Fin Erosion of Salmo salar (Linnaeus 1758) Infested with the Parasite Caligus rogercresseyi (Boxshall & Bravo 2000)
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Maintenance Conditions
2.2. Production of Infective Stages and Fish Infestation
2.3. Experimental Design
2.4. Sampling Procedure
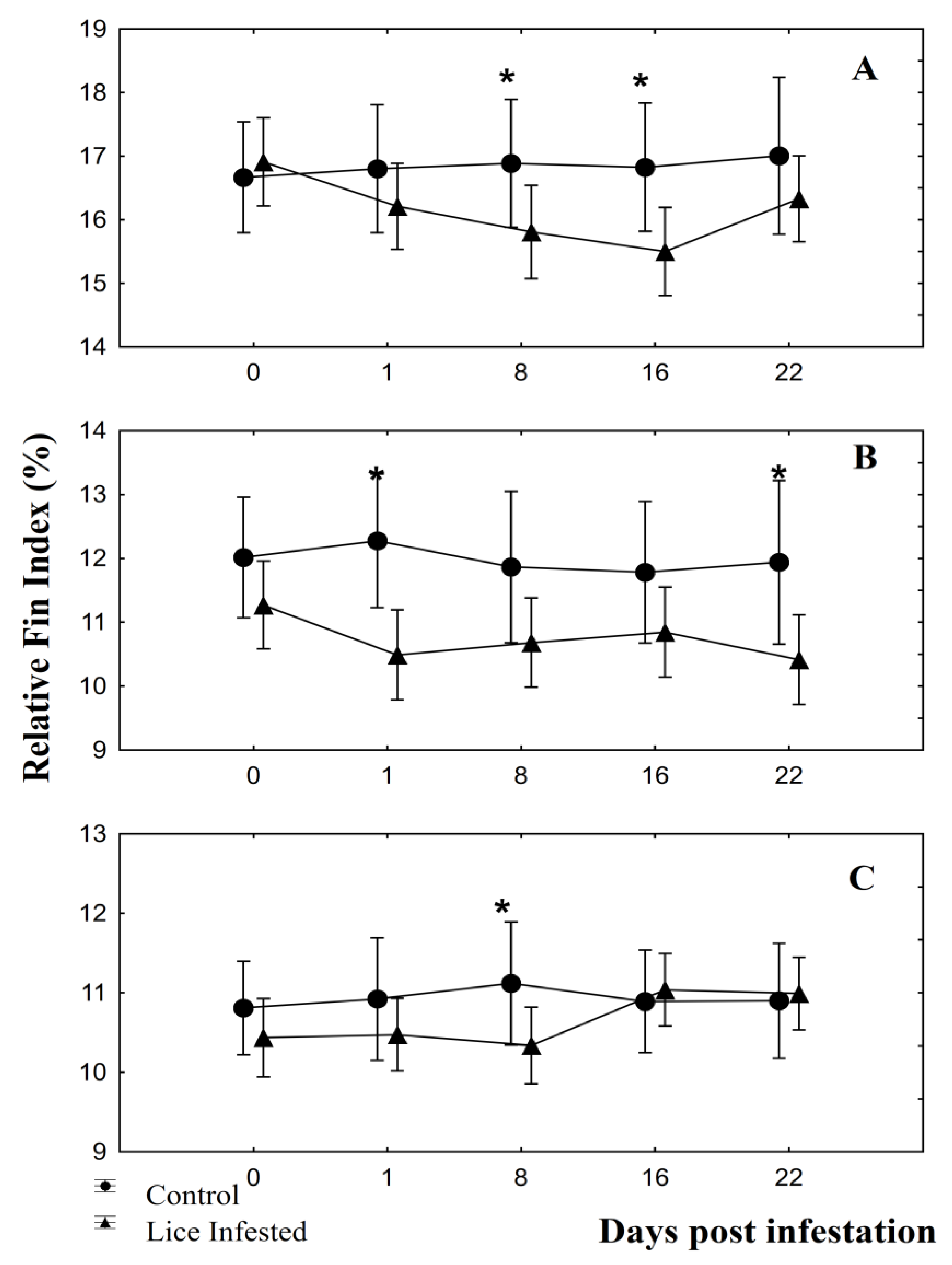
2.5. Fin Damage Quantification
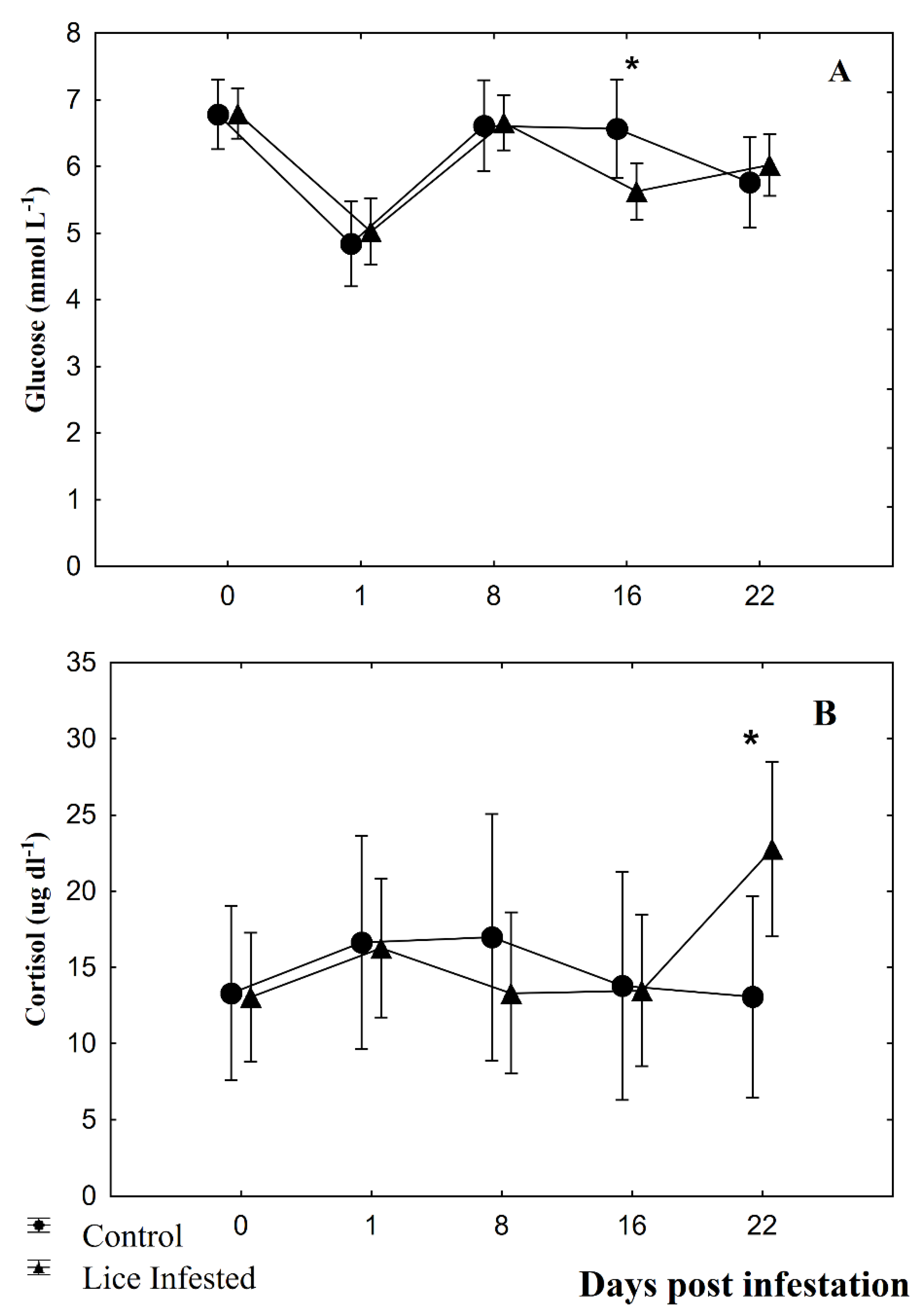
2.6. Plasma Parameters
2.7. Statistical Analysis
3. Results
3.1. Caligus Rogercresseyi Development and Abundances
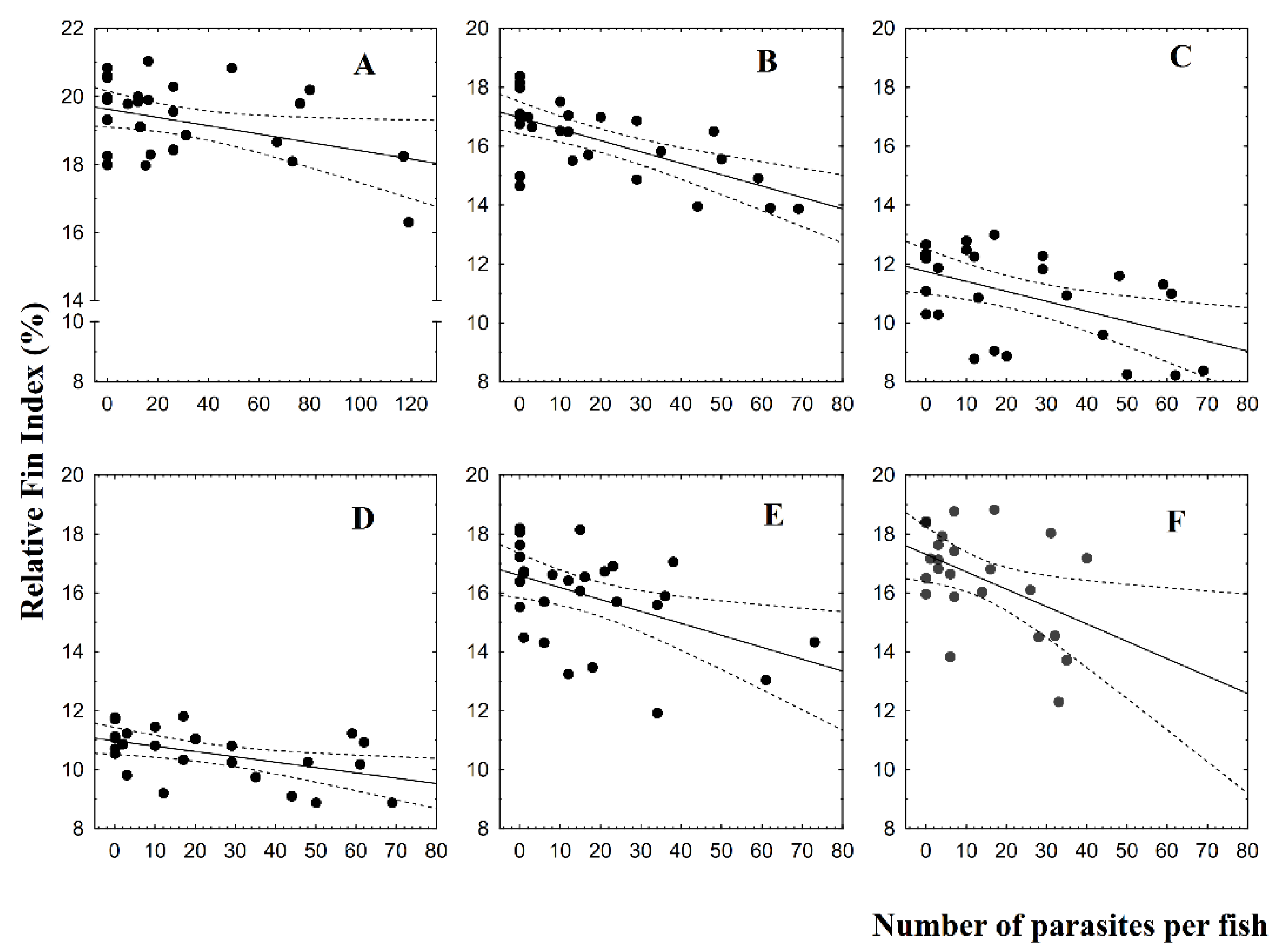
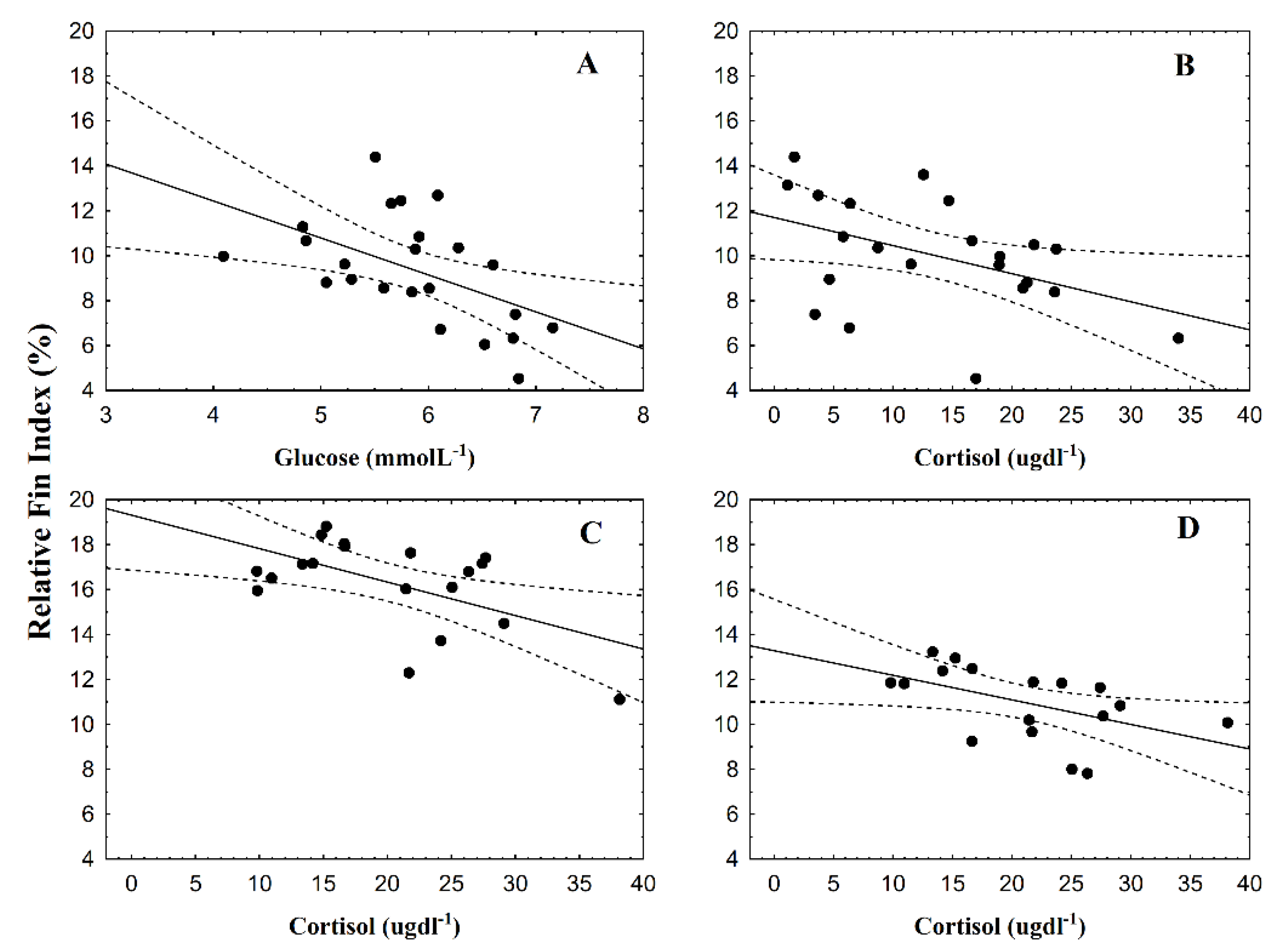
3.2. Fin Damage, Plasma Glucose, and Cortisol Levels
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tadiso, T.M.; Krasnov, A.; Skugor, S.; Afanasyev, S.; Hordvik, I.; Nilsen, F. Gene expression analyses of immune responses in Atlantic salmon during early stages of infection by salmon louse (Lepeophtheirus salmonis) revealed bi-phasic responses coinciding with the copepod-chalimus transition. BMC Genom. 2011, 12, 141. [Google Scholar] [CrossRef]
- Hamilton-West, C.; Arriagada, G.; Yatabe, T.; Valdes, P.; Herve-Claude, L.P.; Urcelay, S. Epidemiological description of the sea lice (Caligus rogercresseyi) situation in southern Chile in August 2007. Prev. Vet. Med. 2012, 104, 341–345. [Google Scholar] [CrossRef] [PubMed]
- Costello, M.J. Ecology of sea lice parasitic on farmed and wild fish. Trends Parasitol. 2006, 22, 475–483. [Google Scholar] [CrossRef]
- Revie, C.W.; Hollinger, E.; Gettinby, G.; Lees, F.; Heuch, P.A. Clustering of parasites within cages on Scottish and Norwegian salmon farms: Alternative sampling strategies illustrated using simulation. Prev. Vet. Med. 2007, 81, 135–147. [Google Scholar] [CrossRef] [PubMed]
- González, L.; Carvajal, J. Life cycle of Caligus rogercresseyi, (Copepoda: Caligidae) parasite of Chilean reared salmonids. Aquaculture 2003, 220, 101–117. [Google Scholar] [CrossRef]
- González, M.P.; Marín, S.L.; Vargas-Chacoff, L. Effects of Caligus rogercresseyi (Boxshall and Bravo, 2000) infestation on physiological response of host Salmo salar (Linnaeus 1758): Establishing physiological thresholds. Aquaculture 2015, 438, 47–54. [Google Scholar] [CrossRef]
- Marín, S.L.; Martin, R.; Lewis, R. Effects of Caligus rogercresseyi (Boxshall and Bravo 2000) chalimus stage condition (dead, moribund, live) on the estimates of Cypermethrin BETAMAX® efficacy. Aquac. Res. 2015, 46, 30–36. [Google Scholar] [CrossRef]
- MacKinnon, B.M. Host response of atlantic salmon (Salmo salar) to Infection by Sea Lice (Caligus elongatus). Can. J. Fish Aquat. Sci. 1993, 50, 789–792. [Google Scholar] [CrossRef]
- Bowers, J.M.; Mustafa, A.; Speare, D.J.; Conboy, G.A.; Brimacombe, M.; Sims, D.E.; Burka, J.F. The physiological response of Atlantic salmon, Salmo salar L.; to a single experimental challenge with sea lice, Lepeophtheirus salmonis. J. Fish Dis. 2000, 23, 165–172. [Google Scholar] [CrossRef]
- Fast, M.D.; Muise, D.M.; Easy, R.E.; Ross, N.W.; Johnson, S.C. The effects of Lepeophtheirus salmonis infections on the stress response and immunological status of Atlantic salmon (Salmo salar). Fish Shellfish Immunol. 2006, 21, 228–241. [Google Scholar] [CrossRef]
- Grimnes, A.; Jakobsen, P.J. The physiological effects of salmon lice infection on post-smolt of Atlantic salmon. J. Fish Biol. 1996, 48, 1179–1194. [Google Scholar] [CrossRef]
- Wells, A.; Grierson, C.E.; MacKenzie, M.; Russon, I.J.; Reinardy, H.; Middlemiss, C.; Bjørn, P.A.; Finstad, B.; Bonga, S.E.W.; Todd, C.D.; et al. Physiological effects of simultaneous, abrupt seawater entry and sea lice (Lepeophtheirus salmonis) infestation of wild, sea-run brown trout (Salmo trutta) smolts. Can. J. Fish Aquat. Sci. 2006, 63, 2809–2821. [Google Scholar] [CrossRef]
- Boxshall, G.A.; Defaye, D. Pathogens of Wild and Farmed Fish: Sea Lice; CRC Press: Boca Raton, FL, USA, 2006. [Google Scholar]
- Treasurer, J.W.; Wadsworth, S.L. Interspecific comparison of experimental and natural routes of Lepeophtheirus salmonis and Caligus elongatus challenge and consequences for distribution of chalimus on salmonids and therapeutant screening. Aquac. Res. 2004, 35, 773–783. [Google Scholar] [CrossRef]
- Person-Le Ruyet, J.; Le Bayon, N.; Gros, S. How to assess fin damage in rainbow trout, Oncorhynchus mykiss? Aquat. Living Resour. 2007, 20, 191–195. [Google Scholar] [CrossRef]
- Noble, C.; Gismervik, K.; Iversen, M.H.; Kolarevic, J.; Nilsson, J.; Stien, L.H.; Turnbull, J.F. Welfare Indicators for Farmed Atlantic Salmon: Tools for Assessing Fish Welfare; FHF The Norwegian Seafood Research Fund: Oslo, The Netherlands, 2018. [Google Scholar]
- Huntingford, F.A.; Kadri, S. Chapter 2: Welfare and Fish. In Fish Welfare; Branson, E., Ed.; Blackwell Publishing: Hoboken, NJ, USA, 2008; p. 316. [Google Scholar]
- Ellis, T.; Yildiz, H.Y.; Lopez-Olmeda, J.; Spedicato, M.T.; Tort, L.; Overli, O.; Martins, C.I. Cortisol and finfish welfare. Fish Physiol. Biochem. 2012, 38, 163–188. [Google Scholar] [CrossRef] [PubMed]
- Treasurer, J.; Feledi, T. The Physical condition and welfare of five species of wild-caught wrasse stocked under aquaculture conditions and when stocked in Atlantic Salmon, Salmo salar, production cages. J. World Aquacult. Soc. 2014, 45, 213–219. [Google Scholar] [CrossRef]
- Ellis, T.; James, J.D.; Stewart, C.; Scott, A.P. A non-invasive stress assay based upon measurement of free cortisol released into the water by rainbow trout. J. Fish Biol. 2004, 65, 1233–1252. [Google Scholar] [CrossRef]
- Huntingford, F.A.; Adams, C.; Braithwaite, V.; Kadri, S.; Pottinger, T.; Sandøe, P.; Turnbull, J. Current issues in fish welfare. J. Fish Biol. 2006, 68, 332–372. [Google Scholar] [CrossRef]
- Ellis, T.; Oidtmann, B.; St-Hilaire, S.; Turnbull, J.; North, B.; MacIntyre, C.; Nikolaidis, J.; Hoyle, I.; Kestin, S.; Knowles, T. Fin Erosion in Farmed Fish. In Fish Welfare; Branson, E.J., Ed.; Wiley Blackwell: Hoboken, NJ, USA, 2008; pp. 121–149. [Google Scholar]
- Noble, C.; Kadri, S.; Mitchell, D.F.; Huntingford, F.A. Growth, production and fin damage in cage-held 0+ Atlantic salmon presmolts (Salmo salar L.) fed either a) on-demand, or b) to a fixed satiation–restriction regime: Data from a commercial farm. Aquaculture 2008, 275, 163–168. [Google Scholar] [CrossRef]
- Hoyle, I.; Oidtmann, B.; Ellis, T.; Turnbull, J.; North, B.; Nikolaidis, J.; Knowles, T.G. A validated macroscopic key to assess fin damage in farmed rainbow trout (Oncorhynchus mykiss). Aquaculture 2007, 270, 142–148. [Google Scholar] [CrossRef]
- Huntingford, F.; Kadri, S. Defining, assessing and promoting the welfare of farmed fish. Rev. Sci. Tech. 2014, 33, 233–244. [Google Scholar] [CrossRef] [PubMed]
- Bosakowski, T.; Wagner, E.J. Assessment of fin erosion by comparison of relative fin length in hatchery and wild trout in Utah. Can. J. Fish Aquat. Sci. 1994, 51, 636–641. [Google Scholar] [CrossRef]
- Araya, A.; Mancilla, M.; Lhorente, J.P.; Neira, R.; Gallardo, J.A. Experimental challenges of Atlantic salmon Salmo salar with incremental levels of copepodids of sea louse Caligus rogercresseyi: Effects on infestation and early development. Aquac. Res. 2012, 43, 1904–1908. [Google Scholar] [CrossRef]
- Close, B.; Banister, K.; Baumans, V.; Bernoth, E.-M.; Bromage, N.; Bunyan, J.; Erhardt, W.; Flecknell, P.; Gregory, N.; Hackbarth, H. Recommendations for euthanasia of experimental animals: Part 1. Lab. Anim. 1996, 30, 293–316. [Google Scholar] [CrossRef]
- Cañon Jones, H.A.; Hansen, L.A.; Noble, C.; Damsgard, B.; Broom, D.M.; Pearce, G.P. Social network analysis of behavioural interactions influencing fin damage development in Atlantic salmon (Salmo salar) during feed-restriction. Appl. Anim. Behav. Sci. 2010, 127, 139–151. [Google Scholar] [CrossRef]
- Vargas-Chacoff, L.; Arjona, F.J.; Polakof, S.; del Río, M.P.M.; Soengas, J.L.; Mancera, J.M. Interactive effects of environmental salinity and temperature on metabolic responses of gilthead sea bream Sparus aurata. Comp. Biochem. Phys. A 2009, 154, 417–424. [Google Scholar] [CrossRef]
- Vargas-Chacoff, L.; Moneva, F.; Oyarzún, R.; Martínez, D.; Muñoz, J.L.P.; Bertrán, C.; Mancera, J.M. Environmental salinity-modified osmoregulatory response in the sub-Antarctic notothenioid fish Eleginops maclovinus. Polar Biol. 2014, 37, 1235–1245. [Google Scholar] [CrossRef]
- Vargas-Chacoff, L.; Saavedra, E.; Oyarzún, R.; Martínez-Montaño, E.; Pontigo, J.P.; Yáñez, A.; Ruiz-Jarabo, I.; Mancera, J.M.; Ortiz, E.; Bertrán, C. Effects on the metabolism, growth, digestive capacity and osmoregulation of juvenile of Sub-Antarctic Notothenioid fish Eleginops maclovinus acclimated at different salinities. Fish Physiol. Biochem. 2015. [Google Scholar] [CrossRef]
- Quinn, G.P.; Keough, M.J. Experimental Design and Data Analysis for Biologists; Cambridge University Press: Cambridge, UK, 2002; p. 537. [Google Scholar]
- Stejskal, V.; PoLicAr, T.; Kristan, J.; Kouril, J.; Hamackova, J. Fin condition in intensively cultured Eurasian perch (Perca fluviatilis). Folia Zool. 2011, 60, 122–128. [Google Scholar] [CrossRef]
- Pelis, R.M.; McCormick, S.D. Fin development in stream- and hatchery-reared Atlantic salmon. Aquaculture 2003, 220, 525–536. [Google Scholar] [CrossRef]
- Turnbull, J.F.; Richards, R.H.; Robertson, D.A. Gross, histological and scanning electron microscopic appearance of dorsal fin rot in farmed Atlantic salmon, Salmo salar L.; parr. J. Fish Dis. 1996, 19, 415–427. [Google Scholar] [CrossRef]
- Brockmark, S.; Neregard, L.; Bohlin, T.; Björnsson, B.T.; Johnsson, J.I. Effects of rearing density and structural complexity on the preand postrelease performance of Atlantic salmon. Trans. Am. Fish Soc. 2007, 136, 1453–1462. [Google Scholar] [CrossRef]
- MacLean, A.; Metcalfe, N.B.; Mitchell, D. Alternative competitive strategies in juvenile Atlantic salmon (Salmo salar): Evidence from fin damage. Aquaculture 2000, 184, 291–302. [Google Scholar] [CrossRef]
- Udomkusonsri, P.; Noga, E.J.; Monteiro-Riviere, N.A. Pathogenesis of acute ulceration response (AUR) in hybrid striped bass. Dis. Aquat. Org. 2004, 61, 199–213. [Google Scholar] [CrossRef]
- Mustafa, A.; MacWilliams, C.; Fernandez, N.; Matchett, K.; Conboy, G.A.; Burka, J.F. Effects of sea lice (Lepeophtheirus salmonis Kroyer, 1837) infestation on macrophage functions in Atlantic salmon (Salmo salar L.). Fish Shellfish Immunol. 2000, 10, 47–59. [Google Scholar] [CrossRef] [PubMed]
- Sutherland, B.; Koczka, K.; Yasuike, M.; Jantzen, S.; Yazawa, R.; Koop, B.; Jones, S. Comparative transcriptomics of Atlantic Salmo salar, chum Oncorhynchus keta and pink salmon O. gorbuscha during infections with salmon lice Lepeophtheirus salmonis. BMC Genom. 2014, 15, 200. [Google Scholar] [CrossRef] [PubMed]
- Dawson, L.; Pike, A.; Houlihan, D.; McVicar, A. Comparison of the susceptibility of sea trout (Salmo trutta L.) and Atlantic salmon (Salmo salar L.) to sea lice (Lepeophtheirus salmonis (Krøyer, 1837)) infections. ICES J. Mar. Sci. 1997, 54, 1129–1139. [Google Scholar] [CrossRef]
- Tully, O.; Poole, W.R.; Whelan, K.F. Infestation parameters for Lepeophtheirus salmonis (Kroyer) (Copepoda: Caligidae) parasitic on sea trout, Salmo trutta L.; off the west coast of Ireland during 1990 and 1991. Aquac. Res. 1993, 24, 545–555. [Google Scholar] [CrossRef]
- Treasurer, J.W.; Bravo, S. The spatial distribution patterns of Caligus rogercresseyi and C. elongatus on Atlantic salmon hosts (Salmo salar). Aquaculture 2011, 320, 154–158. [Google Scholar] [CrossRef]
- Wooten, R.; Smith, J.W.; Needham, E.A. Aspects of the biology of the parasitic copepods Lepeophtheirus salmonis and Caligus elongatus on farmed salmonids, and their treatment. Proc. R. Soc. Edinb. B Biol. Sci. 1982, 81, 185–197. [Google Scholar] [CrossRef]
- Kabata, Z. Mouth and mode of feeding of Caligidae (Copepoda), parasites of fishes, as determined by light and scanning electron microscopy. J. Fish. Res. Board Can. 1974, 31, 1583–1588. [Google Scholar] [CrossRef]
- Johnson, S.; Albright, L. Comparative susceptibility and histopathology oi the response of naive Atlantic, chinook and coho salmon to experimental infection with Lepeophtheirus salmonis (Copepoda: Caligidae). Dis. Aquat. Org. 1992, 14, 179–193. [Google Scholar] [CrossRef]
- Lindsey, C. Form, function and locomotory habits in fish. In Fish physiology: Locomotion; Hoar, W., Randall, D., Eds.; Academic Press: New York, NY, USA, 1978; Volume 7, p. 576. [Google Scholar]
- Sharples, A.D.; Campin, D.N.; Evans, C.W. Fin erosion in a feral population of goldfish, Carassius auratus (L.), exposed to bleached kraft mill effluent. J. Fish Dis. 1994, 17, 483–493. [Google Scholar] [CrossRef]
- Loot, G.; Poulet, N.; Reyjol, Y.; Blanchet, S.; Lek, S. The effects of the ectoparasite Tracheliastes polycolpus (Copepoda: Lernaeopodidae) on the fins of rostrum dace (Leuciscus leuciscus burdigalensis). Parasitol. Res. 2004, 94, 16–23. [Google Scholar] [CrossRef]
- Branson, E. Fish Welfare; Blackwell Publishing: Hoboken, NJ, USA, 2008. [Google Scholar]
- Becerra, J.; Montes, G.; Bexiga, S.; Junqueira, L. Structure of the tail fin in teleosts. Cell Tissue Res. 1983, 230, 127–137. [Google Scholar] [CrossRef]
| dpi | Cop | Ch I–II | Ch III–IV | AF | AM |
|---|---|---|---|---|---|
| 1 | 825 (100) | 0 | 0 | 0 | 0 |
| 8 | 0 | 596 (100) | 0 | 0 | 0 |
| 16 | 0 | 0 | 482 (97) | 10 (1) | 20 (2) |
| 22 | 0 | 0 | 0 | 184 (46) | 218 (54) |
| dpi | Tank 1 | Tank 2 | Tank 3 | Tank 4 | Tank Mean |
|---|---|---|---|---|---|
| 1 | 15.6 (± 6.73) | 29.8 (± 22.35) | 32.0 (± 25.10) | 87.6 (± 30.05) | 41.3 (±31.74) |
| 8 | 8.2 (± 4.87) | 18.4 (± 5.59) | 32.0 (± 15.02) | 60.2 (± 6.83) | 29.8 (±22.55) |
| 16 | 8.4 (± 5.41) | 14.6 (± 6.54) | 34.4 (± 17.10) | 44.6 (± 20.08) | 25.5 (±16.88) |
| 22 | 2.6 (± 2.61) | 10 (± 4.64) | 22.2 (± 10.28) | 44 (± 21.97) | 19.8 (±18.10) |
| Relative Fin Index (%) | Control | Lice Infested |
|---|---|---|
| Dorsal fin | 7.9 (± 3.5) | 8.1 (±2.3) |
| Mean Caudal fins | 19.9 (±1.1) | 19.5 (±0.9) |
| Mean Pectoral fins | 16.7 (±2.2) | 17.0 (±1.9) |
| Mean pelvic fins | 11.7 (±1.3) | 10.9 (±1.8) |
| Anal fin | 10.8 (±1.1) | 10.1 (±2.1) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
González, M.P.; Marín, S.L.; Mancilla, M.; Cañon-Jones, H.; Vargas-Chacoff, L. Fin Erosion of Salmo salar (Linnaeus 1758) Infested with the Parasite Caligus rogercresseyi (Boxshall & Bravo 2000). Animals 2020, 10, 1166. https://doi.org/10.3390/ani10071166
González MP, Marín SL, Mancilla M, Cañon-Jones H, Vargas-Chacoff L. Fin Erosion of Salmo salar (Linnaeus 1758) Infested with the Parasite Caligus rogercresseyi (Boxshall & Bravo 2000). Animals. 2020; 10(7):1166. https://doi.org/10.3390/ani10071166
Chicago/Turabian StyleGonzález, Margarita P., Sandra L. Marín, Melinka Mancilla, Hernán Cañon-Jones, and Luis Vargas-Chacoff. 2020. "Fin Erosion of Salmo salar (Linnaeus 1758) Infested with the Parasite Caligus rogercresseyi (Boxshall & Bravo 2000)" Animals 10, no. 7: 1166. https://doi.org/10.3390/ani10071166
APA StyleGonzález, M. P., Marín, S. L., Mancilla, M., Cañon-Jones, H., & Vargas-Chacoff, L. (2020). Fin Erosion of Salmo salar (Linnaeus 1758) Infested with the Parasite Caligus rogercresseyi (Boxshall & Bravo 2000). Animals, 10(7), 1166. https://doi.org/10.3390/ani10071166





