Egg Production Systems, Open Space Allowance and Their Effects on Physical Parameters and Fatty Acid Profile in Commercial Eggs
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Eggs and Production Systems
2.2. Physical Parameters
2.3. Total Lipids and Fatty Acid Profile
2.4. Statistical Analysis
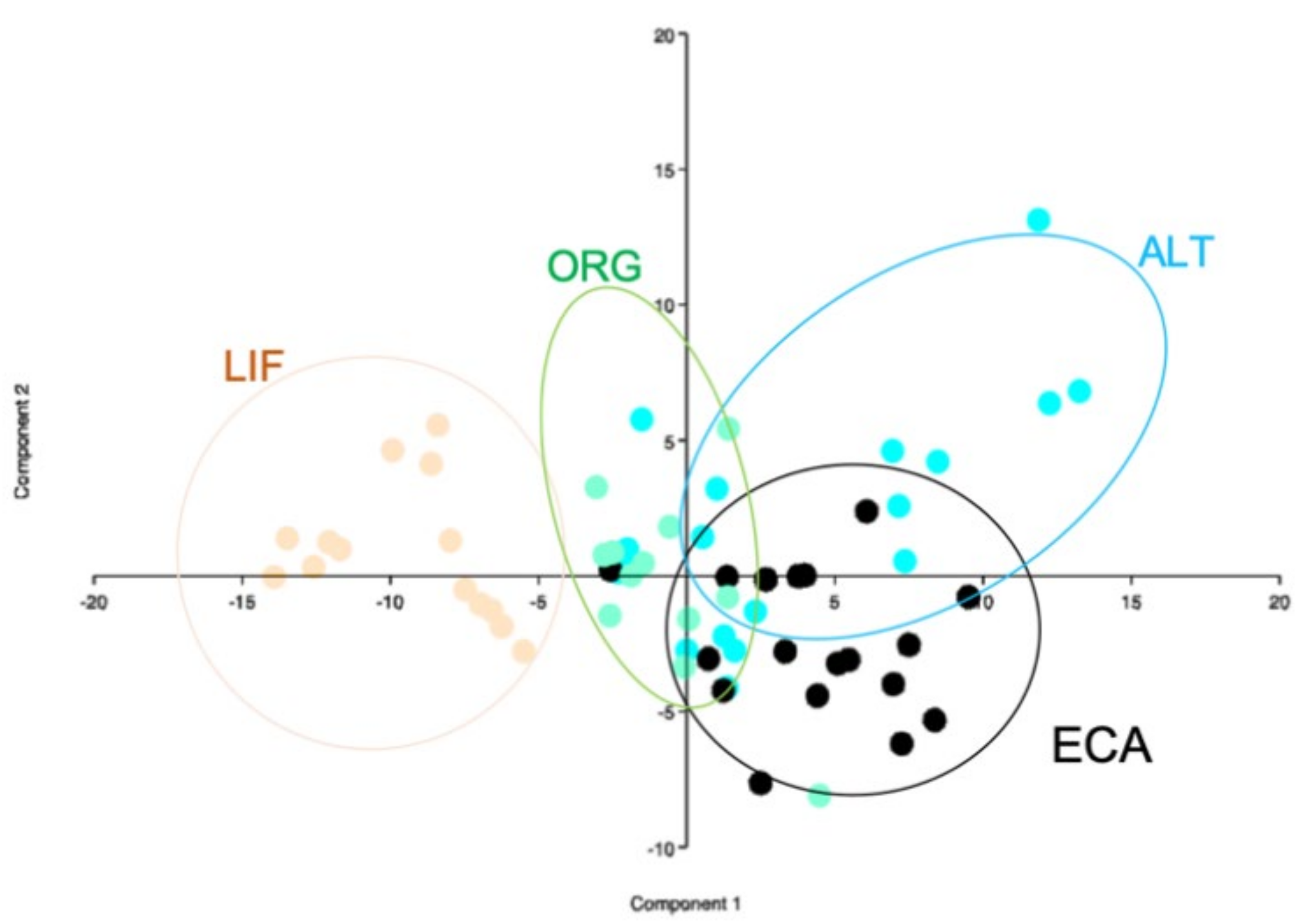
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Grunert, K.G. Food quality and safety: Consumer perception and demand. Eur. Rev. Agric. Econ. 2005, 32, 369–391. [Google Scholar] [CrossRef]
- Brunsø, K.; Fjord, T.A.; Grunert, K.G. Consumers’ Food Choice and Quality Perception; The Aarhus School of Business: Aarhus, Denmark, 2002; pp. 1–60. [Google Scholar]
- Mine, Y.; Kovacs-Nolan, J. Biologically active hen egg components in human health and disease. J. Poult. Sci. 2004, 41, 1–29. [Google Scholar] [CrossRef]
- Nardone, A.; Valfrè, F. Effects of changing production methods on quality of meat, milk and eggs. Livest. Prod. Sci. 1999, 59, 165–182. [Google Scholar] [CrossRef]
- Aumaıtre, A. Quality and safety of animal products. Livest. Prod. Sci. 1999, 59, 113–124. [Google Scholar] [CrossRef]
- Spedding, C.R.W. Sustainability in animal production systems. Anim. Sci. 1995, 61, 1–8. [Google Scholar] [CrossRef]
- Surai, P.F.; Sparks, N.H.C. Designer eggs: From improvement of egg composition to functional food. Trends Food Sci. Technol. 2001, 12, 7–16. [Google Scholar] [CrossRef]
- Noble, R.C.; Speake, B.K.; McCartney, R.; Foggin, C.M.; Deeming, D.C. Yolk lipids and their fatty acids in the wild and captive ostrich (Struthio camelus). Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 1996, 113, 753–756. [Google Scholar] [CrossRef]
- Speake, B.K.; Murray, A.M.B.; Noble, R.C. Transport and transformations of yolk lipids during development of the avian embryo. Prog. Lipid Res. 1998, 37, 1–32. [Google Scholar] [CrossRef]
- Uauy, R.; Castillo, C. Lipid requirements of infants: Implications for nutrient composition of fortified complementary foods. J. Nutr. 2003, 133, 2962S–2972S. [Google Scholar] [CrossRef]
- Drakley, C.; Elson, H.A.; Walker, A.W. Production efficiency of laying hens at four stocking densities in furnished cages of two heights. In Proceedings of the 11th European Poultry Conference, Bremen, Germany, 6–10 September 2002; p. 33. [Google Scholar]
- Rushen, J.; de Passillé, A.M.B. The scientific assessment of the impact of housing on animal welfare: A critical review. Can. J. Anim. Sci. 1992, 72, 721–743. [Google Scholar] [CrossRef]
- Christie, W.W. Lipid Analysis; Pergamon Press: Oxford, UK, 1982; Volume 207. [Google Scholar]
- Folch, J.; Lees, M.; Stanley, G.H.S. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [CrossRef]
- Hamilton, R.J.; Hamilton, S.; Harwood, J. Lipid Analysis: A Practical Approach; IRL Press: Oxford, UK, 1992; ISBN 0199630992. [Google Scholar]
- Christie, W.W.; Noble, R.C.; Moore, J.H. Determination of lipid classes by a gas-chromatographic procedure. Analyst 1970, 95, 940–944. [Google Scholar] [CrossRef]
- IBM SPSS Statistics software: SPSS Statistics, V26; IBMS Statistics: Armonk, NY, USA, 2019.
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological statistics software package for education and data analysis. Palaeontol. Electron. 2001, 4, 9. [Google Scholar]
- Taylor, A.A.; Hurnik, J.F. The long-term productivity of hens housed in battery cages and an aviary. Poult. Sci. 1996, 75, 47–51. [Google Scholar] [CrossRef] [PubMed]
- Rakonjac, S.; Bogosavljević-Bošković, S.; Pavlovski, Z.; Škrbić, Z.; Dosković, V.; Petrović, M.D.; Petričević, V. Laying hen rearing systems: A review of major production results and egg quality traits. Worlds Poult. Sci. J. 2014, 70, 93–104. [Google Scholar] [CrossRef]
- Minelli, G.; Sirri, F.; Folegatti, E.; Meluzzi, A.; Franchini, A. Egg quality traits of laying hens reared in organic and conventional systems. Ital. J. Anim. Sci. 2007, 6, 728–730. [Google Scholar] [CrossRef]
- Campo, J.L.; Cabezas, R.; Torres, O.; Briones, I.G.; Alonso, C. Egg quality and welfare of white-, tinted-, and brown-shell egg layers in three different non-cage housing systems. Arch. Geflugelkd. 2013, 77, 179–188. [Google Scholar]
- Stadelman, W.J.; Pratt, D.E. Factors influencing composition of the hen’s egg. Worlds Poult. Sci. J. 1989, 45, 247–266. [Google Scholar] [CrossRef]
- Milinsk, M.C.; Murakami, A.E.; Gomes, S.T.M.; Matsushita, M.; De Souza, N.E. Fatty acid profile of egg yolk lipids from hens fed diets rich in n-3 fatty acids. Food Chem. 2003, 83, 287–292. [Google Scholar] [CrossRef]
- Cobos, A.; de la Hoz, L.; Cambero, M.I.; Ordóñez, J.A. Dietary modification and hen strain dependence of egg yolk lipids. Food Res. Int. 1995, 28, 71–76. [Google Scholar] [CrossRef]
- Marelli, S.P.; Zaniboni, L.; Madeddu, M.; Sayed, A.A.; Strillacci, M.G.; Mangiagalli, M.G.; Cerolini, S. Physical Parameters and Fatty Acids Profiles in Milanino, Mericanel Della Brianza, Valdarnese Bianca and Commercial Hybrids (Gallus Gallus Domesticus) Table Eggs. Animals 2020, 10, 1533. [Google Scholar] [CrossRef] [PubMed]
- Huneau-Salaün, A.; Michel, V.; Huonnic, D.; Balaine, L.; Le Bouquin, S. Quality of eggs from Polish native Greenleg Partridge chicken-hens maintained in organic vs. backyard production system. Br. Poult. Sci. 2010, 51, 163–169. [Google Scholar] [CrossRef] [PubMed]
- Samman, S.; Kung, F.P.; Carter, L.M.; Foster, M.J.; Ahmad, Z.I.; Phuyal, J.L.; Petocz, P. Fatty acid composition of certified organic, conventional and omega-3 eggs. Food Chem. 2009, 116, 911–914. [Google Scholar] [CrossRef]
- Jiang, Z.; Ahn, D.U.; Sim, J.S. Effects of feeding flax and two types of sunflower seeds on fatty acid compositions of yolk lipid classes. Poult. Sci. 1991, 70, 2467–2475. [Google Scholar] [CrossRef]
- Watkins, B.A. Biochemical and physiological aspects of polyunsaturates. Poult. Avian Biol. Rev. 1995, 6, 1–18. [Google Scholar]
- Bergami, W.; Giavarini, I.; Minoccheri, F.; Negrini, F. Egg composition and breeding technology. Fat contents of the eggs laid by chickens bred in battery or free range. Avicoltura 1978, 47, 35–38. [Google Scholar]
- Simčič, M.; Stibilj, V.; Holcman, A. Fatty acid composition of eggs produced by the Slovenian autochthonous Styrian hen. Food Chem. 2011, 125, 873–877. [Google Scholar] [CrossRef]
- Anderson, K.E. Comparison of fatty acid, cholesterol, and vitamin A and E composition in eggs from hens housed in conventional cage and range production facilities. Poult. Sci. 2011, 90, 1600–1608. [Google Scholar] [CrossRef]
- Karsten, H.D.; Patterson, P.H.; Stout, R.; Crews, G. Vitamins A, E and fatty acid composition of the eggs of caged hens and pastured hens. Renew. Agric. Food Syst. 2010, 25, 45–54. [Google Scholar] [CrossRef]
- Krawczyk, J.; Sokołowicz, Z.; Szymczyk, B. Effect of housing system on cholesterol, vitamin and fatty acid content of yolk and physical characteristics of eggs from Polish native hens. Arch. Geflügelkd 2011, 75, 151–157. [Google Scholar]
- Zaniboni, L.; La Cognata, R.; Cerolini, S. Qualità dell’uovo da consumo nei diversi sistemi di allevamento considerati dalla normativa in vigore. Riv. Avic. 2006, 75, 40–45. [Google Scholar]
- Yenice, G.; Kaynar, O.; Ileriturk, M.; Hira, F.; Hayirli, A. Quality of eggs in different production systems. Czech J. Food Sci. 2016, 34, 370–376. [Google Scholar] [CrossRef]
- Hammershøj, M.; Johansen, N.F. The effect of grass and herbs in organic egg production on egg fatty acid composition, egg yolk colour and sensory properties. Livest. Sci. 2016, 194, 37–43. [Google Scholar] [CrossRef]

| Production System | Space Allowance | Indoor Floor | Oudoor Pen |
|---|---|---|---|
| Alternative—ALT | 1250 cm2/hen | Slat, 15 cm perch/hen, 250 cm2 littered area/hen, 1/3 litter | 2.5 m2/hen |
| Enriched cage—ECA | 750 cm2/hen | Wire mesh cage, 15 cm perch/hen | - |
| Litter floor—LIF | 1389 cm2/hen | Slat, 250 cm2 littered area/hen, 1/3 litter | - |
| Organic—ORG | 1667 cm2/hen | Slat, 18 cm perch/hen; 1/3 litter, max 6 hens/m2 indoor | 4.0 m2/hen |
| Egg Trait 1 | System | Outdoor Space Allowance | ||||
|---|---|---|---|---|---|---|
| ALT | ECA | LIF | ORG | IND | OUT | |
| WE (g) | 67.16 a ± 1.14 | 62.78 b ± 1.40 | 62.11 b ± 1.25 | 58.95 b ± 1.97 | 62.41 ± 1.03 | 65.104 ± 1.09 |
| ALB (g) | 40.73 ± 0.91 | 38.98 ± 1.12 | 39.64 ± 1.001 | 36.15 ± 1.58 | 39.35 ± 0.78 | 39.587 ± 0.826 |
| YOL (g) | 17.68 a ± 0.27 | 15.47 b ± 0.34 | 14.25 b ± 0.30 | 15.42 b ± 0.48 | 14.79 b ± 0.27 | 17.113 a± 0.28 |
| SHE (g) | 8.74 a ± 0.21 | 8.33 a ± 0.26 | 8.23 a ± 0.23 | 7.38 b ± 0.37 | 8.27 ± 0.19 | 8.404 ± 0.20 |
| ALB/WE (%) | 0.60 b ± 0.001 | 0.62 ab ± 0.01 | 0.64 a ± 0.01 | 0.612 ab ± 0.01 | 0.63 a ± 0.001 | 0.607 b ± 0.001 |
| YOLK/WE (%) | 0.26 a ± 0.001 | 0.27 b ± 0.001 | 0.23 c ± 0.001 | 0.26 ab ± 0.01 | 0.24 b ± 0.001 | 0.263 a ± 0.001 |
| SHE/WE (%) | 0.13 ± 0.001 | 0.13 ± 0.001 | 0.13 ± 0.001 | 0.13 ± 0.01 | 0.13 ± 0.001 | 0.129 ± 0.001 |
| TL (g/100 g edi) | 10.06 ac ± 0.23 | 9.53 c ± 0.29 | 8.01 b ± 0.26 | 11.01 a ± 0.41 | 8.69 b ± 0.22 | 10.3 a ± 0.24 |
| Fatty Acid | Housing System | Outdoor Space Allowance | ||||
|---|---|---|---|---|---|---|
| ALT | ECA | LIF | ORG | IND | OUT | |
| C16:0 | 25.7 a ± 0.2 | 24.5 b ± 0.3 | 24.8 ab ± 0.3 | 25.8 ab ± 0.4 | 24.6 b ± 0.2 | 25.7 a ± 0.2 |
| C18:0 | 10.1 a ± 0.2 | 9.4 ab ± 0.2 | 09.1 b ± 0.2 | 10.1 a ± 0.3 | 9.2 b ± 0.1 | 10.1 a ± 0.1 |
| C18:1n9 | 36.0 b ± 0.4 | 35.4 b ± 0.5 | 42.9 a ± 0.5 | 37.9 b ± 0.7 | 39.6 a ± 0.6 | 36.4 b ± 0.7 |
| C18:2n6 | 19.9 a ± 0.5 | 20.7 a ± 0.6 | 14.3 b ± 0.6 | 18.3 a ± 0.9 | 17.2 b ± 0.6 | 19.5 a ± 0.7 |
| C18:3n3 | 0.2 bc ± 0.1 | 0.8 a ± 0.1 | 0.000 c ± 0.001 | 0.4 b ± 0.1 | 0.4 ± 0.1 | 0.3 ± 0.1 |
| C20:4n6 | 3.0 a ± 0.1 | 2.7 ab ± 0.1 | 2.7 ab ± 0.1 | 2.4 b ± 0.2 | 2.7 ± 0.1 | 2.8 ± 0.1 |
| C22:6n3 | 1.2 ± 0.1 | 1.6 ± 0.1 | 1.2 ± 0.1 | 1.4 ± 0.2 | 1.4 ± 0.1 | 1.3 ± 0.1 |
| SFA | 35.7 a ± 0.3 | 33.9 b ± 0.3 | 3.8 b ± 0.3 | 35.9 a ± 0.5 | 33.9 b ± 0.2 | 35.8 a ± 0.2 |
| MUFA | 36.0 b ± 0.4 | 35.4 b ± 0.5 | 42.9 a ± 0.5 | 37.9 b ± 0.7 | 39.6 a ± 0.6 | 36.4 b ± 0.7 |
| PUFA | 24.3 ab ± 0.5 | 25.8 a ± 0.6 | 18.3 c ± 0.6 | 22.4 b ± 0.9 | 21.6 b ± 0.7 | 23.8 a ± 0.7 |
| n6/n3 | 16.2 a ± 0.8 | 10.4 b ± 1.0 | 13.5 ab ± 0.9 | 13.9 ab ± 1.5 | 12.1 b ± 0.7 | 15.5 a ± 0.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marelli, S.P.; Madeddu, M.; Mangiagalli, M.G.; Cerolini, S.; Zaniboni, L. Egg Production Systems, Open Space Allowance and Their Effects on Physical Parameters and Fatty Acid Profile in Commercial Eggs. Animals 2021, 11, 265. https://doi.org/10.3390/ani11020265
Marelli SP, Madeddu M, Mangiagalli MG, Cerolini S, Zaniboni L. Egg Production Systems, Open Space Allowance and Their Effects on Physical Parameters and Fatty Acid Profile in Commercial Eggs. Animals. 2021; 11(2):265. https://doi.org/10.3390/ani11020265
Chicago/Turabian StyleMarelli, Stefano Paolo, Manuela Madeddu, Maria Grazia Mangiagalli, Silvia Cerolini, and Luisa Zaniboni. 2021. "Egg Production Systems, Open Space Allowance and Their Effects on Physical Parameters and Fatty Acid Profile in Commercial Eggs" Animals 11, no. 2: 265. https://doi.org/10.3390/ani11020265
APA StyleMarelli, S. P., Madeddu, M., Mangiagalli, M. G., Cerolini, S., & Zaniboni, L. (2021). Egg Production Systems, Open Space Allowance and Their Effects on Physical Parameters and Fatty Acid Profile in Commercial Eggs. Animals, 11(2), 265. https://doi.org/10.3390/ani11020265






