Koi (Cyprinus rubrofuscus) Seek Out Tactile Interaction with Humans: General Patterns and Individual Differences
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Location and Subjects
2.2. Animal Care
2.2.1. Housing
2.2.2. Feeding
2.2.3. Maintenance
2.3. Study Timeline
2.3.1. Habituation Period (July 2019–October 2019)
2.3.2. Study Period (4 Weeks between October and November 2019)
2.4. Data Collection and Analysis
Video Data
2.5. Coding Scheme
2.6. Statistical Analyses
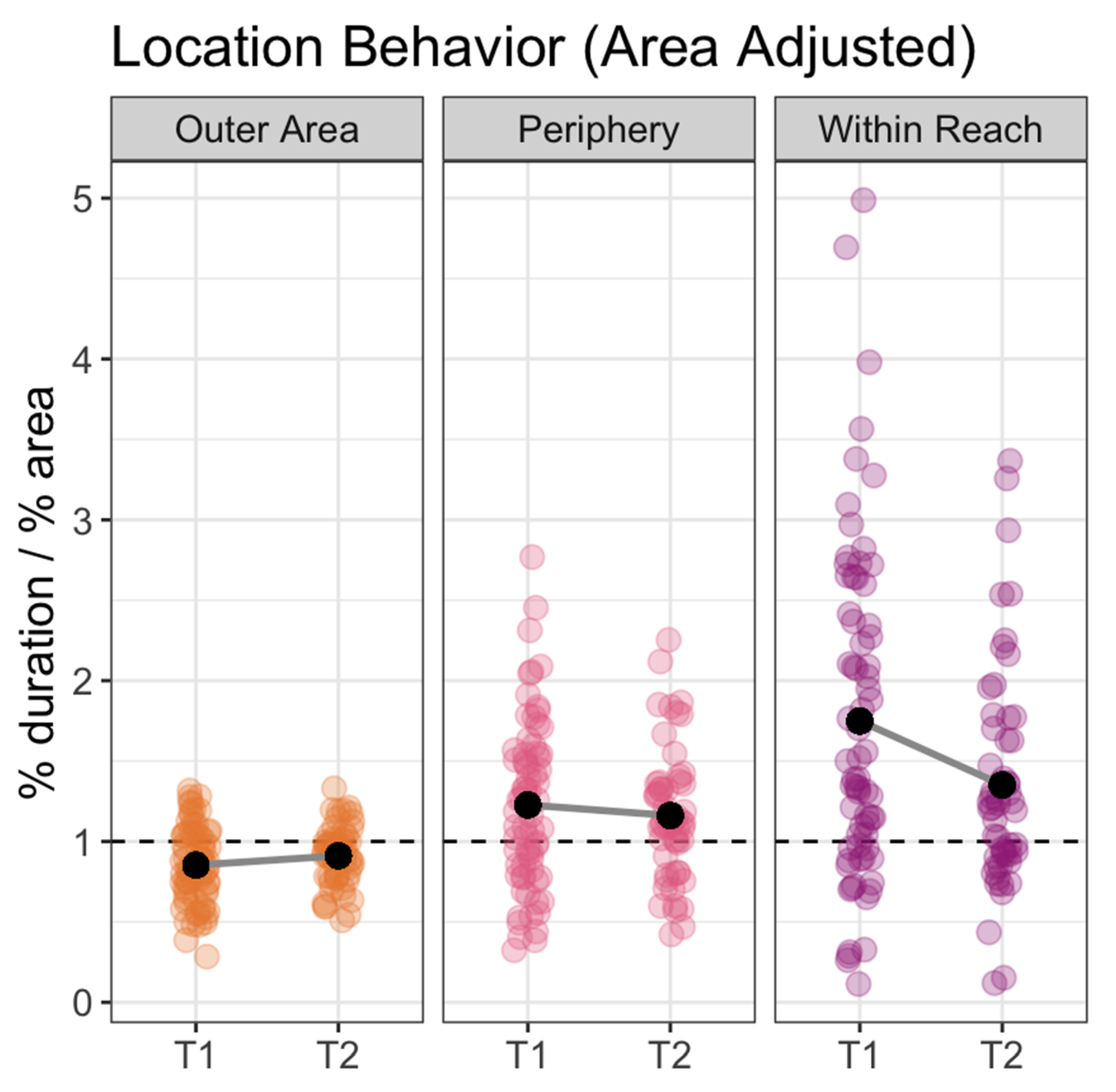
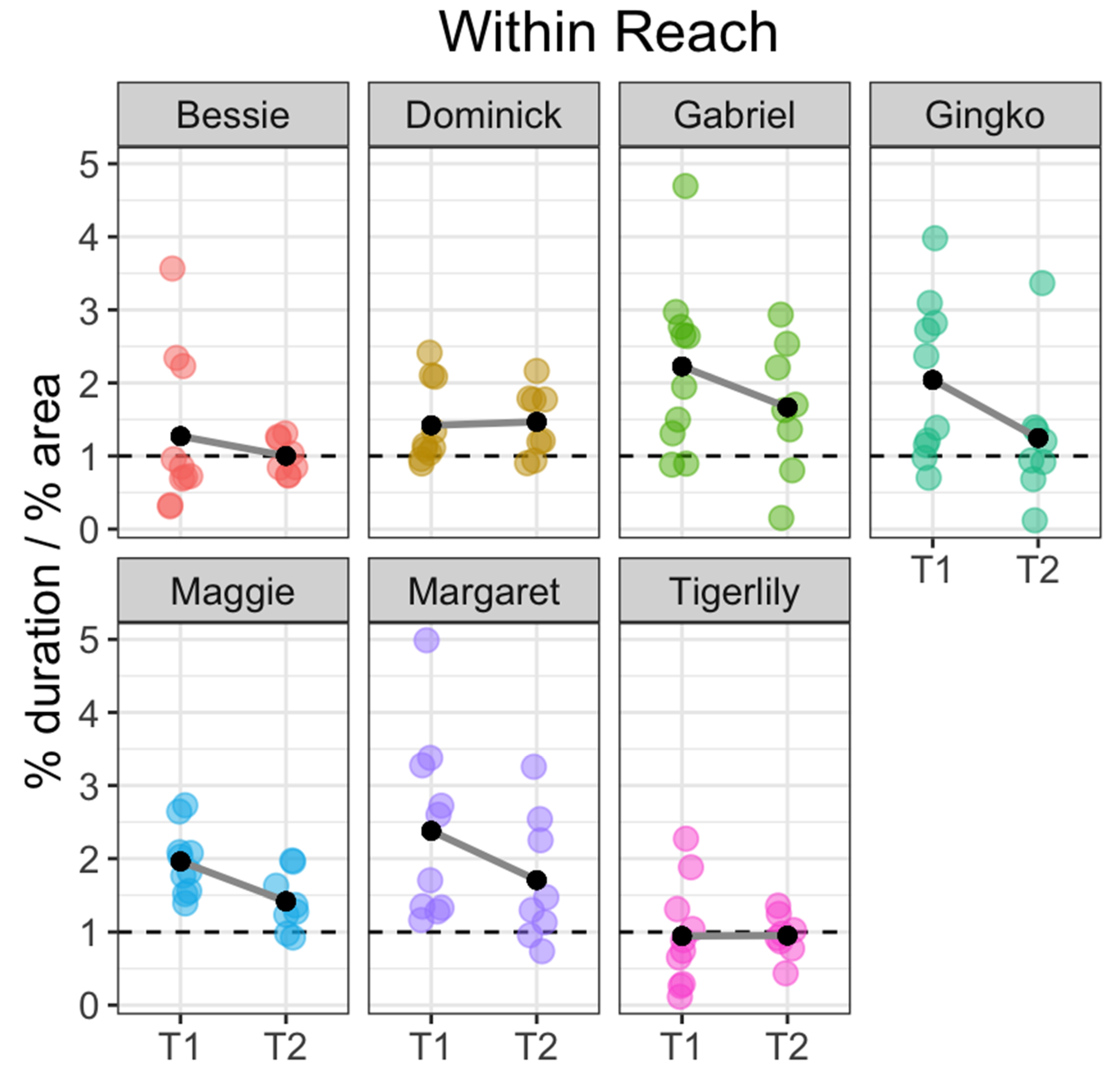
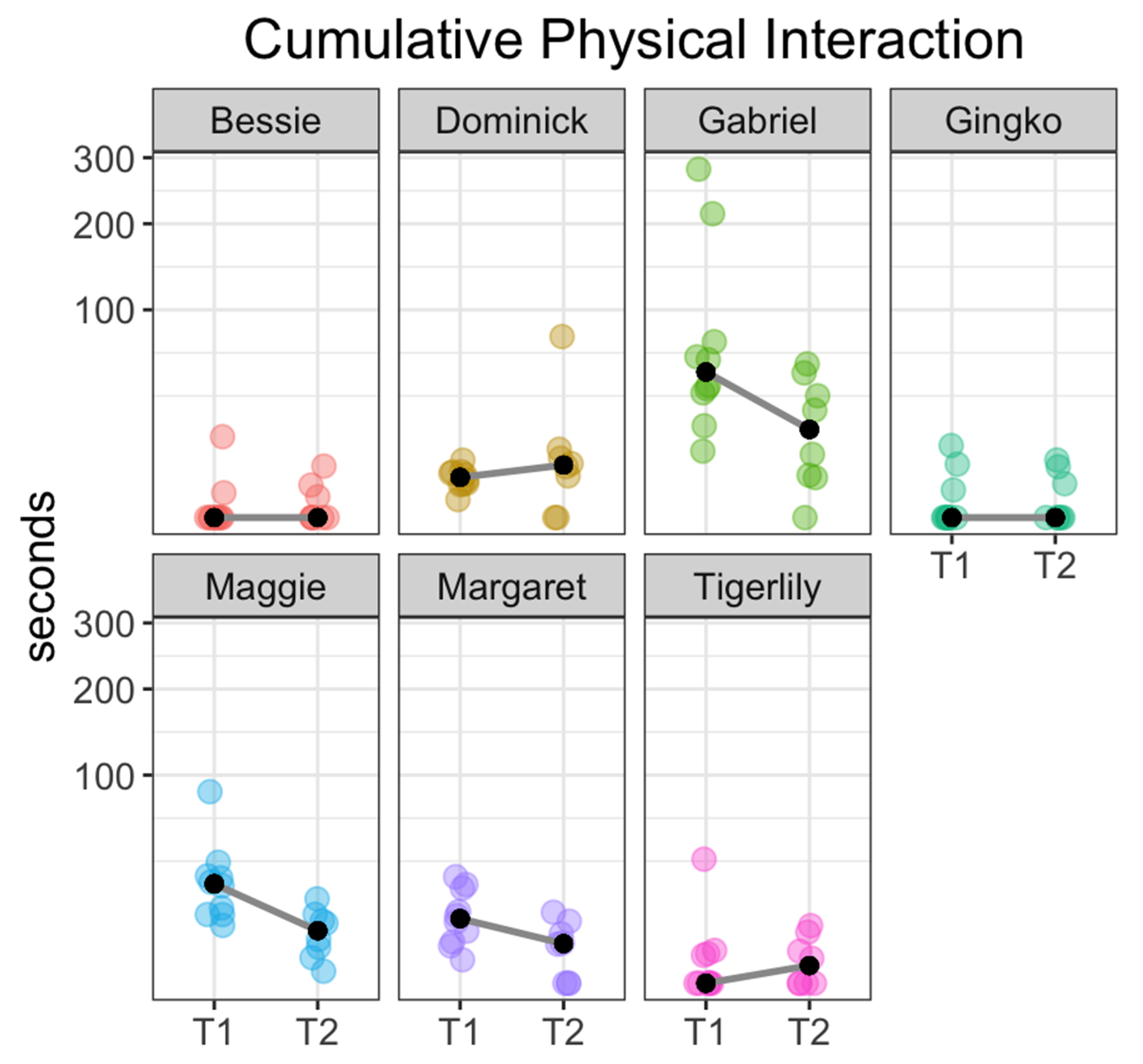
3. Results
4. Discussion
4.1. Reducing Stress through Socialization
4.2. Human–Fish Interactions: A Potential Form of Enrichment for Captive Fishes?
4.2.1. Pleasant Sensory Experience
4.2.2. Cognitive and Social Enrichment
4.3. Koi Personality, Sociality and Future Directions in Carp Welfare Research
4.4. Human–Fish Interactions and Animal Protection—Promoting Human Empathy and Compassion through Positive Interspecies Exchanges
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hosey, G.; Melfi, V. Human-Animal Interactions, Relationships and Bonds: A Review and Analysis of the Literature. Int. J. Comp. Psychol. 2014, 27, 117–142. [Google Scholar]
- Echeverri, A.; Karp, D.S.; Naidoo, R.; Zhao, J.; Chan, K.M.A. Approaching human-animal relationships from multiple angles: A synthetic perspective. Biol. Conserv. 2018, 224, 50–62. [Google Scholar] [CrossRef]
- Tarazona, A.M.; Ceballos, M.C.; Broom, D.M. Human relationships with domestic and other animals: One health, one welfare, one biology. Animals 2020, 10, 43. [Google Scholar] [CrossRef] [PubMed]
- Griffin, J.A.; Hurley, K.; McCune, S. Human-Animal Interaction Research: Progress and Possibilities. Front. Psychol. 2019, 10, 2803. [Google Scholar] [CrossRef] [PubMed]
- Mellor, D.J. Moving beyond the “Five freedoms” by updating the “five provisions” and introducing aligned “animal welfare aims”. Animals 2016, 6, 59. [Google Scholar] [CrossRef]
- Mellor, D.J. Welfare-aligned Sentience: Enhanced Capacities to Experience, Interact, Anticipate, Choose and Survive. Anim. Open Access J. 2019, 9, 440. [Google Scholar] [CrossRef]
- Mellor, D.J. Enhancing animal welfare by creating opportunities for positive affective engagement. N. Z. Vet. J. 2014, 1–18. [Google Scholar] [CrossRef]
- FAO. World Fisheries and Aquaculture; FAO: Rome, Italy, 2018; ISBN 9789251305621. [Google Scholar]
- Lidster, K.; Readman, G.D.; Prescott, M.J.; Owen, S.F. International survey on the use and welfare of zebrafish Danio rerio in research. J. Fish Biol. 2017, 90, 1891–1905. [Google Scholar] [CrossRef]
- Reed, B.; Jennings, M. Guidance on the housing and care of Zebrafish Danio rerio. Res. Anim. Dep. Sci. Group RSPCA 2011, 1, 1–27. [Google Scholar]
- AVMA U.S. Pet Ownership Statistics|American Veterinary Medical Association. Available online: https://www.avma.org/resources-tools/reports-statistics/us-pet-ownership-statistics (accessed on 20 December 2020).
- Brown, C. Fish intelligence, sentience and ethics. Anim. Cogn. 2015, 18, 1–17. [Google Scholar] [CrossRef]
- Brown, C.; Laland, K.; Krause, J. Fish Cognition and Behavior; Brown, C., Laland, K., Krause, J., Eds.; Wiley-Blackwell: Oxford, UK, 2011; ISBN 9781444342536. [Google Scholar]
- Saraiva, J.L.; Castanheira, M.F.; Arechavala-López, P.; Volstorf, J.; Heinzpeter Studer, B. Domestication and Welfare in Farmed Fish. In Animal Domestication; IntechOpen: London, UK, 2019. [Google Scholar]
- Fife-Cook, I.; Franks, B. Positive welfare for fishes: Rationale and areas for future study. Fishes 2019, 4, 31. [Google Scholar] [CrossRef]
- Castanheira, M.F. FishEthoBase: Cyprinus Carpio; Fair Fish International Association: Denens, Switzerland, 2020. [Google Scholar]
- Billard, R. The Carp-Biology and Culture, 1st ed.; Springer: London, UK, 1999; ISBN 9781852331184. [Google Scholar]
- Balon, E.K. About the oldest domesticates among fishes. J. Fish Biol. 2004, 65, 1–27. [Google Scholar] [CrossRef]
- Daniel, W.M.; Morningstar, C.R.; Procopio, J. Cyprinus rubrofuscus Lacepède, 1803: U.S. Geological Survey; Nonindigenous Aquatic Species Database: Gainesville, FL, USA, 2000. [Google Scholar]
- Miller, L.; Patronek, G. Understanding the Effects of Maltreatment on Animal Welfare. In Animal Maltreatment; Oxford University Press: Oxford, UK, 2016; pp. 197–233. [Google Scholar]
- Sah, P.; Méndez, J.D.; Bansal, S. A multi-species repository of social networks. Sci. Data 2019, 6, s41597–s42019. [Google Scholar] [CrossRef]
- Zulkifli, I. Review of human-animal interactions and their impact on animal productivity and welfare. J. Anim. Sci. Biotechnol. 2013, 4, 25. [Google Scholar] [CrossRef] [PubMed]
- Stafford, K.J.; Mellor, D.J. Dehorning and disbudding distress and its alleviation in calves. Vet. J. 2005, 169, 337–349. [Google Scholar] [CrossRef]
- Hemsworth, P.H.; Coleman, G.J.; Barnett, J.L.; Jones, R.B. Behavioural responses to humans and the productivity of commercial broiler chickens. Appl. Anim. Behav. Sci. 1994, 41, 101–114. [Google Scholar] [CrossRef]
- Rushen, J.; Taylor, A.A.; de Passillé, A.M. Domestic animals’ fear of humans and its effect on their welfare. Appl. Anim. Behav. Sci. 1999, 65, 285–303. [Google Scholar] [CrossRef]
- Mota-Rojas, D.; Broom, D.M.; Orihuela, A.; Velarde, A.; Napolitano, F.; Alonso-Spilsbury, M. Effects of human-animal relationship on animal productivity and welfare. J. Anim. Behav. Biometeorol. 2020, 8, 196–205. [Google Scholar] [CrossRef]
- Tallet, C.; Rakotomahandry, M.; Herlemont, S.; Prunier, A. Evidence of Pain, Stress, and Fear of Humans During Tail Docking and the Next Four Weeks in Piglets (Sus scrofa domesticus). Front. Vet. Sci. 2019, 6, 462. [Google Scholar] [CrossRef] [PubMed]
- Meagher, R. Is boredom an animal welfare concern? Anim. Welf. 2019, 28, 21–32. [Google Scholar] [CrossRef]
- Cacioppo, J.T.; Cacioppo, S.; Cole, S.W.; Capitanio, J.P.; Goossens, L.; Boomsma, D.I. Loneliness Across Phylogeny and a Call for Comparative Studies and Animal Models. Perspect. Psychol. Sci. 2015, 10, 202–212. [Google Scholar] [CrossRef]
- Burn, C.C. Bestial boredom: A biological perspective on animal boredom and suggestions for its scientific investigation. Anim. Behav. 2017, 130, 141–151. [Google Scholar] [CrossRef]
- Gourkow, N.; Phillips, C.J.C. Effect of cognitive enrichment on behavior, mucosal immunity and upper respiratory disease of shelter cats rated as frustrated on arrival. Prev. Vet. Med. 2016, 131, 103–110. [Google Scholar] [CrossRef]
- Deldalle, S.; Gaunet, F. Effects of 2 training methods on stress-related behaviors of the dog (Canis familiaris) and on the dog-owner relationship. J. Vet. Behav. Clin. Appl. Res. 2014, 9, 58–65. [Google Scholar] [CrossRef]
- Borgi, M.; Cirulli, F. Pet face: Mechanisms underlying human-animal relationships. Front. Psychol. 2016. [Google Scholar] [CrossRef]
- Špinka, M. Social dimension of emotions and its implication for animal welfare. Appl. Anim. Behav. Sci. 2012, 138, 170–181. [Google Scholar] [CrossRef]
- Serpell, J.A. The Human-Animal Bond. In The Oxford Handbook of Animal Studies; Oxford University Press: Oxford, UK, 2017; pp. 80–97. [Google Scholar]
- Fox, C.; Merali, Z.; Harrison, C. Therapeutic and protective effect of environmental enrichment against psychogenic and neurogenic stress. Behav. Brain Res. 2006, 175, 1–8. [Google Scholar] [CrossRef]
- Chang, F.T.; Hart, L.A. Human-animal bonds in the laboratory: How animal behavior affects the perspective of caregivers. ILAR J. 2002, 43, 10–18. [Google Scholar] [CrossRef]
- Fox, R.; Gee, N.R. Changing conceptions of care: Humanization of the companion animal-human relationship. Soc. Anim. 2016, 24, 107–128. [Google Scholar] [CrossRef]
- Glanville, C.R.; Hemsworth, P.H.; Coleman, G.J. Conceptualising dog owner motivations: The pet care competency model and role of “duty of care”. Anim. Welf. 2020, 29, 271–284. [Google Scholar] [CrossRef]
- Hosey, G.; Melfi, V. Human-Animal Bonds Between Zoo Professionals and the Animals in Their Care. Zoo Biol. 2012, 31, 13–26. [Google Scholar] [CrossRef]
- Mersmann, D.; Schmied-Wagner, C.; Nordmann, E.; Graml, C.; Waiblinger, S. Influences on the avoidance and approach behaviour of dairy goats towards an unfamiliar human-An on-farm study. Appl. Anim. Behav. Sci. 2016. [Google Scholar] [CrossRef]
- Schmidt, K. Concepts of Animal Welfare in Relation to Positions in Animal Ethics. Acta Biotheor. 2011, 59, 153–171. [Google Scholar] [CrossRef]
- Coleman, G.J.; Hemsworth, P.H. Training to improve stockperson beliefs and behaviour towards livestock enhances welfare and productivity. OIE Rev. Sci. Tech. 2014, 33, 131–137. [Google Scholar] [CrossRef] [PubMed]
- LaFollette, M.R.; Cloutier, S.; Brady, C.; Gaskill, B.N.; O’Haire, M.E. Laboratory animal welfare and human attitudes: A cross-sectional survey on heterospecific play or “rat tickling”. PLoS ONE 2019, 14, e0220580. [Google Scholar] [CrossRef] [PubMed]
- Handlin, L.; Hydbring-Sandberg, E.; Nilsson, A.; Ejdebäck, M.; Jansson, A.; Uvnäs-Moberg, K. Short-term interaction between dogs and their owners: Effects on oxytocin, cortisol, insulin and heart rate-an exploratory study. Anthrozoos 2011, 24, 301–315. [Google Scholar] [CrossRef]
- Luebke, J.F.; Watters, J.V.; Packer, J.; Miller, L.J.; Powell, D.M. Zoo Visitors’ Affective Responses to Observing Animal Behaviors. Visit. Stud. 2016, 19, 60–76. [Google Scholar] [CrossRef]
- Mellor, D.J.; Webster, J.R. Development of animal welfare understanding drives change in minimum welfare standards. Rev. Sci. Tech. Off. Int. Epizoot. 2014, 33, 121–130. [Google Scholar] [CrossRef]
- Hemsworth, P.H.; Barnett, J.L.; Coleman, G.J. The integration of human-animal relations into animal welfare monitoring schemes. Anim. Welf. 2009, 18, 335–345. [Google Scholar]
- Luna, D.; Tadich, T. Why Should Human-Animal Interactions Be Included in Research of Working Equids’ Welfare? Animals 2019, 9, 42. [Google Scholar] [CrossRef]
- Hemsworth, P.H.; Sherwen, S.L.; Coleman, G.J. Human contact. In Animal Welfare; CABI: Wallingford, UK, 2018; pp. 294–314. [Google Scholar]
- Mellor, D.J.; Beausoleil, N.J.; Littlewood, K.E.; McLean, A.N.; McGreevy, P.D.; Jones, B.; Wilkins, C. The 2020 Five Domains Model: Including Human–Animal Interactions in Assessments of Animal Welfare. Animals 2020, 10, 1870. [Google Scholar] [CrossRef]
- Brydges, N.M.; Boulcott, P.; Ellis, T.; Braithwaite, V.A. Quantifying stress responses induced by different handling methods in three species of fish. Appl. Anim. Behav. Sci. 2009, 1, 295–301. [Google Scholar] [CrossRef]
- Davie, P.S.; Kopf, R.K. Physiology, behaviour and welfare of fish during recreational fishing and after release. N. Z. Vet. J. 2006, 54, 161–172. [Google Scholar] [CrossRef]
- Rotllant, J.; Tort, L. Cortisol and glucose responses after acute stress by net handling in the sparid red porgy previously subjected to crowding stress. J. Fish Biol. 1997, 51, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Balasch, J.C.; Tort, L. Netting the stress responses in fish. Front. Endocrinol. 2019, 10. [Google Scholar] [CrossRef]
- Friard, O.; Gamba, M. BORIS: A free, versatile open-source event-logging software for video/audio coding and live observations. Methods Ecol. Evol. 2016, 7. [Google Scholar] [CrossRef]
- Gelman, A.; Hill, J. Data Analysis Using Regression and Multilevel/Hierarchical Models; Cambridge University Press: Cambridge, UK, 2006. [Google Scholar]
- Snijders, T.A.B.; Bosker, R.J. Multilevel Analysis An Introduction to Basic and Advanced Multilevel Modeling, 2nd ed.; SAGE Publications: Thousand Oaks, CA, USA, 2012. [Google Scholar]
- R Studio (Ed.) R Studio Team R Studio; R Studio: Boston, MA, USA, 2020; Available online: http//www.rstudio.com/ (accessed on 12 November 2020).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019; Available online: https://www.r-project.org/ (accessed on 31 January 2021).
- Wickham, H.; Averick, M.; Bryan, J.; Chang, W.; McGowan, L.; François, R.; Grolemund, G.; Hayes, A.; Henry, L.; Hester, J.; et al. Welcome to the Tidyverse. J. Open Source Softw. 2019, 4, 1686. [Google Scholar] [CrossRef]
- Bates, D.; Mächler, M.; Bolker, B.M.; Walker, S.C. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Kuznetsova, A.; Brockhoff, P.B.; Christensen, R.H.B. lmerTest Package: Tests in Linear Mixed Effects Models. J. Stat. Softw. 2017, 82, 1–26. [Google Scholar] [CrossRef]
- Hemsworth, P.H.; Verge, J.; Coleman, G.J. Conditioned approach-avoidance responses to humans: The ability of pigs to associate feeding and aversive social experiences in the presence of humans with humans. Appl. Anim. Behav. Sci. 1996, 50, 71–82. [Google Scholar] [CrossRef]
- De Passillé, A.M.; Rushen, J.; Ladewig, J.; Petherick, C. Dairy Calves’ Discrimination of People Based on Previous Handling. J. Anim. Sci. 1996, 74, 969–974. [Google Scholar] [CrossRef]
- Stoskopf, M.K. The physiological effects of psychological stress. Zoo Biol. 1983, 2, 179–190. [Google Scholar] [CrossRef]
- Wielebnowski, N. Stress and distress: Evaluating their impact for the well-being of zoo animals. Proc. J. Am. Vet. Med Assoc. 2003, 223, 973–977. [Google Scholar] [CrossRef]
- Yada, T.; Tort, L. Stress and Disease Resistance: Immune System and Immunoendocrine Interactions. In Fish Physiology; Elsevier Inc.: Amsterdam, The Netherlands, 2016; Volume 35, pp. 365–403. [Google Scholar]
- Demin, K.A.; Taranov, A.S.; Ilyin, N.P.; Lakstygal, A.M.; Volgin, A.D.; de Abreu, M.S.; Strekalova, T.; Kalueff, A.V. Understanding neurobehavioral effects of acute and chronic stress in zebrafish. Stress 2020, 24, 1–18. [Google Scholar] [CrossRef]
- Galhardo, L.; Oliveira, R.F. Psychological stress and welfare in fish. Annu. Rev. Biomed. Sci. 2009, 11, 1–20. [Google Scholar]
- Nardocci, G.; Navarro, C.; Cortés, P.P.; Imarai, M.; Montoya, M.; Valenzuela, B.; Jara, P.; Acuña-Castillo, C.; Fernández, R. Neuroendocrine mechanisms for immune system regulation during stress in fish. Fish Shellfish Immunol. 2014, 40, 531–538. [Google Scholar] [CrossRef]
- Bergamasco, L.; Osella, M.C.; Savarino, P.; Larosa, G.; Ozella, L.; Manassero, M.; Badino, P.; Odore, R.; Barbero, R.; Re, G. Heart rate variability and saliva cortisol assessment in shelter dog: Human-animal interaction effects. Appl. Anim. Behav. Sci. 2010, 125, 56–68. [Google Scholar] [CrossRef]
- Bertenshaw, C.; Rowlinson, P.; Edge, H.; Douglas, S.; Shiel, R. The effect of different degrees of “positive” human-animal interaction during rearing on the welfare and subsequent production of commercial dairy heifers. Appl. Anim. Behav. Sci. 2008, 114, 65–75. [Google Scholar] [CrossRef]
- Schmied, C.; Boivin, X.; Scala, S.; Waiblinger, S. Effect of previous stroking on reactions to a veterinary procedure. Interact. Stud. 2010, 11, 467–481. [Google Scholar] [CrossRef]
- Masud, N.; Ellison, A.; Cable, J. A neglected fish stressor: Mechanical disturbance during transportation impacts susceptibility to disease in a globally important ornamental fish. Dis. Aquat. Organ. 2019, 134, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Stevens, C.H.; Croft, D.P.; Paull, G.C.; Tyler, C.R. Stress and welfare in ornamental fishes: What can be learned from aquaculture? J. Fish Biol. 2017, 91, 409–428. [Google Scholar] [CrossRef]
- Saeij, J.P.J.; Verburg-Van Kemenade, L.B.M.; Van Muiswinkel, W.B.; Wiegertjes, G.F. Daily handling stress reduces resistance of carp to Trypanoplasma borreli: In vitro modulatory effects of cortisol on leukocyte function and apoptosis. Dev. Comp. Immunol. 2003, 27, 233–245. [Google Scholar] [CrossRef]
- Field, T.; Hernandez-Reif, M.; Diego, M.; Schanberg, S.; Kuhn, C. Cortisol decreases and serotonin and dopamine increase following massage therapy. Int. J. Neurosci. 2005, 115, 1397–1413. [Google Scholar] [CrossRef]
- Balcombe, J. Animal pleasure and its moral significance. Appl. Anim. Behav. Sci. 2009, 118, 208–216. [Google Scholar] [CrossRef]
- Waiblinger, S.; Menke, C.; Korff, J.; Bucher, A. Previous handling and gentle interactions affect behaviour and heart rate of dairy cows during a veterinary procedure. Appl. Anim. Behav. Sci. 2004, 85, 31–42. [Google Scholar] [CrossRef]
- Bertenshaw, C.E.; Rowlinson, P. Exploring heifers’ perception of “positive” treatment through their motivation to pursue a retreated human. Anim. Welf. 2008, 17, 313–319. [Google Scholar]
- Boissy, A.; Bouissou, M.F. Effects of early handling on heifers’ subsequent reactivity to humans and to unfamiliar situations. Appl. Anim. Behav. Sci. 1988, 20, 259–273. [Google Scholar] [CrossRef]
- Markowitz, T.M.; Dally, M.R.; Gursky, K.; Price, E.O. Early handling increases lamb affinity for humans. Anim. Behav. 1998, 55, 573–587. [Google Scholar] [CrossRef][Green Version]
- Serrapica, M.; Boivin, X.; Coulon, M.; Braghieri, A.; Napolitano, F. Positive perception of human stroking by lambs: Qualitative behaviour assessment confirms previous interpretation of quantitative data. Appl. Anim. Behav. Sci. 2017, 187, 31–37. [Google Scholar] [CrossRef]
- Zulkifli, I.; Gilbert, J.; Liew, P.K.; Ginsos, J. The effects of regular visual contact with human beings on fear, stress, antibody and growth responses in broiler chickens. Appl. Anim. Behav. Sci. 2002, 79, 103–112. [Google Scholar] [CrossRef]
- Tallet, C.; Brajon, S.; Devillers, N.; Lensink, J. Pig-human interactions: Creating a positive perception of humans to ensure pig welfare. In Advances in Pig Welfare; Elsevier Inc.: Amsterdam, The Netherlands, 2018; pp. 381–398. ISBN 9780081011195. [Google Scholar]
- Terlouw, E.M.C.; Porcher, J. Repeated handling of pigs during rearing. I. Refusal of contact by the handler and reactivity to familiar and unfamiliar humans. J. Anim. Sci. 2005, 83, 1653–1663. [Google Scholar] [CrossRef]
- Gonyou, H.W.; Hemsworth, P.H.; Barnett, J.L. Effects of frequent interactions with humans on growing pigs. Appl. Anim. Behav. Sci. 1986, 16, 269–278. [Google Scholar] [CrossRef]
- Wells, D.L. Sensory stimulation as environmental enrichment for captive animals: A review. Appl. Anim. Behav. Sci. 2009, 118, 1–11. [Google Scholar] [CrossRef]
- Feh, C.; de Mazierès, J. Grooming at a preferred site reduces heart rate in horses. Anim. Behav. 1993, 46, 1191–1194. [Google Scholar] [CrossRef]
- McGreevy, P.D.; Righetti, J.; Thomson, P.C. The reinforcing value of physical contact and the effect on canine heart rate of grooming in different anatomical areas. In Proceedings of the Anthrozoos; Routledge: London, UK, 2005; Volume 18, pp. 236–244. [Google Scholar]
- Kasumyan, A.O. Tactile reception and behavior of fish. J. Ichthyol. 2011, 51, 1035–1103. [Google Scholar] [CrossRef]
- Soares, M.C.; Oliveira, R.F.; Ros, A.F.H.; Grutter, A.S.; Bshary, R. Tactile stimulation lowers stress in fish. Nat. Commun. 2011, 2. [Google Scholar] [CrossRef] [PubMed]
- Bshary, R.; Wurth, M. Cleaner fish Labroides dimidiatus manipulate client reef fish by providing tactile stimulation. Proc. Biol. Sci. 2001, 268, 1495–1501. [Google Scholar] [CrossRef] [PubMed]
- Meehan, C.L.; Mench, J.A. The challenge of challenge: Can problem solving opportunities enhance animal welfare? Appl. Anim. Behav. Sci. 2007, 102, 246–261. [Google Scholar] [CrossRef]
- Nawroth, C.; Langbein, J.; Coulon, M.; Gabor, V.; Oesterwind, S.; Benz-Schwarzburg, J.; von Borell, E. Farm Animal Cognition—Linking Behavior, Welfare and Ethics. Front. Vet. Sci. 2019, 6, 24. [Google Scholar] [CrossRef]
- Clark, F.E. Animal Behavior and Cognition Cognitive Enrichment and Welfare: Current Approaches and Future Directions. ABC 2017, 2017, 52–71. [Google Scholar] [CrossRef]
- Jensvold, M.L.A. Chimpanzee (Pan troglodytes) responses to caregiver use of chimpanzee behaviors. Zoo Biol. 2008, 27, 345–359. [Google Scholar] [CrossRef] [PubMed]
- Chelluri, G.I.; Ross, S.R.; Wagner, K.E. Behavioral correlates and welfare implications of informal interactions between caretakers and zoo-housed chimpanzees and gorillas. Appl. Anim. Behav. Sci. 2013, 147, 306–315. [Google Scholar] [CrossRef]
- Carlstead, K.; Paris, S.; Brown, J.L. Good keeper-elephant relationships in North American zoos are mutually beneficial to welfare. Appl. Anim. Behav. Sci. 2019, 211, 103–111. [Google Scholar] [CrossRef]
- Tetley, C.L.; O’Hara, S.J. Ratings of animal personality as a tool for improving the breeding, management and welfare of zoo mammals. Anim. Welf. 2012, 21, 463–476. [Google Scholar] [CrossRef]
- Williams, E.; Carter, A.; Hall, C.; Bremner-Harrison, S. Social Interactions in Zoo-Housed Elephants: Factors Affecting Social Relationships. Animals 2019, 9, 747. [Google Scholar] [CrossRef]
- Rose, P.E.; Croft, D.P. The potential of social network analysis as a tool for the management of zoo animals. Anim. Welf. 2015, 24, 123–138. [Google Scholar] [CrossRef]
- Afonso, L.O.B. Identifying and managing maladaptive physiological responses to aquaculture stressors. In Fish Physiology; Elsevier Inc.: Amsterdam, The Netherlands, 2020. [Google Scholar]
- Franks, B.; Graham, C.; von Keyserlingk, M.A.G. Is Heightened-Shoaling a Good Candidate for Positive Emotional Behavior in Zebrafish? Anim. Open Access J. 2018, 8, 152. [Google Scholar] [CrossRef]
- Filby, A.L.; Paull, G.C.; Bartlett, E.J.; Van Look, K.J.W.; Tyler, C.R. Physiological and health consequences of social status in zebrafish (Danio rerio). Physiol. Behav. 2010, 101, 576–587. [Google Scholar] [CrossRef]
- Bayne, K. Development of the human-research animal bond and its impact on animal well-being. ILAR J. 2002. [Google Scholar] [CrossRef]
- Rucinque, D.S.; Souza, A.P.O.; Molento, C.F.M. Perception of fish sentience, welfare and humane slaughter by highly educated citizens of Bogotá, Colombia and Curitiba, Brazil. PLoS ONE 2017, 12. [Google Scholar] [CrossRef]
- Message, R.; Greenhough, B. “But It’s Just a Fish”: Understanding the Challenges of Applying the 3Rs in Laboratory Aquariums in the UK. Animals 2019, 9, 75. [Google Scholar] [CrossRef] [PubMed]
- Yue Cottee, S. Are fish the victims of ‘speciesism’? A discussion about fear, pain and animal consciousness. Fish Physiol. Biochem. 2012, 38, 5–15. [Google Scholar] [CrossRef] [PubMed]



| Name | Sex | Length | Distinguishing Features |
|---|---|---|---|
| Bessie | M | 40–45 cm | Primarily grey with lemon yellow coloration on the head and back above lateral line and before dorsal fin. Partially scaled. |
| Dominick | M | 55–60 cm | Deep yellow-orange butterfly koi with dark scale outlines. Mass on left side of abdomen, left eye is disfigured and nonfunctional. Fully scaled. |
| Gabriel | M | 55–60 cm | Yellow-gold body tapering to a deeper shade of squash towards the face with white tipped pectoral and dorsal fins. Partially scaled. |
| Maggie | F | 50–55 cm | White with orange markings on either side of the dorsal fin above the lateral line and anterior to the dorsal fin across the medial line. Fully scaled. |
| Margaret | F | 50–55 cm | Primarily orange and black above lateral line and white below. Partial piebald. Mouth is disfigured but functional. Fully scaled. |
| Gingko | M | 45–50 cm | Primarily light grey-blue with dark crescent-shaped markings along the medial line and dark freckles across body and fins. Partially scaled. |
| Tigerlily | M | 50–55 cm | Primarily orange above lateral line and white underbelly. Black markings on body and fins. Orange head and operculum with white mouth. Partially scaled. |
| Location | Definition | Mean Area | % Total Area |
|---|---|---|---|
| Within Reach | Area of the tank designated by the arc created by the researcher using her submerged arms full range of motion in all directions without leaning forward. | 0.4 m2 | 8% |
| Periphery | Periphery began at the boundary of within reach and extending approximately one arm’s length beyond the within reach area in all directions. In this location, the researcher’s hand was likely still within sight of the fish while also remaining out of immediate reach. | 1.15 m2 | 22% |
| Outer Area | Included all areas of the tank outside of the previously defined locations (including areas that are out of sight in video data, see Figure 2). | 3.1 m2 | 70% |
| Physical Interaction | Any form of tactile interaction between human hand and fish, initiated by the fish (mouthing, brushing, bumping, etc.) A series of tactile interactions were designated as a single period of interaction as long as the fish instigated physical interaction at least once over the course of 3 s and did not move away from the researcher. If interactions occurred more than 3 s apart and/or the fish moved away from the researcher between interactions, they were coded as individual periods. | N/A | N/A |
| Rate of Physical Interaction per Minute | |||
|---|---|---|---|
| Fish | Overall Rate (Mean Times/Min) | Rate by Session Type (Mean Times/Min) | |
| Bessie | 0.02 | T1 | 0.03 |
| T2 | 0.01 | ||
| Diff | −0.02 | ||
| Dominick | 0.16 | T1 | 0.17 |
| T2 | 0.14 | ||
| Diff | −0.03 | ||
| Gabriel | 0.48 | T1 | 0.63 |
| T2 | 0.29 | ||
| Diff | −0.35 | ||
| Gingko | 0.05 | T1 | 0.06 |
| T2 | 0.04 | ||
| Diff | −0.02 | ||
| Maggie | 0.18 | T1 | 0.24 |
| T2 | 0.10 | ||
| Diff | −0.14 | ||
| Margaret | 0.14 | T1 | 0.19 |
| T2 | 0.19 | ||
| Diff | −0.1 | ||
| Tigerlily | 0.05 | T1 | 0.06 |
| T2 | 0.03 | ||
| Diff | −0.02 | ||
| Cumulative Physical Interaction Within a Single Session (20 Min) | |||
|---|---|---|---|
| Fish | Duration Overall (Median and Range in Seconds) | Duration by Session Type (Median and Range in Seconds) | |
| Bessie | 0 (0, 15.2) | T1 | 0 (0, 15.2) |
| T2 | 0 (0, 6.14) | ||
| Dominick | 4.4 (0, 75.9) | T1 | 3.75 (0.75, 7.62) |
| T2 | 6.39 (0, 75.9) | ||
| Gabriel | 37.3 (0, 281) | T1 | 49.1 (10.2, 281) |
| T2 | 18 (0, 54.7) | ||
| Gingko | 0 (0, 12) | T1 | 0 (0, 12) |
| T2 | 0 (0, 7.68) | ||
| Maggie | 11.0 (0.34, 84.7) | T1 | 22.8 (7.75, 84.7) |
| T2 | 6.39 (0.34, 16.5) | ||
| Margaret | 5.96 (0, 26.1) | T1 | 9.66 (1.27, 26.1) |
| T2 | 3.65 (0, 11.7) | ||
| Tigerlily | 0 (0, 35.4) | T1 | 0 (0, 35.4) |
| T2 | 0.72 (0, 7.68) | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fife-Cook, I.; Franks, B. Koi (Cyprinus rubrofuscus) Seek Out Tactile Interaction with Humans: General Patterns and Individual Differences. Animals 2021, 11, 706. https://doi.org/10.3390/ani11030706
Fife-Cook I, Franks B. Koi (Cyprinus rubrofuscus) Seek Out Tactile Interaction with Humans: General Patterns and Individual Differences. Animals. 2021; 11(3):706. https://doi.org/10.3390/ani11030706
Chicago/Turabian StyleFife-Cook, Isabel, and Becca Franks. 2021. "Koi (Cyprinus rubrofuscus) Seek Out Tactile Interaction with Humans: General Patterns and Individual Differences" Animals 11, no. 3: 706. https://doi.org/10.3390/ani11030706
APA StyleFife-Cook, I., & Franks, B. (2021). Koi (Cyprinus rubrofuscus) Seek Out Tactile Interaction with Humans: General Patterns and Individual Differences. Animals, 11(3), 706. https://doi.org/10.3390/ani11030706






