How to Improve Meat Quality and Welfare in Entire Male Pigs by Genetics
Abstract
Simple Summary
Abstract
1. Introduction
2. Genetics for Better Quality in Entire Males
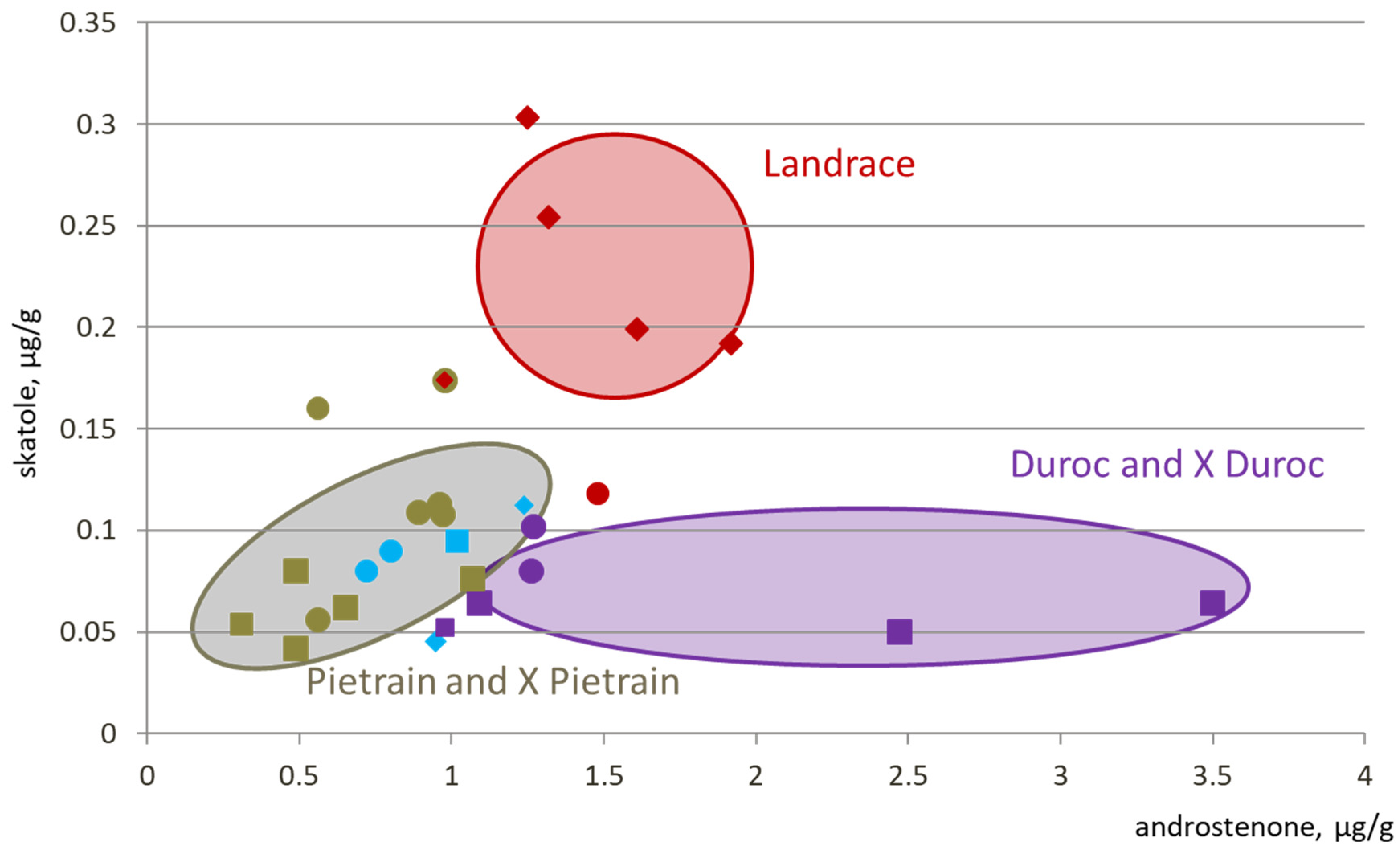
2.1. Breed Effect
2.2. Heritability and Genetic Correlations for Boar Taint Related Traits
2.3. Genetic Relationships between Boar Taint and Traits of Economic Interest
2.4. Selection against Boar Taint
2.5. Selection against Boar Taint in Practice
3. Genetics for Better Entire Male Welfare
3.1. Breed Comparison
3.2. Genetic Parameters for Skin Lesions and Aggressive Behavior and Selection Strategies
4. Perspectives
4.1. New Phenotyping
4.2. Genomic Information
5. Conclusions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Directorate-General for Health and Food Safety; European Union. Establishing Best Practices on the Production, the Processing and the Marketing of Meat from Uncastrated Pigs or Pigs Vaccinated Against Boar Taint (Immunocastrated); Publications Office of the European Union: Luxembourg, 2019. [Google Scholar] [CrossRef]
- Engesser, D.J. Alternatives for Boar Taint Reduction and Elimination Besides Surgical Castration and Destroying Testicular Tissue. Ph.D. Thesis, University of Leipzig, Leipzig, Germany, 2015. [Google Scholar]
- Brinke, I.; Grosse-Brinkhaus, C.; Roth, K.; Proll-Cornelissen, M.J.; Henne, H.; Schellander, K.; Tholen, E. Genomic background and genetic relationships between boar taint and fertility traits in German Landrace and Large White. BMC Genet. 2020, 21, 61. [Google Scholar] [CrossRef] [PubMed]
- Xue, J.L.; Dial, G.D.; Holton, E.E.; Vickers, Z.; Squires, E.J.; Loy, Y.P.; Godbout, D.; Morel, N. Breed differences in boar taint: Relationship between tissue levels of boar taint compounds and sensory analysis of taint. J. Anim. Sci. 1996, 74, 2170–2177. [Google Scholar] [CrossRef]
- Tajet, H.; Andresen, Ø.; Meuwissen, T. Estimation of genetic parameters of boar taint; skatole and androstenone and their correlations with sexual maturation. Acta Vet. Scand. 2006, 48, S9. [Google Scholar] [CrossRef]
- Oskam, I.C.; Lervik, S.; Tajet, H.; Dahl, E.; Ropstad, E.; Andresen, O. Differences in testosterone, androstenone, and skatole levels in plasma and fat between pubertal purebred Duroc and Landrace boars in response to human chorionic gonadotrophin stimulation. Theriogenology 2010, 74, 1088–1098. [Google Scholar] [CrossRef]
- Grindflek, E.; Meuwissen, T.H.E.; Aasmundstad, T.; Hamland, H.; Hansen, M.H.S.; Nome, T.; Kent, M.; Torjesen, P.; Lien, S. Revealing genetic relationships between compounds affecting boar taint and reproduction in pigs. J. Anim. Sci. 2011, 89, 680–692. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Andersen-Ranberg, I.; van Son, M.; Grindflek, E. Genetic parameters and genetic trends for boar taint compounds in Landrace and Duroc. In Proceedings of the World Congress on Genetics Applied to Livestock Production, Montpellier, France, 7–11 February 2018; p. 548. [Google Scholar]
- Grosse-Brinkhaus, C.; Storck, L.C.; Frieden, L.; Neuhoff, C.; Schellander, K.; Looft, C.; Tholen, E. Genome-wide association analyses for boar taint components and testicular traits revealed regions having pleiotropic effects. BMC Genet. 2015, 16, 36. [Google Scholar] [CrossRef] [PubMed]
- Windig, J.J.; Mulder, H.A.; Napel, J.T.; Knol, E.F.; Mathur, P.K.; Crump, R.E. Genetic parameters for androstenone, skatole, indole, and human nose scores as measures of boar taint and their relationship with finishing traits. J. Anim. Sci. 2012, 90, 2120–2129. [Google Scholar] [CrossRef][Green Version]
- Bonneau, M.; Desmoulin, B.; Dumont, B.L. Organoleptic quality of meat from entire or castrated male pigs—Composition of fatty tissues and boar taint in double muscled breeds. Ann. Zootech. 1979, 28, 53–72. [Google Scholar] [CrossRef]
- Sellier, P. Genetics of meat and carcass traits. In The Genetics of the Pig; Rothschild, M.F., Ruvinsky, A., Eds.; CAB International: Wallingford, UK, 1998; pp. 463–510. [Google Scholar]
- Frieden, L.; Neuhoff, C.; Grosse-Brinkhaus, C.; Cinar, M.U.; Schellander, K.; Looft, C.; Tholen, E. Breeding potential of selection against boar taint. Zuchtungskunde 2012, 84, 394–411. [Google Scholar]
- Mathur, P.K.; Napel, J.T.; Crump, R.E.; Mulder, H.A.; Knol, E.F. Genetic relationship between boar taint compounds, human nose scores, and reproduction traits in pigs. J. Anim. Sci. 2013, 91, 4080–4089. [Google Scholar] [CrossRef]
- Dugue, C.; Prunier, A.; Mercat, M.J.; Monziols, M.; Blanchet, B.; Larzul, C. Genetic determinism of boar taint and relationship with growth traits, meat quality and lesions. Animal 2020, 14, 1333–1341. [Google Scholar] [CrossRef]
- Robic, A.; Larzul, C.; Bonneau, M. Genetic and metabolic aspects of androstenone and skatole deposition in pig adipose tissue: A review. Genet. Sel. Evol. 2008, 40, 129–143. [Google Scholar] [CrossRef] [PubMed]
- Parois, S.; Bonneau, M.; Chevillon, P.; Larzul, C.; Quiniou, N.; Robic, A.; Prunier, A. Boar taint in the meat of entire male pigs: The problems and the potential solutions. INRA Prod. Anim. 2018, 31, 23–35. [Google Scholar] [CrossRef]
- Wismer-Pedersen, J.; Jonsson, P.; Jensen, P.; Banyai, A. The occurrence of sex odour in Danish Landrace boars. In Meat Production from Entire Male Animals; Rhodes, D.N., Ed.; J&A Churchill Ltd.: London, UK, 1969; pp. 285–295. [Google Scholar]
- Alsing, W.; Claus, R.; Pirchner, F.; Willeke, H. Selektionsexperiment auf Ebergeruch. Genet. Sel. Evol. 1978, 10, 148. [Google Scholar] [CrossRef]
- Ciobanu, D.C.; Lonergan, S.M.; Huff-Lonergan, E.J. Genetics of meat quality and carcass traits. In The Genetics of the Pig, 2nd ed.; Rothschild, M.F., Ruvinsky, A., Eds.; CAB International: Wallingford, UK, 2011; pp. 355–389. [Google Scholar] [CrossRef]
- Willeke, H.; Claus, R.; Muller, E.; Pirchner, F.; Karg, H. Selection for high and low-level of 5-α-androst-16-en-3-one in boars, 1: Direct and correlated response of endocrinologic traits. J. Anim. Breed. Genet. 1987, 104, 64–73. [Google Scholar] [CrossRef]
- Tusell, L.; Gilbert, H.; Vitezica, Z.G.; Mercat, M.J.; Legarra, A.; Larzul, C. Dissecting total genetic variance into additive and dominance components of purebred and crossbred pig traits. Animal 2019, 13, 2429–2439. [Google Scholar] [CrossRef]
- Parois, S.P.; Prunier, A.; Mercat, M.J.; Merlot, E.; Larzul, C. Genetic relationships between measures of sexual development, boar taint, health, and aggressiveness in pigs. J. Anim. Sci. 2015, 93, 3749–3758. [Google Scholar] [CrossRef]
- Azevedo, C.F.; Silva, F.F.E.; de Resende, M.D.V.; Lopes, M.S.; Duijvesteijn, N.; Guimaraes, S.E.F.; Lopes, P.S.; Kelly, M.J.; Viana, J.M.S.; Knol, E.F. Supervised independent component analysis as an alternative method for genomic selection in pigs. J. Anim. Breed. Genet. 2014, 131, 452–461. [Google Scholar] [CrossRef] [PubMed]
- Lukic, B.; Pong-Wong, R.; Rowe, S.J.; de Koning, D.J.; Velander, I.; Haley, C.S.; Archibald, A.L.; Woolliams, J.A. Efficiency of genomic prediction for boar taint reduction in Danish Landrace pigs. Anim. Genet. 2015, 46, 607–616. [Google Scholar] [CrossRef]
- de Campos, C.F.; Lopes, M.S.; Silva, F.F.E.; Veroneze, R.; Knol, E.F.; Lopes, P.S.; Guimaraes, S.E.F. Genomic selection for boar taint compounds and carcass traits in a commercial pig population. Livest. Sci. 2015, 174, 10–17. [Google Scholar] [CrossRef]
- Duijvesteijn, N.; Knol, E.F.; Bijma, P. Direct and associative effects for androstenone and genetic correlations with backfat and growth in entire male pigs. J. Anim. Sci. 2012, 90, 2465–2475. [Google Scholar] [CrossRef]
- Sellier, P.; Le Roy, P.; Fouilloux, M.N.; Gruand, J.; Bonneau, M. Responses to restricted index selection and genetic parameters for fat androstenone level and sexual maturity status of young boars. Livest. Prod. Sci. 2000, 63, 265–274. [Google Scholar] [CrossRef]
- Rostellato, R.; Bonfatti, V.; Larzul, C.; Bidanel, J.P.; Carnier, P. Estimates of genetic parameters for content of boar taint compounds in adipose tissue of intact males at 160 and 220 days of age. J. Anim. Sci. 2015, 93, 4267–4276. [Google Scholar] [CrossRef]
- Jonsson, P.; Andresen, O. Experience during two generations of within lines boar performance testing, using 5-α-androst-16-ene-3-one (5α-androstenone) and an olfactory judgement of boar taint. Ann. Genet. Sel. Anim. 1979, 11, 241–250. [Google Scholar] [CrossRef]
- Strathe, A.B.; Velander, I.H.; Mark, T.; Kadarmideen, H.N. Genetic parameters for androstenone and skatole as indicators of boar taint and their relationship to production and litter size traits in Danish Landrace. J. Anim. Sci. 2013, 91, 2587–2595. [Google Scholar] [CrossRef]
- Sellier, P.; Bonneau, M. Genetic-relationships between fat androstenone level in males and development of male and female genital-tract in pigs. J. Anim. Breed. Genet. 1988, 105, 11–20. [Google Scholar] [CrossRef]
- Haberland, A.M.; Luther, H.; Hofer, A.; Tholen, E.; Simianer, H.; Lind, B.; Baes, C. Efficiency of different selection strategies against boar taint in pigs. Animal 2014, 8, 11–19. [Google Scholar] [CrossRef]
- Moerlein, D.; Tholen, E. Fatty acid composition of subcutaneous adipose tissue from entire male pigs with extremely divergent levels of boar taint compounds—An exploratory study. Meat Sci. 2015, 99, 1–7. [Google Scholar] [CrossRef]
- Robic, A.; Faraut, T.; Prunier, A. Pathways and genes involved in steroid hormone metabolism in male pigs: A review and update. J. Steroid Biochem. Mol. Biol. 2014, 140, 44–55. [Google Scholar] [CrossRef]
- Jonsson, P.; Wismer-Pedersen, J. Genetics of sex odour in boars. Livest. Prod. Sci. 1974, 1, 53–66. [Google Scholar] [CrossRef]
- Lervik, S.; Oskam, I.; Krogenaes, A.; Andresen, O.; Dahl, E.; Haga, H.A.; Tajet, H.; Olsaker, I.; Ropstad, E. Androstenone and testosterone levels and testicular morphology of Duroc boars related to estimated breeding value for androstenone. Theriogenology 2013, 79, 986–994. [Google Scholar] [CrossRef]
- Strathe, A.B.; Velander, I.H.; Mark, T.; Ostersen, T.; Hansen, C.; Kadarmideen, H.N. Genetic parameters for male fertility and its relationship to skatole and androstenone in Danish Landrace boars. J. Anim. Sci. 2013, 91, 4659–4668. [Google Scholar] [CrossRef]
- Ducro-Steverink, D. Selection against boar taint: A simulation study. Acta Vet. Scand. 2006, 48 (Suppl. 1), 6. [Google Scholar] [CrossRef]
- Legarra, A.; Christensen, O.F.; Aguilar, I.; Misztal, I. Single Step, a general approach for genomic selection. Livest. Sci. 2014, 166, 54–65. [Google Scholar] [CrossRef]
- Bonneau, M.; Weiler, U. Pros and Cons of alternatives to piglet castration: Welfare, boar taint, and other meat quality traits. Animals 2019, 9, 884. [Google Scholar] [CrossRef]
- Rydhmer, L.; Canario, L. Behavioral Genetics in Pigs and Relations to Welfare. In Genetics and the Behavior of Domestic Animals; Grandin, T., Deesing, M.J., Eds.; Academic Press: Cambridge, MA, USA, 2014; pp. 397–434. [Google Scholar] [CrossRef]
- Hemsworth, P.H.; Tilbrook, A.J. Sexual behavior of male pigs. Horm. Behav. 2007, 52, 39–44. [Google Scholar] [CrossRef]
- Fredriksen, B.; Lium, B.M.; Marka, C.H.; Mosveen, B.; Nafstad, O. Entire male pigs in farrow-to-finish pens—Effects on animal welfare. Appl. Anim. Behav. Sci. 2008, 110, 258–268. [Google Scholar] [CrossRef]
- Signoret, J.P. Le comportement sexuel des ongulés domestiques. Ann. Med. Vet. 1976, 120, 531–540. [Google Scholar]
- D’Eath, R.B.; Turner, S.P.; Kurt, E.; Evans, G.; Thoelking, L.; Looft, H.; Wimmers, K.; Murani, E.; Klont, R.; Foury, A.; et al. Pigs’ aggressive temperament affects pre-slaughter mixing aggression, stress and meat quality. Animal 2010, 4, 604–616. [Google Scholar] [CrossRef]
- Turner, S.P. Breeding against harmful social behaviours in pigs and chickens: State of the art and the way forward. Appl. Anim. Behav. Sci. 2011, 134, 1–9. [Google Scholar] [CrossRef]
- Peden, R.S.E.; Turner, S.P.; Boyle, L.A.; Camerlink, I. The translation of animal welfare research into practice: The case of mixing aggression between pigs. Appl. Anim. Behav. Sci. 2018, 204, 1–9. [Google Scholar] [CrossRef]
- Canario, L.; Bijma, P.; David, I.; Camerlink, I.; Martin, A.; Rauw, W.M.; Flatres-Grall, L.; van der Zande, L.; Turner, S.P.; Larzul, C.; et al. Prospects for the analysis and reduction of damaging behaviour in group-housed livestock, with application to pig breeding. Front. Genet. 2020, 11, 611073. [Google Scholar] [CrossRef]
- McBride, G.; James, W.; Hodgens, N. Social behaviour of domestic animals. IV. Growing pigs. Anim. Sci. 1964, 6, 129–139. [Google Scholar] [CrossRef]
- Lund, A.; Simonen, H.B. Aggression and stimulus-directed activities in two breeds of finishing pig. Pig J. 2000, 45, 123–130. [Google Scholar]
- Kim, D.-H.; Ha, D.-M.; Jeon, J.-H.; Song, J.-I.; Moon, S.-H. A study on the expression of harmful social behaviours in breeding pig. J. Anim. Environ. Sci. 2013, 19, 149–154. [Google Scholar] [CrossRef]
- Turner, S.P.; Roehe, R.; Mekkawy, W.; Farnworth, M.J.; Knap, P.W.; Lawrence, A.B. Bayesian analysis of genetic associations of skin lesions and behavioural traits to identify genetic components of individual aggressiveness in pigs. Behav. Genet. 2008, 38, 67–75. [Google Scholar] [CrossRef]
- Turner, S.P.; Roehe, R.; D’Eath, R.B.; Ison, S.H.; Farish, M.; Jack, M.C.; Lundeheim, N.; Rydhmer, L.; Lawrence, A.B. Genetic validation of post mixing skin injuries in pigs as an indicator of aggressiveness and the relationship with injuries under more stable social conditions. J. Anim. Sci. 2009, 87, 3076–3082. [Google Scholar] [CrossRef]
- Prunier, A.; Muller, N.; Courboulay, V.; Udin, L.; Larzul, C. Body lesions in entire male pigs during growth and on the carcass. Journées Rech. Porc. Fr. 2013, 45, 57–62. [Google Scholar]
- Desire, S.; Turner, S.P.; D’Eath, R.B.; Doeschl-Wilson, A.B.; Lewis, C.R.G.; Roehe, R. Genetic associations of short- and long-term aggressiveness identified by skin lesion with growth, feed efficiency, and carcass characteristics in growing pigs. J. Anim. Sci. 2015, 93, 3303–3312. [Google Scholar] [CrossRef]
- Chu, Q.; Liang, T.; Fu, L.; Li, H.; Zhou, B. Behavioural genetic differences between Chinese and European pigs. J. Genet. 2017, 96, 707–715. [Google Scholar] [CrossRef] [PubMed]
- Turner, S.P.; D’Eath, R.B.; Roehe, R.; Lawrence, A.B. Selection against aggressiveness in pigs at re-grouping: Practical application and implications for long-term behavioural patterns. Anim. Welf. 2010, 19, 123–132. [Google Scholar]
- Scheffler, K.; Stamer, E.; Traulsen, I.; Krieter, J. Estimation of genetic parameters for agonistic behaviour of pigs at different ages. J. Agric. Sci. 2016, 154, 732–741. [Google Scholar] [CrossRef]
- Turner, S.P.; Nevison, I.M.; Desire, S.; Camerlink, I.; Roehe, R.; Ison, S.H.; Farish, M.; Jack, M.C.; D’Eath, R.B. Aggressive behaviour at regrouping is a poor predictor of chronic aggression in stable social groups. Appl. Anim. Behav. Sci. 2017, 191, 98–106. [Google Scholar] [CrossRef]
- Canario, L.; Turner, S.P.; Roehe, R.; Lundeheim, N.; D’Eath, R.B.; Lawrence, A.B.; Knol, E.; Bergsma, R.; Rydhmer, L. Genetic associations between behavioral traits and direct-social effects of growth rate in pigs. J. Anim. Sci. 2012, 90, 4706–4715. [Google Scholar] [CrossRef] [PubMed]
- Camerlink, I.; Turner, S.P.; Farish, M.; Arnott, G. Aggressiveness as a component of fighting ability in pigs using a game-theoretical framework. Anim. Behav. 2015, 108, 183–191. [Google Scholar] [CrossRef]
- Hong, J.K.; Kim, K.H.; Song, N.R.; Choi, T.; Hwang, H.S.; Lee, J.K.; Eom, T.K.; Rhim, S.J. Social behavior and group growth of finishing pigs with divergent social breeding values. Rev. Colomb. Cienc. Pecu. 2018, 31, 267–275. [Google Scholar] [CrossRef]
- Nielsen, H.M.; Ask, B.; Madsen, P. Social genetic effects for growth in pigs differ between boars and gilts. Genet. Sel. Evol. 2018, 50, 4. [Google Scholar] [CrossRef]
- Angarita, B.K.; Cantet, R.J.C.; Wurtz, K.E.; O’Malley, C.I.; Siegford, J.M.; Ernst, C.W.; Turner, S.P.; Steibel, J.P. Estimation of indirect social genetic effects for skin lesion count in group-housed pigs by quantifying behavioral interactions. J. Anim. Sci. 2019, 97, 3658–3668. [Google Scholar] [CrossRef]
- Farine, D.R.; Whitehead, H. Constructing, conducting and interpreting animal social network analysis. J. Anim. Ecol. 2015, 84, 1144–1163. [Google Scholar] [CrossRef] [PubMed]
- Foister, S.; Doeschl-Wilson, A.; Roehe, R.; Arnott, G.; Boyle, L.; Turner, S. Social network properties predict chronic aggression in commercial pig systems. PLoS ONE 2018, 13, e0205122. [Google Scholar] [CrossRef]
- Larzul, C.; Hassenfratz, C.; Blanchet, B.; Prunier, A. Analyse des réseaux sociaux appliquée aux données DAC porcins. Journées Sci. Département Génétique Anim. 2018, S9, 1. [Google Scholar]
- Wurtz, K.; Camerlink, I.; D’Eath, R.B.; Fernandez, A.P.; Norton, T.; Steibel, J.; Siegford, J. Recording behaviour of indoor-housed farm animals automatically using machine vision technology: A systematic review. PLoS ONE 2019, 14, e0226669. [Google Scholar] [CrossRef]
- Benjamin, M.; Yik, S. Precision Livestock Farming in Swine Welfare: A Review for Swine Practitioners. Animals 2019, 9, 133. [Google Scholar] [CrossRef]
- Nasirahmadi, A.; Hensel, O.; Edwards, S.A.; Sturm, B. Automatic detection of mounting behaviours among pigs using image analysis. Comput. Electron. Agric. 2016, 124, 295–302. [Google Scholar] [CrossRef]
- Li, D.; Chen, Y.; Zhang, K.; Li, Z. Mounting Behaviour Recognition for Pigs Based on Deep Learning. Sensors 2019, 19, 4924. [Google Scholar] [CrossRef]
- Oczak, M.; Viazzi, S.; Ismayilova, G.; Sonoda, L.T.; Roulston, N.; Fels, M.; Bahr, C.; Hartung, J.; Guarino, M.; Berckmans, D.; et al. Classification of aggressive behaviour in pigs by activity index and multilayer feed forward neural network. Biosyst. Eng. 2014, 119, 89–97. [Google Scholar] [CrossRef]
- Kapun, A.; Adrion, F.; Tomalia, M.; Gallmann, E. Recording of behavior and activity patterns of growing-finishing pigs based on a UHF-RFID system. In Proceedings of the 13th Conference Construction, Engineering and Environment in Livestock Farming, Hohenheim, Germany, 18–20 September 2017. [Google Scholar]
- Boileau, A.; Farish, M.; Turner, S.P.; Camerlink, I. Infrared thermography of agonistic behaviour in pigs. Physiol. Behav. 2019, 210, 112637. [Google Scholar] [CrossRef]
- Rodriguez-Baena, D.S.; Gomez-Vela, F.A.; Garcia-Torres, M.; Divina, F.; Barranco, C.D.; Daz-Diaz, N.; Jimenez, M.; Montalvo, G. Identifying livestock behavior patterns based on accelerometer dataset. J. Comput. Sci. 2020, 41, 101076. [Google Scholar] [CrossRef]
- Falková, L.; Vrtková, I. Molecular Genetic Markers Associated with Boar Taint—Could Molecular Genetics Contribute to Its Reduction? Res. Pig Breed. 2019, 13, 7–13. [Google Scholar]
- Squires, J.; Bone, C.; Cameron, J. Pork Production with Entire Males: Directions for Control of Boar Taint. Animals 2020, 10, 1665. [Google Scholar] [CrossRef]
- Duarte, D.A.S.; Fortes, M.R.S.; Duarte, M.D.; Guimaraes, S.E.F.; Verardo, L.L.; Veroneze, R.; Ribeiro, A.M.F.; Lopes, P.S.; de Resende, M.D.V.; Silva, F.F.E. Genome-wide association studies, meta-analyses and derived gene network for meat quality and carcass traits in pigs. Anim. Prod. Sci. 2018, 58, 1100–1108. [Google Scholar] [CrossRef]
- Munoz, M.; Bozzi, R.; Garcia, F.; Nunez, Y.; Geraci, C.; Crovetti, A.; Garcia-Casco, J.; Alves, E.; Skrlep, M.; Charneca, R.; et al. Diversity across major and candidate genes in European local pig breeds. PLoS ONE 2018, 13, e0207475. [Google Scholar] [CrossRef]
- Pong-Wong, R.; Nagamine, Y.; D’Eath, R.B.; Turner, S.P.; Lawrence, A.B.; Haley, C.S. Dissecting the genetic variation of aggressive behaviour in pigs using dense SNP data. In Proceedings of the XV Reunion Nacional de Mejora Genetica Animal, Vigo, Spain, 16–18 June 2010. [Google Scholar]
- Wurtz, K.E.; Siegford, J.M.; Ernst, C.W.; Raney, N.E.; Bates, R.O.; Steibel, J.P. Genome-wide association analyses of lesion counts in group-housed pigs. Anim. Genet. 2018, 49, 628–631. [Google Scholar] [CrossRef] [PubMed]
- Murani, E.; Ponsuksili, S.; D’Eath, R.B.; Turner, S.P.; Kurt, E.; Evans, G.; Thoelking, L.; Klont, R.; Foury, A.; Mormede, P.; et al. Association of HPA axis-related genetic variation with stress reactivity and aggressive behaviour in pigs. BMC Genet. 2010, 11, 74. [Google Scholar] [CrossRef]
- Van den Broeke, A.; Aluwe, M.; Janssens, S.; Wauters, J.; Vanhaecke, L.; Buys, N.; Millet, S.; Tuyttens, F.A.M. The effect of the MC4R gene on boar taint compounds, sexual maturity and behaviour in growing-finishing boars and gilts. Animal 2015, 9, 1688–1697. [Google Scholar] [CrossRef] [PubMed]
- Terenina, E.; Bazovkina, D.; Rousseau, S.; Salin, F.; D’Eath, R.; Turner, S.; Kulikov, A.; Mormede, P. Association entre Polymorphismes de Gènes Candidats et Comportements Agressifs chez le Porc. Journées Rech. Porc. Fr. 2012, 44, 45–46. [Google Scholar]
- Chen, R.; Chu, Q.; Shen, C.; Tong, X.; Gao, S.; Liu, X.; Zhou, B.; Schinckel, A.P. Identification of Single Nucleotide Polymorphisms in Porcine MAOA Gene Associated with Aggressive Behavior of Weaned Pigs after Group Mixing. Animals 2019, 9, 952. [Google Scholar] [CrossRef] [PubMed]
| Breed/Population | Heritability | Genetic Correlation | Reference | |
|---|---|---|---|---|
| Androstenone | Skatole | |||
| French Large White × Landrace | 0.49; 0.55 | [28] | ||
| French Pietrain | 0.63 | 0.37 | 0.61 | [22] |
| French Pietrain × Large White | 0.70 | 0.24 | 0.36 | |
| French Pietrain | 0.38 * | 0.14 * | [15] | |
| French Pietrain × Large White | 0.54 * | 0.27 * | ||
| French Pietrain | 0.57 | [23] | ||
| French Pietrain × Large White | 0.71 | |||
| Italian Large White (160 days) | 0.39 | 0.60 | 0.30 | [29] |
| Italian Large White (220 days) | 0.58 | 0.60 | 0.56 | |
| German Landrace | 0.56 | [20] | ||
| German Landrace | 0.50 | 0.52 | 0.29 | [3] |
| German Large White | 0.39 | 0.32 | 0.41 | |
| Norwegian Landrace | 0.54 | 0.55 | 0.36 | [5] |
| Norwegian Duroc | 0.56 | 0.23 | 0.62 | |
| Norwegian Landrace | 0.49 | 0.41 | 0.32 | [7] |
| Norwegian Duroc | 0.67 | 0.37 | 0.33 | |
| Norwegian Landrace | 0.45 | 0.53 | 0.32 | [8] |
| Norwegian Duroc | 0.50 | 0.33 | 0.42 | |
| Dutch synthetic | 0.75 | 0.53 | 0.22 | From [2] |
| Dutch Duroc | 0.46 | 0.26 | 0.24 | [26] |
| Dutch Duroc | 0.63 | [27] | ||
| Multibreed | 0.69 | 0.53 | From [2] | |
| Multibreed | 0.64 | 0.26 | From [2] | |
| Multibreed | 0.54 | 0.41 | 0.37 | [10] |
| Dutch Pietrain | 0.72 | 0.69 | [14] | |
| Dutch Landrace and Yorkshire | 0.73 | 0.82 | ||
| Swiss Large White | 0.45 | 0.50 | 0.11 | From [2] |
| Danish Landrace | 0.25; 0.54 | [30] | ||
| Danish Landrace | 0.47–0.49 | 0.54–0.56 | From [2] | |
| Danish Landrace | 0.59 | 0.33 | 0.35 | [31] |
| Danish Landrace | 0.27–0.31 | 0.06–0.11 | [25] | |
| Danish Landrace | 0.52 | 0.28 | 0.38 | From [2] |
| Landrace | 0.27 | From [2] | ||
| Large White | 0.19 | |||
| Duroc | 0.19 | |||
| Hampshire | 0.21 | |||
| Pietrain | 0.61 | 0.50 | From [2] | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Larzul, C. How to Improve Meat Quality and Welfare in Entire Male Pigs by Genetics. Animals 2021, 11, 699. https://doi.org/10.3390/ani11030699
Larzul C. How to Improve Meat Quality and Welfare in Entire Male Pigs by Genetics. Animals. 2021; 11(3):699. https://doi.org/10.3390/ani11030699
Chicago/Turabian StyleLarzul, Catherine. 2021. "How to Improve Meat Quality and Welfare in Entire Male Pigs by Genetics" Animals 11, no. 3: 699. https://doi.org/10.3390/ani11030699
APA StyleLarzul, C. (2021). How to Improve Meat Quality and Welfare in Entire Male Pigs by Genetics. Animals, 11(3), 699. https://doi.org/10.3390/ani11030699






