Effects of Feeding a Hypoallergenic Diet with a Nutraceutical on Fecal Dysbiosis Index and Clinical Manifestations of Canine Atopic Dermatitis
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Selection of Animals
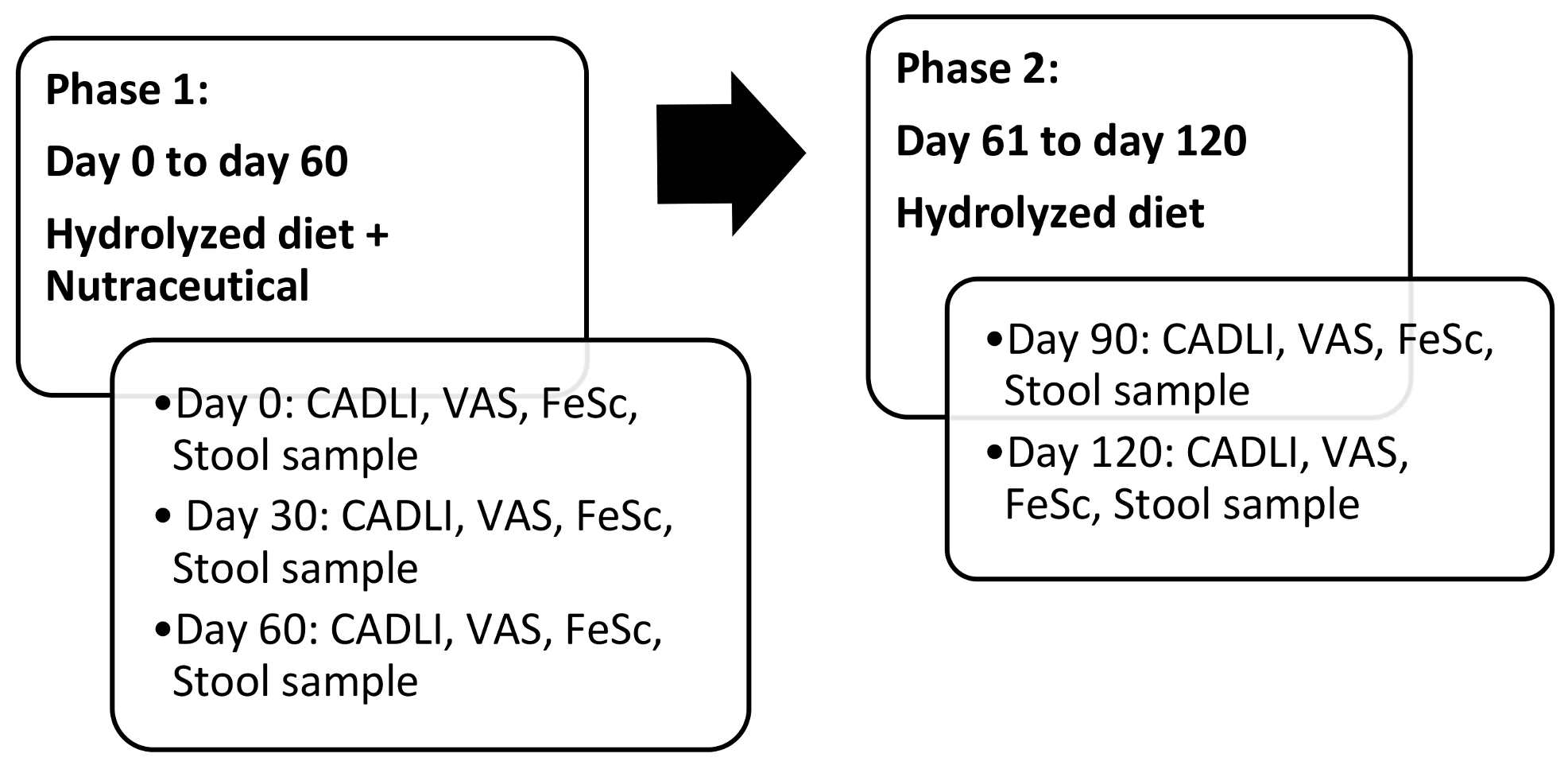
2.2. Study Design
2.3. Evaluation of Stool and Fecal Microbiome
2.4. Statistical Analysis
3. Results
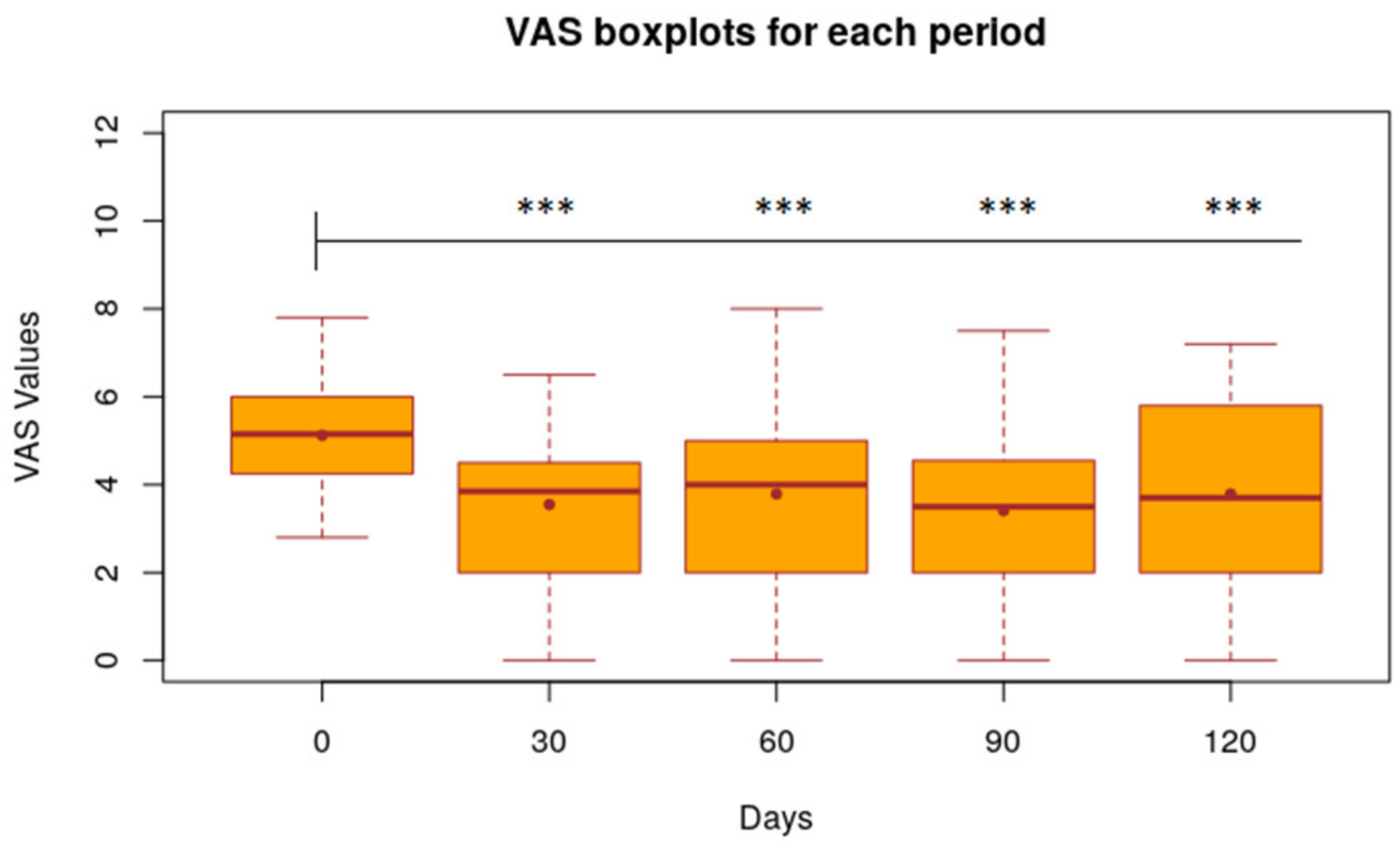
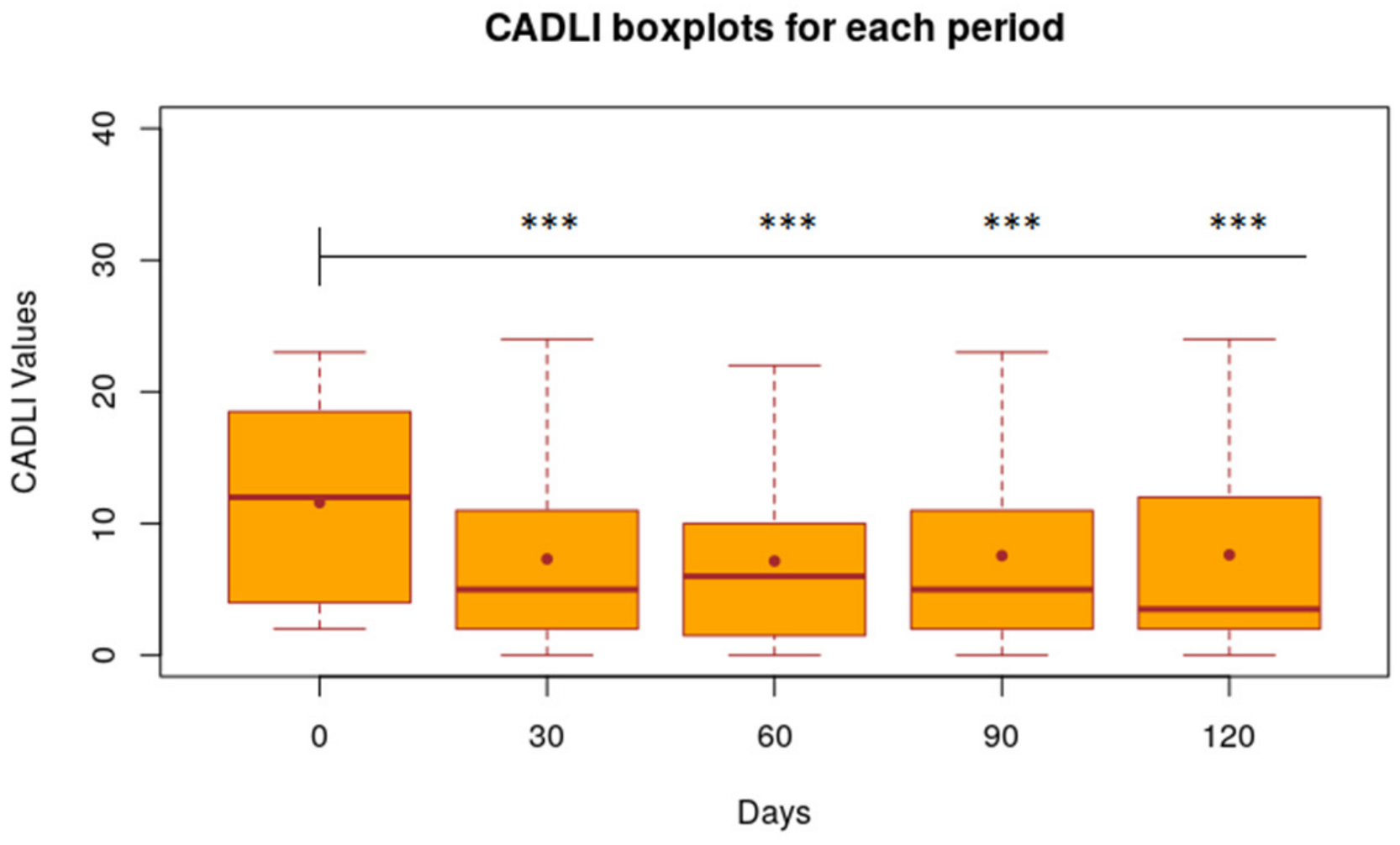
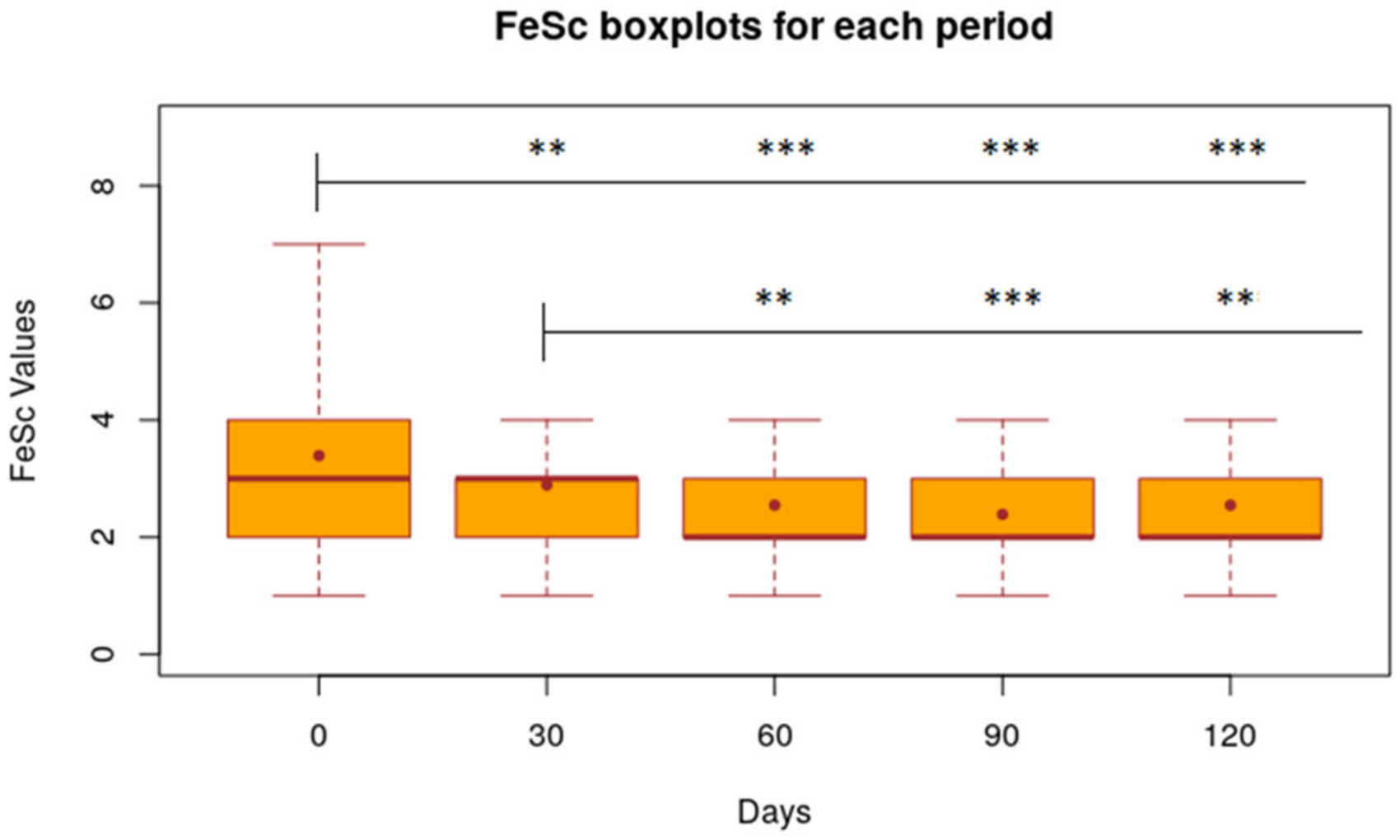
3.1. VAS, CADLI and FeSc
3.2. Medical Therapy
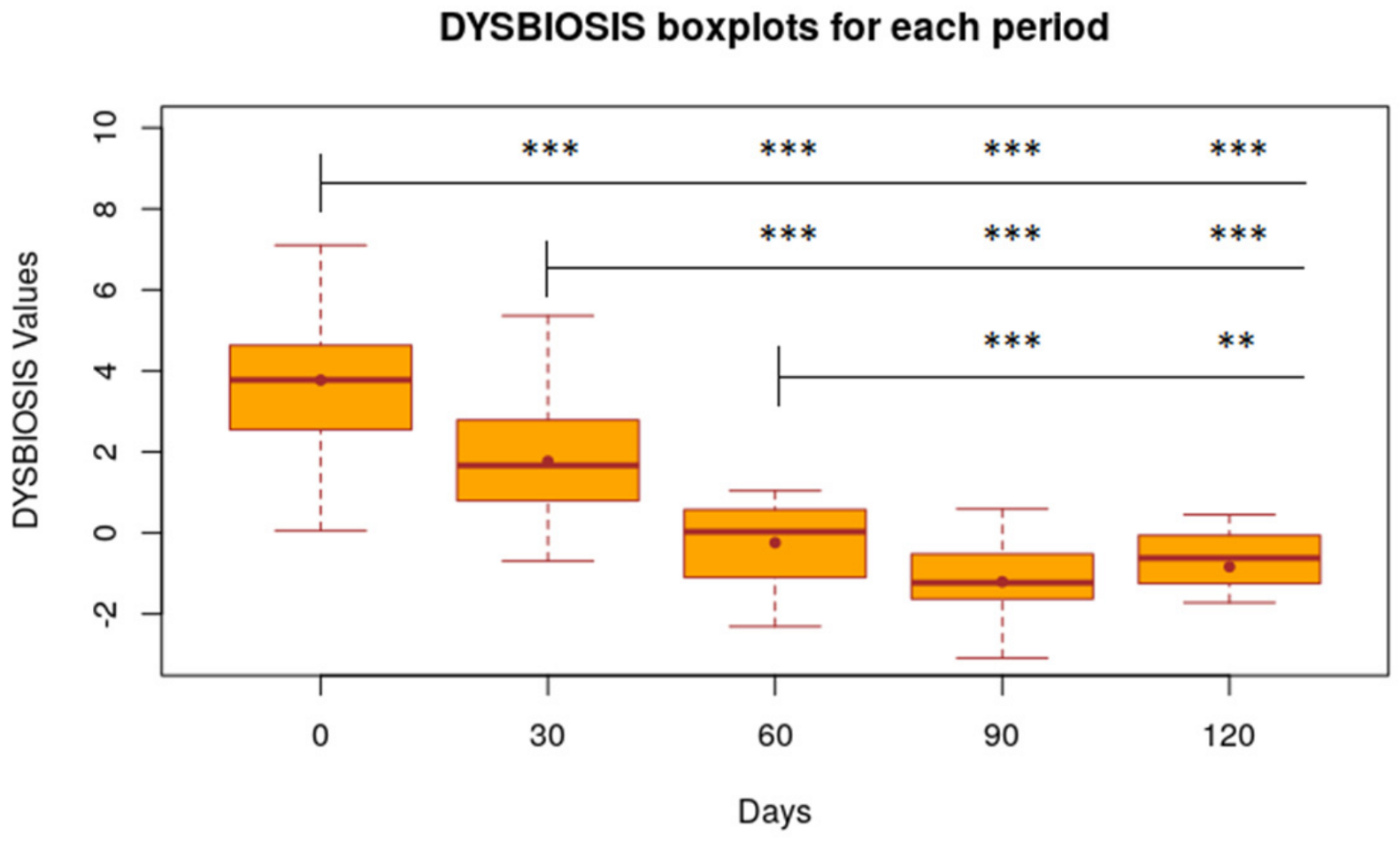
3.3. Dysbiosis Index
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Marsella, R.; De Benedetto, A. Atopic Dermatitis in Animals and People: An Update and Comparative Review. Vet. Sci. 2017, 4, 37. [Google Scholar] [CrossRef] [PubMed]
- Miller, W.H.; Griffin, C.E.; Campbell, K.L. Dermatologic therapy. In Muller & Kirk’s Small Animal Dermatology; Elsevier Mosby: Maryland Heights, MO, USA, 2013. [Google Scholar]
- Hensel, P.; Santoro, D.; Favrot, C.; Hill, P.B.; E Griffin, C. Canine atopic dermatitis: Detailed guidelines for diagnosis and allergen identification. BMC Vet. Res. 2015, 11, 196. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Olivry, T.; for the International Committee on Allergic Diseases of Animals; DeBoer, D.J.; Favrot, C.; Jackson, H.A.; Mueller, R.S.; Nuttall, T.; Prélaud, P. Treatment of canine atopic dermatitis: 2015 updated guidelines from the International Committee on Allergic Diseases of Animals (ICADA). BMC Vet. Res. 2015, 11, 210. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santoro, D. Therapies in Canine Atopic Dermatitis: An Update. Vet. Clin. N. Am. Small Anim. Pract. 2019, 49, 9–26. [Google Scholar] [CrossRef]
- Campbell, K.L. Clinical Use of Fatty Acid Supplements in Dogs. Vet. Dermatol. 1993, 4, 167–173. [Google Scholar] [CrossRef]
- Vaughn, D.M.; Reinhart, G.A.; Swaim, S.F.; Lauten, S.D.; Garner, C.A.; Boudreaux, M.K.; Spano, J.S.; Hoffman, C.E.; Conner, B. Evaluation of Effects of Dietary n-6 to n-3 Fatty Acid Ratios on Leukotriene B Synthesis in Dog Skin and Neutrophils. Vet. Dermatol. 1994, 5, 163–173. [Google Scholar] [CrossRef]
- Olivry, T.; Marsella, R.; Hillier, A. The ACVD task force on canine atopic dermatitis (XXIII): Are essential fatty acids effective? Vet. Immunol. Immunopathol. 2001, 81, 347–362. [Google Scholar] [CrossRef]
- Nesbitt, G.H.; Freeman, L.M.; Hannah, S.S. Effect of n-3 fatty acid ratio and dose on clinical manifestations, plasma fatty acids and inflammatory mediators in dogs with pruritus. Vet. Dermatol. 2003, 14, 67–74. [Google Scholar] [CrossRef]
- Noli, C.; Scarampella, F. Efficacia dell’olio di semi di ribes nero nella dermatite atopica del cane: Uno studio in doppio cieco controllato da placebo. Veterinaria 2002, 16, 60–66. [Google Scholar]
- Baddaky-Taugbol, B.; Vroom, M.V.; Nordberg, L. A randomised, controlled, double-blinded, multicentric study on the efficacy of a diet rich in fish oil and borage oil in the control of canine atopic dermatitis. In Proceedings of the 5th World Congress of Veterinary Dermatology, Vienna, Austria, 25–28 August 2004; pp. 173–187. [Google Scholar]
- Noli, C.; Carta, G.; Cordeddu, L.; Melis, M.; Murru, E.; Banni, S. Conjugated linoleic acid and black currant seed oil in the treatment of canine atopic dermatitis: A preliminary report. Vet. J. 2007, 173, 413–421. [Google Scholar] [CrossRef]
- Brown, K.; DeCoffe, D.; Molcan, E.; Gibson, D.L. Diet-Induced Dysbiosis of the Intestinal Microbiota and the Effects on Immunity and Disease. Nutrients 2012, 4, 1095–1119. [Google Scholar] [CrossRef] [Green Version]
- Suchodolski, J.S. Intestinal Microbiota of Dogs and Cats: A Bigger World than We Thought. Vet. Clin. N. Am. Small Anim. Pr. 2011, 41, 261–272. [Google Scholar] [CrossRef]
- Suchodolski, J.S.; Markel, M.E.; Garcia-Mazcorro, J.; Unterer, S.; Heilmann, R.M.; Dowd, S.; Kachroo, P.; Ivanov, I.; Minamoto, Y.; Dillman, E.M.; et al. The Fecal Microbiome in Dogs with Acute Diarrhea and Idiopathic Inflammatory Bowel Disease. PLoS ONE 2012, 7, e51907. [Google Scholar] [CrossRef] [Green Version]
- Campbell, D.E.; Mehr, S. Fifty years of allergy: 1965–2015. J. Paediatr. Child Health 2015, 5, 91–93. [Google Scholar] [CrossRef]
- Bizikova, P.; Pucheu-Haston, C.M.; Eisenschenk, M.N.C.; Marsella, R.; Nuttall, T.; Santoro, D. Review: Role of genetics and the environment in the pathogenesis of canine atopic dermatitis. Vet. Dermatol. 2015, 26, 95-e26. [Google Scholar] [CrossRef]
- Salzman, N.H. The role of the microbiome in immune cell development. Ann. Allergy Asthma Immunol. 2014, 113, 593–598. [Google Scholar] [CrossRef]
- Parodi, A.; Paolino, S.; Greco, A.; Drago, F.; Mansi, C.; Rebora, A.; Savarino, V. Small Intestinal Bacterial Overgrowth in Rosacea: Clinical Effectiveness of Its Eradication. Clin. Gastroenterol. Hepatol. 2008, 6, 759–764. [Google Scholar] [CrossRef]
- Watanabe, S.; Narisawa, Y.; Arase, S.; Okamatsu, H.; Ikenaga, T.; Tajiri, Y.; Kumemura, M. Differences in fecal microflora between patients with atopic dermatitis and healthy control subjects. J. Allergy Clin. Immunol. 2003, 111, 587–591. [Google Scholar] [CrossRef]
- Katta, R.; Desai, S.P. Diet and dermatology: The role of dietary intervention in skin disease. J. Clin. Aesthet. Dermatol. 2014, 7, 46–51. [Google Scholar]
- Marsella, R.; Santoro, D.; Ahrens, K. Early exposure to probiotics in a canine model of atopic dermatitis has long-term clinical and immunological effects. Vet. Immunol. Immunopathol. 2012, 146, 185–189. [Google Scholar] [CrossRef]
- Betsi, G.I.; Papadavid, E.; Falagas, M.E. Probiotics for the treatment or prevention of atopic dermatitis: A review of the evidence from randomized controlled trials. Am. J. Clin. Dermatol. 2008, 9, 93–103. [Google Scholar] [CrossRef]
- Rybníĉek, J.; Harvey, R.; Hill, P.B.; Lau-Gillard, P.J. Further validation of a pruritus severity scale for use in dogs. Vet. Dermatol. 2009, 20, 115–122. [Google Scholar] [CrossRef]
- Olivry, T.; Saridomichelakis, M.; Nuttall, T.; Bensignor, E.; Griffin, C.E.; Hill, P.B.; the International Committee on Allergic Diseases of Animals (ICADA). Validation of the Canine Atopic Dermatitis Extent and Severity Index (CADESI)-4, a simplified severity scale for assessing skin lesions of atopic dermatitis in dogs. Vet. Dermatol. 2014, 25, 77–85.e25. [Google Scholar] [CrossRef]
- Moxham, G. Waltham feces scoring system- A tool for veterinarians and pet owners: How does your pet rate? WALTHAM® Focus 2001, 11, 24–25. [Google Scholar]
- Alshawaqfeh, M.K.; Wajid, B.; Minamoto, Y.; Markel, M.; Lidbury, J.A.; Steiner, J.M.; Serpedin, E.; Suchodolski, J.S. A dysbiosis index to assess microbial changes in fecal samples of dogs with chronic inflammatory enteropathy. FEMS Microbiol. Ecol. 2017, 93. [Google Scholar] [CrossRef] [Green Version]
- Lozupone, C.A.; Stombaugh, J.; Gordon, J.I.; Jansson, J.K.; Knight, R. Diversity, stability and resilience of the human gut microbiota. Nature 2012, 489, 220–230. [Google Scholar] [CrossRef] [Green Version]
- Olszak, T.; An, D.; Zeissig, S.; Pinilla-Vera, M.; Richter, J.; Franke, A.; Glickman, J.N.; Siebert, R.; Baron, R.M.; Kasper, D.L.; et al. Microbial Exposure During Early Life Has Persistent Effects on Natural Killer T Cell Function. Science 2012, 336, 489–493. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arpaia, N.; Campbell, C.; Fan, X.; Dikiy, S.; Van Der Veeken, J.; DeRoos, P.; Liu, H.; Cross, J.R.; Pfeffer, K.; Coffer, P.J.; et al. Metabolites produced by commensal bacteria promote peripheral regulatory T-cell generation. Nature 2013, 504, 451–455. [Google Scholar] [CrossRef]
- Liong, M.-T. Probiotics: Biology, Genetics and Health Aspects; Springer: Berlin/Heidelberg, Germany, 2011. [Google Scholar]
- Plaza-Diaz, J.; Gomez-Llorente, C.; Fontana, L.; Gil, A. Modulation of immunity and inflammatory gene expression in the gut, in inflammatory diseases of the gut and in the liver by probiotics. World J. Gastroenterol. 2014, 20, 15632–15649. [Google Scholar] [CrossRef] [PubMed]
- Kalliomäki, M.; Salminen, S.; Arvilommi, H.; Kero, P.; Koskinen, P.; Isolauri, E. Probiotics in primary prevention of atopic disease: A randomised placebo-controlled trial. Lancet 2001, 357, 1076–1079. [Google Scholar] [CrossRef]
- Akter, S.; Part, J.H.; Jung, H.K. Potential health promoting beneficts of parabiotics, inactivated probiotic cells. J. Microbiol. Biotechnol. 2020, 30, 477–481. [Google Scholar] [CrossRef]
- Teame, T.; Wang, A.; Xie, M.; Zhang, Z.; Yang, Y.; Ding, Q.; Gao, C.; Olsen, R.E.; Ran, C.; Zhou, Z. Paraprobiotics and Postbiotics of Probiotic Lactobacilli, Their Positive Effects on the Host and Action Mechanisms: A Review. Front. Nutr. 2020, 7, 570344. [Google Scholar] [CrossRef]
- Lee, S.-Y.; Lee, E.; Park, Y.M.; Hong, S.-J. Microbiome in the Gut-Skin Axis in Atopic Dermatitis. Allergy, Asthma Immunol. Res. 2018, 10, 354–362. [Google Scholar] [CrossRef]
- Unsal, H.; Balkaya, H.U.A.M. Glucocorticoids and the Intestinal Environment. In Lucocorticoids—New Recognition of Our Familiar Friend; Qian, X., Ed.; InTech: Rijeka, Croatia, 2012; pp. 7–150. [Google Scholar]
- Navarro-López, V.; Núñez-Delegido, E.; Ruzafa-Costas, B.; Sánchez-Pellicer, P.; Agüera-Santos, J.; Navarro-Moratalla, L. Probiotics in the Therapeutic Arsenal of Dermatologists. Microorganisms 2021, 9, 1513. [Google Scholar] [CrossRef]
- Park, D.; Kim, J.; Park, H.-J.; Hahm, D.-H. Comparative Analysis of the Microbiome across the Gut–Skin Axis in Atopic Dermatitis. Int. J. Mol. Sci. 2021, 22, 4228. [Google Scholar] [CrossRef]
- Cornegliani, L.; Persico, P.; Di Prinzio, R.; Gramenzi, A. Valutazione del microbiota intestinale in cani sani e cani affetti da dermatite atopica cronica. Evaluation of intestinal microbiota in atopica and healthy dogs. Veterinaria 2021, 35, 195–200. [Google Scholar]
| Breed | Age | Gender | Weight | Therapy | Previous Diet |
|---|---|---|---|---|---|
| Abruzzese Maremma shepherd dog | 8 | nf | 45 | immunotherapy | hydrolised diet (Z/D Hill’s) |
| French bulldog | 3 | nf | 10 | immunotherapy | hydrolised diet (Anallergenic, Royal Canin) |
| French bulldog | 4.5 | f | 15.5 | immunotherapy | monoproteic diet (Exclusion, Pork) |
| English bulldog | 4 | m | 31 | immunotherapy | hydrolised diet (Trainer, Rabbit) |
| Labrador retriever | 2.5 | f | 40 | immunotherapy | hydrolised diet (Z/D Hill’s) |
| Mongrel | 6 | nf | 38.5 | immunotherapy | hydrolised diet (Z/D Hill’s) |
| Newfoundland | 5 | m | 68 | immunotherapy | monoproteic diet (Forza 10, Beef) |
| Labrador retriever | 2 | f | 25 | lokivetmab | hydrolised diet (Hypoallergenic Royal Canin) |
| Border collie | 4 | f | 24 | lokivetmab | monoproteic diet (Trainer, Beef) |
| Boxer | 2 | m | 20 | lokivetmab | monoproteic diet (Exclusion, Rabbit) |
| French bulldog | 2 | f | 10 | lokivetmab | monoproteic diet (Exclusion, Salmon) |
| German shepherd | 7 | f | 37 | lokivetmab | hydrolised diet (H/A Purina) |
| Chinese shar-pei | 2.5 | f | 17 | lokivetmab | hydrolised diet (H/A Purina) |
| American Stafforshire terrier | 2 | m | 35 | none | monoproteic diet (Trainer, Pork) |
| English bulldog | 2 | m | 28 | none | monoproteic diet (Trainer, Rabbit) |
| Mongrel | 6 | f | 18.5 | none | limited-antigen diet (Acana, Pork and Beef) |
| Border collie | 8 | m | 21 | oclacitinib | hydrolised diet (Z/D Hill’s) |
| Boxer | 3.5 | f | 23.8 | oclacitinib | hydrolised diet (Hypoallergenic Royal Canin) |
| Cane corso | 8 | nf | 54 | oclacitinib | hydrolised diet (H/A Purina) |
| Dogue de Bordeaux | 6 | nf | 62 | oclacitinib | monoproteic diet (Trainer, Rabbit) |
| Dogue de Bordeaux | 2 | m | 52 | oclacitinib | monoproteic diet (Forza 10, Beef) |
| Golden retriever | 4.7 | f | 33 | oclacitinib | hydrolised diet (Z/D Hill’s) |
| Golden retriever | 4 | m | 35 | oclacitinib | monoproteic diet (Exclusion, Pork) |
| Jack Russel terrier | 2.8 | f | 8.1 | oclacitinib | monoproteic diet (Trainer, Pork) |
| Maltese | 2.3 | f | 5 | oclacitinib | monoproteic diet (Prolife, Pork) |
| Mongrel | 8 | nm | 24 | oclacitinib | monoproteic diet (Exclusion, Pork) |
| Mongrel | 5 | nf | 20 | oclacitinib | monoproteic diet (Prolife, Beef) |
| Mongrel | 9 | nf | 14 | oclacitinib | monoproteic diet (Prolife, Rabbit) |
| Chinese shar-pei | 2.5 | f | 14.5 | oclacitinib | fatty acid-enriched diet (Derm Defence Hill’s) |
| West Higland white terrier | 9 | f | 8.3 | oclacitinib | limited-antigen diet (Sensitivity Royal Canin) |
| West Higland white terrier | 8 | f | 8.9 | oclacitinib | limited-antigen diet (Sensitivity Royal Canin) |
| West Higland white terrier | 2 | m | 11 | oclacitinib | hydrolised diet (Anallergenic Royal Canin) |
| Components | Dose |
|---|---|
| Black currant seed oil | 625 mg/capsule |
| Heat-inactivated Lactobacillus reuteri NBF 1 | 200 mg/capsule |
| Zinc oxide | 25 mg/capsule |
| Nucleotides | 50 mg/capsule |
| Dogs | Therapy | Composition of Immunotherapy | Dose |
|---|---|---|---|
| 7 | Specific immunotherapy | 2 DF, TP; 4 DF, TP, AS; 1 DF | Maintenance therapy 1 mL/sc once a month |
| 6 | Lokivetmab | 1 mg/kg/sc once a month | |
| 3 | None | ||
| 16 | Oclacitinib | 0.4–0.6 mg/kg/q24 h orally |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guidi, E.E.A.; Gramenzi, A.; Persico, P.; Di Prinzio, R.; Di Simone, D.; Cornegliani, L. Effects of Feeding a Hypoallergenic Diet with a Nutraceutical on Fecal Dysbiosis Index and Clinical Manifestations of Canine Atopic Dermatitis. Animals 2021, 11, 2985. https://doi.org/10.3390/ani11102985
Guidi EEA, Gramenzi A, Persico P, Di Prinzio R, Di Simone D, Cornegliani L. Effects of Feeding a Hypoallergenic Diet with a Nutraceutical on Fecal Dysbiosis Index and Clinical Manifestations of Canine Atopic Dermatitis. Animals. 2021; 11(10):2985. https://doi.org/10.3390/ani11102985
Chicago/Turabian StyleGuidi, Eleonora Elisa Alice, Alessandro Gramenzi, Paola Persico, Roberta Di Prinzio, Daniele Di Simone, and Luisa Cornegliani. 2021. "Effects of Feeding a Hypoallergenic Diet with a Nutraceutical on Fecal Dysbiosis Index and Clinical Manifestations of Canine Atopic Dermatitis" Animals 11, no. 10: 2985. https://doi.org/10.3390/ani11102985
APA StyleGuidi, E. E. A., Gramenzi, A., Persico, P., Di Prinzio, R., Di Simone, D., & Cornegliani, L. (2021). Effects of Feeding a Hypoallergenic Diet with a Nutraceutical on Fecal Dysbiosis Index and Clinical Manifestations of Canine Atopic Dermatitis. Animals, 11(10), 2985. https://doi.org/10.3390/ani11102985







