Activity Time Budgets—A Potential Tool to Monitor Equine Welfare?
Abstract
Simple Summary
Abstract
1. Introduction
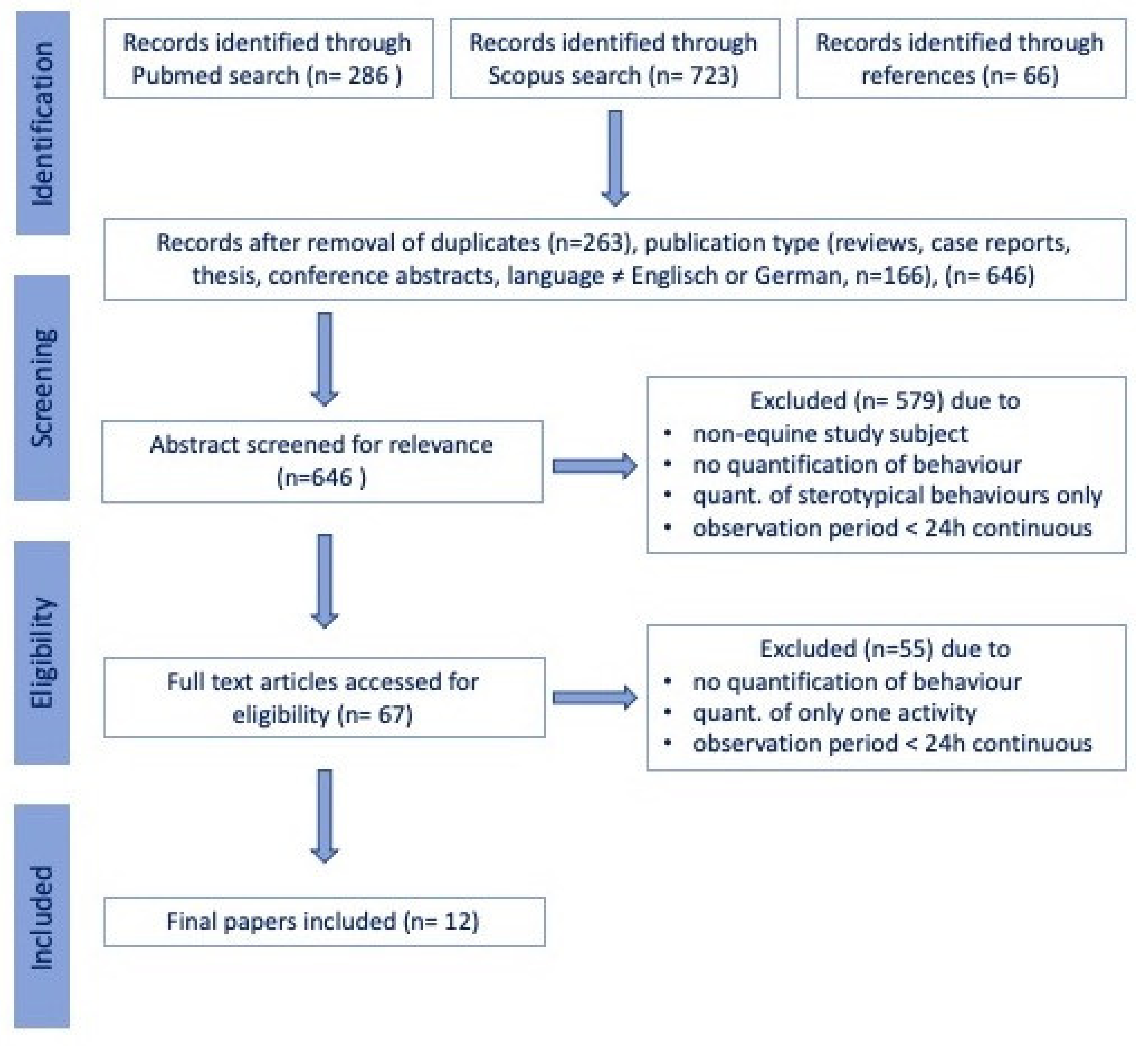
2. Materials and Methods
2.1. Data Sources and Searches
2.2. Data Extraction and Risk of Bias Assessment
3. Results
4. Discussion
4.1. Time Budget Measurement: Methods and Use
4.2. Time Budget for Feeding and Foraging
4.3. Time Budget for Resting and Sleeping
4.4. Time Budget for Locomotion and Movement
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statements
Acknowledgments
Conflicts of Interest
References
- Fraser, D. Understanding animal welfare. Acta Veter Scand. 2008, 50, S1–S7. [Google Scholar] [CrossRef]
- Ohl, F.; Van Der Staay, F. Animal welfare: At the interface between science and society. Veter J. 2012, 192, 13–19. [Google Scholar] [CrossRef]
- Green, T.C.; Mellor, D.J. Extending ideas about animal welfare assessment to include ‘quality of life’ and related concepts. N. Z. Veter J. 2011, 59, 263–271. [Google Scholar] [CrossRef]
- Lesimple, C. Indicators of Horse Welfare: State-of-the-Art. Animals 2020, 10, 294. [Google Scholar] [CrossRef] [PubMed]
- Lesimple, C.; Reverchon-Billot, L.; Galloux, P.; Stomp, M.; Boichot, L.; Coste, C.; Henry, S.; Hausberger, M. Free movement: A key for welfare improvement in sport horses? Appl. Anim. Behav. Sci. 2020, 225, 104972. [Google Scholar] [CrossRef]
- Hausberger, M.; Lerch, N.; Guilbaud, E.; Stomp, M.; Grandgeorge, M.; Henry, S.; Lesimple, C. On-Farm Welfare Assessment of Horses: The Risks of Putting the Cart before the Horse. Animals 2020, 10, 371. [Google Scholar] [CrossRef] [PubMed]
- Broom, D.M. Animal Welfare: The Concept of the Issues. In Attitudes to Animals; Dollins, F., Ed.; Cambridge University Press: Cambridge, UK, 1999; pp. 129–142. ISBN 9780511608476. [Google Scholar]
- McGreevy, P.; Berger, J.; De Brauwere, N.; Doherty, O.; Harrison, A.; Fiedler, J.; Jones, C.; McDonnell, S.; McLean, A.; Nakonechny, L.; et al. Using the Five Domains Model to Assess the Adverse Impacts of Husbandry, Veterinary, and Equitation Interventions on Horse Welfare. Animals 2018, 8, 41. [Google Scholar] [CrossRef] [PubMed]
- Wolfensohn, S. Too Cute to Kill? The Need for Objective Measurements of Quality of Life. Animals 2020, 10, 1054. [Google Scholar] [CrossRef]
- Lesimple, C.; Gautier, E.; Benhajali, H.; Rochais, C.; Lunel, C.; Bensaïd, S.; Khalloufi, A.; Henry, S.; Hausberger, M. Stall architecture influences horses’ behaviour and the prevalence and type of stereotypies. Appl. Anim. Behav. Sci. 2019, 219, 104833. [Google Scholar] [CrossRef]
- Waran, N.; Randle, H. What we can measure, we can manage: The importance of using robust welfare indicators in Equitation Science. Appl. Anim. Behav. Sci. 2017, 190, 74–81. [Google Scholar] [CrossRef]
- Asher, L.; Collins, L.M.; Ortiz-Pelaez, A.; Drewe, J.A.; Nicol, C.J.; Pfeiffer, D.U. Recent advances in the analysis of behavioural organization and interpretation as indicators of animal welfare. J. R. Soc. Interface 2009, 6, 1103–1119. [Google Scholar] [CrossRef]
- Hockenhull, J.; Whay, H.R. A review of approaches to assessing equine welfare. Equine Veter Educ. 2014, 26, 159–166. [Google Scholar] [CrossRef]
- Goodwin, D. The importance of ethology in understanding the behaviour of the horse. Equine Veter J. 2010, 31, 15–19. [Google Scholar] [CrossRef]
- Yarnell, K.; Hall, C.; Royle, C.; Walker, S.L. Domesticated horses differ in their behavioural and physiological responses to isolated and group housing. Physiol. Behav. 2015, 143, 51–57. [Google Scholar] [CrossRef]
- Green, N.F.; Green, H.D. The Wild Horse Population of Stone Cabin Valley, Nevada: A Preliminary Report. In Proceedings of the National Wild Horse Forum, Reno, NV, USA, 5–7 April 1977. [Google Scholar]
- Murphy, B.A. Circadian and Circannual Regulation in the Horse: Internal Timing in an Elite Athlete. J. Equine Veter Sci. 2019, 76, 14–24. [Google Scholar] [CrossRef]
- Boy, V.; Duncan, P. Time-Budgets of Camargue Horses I. Developmental Changes in the Time-Budgets of Foals. Behavior 1979, 71, 187–201. [Google Scholar] [CrossRef]
- Berger, A.; Scheibe, K.-M.; Michaelis, S.; Streich, W.J. Evaluation of living conditions of free-ranging animals by automated chronobiological analysis of behavior. Behav. Res. Methods Instrum. Comput. 2003, 35, 458–466. [Google Scholar] [CrossRef]
- Veasey, J.; Waran, N.; Young, R. On comparing the behaviour of zoo housed animals with wild conspecifics as a welfare indicator. Anim. Welf. 1996, 5, 13–24. [Google Scholar]
- Waran, N.K. Can studies of feral horse behaviour be used for assessing domestic horse welfare? Equine Veter J. 1997, 29, 249–251. [Google Scholar] [CrossRef]
- Yeates, J. Naturalness and Animal Welfare. Animals 2018, 8, 53. [Google Scholar] [CrossRef]
- Flannigan, G.; Stookey, J. Day-time time budgets of pregnant mares housed in tie stalls: A comparison of draft versus light mares. Appl. Anim. Behav. Sci. 2002, 78, 125–143. [Google Scholar] [CrossRef]
- Sarrafchi, A.; Blokhuis, H.J. Equine stereotypic behaviors: Causation, occurrence, and prevention. J. Veter Behav. 2013, 8, 386–394. [Google Scholar] [CrossRef]
- Heleski, C.; Shelle, A.; Nielsen, B.; Zanella, A. Influence of housing on weanling horse behavior and subsequent welfare. Appl. Anim. Behav. Sci. 2002, 78, 291–302. [Google Scholar] [CrossRef]
- Benhajali, H.; Richard-Yris, M.-A.; Leroux, M.; Ezzaouïa, M.; Charfi, F.; Hausberger, M. A note on the time budget and social behaviour of densely housed horses. Appl. Anim. Behav. Sci. 2008, 112, 196–200. [Google Scholar] [CrossRef]
- Benhajali, H.; Richard-Yris, M.-A.; Ezzaouia, M.; Charfi, F.; Hausberger, M. Foraging opportunity: A crucial criterion for horse welfare? Animals 2009, 3, 1308–1312. [Google Scholar] [CrossRef]
- Berger, A.; Scheibe, K.-M.; Eichhorn, K.; Scheibe, A.; Streich, J. Diurnal and ultradian rhythms of behaviour in a mare group of Przewalski horse (Equus ferus przewalskii), measured through one year under semi-reserve conditions. Appl. Anim. Behav. Sci. 1999, 64, 1–17. [Google Scholar] [CrossRef]
- Martin, A.-M.; Elliott, J.A.; Duffy, P.; Blake, C.M.; Ben Attia, S.; Katz, L.M.; Browne, J.A.; Gath, V.; McGivney, B.A.; Hill, E.W.; et al. Circadian regulation of locomotor activity and skeletal muscle gene expression in the horse. J. Appl. Physiol. 2010, 109, 1328–1336. [Google Scholar] [CrossRef]
- Ladds, M.A.; Salton, M.; Hocking, D.P.; McIntosh, R.R.; Thompson, A.P.; Slip, D.J.; Harcourt, R.G. Using accelerometers to develop time-energy budgets of wild fur seals from captive surrogates. PeerJ 2018, 6, e5814. [Google Scholar] [CrossRef]
- Lind, N.M.; Vinther, M.; Hemmingsen, R.P.; Hansen, A.K. Validation of a digital video tracking system for recording pig locomotor behaviour. J. Neurosci. Methods 2005, 143, 123–132. [Google Scholar] [CrossRef]
- Eerdekens, A.; Deruyck, M.; Fontaine, J.; Martens, L.; De Poorter, E.; Joseph, W. Automatic equine activity detection by convolutional neural networks using accelerometer data. Comput. Electron. Agric. 2020, 168, 105139. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Prisma Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Methley, A.M.; Campbell, S.; Chew-Graham, C.; McNally, R.; Cheraghi-Sohi, S. PICO, PICOS and SPIDER: A comparison study of specificity and sensitivity in three search tools for qualitative systematic reviews. BMC Health Serv. Res. 2014, 14, 579. [Google Scholar] [CrossRef]
- Kennedy, C.E.; Fonner, V.A.; Armstrong, K.A.; Denison, J.A.; Yeh, P.T.; O’Reilly, K.R.; Sweat, M.D. The Evidence Project risk of bias tool: Assessing study rigor for both randomized and non-randomized intervention studies. Syst. Rev. 2019, 8, 3. [Google Scholar] [CrossRef]
- Villafaina-Domínguez, B.; Collado-Mateo, D.; Merellano-Navarro, E.; Villafaina, S. Effects of Dog-Based Animal-Assisted Interventions in Prison Population: A Systematic Review. Animals 2020, 10, 2129. [Google Scholar] [CrossRef] [PubMed]
- Duncan, P. Time-Budgets of Camargue Horses Ii. Time-Budgets of Adult Horses and Weaned Sub-Adults. Behavior 1980, 72, 26–48. [Google Scholar] [CrossRef]
- Duncan, P. Time-Budgets of Camargue Horses Iii. Environmental Influences. Behavior 1985, 92, 188–208. [Google Scholar] [CrossRef]
- Boyd, L.E.; Carbonaro, D.A.; Houpt, K.A. The 24-hour time budget of Przewalski horses. Appl. Anim. Behav. Sci. 1988, 21, 5–17. [Google Scholar] [CrossRef]
- Price, J.; Catriona, S.; Welsh, E.M.; Waran, N.K. Preliminary evaluation of a behaviour–based system for assessment of post–operative pain in horses following arthroscopic surgery. Veter Anaesth. Analg. 2003, 30, 124–137. [Google Scholar] [CrossRef]
- Elia, J.B.; Erb, H.N.; Houpt, K.A. Motivation for hay: Effects of a pelleted diet on behavior and physiology of horses. Physiol. Behav. 2010, 101, 623–627. [Google Scholar] [CrossRef]
- Aristizabal, F.; Nieto, J.; Yamout, S.; Snyder, J. The effect of a hay grid feeder on feed consumption and measurement of the gastric pH using an intragastric electrode device in horses: A preliminary report. Equine Veter J. 2013, 46, 484–487. [Google Scholar] [CrossRef] [PubMed]
- Sartori, C.; Guzzo, N.; Normando, S.; Bailoni, L.; Mantovani, R. Evaluation of behaviour in stabled draught horse foals fed diets with two protein levels. Animals 2017, 11, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Maisonpierre, I.N.; Sutton, M.A.; Harris, P.; Menzies-Gow, N.; Weller, R.; Pfau, T. Accelerometer activity tracking in horses and the effect of pasture management on time budget. Equine Veter J. 2019, 51, 840–845. [Google Scholar] [CrossRef] [PubMed]
- Correa, M.G.; e Silva, C.F.R.; Dias, L.A.; Junior, S.D.S.R.; Thomes, F.R.; Lago, L.A.D.; Carvalho, A.D.M.; Faleiros, R.R. Welfare benefits after the implementation of slow-feeder hay bags for stabled horses. J. Veter Behav. 2020, 38, 61–66. [Google Scholar] [CrossRef]
- Raspa, F.; Tarantola, M.; Bergero, D.; Nery, J.; Visconti, A.; Mastrazzo, C.M.; Cavallini, D.; Valvassori, E.; Valle, E. Time-Budget of Horses Reared for Meat Production: Influence of Stocking Density on Behavioural Activities and Subsequent Welfare. Animals 2020, 10, 1334. [Google Scholar] [CrossRef]
- Rose-Meierhöfer, S.; Klaer, S.; Ammon, C.; Brunsch, R.; Hoffmann, G. Activity Behavior of Horses Housed in Different Open Barn Systems. J. Equine Veter Sci. 2010, 30, 624–634. [Google Scholar] [CrossRef]
- Bonneau, M.; Vayssade, J.-A.; Troupe, W.; Arquet, R. Outdoor animal tracking combining neural network and time-lapse cameras. Comput. Electron. Agric. 2020, 168, 105150. [Google Scholar] [CrossRef]
- Nathan, R.; Spiegel, O.; Fortmann-Roe, S.; Harel, R.; Wikelski, M.; Getz, W.M. Using tri-axial acceleration data to identify behavioral modes of free-ranging animals: General concepts and tools illustrated for griffon vultures. J. Exp. Biol. 2012, 215, 986–996. [Google Scholar] [CrossRef]
- Egan, S.; Kearney, C.M.; Brama, P.A.; Parnell, A.C.; McGrath, D. Exploring stable-based behaviour and behaviour switching for the detection of bilateral pain in equines. Appl. Anim. Behav. Sci. 2021, 235, 105214. [Google Scholar] [CrossRef]
- Pritchett, L.C.; Ulibarri, C.; Roberts, M.C.; Schneider, R.K.; Sellon, D.C. Identification of potential physiological and behavioral indicators of postoperative pain in horses after exploratory celiotomy for colic. Appl. Anim. Behav. Sci. 2003, 80, 31–43. [Google Scholar] [CrossRef]
- Dubois, C.; Zakrajsek, E.; Haley, D.B.; Merkies, K. Validation of triaxial accelerometers to measure the lying behaviour of adult domestic horses. Animals 2015, 9, 110–114. [Google Scholar] [CrossRef] [PubMed]
- Clothier, J.; Small, A.; Hinch, G.; Barwick, J.; Brown, W.Y. Using Movement Sensors to Assess Lying Time in Horses With and Without Angular Limb Deformities. J. Equine Veter Sci. 2019, 75, 55–59. [Google Scholar] [CrossRef]
- Hausberger, M.; Fureix, C.; Lesimple, C. Detecting horses’ sickness: In search of visible signs. Appl. Anim. Behav. Sci. 2016, 175, 41–49. [Google Scholar] [CrossRef]
- Ashley, F.H.; Waterman-Pearson, A.E.; Whay, H.R. Behavioural assessment of pain in horses and donkeys: Application to clinical practice and future studies. Equine Veter J. 2010, 37, 565–575. [Google Scholar] [CrossRef]
- Houpt, K.A. Ingestive Behavior. Veter Clin. N. Am. Equine Pr. 1990, 6, 319–337. [Google Scholar] [CrossRef]
- Harris, P. Nutrition, behaviour and the role of supplements for calming horses: The veterinarian’s dilemma. Veter J. 2005, 170, 10–11. [Google Scholar] [CrossRef]
- Mayes, E.; Duncan, P. Temporal Patterns of Feeding Behaviour in Free-Ranging Horses. Behavior 1986, 96, 105–129. [Google Scholar] [CrossRef]
- Thorne, J.; Goodwin, D.; Kennedy, M.; Davidson, H.; Harris, P. Foraging enrichment for individually housed horses: Practicality and effects on behaviour. Appl. Anim. Behav. Sci. 2005, 94, 149–164. [Google Scholar] [CrossRef]
- Ellis, A.D.; Fell, M.; Luck, K.; Gill, L.; Owen, H.; Briars, H.; Barfoot, C.; Harris, P. Effect of forage presentation on feed intake behaviour in stabled horses. Appl. Anim. Behav. Sci. 2015, 165, 88–94. [Google Scholar] [CrossRef]
- Ellis, A.D.; Redgate, S.; Zinchenko, S.; Owen, H.; Barfoot, C.; Harris, P. The effect of presenting forage in multi-layered haynets and at multiple sites on night time budgets of stabled horses. Appl. Anim. Behav. Sci. 2015, 171, 108–116. [Google Scholar] [CrossRef]
- Rochais, C.; Henry, S.; Hausberger, M. “Hay-bags” and “Slow feeders”: Testing their impact on horse behaviour and welfare. Appl. Anim. Behav. Sci. 2018, 198, 52–59. [Google Scholar] [CrossRef]
- Christie, J.L.; Hewson, C.J.; Riley, C.B.; McNiven, M.A.; Dohoo, I.R.; Bate, L.A. Management factors affecting stereotypies and body condition score in nonracing horses in Prince Edward Island. Can. Veter J. Rev. Veter Can. 2006, 47, 136–143. [Google Scholar]
- Hothersall, B.; Nicol, C. Role of Diet and Feeding in Normal and Stereotypic Behaviors in Horses. Veter Clin. N. Am. Equine Pr. 2009, 25, 167–181. [Google Scholar] [CrossRef] [PubMed]
- Hothersall, B.; Casey, R. Undesired behaviour in horses: A review of their development, prevention, management and association with welfare. Equine Veter Educ. 2011, 24, 479–485. [Google Scholar] [CrossRef]
- Hanis, F.; Chung, E.L.T.; Kamalludin, M.H.; Idrus, Z. The Influence of Stable Management and Feeding Practices on the Abnormal Behaviors among Stabled Horses in Malaysia. J. Equine Veter Sci. 2020, 94, 103230. [Google Scholar] [CrossRef]
- Waters, A.J.; Nicol, C.J.; French, N.P. Factors influencing the development of stereotypic and redirected behaviours in young horses: Findings of a four year prospective epidemiological study. Equine Veter J. 2010, 34, 572–579. [Google Scholar] [CrossRef] [PubMed]
- Ninomiya, S.; Kusunose, R.; Sato, S.; Terada, M.; Sugawara, K. Effects of feeding methods on eating frustration in stabled horses. Anim. Sci. J. 2004, 75, 465–469. [Google Scholar] [CrossRef]
- Burla, J.-B.; Ostertag, A.; Patt, A.; Bachmann, I.; Hillmann, E. Effects of feeding management and group composition on agonistic behaviour of group-housed horses. Appl. Anim. Behav. Sci. 2016, 176, 32–42. [Google Scholar] [CrossRef]
- Zupan, M.; Štuhec, I.; Jordan, D. The Effect of an Irregular Feeding Schedule on Equine Behavior. J. Appl. Anim. Welf. Sci. 2019, 23, 156–163. [Google Scholar] [CrossRef]
- Baumgartner, M.; Boisson, T.; Erhard, M.H.; Zeitler-Feicht, M.H. Common Feeding Practices Pose A Risk to the Welfare of Horses When Kept on Non-Edible Bedding. Animals 2020, 10, 411. [Google Scholar] [CrossRef]
- Burla, J.-B.; Rufener, C.; Bachmann, I.; Gygax, L.; Patt, A.; Hillmann, E. Space Allowance of the Littered Area Affects Lying Behavior in Group-Housed Horses. Front. Veter Sci. 2017, 4, 23. [Google Scholar] [CrossRef]
- Arnold, G. Comparison of the time budgets and circadian patterns of maintenance activities in sheep, cattle and horses grouped together. Appl. Anim. Behav. Sci. 1984, 13, 19–30. [Google Scholar] [CrossRef]
- Sweeting, M.P.; Houpt, C.; Houpt, K.A. Social Facilitation of Feeding and Time Budgets in Stabled Ponies. J. Anim. Sci. 1985, 60, 369–374. [Google Scholar] [CrossRef]
- Ogilvie-Graham, T. Time Budget Studies in Stalled Horses. 1994. Available online: https://era.ed.ac.uk/handle/1842/29924 (accessed on 2 January 2021).
- Greening, L.; Downing, J.; Amiouny, D.; Lekang, L.; McBride, S. The effect of altering routine husbandry factors on sleep duration and memory consolidation in the horse. Appl. Anim. Behav. Sci. 2021, 236, 105229. [Google Scholar] [CrossRef]
- Wei, H.; Zhao, W.; Wang, Y.-X.; Pertovaara, A. Pain-related behavior following REM sleep deprivation in the rat: Influence of peripheral nerve injury, spinal glutamatergic receptors and nitric oxide. Brain Res. 2007, 1148, 105–112. [Google Scholar] [CrossRef]
- May, M.E.; Harvey, M.T.; Valdovinos, M.G.; Kline, R.H.; Wiley, R.G.; Kennedy, C.H. Nociceptor and age specific effects of REM sleep deprivation induced hyperalgesia. Behav. Brain Res. 2005, 159, 89–94. [Google Scholar] [CrossRef]
- Fader, C.; Sambraus, H. The Resting Behaviour of Horses in Loose Housing Systems. Tierärztliche Umsch. 2004, 59, 320–327. [Google Scholar]
- Raabymagle, P.; Ladewig, J. Lying behavior in horses in relation to box size. J. Equine Veter Sci. 2006, 26, 11–17. [Google Scholar] [CrossRef]
- Pedersen, G.R.; Søndergaard, E.; Ladewig, J. The influence of bedding on the time horses spend recumbent. J. Equine Veter Sci. 2004, 24, 153–158. [Google Scholar] [CrossRef]
- Burla, J.-B.; Ostertag, A.; Westerath, H.S.; Hillmann, E. Gait determination and activity measurement in horses using an accelerometer. Comput. Electron. Agric. 2014, 102, 127–133. [Google Scholar] [CrossRef]
| Author(s) and Publication Year | Study Design | Control Group | Intervention | Observation Method(s) | Observation: Number of Days |
|---|---|---|---|---|---|
| Boy & Duncan 1979 [18] | Observational study (field) | no | no | manual, scan sampling 5-min blocks | 24 h for two day in two weeks each month |
| Duncan 1980 [37] | Observational study (field) | no | no | manual, scan sampling 5-min blocks | 24 h for two day in two weeks each month |
| Duncan 1985 [38] | Observational study (field) | no | no | manual, scan sampling 5-min blocks | 24 h two day in two weeks each month |
| Boyd et al. 1988 [39] | Observational study (field) | no | no | manual, focal sampling | 24 h |
| Berger et al. 1999 [28] | Observational study (field) | no | no | telemetry system ETHOSYS | one year |
| Price et al. 2003 [40] | Prospective non-blinded, non-randomized controlled trial | yes | elective arthroscopy | CVI, time lapse video recorder, manual, instantaneous sampling | continuously 72 h |
| Elia et al. 2010 [41] | Prospective non-blinded, non-randomized case series | no | feeding test | video time-lapse mode, manual, instantaneous sampling, Observer TM program | 24 h 3 days per week |
| Aristizabal et al. 2014 [42] | Prospective non-blinded, non-randomized case series | no | two feeding devices | video, manual | 24 h |
| Sartori et al. 2017 [43] | Prospective non-blinded, non-randomized case series | no | two isoenergetic diets | video, manual, focal sampling continuous | 2 days |
| Maisonpierre et al. 2019 [44] | Prospective non-blinded, non-randomized case series | no | no | accelerometer (Actigraph) | 24 h, 20 days |
| Correa et al. 2020 [45] | Observational study (stable) | no | additional hay bags | video, manual, ad libitum sampling, pedometer | 24 h |
| Raspa et al. 2020 [46] | prospective non-blinded, non-randomized case series | no | three stocking density’s | video, manual, scan sampling at 15 min intervals | 3 consecutive days (72 h) |
| Author(s) and Publication Year | Horses (n) | Feral/Semiferal/ Domesticated | Breed | Gender | Age |
|---|---|---|---|---|---|
| Boy & Duncan 1979 [18] | 11 | semiferal, free-ranging | Camargue | Foals | n.a. |
| Duncan 1980 [37] | 18 | semiferal, free-ranging | Camargue | 4 males, 9 mares, 5 yearlings | n.a. |
| Duncan 1985 [38] | 18 | semiferal, free-ranging | Camargue | 9 males, 9 mares | n.a. |
| Boyd et al., 1988 [39] | 8 | semiferal, semi-reserve | Przewalski | 1 stallion, 6 mares, 1 foal | n.a. |
| Berger et al. 1999 [28] | 4 | semiferal, semi-reserve | Przewalski | mares | n.a. |
| Price et al. 2003 [40] | 12 | domesticated | mixed breed | 6 geldings, 6 mares | 9 (4–15) years |
| Elia et al. 2010 [41] | 8 | domesticated | mixed breed | 8 mares | (6–14) years |
| Aristizabal et al. 2014 [42] | 10 | domesticated | mixed breed | 4 geldings, 6 mares | 23 (20–27) years |
| Sartori et al. 2017 [43] | 20 | domesticated | Italian draft horse | 10 males, 10 mares | 12–18 months |
| Maisonpierre et al. 2019 [44] | 6 | domesticated | mixed breed | 4 geldings, 2 mares | 14 (4–22) years |
| Correa et al. 2020 [45] | 7 | domesticated | Brazilian jumper | 3 males, 4 mares | 10 ± 5 years |
| Raspa et al. 2020 [46] | 22 | domesticated | Comtois | 19 males, 3 mares | 22 ± 2 months |
| Author(s) and Publication Year | TB in % Standing or Resting | TB in % Lying | TB in % Eating | TB in % Movement/Locomotion | TB in % Other Behaviour e.g. Drinking, Playing | Comments |
|---|---|---|---|---|---|---|
| Boy & Duncan 1979 [18] | 8.1–11.8% | 2.7–15% | 13–62% | Data of foals during developing | ||
| Duncan 1980 [37] | 12.9–19.52% | 4.25–13.76% | 50.82–63.89% | 5.45–9.3% | n.a. | TB ranges based on the TB detailed in Table 9 of the publication, TB variation due to sex and age |
| Duncan 1985 [38] | 13.4–29.3% | 4.2–15.5% | 60.8–66.6% | 4.3–13.4% | n.a. | TB ranges based on the TB detailed in Table 8 of the publication; TB depending on season and gender |
| Boyd et al. 1988 [39] | 36.6% ± 5.4% | 5.3% ± 2.5% | 46.4% ± 5.9% | 7.4% ± 1% | 10.2% ± 0.5% | Variation during daytime and season for feeding and standing |
| Berger et al. 1999 [28] | 36.4% ± 15.7% winter: 48.4% ± 15% summer: 30.7% ± 29% | n.a. | 29.8% ± 13% | n.a. | n.a. | Fluctuation over 24 h for feeding behaviour |
| Price et al. 2003 [40] | 54 ± 9: control group 66 ± 12: post-surgery | 8 ± 6: control group 4 ± 6: post-surgery | 34 ± 6: control group 20 ± 9: post-surgery | 0.015 ± 0.005: control group 3 ± 2: post-surgery | 2% ± 0%: control 2% ± 1%: post-surgery | Values 0–24 h post-surgery, horses are housed in a stable |
| Elia et al. 2010 [41] | in stall: pellet-fed group: 58% in stall: hay-fed group: 36.6% paddock: pellet-fed group: 47.5 % paddock: hay-fed group: 32.4% | n.a. | In stall: pellet-fed group: 10% In stall: hay-fed group: 64% | paddock: pellet-fed group:12.3% paddock: hay-fed group 19.1% | searching: in stall: pellet-fed group: 11.5% in stall: hay-fed group: 1.2% | TB are provided separately for the time spent in the stable resp. the paddock for both groups |
| Aristizabal et al. 2014 [42] | ground feeding: 68% ± 8.6%. feeder: 65% ± 8.2% | ground feeding: 3% ± 5.5% feeder: 5% ± 6.68% | ground feeding: 28% ± 5.5% feeder: 31% ± 8.4% | n.a. | n.a. | Increased hay intake during daytime |
| Sartori et al. 2017 [43] | 15.58% ± 5.02% | 25.72% ± 4% | 32.47% ± 3.75% | 15.32% ± 2.37% | 11.31 ± 3.32% | Details for gender and diet |
| Maisonpierre et al. 2019 [44] | 33% (27.5–31.1) daytime51% (47.1–55.2) night-time 36% (33.3–39.2) standard paddock 42.9% (36.6–47.1) small paddock | n.a. | 60.8% (58.2 65–4) daytime 46.8% (43.3–50.2) night-time 50.8% (47.9–55) standard paddock 48.6% (42.9–56.7) small paddock | 4.6% (3.7–6.9) daytime2.4% (0.8–3.4) night-time 4.1% (3.1–5.8) standard paddock 2.5% (1.9–4.2) small paddock | n.a. | TBs per paddock size were calculated based on the hours spent for each activity provided in the publication |
| Correa et al. 2020 [45] | Basal: 62.7% Hay bag: 65% | Basal: 10.7% Hay bag: 9.9% | Basal: 12.5%Hay bag: 15.9% | Abnormal behaviour: Basal: 9% Hay bag: 5.9% | Leisure combines movement, standing, investigation | |
| Raspa et al. 2020 [46] | 30.56% ± 6.56% | 27.33% ± 2.05% | 30.55% ± 3.59% | 4.07% ± 1.06% | <2% | reduced stocking density increased locomotion and playing, this change in TB was not quantified |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Auer, U.; Kelemen, Z.; Engl, V.; Jenner, F. Activity Time Budgets—A Potential Tool to Monitor Equine Welfare? Animals 2021, 11, 850. https://doi.org/10.3390/ani11030850
Auer U, Kelemen Z, Engl V, Jenner F. Activity Time Budgets—A Potential Tool to Monitor Equine Welfare? Animals. 2021; 11(3):850. https://doi.org/10.3390/ani11030850
Chicago/Turabian StyleAuer, Ulrike, Zsofia Kelemen, Veronika Engl, and Florien Jenner. 2021. "Activity Time Budgets—A Potential Tool to Monitor Equine Welfare?" Animals 11, no. 3: 850. https://doi.org/10.3390/ani11030850
APA StyleAuer, U., Kelemen, Z., Engl, V., & Jenner, F. (2021). Activity Time Budgets—A Potential Tool to Monitor Equine Welfare? Animals, 11(3), 850. https://doi.org/10.3390/ani11030850






