Animal-Assisted Therapy as a Non-Pharmacological Approach in Alzheimer’s Disease: A Retrospective Study
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Team
2.2. Operative Method
2.2.1. Study Design and Participants
2.2.2. Methodology
2.2.3. Measurements
2.3. Data Analysis
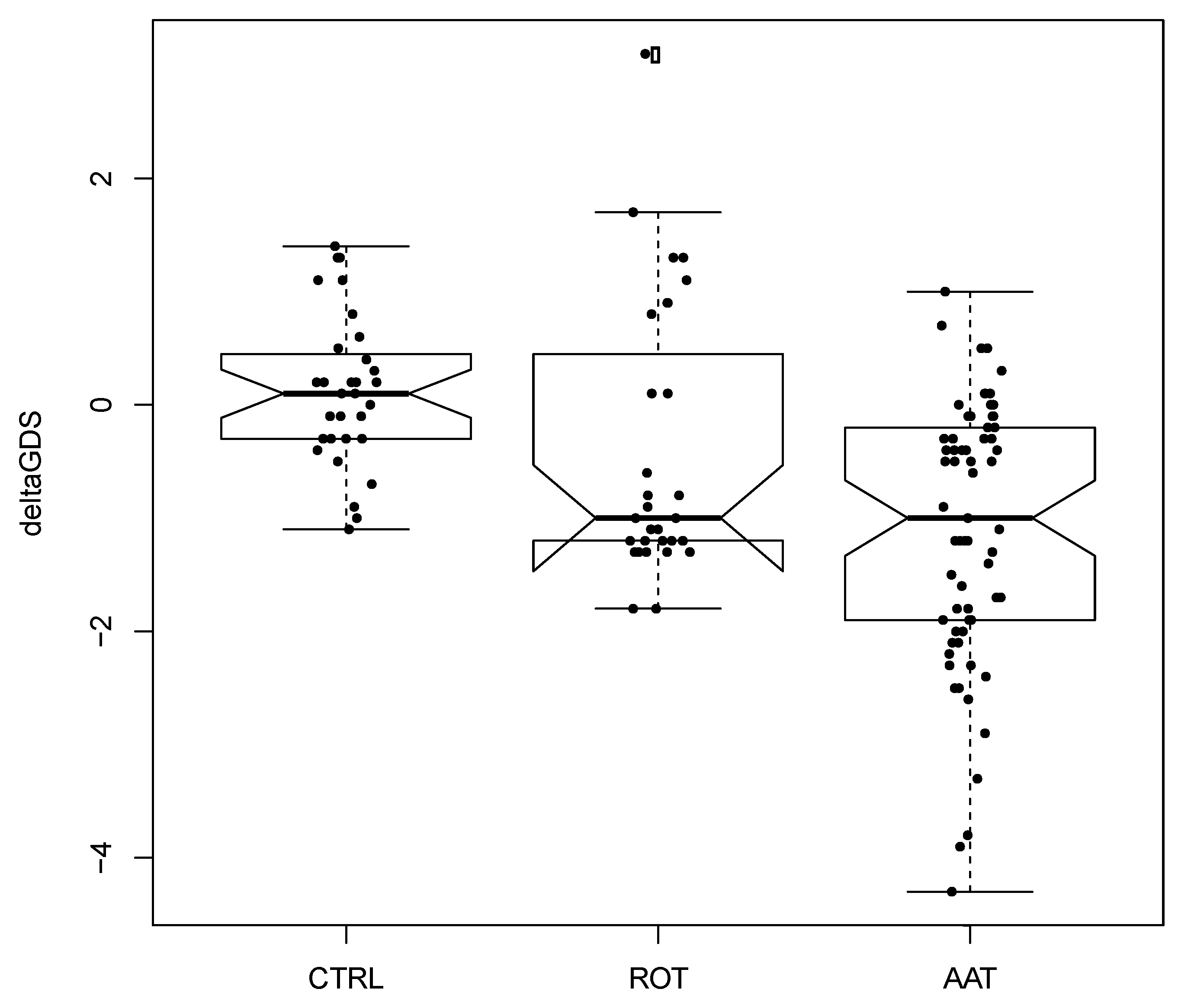
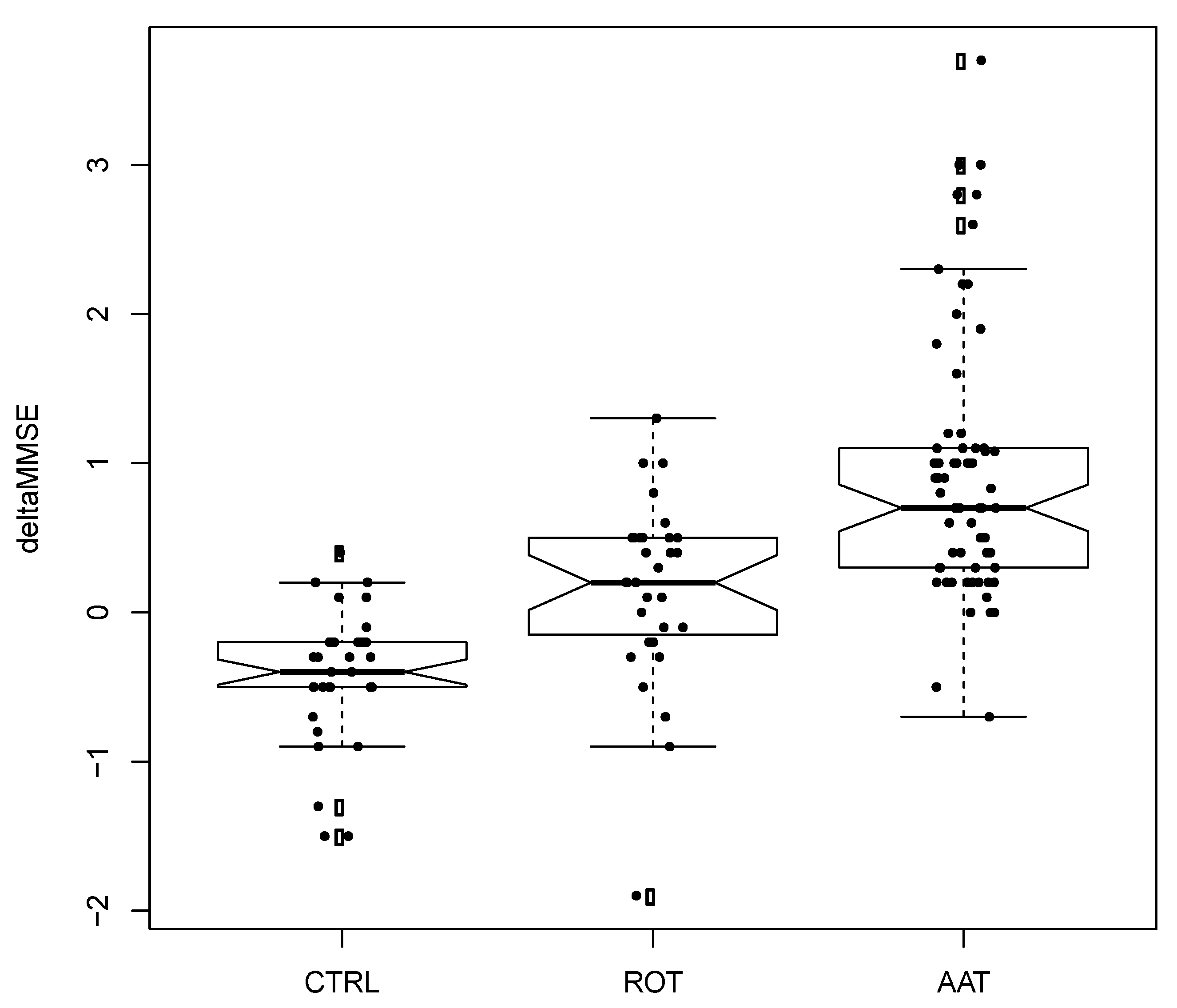
3. Results
4. Discussion
Limits
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Serrano-Pozo, A.; Frosch, M.P.; Masliah, E.; Hyman, B.T. Neuropathological Alterations in Alzheimer Disease. Cold Spring Harb. Perspect. Med. 2011, 1, a006189. [Google Scholar] [CrossRef]
- Prince, M.; Wimo, A.; Ali, G.; Guerchet, M.; Prina, A.M.; Albanese, E.; Wu, Y. Recent global trends in the prevalence and incidence of dementia, and survival with dementia. Alzheimer’s Res. Ther. 2016, 8, 23. [Google Scholar] [CrossRef]
- Day, G.; Musiek, E.; Roe, C.; Norton, J.; Goate, A.; Cruhaga, C.; Cairns, N.; Morris, J. Phenotypic similarities between late-onset autosomal dominant and sporadic Alzheimer disease: A single-family case-control study. JAMA Neurol. 2016, 73, 1125–1132. [Google Scholar] [CrossRef]
- Reitz, C.; Brayne, C.; Mayeux, R. Epidemiology of Alzheimer disease. Nat. Rew. Neurol. 2011, 7, 137–152. [Google Scholar] [CrossRef]
- Sun, B.; Li, W.; Zhu, C.; Jin, W.; Zeng, F.; Liu, Y.; Bu, X.; Zhu, J.; Yao, X.; Wang, Y. Clinical research on Alzheimer’s disease: Progress and perspectives. Neurosci. Bull. 2018, 34, 1111–1118. [Google Scholar] [CrossRef] [PubMed]
- Pietrzak, K.; Czarnecka, K.; Mikiciuk-Olasik, E.; Szymanski, P. New perspectives of Alzheimer disease diagnosis—The most popular and future methods. Med. Chem. 2018, 14, 34–43. [Google Scholar] [CrossRef]
- Apostolova, L.G. Alzheimer Disease. Continuum (Minneap. Minn.) 2016, 22, 419–434. [Google Scholar] [CrossRef] [PubMed]
- Menna, L.F.; Santaniello, A.; Amato, A.; Ceparano, G.; Di Maggio, A.; Sansone, M.; Formisano, P.; Cimmino, I.; Perruolo, G.; Fioretti, A. Changes of Oxytocin and Serotonin Values in Dialysis Patients after Animal Assisted Activities (AAAs) with a Dog- A Preliminary Study. Animals 2019, 9, 526. [Google Scholar] [CrossRef]
- Gan, G.Z.H.; Hill, A.; Hill, A.M.; Yeung, P.; Keesing, S.; Netto, J.A. Pet ownership and its influence on mental health in older adults. Aging Ment. Health 2019, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Sollami, A.; Gianferrari, E.; Alfieri, E.; Artioli, G.; Tuffarelli, C. Pet therapy: An effective strategy to care for the elderly? An experimental study in a nursing home. Acta Biomed. 2017, 88, 25–31. [Google Scholar] [CrossRef]
- Menna, L.F.; Santaniello, A.; Gerardi, F.; Di Maggio, A.; Milan, G. Evaluation of the efficacy of animal-assisted therapy based on the reality orientation therapy protocol in Alzheimer’s disease patients: A pilot study. Psychogeriatrics 2016, 16, 240–246. [Google Scholar] [CrossRef] [PubMed]
- Menna, L.F.; Santaniello, A.; Federica, G.; Sansone, M.; Di Maggio, A.; Di Palma, A.; Perruolo, G.; D’Esposito, V.; Formisano, P. Efficacy of animal-assisted therapy adapted to reality orientation therapy: Measurement of salivary cortisol. Psycogeriatrics 2019, 19, 510–512. [Google Scholar] [CrossRef] [PubMed]
- Nordgren, L.; Engstrom, G. Effects of dog-assisted intervention on behavioural and psychological symptoms of dementia. Nurs. Older People 2014, 26, 31–33. [Google Scholar] [CrossRef]
- Friedmann, E.; Galik, E.; Thomas, S.A.; Hall, S.P.; Chung, S.Y.; McCune, S. Evaluation of a pet-assisted living intervention for improving functional status in assisted living residents with mild to moderate cognitive impairment: A pilot study. Am. J. Alzheimer’s Dis. Other Demen. 2015, 30, 276–289. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Valdeón, L.; Fernández-Martínez, E.; Loma-Ramos, S.; López-Alonso, A.I.; Darkistade, E.B.; Ladera, V. Canine-assisted therapy and quality of life in people with Alzheimer-type dementia: Pilot study. Front. Psychol. 2019, 10, 1332. [Google Scholar] [CrossRef] [PubMed]
- Wesenberg, S.; Mueller, C.; Nestmann, F.; Holthoff-Detto, V. Effects of an animal-assisted intervention on social behaviour, emotions, and behavioural and psychological symptoms in nursing home residents with dementia. Psychogeriatrics 2019, 19, 219–227. [Google Scholar] [CrossRef]
- Bono, A.V.; Benvenuti, C.; Buzzi, M.; Ciatti, R.; Chiarelli, V.; Chiambretto, P.; Morelli, C.; Pinciroli, M.; Pini, A.; Prestigiacomo, T.; et al. Effects of animal assisted therapy (AAT) carried out with dogs on the evolution of mild cognitive impairment. G Gerontol. 2015, 63, 32–36. [Google Scholar]
- Olsen, C.; Pedersen, I.; Bergland, A.; Enders-Slegers, M.; Ihlebæk, C. Effect of animal-assisted activity on balance and quality of life in home-dwelling persons with dementia. Geriatr. Nurs. 2016, 37, 284–291. [Google Scholar] [CrossRef]
- Menna, L.F. The Scientific Approach to Pet Therapy. In The Method and Training According to the Federiciano Model, 1st ed.; University of Naples Federico II: Naples, Italy, 2016; ISBN 979-12-200-3991-8. [Google Scholar]
- Menna, L.F.; Santaniello, A.; Todisco, M.; Amato, A.; Borrelli, L.; Scandurra, C.; Fioretti, A. The human-animal relationship as the focus of animal-assisted interventions: A one-health approach. Int. J. Environ. Res. Public Health 2019, 16, 3660. [Google Scholar] [CrossRef] [PubMed]
- Santaniello, A.; Sansone, M.; Fioretti, A.; Menna, L.F. Systematic Review and Meta-Analysis of the Occurrence of ESKAPE Bacteria Group in Dogs, and the Related Zoonotic Risk in Animal-Assisted Therapy, and in Animal-Assisted Activity in the Health Context. Int. J. Environ. Res. Public Health 2020, 17, 3278. [Google Scholar] [CrossRef]
- Maurelli, M.P.; Santaniello, A.; Fioretti, A.; Cringoli, G.; Rinaldi, L.; Menna, L.F. The presence of Toxocara eggs on dog’s fur as potential zoonotic risk in animal-assisted interventions: A systematic review. Animals 2019, 19, 827. [Google Scholar] [CrossRef] [PubMed]
- Gerardi, F.; Santaniello, A.; Del Prete, L.; Maurelli, M.P.; Menna, L.F.; Rinaldi, L. Parasitic infections in dogs involved in animal-assisted interventions. Ital. J. Anim. Sci. 2018, 1, 269–272. [Google Scholar] [CrossRef]
- Ghasemzadeh, I.; Namazi, S.H. Review of bacterial and viral zoonotic infections transmitted by dogs. J. Med. Life 2015, 8, 1–5. [Google Scholar] [PubMed]
- Zanetti, O.; Frisoni, G.B.; De Leo, D.; Dello Buono, M.; Bianchetti, A.; Trabucchi, M. Reality orientation therapy in Alzheimer’s disease: Useful or not? A controlled study. Alzheimer Dis. Assoc. Disord. 1995, 9, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Zanetti, O.; Calabria, M.; Cotelli, M. Effectiveness of the association of donepezil and Reality Orientation Therapy. G Gerontol. 2004, 52, 408–411. [Google Scholar]
- Aguirre, E.; Woods, R.; Spector, A.; Orrell, M. Cognitive stimulation for dementia: A systematic review of the evidence of effectiveness from randomised controlled trials. Ageing Res. Rev. 2013, 12, 253–262. [Google Scholar] [CrossRef]
- Folstein, M.F.; Folstein, S.E. ‘Mini-Mental Stat’: A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Mimura, M.; Komatsu, S. Cognitive rehabilitation and cognitive training for mild dementia. Psychogeriatrics 2007, 7, 137–143. [Google Scholar] [CrossRef]
- Aikman, G.G.; Oehlert, M.E. Geriatric depression scale: Long form versus short form. Clin. Gerontol. 2001, 22, 63–70. [Google Scholar] [CrossRef]
- O’Connell, N.S.; Dai, L.; Jiang, Y.; Speiser, J.L.; Ward, R.; Wei, W.; Carroll, R.; Gebregziabher, M. Methods for Analysis of Pre-Post Data in Clinical Research: A Comparison of Five Common Methods. J. Biom. Biostat. 2017, 8, 1–8. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. Available online: https://www.R-project.org/ (accessed on 5 May 2020).
- Heiberger, R.M. HH: Statistical Analysis and Data Display: Heiberger and Holland, R package version 3.1-37; 2019. Available online: https://CRAN.R-project.org/package=HH (accessed on 5 May 2020).
- Kanamori, M.; Suzuki, M.; Yamamoto, K.; Kanda, M.; Matsui, Y.; Kojima, E.; Fukawa, H.; Sugita, T.; Oshiro, H. A day care program and evaluation of animal-assisted therapy (AAT) for the elderly with senile dementia. Am. J. Alzheimer’s Dis. Other Dement. 2001, 16, 234–239. [Google Scholar] [CrossRef] [PubMed]
- Moretti, F.; De Ronchi, D.; Bernabei, V.; Marchetti, L.; Ferrari, B.; Forlani, C.; Negretti, F.; Sacchetti, C.; Atti, A.R. Pet therapy in elderly patients with mental illness. Psychogeriatrics 2011, 11, 125–129. [Google Scholar] [CrossRef] [PubMed]
- Motomura, N.; Yagi, T.; Ohyama, H. Animal assisted therapy for people with dementia. Psychogeriatrics 2004, 4, 40–42. [Google Scholar] [CrossRef]
- Thodberg, K.; Sorensen, L.U.; Christensen, J.W.; Poulsen, P.H.; Houbak, B.; Damgaard, V.; Keseler, I.; Edwards, D.; Vibedech, P.B. Therapeutic effects of dog visits in nursing homes for the elderly. Psychogeriatrics 2016, 16, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Marks, G.; McVilly, K. Trained assistance dogs for people with dementia: A systematic review. Psychogeriatrics 2020. [Google Scholar] [CrossRef]
| Formal ROT | AAT Session | Duration | |
|---|---|---|---|
| Step 1 | 1. Structuring the setting 2. Presenting the therapist/patient 3. Stimulating cognitive function | 1. Structuring the setting 2. Presenting the zootherapist/dog/patient 3. Stimulating cognitive function through repeated requests for information about the dog (e.g., name, breed, age, and sex) | 10′ |
| Step 2 | 1. Temporal orientation (day, month, year, and season) 2. Spatial orientation (place, structure, floor, room, city, country, and region) 3. Stimulation of memory | 1. Temporal orientation (day, month, year, and season) 2. Spatial orientation (place, structure, floor, room, city, country, and region); 5 min of play structured activities with the dog (e.g., hide the ball) 3. Stimulation of memory by telling a story about their own pets | 15′ |
| Step 3 | 1. Stimulation of memory (attention) 2. Understanding of language (story) | 1. Structured play/interaction with the dog (attention: fetch, hide the ball, caring for the dog) 2. Understanding of language (story: giving the dog commands and waiting for the execution of the command) | 15′ |
| Step 4 | 1. Closing speech (ritualized) | 1. Closing speech (ritualized: washing hands) | 5′ |
| Group | No. of Patients | MMSE 1 (Mean ± SD) | GDS 2 (Mean ± SD) | Age (Years) Mean ± SD 9 | |
|---|---|---|---|---|---|
| F 6 | M 7 | T0 8 | |||
| AAT 3 | 52 | 13 | 19.2 ± 2.39 | 11.8 ± 2.95 | 76.6 ± 5.0 |
| ROT 4 | 22 | 9 | 20.1 ± 0.94 | 12.4 ± 1.46 | 75.6 ± 6.0 |
| CTRL 5 | 21 | 10 | 20.4 ± 0.53 | 11.5 ± 0.73 | 75.0 ± 6.3 |
| Total | 95 | 32 | 19.7 ± 1.9 | 11.9 ± 2.3 | 76 ± 5.8 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santaniello, A.; Garzillo, S.; Amato, A.; Sansone, M.; Di Palma, A.; Di Maggio, A.; Fioretti, A.; Menna, L.F. Animal-Assisted Therapy as a Non-Pharmacological Approach in Alzheimer’s Disease: A Retrospective Study. Animals 2020, 10, 1142. https://doi.org/10.3390/ani10071142
Santaniello A, Garzillo S, Amato A, Sansone M, Di Palma A, Di Maggio A, Fioretti A, Menna LF. Animal-Assisted Therapy as a Non-Pharmacological Approach in Alzheimer’s Disease: A Retrospective Study. Animals. 2020; 10(7):1142. https://doi.org/10.3390/ani10071142
Chicago/Turabian StyleSantaniello, Antonio, Susanne Garzillo, Alessia Amato, Mario Sansone, Annalisa Di Palma, Annamaria Di Maggio, Alessandro Fioretti, and Lucia Francesca Menna. 2020. "Animal-Assisted Therapy as a Non-Pharmacological Approach in Alzheimer’s Disease: A Retrospective Study" Animals 10, no. 7: 1142. https://doi.org/10.3390/ani10071142
APA StyleSantaniello, A., Garzillo, S., Amato, A., Sansone, M., Di Palma, A., Di Maggio, A., Fioretti, A., & Menna, L. F. (2020). Animal-Assisted Therapy as a Non-Pharmacological Approach in Alzheimer’s Disease: A Retrospective Study. Animals, 10(7), 1142. https://doi.org/10.3390/ani10071142






