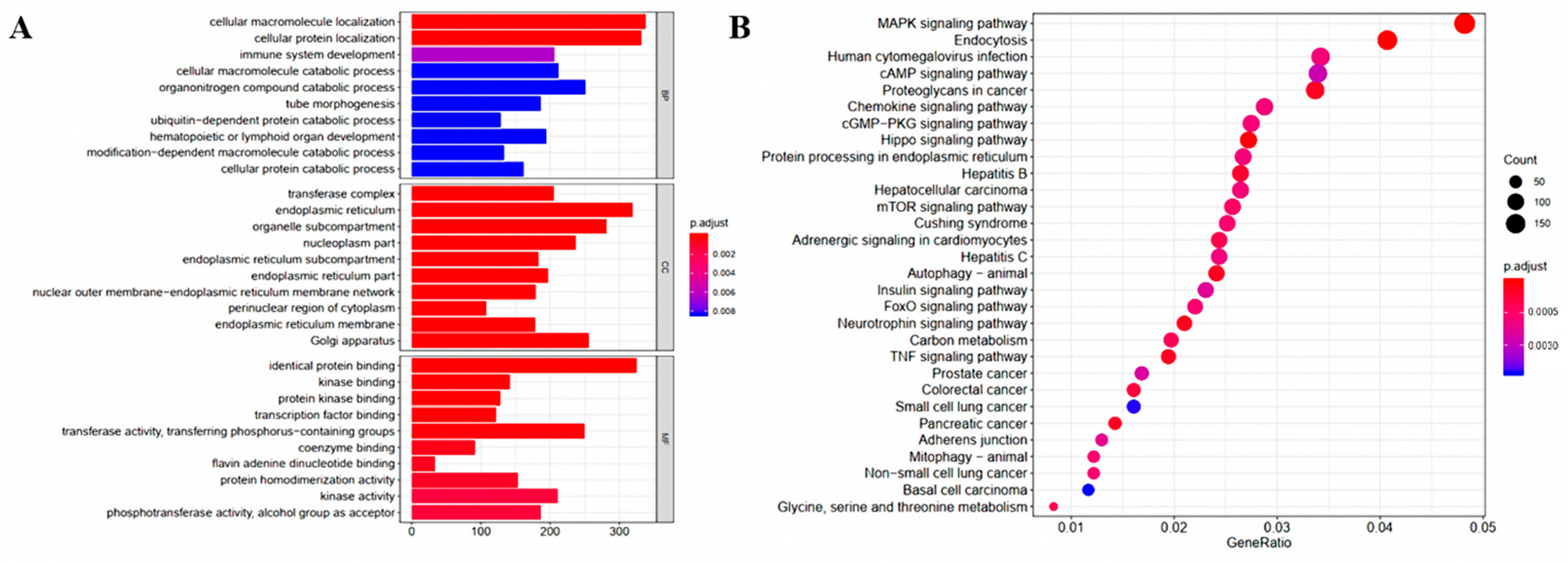
3.2. Analysis of the Liver Transcriptome under the Influence of G. elegans
3.3. Analysis of the Plasma Metabolome under the Influence of G. elegans
3.4. Integrated Enrichment Analysis of Transcript and Metabolite Profiles
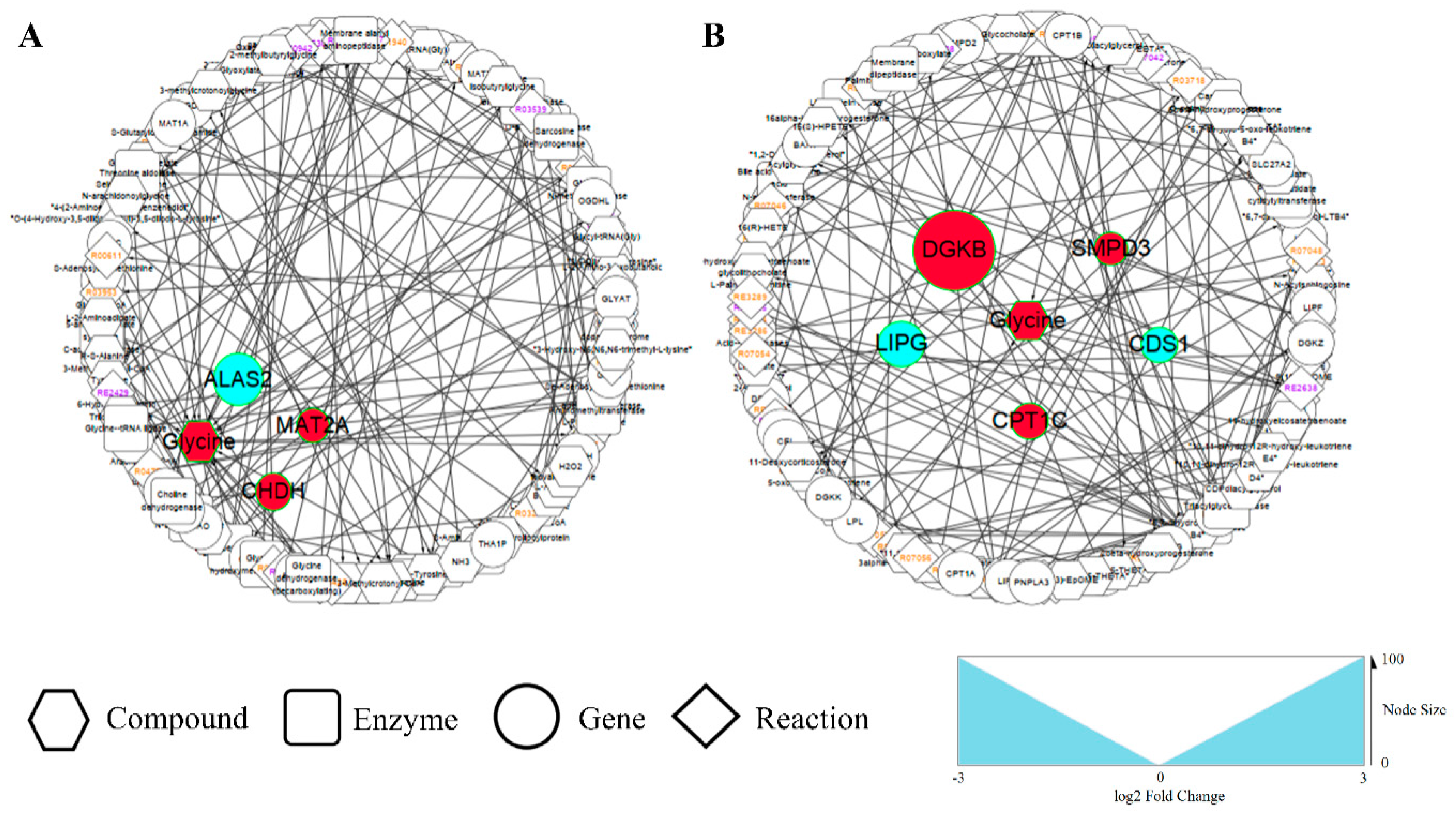
3.4.1. Amino Acid Metabolism Alterations
3.4.2. Lipid Metabolism Alterations
4. Discussion
4.1. Effect of G. elegans on Intake and Average Gain
4.2. Effect of G. elegans on Physiological Function of Weaned Piglets
4.3. Evidence That G. elegans Regulates Amino Acid Metabolism and Decreases p38 Activation
4.4. G. elegans Regulates Lipid Metabolism
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BP | biological processes |
| CD | Compound Discoverer |
| DEGs | differentially expressed genes |
| ERK | extracellular signal-regulated kinase |
| G. elegans | Gelsemium elegans |
| GO | genetic ontology |
| JNK | c-Jun N-terminal kinase |
| KEGG | Kyoto Encyclopedia of Genes and Genomes |
| LPS | lipopolysaccharide |
| MF | molecular function |
| MAPKs | mitogen-activated protein kinases |
| NAC | N-acetylcysteine |
| NAFLD | Nonalcoholic fatty liver disease |
| PCA | principal component analysis |
| PLS-DA | partial least squares-discriminant analysis |
References
- Yuan, Z.; Matias, F.B.; Wu, J.; Liang, Z.; Sun, Z. Koumine Attenuates Lipopolysaccaride-Stimulated Inflammation in RAW264.7 Macrophages, Coincidentally Associated with Inhibition of NF-κB, ERK and p38 Pathways. Int. J. Mol. Sci. 2016, 17, 430. [Google Scholar] [CrossRef] [PubMed]
- Rujjanawate, C.; Kanjanapothi, D.; Panthong, A. Pharmacological effect and toxicity of alkaloids from Gelsemium elegans Benth. J. Ethnopharmacol. 2003, 89, 91–95. [Google Scholar] [CrossRef]
- Xu, Y.; Qiu, H.-Q.; Liu, H.; Liu, M.; Huang, Z.-Y.; Yang, J.; Su, Y.-P.; Yu, C.-X. Effects of koumine, an alkaloid of Gelsemium elegans Benth., on inflammatory and neuropathic pain models and possible mechanism with allopregnanolone. Pharm. Biochem. Behav. 2012, 101, 504–514. [Google Scholar] [CrossRef] [PubMed]
- Dao-Rong, L.; Tai-Kang, H.; You-Shun, W.; Yu-Min, F.; Fu-Qing, Z.; Qi-An, L. Experimental study on feeding ginseng to pigs. Chin. Anim. Husb. Vet. Med. 1994, 02, 67–68. [Google Scholar]
- Lin, Q.-Y. Compound pig ginseng feed additive fattening test report. Chin. Anim. Husb. Vet. Med. 1993, 05, 207–208. [Google Scholar]
- Chen, X.J.; Wang, Y.; Wang, S.S.; Wu, Y.; Li, Y.Y.; Sun, Z.L. Effects of Gelsemium elegans Extract on Growth Performance, Intestinal Morphology and Cecal Flora of Growing Pigs. Chin. J. Anim. Nutr. 2018, 30, 3626–3633. [Google Scholar]
- Cao, J.J.; Yang, K.; Huang, C.Y.; Li, Y.J.; Yu, H.; Wu, Y.; Sun, Z.L.; Liu, Z.Y. Pharmacokinetic Study of Multiple Components of Gelsemium elegans in Goats by Ultra-Performance Liquid Chromatography Coupled to Tandem Mass Spectrometry. J. Anal. Toxicol. 2020, 44, 378–390. [Google Scholar] [CrossRef]
- Xin, H.; YiFei, G.; Ke, Y.; YiYu, C. Gas Chromatography-Mass Spectrometry Based on Metabonomics Study of Carbon Tetrachloride-Induced Acute Liver Injury in Mice. Chin. J. Anal. Chem. 2007, 35, 1736–1740. [Google Scholar] [CrossRef]
- Sun, R.; Chen, M.; Hu, Y.; Lan, Y.; Gan, L.; You, G.; Yue, M.; Wang, H.; Xia, B.; Zhao, J.; et al. CYP3A4/5 mediates the metabolic detoxification of humantenmine, a highly toxic alkaloid from Gelsemium elegans Benth. J. Appl. Toxicol. JAT 2019, 39, 1283–1292. [Google Scholar] [CrossRef]
- Yue, R.; Jin, G.; Wei, S.; Huang, H.; Su, L.; Zhang, C.; Xu, Y.; Yang, J.; Liu, M.; Chu, Z.; et al. Immunoregulatory Effect of Koumine on Nonalcoholic Fatty Liver Disease Rats. J. Immunol. Res. 2019, 2019, 8325102. [Google Scholar] [CrossRef]
- Gessner, D.K.; Schwarz, A.; Meyer, S.; Wen, G.; Most, E.; Zorn, H.; Ringseis, R.; Eder, K. Insect Meal as Alternative Protein Source Exerts Pronounced Lipid-Lowering Effects in Hyperlipidemic Obese Zucker Rats. J. Nutr. 2019, 149, 566–577. [Google Scholar] [CrossRef]
- Huang, C.-Y.; Yang, K.; Cao, J.-J.; Li, Y.-J.; Wang, Z.-Y.; Yu, H.; Sun, Z.-L.; Zhang, X.; Liu, Z.-Y. Gene expression profile analysis of ileum transcriptomes in pigs fed Gelsemium elegans plants. Sci. Rep. 2019, 9, 1–8. [Google Scholar]
- Cabañas-García, E.; Areche, C.; Jáuregui-Rincón, J.; Cruz-Sosa, F.; Pérez-Molphe Balch, E. Phytochemical Profiling of (Cactaceae) Growing in Greenhouse Conditions Using Ultra-High-Performance Liquid Chromatography⁻Tandem Mass Spectrometry. Molecules 2019, 24, 705. [Google Scholar] [CrossRef] [PubMed]
- Bennett, B.J.; Vallim, T.Q.d.A.; Wang, Z.; Shih, D.M.; Meng, Y.; Gregory, J.; Allayee, H.; Lee, R.; Graham, M.; Crooke, R. Trimethylamine-N-Oxide, a Metabolite Associated with Atherosclerosis, Exhibits Complex Genetic and Dietary Regulation. Cell Metab. 2013, 17, 49–60. [Google Scholar] [CrossRef] [PubMed]
- Wu, J. Experimental study on promoting pig growth by Gelsemium elegans Benth. Chin. Fujian Agric. For. Univ. 2015, 828, 5. [Google Scholar]
- Wang, J.; Sun, Z.L. Effect and mechanism of adding Gelsemium elegans dry powder to feed on growth performance of piglets. Chin. J. Tradit. Vet. Sci. 2019, 002, 3–4. [Google Scholar]
- Wu, G. Glycine Stimulates Protein Synthesis and Inhibits Oxidative Stress in Pig Small Intestinal Epithelial Cells. J. Nutr. 2014, 144, 1540–1548. [Google Scholar]
- Jin, G.-L.; Su, Y.-P.; Liu, M.; Xu, Y.; Yang, J.; Liao, K.-J.; Yu, C.-X. Medicinal plants of the genus Gelsemium (Gelsemiaceae, Gentianales)--a review of their phytochemistry, pharmacology, toxicology and traditional use. J. Ethnopharmacol. 2014, 152, 33–52. [Google Scholar] [CrossRef]
- Yang, K.; Long, X.-M.; Cao, J.-J.; Li, Y.-J.; Wu, Y.; Bai, X.; Sun, Z.-L.; Liu, Z.-Y. An analytical strategy to explore the multicomponent pharmacokinetics of herbal medicine independently of standards: Application in Gelsemium elegans extracts. J. Pharm. Biomed. Anal. 2019, 176, 112833. [Google Scholar] [CrossRef] [PubMed]
- Ye, Q.; Feng, Y.; Wang, Z.; Zhou, A.; Xie, S.; Zhang, Y.; Xiang, Q.; Song, E.; Zou, J. Effects of dietary Gelsemium elegans alkaloids on growth performance, immune responses and disease resistance of Megalobrama amblycephala. Fish Shellfish. Immunol. 2019, 91, 29–39. [Google Scholar] [CrossRef]
- Legendre, P.; Förstera, B.; Jüttner, R.; Meier, J.C. Glycine Receptors Caught between Genome and Proteome-Functional Implications of RNA Editing and Splicing. Front. Mol. Neurosci. 2009, 2, 23. [Google Scholar] [CrossRef] [PubMed]
- Grotz, M.R.; Pape, H.C.; van Griensven, M.; Stalp, M.; Rohde, F.; Bock, D.; Krettek, C. Glycine reduces the inflammatory response and organ damage in a two-hit sepsis model in rats. Shock 2001, 16, 116–121. [Google Scholar] [CrossRef] [PubMed]
- Uraz, S.; Tahan, G.; Aytekin, H.; Tahan, V. N-acetylcysteine expresses powerful anti-inflammatory and antioxidant activities resulting in complete improvement of acetic acid-induced colitis in rats. Scand. J. Clin. Lab. Investig. 2013, 73, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Harrison, P.; Wendon, J.; Williams, R. Evidence of increased guanylate cyclase activation by acetylcysteine in fulminant hepatic failure. Hepatology 1996, 23, 1067–1072. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Hou, Y.; Yi, D.; Wang, L.; Ding, B.; Chen, X.; Long, M.; Liu, Y.; Wu, G. Protective effects of N-acetylcysteine on acetic acid-induced colitis in a porcine model. BMC Gastroenterol. 2013, 13, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Zhou, N.; Sun, Y.-P.; Zheng, X.-K.; Wang, Q.-H.; Yang, Y.-Y.; Bai, Z.-Y.; Kuang, H.-X.; Feng, W.-S. A Metabolomics-Based Strategy for the Mechanism Exploration of Traditional Chinese Medicine: Seeds Extract and Fractions as a Case Study. Evid.-Based Complement. Altern. Med. 2017, 2017, 2845173. [Google Scholar] [CrossRef]
- Vuolteenaho, K.; Koskinen, A.; Kukkonen, M.; Nieminen, R.; Päivärinta, U.; Moilanen, T.; Moilanen, E. Leptin enhances synthesis of proinflammatory mediators in human osteoarthritic cartilage--mediator role of NO in leptin-induced PGE2, IL-6, and IL-8 production. Mediat. Inflamm. 2009, 2009, 345838. [Google Scholar] [CrossRef]
- Xia, C.; Braunstein, Z.; Toomey, A.C.; Zhong, J.; Rao, X. S100 Proteins As an Important Regulator of Macrophage Inflammation. Front. Immunol. 2017, 8, 1908. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Hu, X.; Long, K.; Gao, C.; Dong, H.-L.; Zhong, Q.; Gao, X.-M.; Gong, F.-Y. Extraordinarily potent proinflammatory properties of lactoferrin-containing immunocomplexes against human monocytes and macrophages. Sci. Rep. 2017, 7, 4230. [Google Scholar] [CrossRef]
- He, F.; Wu, C.; Li, P.; Li, N.; Zhang, D.; Zhu, Q.; Ren, W.; Peng, Y. Functions and Signaling Pathways of Amino Acids in Intestinal Inflammation. Biomed. Res. Int. 2018, 2018, 9171905. [Google Scholar] [CrossRef]
- Wang, L.; Zhou, J.; Hou, Y.; Yi, D.; Ding, B.; Xie, J.; Zhang, Y.; Chen, H.; Wu, T.; Zhao, D.; et al. N-Acetylcysteine supplementation alleviates intestinal injury in piglets infected by porcine epidemic diarrhea virus. Amino. Acids 2017, 49, 1931–1943. [Google Scholar] [CrossRef]
- Yi, D.; Hou, Y.; Xiao, H.; Wang, L.; Zhang, Y.; Chen, H.; Wu, T.; Ding, B.; Hu, C.-A.A.; Wu, G. N-Acetylcysteine improves intestinal function in lipopolysaccharides-challenged piglets through multiple signaling pathways. Amino. Acids 2017, 49, 1915–1929. [Google Scholar] [CrossRef] [PubMed]
- Docena, G.; Rovedatti, L.; Kruidenier, L.; Fanning, A.; Leakey, N.A.B.; Knowles, C.H.; Lee, K.; Shanahan, F.; Nally, K.; McLean, P.G.; et al. Down-regulation of p38 mitogen-activated protein kinase activation and proinflammatory cytokine production by mitogen-activated protein kinase inhibitors in inflammatory bowel disease. Clin. Exp. Immunol. 2010, 162, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Coskun, M.; Olsen, J.; Seidelin, J.B.; Nielsen, O.H. MAP kinases in inflammatory bowel disease. Clin. Chim Acta 2011, 412, 513–520. [Google Scholar] [CrossRef] [PubMed]
- Raza, A.; Crothers, J.W.; McGill, M.M.; Mawe, G.M.; Teuscher, C.; Krementsov, D.N. Anti-inflammatory roles of p38α MAPK in macrophages are context dependent and require IL-10. J. Leukoc. Biol. 2017, 102, 1219–1227. [Google Scholar] [CrossRef] [PubMed]
- You, B.H.; Chae, H.-S.; Song, J.; Ko, H.W.; Chin, Y.-W.; Choi, Y.H. α-Mangostin ameliorates dextran sulfate sodium-induced colitis through inhibition of NF-κB and MAPK pathways. Int. Immunopharmacol. 2017, 49, 212–221. [Google Scholar] [CrossRef]
- Karnovsky, A.; Weymouth, T.; Hull, T.; Tarcea, V.G.; Scardoni, G.; Laudanna, C.; Sartor, M.A.; Stringer, K.A.; Jagadish, H.V.; Burant, C. Metscape 2 bioinformatics tool for the analysis and visualization of metabolomics and gene expression data. Bioinformatics 2012, 28, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Maclouf, J.; Fltzpatrlck, F.; Murphy, R. Transcellular biosynthesis of eicosanoids. Pharmacol. Res. Off. J. Ital. Pharmacol. Soc. 1989, 21, 1–7. [Google Scholar] [CrossRef]
- Ilan, Y. Compounds of the sphingomyelin-ceramide-glycosphingolipid pathways as secondary messenger molecules: New targets for novel therapies for fatty liver disease and insulin resistance. Am. J. Physiol.-Gastrointest. Liver Physiol. 2016, 310, G1102–G1117. [Google Scholar] [CrossRef]
- Padberg, I.; Peter, E.; González-Maldonado, S.; Witt, H.; Mueller, M.; Weis, T.; Bethan, B.; Liebenberg, V.; Wiemer, J.; Katus, H.A.; et al. A new metabolomic signature in type-2 diabetes mellitus and its pathophysiology. PLoS ONE 2014, 9, e85082. [Google Scholar] [CrossRef] [PubMed]
- Guasch-Ferré, M.; Hruby, A.; Toledo, E.; Clish, C.B.; Martínez-González, M.A.; Salas-Salvadó, J.; Hu, F.B. Metabolomics in Prediabetes and Diabetes: A Systematic Review and Meta-analysis. Diabetes Care 2016, 39, 833–846. [Google Scholar] [CrossRef] [PubMed]
- Dunning, K.R.; Anastasi, M.R.; Zhang, V.J.; Russell, D.L.; Robker, R.L. Regulation of fatty acid oxidation in mouse cumulus-oocyte complexes during maturation and modulation by PPAR agonists. PLoS ONE 2014, 9, e87327. [Google Scholar] [CrossRef] [PubMed]
- Kaushal, C. The peroxisome proliferator-activated receptor: A family of nuclear receptors role in various diseases. J. Adv. Pharm. Technol. Res. 1999, 2, 236. [Google Scholar]
- Sander, K. Integrated physiology and systems biology of PPARα. Mol. Metab. 2014, 3, 354–371. [Google Scholar]
- McGarry, J.D.; Brown, N.F. The mitochondrial carnitine palmitoyltransferase system. From concept to molecular analysis. Eur. J. Biochem. 1997, 244, 1–14. [Google Scholar] [CrossRef]
- Thupari, J.N.; Landree, L.E.; Ronnett, G.V.; Kuhajda, F.P. C75 increases peripheral energy utilization and fatty acid oxidation in diet-induced obesity. Proc. Natl. Acad. Sci. USA 2002, 99, 9498–9502. [Google Scholar] [CrossRef] [PubMed]
- Zong, J.-Y.; Sun, R.-X.; Yu, M. Regulation of gluconeogenesis by adipose tissue. Chin. Chem. life 2017, 37, 842–848. [Google Scholar]

| Items | Treatment | SEM 1 | p-Value 2 | |
|---|---|---|---|---|
| Control | G. elegans-Treated | L | ||
| Num | 10 | 30 | - | - |
| Average feed intake, kg | 13.58 | 9.02 | 0.165 | <0.001 |
| Average gain, kg | 5.32 | 4.06 | 0.038 | <0.001 |
| Feed conversion ratio | 2.55 | 2.22 | 1.176 | <0.001 |
| Items | Treatment | p-Value | |
|---|---|---|---|
| Control | G. elegans-Treated | ||
| WBC, 109/L | 18.13 | 25.22 | 0.040 |
| LYMPH, 109/L | 8.51 | 12.13 | 0.023 |
| EOS, 109/L | 1.14 | 1.76 | 0.058 |
| LYMPH, % | 47.01 | 48.63 | 0.304 |
| EOS, % | 8.07 | 7.07 | 0.667 |
| RBC, 1012/L | 7.919 | 9.188 | 0.852 |
| HGB, g/L | 122.33 | 140 | 0.504 |
| MCV, fL | 81.75 | 78.58 | 0.027 |
| MCH, pg | 16.3 | 18.7 | 0.611 |
| MCHC, g/L | 202 | 236.5 | 0.287 |
| RDW_CV, % | 16.09 | 16.62 | 0.413 |
| RDW_SD, fL | 52.6 | 52.1 | 0.077 |
| HCT, % | 64.82 | 71.77 | 0.163 |
| PLT, 109/L | 256.2 | 984 | 0.785 |
| MPV, fL | 10.56 | 11.23 | 0.096 |
| PDW | 14.24 | 15.08 | 0.106 |
| PCT, % | 0.273 | 1.111 | 0.186 |
| Gene Symbol | Tissue | RNA-Seq | qRT-PCR | ||
|---|---|---|---|---|---|
| p-Value | Fold Change | p-Value | Fold Change | ||
| Atp2b3 | Liver | <0.001 | 3.57 | 0.014 | 2.39 |
| Mat2a | Liver | <0.001 | 2.18 | 0.008 | 1.70 |
| Chdh | Liver | 0.026 | 2.35 | 0.024 | 1.48 |
| Slc20a2 | Ileum | 0.006 | 2.05 | 0.010 | 1.89 |
| Slc28a1 | Ileum | 0.008 | 2.02 | 0.028 | 2.02 |
| Mt3 | Ileum | <0.001 | 3.49 | 0.008 | 3.75 |
| Metabolites | VIP | log2(Fold Change) | p-Value |
|---|---|---|---|
| Methylsulfonylmethane | 2.24 | 2.44 | <0.001 |
| 3-(4-Hydroxy-5-oxo-3-phenyl-2,5-dihydro-2-furanyl) propanoic acid | 1.46 | 1.58 | <0.001 |
| Visnagin | 1.53 | 1.66 | <0.001 |
| Beta-Naphthoxyacetic Acid | 1.57 | 1.70 | <0.001 |
| Menadiol | 1.41 | 1.52 | <0.001 |
| N-Feruloylserotonin | 2.29 | 2.52 | <0.001 |
| Dibenzo-1,4-dioxin | 1.45 | 1.55 | 0.001 |
| (-)-Akuammicine | 1.98 | 2.20 | 0.001 |
| 1-Naphthol | 1.38 | 1.51 | 0.002 |
| 4-Methylene-2-oxoglutarate | 1.45 | 1.58 | 0.002 |
| 5-Formyl-2-furoic acid | 1.45 | 1.60 | 0.003 |
| N-Acetyl-L-phenylalanine | 1.66 | 1.75 | 0.003 |
| Menadione | 1.18 | 1.32 | 0.005 |
| Phenylacetylglycine | 1.18 | 1.25 | 0.006 |
| Indole-3-carbidol | 1.47 | 1.53 | 0.010 |
| Furathiazole | 1.32 | 1.39 | 0.016 |
| 1-Stearoyl-2-arachidonoyl-sn-glycero-3-phosphoserine | 1.44 | 1.72 | 0.023 |
| Spirodiclofen | 2.35 | 2.35 | 0.028 |
| Pelargonidin | 1.19 | −1.20 | 0.030 |
| 2,5-Dimethyl-4-ethoxy-3(2H)-furanone | 1.11 | 1.35 | 0.041 |
| 7-alpha-Hydroxy-3-oxochol-4-en-24-oic acid | 1.31 | 1.47 | 0.047 |
| Metabolites | VIP | log2(Fold Change) | p-Value |
|---|---|---|---|
| Maslinic acid | 2.54 | 2.19 | 0.001 |
| 10-Acetyl-9-hydroxy-7,7-dimethyl-2,6,6a,7,11a,11b-hexahydro-11H-pyrrolo[1′,2′:2,3]isoindolo[4,5,6-cd]indol-11-one | 2.23 | 1.99 | 0.003 |
| Adipic acid | 2.17 | 1.94 | 0.004 |
| 4-[(2-Isopropyl-5-methylcyclohexyl)oxy]-4-oxobutanoic acid | 1.14 | −0.90 | 0.008 |
| Glycine | 1.23 | 1.02 | 0.009 |
| Imazamethabenz | 1.39 | 1.13 | 0.009 |
| p-Tolyl beta-d-glucuronide | 1.50 | 1.23 | 0.011 |
| Phenylacetylglycine | 1.45 | 1.18 | 0.011 |
| (+)-CP 55,940 | 1.71 | 1.42 | 0.011 |
| d-(-)-Quinic acid | 2.08 | 1.89 | 0.013 |
| (-)-CP 55,940 | 2.00 | 1.65 | 0.013 |
| Cascarillin | 1.07 | −0.81 | 0.020 |
| Picrasin C | 1.14 | −0.84 | 0.026 |
| 10-[4-(2,4,4-Trimethyl-2-pentanyl) phenoxy]-1-decanol | 2.08 | −1.48 | 0.038 |
| N-Acetyl-L-cysteine | 1.06 | 0.95 | 0.041 |
| 4-Methylphenol | 1.47 | 1.16 | 0.043 |
| Actinoquinol | 1.11 | −0.81 | 0.046 |
| Glycol stearate | 2.35 | −1.79 | 0.49 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, C.-Y.; Yang, K.; Cao, J.-J.; Wang, Z.-Y.; Wu, Y.; Sun, Z.-L.; Liu, Z.-Y. Integration of Metabolomics and Transcriptomicsto Comprehensively Evaluate the Metabolic Effects of Gelsemium elegans on Pigs. Animals 2021, 11, 1192. https://doi.org/10.3390/ani11051192
Huang C-Y, Yang K, Cao J-J, Wang Z-Y, Wu Y, Sun Z-L, Liu Z-Y. Integration of Metabolomics and Transcriptomicsto Comprehensively Evaluate the Metabolic Effects of Gelsemium elegans on Pigs. Animals. 2021; 11(5):1192. https://doi.org/10.3390/ani11051192
Chicago/Turabian StyleHuang, Chong-Yin, Kun Yang, Jun-Jie Cao, Zi-Yuan Wang, Yong Wu, Zhi-Liang Sun, and Zhao-Ying Liu. 2021. "Integration of Metabolomics and Transcriptomicsto Comprehensively Evaluate the Metabolic Effects of Gelsemium elegans on Pigs" Animals 11, no. 5: 1192. https://doi.org/10.3390/ani11051192
APA StyleHuang, C.-Y., Yang, K., Cao, J.-J., Wang, Z.-Y., Wu, Y., Sun, Z.-L., & Liu, Z.-Y. (2021). Integration of Metabolomics and Transcriptomicsto Comprehensively Evaluate the Metabolic Effects of Gelsemium elegans on Pigs. Animals, 11(5), 1192. https://doi.org/10.3390/ani11051192






