Simple Summary
Three weighted-stratified random-effects meta-analyses were performed to estimate the worldwide neonatal calf diarrhoea prevalence of mixed infections of the causative agents bovine rotavirus (BRV), bovine coronavirus (BCoV), Escherichia coli K99 (ETEC) and Cryptosporidium spp. (Crypto). The highest worldwide mean pooled prevalence was identified for BRV-Crypto (6.69%; confidence interval (CI): 4.27–9.51), followed by BRV-BCoV (2.84%; CI: 1.78–4.08) and BRV-ETEC (1.64%; CI: 0.76–2.75). In all concurrent infections with BRV, the highest mean prevalence was identified in calves with diarrhoea, in dairy herds and in the age classes of sampled animals between 0–14 days. The prevalence of the BRV-BCoV mixed infection is higher than expected based on the ratio of the occurrence of both individual infections in calves with diarrhoea.
Abstract
Multiple enteropathogens such as bovine rotavirus (BRV), bovine coronavirus (BCoV), Escherichia coli K99 (ETEC) and Cryptosporidium spp. (Crypto) are the most common causes of calf diarrhoea during the first 30 days of animal age. Three weighted-stratified random-effects meta-analyses were performed to calculate the worldwide prevalence of mixed infections of the causative agents (i.e., BRV-BCoV, BRV-ETEC, BRV-Crypto) and their potential influencing factors. The meta-analysis covered 41 studies (94 sub-studies) in 21 countries that determined the presence or absence of mixed infections in global calf populations. The highest worldwide estimated pooled prevalence was identified for BRV-Crypto (6.69%), followed by BRV-BCoV (2.84%), and BRV-ETEC (1.64%). The chance of detecting BCoV in calves with diarrhoea was 1.83 higher in the presence of BRV compared to calves without BRV, whereby an inhibition effect (odds ratio: 0.77) was determined between BRV and Crypto infections. The diagnostic methods were identified as a significant influencing factor in the detection of all considered mixed infections, while the other analysed factors differed in relation to their effect on prevalence. In contrast to BRV-BCoV, the prevalence of BRV-ETEC and BRV-Crypto mixed infections followed the course of individual ETEC and Crypto prevalence related to the age class of the sampled animals.
1. Introduction
Neonatal calf diarrhoea (NCD) is a well-known worldwide disease in the cattle industry which causes substantial economic losses due to high morbidity, mortality, growth retardation and treatment costs, as well as serious long-term consequences such as delayed first calving [1,2,3,4,5,6,7,8,9]. NCD is the most common cause of death in dairy calves during their first 30 days of age with a case fatality risk of approximately 5% [10,11].
Multiple enteric pathogens, viral (e.g., bovine rotavirus, bovine coronavirus, bovine viral diarrhoea virus), parasitic (e.g., Cryptosporidium parvum, Giardia duodenalis, Eimeria spp.) and bacterial (e.g., Escherichia coli K99, Salmonella spp., Clostridium perfringens) are infectious causative agents of NCD [1,12,13,14]. Bovine rotavirus (BRV), bovine coronavirus (BCoV), enterotoxigenic Escherichia coli K99 (ETEC) and Cryptosporidium parvum are the most frequently identified causative factors of calf diarrhoea during the first 30 days of age [15,16,17,18], whilst BRV is the most commonly involved pathogen in mixed infections [14,19,20]. Neonatal calves are most susceptible to infections with ETEC in the first four days of life [15,21]. In the first to second week of age, infections with BRV are most common, whilst infections with BCoV occur more frequently from days five to 20 [22]. Between the first and the third week, calves are most susceptible to infections with Cryptosporidium parvum [23]. To the best of our knowledge, an overview about mixed infections of these pathogens related to animal age across the literature is not available yet.
Concurrent infections of these infectious causative agents are often observed, in particular in calves with diarrhoea compared to healthy calves [17,24,25]. Besides pathogens themselves, there are other factors such as applied diagnostic methods, management-related factors (e.g., dam vaccination, colostral consumption, herd size, biosecurity practice, calf housing, hygienic condition, separation of animal based on age, feeding), and environmental factors (e.g., season of birth) that may influence recording of the occurrence and/or prevalence of enteropathogens [10,26,27,28,29,30].
The objectives of this study were (i) to review the literature systematically regarding the prevalence of BRV infections in combination with BCoV (i.e., BRV-BCoV), ETEC (i.e., BRV-ETEC) and Cryptosporidium spp. (Crypto) (i.e., BRV-Crypto; N.B. a differentiation of the species is not possible with formerly commonly used diagnostic methods like acid-fast staining, the analysis here is based on the genus level of Cryptosporidium spp. [31]) and potential influencing factors; (ii) to perform weighted random-effects meta-analyses to estimate the overall pooled prevalences across the worldwide studies and to identify sources of heterogeneity of prevalences among the study outcomes (referred as subgroup-analysis), (iii) to determine the statistical influence of potential influencing factors on the reported prevalences of concurrent infections; (iv) to analyse the chance that one of the three pathogens occur in the presence of BRV; (v) to determine the expected prevalence of mixed infection in calves with diarrhoea, assuming that both considered causative agents occur independent from each other and (vi) to model the worldwide prevalence of mixed-infection depending on the age class of sampled animals.
2. Materials and Methods
A systematic literature search was conducted to identify studies focusing on the prevalence of mixed infections (i.e., BRV-BCoV, BRV-ETEC, BRV-Crypto). Three online databases were used, considering publications until June 2020: PubMed, Scopus and Web of Science. The following predefined search terms were used to identify the greatest possible number of publications: (neonatal calf diarrhea OR calf diarrhoea OR diarrheic calves OR diarrhoeic calves OR pre-weaned) AND (prevalence) AND (mixed infection OR concurrent infection OR co-infection). Due to the large number of articles returned in Scopus and Web of Science, the search terms were set in quotation marks to ensure that the online databases only return publications with the exact sequence of words. Studies returned by the online databases were defined as ‘primary literature’ and were screened in full by one reviewer (MB) regarding the predefined criteria shown in Table 1 and were reviewed again for validation by one reviewer (FR). Additionally, the reference lists of the primary literature were reviewed regarding article title and abstract for further appropriate studies (MB). Studies from the reference lists were defined as ‘secondary literature’. Uncertainties regarding the inclusion and/or recording of data from the studies were discussed between all authors until a consensus was reached.

Table 1.
Collected data of the neonatal calf diarrhoea prevalence studies and analysed criteria in the meta-analysis and multivariate regression analysis.
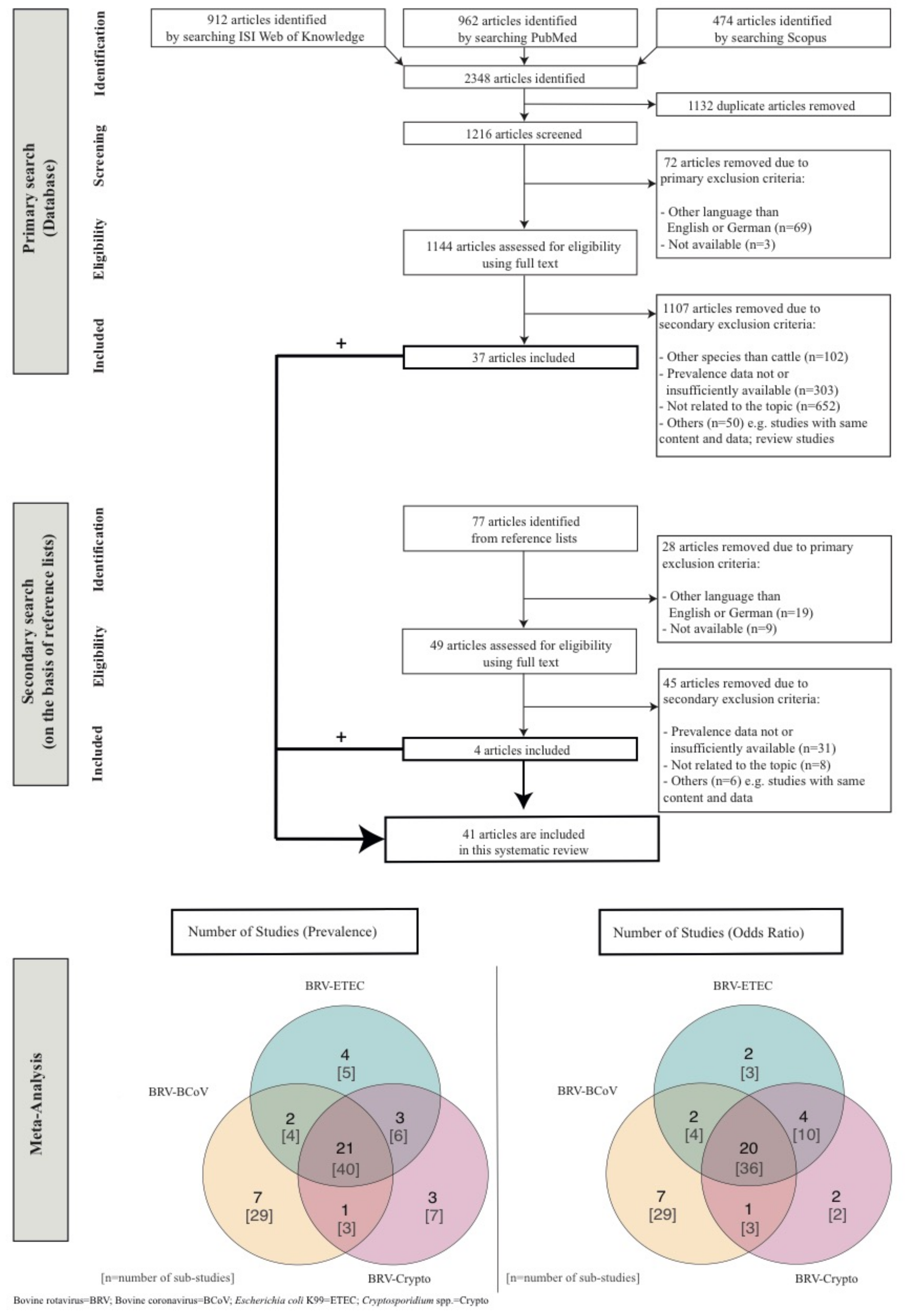
The number of identified studies (primary and secondary literature) and the study selection workflow, in accordance with the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analysis) guidelines, are presented in Figure 1. The data collected (Table 1) from the studies (i.e., prevalences of concurrent infections (i.e., BRV-BCoV, BRV-ETEC, BRV-Crypto), occurrence of the individual enteropathogens (BRV, BCoV, ETEC, Crypto), geographical region, sampling period, number of herds, herd type, age of sampled animals, health status, number of tested animals, sample type, genotypes, vaccination status, colostrum intake, diagnostic methods, study type) were entered into a Microsoft Excel datasheet Version 16.16.27 (2016). A study was divided into sub-studies if the study covered differences in e.g., herd type, health status and animal age. Because of the consideration of sub-studies, the total number of publications included in the presented study is thus not identical to the total number of observations. The criteria for study inclusion were (i) focusing on diarrhoea in calves aged ≤ 60 days; (ii) reporting prevalences of BRV-BCoV, BRV-ETEC and/or BRV-Crypto as percentage and/or total number of tested and positively tested calves; (iii) consideration of more than one herd and (iv) only original studies on prevalence data. Although NCD is the most common cause of death in dairy calves during their first 30 days of age, we considered studies with age ranges up to 60 days in the analysis because many of the studies published age ranges including animals older than 30 days. Further, age ranges up to 60 days were considered to get a better impression of the development of mixed infection in both dairy and beef production systems instead of single infection in one production system, and to confirm the knowledge in the literature that NCD most frequently appears in the first 30 days of age. A tested animal corresponds to one sample in the analysed studies. All published mixed infections (i.e., double, triple and quadruple) were considered. For instance, BRV-BCoV-Crypto triple infections were incorporated in the analyses for BRV-BCoV and BRV-Crypto, respectively.

Figure 1.
Flow chart of studies incorporated in the systematic review and meta-analysis.
The prevalence of mixed infections with BRV (i.e., BRV-BCoV, BRV-ETEC, BRV-Crypto) were analysed in three weighted-stratified meta-analyses using random effect models. The meta-analyses were used to estimate the worldwide pooled prevalences of the mixed infections in the sampled animals. The prevalences were weighted on the inverse of within-study variance and the variability across the studies, according to the PM (Paule and Mandel) method (Supplementary Material I) [32,33]. For variance-stabilisation of the prevalence data distribution, Freeman–Tukey double arcsine transformation was used [34]. The corresponding back-transformation was conducted according to the approach by Miller (Supplementary Material I) [35]. To validate our approach, we used the REML (restricted maximum likelihood) method instead of the PM, for model fitting, whereas both sub-studies and studies were used simultaneously as random factors. To determine the heterogeneity of the incorporated studies in the meta-analysis, i) the Higgins inverse variance (I2) index (i.e., the percentage of total variation across the studies) and ii) the Cochran’s Q-Test (i.e., degree of between study variance, whereby p < 0.05 indicated heterogeneity) was calculated. I2 greater than 50% indicated substantial heterogeneity between studies (I2 lay between 0 and 100%) [36,37]. Both, I2 and Cochran’s Q-Test provide no information about the factors which cause the heterogeneity [38]. Thus, a weighted-stratified random-effects meta-analysis (subgroup-analysis) based on the factors in Table 1 was performed in order to identify the possible source of heterogeneity. To avoid imprecise calculation, factors incorporating less than 75% data were excluded from the subgroup-analysis (Table 1). The Egger test and a regression test for funnel plot asymmetry were conducted to identify publication bias. The outliers were identified by performing an influential case diagnostic (i.e., DFFITS (difference in fits) value, covariance ratios, estimates of τ2, Cook’s distances and test statistics for (residual) heterogeneity, see Supplementary Figure S1) [39,40]. The pooled prevalences for concurrent infections of each study and their weight contribution proportion to the meta-analyses was stratified by the health status of the calves and bounded by 95% confidence intervals (CIs) (Supplementary Figures S2–S4).
Uni- and multivariate meta-regression analyses were performed based on the approach by Scharnböck et al., 2018 to determine the potential significant influence of factors in Table 1 and their explainable proportion on the variability (R2) of prevalences of BRV-BCoV, BRV-ETEC and BRV-Crypto [13]. The final multivariate regression analysis includes only most relevant factors without declining the model-fit accuracy. The most relevant factors were non-correlated (N.B. association between the factors were analysed using Goodman–Kruskal–tau), significant factors from the univariate meta-regression analysis not altering the R2 by more than 10% of the full multivariate regression model and provided the lowest Akaike information criteria, corrected for small sample size (AICc). The estimated overall mean prevalences of the concurrent infections from the sub-group meta-analysis were used to model the prevalences of mixed-infection depending on the age with the Loess algorithm (Supplementary Material I). The same approach was applied for the prevalence of each of the four considered pathogens because in contrast to mixed infection, knowledge about the prevalence of the individual four pathogens as a function of age is already known. Thus, if the course of the individual prevalences related to animal age matched the knowledge in the literature, we considered the approach as valid for the mixed-infections.
Additionally, we calculated the expected prevalence of each mixed infection under assumption of independency of both considered causative agents. We investigated whether the expected prevalence of the mixed infections was higher or lower as expected based on the ratio of the occurrence of both individual infections in calves with diarrhoea. In order to analyse the association between two individual pathogens, the OR (odds ratio) based on the absolute frequencies of the detected individual pathogens was used as an effect size for the meta-analysis. This allows us to quantify the OR for one pathogen (i.e., BCoV, ETEC, Crypto) when BRV was present. Both pathogens occur independently, if the OR = 1, while OR >1 or <1 indicated dependency. The meta-analyses were implemented in R (Version 3.4.1 R Foundation for Statistical Computing, Vienna, Austria) using the “metafor” and “GoodmanKruskal” package [41,42].
3. Results
In total, 41 (94 sub-studies) from 1293 studies in 21 different countries were included in the meta-analysis (Figure 1). In total, 12,208 animals in approximately 2110 herds were tested for concurrent infections worldwide. The highest worldwide mean pooled prevalence (Table 2, Table 3 and Table 4) was identified for BRV-Crypto (6.69%; CI: 4.27–9.51), followed by BRV-BCoV (2.84%; CI: 1.78–4.08) and BRV-ETEC (1.64%; CI: 0.76–2.75). The regression test for funnel plot asymmetry shows no publication bias (BRV-BCoV: z = 0.41, p = 0.67; BRV-ETEC: z = 1.59, p = 0.11; BRV-Crypto: z = −0.25, p = 0.79), no outliers (Supplementary Figure S1) and no multicollinearity issues across all mixed infections. The validation of the meta-regression analysis with the restricted maximum likelihood (REML) instead of Paule and Mandel (PM) shows no significant differences in the meta-results, no outliers and no publication bias.

Table 2.
Subgroup meta-analysis of studies reporting the concurrent prevalence of bovine rotavirus (BRV) and bovine coronavirus (BCoV). N.B. Detailed description of the factors is provided in Table 1.

Table 3.
Subgroup meta-analysis of studies reporting the concurrent prevalence of bovine rotavirus (BRV) and Escherichia coli K99 (ETEC) N.B. Detailed description of the factors is provided in Table 1.

Table 4.
Subgroup meta-analysis of studies reporting the concurrent prevalence of bovine rotavirus (BRV) and Cryptosporidium spp. (Crypto) N.B. Detailed description of the factors is provided in Table 1.
The geographical distribution demonstrated that the majority of BRV-BCoV infections were identified in Europe (4.72%; CI: 2.49–7.45), while the highest prevalences of BRV-ETEC (3.70%; CI: 0.32–9.39) and BRV-Crypto (16.61%; CI: 8.03–27.19) were determined in West Asia. In all concurrent infections with BRV, the highest mean prevalence was identified in calves with diarrhoea, in dairy herds and in the age classes of sampled animals between 0–14 days (Table 2, Table 3 and Table 4). The lowest pooled prevalences of the mixed infections were identified in case-control studies. In contrast to BRV-BCoV, the prevalence of BRV-Crypto increased over time (from 1980: 2.01%; CI: 0.00–11.65 to 2011–2019: 9.07%; CI: 4.72–14.44). In this context, the highest pooled prevalence was identified for the more recent diagnostic methods such as lateral flow immunochromatographic assay (BRV-Crypto; RA: 13.49%; CI: 6.80–21.74) in contrast to methods frequently applied in the past such as acid-fast staining (BRV-Crypto; MS: 3.44%; CI: 0.85–7.16; Table 4). Diagnostic methods were identified as a significant influencing factor in the uni- and/or multivariate-meta-regression analyses over all considered mixed infections. The significance and explained variance of the remaining factors on the worldwide prevalences differ between the concurrent infections and is shown in Table 5.

Table 5.
Uni- and multivariate meta-regression analysis stratified by factors and type of double mixed infection.
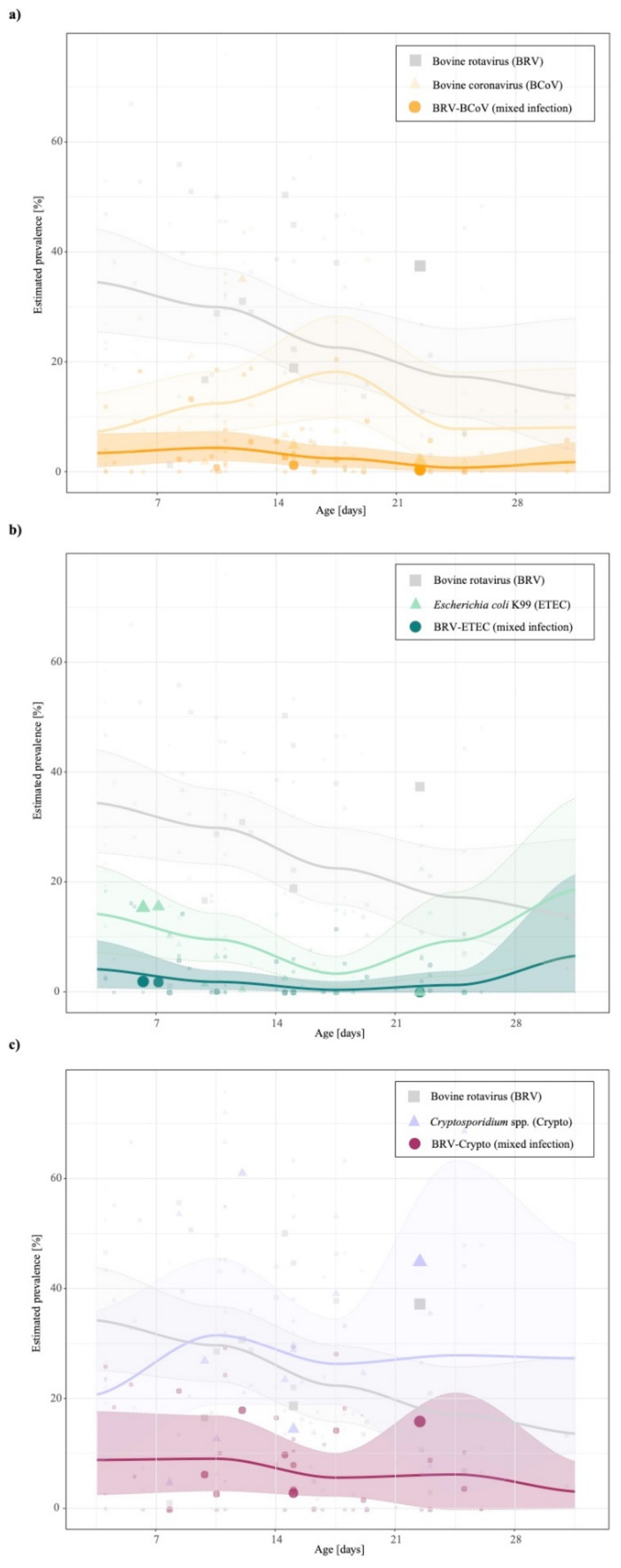
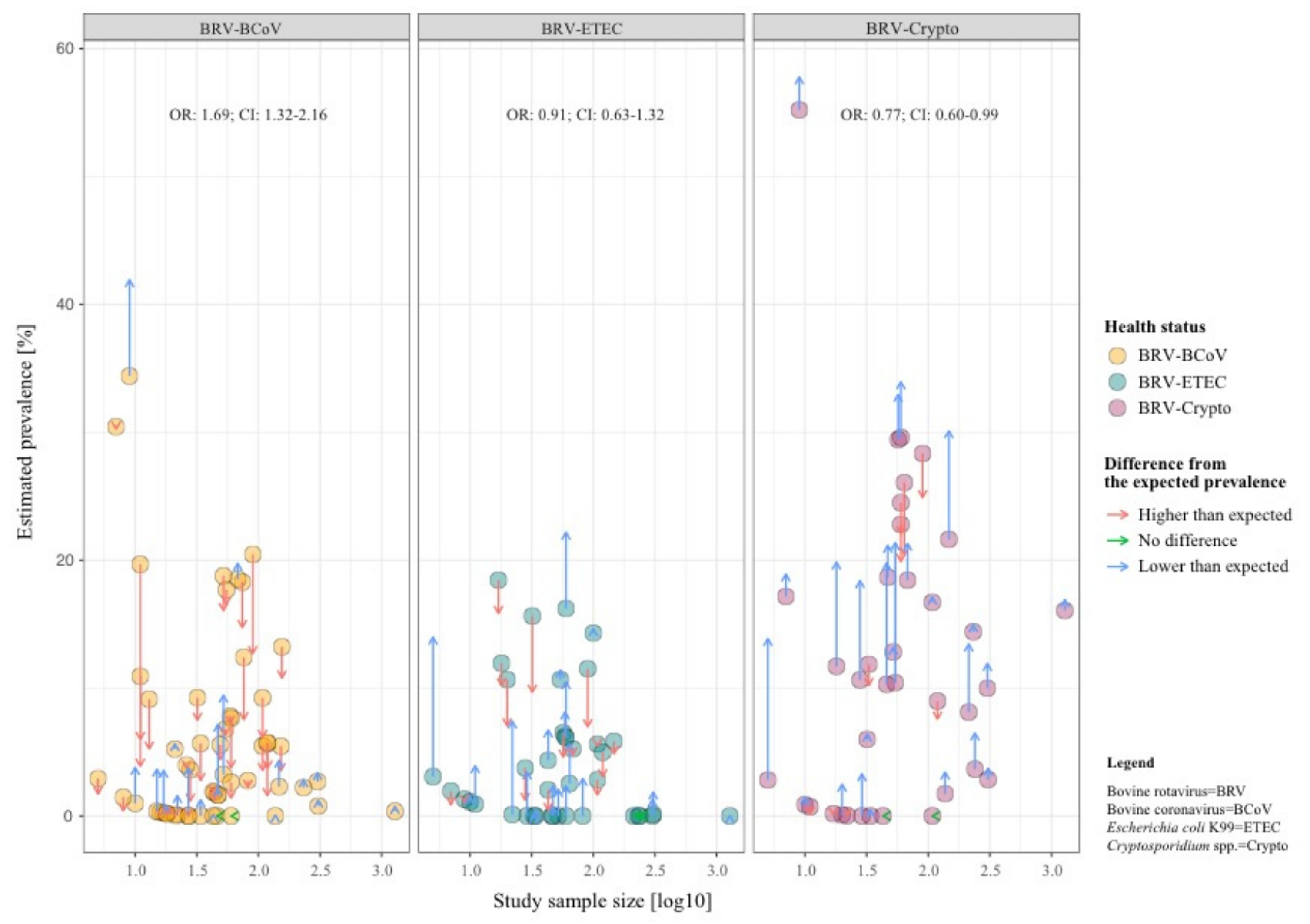
Our study results confirm that the most concurrent infections occur in dairy and beef production systems in an age range up to 30 days (see Table 2, Table 3 and Table 4). The highest mean prevalence of BRV-BCoV (BRV-ETEC and BRV-Crypto) was identified in animals aged between 7–14 days under consideration the sample size (BRV-ETEC: 0–7 days and BRV-Crypto: 7–14 days). Figure 2 shows that in contrast to BRV-BCoV, the prevalence of BRV-ETEC and BRV-Crypto mixed infections follow the course of the individual ETEC and Crypto prevalence related to the age class of sampled animals. The prevalence of the BRV-BCoV mixed infection is higher than expected based on the ratio of the occurrence of both individual infections in calves with diarrhoea (Figure 3). The chance/odds ratio (OR) to detect BCoV in calves was 1.83 (CI: 1.48–2.27) times higher in the presence of BRV compared to calves without BRV, whereby an opposite effect was identified for BRV-Crypto infections (OR 0.77; CI: 0.60–0.99).

Figure 2.
Temporal analysis of the individual (a) grey: bovine rotavirus; (b) orange: bovine coronavirus; (c) green: Escherichia coli K99; purple: Cryptosporidium spp.) and mixed prevalences stratified by age class of sampled animals until 30 days. The lines represent the mean prevalence estimates of all considered studies with the corresponding 95% CI (area) and individual prevalence points of studies (dots) during the period observed. The more prevalence estimates available at a certain age class of sampled animals, the wider the dots. N.B. To avoid imprecise model predictions, studies in the age groups (28–49 days) were excluded from the temporal curve fitting due to the small number of available studies (see number of available studies in Table 2, Table 3 and Table 4).

Figure 3.
Comparison of the detected prevalence of mixed infections (dots) and the expected prevalence of infections (arrowheads) in calves with diarrhoea under the assumption of independency between both pathogens (e.g., P (BRV ∩ BCoV) = P(BRV) × P(BCoV)). Dots with blue arrows represent data with a lower prevalence than we would expect in an independent co-infection, while dots with red arrows represent data with a higher prevalence, as expected.
4. Discussion
To assess studies with specific focus on NCD prevalences caused by BRV in combination with BCoV, ETEC and Crypto, we reviewed 1193 studies in full, of which 41 studies were incorporated in the meta-analysis presented here. BRV was used as reference for the comparison because it is the most common infectious causative agent in combination with other pathogens of NCD [14,19,20]. As far as the authors are aware, this is the first worldwide meta-analysis to be carried out regarding the concurrent infections of NCD. In contrast to other systematic reviews and meta-analyses with similar focus, our study focused on calves in the most vulnerable age class and took into account the interaction of several pathogens instead of pathogens tested individually [44,45,46].
The results presented here revealed a wide variation in the prevalence of considered mixed infections and their significant influencing factors. The considered causative agents in the presented study cover three (i.e., viruses, bacteria, parasites) of five classes of pathogens in different combinations which differ in their pathogenicity, virulence, infectivity and environmental resistance [47]. This might explain the heterogeneous distribution of the prevalences as well as why the factors differ regarding their significant influence and explained variance on the worldwide prevalences (Table 5).
It is only useful to a limited extent to discuss in detail specific factors on the level of prevalence. For instance, the factor “geographical region” covered several country-specific factors such as average herd size, general law standards, typical husbandry systems, trading systems. All these factors might have a direct or indirect effect on the biosecurity level on farms. For example, Sahlström and colleagues showed that larger farms tend to have higher levels of biosecurity [48], resulting in potentially higher prevalence of infections for areas with smaller farm sizes on average. We assume that this effect might explain the higher prevalence of BRV-BCoV (4.72%; CI: 2.49–7.45) and BRV-Crypto (8.90%; CI: 4.98–13.65) in Europe with smaller structured holdings [49] compared to other regions (Table 2 and Table 4). However, it has been described that factors which influence biosecurity, such as herd size, can also have a direct influence on the incidence of infection [50]. As an example, an accurate uptake of colostrum reduces infections with ETEC [51]. Barry and colleagues showed that calves in smaller herds tend to have higher immunoglobulin G levels [52] which might be a consequence of better colostrum management and/or quality [53].
The results of the meta-analysis presented here confirm the results of several studies [17,24,25] that mixed infections are more common in calves with diarrhoea (BRV-BCoV: 4.22%; BRV-ETEC: 2.26%; BRV-Crypto: 9.41%) than in healthy calves (BRV-BCoV: 0.00%; BRV-ETEC: 0.13%; BRV-Crypto: 0.00%). The lowest prevalences were found across all mixed infections in case-control studies compared to case studies. This can be explained by the fact that as well as calves with diarrhoea, healthy animals were also included. In contrast to BRV-BCoV and BRV-ETEC, an increase in BRV-Crypto prevalence was identified during the period. This can primarily be explained by the use of more sensitive diagnostic methods from 2011 onwards. For instance, the use of microscopy (MS) for Crypto detection (as a single detection method) was mainly found in the studies dated before 1991 and is less sensitive than other diagnostic methods (see diagnostic factor: Several in Table 2, Table 3 and Table 4 and Table 1) used since then. Several authors also reported increasing Crypto prevalence due to more sensitive diagnostic methods [45,54]. For example, the prevalence determined with RA was approximately four times higher than that of MS (Table 4).
The uni- and multivariate regression analysis revealed that the factor “diagnostic method” had a significant impact on the detected prevalence of BRV-BCoV, BRV-ETEC, BRV-Crypto. Although the collected factors in the study presented here can explain a high variance of BRV-BCoV prevalences (R2 = 61.23%), it is much less appropriate in the case of BRV-ETEC (R2 = 47.83) and BRV-Crypto (R2 = 46.20%), which might indicate the presence of other essential factors which were not considered in this study presented here due to the lack of reporting in the literature. These could include factors such as (i) vaccination status of the dam, because colostrum of immunized dams could increase the antibody titre of calves against BRV, BCoV, ETEC in the first month of the animal life [30]. Thus, colostral consumption can decrease the neonatal diarrhoea prevalence, and also reduce shedding of Cryptosporidium parvum [55,56]. Information about colostral consumption of calves and vaccination status of dams were specified in 4 (9.76%) and 11 studies (23.83%) respectively. Both factors were not incorporated in the meta-analysis due to the low number of studies (Table 1); (ii) season of sampling, because calves born in the winter season have higher risk of diarrhoea [29] due to lower colostrum quality of the dam [57] and a higher shedding of pathogens (e.g., Cryptosporidium oocysts) throughout the winter season compared to summer [58,59]. Further factors which might influence the prevalences are (iii) e.g., feeding, animal stock intensity and regulations to protect calves [60]. As already mentioned, some of these factors could be indirectly included in the factor “geographical region”. One of the influencing factors that was also not considered in the study presented here is the role of the gut microbiome during pathogenesis at the site of infection in the early life of an animal, and the host-microbial interactions with dietary interventions. A number of studies have analysed the effect of the microbiome composition on new-born health such as on the calf gastrointestinal tract [61,62,63,64,65]. A limitation of our meta-analysis is that the reported prevalences in the studies were not corrected for the varying levels of sensitivity and specificity of the diagnostic tests used (also referred to as apparent prevalence). N.B. only nine studies (21.95%) provided information on the sensitivity and specificity of the applied diagnostic methods (Table 1). Consequently, the worldwide estimated prevalences could be under- and/or overestimated in the presented meta-analysis. Additionally, the estimated worldwide prevalences could be under- and/or overestimated due to our predefined exclusion criteria and/or because studies may not have been identified by the chosen database, search terms and language restrictions. Furthermore, the relatively small number of studies per factor does not allow us to take into account the interaction between the factors. Such interaction would be essential to interpret the results of the subgroup analysis more accurately. For instance, the main reason why the implementation of the antibody-based methods is not reflected in the level of BRV-BCoV infections (Table 2) might be explained by the increasing number of case-control studies since 2001 and thus would explain the decrease in BRV-BCoV prevalence from this year onwards. In general, the results of the subgroup analysis should be interpreted with caution concerning the sample size, the number of studies and broad definition of subgroup factors. The broad definition of factors was used to avoid an unbalanced number of studies per analysed factor. For instance, instead of a range or mean of animal age, it would be more appropriate for epidemiological prevalence studies to summarise the age groups at intervals of seven days and, especially in the first week of age, on a daily basis. This suggestion is supported by other studies [15,21] reporting for example that ETEC frequently occurs in the first four days of animal age. The ability of the Escherichia coli K99 antigen to bind on the mucous membrane of the small intestine is age-dependent and gradually decreases from 12 h of animal age [66]. The latter might also explain the course of the prevalence level illustrated in Figure 2. An increase of ETEC after the 3rd week of animal life was observed in the study presented here, a result which was also reported by Izzo and colleagues [67]. This could be related to an immunological gap caused by the decrease of maternal antibodies, while the antibody protection of the calf is not yet sufficient [68]. This decrease in maternal antibodies could also explain the increase of BCoV prevalences in the third week of animal life. Figure 2 shows that BRV prevalence peaks in the first age class of sampled animals, which is a consequence of the short incubation period of 24 h of BRV in combination with a higher susceptibility in this age class [22].
The course of the prevalences of the individual pathogens related to the age class of the sampled animals presented in this study is in accordance with several studies, testing the age dependencies of prevalences of these pathogens [21,23,69,70]. Figure 2 shows that in contrast to BRV-BCoV, the BRV-ETEC and BRV-Crypto mixed infection follows the course of the individual ETEC and Crypto prevalences related to the age class of sampled animals. A prolonged susceptibility and a synergistic interaction between BRV-ETEC has been proven experimentally [71,72,73,74,75]. This observation could be explained by taking into account the fact that rotavirus infection induces important changes in the cytoskeleton which correlate with a decrease in apical expression of disaccharidase [76]. This reduced disaccharidase activity on the cell surface, regardless of whether there is cell damage or not [77], could encourage the growth of bacteria, as described in several studies [15,78]. This synergistic effect has been described in the literature for the youngest age group which we could not analyse due to the broad and insufficient detail description of animal ages in the analysed studies. The results of our meta-analysis did not indicate a synergistic effect across all age groups, since the OR of a simultaneous infection of BRV and ETEC was not significant (OR: 0.94; 95% CI: 0.67–1.31; Figure 3).
Figure 3 indicates a synergistic effect between BRV and BCoV (OR:1.69; 95% CI: 1.32–2.16) and an inhibitory effect between BRV and Crypto (OR:0.77; 95% CI: 0.60–0.99). The former could be related to the fact that both BRV and BCoV increasingly cause diarrhoea in calves with failure of passive transfer [79], whereby weakening of the calf by one pathogen could also have a beneficial effect on other pathogens. In contrast to BRV, BCoV does not only infect the mature enterocytes in the small intestine but also the crypt cells and colonocytes [22,80]. The latter could be related to the fact that in the event of an infection with rotavirus endotoxin non-structural protein 4 (NSP4) is produced intracellularly and the upregulation of Ca2+ has an influence on the Ca2+-sensitive proteins F-actin, villin, and tubulin, resulting in damage of the microvillar cytoskeleton of the cell [77,81,82]. This or a similar pathophysiological effect might have an influence on the Crypto-binding capacity on the cell damaged by BRV. For example, Chen and colleagues described a decrease in infection of up to 70% with Cryptosporidium parvum induced by 2-actin depolymerisation in a vivo experiment [83]. This does not apply to a reverse appearance of infection as Tzipori and colleagues showed in lambs, where a previous infection with Crypto had no effect on BRV [84].
It is desirable to analyse prevalence data from numerous studies within the meta-analysis as it provides a more general overview of the influencing factors across the literature and countries. Thus, results of the meta-analysis are more powerful and less biased than conventional statistical methods and/or results of an individual study regarding NCD prevalences [13,38,85]. However, our study indicates the need for more standardised epidemiological studies to provide more robust conclusions regarding the importance of pathogens and their influencing factors. For instance, knowledge about the impact of other factors (e.g., vaccination of dams [30,55], supplementation of colostrum in the first two weeks of animal life [86,87], calf housing (place and individual vs. group pens) [28,29], routinely disposal and cleaning of bedding [26] as well as cleaning of feeding utensils [88] or quarantine of purchased animals [48]) on prevalence could help the livestock owner to reduce the direct production losses caused by NCD. The analysis of the effectiveness of specific measures against pathogens for which no vaccines are available yet (e.g., Crypto) would be essential to reduce the spread of zoonotic pathogens (in particular to minimise the health risk to farmers) and/or to reduce the use of drugs (in particular antibiotics). The authors also recommend taking into account the genotyping of the pathogens to identify possible mutations, to reassess if the vaccination strains match the field strains and to increase the understanding of the transmission of (zoonotic) pathogens. N.B. BRV and Crypto are important pathogens of diarrhoea in children and immunocompromised adults [89,90]. To determine the impact of potential influencing factors on the level of reported prevalences incurred by BRV-BCoV, BRV-ETEC, BRV-Crypto infections, it is desirable to have detailed and additional information on the prevalence, pathogens and animals, as shown in the study presented here.
5. Conclusions
As far as the authors are aware, this is the first worldwide meta-analysis to be carried out regarding the mixed infections of NCD. The results presented here revealed (i) a wide variation in the prevalence of the considered concurrent infections. The global prevalence of BRV-Crypto in calves (6.9%) was twice as high compared to that of BRV-BCoV (2.84%) and four times higher than BRV-ETEC (1.64%); (ii) calves with diarrhoea and in the age classes of sampled animals between 0–14 days showed the highest worldwide prevalence; iii) the chance to detect BCoV in calves with diarrhoea was higher in the presence of BRV compared to calves without BRV, whereby an inhibition effect was determined between BRV and Crypto infections; iv) diagnostic methods were identified as a significant influencing factor in detecting the considered mixed infections, while other factors differ related to their significance and explained variance on prevalences.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/ani11041014/s1, Supplementary Material I: Detail description of the methodology, Figure S1: Funnel plot and influential case diagnostic for studies covering (a) BRV-BCoV mixed infections, (b) BRV-ETEC mixed infections and (c) BRV-Crypto mixed infections. N.B. no outliers were identified for all mixed infections., Figure S2: Forest plot of studies with BRV-BCoV prevalences ordered by health status of the calves and publication year. N.B. The full references are provided at the end of the Supplementary Material., Figure S3: Forest plot of studies with BRV-ETEC prevalences ordered by health status of the calves and publication year. N.B. The full references are provided at the end of the Supplementary Material., Figure S4: Forest plot of studies with BRV-Crypto prevalences ordered by health status of the calves and publication year. N.B. The full references are provided at the end of the Supplementary Material.
Author Contributions
Conceptualization, M.B., F.-F.R. and B.C.; methodology, M.B., F.-F.R. and B.C., software, F.-F.R. and B.C.; validation, M.B. and F.-F.R.; formal analysis, M.B.; investigation, M.B., F.-F.R. and B.C.; resources, B.C.; data curation, M.B., F.-F.R.; writing—original draft preparation, M.B. and B.C.; writing—review and editing, M.B., F.-F.R. and B.C., visualization, F.-F.R. and B.C.; supervision, B.C.; project administration, B.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable for studies not involving humans or animals.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available in the supplementary material. The digital datasets and analyses of the present study are available from the corresponding author upon request.
Acknowledgments
We would like to show our appreciation to Isabella Schatzer and Werner Hirschmugl from the Library of the University of Veterinary Medicine Vienna for they support of the procurement of the literature. Open Access Funding by the University of Veterinary Medicine Vienna.
Conflicts of Interest
The authors declare no conflict of interest. None of the authors of this paper has a financial or personal relationship with other people or organisations that could inappropriately influence or bias the content of the paper.
References
- Cho, Y.-I.I.; Yoon, K.-J.J. An overview of calf diarrhea—Infectious etiology, diagnosis, and intervention. J. Vet. Sci. 2014, 15, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Østerås, O.; Gjestvang, M.S.; Vatn, S.; Sølverød, L. Perinatal death in production animals in the Nordic countries—Incidence and costs. Acta Vet. Scand. 2007, 49, 1–4. [Google Scholar] [CrossRef]
- Donovan, G.A.; Dohoo, I.R.; Montgomery, D.M.; Bennett, F.L. Calf and disease factors affecting growth in female Holstein calves in Florida, USA. Prev. Vet. Med. 1998, 33, 1–10. [Google Scholar] [CrossRef]
- Waltner-Toews, D.; Martin, S.W.; Meek, A.H. The effect of early calfhood health status on survivorship and age at first calving. Can. J. Vet. Res. 1986, 50, 314–317. [Google Scholar]
- Pinior, B.; Firth, C.L.; Richter, V.; Lebl, K.; Trauffler, M.; Dzieciol, M.; Hutter, S.E.; Burgstaller, J.; Obritzhauser, W.; Winter, P.; et al. A systematic review of financial and economic assessments of bovine viral diarrhea virus (BVDV) prevention and mitigation activities worldwide. Prev. Vet. Med. 2017, 137, 77–92. [Google Scholar] [CrossRef]
- Richter, V.; Lebl, K.; Baumgartner, W.; Obritzhauser, W.; Käsbohrer, A.; Pinior, B. A systematic worldwide review of the direct monetary losses in cattle due to bovine viral diarrhoea virus infection. Vet. J. 2017, 220, 80–87. [Google Scholar] [CrossRef]
- Marschik, T.; Obritzhauser, W.; Wagner, P.; Richter, V.; Mayerhofer, M.; Egger-Danner, C.; Käsbohrer, A.; Pinior, B. A cost-benefit analysis and the potential trade effects of the bovine viral diarrhoea eradication programme in Styria, Austria. Vet. J. 2018, 231, 19–29. [Google Scholar] [CrossRef]
- Richter, V.; Kattwinkel, E.; Firth, C.L.; Marschik, T.; Dangelmaier, M.; Trauffler, M.; Obritzhauser, W.; Baumgartner, W.; Käsbohrer, A.; Pinior, B. Mapping the global prevalence of bovine viral diarrhoea virus infection and its associated mitigation programmes. Vet. Rec. 2019, 184, 711. [Google Scholar] [CrossRef]
- Burgstaller, J.; Obritzhauser, W.; Kuchling, S.; Kopacka, I.; Pinior, B.; Köfer, J. The effect of bovine viral diarrhoea virus on fertility in dairy cows: Two case-control studies in the province of Styria, Austria. Berl. Munch. Tierarztl. Wochenschr. 2015, 129, 103–110. [Google Scholar] [CrossRef]
- Windeyer, M.C.; Leslie, K.E.; Godden, S.M.; Hodgins, D.C.; Lissemore, K.D.; LeBlanc, S.J. Factors associated with morbidity, mortality, and growth of dairy heifer calves up to 3 months of age. Prev. Vet. Med. 2014, 113, 231–240. [Google Scholar] [CrossRef]
- Svensson, C.; Linder, A.; Olsson, S. Mortality in Swedish Dairy Calves and Replacement Heifers. J. Dairy Sci. 2006, 89, 4769–4777. [Google Scholar] [CrossRef]
- Foster, D.M.; Smith, G.W. Pathophysiology of diarrhea in calves. Vet. Clin. North Am. Food Anim. Pract. 2009, 25, 13–36. [Google Scholar] [CrossRef]
- Scharnböck, B.; Roch, F.-F.; Richter, V.; Funke, C.; Firth, C.L.; Obritzhauser, W.; Baumgartner, W.; Käsbohrer, A.; Pinior, B. A meta-analysis of bovine viral diarrhoea virus (BVDV) prevalences in the global cattle population. Sci. Rep. 2018, 8, 14420. [Google Scholar] [CrossRef]
- Gillhuber, J.; Rügamer, D.; Pfister, K.; Scheuerle, M.C. Giardiosis and other enteropathogenic infections: A study on diarrhoeic calves in Southern Germany. BMC Res. Notes 2014, 7, 1–9. [Google Scholar] [CrossRef]
- Acres, S.D. Enterotoxigenic Escherichia coli infections in newborn calves: A review. J. Dairy Sci. 1985, 68, 229–256. [Google Scholar] [CrossRef]
- Tzipori, S. The relative importance of enteric pathogens affecting neonates of domestic animals. Adv. Vet. Sci. Comp. Med. 1985, 29, 103–206. [Google Scholar]
- Snodgrass, D.R.R.; Terzolo, H.R.; Sherwood, D.; Campbell, I.; Menzies, J.D.; Synge, B.A. Aetiology of diarrhoea in young calves. Vet. Rec. 1986, 119, 31–34. [Google Scholar] [CrossRef]
- Krogh, H.V.; Henriksen, S.A. Bovine cryptosporidiosis in Denmark. 2. Cryptosporidia associated with neonatal calf diarrhea. Nord. Vet. Med. 1985, 37, 42–47. [Google Scholar]
- Lanz Uhde, F.; Kaufmann, T.; Sager, H.; Albini, S.; Zanoni, R.; Schelling, E.; Meylan, M. Prevalence of four enteropathogens in the faeces of young diarrhoeic dairy calves in Switzerland. Vet. Rec. 2008, 163, 362–366. [Google Scholar] [CrossRef]
- Al Mawly, J.; Grinberg, A.; Prattley, D.; Moffat, J.; French, N. Prevalence of endemic enteropathogens of calves in New Zealand dairy farms. N. Z. Vet. J. 2015, 63, 147–152. [Google Scholar] [CrossRef]
- Krogh, H.V. Occurrence of enterotoxigenic Escherichia coli in calves with acute neonatal diarrhoea. Nord. Vet. Med. 1983, 35, 346–352. [Google Scholar]
- Torres-Medina, A.; Schlafer, D.H.; Mebus, C.A. Rotaviral and coronaviral diarrhea. Vet. Clin. North Am. Food Anim. Pract. 1985, 1, 471–493. [Google Scholar] [CrossRef]
- Santín, M.; Trout, J.M.; Fayer, R. A longitudinal study of cryptosporidiosis in dairy cattle from birth to 2 years of age. Vet. Parasitol. 2008, 155, 15–23. [Google Scholar] [CrossRef]
- Sherwood, D.; Snodgrass, D.R.; Lawson, G.H. Prevalence of enterotoxigenic Escherichia coli in calves in Scotland and northern England. Vet. Rec. 1983, 113, 208–212. [Google Scholar] [CrossRef]
- Reynolds, D.J.; Morgan, J.H.; Chanter, N.; Jones, P.W.; Bridger, J.C.; Debney, T.G.; Bunch, K.J. Microbiology of calf diarrhoea in southern Britain. Vet. Rec. 1986, 119, 34–39. [Google Scholar] [CrossRef]
- Mohammed, H.O.; Wade, S.E.; Schaaf, S. Risk factors associated with Cryptosporidium parvum infection in dairy cattle in southeastern New York State. Vet. Parasitol. 1999, 83, 1–13. [Google Scholar] [CrossRef]
- Klein-Jöbstl, D.; Arnholdt, T.; Sturmlechner, F.; Iwersen, M.; Drillich, M. Results of an online questionnaire to survey calf management practices on dairy cattle breeding farms in Austria and to estimate differences in disease incidences depending on farm structure and management practices. Acta Vet. Scand. 2015, 57, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Silverlås, C.; Emanuelson, U.; de Verdier, K.; Björkman, C. Prevalence and associated management factors of Cryptosporidium shedding in 50 Swedish dairy herds. Prev. Vet. Med. 2009, 90, 242–253. [Google Scholar] [CrossRef] [PubMed]
- Gulliksen, S.M.; Jor, E.; Lie, K.I.; Hamnes, I.S.; Løken, T.; Akerstedt, J.; Osterås, O.; Åkerstedt, J.; Østerås, O. Enteropathogens and risk factors for diarrhea in Norwegian dairy calves. J. Dairy Sci. 2009, 92, 5057–5066. [Google Scholar] [CrossRef] [PubMed]
- Kohara, J.; Hirai, T.; Mori, K.; Ishizaki, H.; Tsunemitsu, H. Enhancement of passive immunity with maternal vaccine against newborn calf diarrhea. J. Vet. Med. Sci. 1997, 59, 1023–1025. [Google Scholar] [CrossRef] [PubMed]
- OIE. Cryptosporidiosis. In Terrestrial Manual, 8th ed.; OIE: Paris, France, 2018; pp. 1678–1692. [Google Scholar]
- Paule, R.C.; Mandel, J. Consensus values, regressions, and weighting factors. J. Res. Natl. Inst. Stand. Technol. 1989, 94, 197–203. [Google Scholar] [CrossRef]
- Veroniki, A.A.; Jackson, D.; Viechtbauer, W.; Bender, R.; Bowden, J.; Knapp, G.; Kuss, O.; Higgins, J.P.T.; Langan, D.; Salanti, G. Methods to estimate the between-study variance and its uncertainty in meta-analysis. Res. Synth. Methods 2016, 7, 55–79. [Google Scholar] [CrossRef]
- Freeman, M.F.; Tukey, J.W. Transformations related to the angular and the square root. Ann. Math. Stat. 1950, 607–611. [Google Scholar] [CrossRef]
- Miller, J.J. The inverse of the Freeman—Tukey double arcsine transformation. Am. Stat. 2012, 32, 3–4. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef]
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef]
- Pinior, B.; Garcia, S.; Minviel, J.J.; Raboisson, D. Epidemiological factors and mitigation measures influencing production losses in cattle due to bovine viral diarrhoea virus infection: A meta-analysis. Transbound. Emerg. Dis. 2019, 66, 2426–2439. [Google Scholar] [CrossRef]
- Belsley, D.A.; Kuh, E.; Welsch, R.E. Detecting influential observations and outliers. In Regression Diagnostics; Belsley, D.A., Kuh, E., Welsch, R.E., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 1980; Volume 6, p. 50. [Google Scholar] [CrossRef]
- Cook, R.D. Detection of influential observation in linear regression. Technometrics 1977, 19, 15–18. [Google Scholar] [CrossRef]
- Viechtbauer, W. Conducting meta-analyses in R with the metafor package. J. Stat. Softw. 2010, 36, 1–48. [Google Scholar] [CrossRef]
- Pearson, R. GoodmanKruskal: Association Analysis for Categorical Variables. R Packag. Version 0.0.3. 2020. Available online: https://cran.r-project.org/web/packages/GoodmanKruskal/vignettes/GoodmanKruskal.html (accessed on 3 April 2021).
- Borenstein, M.; Hedges, L.V.; Higgins, J.P.T.; Rothstein, H.R. Identifying and quantifying heterogeneity. In Introduction to Meta-Analysis; Borenstein, M., Hedges, L.V., Higgins, J.P.T., Rothstein, H.R., Eds.; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2009; pp. 107–125. [Google Scholar] [CrossRef]
- Kolenda, R.; Burdukiewicz, M.; Schierack, P. A systematic review and meta-analysis of the epidemiology of pathogenic Escherichia coli of calves and the role of calves as reservoirs for human pathogenic E. coli. Front. Cell. Infect. Microbiol. 2015, 5, 23. [Google Scholar] [CrossRef]
- Hatam-Nahavandi, K.; Ahmadpour, E.; Carmena, D.; Spotin, A.; Bangoura, B.; Xiao, L. Cryptosporidium infections in terrestrial ungulates with focus on livestock: A systematic review and meta-analysis. Parasites Vectors 2019, 12, 1–23. [Google Scholar] [CrossRef]
- Papp, H.; László, B.; Jakab, F.; Ganesh, B.; De Grazia, S.; Matthijnssens, J.; Ciarlet, M.; Martella, V.; Bányai, K. Review of group A rotavirus strains reported in swine and cattle. Vet. Microbiol. 2013, 165, 190–199. [Google Scholar] [CrossRef]
- Kiehl, W. RKI-Fachwörterbuch Infektionsschutz Und Infektionsepidemiologie; Robert-Koch-Institut: Berlin, Germany, 2015. [Google Scholar]
- Sahlström, L.; Virtanen, T.; Kyyrö, J.; Lyytikäinen, T. Biosecurity on Finnish cattle, pig and sheep farms—results from a questionnaire. Prev. Vet. Med. 2014, 117, 59–67. [Google Scholar] [CrossRef]
- Bokusheva, R.; Kimura, S. Cross-country comparison of farm size distribution. OECD Food Agric. Fish. Pap. 2016, 94. [Google Scholar] [CrossRef]
- Frank, N.A.; Kaneene, J.B. Management risk factors associated with calf diarrhea in michigan dairy herds. J. Dairy Sci. 1993, 76, 1313–1323. [Google Scholar] [CrossRef]
- Logan, E.F.; Pearson, G.R.; McNulty, M.S. Studies on the immunity of the calf to colibacillosis--VII: The experimental reproduction of enteric colibacillosis in colostrum-fed calves. Vet. Rec. 1977, 101, 443–446. [Google Scholar] [CrossRef]
- Barry, J.; Bokkers, E.A.M.; Berry, D.P.; de Boer, I.J.M.; McClure, J.; Kennedy, E. Associations between colostrum management, passive immunity, calf-related hygiene practices, and rates of mortality in preweaning dairy calves. J. Dairy Sci. 2019, 102, 10266–10276. [Google Scholar] [CrossRef]
- Kehoe, S.I.; Jayarao, B.M.; Heinrichs, A.J. A survey of bovine colostrum composition and colostrum management practices on Pennsylvania dairy farms. J. Dairy Sci. 2007, 90, 4108–4116. [Google Scholar] [CrossRef]
- Cho, Y.-I.I.; Han, J.-I.I.; Wang, C.; Cooper, V.; Schwartz, K.; Engelken, T.; Yoon, K.-J.J. Case-control study of microbiological etiology associated with calf diarrhea. Vet. Microbiol. 2013, 166, 375–385. [Google Scholar] [CrossRef]
- Meganck, V.; Hoflack, G.; Piepers, S.; Opsomer, G. Evaluation of a protocol to reduce the incidence of neonatal calf diarrhoea on dairy herds. Prev. Vet. Med. 2015, 118, 64–70. [Google Scholar] [CrossRef]
- Trotz-Williams, L.A.; Wayne Martin, S.; Leslie, K.E.; Duffield, T.; Nydam, D.V.; Peregrine, A.S. Calf-level risk factors for neonatal diarrhea and shedding of Cryptosporidium parvum in Ontario dairy calves. Prev. Vet. Med. 2007, 82, 12–28. [Google Scholar] [CrossRef] [PubMed]
- Gulliksen, S.M.; Lie, K.I.; Sølverød, L.; Østerås, O. Risk factors associated with colostrum quality in Norwegian dairy cows. J. Dairy Sci. 2008, 91, 704–712. [Google Scholar] [CrossRef] [PubMed]
- Maddox-Hyttel, C.; Langkjaer, R.B.; Enemark, H.L.; Vigre, H. Cryptosporidium and Giardia in different age groups of Danish cattle and pigs--occurrence and management associated risk factors. Vet. Parasitol. 2006, 141, 48–59. [Google Scholar] [CrossRef] [PubMed]
- Hamnes, I.S.; Gjerde, B.; Robertson, L. Prevalence of Giardia and Cryptosporidium in dairy calves in three areas of Norway. Vet. Parasitol. 2006, 140, 204–216. [Google Scholar] [CrossRef]
- Council Directive 2008/119/EC of 18 December 2008 Laying Down Minimum Standards for the Protection of Calves [2008] OJ L 10. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32008L0119&rid=2 (accessed on 3 April 2021).
- Malmuthuge, N.; Griebel, P.J.; Guan, L.L. The Gut Microbiome and Its Potential Role in the Development and Function of Newborn Calf Gastrointestinal Tract. Front. Vet. Sci. 2015, 2, 36. [Google Scholar] [CrossRef]
- Malmuthuge, N.; Guan, L.L. Understanding the gut microbiome of dairy calves: Opportunities to improve early-life gut health. J. Dairy Sci. 2017, 100, 5996–6005. [Google Scholar] [CrossRef]
- Alipour, M.J.; Jalanka, J.; Pessa-Morikawa, T.; Kokkonen, T.; Satokari, R.; Hynönen, U.; Iivanainen, A.; Niku, M. The composition of the perinatal intestinal microbiota in cattle. Sci. Rep. 2018, 8, 10437. [Google Scholar] [CrossRef]
- Osorio, J.S. Gut health, stress, and immunity in neonatal dairy calves: The host side of host-pathogen interactions. J. Anim. Sci. Biotechnol. 2020, 11, 105. [Google Scholar] [CrossRef]
- Hang, B.P.T.; Wredle, E.; Dicksved, J. Analysis of the developing gut microbiota in young dairy calves-impact of colostrum microbiota and gut disturbances. Trop. Anim. Health Prod. 2020, 53, 50. [Google Scholar] [CrossRef]
- Runnels, P.L.; Moon, H.W.; Schneider, R.A. Development of resistance with host age to adhesion of K99+ Escherichia coli to isolated intestinal epithelial cells. Infect. Immun. 1980, 28, 298–300. [Google Scholar] [CrossRef]
- Izzo, M.M.; Kirkland, P.D.; Mohler, V.L.; Perkins, N.R.; Gunn, A.A.; House, J.K. Prevalence of major enteric pathogens in Australian dairy calves with diarrhoea. Aust. Vet. J. 2011, 89, 167–173. [Google Scholar] [CrossRef]
- Hulbert, L.E.; Moisá, S.J. Stress, immunity, and the management of calves. J. Dairy Sci. 2016, 99, 3199–3216. [Google Scholar] [CrossRef]
- Bulgin, M.S.; Anderson, B.C.; Ward, A.C.S.; Evermann, J.F. Infectious agents associated with neonatal calf disease in southwestern Idaho and eastern Oregon. J. Am. Vet. Med. Assoc. 1982, 180, 1222–1226. [Google Scholar]
- García, A.; Ruiz-Santa-Quiteria, J.A.; Orden, J.A.; Cid, D.; Sanz, R.; Gómez-Bautista, M.; de la Fuente, R. Rotavirus and concurrent infections with other enteropathogens in neonatal diarrheic dairy calves in Spain. Comp. Immunol. Microbiol. Infect. Dis. 2000, 23, 175–183. [Google Scholar] [CrossRef]
- Snodgrass, D.R.R.; Smith, M.L.; Krautil, F.L. Interaction of rotavirus and enterotoxigenic Escherichia coli in conventionally-reared dairy calves. Vet. Microbiol. 1982, 7, 51–60. [Google Scholar] [CrossRef]
- Gouet, P.; Contrepois, M.; Dubourguier, H.C.; Riou, Y.; Scherrer, R.; Laporte, J.; Vautherot, J.F.; Cohen, J.; L’Haridon, R. The experimental production of diarrhoea in colostrum deprived axenic and gnotoxenic calves with enteropathogenic Escherichia coli, rotavirus, coronavirus and in a combined infection of rotavirus and E. coli. Ann. Rech. Vet. 1978, 9, 433–440. [Google Scholar]
- Hess, R.G.; Bachmann, P.A.; Baljer, G.; Mayr, A.; Pospischil, A.; Schmid, G. Synergism in experimental mixed infections of newborn colostrum-deprived calves with bovine rotavirus and enterotoxigenic Escherichia coli (ETEC). Zent. für Veterinärmedizin B 1984, 31, 585–596. [Google Scholar] [CrossRef]
- Runnels, P.L.; Moon, H.W.; Matthews, P.J.; Whipp, S.C.; Woode, G.N. Effects of microbial and host variables on the interaction of rotavirus and Escherichia coli infections in gnotobiotic calves. Am. J. Vet. Res. 1986, 47, 1542–1550. [Google Scholar]
- Tzipori, S.R.; Makin, T.J.; Smith, M.L.; Krautil, F.L.I. Clinical manifestations of diarrhea in calves infected with rotavirus and enterotoxigenic Escherichia coli. J. Clin. Microbiol. 1981, 13, 1011–1016. [Google Scholar] [CrossRef]
- Collins, J.; Starkey, W.G.; Wallis, T.S.; Clarke, G.J.; Worton, K.J.; Spencer, A.J.; Haddon, S.J.; Osborne, M.P.; Candy, D.C.; Stephen, J. Intestinal enzyme profiles in normal and rotavirus-infected mice. J. Pediatr. Gastroenterol. Nutr. 1988, 7, 264–272. [Google Scholar] [CrossRef]
- Jourdan, N.; Brunet, J.P.; Sapin, C.; Blais, A.; Cotte-Laffitte, J.; Forestier, F.; Quero, A.M.; Trugnan, G.; Servin, A.L. Rotavirus infection reduces sucrase-isomaltase expression in human intestinal epithelial cells by perturbing protein targeting and organization of microvillar cytoskeleton. J. Virol. 1998, 72, 7228–7236. [Google Scholar] [CrossRef]
- Morin, M.; Larivière, S.; Lallier, R.; Lariviere, S.; Lallier, R. Pathological and microbiological observations made on spontaneous cases of acute neonatal calf diarrhea. Can. J. Comp. Med. Rev. Can. Med. Comp. 1976, 40, 228–240. [Google Scholar]
- Durham, P.J.K.; Farquharson, B.C.; Stevenson, B.J. Rotavirus and coronavirus associated diarrohoea in calves. N. Z. Vet. J. 1979, 27, 266–272. [Google Scholar] [CrossRef][Green Version]
- Crawford, S.E.; Ramani, S.; Tate, J.E.; Parashar, U.D.; Svensson, L.; Hagbom, M.; Franco, M.A.; Greenberg, H.B.; O’Ryan, M.; Kang, G.; et al. Rotavirus infection. Nat. Rev. Dis. Prim. 2017, 3, 17083. [Google Scholar] [CrossRef]
- Brunet, J.P.; Jourdan, N.; Cotte-Laffitte, J.; Linxe, C.; Géniteau-Legendre, M.; Servin, A.; Quéro, A.M. Rotavirus infection induces cytoskeleton disorganization in human intestinal epithelial cells: Implication of an increase in intracellular calcium concentration. J. Virol. 2000, 74, 10801–10806. [Google Scholar] [CrossRef]
- Brunet, J.P.; Cotte-Laffitte, J.; Linxe, C.; Quero, A.M.; Géniteau-Legendre, M.; Servin, A. Rotavirus infection induces an increase in intracellular calcium concentration in human intestinal epithelial cells: Role in microvillar actin alteration. J. Virol. 2000, 74, 2323–2332. [Google Scholar] [CrossRef]
- Chen, X.M.; LaRusso, N.F. Mechanisms of attachment and internalization of Cryptosporidium parvum to biliary and intestinal epithelial cells. Gastroenterology 2000, 118, 368–379. [Google Scholar] [CrossRef]
- Tzipori, S.; Sherwood, D.; Angus, K.W.; Campbell, I.; Gordon, M. Diarrhea in lambs: Experimental infections with enterotoxigenic Escherichia coli, rotavirus, and Cryptosporidium sp. Infect. Immun. 1981, 33, 401–406. [Google Scholar] [CrossRef]
- Gurevitch, J.; Koricheva, J.; Nakagawa, S.; Stewart, G. Meta-analysis and the science of research synthesis. Nature 2018, 555, 175–182. [Google Scholar] [CrossRef]
- Berge, A.C.B.B.; Besser, T.E.; Moore, D.A.; Sischo, W.M. Evaluation of the effects of oral colostrum supplementation during the first fourteen days on the health and performance of preweaned calves. J. Dairy Sci. 2009, 92, 286–295. [Google Scholar] [CrossRef]
- Gutzwiller, A. Effect of colostrum intake on diarrhoea incidence in new-born calves. Schweiz. Arch. Tierheilkd. 2002, 144, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Trotz-Williams, L.A.; Martin, S.W.; Leslie, K.E.; Duffield, T.; Nydam, D.V.; Peregrine, A.S. Association between management practices and within-herd prevalence of Cryptosporidium parvum shedding on dairy farms in southern Ontario. Prev. Vet. Med. 2008, 83, 11–23. [Google Scholar] [CrossRef] [PubMed]
- Anderson, E.J.; Weber, S.G. Rotavirus infection in adults. Lancet. Infect. Dis. 2004, 4, 91–99. [Google Scholar] [CrossRef]
- Checkley, W.; White, A.C.J.; Jaganath, D.; Arrowood, M.J.; Chalmers, R.M.; Chen, X.-M.; Fayer, R.; Griffiths, J.K.; Guerrant, R.L.; Hedstrom, L.; et al. A review of the global burden, novel diagnostics, therapeutics, and vaccine targets for cryptosporidium. Lancet. Infect. Dis. 2015, 15, 85–94. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).