Morphometric Characteristics of the Skull in Horses and Donkeys—A Pilot Study
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects and Head Preparation
2.2. Skull Measurements
2.3. Statistical Analyses
3. Results
3.1. Horses
3.2. Donkeys
3.3. Comparison of Measurements between Donkeys and Horses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Grilz-Seger, G.; Reiter, S.; Neuditschko, M.; Wallner, B.; Rieder, S.; Leeb, T.; Jagannathan, V.; Mesarič, M.; Cotman, M.; Pausch, H.; et al. A genome-wide association analysis in noriker horses identifies a snp associated with roan coat color. J. Equine Vet. Sci. 2020, 88, 102950. [Google Scholar] [CrossRef] [PubMed]
- Aberle, K.S.; Hamann, H.; Drögemüller, C.; Distl, O. Genetic diversity in german draught horse breeds compared with a group of primitive, riding and wild horses by means of microsatellite dna markers. Anim. Genet. 2004, 35, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Robert, C.; Valette, J.-P.; Denoix, J.-M. Longitudinal development of equine forelimb conformation from birth to weaning in three different horse breeds. Vet. J. 2013, 198, e75–e80. [Google Scholar] [CrossRef] [PubMed]
- Malinowski, K.; Christensen, R.A.; Hafs, H.D.; Scanes, C.G. Age and breed differences in thyroid hormones, insulin-like growth factor (igf)-i and igf binding proteins in female horses. J. Anim. Sci. 1996, 74, 1936–1942. [Google Scholar] [CrossRef] [PubMed]
- Gastal, E.L.; Gastal, M.O.; Beg, M.A.; Neves, A.P.; Petrucci, B.P.L.; Mattos, R.C.; Ginther, O.J. Miniature ponies: Similarities and differences from larger breeds in follicles and hormones during the estrous cycle. J. Equine Vet. Sci. 2008, 28, 508–517. [Google Scholar] [CrossRef]
- Janczarek, I.; Stachurska, A.; Kedzierski, W.; Wilk, I. Responses of horses of various breeds to a sympathetic training method. J. Equine Vet. Sci. 2013, 33, 794–801. [Google Scholar] [CrossRef]
- Lloyd, A.S.; Martin, J.E.; Bornett-Gauci, H.L.I.; Wilkinson, R.G. Horse personality: Variation between breeds. Appl. Anim. Behav. Sci. 2008, 112, 369–383. [Google Scholar] [CrossRef]
- McGreevy, P.D.; Thomson, P.C. Differences in motor laterality between breeds of performance horse. Appl. Anim. Behav. Sci. 2006, 99, 183–190. [Google Scholar] [CrossRef]
- Druml, T.; Baumung, R.; Sölkner, J. Morphological analysis and effect of selection for conformation in the noriker draught horse population. Livest. Sci. 2008, 115, 118–128. [Google Scholar] [CrossRef]
- Goodwin, D.; Levine, M.; McGreevy, P.D. Preliminary investigation of morphological differences between ten breeds of horses suggests selection for paedomorphosis. J. Appl. Anim. Welf. Sci. 2008, 11, 204–212. [Google Scholar] [CrossRef]
- Folla, F.; Sartori, C.; Guzzo, N.; Pigozzi, G.; Mantovani, R. Genetics of linear type traits scored on young foals belonging to the italian heavy draught horse breed. Livest. Sci. 2019, 219, 91–96. [Google Scholar] [CrossRef]
- Merkies, K.; Alebrand, J.; Harwood, B.; LaBarge, K.; Scott, L. Investigation into thoracic asymmetry in ridden horses. Comp. Exerc. Physiol. 2020, 16, 55–62. [Google Scholar] [CrossRef]
- Evans, K.E.; McGreevy, P.D. Conformation of the equine skull: A morphometric study. J. Vet. Med. Ser. C Anat. Histol. Embryol. 2006, 35, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Nolte, W.; Thaller, G.; Kuehn, C. Selection signatures in four german warmblood horse breeds: Tracing breeding history in the modern sport horse. PLoS ONE 2019, 14, e0215913. [Google Scholar] [CrossRef] [PubMed]
- Górecka-Bruzda, A.; Jastrzebska, E.; Sosnowska, Z.; Jaworski, Z.; Jezierski, T.; Chruszczewski, M.H. Reactivity to humans and fearfulness tests: Field validation in polish cold blood horses. Appl. Anim. Behav. Sci. 2011, 133, 207–215. [Google Scholar] [CrossRef]
- Sackman, J.E.; Houpt, K.A. Equine personality: Association with breed, use, and husbandry factors. J. Equine Vet. Sci. 2019, 72, 47–55. [Google Scholar] [CrossRef]
- Fenner, K.; Freire, R.; McLean, A.; McGreevy, P. Behavioral, Demographic, and Management influences on equine responses to negative reinforcement. J. Vet. Behav. 2019, 29, 11–17. [Google Scholar] [CrossRef]
- Proops, L.; Osthaus, B.; Bell, N.; Long, S.; Hayday, K.; Burden, F. Shelter-seeking behavior of donkeys and horses in a temperate climate. J. Vet. Behav. 2019, 32, 16–23. [Google Scholar] [CrossRef]
- Burden, F.; Thiemann, A. Donkeys are different. J. Eq. Vet. Sci. 2015, 35, 376–382. [Google Scholar] [CrossRef]
- Davis, E. Donkey and mule welfare. Vet. Clin. N. Am.-Eq. Pract. 2019, 35, 481–491. [Google Scholar] [CrossRef]
- McLean, A.K.; Navas González, F.J.; Canisso, I.F. Donkey and mule behavior. Vet. Clin. N. Am.-Eq. Pract. 2019, 35, 575–588. [Google Scholar] [CrossRef] [PubMed]
- Grint, N.J.; Beths, T.; Yvorchuk-St Jean, K.; Whay, H.R.; Murrell, J.C. Analysis of behaviors observed during mechanical nociceptive threshold testing in donkeys and horses. J. Equine Vet. Sci. 2017, 50, 102–109. [Google Scholar] [CrossRef]
- van Dierendonck, M.C.; van Loon, J.P.A.M.; Burden, F.A.; Rickards, K. Monitoring acute pain in donkeys with the equine utrecht university scale for donkeys composite pain assessment (equus-donkey-compass) and the equine utrecht university scale for donkey facial assessment of pain (Equus-Donkey-Fap). Animals 2020, 10, 354. [Google Scholar] [CrossRef] [PubMed]
- Corrales-Hernández, A.; Mota-Rojas, D.; Guerrero-Legarreta, I.; Roldan-Santiago, P.; Rodríguez-Salinas, S.; Yáñez-Pizaña, A.; de la Cruz, L.; González-Lozano, M.; Mora-Medina, P. Physiological responses in horses, donkeys and mules sold at livestock markets. Int. J. Vet. Sci. Med. 2018, 6, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Georgevsky, D.; Carrasco, J.J.; Valenzuela, M.; McGreevy, P.D. Domestic dog skull diversity across breeds, breed groupings, and genetic clusters. J. Vet. Behav. 2013, 9, 228–234. [Google Scholar] [CrossRef]
- McGreevy, P.D.; Georgevsky, D.; Carrasco, J.; Valenzuela, M.; Duffy, D.L.; Serpell, J.A. Dog behavior co-varies with height, bodyweight and skull shape. PLoS ONE 2013, 8. [Google Scholar] [CrossRef]
- Stone, H.R.; McGreevy, P.D.; Starling, M.J.; Forkman, B. Associations between domestic-dog morphology and behaviour scores in the dog mentality assessment. PLoS ONE 2016, 11, e0149403. [Google Scholar] [CrossRef]
- Cervantes, I.; Baumung, R.; Molina, A.; Druml, T.; Gutiérrez, J.P.; Sölkner, J.; Valera, M. Size and shape analysis of morphofunctional traits in the spanish arab horse. Livest. Sci. 2009, 125, 43–49. [Google Scholar] [CrossRef]
- Radinsky, L. Ontogeny and phylogeny in horse skull. Evolution 1984, 38, 1–15. [Google Scholar] [CrossRef]
- Komosa, M.; Molinski, K.; Godynicki, S. The variability of cranial morphology in modern horses. Zoolog. Sci. 2006, 23, 289–298. [Google Scholar] [CrossRef]
- McGreevy, P.; Grassi, T.D.; Harman, A.M. A strong correlation exists between the distribution of retinal ganglion cells and nose length in the dog. Brain. Behav. Evol. 2004, 63, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Roberts, T.; McGreevy, P.; Valenzuela, M. Human induced rotation and reorganization of the brain of domestic dogs. PLoS ONE 2010, 5, e11946. [Google Scholar] [CrossRef] [PubMed]
- Evans, K.E.; McGreevy, P.D. The distribution of ganglion cells in the equine retina and its relationship to skull morphology. J. Vet. Med. Ser. C Anat. Histol. Embryol. 2007, 36, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, B.L. Making Sense of It All: The importance of taking into account the sensory abilities of animals in their housing and management. Appl. Anim. Behav. Sci. 2018, 205, 175–180. [Google Scholar] [CrossRef]
- Mazzatenta, A.; Veronesi, M.C.; Vignola, G.; Ponzio, P.; Carluccio, A.; De Amicis, I. Behavior of martina franca donkey breed jenny-and-foal dyad in the neonatal period. J. Vet. Behav. 2019, 33, 81–89. [Google Scholar] [CrossRef]
- Simcock, N.K.; Gray, H.; Bouchebti, S.; Wright, G.A. Appetitive olfactory learning and memory in the honeybee depend on sugar reward identity. J. Insect Physiol. 2018, 106, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Patin, A.; Pause, B.M. Human amygdala activations during nasal chemoreception. Neuropsychologia 2015, 78, 171–194. [Google Scholar] [CrossRef]
- Soudry, Y.; Lemogne, C.; Malinvaud, D.; Consoli, S.M.; Bonfils, P. Olfactory system and emotion: Common substrates. Eur. Ann. Otorhinolaryngol. Head Neck Dis. 2011, 128, 18–23. [Google Scholar] [CrossRef]
- Cardinal, R.N.; Parkinson, J.A.; Hall, J.; Everitt, B.J. Emotion and motivation: The role of the amygdala, ventral striatum, and prefrontal cortex. Neurosci. Biobehav. Rev. 2002, 26, 321–352. [Google Scholar] [CrossRef]
- Corbit, L.H.; Leung, B.K.; Balleine, B.W. The role of the amygdala-striatal pathway in the acquisition and performance of goal-directed instrumental actions. J. Neurosci. 2013, 33, 17682–17690. [Google Scholar] [CrossRef]
- Pena, R.R.; Medeiros, D. de C.; Guarnieri, L. de O.; Guerra, J.B.; Carvalho, V.R.; Mendes, E.M.A.M.; Pereira, G.S.; Moraes, M.F.D. Home-Cage odors spatial cues elicit theta phase/gamma amplitude coupling between olfactory bulb and dorsal hippocampus. Neuroscience 2017, 363, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Saslow, C.A. Understanding the perceptual world of horses. Appl. Anim. Behav. Sci. 2002, 78, 209–224. [Google Scholar] [CrossRef]
- McGreevy, P.D.; Rogers, L. Motor and sensory laterality in thoroughbred horses. Appl. Anim. Behav. Sci. 2005, 92, 337–352. [Google Scholar] [CrossRef]
- Grandin, T.; Deesing, M.J.; Struthers, J.J.; Swinker, A.M. Cattle with hair whorl patterns above the eyes are more behaviorally agitated during restraint. Appl. Anim. Behav. Sci. 1995, 46, 117–123. [Google Scholar] [CrossRef]
- Meyer, J.F. What’s in a whorl (hair swirls of horses). Horse Rider 2008, 47, 46–53. [Google Scholar]
- Górecka, A.; Słoniewski, K.; Golonka, M.; Jaworski, Z.; Jezierski, T. Heritability of hair whorl position on the forehead in konik horses. J. Anim. Breed. Genet. 2006, 123, 396–398. [Google Scholar] [CrossRef]
- Górecka, A.; Golonka, M.; Chruszczewski, M.; Jezierski, T. A note on behaviour and heart rate in horses differing in facial hair whorl. Appl. Anim. Behav. Sci. 2007, 105, 244–248. [Google Scholar] [CrossRef]
- Shivley, C.; Grandin, T.; Deesing, M. Behavioral laterality and facial hair whorls in horses. J. Equine Vet. Sci. 2016, 44, 62–66. [Google Scholar] [CrossRef]
- Graf, P.; König von Borstel, U.; Gauly, M. Importance of personality traits in horses to breeders and riders. J. Vet. Behav. 2013, 8, 316–325. [Google Scholar] [CrossRef]
- Starling, M.J.; Branson, N.; Thomson, P.C.; McGreevy, P.D. “Boldness” in the domestic dog differs among breeds and breed groups. Behav. Process. 2013, 97, 53–62. [Google Scholar] [CrossRef]
- Mehrkam, L.R.; Wynne, C.D.L. Behavioral Differences among breeds of domestic dogs (Canis Lupus Familiaris): Current status of the science. Appl. Anim. Behav. Sci. 2014, 155, 12–27. [Google Scholar] [CrossRef]
- Janiszewska, J.; Ignor, J.; Cieśla, A. Einfluss eines 11-monatigen trainings auf die ergebnisse des “Ängstlichkeitstests” von jungen halbblut-hengsten. Arch. Anim. Breed. 2004, 47, 7–13. [Google Scholar] [CrossRef]
- Borstel, U.; Pirsich, W.; Gauly, M.; Bruns, E. Repeatability and reliability of scores from ridden temperament tests conducted during performance tests. Appl. Anim. Behav. Sci. 2012, 139, 251–263. [Google Scholar] [CrossRef]
- Kuhnke, S.; Bär, K.; Bosch, P.; Rensing, M.; Borstel, U.K.V. Evaluation of a novel system for linear conformation, gait, and personality trait scoring and automatic ranking of horses at breed shows: A pilot study in american quarter horses. J. Equine Vet. Sci. 2019, 78, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Osthaus, B.; Proops, L.; Hocking, I.; Burden, F. Spatial cognition and perseveration by horses, donkeys and mules in a simple a-not-b detour task. Anim. Cogn. 2013, 16, 301–305. [Google Scholar] [CrossRef]
- Baragli, P.; Regolin, L. Cognition tests in equids (Equus Caballus and Equus Asinus). In Proceedings of the International Equine Science Meeting, Regensburg, Germany, 3–5 October 2008. [Google Scholar]
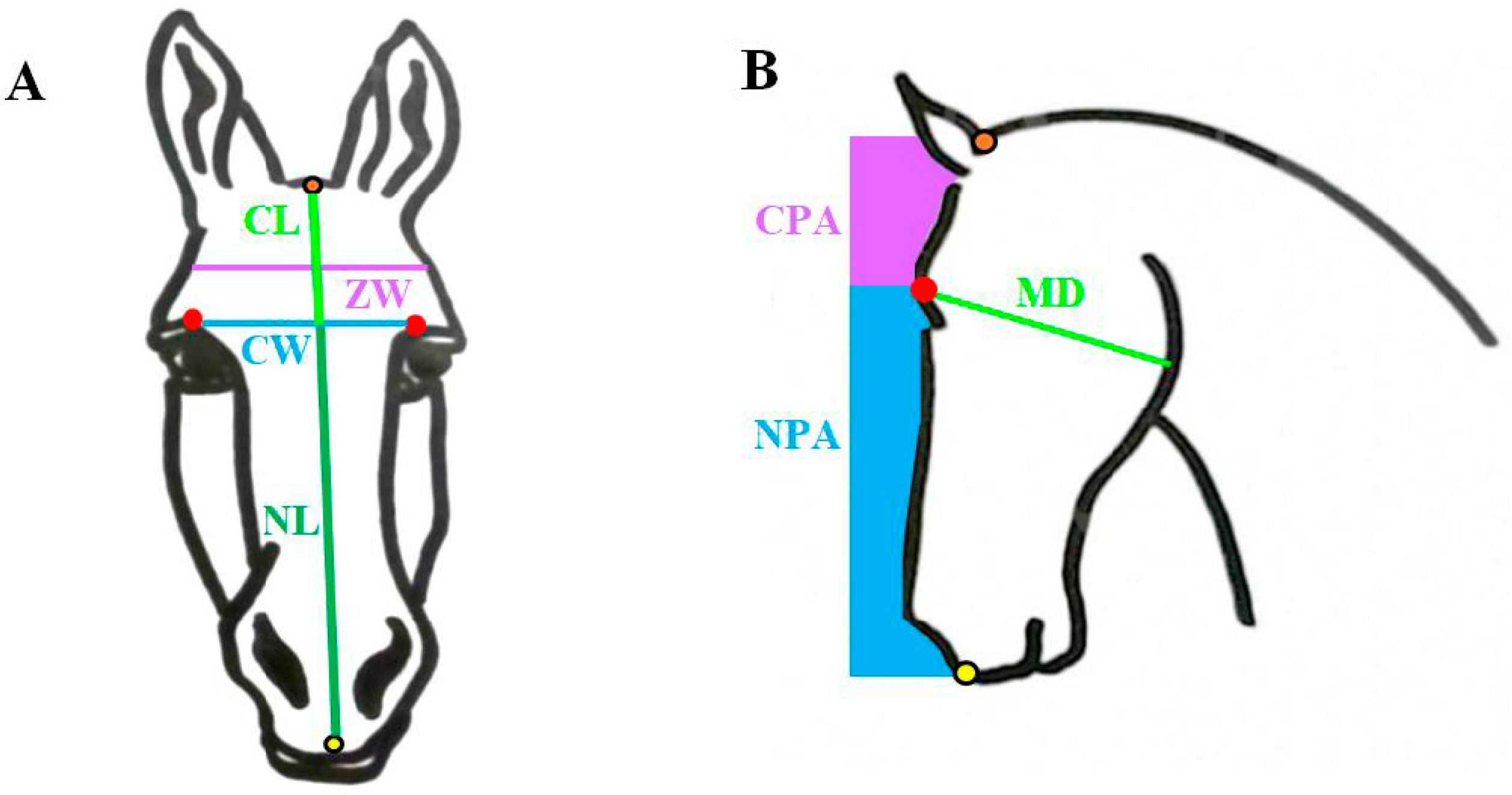
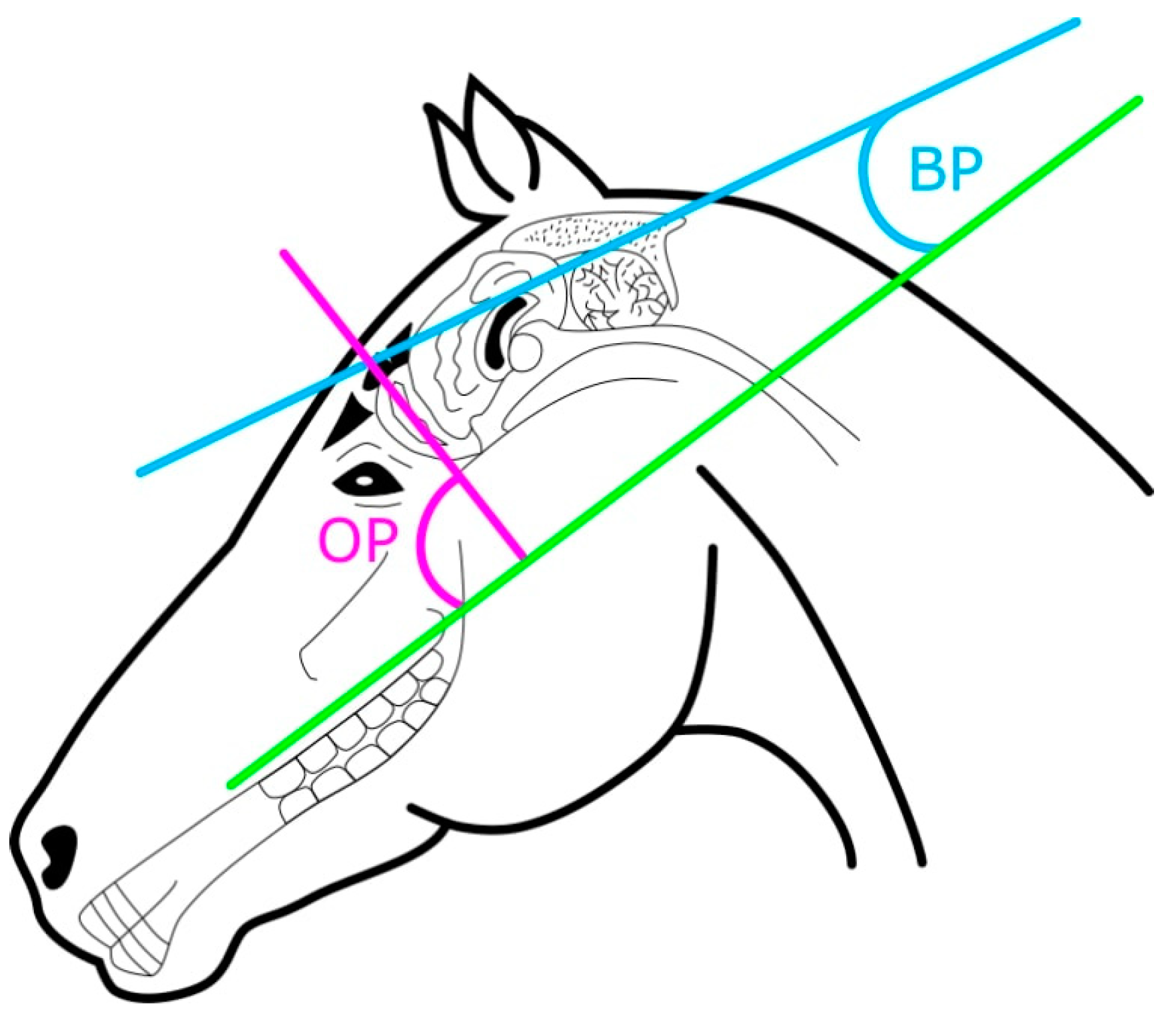

| Variable | Horse | Donkey | p-Value |
|---|---|---|---|
| Mean | Mean | ||
| Head weight (kg) | 22.5 ± 4.9 | 13.6 ± 3.7 | <0.0001 * |
| Skull length (cm) | 53.6 ± 2.6 | 46.6 ± 5.0 | 0.0080 * |
| Cranial length (cm) | 23.6 ± 1.4 | 20.4 ± 2.7 | 0.0002 * |
| Nasal length (cm) | 30.4 ± 3.9 | 27.5 ± 3.1 | 0.1919 |
| Zygomatic width (cm) | 22.6 ± 1.8 | 21.9 ± 2.2 | 0.0776 |
| Cranial width (cm) | 19.0 ± 0.8 | 16.3 ± 2.4 | <0.0001 * |
| Mandibular depth (cm) | 27.2 ± 1.1 | 24.5 ± 2.4 | 0.0019 * |
| Skull index | 42.2 ± 3.2 | 46.2 ± 3.4 | 0.0025 * |
| Cranial index | 80.9 ± 6.2 | 77.2 ± 9.1 | 0.1399 |
| Nasal index | 76.0 ± 14.5 | 79.6 ± 7.5 | 0.6600 |
| Mandibular index | 50.8 ± 2.6 | 52.2 ± 5.2 | 0.5791 |
| Cranial profile index | 8.8 ± 1.0 | 7.6 ± 1.2 | 0.0335 * |
| Nasal profile index | 9.1 ± 2.8 | 7.3 ± 0.8 | 0.8359 |
| Olfactory bulb area (cm2) | 2.3 ± 1.3 | 1.98 ± 1.3 | 0.0478 * |
| Olfactory bulb pitch (°) | 66.0 ± 11.8 | 56.6 ± 1318 | 0.0163 * |
| Brain pitch (°) | 18.3 ± 45.7 | 16.7 ± 37.8 | 0.4338 |
| Whorl location (cm) | 0.1 ± 1.5 | 8.0 ± 6.5 | 0.0001 * |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Merkies, K.; Paraschou, G.; McGreevy, P.D. Morphometric Characteristics of the Skull in Horses and Donkeys—A Pilot Study. Animals 2020, 10, 1002. https://doi.org/10.3390/ani10061002
Merkies K, Paraschou G, McGreevy PD. Morphometric Characteristics of the Skull in Horses and Donkeys—A Pilot Study. Animals. 2020; 10(6):1002. https://doi.org/10.3390/ani10061002
Chicago/Turabian StyleMerkies, Katrina, Georgios Paraschou, and Paul Damien McGreevy. 2020. "Morphometric Characteristics of the Skull in Horses and Donkeys—A Pilot Study" Animals 10, no. 6: 1002. https://doi.org/10.3390/ani10061002
APA StyleMerkies, K., Paraschou, G., & McGreevy, P. D. (2020). Morphometric Characteristics of the Skull in Horses and Donkeys—A Pilot Study. Animals, 10(6), 1002. https://doi.org/10.3390/ani10061002





