Efficacy of Different Drenching Regimens of Gluconeogenic Precursors during Transition Period on Body Condition Score, Production, Reproductive Performance, Subclinical Ketosis and Economics of Dairy Cows
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
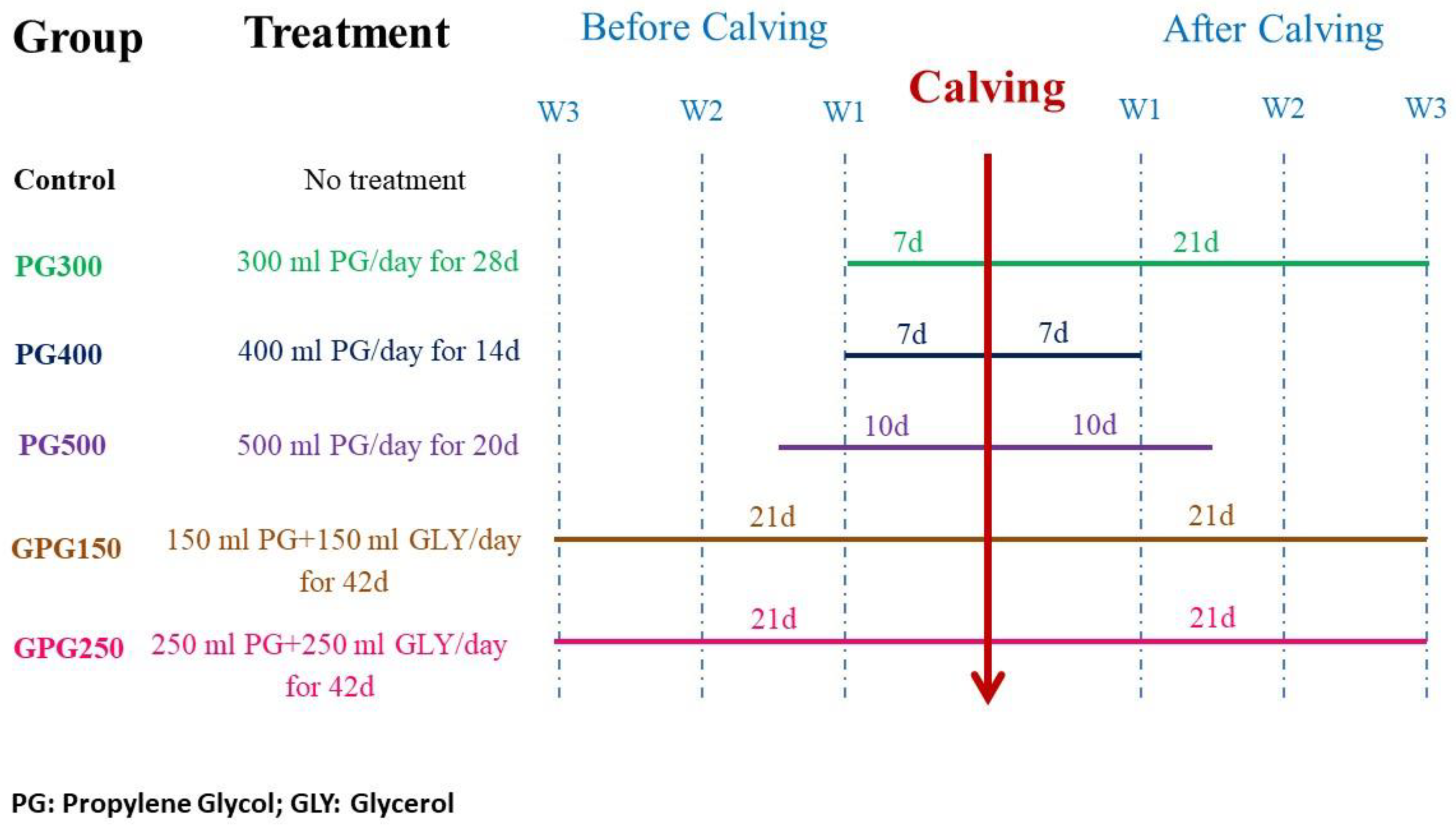
2.1. Animals, Management, Experimental Design and Treatment Regimen
2.2. Evaluation of Body Condition Score (BCS), Milk Productivity and Reproductive Traits
2.3. Blood Metabolites
2.4. Economic Assessments
2.5. Statistical Analyses
3. Results
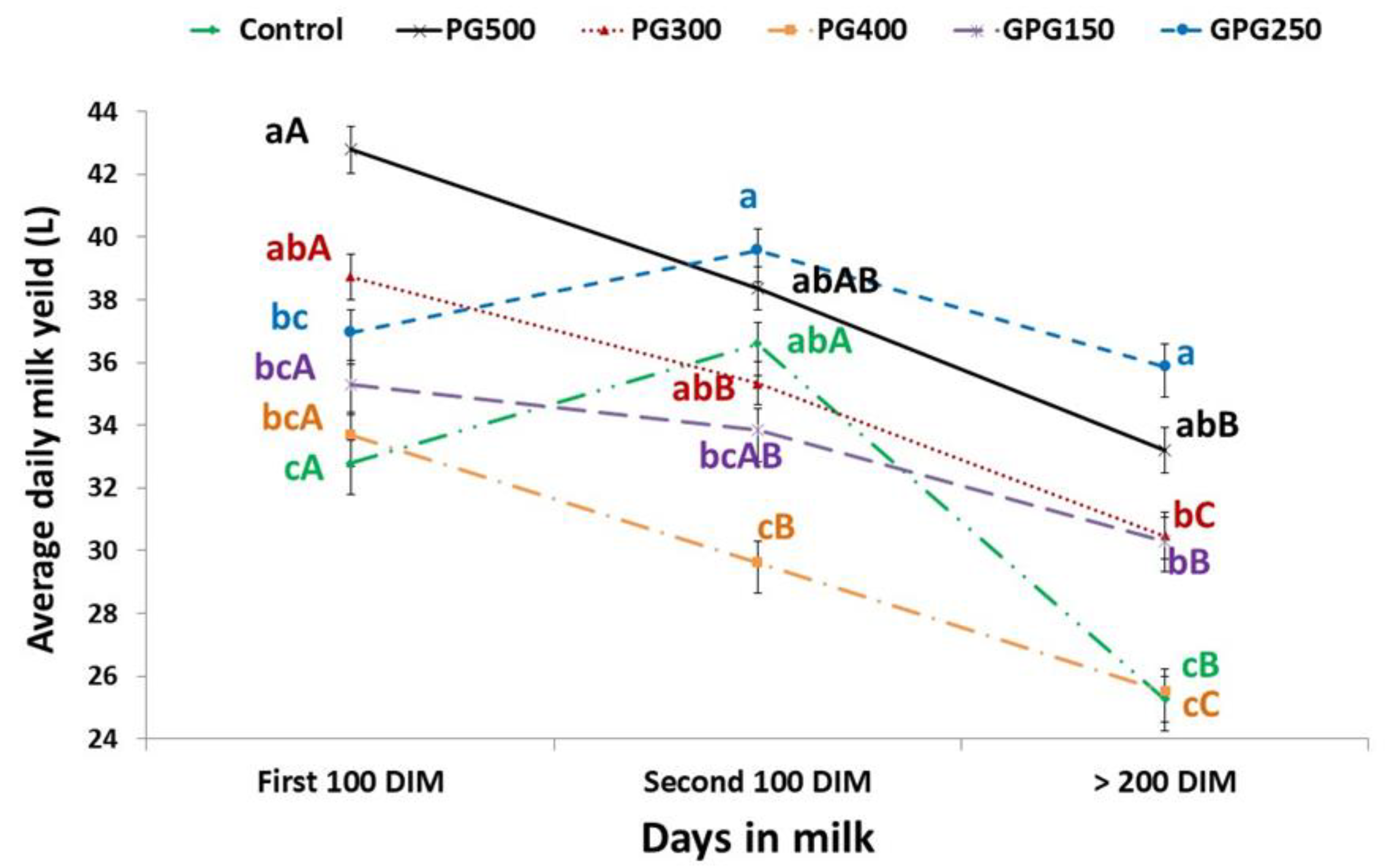
3.1. Body Condition Score (BCS), Milk Productivity and Reproductive Traits
3.2. Blood Metabolites
3.3. Economic Parameters
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Grummer, R.R. Impact of changes in organic nutrient metabolism on feeding the transition dairy cow. J. Anim. Sci. 1995, 73, 2820–2833. [Google Scholar] [CrossRef]
- Van Saun, R.J. Indicators of dairy cow transition risks: Metabolic profiling revisited. Tierarztl Prax Ausg G Grosstiere/Nutztiere 2016, 44, 118–126. [Google Scholar] [CrossRef]
- Wankhade, P.R.; Manimaran, A.; Kumaresan, A.; Jeyakumar, S.; Ramesha, K.P.; Sejian, V.; Rajendran, D.; Varghese, M.R. Metabolic and immunological changes in transition dairy cows: A review. Vet. World 2017, 10, 1367–1377. [Google Scholar] [CrossRef]
- Bauman, D.E.; Currie, W.B. Partitioning of Nutrients During Pregnancy and Lactation: A Review of Mechanisms Involving Homeostasis and Homeorhesis. J. Dairy Sci. 1980, 63, 1514–1529. [Google Scholar] [CrossRef]
- Butler, S.T.; Pelton, S.H.; Butler, W.R. Energy balance, metabolic status, and the first postpartum ovarian follicle wave in cows administered propylene glycol. J. Dairy Sci. 2006, 89, 2938–2951. [Google Scholar] [CrossRef]
- Drackley, J.K. Biology of Dairy Cows During the Transition Period: The Final Frontier? J. Dairy Sci. 1999, 82, 2259–2273. [Google Scholar] [CrossRef]
- DeFrain, J.M.; Hippen, A.R.; Kalscheur, K.F.; Jardon, P.W. Feeding Glycerol to Transition Dairy Cows: Effects on Blood Metabolites and Lactation Performance. J. Dairy Sci. 2004, 87, 4195–4206. [Google Scholar] [CrossRef]
- Chung, Y.-H.; Rico, D.; Martinez, C.; Cassidy, T.; Noirot, V.; Ames, A.; Varga, G. Effects of Feeding Dry Glycerin to Early Postpartum Holstein Dairy Cows on Lactational Performance and Metabolic Profiles. J. Dairy Sci. 2007, 90, 5682–5691. [Google Scholar] [CrossRef]
- Piantoni, P.; Allen, M.S. Evaluation of Propylene Glycol and Glycerol Infusions as Treatments for Ketosis in Dairy Cows. J. Dairy Sci. 2015, 98, 5429–5439. [Google Scholar] [CrossRef]
- Herdt, T.H.; Emery, R.S. Therapy of Diseases of Ruminant Intermediary Metabolism. Vet. Clin. N. Am. Food Anim. Pr. 1992, 8, 91–106. [Google Scholar] [CrossRef]
- Johnson, R.B. The Treatment of Ketosis with Glycerol and Propylene Glycol. Cornell Vet. 1954, 44, 6–21. [Google Scholar]
- McArt, J.; Nydam, D.; Ospina, P.; Oetzel, G. A Field Trial on the Effect of Propylene Glycol on Milk Yield and Resolution of Ketosis in Fresh Cows Diagnosed with Subclinical Ketosis. J. Dairy Sci. 2011, 94, 6011–6020. [Google Scholar] [CrossRef] [PubMed]
- McArt, J.; Nydam, D.; Oetzel, G. A Field Trial on the Effect of Propylene Glycol on Displaced Abomasum, Removal from Herd, and Reproduction in Fresh Cows Diagnosed with Subclinical Ketosis. J. Dairy Sci. 2012, 95, 2505–2512. [Google Scholar] [CrossRef] [PubMed]
- Gordon, J.L.; Leblanc, S.; Duffield, T.F. Ketosis Treatment in Lactating Dairy Cattle. Vet. Clin. N. Am. Food Anim. Pr. 2013, 29, 433–445. [Google Scholar] [CrossRef] [PubMed]
- Rizos, D.; Kenny, D.A.; Griffin, W.; Quinn, K.M.; Duffy, P.; Mulligan, F.J.; Roche, J.F.; Boland, M.P.; Lonergan, P. The effect of feeding propylene glycol to dairy cows during the early postpartum period on follicular dynamics and on metabolic parameters related to fertility. Theriogenology 2008, 69, 688–699. [Google Scholar] [CrossRef] [PubMed]
- Osman, M.A.; Allen, P.S.; Mehyar, N.A.; Bobe, G.; Coetzee, J.F.; Koehler, K.J.; Beitz, D.C. Acute Metabolic Responses of Postpartal Dairy Cows to Subcutaneous Glucagon Injections, Oral Glycerol, or Both. J. Dairy Sci. 2008, 91, 3311–3322. [Google Scholar] [CrossRef]
- Nielsen, N.I.; Ingvartsen, K.L. Propylene glycol for dairy cows: A review of the metabolism of propylene glycol and its effects on physiological parameters, feed intake, milk production and risk of ketosis. Anim. Feed Sci. Technol. 2004, 115, 191–213. [Google Scholar] [CrossRef]
- Trabue, S.; Scoggin, K.; Tjandrakusuma, S.; Rasmussen, M.A.; Reilly, P.J. Ruminal Fermentation of Propylene Glycol and Glycerol. J. Agric. Food Chem. 2007, 55, 7043–7051. [Google Scholar] [CrossRef]
- Studer, V.A.; Grummer, R.R.; Bertics, S.J.; Reynolds, C.K. Effect of Prepartum Propylene Glycol Administration on Periparturient Fatty Liver in Dairy Cows. J. Dairy Sci. 1993, 76, 2931–2939. [Google Scholar] [CrossRef]
- Christensen, J.O.; Grummer, R.R.; Rasmussen, F.E.; Bertics, S.J. Effect of method of delivery of propylene glycol on plasma metabolites of feed-restricted cattle. J. Dairy Sci. 1997, 80, 563–568. [Google Scholar] [CrossRef]
- Pickett, M.M.; Piepenbrink, M.S.; Overton, T.R. Effects of Propylene Glycol or Fat Drench on Plasma Metabolites, Liver Composition, and Production of Dairy Cows during the Periparturient Period1. J. Dairy Sci. 2003, 86, 2113–2121. [Google Scholar] [CrossRef]
- Roche, J.R.; Friggens, N.C.; Kay, J.K.; Fisher, M.W.; Stafford, K.J.; Berry, D.P. Invited review: Body condition score and its association with dairy cow productivity, health, and welfare. J. Dairy Sci. 2009, 92, 5769–5801. [Google Scholar] [CrossRef]
- McMurray, C.H.; Blanchflower, W.J.A.; Rice, D. Automated Kinetic Method for D-3-Hydroxybutyrate in Plasma or Serum. Clin. Chem. 1984, 30, 421–425. [Google Scholar] [CrossRef]
- Ospina, P.A.; Nydam, D.V.; Stokol, T.; Overton, T.R. Associations of elevated nonesterified fatty acids and β-hydroxybutyrate concentrations with early lactation reproductive performance and milk production in transition dairy cattle in the northeastern United States. J. Dairy Sci. 2010, 93(4), 1596–1603. [Google Scholar] [CrossRef]
- Trinder, P. Determination of blood glucose using an oxidase-peroxidase system with a non-carcinogenic chromogen. J. Clin. Pathol. 1969, 22, 158–161. [Google Scholar] [CrossRef] [PubMed]
- Mair, B.; Drillich, M.; Klein-Jöbst, D.; Kanz, P.; Borchardt, S.; Meyer, L.; Schwendenwein, I.; Iwersen, M. Glucose concentration in capillary blood of dairy cows obtained by a minimally invasive lancet technique and determined with three different hand-held devices. BMC Vet. Res. 2016, 12, 34. [Google Scholar] [CrossRef] [PubMed]
- Jagannatha, S.; Keown, J.; Van Vleck, L.D.; Lewis, A. Effects of Days Open Days Dry, and Season of Freshening on Income over Feed Cost and 305-Day Mature Equivalent Milk Yield, for Three Different Production Levels. Prof. Anim. Sci. 1995, 11, 223–229. [Google Scholar] [CrossRef]
- Fisher, L.; Erfle, J.; Lodge, G.; Sauer, F. Effects of propylene glycol or glycerol supplementation of the diet of dairy cows on feed intake, milk yield and composition, and incidence of ketosis. Can. J. Anim. Sci. 1973, 53, 289–296. [Google Scholar] [CrossRef]
- Sauer, F.D.; Erfle, J.D.; Fisher, L.J. Propylene glycol and glycerol as a feed additive for lactating dairy cows: An evaluation of blood metabolite parameters. Can. J. Anim. Sci. 1973, 53, 265–271. [Google Scholar] [CrossRef]
- Kristensen, N.B.; Raun, B.M.L. Ruminal and Intermediary Metabolism of Propylene Glycol in Lactating Holstein Cows. J. Dairy Sci. 2007, 90, 4707–4717. [Google Scholar] [CrossRef]
- Kass, M.; Ariko, T.; Samarütel, J.; Ling, K.; Jaakson, H.; Kaart, T.; Arney, D.; Kärt, O.; Ots, M. Long-term oral drenching of crude glycerol to primiparous dairy cows in early lactation. Anim. Feed Sci. Technol. 2013, 184, 58–66. [Google Scholar] [CrossRef]
- Kalyesubula, M.; Rosov, A.; Alon, T.; Moallem, U.; Dvir, H. Intravenous Infusions of Glycerol Versus Propylene Glycol for the Regulation of Negative Energy Balance in Sheep: A Randomized Trial. Animals 2019, 9, 731. [Google Scholar] [CrossRef] [PubMed]
- Zachut, M.; Moallem, U. Consistent magnitude of postpartum body weight loss within cows across lactations and the relation to reproductive performance. J. Dairy Sci. 2017, 100, 3143–3154. [Google Scholar] [CrossRef]
- Carvalho, P.; Souza, A.H.; Amundson, M.C.; Hackbart, K.S.; Fuenzalida, M.; Herlihy, M.M.; Ayres, H.; Dresch, A.R.; Vieira, L.; Guenther, J.N.; et al. Relationships between fertility and postpartum changes in body condition and body weight in lactating dairy cows. J. Dairy Sci. 2014, 97, 3666–3683. [Google Scholar] [CrossRef]
- Shin, E.-K.; Jeong, J.-K.; Choi, I.-S.; Kang, H.-G.; Hur, T.-Y.; Jung, Y.-H.; Kim, I.-H. Relationships Among Ketosis, Serum Metabolites, Body Condition, and Reproductive Outcomes in Dairy Cows. Theriogenology 2015, 84, 252–260. [Google Scholar] [CrossRef]
- Saby-Chaban, C.; Zhang, W.; Fournier, R.; Servien, R.; Villa-Vialaneix, N.; Corbière, F.; Chastant-Maillard, S. Progesterone and betahydroxybutyrate in line measurements for a better description and understanding of Holstein cows fertility in field conditions. In Proceedings of the 8th European Conference on Precision Livestock Farming (ECPLF), Nantes, France, 12–14 September 2017; p. 7. [Google Scholar]
- Miyoshi, S.; Pate, J.L.; Palmquist, D.L. Effects of propylene glycol drenching on energy balance, plasma glucose, plasma insulin, ovarian function and conception in dairy cows. Anim. Reprod. Sci. 2001, 68, 29–43. [Google Scholar] [CrossRef]
- Berlinguer, F.; Gonzalez-Bulnes, A.; Contreras-Solis, I.; Spezzigu, A.; Torres-Rovira, L.; Succu, S.; Naitana, S.; Leoni, G.G. Glucogenic supply increases oocyte developmental competence in sheep. Reprod. Fertil. Dev. 2012, 24, 1055–1062. [Google Scholar] [CrossRef]
- Lucy, M.C.; Escalante, R.C.; Keisler, D.H.; Lamberson, W.R.; Mathew, D.J. Short communication: Glucose infusion into early postpartum cows defines an upper physiological set point for blood glucose and causes rapid and reversible changes in blood hormones and metabolites. J. Dairy Sci. 2013, 96, 5762–5768. [Google Scholar] [CrossRef]
- Melendez, P.; Severino, K.; Marin, M.P.; Duchens, M. The effect of a product with three gluconeogenic precursors during the transition period on blood metabolites and milk yield in Chilean Holstein cattle. J. Appl. Anim. Res. 2018, 46, 613–617. [Google Scholar] [CrossRef]
- Cozzi, G.; Berzaghi, P.; Gottardo, F.; Gabai, G.; Andrighetto, I. Effects of feeding propylene glycol to mid-lactating dairy cows. Anim. Feed Sci. Technol. 1996, 64, 43–51. [Google Scholar] [CrossRef]
- Omazic, A.W.; Kronqvist, C.; Zhongyan, L.; Martens, H.; Holtenius, K. The Fate of Glycerol Entering the Rumen of Dairy Cows and Sheep. J. Anim. Physiol. Anim. Nutr. 2014, 99, 258–264. [Google Scholar] [CrossRef] [PubMed]
- Ferraro, S.; Mendoza, G.; Miranda, A.; Gutierrez, C. In vitro ruminal fermentation of glycerol, propylene glycol and molasses combined with forages and their effect on glucose and insulin blood plasma concentrations after an oral drench in sheep. Anim. Feed Sci. Technol. 2016, 213, 74–80. [Google Scholar] [CrossRef]
- Kelton, D.F.; Lissemore, K.D.; Martin, R.E. Recommendations for recording and calculating the incidence of selected clinical diseases of dairy cattle. J. Dairy Sci. 1998, 81, 2502–2509. [Google Scholar] [CrossRef]
- Pechova, A.; Pecinka, P.; Kudrnacova, J.; Pavlata, L. The comparison of propylene glycol and glycerol as feed additives in early lactation of high producing dairy cows. J. Anim. Feed Sci. 2014, 23, 285–292. [Google Scholar] [CrossRef]
| Ingredient as Fed (Kg/Head/Day) | Close Up | Milking |
|---|---|---|
| Alfalfa hay | 2.25 | 4.00 |
| Berseem fresh | 5.00 | 18.00 |
| Corn Silage | 16.00 | 15.25 |
| Wheat straw | - | 0.75 |
| Total Forage | 23.25 | 38.00 |
| Ground yellow corn | 3.50 | 7.00 |
| Soybean 48% | 0.20 | 3.00 |
| Corn gluten feed | 1.50 | 3.50 |
| Rice Polish | 0.70 | 2.00 |
| Wheat bran | 0.25 | 1.90 |
| Molasses, cane | - | 0.50 |
| Magnapac | - | 0.45 |
| Limestone | - | 0.090 |
| Sodium bicarbonate | - | 0.22 |
| Mentrax (Chelated) | 0.01 | 0.001 |
| NEW T-NIL® Dry 1 (Antimycotoxin) | 0.03 | 0.04 |
| Premix 2 | 0.03 | 0.1 |
| Nutrients (%) according to feed analysis | ||
| Crude Protein | 15.2 | 18.2 |
| NDF 3 | 34.43 | 33.71 |
| ADF 4 | 23.01 | 17.63 |
| Cellulose | 18.6 | 15.2 |
| Hemicellulose | 11.42 | 16.08 |
| Lignin | 4.41 | 2.43 |
| NDICP 5 | 2.84 | 2.26 |
| ADICP 6 | 1.75 | 0.71 |
| Crude Fat | 2.29 | 3.88 |
| Ash | 10.96 | 9.03 |
| NFC 7 | 34.38 | 34.83 |
| NEl(Mcal/kg) 8 | 1.5 | 1.64 |
| Item | Control | Gluconeogenic Precursors Supplementation | SEM | p-Value * | ||||
|---|---|---|---|---|---|---|---|---|
| PG300 | PG400 | PG500 | GPG150 | GPG250 | ||||
| BCS 0 DIM | 3.56 A | 3.53 A | 3.63 A | 3.59 A | 3.69 A | 3.61 A | 0.03 | 0.116 |
| BCS 30 DIM | 2.45 d,B | 2.62 b,c,B | 2.55 c,B | 2.77 a,b,B | 2.78 a,b,B | 2.81 a,B | 0.02 | 0.008 |
| BCS 75 DIM | 2.42 b,B | 2.52 a,b,B | 2.50 a,b,B | 2.59 a,b,B | 2.56 a,b,B | 2.65 a,B | 0.02 | 0.011 |
| SEM | 0.07 | 0.08 | 0.06 | 0.09 | 0.10 | 0.06 | ||
| p-Value ** | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | ||
| Item | Control | Gluconeogenic Precursors Supplementation | SEM | p-Value * | ||||
|---|---|---|---|---|---|---|---|---|
| PG300 | PG400 | PG500 | GPG150 | GPG250 | ||||
| LL | 377.83 a | 342.31 a,b | 346.39 a,b | 308.10 b,c | 335.67 b,c | 299.50 c | 5.69 | <0.001 |
| TMY | 11433.14 | 11744.65 | 10069.55 | 11701.63 | 11030.11 | 11225.26 | 213.18 | 0.216 |
| 305 MY | 9229.30 b,c | 10464.55 a,b | 8866.35 c | 11583.90 a | 10022.30 b,c | 11431.14 a | 210.78 | <0.001 |
| DMY | 30.26 b,c | 34.31 a,b | 29.07 c | 37.98 a | 32.86 b,c | 37.48 a | 0.69 | <0.001 |
| Item | Control | Gluconeogenic Precursors Supplementation | SEM | p-Value * | ||||
|---|---|---|---|---|---|---|---|---|
| PG300 | PG400 | PG500 | GPG150 | GPG250 | ||||
| DIMFB | 92.89 a | 65.85 b | 69.17 b | 62.70 b | 65.44 b | 54.11 b | 2.87 | <0.001 |
| DO | 185.89 a | 127.23 b,c | 141.89 b | 97.50 b,c | 115.67 b,c | 81.78 c | 7.03 | <0.001 |
| CI | 467.89 a | 409.23 b,c | 423.89 b | 379.50 b,c | 397.67 b,c | 363.78 c | 7.03 | <0.001 |
| S/C | 3.83 a | 2.62 b | 2.78 a,b | 1.90 b | 2.67 a,b | 2.00 b | 0.16 | 0.004 |
| Item | Control | Gluconeogenic Precursors Supplementation | SEM | p-Value | Chi Square Value: | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| PG300 | PG400 | PG500 | GPG150 | GPG250 | ||||||
| At 7 DIM | % of cows suffered from SCK (number) | 61.1 (11) | 27.7 (5) | 33.3 (6) | 16.6 (3) | 0 (0) | 0 (0) | 0.012 | 14.56 | |
| BHB | 13.71 a | 12.16 a,b | 12.02 a,b | 10.26 b,c | 9.52 c | 7.42 d | 0.35 | <0.001 | ||
| At 14 DIM | % of cows suffered from hypoglycemia (number) | 18.2 (4) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0.005 | 16.97 | |
| Glucose | 43.85 b | 51.77 a | 49.16 a,b | 53.33 a | 54.50 a | 51.75 a | 0.96 | 0.006 | ||
| Item. | Control | Gluconeogenic Precursors Supplementation | SEM | p-Value | ||||
|---|---|---|---|---|---|---|---|---|
| PG300 | PG400 | PG500 | GPG150 | GPG250 | ||||
| Feed Cost/Animal (US$) | 2779.95 a | 2526.20 a,b | 2520.86 a,b | 2344.21 b | 2486.60 b | 2271.12 b | 38.45 | 0.003 |
| Insemination Cost/Animal (US$) | 31.98 a | 21.87 b | 23.21 a,b | 15.87 b | 22.29 a,b | 16.70 b | 1.39 | 0.007 |
| Total Cost/Animal (US$) | 3510.33 a | 3176.91 b | 3188.78 b | 2948.00 b | 3124.97 b | 2868.37 b | 49.10 | <0.001 |
| Cost per kg Milk (US$) | 0.31 a | 0.27 a,b | 0.32 a | 0.25 b | 0.28 a,b | 0.26 b | 0.01 | <0.001 |
| Feed cost per kg Milk (US$) | 0.24 a | 0.22 a,b | 0.25 a | 0.20 b | 0.23 ab | 0.20 b | 0.04 | <0.001 |
| Milk Return (US$) | 4950.54 | 5085.43 | 4360.12 | 5066.8 | 4776.04 | 4860.53 | 92.30 | 0.213 |
| Net Return (US$) | 1440.21 b,c | 1908.52 a,b | 1171.34 c | 2118.80 a | 1651.07 b,c | 1992.16 a | 83.00 | <0.001 |
| IOFC (US$) | 2170.59 a,b | 2559.23 a | 1839.26 b | 2722.59 a | 2289.44 a,b | 2589.41 a | 81.44 | 0.029 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
El-Kasrawy, N.I.; Swelum, A.A.; Abdel-Latif, M.A.; Alsenosy, A.E.-W.A.; Beder, N.A.; Alkahtani, S.; Abdel-Daim, M.M.; Abd El-Aziz, A.H. Efficacy of Different Drenching Regimens of Gluconeogenic Precursors during Transition Period on Body Condition Score, Production, Reproductive Performance, Subclinical Ketosis and Economics of Dairy Cows. Animals 2020, 10, 937. https://doi.org/10.3390/ani10060937
El-Kasrawy NI, Swelum AA, Abdel-Latif MA, Alsenosy AE-WA, Beder NA, Alkahtani S, Abdel-Daim MM, Abd El-Aziz AH. Efficacy of Different Drenching Regimens of Gluconeogenic Precursors during Transition Period on Body Condition Score, Production, Reproductive Performance, Subclinical Ketosis and Economics of Dairy Cows. Animals. 2020; 10(6):937. https://doi.org/10.3390/ani10060937
Chicago/Turabian StyleEl-Kasrawy, Nagwa I., Ayman A. Swelum, Mervat A. Abdel-Latif, Abd El-Wahab A. Alsenosy, Noha A. Beder, Saad Alkahtani, Mohamed M. Abdel-Daim, and Ayman H. Abd El-Aziz. 2020. "Efficacy of Different Drenching Regimens of Gluconeogenic Precursors during Transition Period on Body Condition Score, Production, Reproductive Performance, Subclinical Ketosis and Economics of Dairy Cows" Animals 10, no. 6: 937. https://doi.org/10.3390/ani10060937
APA StyleEl-Kasrawy, N. I., Swelum, A. A., Abdel-Latif, M. A., Alsenosy, A. E.-W. A., Beder, N. A., Alkahtani, S., Abdel-Daim, M. M., & Abd El-Aziz, A. H. (2020). Efficacy of Different Drenching Regimens of Gluconeogenic Precursors during Transition Period on Body Condition Score, Production, Reproductive Performance, Subclinical Ketosis and Economics of Dairy Cows. Animals, 10(6), 937. https://doi.org/10.3390/ani10060937








