Early Diagnosis of Canine Hip Laxity: Correlation between Clinical Orthopedic Examinations and the FCI Scoring Method in a Closed Cohort of Rottweilers
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Approval and Consent
2.2. Anesthesia
2.3. Orthopedic Hip Examinations under General Anesthesia
2.3.1. Barlow Maneuver
2.3.2. Ortolani Maneuver
2.3.3. Bardens Maneuver
2.4. Goniometric Assessments
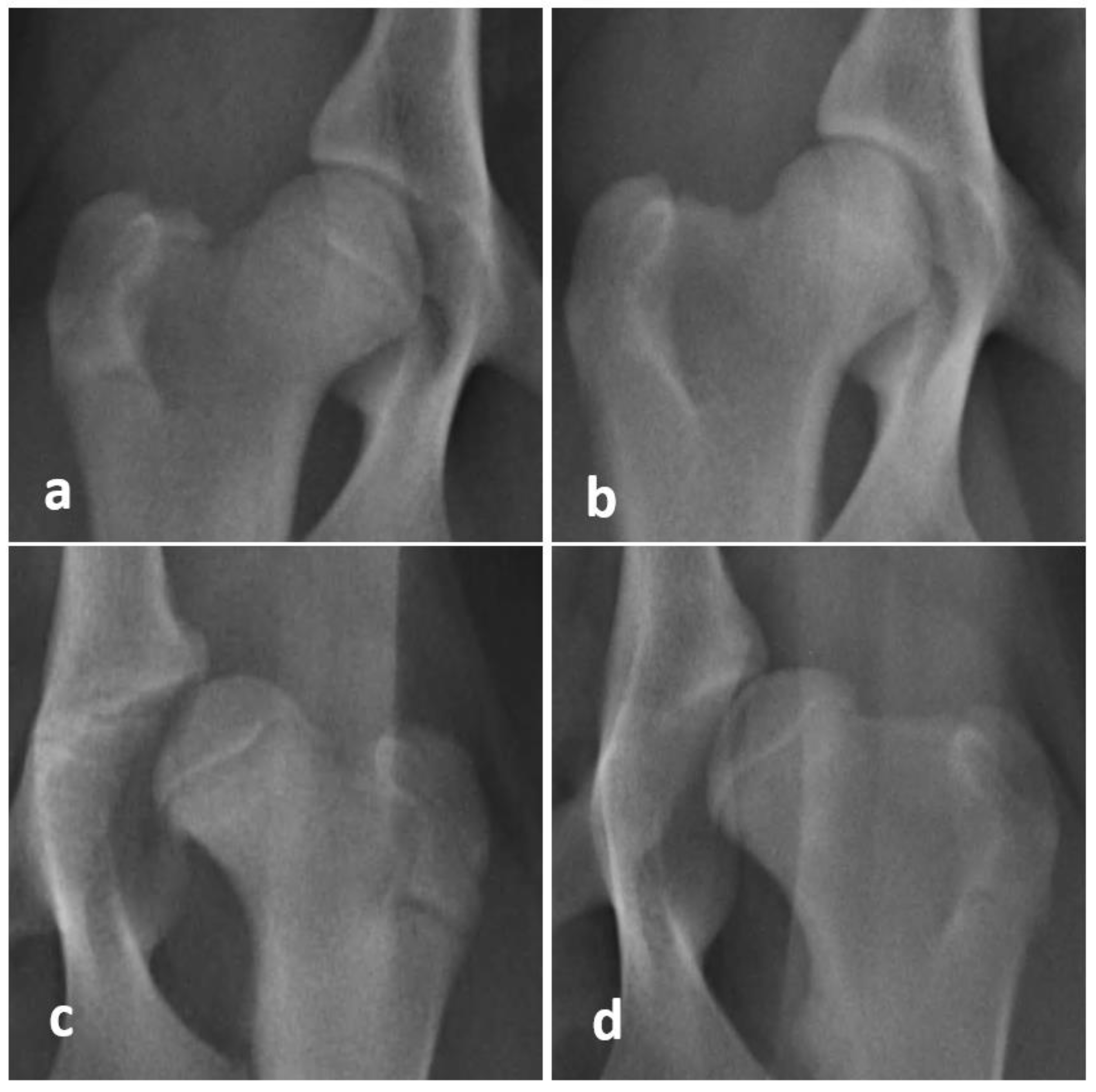
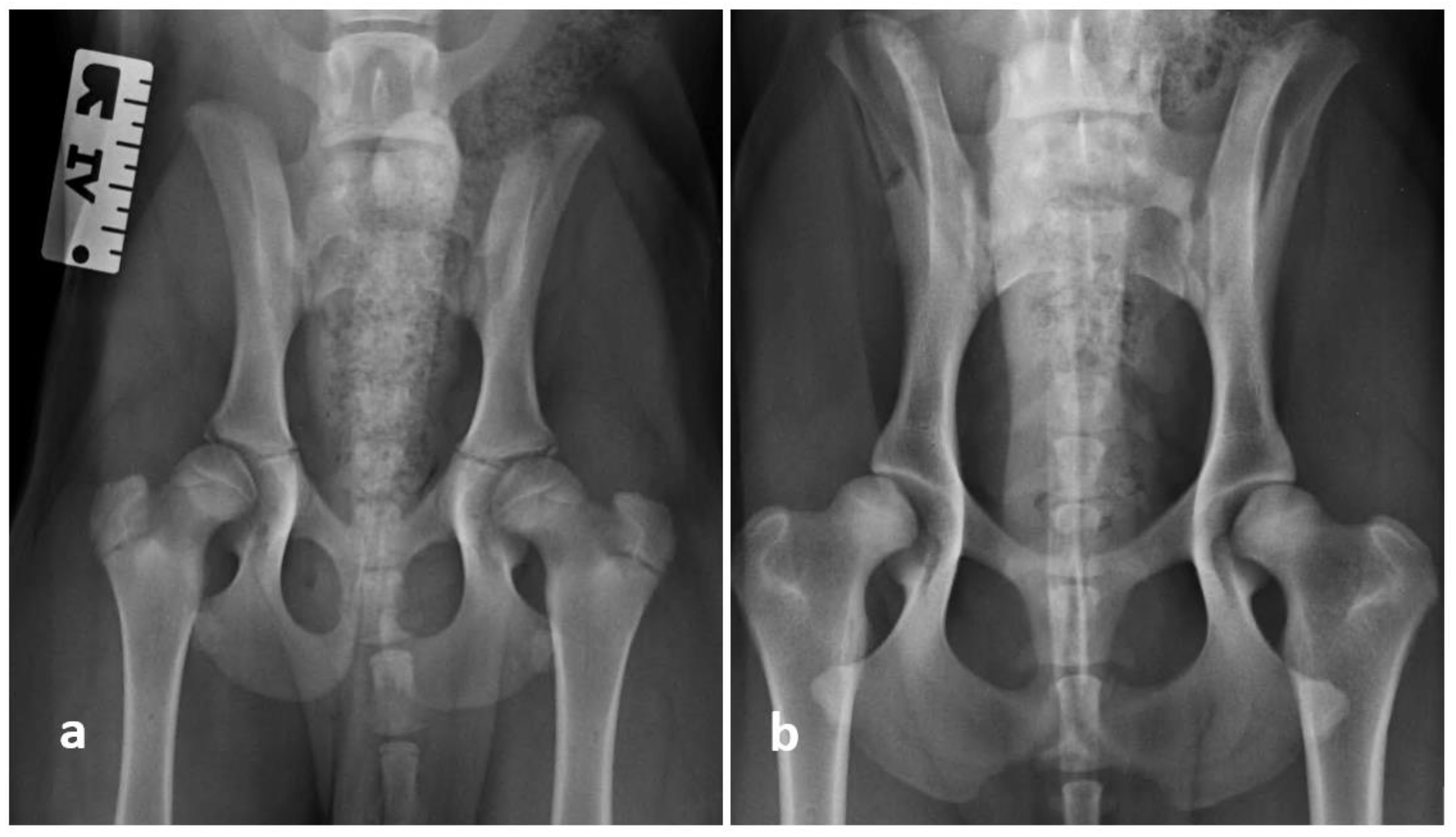
2.5. Radiographic Evaluation
2.6. Statistical Analysis
3. Results
3.1. Ortolani Findings
3.2. Barlow Findings
3.3. Bardens Findings
3.4. Subluxation Angle
3.5. Reduction Angle
3.6. FCI Scoring
3.7. Correlation between Ortolani Findings and RA/SA
3.8. Correlation between the FCI Score and RA/SA
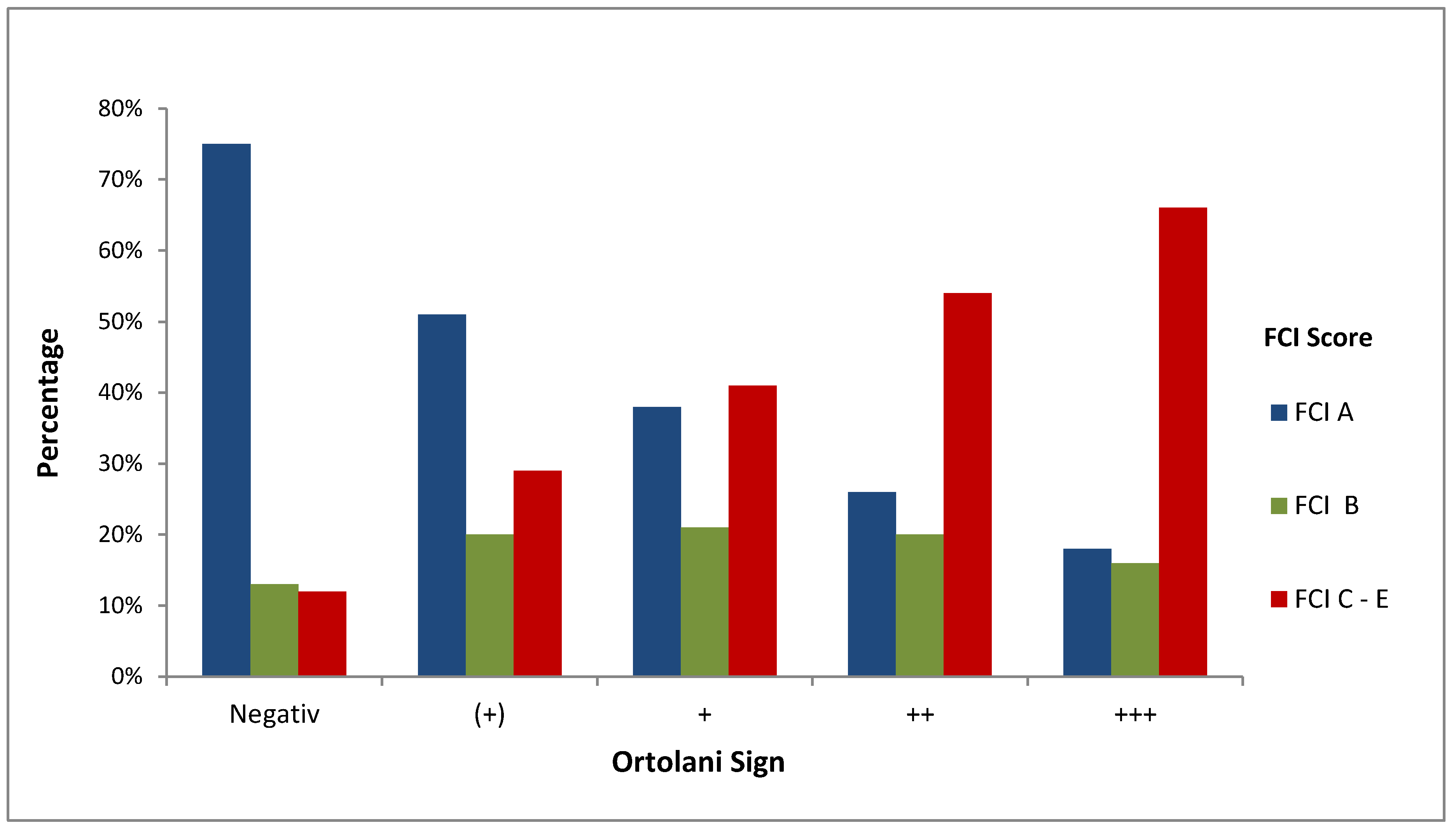
3.9. Correlation between the FCI Score and Ortolani Findings in Dorsal Recumbency
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Henrigson, B.; Norberg, I.; Olssons, S.-E. On the Etiology and Pathogenesis of Hip Dysplasia: A Comparative Review. J. Small Anim. Pract. 1966, 7, 673–688. [Google Scholar] [CrossRef] [PubMed]
- Lust, G. An overview of the pathogenesis of canine hip dysplasia. J. Am. Veter Med. Assoc. 1997, 210, 1443–1445. [Google Scholar]
- Lafond, E.; Breur, G.J.; Austin, C.C. Breed Susceptibility for Developmental Orthopedic Diseases in Dogs. J. Am. Anim. Hosp. Assoc. 2002, 38, 467–477. [Google Scholar] [CrossRef] [PubMed]
- Kealy, R.D.; Lawler, D.F.; Ballam, J.M.; Mantz, S.L.; Biery, D.N.; Greeley, E.H.; Lust, G.; Segre, M.; Smith, G.K.; Stowe, H.D. Effects of diet restriction on life span and age-related changes in dogs. J. Am. Veter Med. Assoc. 2002, 220, 1315–1320. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.K.; Paster, E.R.; Powers, M.Y.; Lawler, D.F.; Biery, D.N.; Shofer, F.S.; McKelvie, P.J.; Kealy, R.D. Lifelong diet restriction and radiographic evidence of osteoarthritis of the hip joint in dogs. J. Am. Veter Med. Assoc. 2006, 229, 690–693. [Google Scholar] [CrossRef]
- Riser, W.H. The Dysplastic Hip Joint: Its Radiographic and Histologic Development1. Veter Radiol. 1973, 14, 35–50. [Google Scholar] [CrossRef]
- Smith, G.K.; Gregor, T.P.; Rhodes, W.H.; Biery, D.N. Coxofemoral joint laxity from distraction radiography and its contemporaneous and prospective correlation with laxity, subjective score, and evidence of degenerative joint disease from conventional hip-extended radiography in dogs. Am. J. Veter Res. 1993, 54, 1021–1042. [Google Scholar]
- Smith, G.K.; A Popovitch, C.; Gregor, T.P.; Shofer, F.S. Evaluation of risk factors for degenerative joint disease associated with hip dysplasia in dogs. J. Am. Veter Med. Assoc. 1995, 206, 642–647. [Google Scholar]
- Runge, J.J.; Kelly, S.P.; Gregor, T.P.; Kotwal, S.; Smith, G. Distraction index as a risk factor for osteoarthritis associated with hip dysplasia in four large dog breeds *. J. Small Anim. Pract. 2010, 51, 264–269. [Google Scholar] [CrossRef]
- Köppel, E. Zur Entwicklung der Articulatiocoxae und radiologischen Frühdiagnostik der Hüftgelenkdysplasie des Hundes. In Eine Morphologische Studie; Enke Verlag: Stuttgart, Germany, 1991; ISBN 978-3432993416. [Google Scholar]
- Barlow, T.G. Early diagnosis and treatment of congenital dislocation of the hip. J. Bone Jt. Surgery. Br. Vol. 1962, 44, 292–301. [Google Scholar] [CrossRef]
- Chalman, J.A.; Butler, H.C. Coxofemoral joint laxity and the Ortolani sign. J. Am. Anim. Hosp. Assoc. 1985, 21, 671–676. [Google Scholar]
- Bardens, J.W.; Hardwick, H. New observations on the diagnosis and cause of hip dysplasia. Veter Med. Small Anim. Clin. VM SAC 1968, 63, 238–245. [Google Scholar]
- Linnmann, S.M. Die Hueftgelenksdysplasie des Hundes, 2nd ed.; Veterinaerspiegel: Schaefermueller Publishing: Berlin, Germany, 2012; ISBN 9783865420138. [Google Scholar]
- Ginja, M.M.; Gonzalo-Orden, J.M.; Melo-Pinto, P.; Bulas-Cruz, J.; Orden, M.A.; San Roman, F.; Llorens-Pena, M.P.; Ferreira, A.J. Early hip laxity examination in predicting moderate and severe hip dysplasia in Estrela mountain dog. J. Small Anim. Pract. 2008, 49, 641–646. [Google Scholar] [CrossRef] [PubMed]
- Schulz, K.S.; Dejardin, L.M. Surgical treatment of canine hip dysplasia. In Textbook of Small Animal Surgery, 3rd ed.; Slatter, D.H., Ed.; Saunders: Philadelphia, PA, USA, 2003; pp. 2029–2059. ISBN 0-7216-8607-9. [Google Scholar]
- Vezzoni, A. Is early evaluation reliable for CHD diagnosis? In Proceedings of the12th ESVOT Congress, Munich, Germany, 10–12 September 2004; pp. 145–151. [Google Scholar]
- Vezzoni, A.; Boiocchi, S.; Vanelli, A.B.; Bronzo, V.; Vezzoni, L.; Bronzo, V. Double pelvic osteotomy for the treatment of hip dysplasia in young dogs. Veter Comp. Orthop. Traumatol. 2010, 23, 444–452. [Google Scholar] [CrossRef] [PubMed]
- Gatineau, M.; Dupuis, J.; Beauregard, G.; Charette, B.; Breton, L.; Beauchamp, G.; D’Anjou, M.-A. Palpation and Dorsal Acetabular Rim Radiographic Projection for Early Detection of Canine Hip Dysplasia: A Prospective Study. Veter Surg. 2011, 41, 42–53. [Google Scholar] [CrossRef]
- DeCamp, C.E.; Johnston, S.A.; Dejardin, L.M.; Schaefer, S.L. Fractures and orthopedic conditions of the himdlimb. In Brinker, Piermattei and Flo’s Handbook of Small Animal Orthopedics and Fracture Repair, 5th ed.; Elsevier Health Sciences: St. Louis, MO, USA, 2016; pp. 468–517. ISBN 978-1-4377-2364-9. [Google Scholar]
- Vezzoni, A.; Dravelli, G.; Corbari, A.; De Lorenzi, M.; Cirla, A.; Tranquilo, V. Early diagnosis of canine hip dysplasia. Eur. J. Comp. Anim. Prac. 2005, 15, 173–183. [Google Scholar]
- Slocum, B.; Devine, T.M. Dorsal acetabular rim radiographic view for evaluation of the canine hip. J. Am. Anim. Hosp. Assoc. 1990, 26, 289–296. [Google Scholar]
- Federation Cynologique Internationale. Available online: http://www.fci.be/en/Hip-and-Elbow-Dysplasia-162.html (accessed on 16 November 2020).
- Flückiger, M. Scoring radiographs for canine hip dysplasia-The big three organisations in the world. Eur. J. Comp. Anim. Prac. 2007, 17, 135–140. [Google Scholar]
- Smith, G.K.; Biery, D.N.; Gregor, T.P. New concepts of coxofemoral joint stability and the development of a clinical stress-radiographic method for quantitating hip joint laxity in the dog. J. Am. Veter Med. Assoc. 1990, 196, 59–70. [Google Scholar]
- A Puerto, D.; Smith, G.K.; Gregor, T.P.; Lafond, E.; Conzemius, M.G.; Cabell, L.W.; McKelvie, P.J. Relationships between results of the Ortolani method of hip joint palpation and distraction index, Norberg angle, and hip score in dogs. J. Am. Veter Med. Assoc. 1999, 214, 497–501. [Google Scholar]
- Lust, G.; Williams, A.J.; Burton-Wurster, N.; Pijanowski, G.J.; A Beck, K.; Rubin, G.; Smith, G.K. Joint laxity and its association with hip dysplasia in Labrador retrievers. Am. J. Veter Res. 1993, 54, 1990–1999. [Google Scholar]
- Slocum, B.; Slocum, T.D. Definitions of hip terms. In Current Techniques in Small Animal Surgery; Bojrab, M.J., Ellison, G.W., Slocum, B., Eds.; Williams and Wilkins: Baltimore, MD, USA, 1998; pp. 1151–1154. ISBN 9780683008906. [Google Scholar]
- Genevois, J.-P.; Viguier, E.; Carozzo, C.; Livet, V.; Baldinger, A.; Cachon, T.; Taroni, M. Evolution of Radiographic Parameters of Canine Passive Hip Laxity at 4, 6 and 12 months: A Study of 306 Dogs. Veter Comp. Orthop. Traumatol. 2018, 31, 321–326. [Google Scholar] [CrossRef] [PubMed]
- Schachner, E.R.; Lopez, M.J. Diagnosis, prevention, and management of canine hip dysplasia: A review. Vet. Med. Res. Rep. 2015, 6, 181. [Google Scholar]
- Adams, W.M.; Dueland, R.T.; Meinen, J.; O’Brien, R.T.; Giuliano, E.; Nordheim, E.V. Early detection of canine hip dysplasia: Comparison of two palpation and five radiographic methods. J. Am. Anim. Hosp. Assoc. 1998, 34, 339–347. [Google Scholar] [CrossRef]
- Corfield, G.; Read, R.; Eastley, K.; Richardson, J.; Robertson, I.D.; Day, R.E. Assessment of the hip reduction angle for predicting osteoarthritis of the hip in the Labrador Retriever. Aust. Veter J. 2007, 85, 212–216. [Google Scholar] [CrossRef]
- Taroni, M.; Viguier, E.; Pillard, P.; Livet, V.; Cachon, T.; Carozzo, C.; Genevois, J.-P. Comparison of Early Measurements of the Distraction Index, Norberg Angle on Distracted View and the Official Radiographic Evaluation of the Hips of 215 Dogs from Two Guide Dog Training Schools. Veter Comp. Orthop. Traumatol. 2018, 31, 445–451. [Google Scholar] [CrossRef]
- Klever, J.; Brühschwein, A.; Wagner, S.; Reese, S.; Meyer-Lindenberg, A. Comparison of Reliability of Norberg Angle and Distraction Index as Measurements for Hip Laxity in Dogs. Veter Comp. Orthop. Traumatol. 2020, 33, 274–278. [Google Scholar] [CrossRef]
- Gulanber, E.G.; Gulanber, N.G.; Albayrak, N.R.; Ozer, K.; Aktas, M.; Aksoy, O. Use of distraction radiography in canine hip dysplasia: Comparison of early and late results with two different distractors. Med. Weter. 2006, 62, 1245–1248. [Google Scholar]
- Adams, W.M.; Dueland, R.T.; Daniels, R.; Fialkowski, J.P.; Nordheim, E.V. Comparison of two palpation, four radiographic and three ultrasound methods for early detection of mild to moderate canine hip dysplasia. Veter Radiol. Ultrasound 2000, 41, 484–490. [Google Scholar] [CrossRef] [PubMed]
- Online Mendelian Inheritance in Animals, OMIA. Sydney School of Veterinary Science. Available online: https://omia.org/OMIA000473/9615/ (accessed on 29 January 2021).
- Coopman, F.; Verhoeven, G.; Saunders, J.; Duchateau, L.; Van Bree, H. Prevalence of hip dysplasia, elbow dysplasia and humeral head osteochondrosis in dog breeds in Belgium. Veter Rec. 2008, 163, 654–658. [Google Scholar] [CrossRef] [PubMed]
- Witsberger, T.H.; Villamil, J.A.; Schultz, L.G.; Hahn, A.W.; Cook, J.L. Prevalence of and risk factors for hip dysplasia and cranial cruciate ligament deficiency in dogs. J. Am. Veter Med. Assoc. 2008, 232, 1818–1824. [Google Scholar] [CrossRef] [PubMed]
- Kapatkin, A.S.; Gregor, T.P.; Hearon, K.; Richardson, R.W.; McKelvie, P.J.; Fordyce, H.H.; Smith, G. Comparison of two radiographic techniques for evaluation of hip joint laxity in 10 breeds of dogs. J. Am. Veter Med. Assoc. 2004, 224, 542–546. [Google Scholar] [CrossRef] [PubMed]
- Ohlerth, S.; Geiser, B.; Flückiger, M.; Geissbühler, U. Prevalence of Canine Hip Dysplasia in Switzerland Between 1995 and 2016—A Retrospective Study in 5 Common Large Breeds. Front. Veter Sci. 2019, 6, 378. [Google Scholar] [CrossRef] [PubMed]

| Examination Date 1 | Recumbency | N (Dogs/Hips) | Frequency (Percentage) 2 | ||||
|---|---|---|---|---|---|---|---|
| − | (+) | + | ++ | +++ | |||
| M1 | Dorsal | 28/56 | 10 (17.9%) | 24 (42.9%) | 17 (30.4%) | 5 (8.9%) | 0 (0%) |
| M1 | Lateral | 28/56 | 44 (78.6%) | 3 (5.4%) | 7 (12.5%) | 1 (1.8%) | 1 (1.8%) |
| M2 | Dorsal | 30/60 | 27 (45.0%) | 12 (20.0%) | 15 (25.0%) | 4 (6.7%) | 2 (3.3%) |
| M2 | Lateral | 30/60 | 53 (88.3%) | 3 (5.0%) | 2 (3.3%) | 2 (3.3%) | 0 (0%) |
| M3 | Dorsal | 29/58 | 37 (63.8%) | 8 (13.8%) | 7 (12.1%) | 5 (8.6%) | 1 (1.7%) |
| M3 | Lateral | 29/58 | 51 (87.9%) | 3 (5.2%) | 3 (5.2%) | 0 (0%) | 1 (1.7%) |
| Examination Date 1 | Recumbency | N (Dogs/Hips) | Frequency (Percentage) 2 | ||
|---|---|---|---|---|---|
| − | + | Not definable | |||
| M1 | Dorsal | 28/56 | 10 (17.9%) | 23 (41.1%) | 23 (41.1%) |
| M1 | Lateral | 28/56 | 44 (78.6%) | 9 (16.1%) | 3 (5.4%) |
| M2 | Dorsal | 30/60 | 27 (45.0%) | 21 (35.0%) | 12 (20.0%) |
| M2 | Lateral | 30/60 | 53 (88.3%) | 5 (8.3%) | 2 (3.3%) |
| M3 | Dorsal | 29/58 | 38 (65.5%) | 12 (20.7%) | 8 (13.8%) |
| M3 | Lateral | 29/58 | 51 (87.9%) | 4 (6.9%) | 3 (5.2%) |
| Examination Date 1 | Recumbency | N (Dogs/Hips) | Frequency (Percentage) 2 | |
|---|---|---|---|---|
| − | + | |||
| M1 | Lateral | 28/56 | 42 (75.0%) | 14 (25.0%) |
| M2 | Lateral | 30/60 | 53 (88.3%) | 7 (11.7%) |
| M3 | Lateral | 29/58 | 55 (94.8%) | 3 (5.2%) |
| Examination Date 1 | Recumbency | r | p-Value |
|---|---|---|---|
| M1 | Dorsal | 0.564 | 0.001 |
| M1 | Lateral | 0.473 | 0.001 |
| M2 | Dorsal | 0.327 | 0.063 |
| M2 | Lateral | 0.495 | 0.003 |
| M3 | Dorsal | 0.392 | 0.087 |
| M3 | Lateral | 0.657 | 0.001 |
| Examination Date 1 | r | p-Value |
|---|---|---|
| M1 | 0.214 | 0.120 |
| M2 | 0.393 | 0.002 |
| M3 | 0.527 | 0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vidoni, B.; Bauer, V.; Bockstahler, B.; Gumpenberger, M.; Tichy, A.; Aghapour, M. Early Diagnosis of Canine Hip Laxity: Correlation between Clinical Orthopedic Examinations and the FCI Scoring Method in a Closed Cohort of Rottweilers. Animals 2021, 11, 416. https://doi.org/10.3390/ani11020416
Vidoni B, Bauer V, Bockstahler B, Gumpenberger M, Tichy A, Aghapour M. Early Diagnosis of Canine Hip Laxity: Correlation between Clinical Orthopedic Examinations and the FCI Scoring Method in a Closed Cohort of Rottweilers. Animals. 2021; 11(2):416. https://doi.org/10.3390/ani11020416
Chicago/Turabian StyleVidoni, Britta, Veronika Bauer, Barbara Bockstahler, Michaela Gumpenberger, Alexander Tichy, and Masoud Aghapour. 2021. "Early Diagnosis of Canine Hip Laxity: Correlation between Clinical Orthopedic Examinations and the FCI Scoring Method in a Closed Cohort of Rottweilers" Animals 11, no. 2: 416. https://doi.org/10.3390/ani11020416
APA StyleVidoni, B., Bauer, V., Bockstahler, B., Gumpenberger, M., Tichy, A., & Aghapour, M. (2021). Early Diagnosis of Canine Hip Laxity: Correlation between Clinical Orthopedic Examinations and the FCI Scoring Method in a Closed Cohort of Rottweilers. Animals, 11(2), 416. https://doi.org/10.3390/ani11020416









