Salivary Chromogranin A (CgA) Response to the Noradrenaline Transporter Blocker Atomoxetine in Dogs
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Study Protocol
2.3. Saliva Sample Collection
2.4. CgA and Cortisol Assay
2.5. Statistical Analysis
3. Results
3.1. Salivary CgA
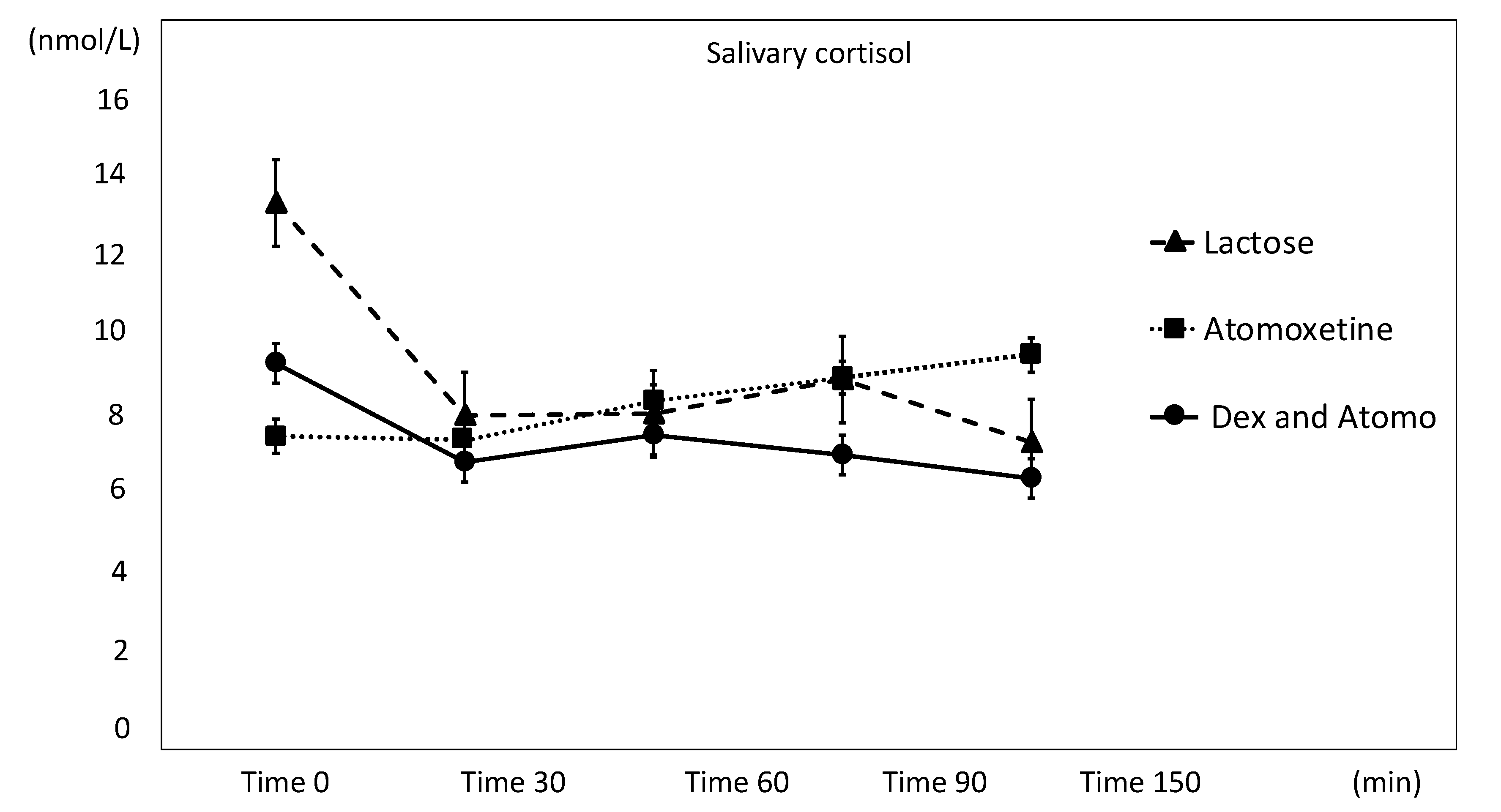
3.2. Salivary Cortisol
3.3. Heart Rate and Behavior Responses
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chmelíková, E.; Bolechová, P.; Chaloupková, H.; Svobodová, I.; Jovičić, M.; Sedmíková, M. Salivary cortisol as a marker of acute stress in dogs: A review. Domest. Anim. Endocrinol. 2020, 72, 106428. [Google Scholar] [CrossRef]
- Hekman, J.P.; Karas, A.Z.; Dreschel, N.A. Salivary cortisol concentrations and behavior in a population of healthy dogs hospitalized for elective procedures. Appl. Anim. Behav. Sci. 2012, 141, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Lensen, R.C.M.M.; Moons, C.P.H.; Diederich, C. Physiological stress reactivity and recovery related to behavioral traits in dogs (Canis familiaris). PLoS ONE 2019, 14, e0222581. [Google Scholar] [CrossRef]
- Polgár, Z.; Blackwell, E.J.; Rooney, N.J. Assessing the welfare of kennelled dogs—A review of animal-based measures. Appl. Anim. Behav. Sci. 2019, 213, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Accorsi, P.A.; Carloni, E.; Valsecchi, P.; Viggiani, R.; Gamberoni, M.; Tamanini, C.; Seren, E. Cortisol determination in hair and faeces from domestic cats and dogs. Gen. Comp. Endocrinol. 2008, 155, 398–402. [Google Scholar] [CrossRef] [PubMed]
- Beerda, B.; Schilder, M.B.; Janssen, N.S.; Mol, J.A. The use of saliva cortisol, urinary cortisol, and catecholamine measurements for a noninvasive assessment of stress responses in dogs. Horm. Behav. 1996, 30, 272–279. [Google Scholar] [CrossRef] [PubMed]
- Dreschel, N.A.; Granger, D.A. Methods of collection for salivary cortisol measurement in dogs. Horm. Behav. 2009, 55, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Palme, R. Non-invasive measurement of glucocorticoids: Advances and problems. Physiol. Behav. 2019, 199, 229–243. [Google Scholar] [CrossRef] [PubMed]
- Rooney, N.J.; Gaines, S.A.; Bradshaw, J.W.S. Behavioural and glucocorticoid responses of dogs (Canis familiaris) to kennelling: Investigating mitigation of stress by prior habituation. Physiol. Behav. 2007, 92, 847–854. [Google Scholar] [CrossRef] [PubMed]
- Steiss, J.E.; Schaffer, C.; Ahmad, H.A.; Voith, V.L. Evaluation of plasma cortisol levels and behavior in dogs wearing bark control collars. Appl. Anim. Behav. Sci. 2007, 106, 96–106. [Google Scholar] [CrossRef]
- Akiyoshi, H.; Aoki, M.; Kumagai, D.; Saleh, N.; Noda, K.; Shimada, T.; Sugii, S.; Ohashi, F. Emzyme-linked immunosorbent assay for detection of canine chromogranin A by use of immunological cross-reactivity of rabbit anti-bovine chromogranin A antibody. J. Vet. Med Sci. 2005, 67, 535–537. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Escribano, D.; Soler, L.; Gutiérrez, A.M.; Martínez-Subiela, S.; Cerón, J.J. Measurement of chromogranin A in porcine saliva: Validation of a time-resolved immunofluorometric assay and evaluation of its application as a marker of acute stress. Animal 2013, 7, 640–647. [Google Scholar] [CrossRef] [PubMed]
- Stridsberg, M.; Pettersson, A.; Hagman, R.; Westin, C.; Höglund, O. Chromogranins can be measured in samples from cats and dogs. BMC Res. Notes 2014, 7, 336. [Google Scholar] [CrossRef] [PubMed]
- Taupenot, L.; Harper, K.L.; O’Connor, D.T. The Chromogranin–Secretogranin Family. N. Engl. J. Med. 2003, 348, 2579–2580. [Google Scholar] [CrossRef] [PubMed]
- Banks, P.; Helle, K. The release of protein from the stimulated adrenal medulla. Biochem. J. 1965, 97, 40C–41C. [Google Scholar] [CrossRef] [PubMed]
- Helle, K.B. Some chemical and physical properties of the soluble protein fraction of bovine adrenal chromaffin granules. Mol. Pharmacol. 1966, 2, 298–310. [Google Scholar]
- Saruta, J.; Tsukinoki, K.; Sasaguri, K.; Ishii, H.; Yasuda, M.; Osamura, Y.R.; Watanabe, Y.; Sato, S. Expression and localization of chromogranin A gene and protein in human submandibular gland. Cells Tissues Organs 2005, 180, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Kanai, K.; Hino, M.; Hori, Y.; Nakao, R.; Hoshi, F.; Itoh, N.; Higuchi, S. Circadian variations in salivary chromogranin a concentrations during a 24-hour period in dogs. J. Vet. Sci. 2008, 9, 423–425. [Google Scholar] [CrossRef] [PubMed]
- Escribano, D.; Fuentes-Rubio, M.; Cerón, J.J. Salivary testosterone measurements in growing pigs: Validation of an automated chemiluminescent immunoassay and its possible use as an acute stress marker. Res. Vet. Sci. 2014, 97, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Liu, Z.; Liu, W.; Yin, C.; Ci, L.; Zhao, R.; Yang, X. Short communication: Salivary haptoglobin and chromogranin A as non-invasive markers during restraint stress in pigs. Res. Vet. Sci. 2017, 114, 27–30. [Google Scholar] [CrossRef] [PubMed]
- Gehlert, D.R.; Gackenheimer, S.L.; Robertson, D.W. Localization of rat brain binding sites for [3H] tomoxetine, an enantiomerically pure ligand for norepinephrine reuptake sites. Neurosci. Lett. 1993, 157, 203–206. [Google Scholar] [CrossRef]
- Wong, D.T.; Threlkeld, P.G.; Best, K.L.; Bymaster, F.P. A new inhibitor of norepinephrine uptake devoid of affinity for receptors in rat brain. J. Pharmacol. Exp. Ther. 1982, 222, 61–65. [Google Scholar]
- Mattiuz, E.L.; Ponsler, G.D.; Barbuch, R.J.; Wood, P.G.; Mullen, J.H.; Shugert, R.L.; Li, Q.; Wheeler, W.J.; Kuo, F.; Conrad, P.C.; et al. Disposition and metabolic fate of atomoxetine hydrochloride: Pharmacokinetics, metabolism, and excretion in the Fischer 344 rat and beagle dog. Drug Metab. Dispos. 2003, 31, 88–97. [Google Scholar] [CrossRef] [PubMed]
- Cobb, M.L.; Iskandarani, K.; Chinchilli, V.M.; Dreschel, N.A. A systematic review and meta-analysis of salivary cortisol measurement in domestic canines. Domest. Anim. Endocrinol. 2016, 57, 31–42. [Google Scholar] [CrossRef]
- Csoltova, E.; Martineau, M.; Boissy, A.; Gilbert, C. Behavioral and physiological reactions in dogs to a veterinary examination: Owner-dog interactions improve canine well-being. Physiol. Behav. 2017, 177, 270–281. [Google Scholar] [CrossRef] [PubMed]
- Stollar, O.; Moore, G.; Mukhopadhyay, A.; Gwin, W.; Ogata, N. Effects of a single dose of oral gabapentin in dogs during a veterinary visit: A double-blind, placebo-controlled study. J. Am. Vet. Med Assoc. 2021. accepted. [Google Scholar]
- Hekman, J.P.; Karas, A.Z.; Sharp, C.R.; Grafton, N. Psychogenic stress in hospitalized dogs: Cross species comparisons, implications for health care, and the challenges of evaluation. Animals 2014, 4, 331–347. [Google Scholar] [CrossRef] [PubMed]
- Den, R.; Toda, M.; Nagasawa, S.; Kitamura, K.; Morimoto, K. Circadian rhythm of human salivary chromogranin A. Biomed. Res. 2007, 28, 57–60. [Google Scholar] [CrossRef] [PubMed]
- ZoetisUS.com. Available online: https://www.zoetisus.com/contact/pages/product_information/msds_pi/pi/sileo.pdf (accessed on 15 August 2021).
- Warren, C.M.; van den Brink, R.L.; Nieuwenhuis, S.; Bosch, J.A. Norepinephrine transporter blocker atomoxetine increases salivary alpha amylase. Psychoneuroendocrinology 2017, 78, 233–236. [Google Scholar] [CrossRef] [PubMed]
- Chamberlain, S.R.; Müller, U.; Cleary, S.; Robbins, T.W.; Sahakian, B.J. Atomoxetine increases salivary cortisol in healthy volunteers. J. Psychopharmacol. 2007, 21, 545–549. [Google Scholar] [CrossRef] [PubMed]
- Stern, L.; Schell, M. Management of Attention-Deficit Disorder and Attention-Deficit/Hyperactivity Disorder Drug Intoxication in Dogs and Cats: An Update. Vet. Clin. North Am.-Small Anim. Pract. 2018, 48, 959–968. [Google Scholar] [CrossRef] [PubMed]
- Tiira, K.; Sulkama, S.; Lohi, H. Prevalence, comorbidity, and behavioral variation in canine anxiety. J. Vet. Behav. 2016, 16, 36–44. [Google Scholar] [CrossRef]
- Kurachi, T.; Irimajiri, M.; Mizuta, Y.; Satoh, T. Dogs predisposed to anxiety disorders and related factors in Japan. Appl. Anim. Behav. Sci. 2017, 196, 69–75. [Google Scholar] [CrossRef]
- Dinwoodie, I.R.; Dwyer, B.; Zottola, V.; Gleason, D.; Dodman, N.H. Demographics and comorbidity of behavior problems in dogs. J. Vet. Behav. 2019, 32, 62–71. [Google Scholar] [CrossRef]
- Salonen, M.; Sulkama, S.; Mikkola, S.; Puurunen, J.; Hakanen, E.; Tiira, K.; Araujo, C.; Lohi, H. Prevalence, comorbidity, and breed differences in canine anxiety in 13,700 Finnish pet dogs. Sci. Rep. 2020, 10, 2962. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kooriyama, T.; Mukhopadhyay, A.; Moore, G.E.; Ogata, N. Salivary Chromogranin A (CgA) Response to the Noradrenaline Transporter Blocker Atomoxetine in Dogs. Animals 2021, 11, 2844. https://doi.org/10.3390/ani11102844
Kooriyama T, Mukhopadhyay A, Moore GE, Ogata N. Salivary Chromogranin A (CgA) Response to the Noradrenaline Transporter Blocker Atomoxetine in Dogs. Animals. 2021; 11(10):2844. https://doi.org/10.3390/ani11102844
Chicago/Turabian StyleKooriyama, Takanori, Abhijit Mukhopadhyay, George E. Moore, and Niwako Ogata. 2021. "Salivary Chromogranin A (CgA) Response to the Noradrenaline Transporter Blocker Atomoxetine in Dogs" Animals 11, no. 10: 2844. https://doi.org/10.3390/ani11102844
APA StyleKooriyama, T., Mukhopadhyay, A., Moore, G. E., & Ogata, N. (2021). Salivary Chromogranin A (CgA) Response to the Noradrenaline Transporter Blocker Atomoxetine in Dogs. Animals, 11(10), 2844. https://doi.org/10.3390/ani11102844




